Abstract
Evaluation of skeletal muscle frequently combines morphologic and morphometric techniques. As is the case with many organ systems, skeletal muscle has limited responses to insult or injury. Over the past several years, crucial interactions between skeletal muscle, bone, and the nervous system have been described. The aim of this lecture was to give attendees the necessary background information in basic skeletal muscle morphology, important species differences, introduction to skeletal muscle biomarkers, approaches to morphologic and morphometric evaluation, and examples of background findings and typical responses of skeletal muscle to insult or injury.
Introduction to Skeletal Muscle Biomarkers
Biomarkers used to assess skeletal muscle are generally markers of skeletal muscle damage, not other pathology such as atrophy. They are typically cellular enzymes (such as creatine kinase and aspartame amino transferase) or components of the contractile apparatus (including troponin, actin, myosin, and tropomyosin); most have isoforms with variable specificity for skeletal muscle and can be found in other organs (notably heart, liver, and kidney; Berridge, Van Vleet, and Herman, 2013; Gordon, Regnier, and Homsher, 2001). Panels of various skeletal muscle biomarkers have been developed to provide better sensitivity and selectivity than interpreting data from only 1 or 2 biomarkers. The reader is directed to R. Goldstein’s synopsis of recent advances in biomarker development for skeletal muscle in this same symposium issue for further details.
Handling and Processing of Skeletal Muscle Samples
Because skeletal muscles are anisotropic in fiber-type distribution and prone to handling and fixation artifacts, advance planning and due care are critical to successful evaluation. Appropriate orientation of skeletal muscle at trimming is vital for myofiber analysis. Cut sections should be either true cross sections (necessary for determination of fiber size) or true longitudinal sections (in order to follow myofibers for a distance). Oblique sections are not suitable for image analysis, although they can be used for routine toxicologic pathology evaluation. The importance of avoiding handling artifacts including myofiber contracture and/or swelling from sudden exposure to formalin or artifacts related to freezing of muscle cannot be overstated. Finally, consistency in the choice of skeletal muscle and precise location of sampling is vital to producing interpretable results.
Points to keep in mind during sampling of skeletal muscle for routine formalin fixation include controlling the length of the sample to minimize artifactual contracture, for example, by clamping or otherwise securing the muscle sample and avoiding overly sudden exposure of fresh muscle to cold formalin (which will also induce contracture). For skeletal muscle sections to be frozen, avoidance of freeze artifact is crucially important. In order to avoid freeze artifact, the skeletal muscle sample must freeze fast enough that water ice crystals do not form in the myofiber cytoplasm and that the frozen blocks be stored in an ultracold freezer (−70°C to −80°C). One way to accomplish a very fast freeze is to submerge the muscle sample into a container of a freezing solvent (such as isopentane) that is surrounded by liquid nitrogen (vs. submergence of the sample directly into liquid nitrogen). The chilled solvent prevents formation of a nitrogen gas bubble around the sample which would delay the speed of freezing and increase the likelihood of intracellular ice crystal formation. Keep in mind that repeated freeze–thaw cycles will also induce ice crystal formation, so a single frozen tissue block may only be cut in a few times before ice crystal formation decreases the quality of the sections obtained. Production of more than 1 tissue block from the same sample is recommended to increase the number of cutting sessions available for a muscle sample.
Muscle Fiber Types and Topologic Variability
Myosin light chain and heavy chain isoforms vary in skeletal muscle based on functionality. Historically, muscle fiber types were divided into 2 basic categories, slow twitch and fast twitch, which was based on speed of contraction. It is now known that slow-twitch muscles (also called type I) are often found in muscles used for maintenance of posture and that they were resistant to fatigue by relying mainly on oxidative metabolism, while fast-twitch muscles (also called type II) are typically found in muscles used for motion, are fatigable, and rely primarily on glycolytic metabolism (see Table 1). The type II fast-twitch fibers are further divided into types IIA, IIX, IIB, and IIC depending on staining characteristics for myosin adenosine triphosphatase (ATPase) or myosin isoforms and on physiologic characteristics of twitch speed and fatigability. Type IIC fibers are a transitional form of myofiber present in foci of regenerating myofibers and also during postnatal muscle development. More recently, hybrid myofibers that combine attributes of different classical fiber types (such as IIA-X, IIX-B, etc.) have been reported in several species (Salviati, Betto, and Danieli Betto, 1982; Schiaffino et al., 2001).
Classic Muscle Fiber-type Characteristics.

Muscle fiber types are not randomly or evenly distributed within a muscle. Slow-twitch fibers tend to be present in higher amounts deep in a muscle (shown in many species) and more proximally (reported in rat hindlimb muscles; Wang et al., 2000, 2001).
Some Specific Muscle and Species Differences
Humans and dogs do not have type IIB myofibers (but do have type IIX). While the soleus muscle is often considered to contain only type I myofibers, it actually contains a mixture of type I and IIA myofibers in the mouse, and the soleus muscle is not present in the dog.
Choice of Skeletal Muscle for Analysis
The ideal skeletal muscle would have either entirely 1 fiber type or a consistent mixture of fast- and slow-twitch myofibers with a consistent orientation, so that true cross sections and longitudinal sections are easy to produce. For routine toxicity studies, thigh muscles (biceps femoris, and quadriceps) or hindlimb muscles (gastrocnemius, long digital extensor, and anterior tibial) are commonly chosen. Most of these muscles contain more fast-twitch fibers than slow-twitch fibers, particularly superficially. An advantage of using the biceps femoris is that it can be used to protect the sciatic nerve prior to trimming, decreasing handling artifacts in peripheral nerve sections. A disadvantage in using the gastrocnemius is that the fiber orientation is so variable that it is extremely difficult to obtain true cross sections or longitudinal sections.
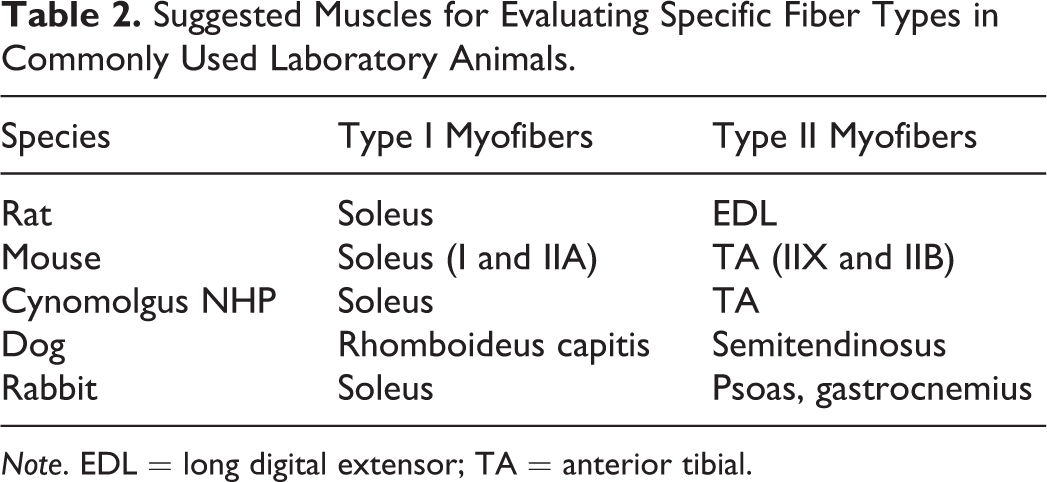
For investigations of a particular fiber type, some muscles are predominately type I or type II (see Table 2 for suggestions of commonly used laboratory animals).
Suggested Muscles for Evaluating Specific Fiber Types in Commonly Used Laboratory Animals.
Light Microscopic Stains for Evaluation of Skeletal Muscle
Historically, most investigative microscopic evaluations on skeletal muscle have been performed on frozen sections. Many of these procedures require intact cytoplasmic enzymes and therefore chemical fixation is contraindicated. Lack of chemical fixation also results in fewer cellular artifacts (contracted fibers and swollen fibers in particular) than in routinely fixed tissue. Common enzyme histochemical stains for frozen sections are myosin ATPase (run under acid and alkaline conditions) for muscle fiber typing and stains for mitochondria (including nicotinamide adenine dinucleotide [NADH]-tetrazolium reductase, cytochrome oxidase, and succinate dehydrogenase). Other stains used on frozen sections include modified Gömöri’s trichrome, hematoxylin and eosin (H&E), periodic acid–Schiff’s base (PAS), Alcian blue, and so on. Fluorescent labeled lectins can be used to illuminate cell membranes and intracellular carbohydrates. Laminin in the myofiber basement membrane can also be easily detected. Immunohistochemistry for myosin fiber typing on frozen sections has increased sensitivity (particularly the identification of hybrid fibers) compared with myosin enzyme histochemistry (Kammoun et al., 2014). Oil red O as a lipid stain can be challenging and the use of alternative stains, such as Paraphenylenediamine (PPD), gives much better resolution and localization (Shirai et al., 2016).
For routine skeletal muscle evaluation, however, formalin-fixed paraffin embedded (FFPE) material is the standard, even though cellular artifacts (due to chemical fixation) are much more common in FFPE sections than in frozen sections. The standard stains for FFPE include H&E, Masson’s trichrome, PAS, and Picrosirius Red. Immunohistochemistry for myosin isoforms has become increasingly available for FFPE skeletal muscle, which will provide the ability for higher throughput than fiber typing on frozen sections.
Common Light Microscopic Findings in Skeletal Muscle
The light microscopic appearance of degenerating myocytes is characterized by cytoplasmic vacuolation, disassociation of myofilaments (or alternately, hypercontraction), multiple internal nuclei, and infiltration of inflammatory cells (most often macrophages). H&E staining may show increased eosinophilia if myofibers are hypercontracted or cytoplasmic pallor if myofibrillar degeneration is significant.
The light microscopic appearance of regenerating myocytes is characterized by cytoplasmic basophilia due to increased RNA expression, internal nuclei, and smaller size than regular myofibers. These fibers are generally type IIC, a transitional fiber type. Small foci of regenerating cells can often be seen in striated muscle in the esophagus as a sequelae to oral gavage trauma.
Incidental findings are often seen near fibrous structures, including tendons, epimysium, perimysium, connective tissue fascia, and so on. They include individual or very small numbers of degenerating or regenerating fibers or small cellular infiltrates. A small number of these are typical in normal skeletal muscle and do not indicate a pathologic process.
Other structures of note in skeletal muscles include peripheral nerve fibers and muscle spindles, which should be evaluated along with myofibers, vascular supply, and connective tissue. For all recorded findings, the use of International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) terminology is strongly encouraged.
Image Analysis and Electron Microscopy
Image analysis is commonly performed on sections of skeletal muscle to determine fiber size such as a cross-sectional area (CSA) or a diameter (Pertl et al., 2013). Measurement of CSA requires true cross (transverse) sections, while measuring a diameter (such as Feret’s or a “lesser” diameter) allows for a small degree of section obliquity. In order to optimize color thresholding by an image analysis program, it is important to use high-contrast stains whose colors can be separated easily. The stains can be chemical, histochemical, fluorescent, or a combination.
Appropriate stereological techniques must be followed. For determination of fiber size, it is imperative that only true cross sections be measured, that randomly selected areas be measured (while keeping in mind the inherent variability in skeletal muscle fiber-type distribution), and that a predetermined total area or total number of muscle fibers be measured.
Cross sections or longitudinal sections can be used to measure other parameters such as myofiber nuclear size and/or number. Longitudinal sections are more frequently evaluated by transmission electron microscopy for ultrastructure, including myofibrillar contractile components or abnormal content.
Ancillary Methods
Beyond the scope of this summary are a number of ancillary methods of assessing skeletal muscles, including electromyography/electrophysiology for functional assessment of the nerve/muscle axis, imaging technologies including magnetic resonance imaging (MRI) and computed tomography for determination of total muscle size and adiposity and weights of individual skeletal muscles (although collection techniques must be extremely consistent), and molecular biology techniques such as proteomics, genomics, and so on, to determine upregulation or downregulation of genes and gene products.
Summary: Take-home Points
Current biomarkers are more indicative of damage, not atrophy; new biomarker panels are being developed as adjuncts to improve sensitivity and selectivity. Good tissue handling and processing are vital for minimizing artifacts and conducting quantitative analysis. Awareness of species, muscle, and topological variations of fiber types are key concepts. Use stereological principles for quantitation of myofiber size and type.
Use INHAND terminology for describing light microscopic findings in skeletal muscle.
Footnotes
Author’s Note
All studies presented were conducted in accordance with current guidelines for animal welfare and all procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC).
Acknowledgments
The author would like to thank Rani Sellers, Andrew Robertson, Alan Opsahl, Walt Bobrowski, and Christopher Dubois, all at Pfizer Inc., in Pearl River, NY (R.S.) or Groton, CT (A.R., A.O., W.B., and C.D.).
Author Contribution
The author (KG) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. The author gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
