Abstract
Because articular cartilage has very limited healing potential, most symptomatic cartilage injuries eventually result in end-stage osteoarthritis and are treated with artificial joint replacement. Our interdisciplinary, comparative orthopedic research performed by a team of DVMs, MDs, engineers, and basic scientists has yielded marked progress toward effective biologic joint restoration strategies by bringing bench-side ideas to fruition in bedside applications in both canine and human patients. This mini-review summarizes the progress of biologic joint restoration strategies at our center.
Articular cartilage has very limited intrinsic healing potential (Huey, Hu, and Athanasiou 2012), and its damage often leads to the development of osteoarthritis, the most common form of arthritis and a leading cause of disability throughout the world (Neogi 2013). Currently, there is no cure for osteoarthritis, and medical treatments are mainly focused on decreasing inflammation and pain. Surgical intervention is often used in an attempt to treat irrevocably damaged articular cartilage by removal (debridement), repair, and/or replacement. Total joint replacement (TJR) using synthetic prosthetic implants is often considered the definitive treatment option for patients with extensive articular cartilage damage. Although TJR surgery is appropriately considered to be one of the greatest surgical advances in recent times based on consistent improvements in patients’ pain, function, and quality of life, complications, morbidity, and revisions rates are still significant such that TJR is ideally reserved for patients older than 65 years and not involved in high-impact activities (Lachiewicz and Soileau 2009; Ritter and Meneghini 2010; Swanson, Schmalzried, and Dorey 2009).
Microfracture and subchondral drilling are common procedures aimed at encouraging cartilage repair by marrow stimulation (McNickle, Provencher, and Cole 2008). Marrow stimulation allows bone marrow cells to reach to the avascular cartilage lesion to mount a healing response; however, unlike hyaline cartilage, the resultant repaired cartilage is type I collagen-rich fibrocartilage and deteriorates over time in response to the mechanical loading of the joint (Kaul et al. 2012; Murawski, Foo, and Kennedy 2010). Autologous chondrocyte implantation (ACI) techniques use the patient’s own cells with or without scaffolds to enhance repair in cartilage defects and can yield more robust repair tissue than marrow stimulation and good clinical outcomes (Biant et al. 2014; Brittberg et al. 1994; Peterson et al. 2002). However, ACI requires 2 surgeries: harvest of the patient’s chondrocytes and implantation of culture-expanded chondrocytes. The resultant repair tissue is predominantly fibrocartilage (Gikas et al. 2009; Roberts et al. 2009), the procedure is costly, and graft failure is not an uncommon complication (Gikas et al. 2009; Niemeyer et al. 2008).
Regeneration of articular cartilage using tissue engineering strategies has demonstrated promising outcomes in animals. Large tissue-engineered constructs with hyaline cartilage architecture and native biomechanical properties have been successfully elaborated in vitro (Bian et al. 2010; Roach et al. 2015). Our collaborative studies with Professor Hung at Columbia University have demonstrated that agarose constructs seeded with canine chondrocytes and subjected to dynamic deformation produce tissue-engineered cartilage with material properties and biochemical composition matching native canine hyaline cartilage (Bian et al. 2010). After safety and efficacy testing in preclinical animal models (Ng et al. 2010), 6 client-owned canine patients with symptomatic stifle (knee) joint articular cartilage defects were treated with agarose-based tissue-engineered osteochondral constructs with informed client consent and have shown good to excellent long-term (4–8 years) clinical outcomes (unpublished data). Cell-free scaffolds aimed at homing endogenous host cells for self-repair is another attractive articular cartilage tissue engineering approach with evidence for applicability. In collaboration with Professor Ma at Columbia University, our research demonstrated that the entire articular surface of rabbit humeral heads can be restored by homing host cells to a patient-specific scaffold using a transforming growth factor (TGF) beta3-infused polycaprolactone with hydroxyapatite bioscaffold (Lee et al. 2010). Although these and other articular cartilage tissue engineering strategies hold great potential for improving cartilage regeneration and repair treatments, these technologies are not yet approved for use in human patients, and the regulatory and financial hurdles to their clinical application are daunting.
Osteochondral graft transfer and transplantation are the only currently available methods for restoring articular defects with hyaline cartilage. Osteochondral autograft is a viable option for treating smaller (<2 cm2) articular defects, primarily in knees, without risk of disease transmission or immune response. Reported clinical success rates range between 72% and 92% in long-term follow-up studies (Hangody et al. 2008; Pareek et al. 2016). However, osteochondral autograft is associated with donor site morbidity and is limited by defect size and location (Camp, Stuart, and Krych 2014). Osteochondral allograft (OCA) transplantation is another approved option for restoring articular defects with hyaline articular cartilage that is far less limited by lesion size and location. The first clinical OCA transplantation was reported more than 100 years ago (Lexer 1908), and it has been used clinically for more than 40 years since its modern descriptions by Gross et al. (1975) and Meyers, Akeson, and Convery 1989. OCA transplantation has been associated with 88% return to sport (Krych, Robertson, and Williams 2012) and greater than 80% 10-year graft survivorship for treatment of large femoral condyle lesions (Aubin et al. 2001; Gross, Shasha, and Aubin 2005). However, the use of OCAs in clinical practice is limited by availability (quantity) of acceptable donor tissues for eligible patients. One of the major limitations to availability is the capability of tissue banks to preserve OCAs with essential chondrocyte viability (quality) for sufficient time after procurement to complete mandatory disease screening protocols and identify, match, and place the tissue at a center for transplantation into an eligible patient (Capeci et al. 2013; Demange and Gomoll 2012). Studies have shown that human OCAs stored at 4°C using the current standard of care (SOC) method at tissue banks for more than 14 days undergo significant decrease in chondrocyte viability such that the majority of grafts fall below the minimum essential chondrocyte viability level (70% of day 0 viable chondrocyte density [VCD]) by day 28 after procurement (Allen et al. 2005; Ball et al. 2004; S. K. Williams et al. 2003). Data from our laboratory revealed that mean chondrocyte viability in SOC OCAs (n = 24, storage days ranging from 16 to 21 days) obtained from 2 tissue banks and designated for transplantation into patients was only 62% (unpublished data). Clinical data from 75 patients who underwent OCA transplantation at our center for treatment of large (>2 cm2) articular defects of the femoral condyle showed that graft storage of >28 days at 4°C prior to implantation was associated with a significantly and 2.6 times lower likelihood of a successful outcome (Nuelle et al. 2017), which matches data from others (LaPrade et al. 2009). The other major factors to alter clinical outcomes after OCA transplantation include patients’ preoperative activity levels, body mass index (Nuelle et al. 2017), and surgical techniques for graft creation and implantation. Therefore, our team of orthopedic clinicians and scientists designed and implemented a comparative translational research approach to address quantity, quality, and technique limitations for successful OCA transplantation in canine and human patients.
Materials and Methods
All procedures were performed under institutional Animal Care and Use Committee approvals (8235, 8236, and 8285) for canine studies and institutional review board (IRB) approvals (2003053, 2002628, and 2005936) for human studies. As a critical step toward improving maintenance of essential chondrocyte viability of OCAs during preservation, a series of experiments were performed using medial and lateral femoral condyles aseptically harvested from stifle (knee) joints of adult canine cadavers within 4 hr of euthanasia performed for unrelated reasons (Garrity et al. 2012; A. Stoker et al. 2012; A. M. Stoker et al. 2011). The femoral condyles were either used as time 0 (at harvest) controls or randomly assigned to one of more than 40 different combinations of media, temperature, and container characteristics to evaluate the effects of preservation methods for extending the duration of OCA preservation.
After optimizing temperature, media, and container characteristics for OCA quality during extended preservation (Garrity et al. 2012; A. Stoker et al. 2012; A. M. Stoker et al. 2011), functional outcomes of OCAs were evaluated using a preclinical canine model (Cook et al. 2014, 2016). Then, the effectiveness of our novel protocol to maintain sufficient chondrocyte viability, extracellular matrix composition, and material properties was evaluated using human femoral condyle OCAs (A. M. Stoker, Stannard, et al. 2017).
For all experiments, chondrocyte viability was determined using Calcein AM (Invitrogen, Carlsbad, CA) for the live cell stain and either ethidium homodimer-1 (Invitrogen, Carlsbad, CA) or SYTOX Blue (Invitrogen, Carlsbad, CA) for visualizing dead cells as described elsewhere (Cook et al. 2014, 2016; Garrity et al. 2012; A. M. Stoker, Stannard, et al. 2017; A. Stoker et al. 2012). In addition, histologic integrity of OCAs was evaluated using the Osteoarthritis Research Society International (OARSI) system (Cook, Kuroki, et al. 2010). For histological processing, tissues were fixed in 10% buffered formalin fixative for 48 hr and decalcified in 10% EDTA solution. Furthermore, biochemical evaluation of extracellular matrix composition of OCAs was assessed using dimethylmethylene blue assay (DMMB) for glycosaminoglycan (Farndale, Buttle, and Barrett 1986) and hydroxyproline assay for collagen (Reddy and Enwemeka 1996), and biomechanical properties including dynamic modules and instantaneous tissue modules of OCAs were assessed as described previously (Cook et al. 2016; Garrity et al. 2012). In the clinical study, outcomes were assessed by using multiple patient-reported measures including Visual Analog Scale for pain (VAS pain), International Knee Documentation Committee (IKDC; Irrgang et al. 2001), Single Assessment Numerical Evaluation (SANE; Winterstein et al. 2013), Tegner (Tegner and Lysholm 1985), and Patient-Reported Outcomes Measurement Information System (PROMIS) Mobility (Kratz et al. 2013).
Results
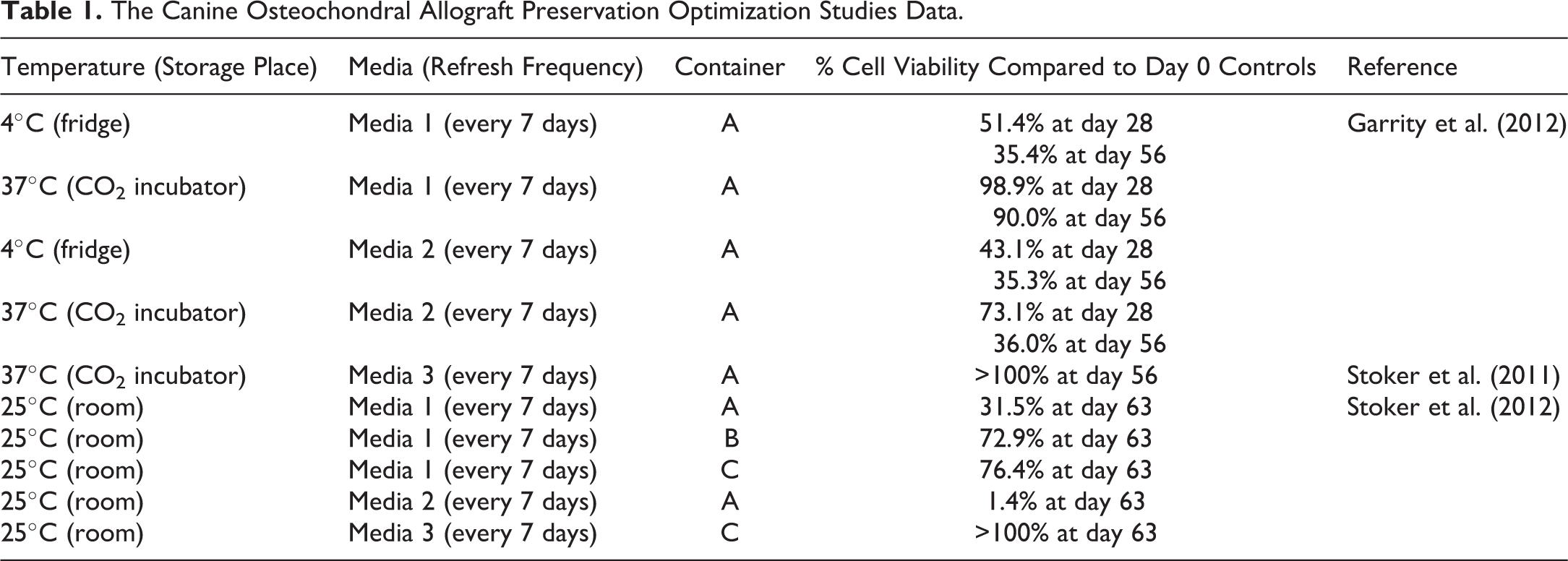
In vitro study using canine femoral condyle OCAs (n = 45) conducted by Garrity et al. (2012) showed that chondrocyte viability in OCAs was well maintained after 28 and 56 days storage at 37°C in a serum-free media named Media 1. This study also showed that an anti-inflammatory and chondrogenic media named Media 2 was not a good preservation media with mean OCA viability markedly dropped down after 28 days. Simultaneously, A. M. Stoker et al. (2011) demonstrated that culture canine femoral condyles (n = 5) preserved at 37°C in a proprietary media named Media 3 maintained chondrocyte viability. A subsequent study conducted by A. Stoker et al. (2012) demonstrated that nearly day 0 chondrocyte viability (n = 7) can be maintained for up to 63 days when canine femoral condyle OCAs are stored at room temperature (∼25°C) in a proprietary container named “C” with Media 3 (n = 8). The canine OCA preservation optimization in vitro studies data are summarized in Table 1.
The Canine Osteochondral Allograft Preservation Optimization Studies Data.
In order to validate these in vitro study findings, Cook et al. (2014, 2016) conducted a preclinical canine model study. In this study, canine femoral condyle OCAs were stored for 28 or 60 days after procurement at room temperature using our novel protocol with a proprietary media named Media 3 and a proprietary container named “C” compared to the SOC tissue bank protocol at 4°C. Mean chondrocyte viability in OCAs stored in our protocol was 82% at day 28 and 89% at day 60 while for those stored using the SOC protocol was 60% at day 28 and 52% at day 60. In this study, all successful OCAs as determined by radiographs, arthroscopy, histology, extracellular matrix biochemistry, and biomechanics had greater than 70% chondrocyte viability at the time of implantation regardless of preservation protocol or storage duration. OCA plugs stored at room temperature with our novel protocol had a successful outcome rate of 85% (12 successful grafts of 14) while those stored using the SOC protocol had a successful outcome rate of 28% (4 successful grafts of 14; Figure 1).
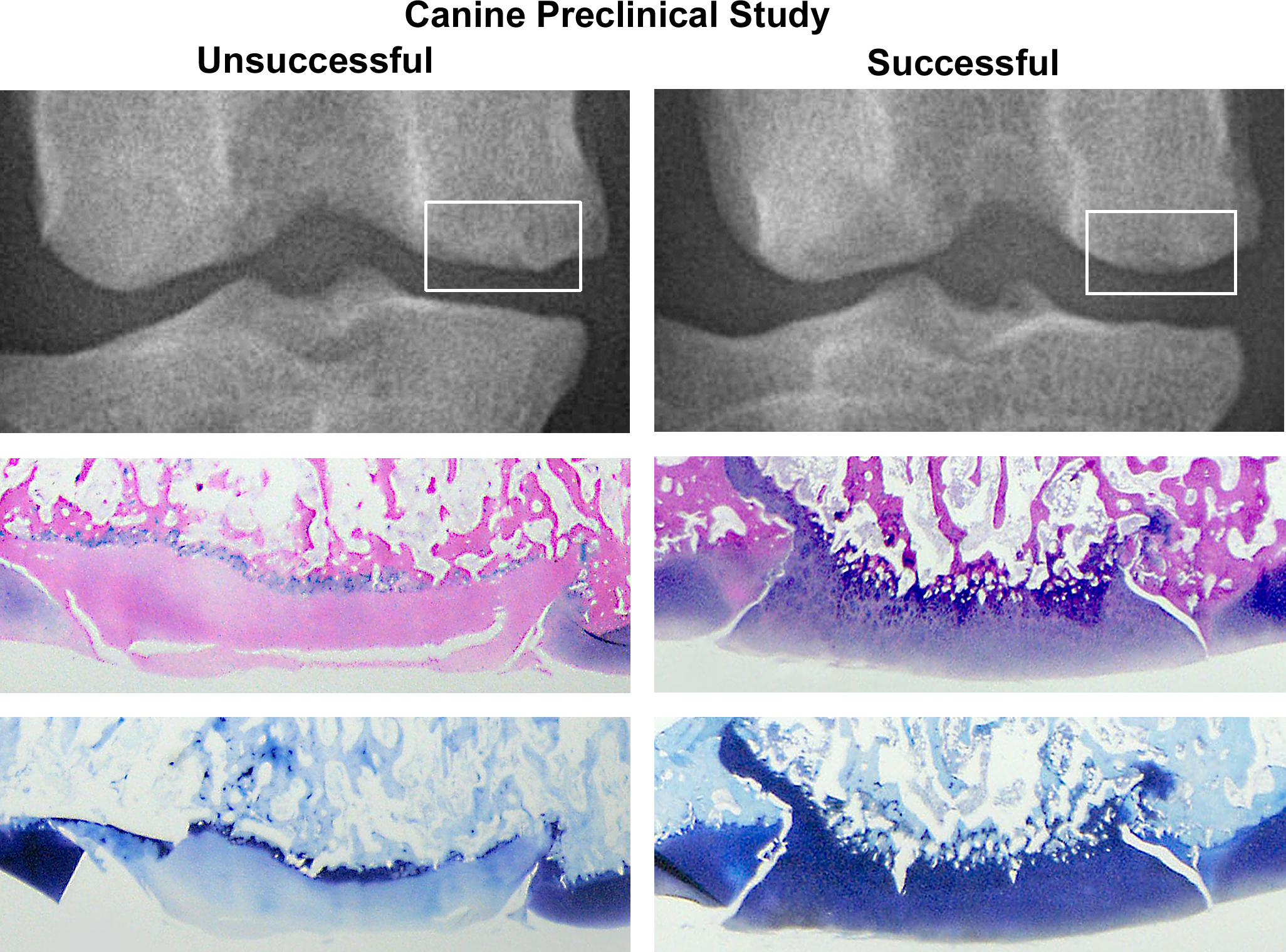
Radiographic and histologic comparison between an unsuccessful osteochondral allograft (OCA) preserved 60 days with a current standard of care protocol and a successful OCA preserved 60 days in our novel protocol (MOPS™) 6 months after press-fit implantation in femoral condyles from a preclinical canine model study (Cook et al., 2016; Cook et al., 2014). Radiographically, an unsuccessful OCA in medial femoral condyle shows mild articular surface irregularities (box) while a successful OCA in medial femoral condyle shows smooth articular surface (box). Histologically, an unsuccessful OCA graft is characterized by an irregular surface, loss of viable chondrocytes, and depletion of toluidine blue staining in cartilage while a successful OCA graft has a smooth surface, intact chondrocytes, and abundant glycosaminoglycan (hematoxylin and eosin and toluidine blue). MOPS™ = Missouri Osteochondral Allograft Preservation System.
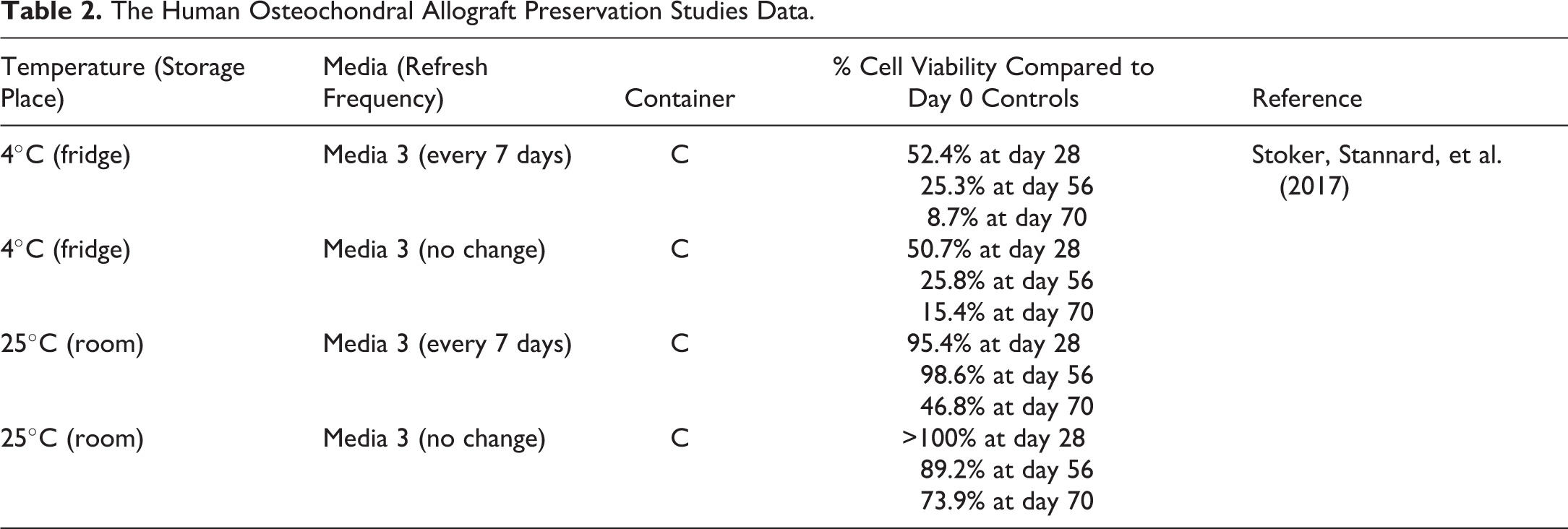
Based on the results of the canine studies, patent protection has been achieved and continues to be pursued for the novel OCA preservation system, which has also been trademarked as the MOPS™, acronym for Missouri Osteochondral Allograft Preservation System. A. M. Stoker, Stannard, et al. (2017) designed and conducted a study to evaluate the effectiveness of MOPS for maintaining essential chondrocyte viability in human femoral condyle OCAs. This study revealed that human OCAs stored using MOPS at room temperature maintained excellent VCD, with mean %VCD at 95.4% of day 0 controls at day 28 and 98.6% at day 56 with weekly media changes and mean %VCD of 102.9% of day 0 controls at day 28 and 89.2% at day 56 without media changes (Table 2). Moreover, in OCAs stored using MOPS protocol without media changes, %VCD was maintained above the minimum sufficient viability level at day 70 (73.9%). Importantly, all OCAs were negative for microbial growth at all time points when tested according to the current U.S. Pharmacopeia <71> protocol (www.pharmawebinars.com/usp-71-pharma-webinars/), and extracellular matrix composition and biomechanical properties were maintained in MOPS OCAs for 70 days after procurement (A. M. Stoker, Stannard, et al. 2017).
The Human Osteochondral Allograft Preservation Studies Data.
Osteochondral grafts including MOPS OCAs are regulated under U.S. Food and Drug Administration section 361 of the Public Health Service Act and 21 Code of Federal Regulation 1271 that defines human cells, tissues, or cellular- or tissue-based products. MOPS technology is licensed to Musculoskeletal Transplant Foundation for use in preparation of OCAs. In conjunction with the implementation of MOPS grafts at our center, we have also optimized the surgical techniques for graft creation and implantation to include patient-specific graft contouring, autogenous bone marrow aspirate concentrate (BMC)-treated donor bone (Oladeji et al. 2017; Stoker, Baumann, et al. 2017), and treatment-specific postoperative rehabilitation. Patients receiving MOPS OCA transplants are enrolled in a dedicated IRB-approved registry for prospective assessment of outcomes. Outcome assessments include VAS pain, IKDC, SANE, Tegner, and PROMIS Mobility at 6 months and yearly after surgery. All complications and reoperations are recorded. OCA survival is determined based on maintenance of acceptable levels of pain and function and/or need for revision surgery. To date, mean %VCD for MOPS allografts transplanted into this cohort of patients (n = 42) is 98% (range 86–100%). Mean age of patients is 34.8 years. Mean follow-up is 9 months with a range of 6–15 months. At 6 months postoperatively, all outcome measures were improved from preoperative status with pain and SANE scores reaching significantly (p < .05) higher levels. At 1 year postoperatively, all outcome measures were significantly (p < .05) improved compared to preoperative status. Revision surgery has been required in 4.8% of patients, and 11.9% of patients required minor reoperations for lysis of adhesions to restore range of motion and/or for screw removal. No patients have required TJR, making initial MOPS grafts survival 95.2%, and no surgery-related infections, deep vein thromboses, or pulmonary emboli have been encountered in this cohort. All patients returned to work by 6 months after surgery and 83% returned to preinjury level of activity (Tegner) by 1 year after surgery. All outcome assessments are ongoing as part of the registry. Figure 2 illustrates our One Health–One Medicine approach to develop a novel OCA transplantation protocol.
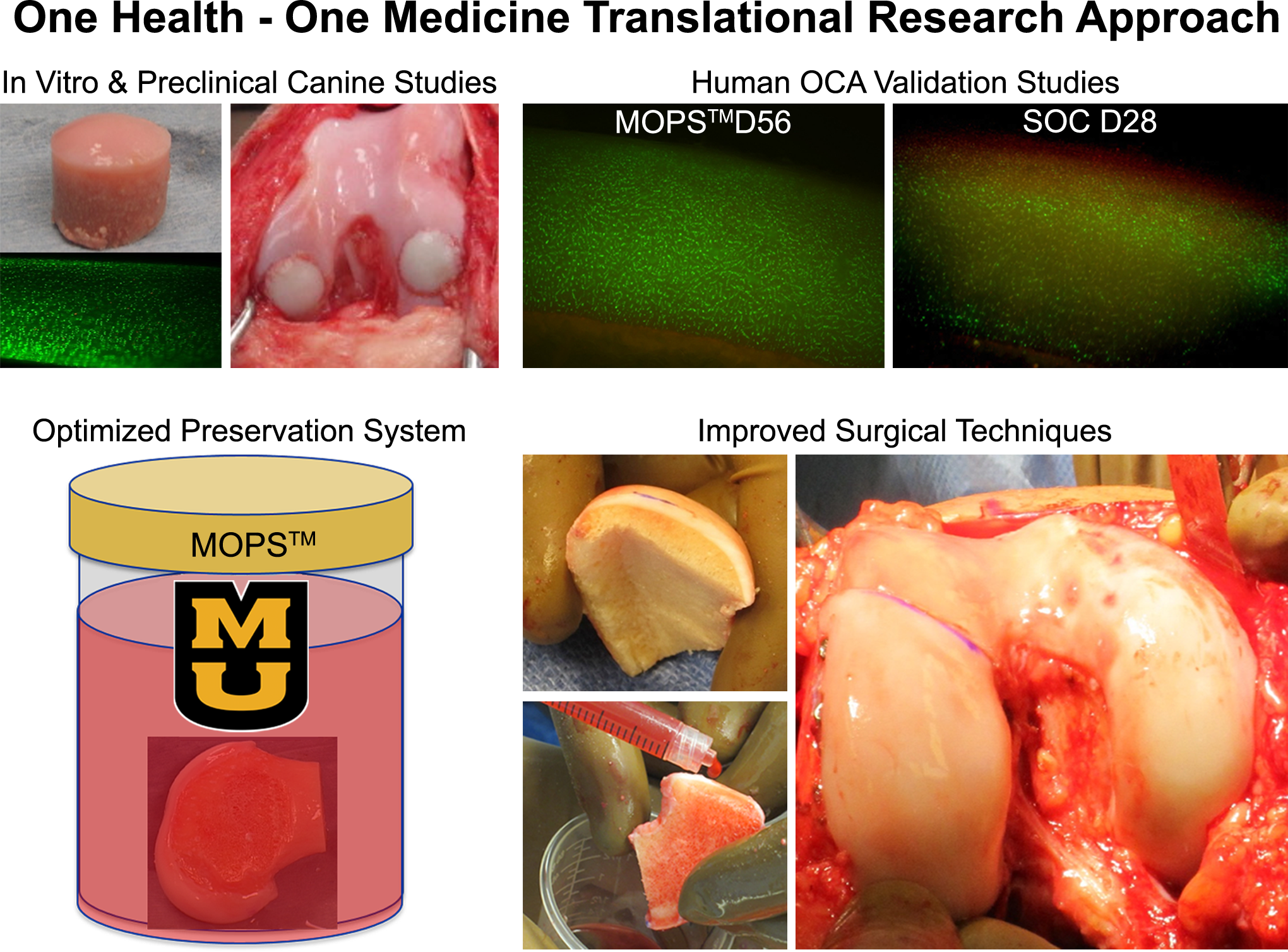
Illustration summary of One Health–One Medicine approach and development of a novel osteochondral allograft (OCA) transplantation protocol in our laboratory. OCA preservation method was optimized by in vitro studies using canine tissues and validated by preclinical canine studies. Based on the results of those studies, the novel OCA preservation method, MOPS™ protocol, was applied to human tissues for validation. Photomicrographs for live and dead staining (Calcein AM-Ethidium homodimer) show excellent cell viability in human femoral condyle OCA preserved in MOPS at day 56 of storage while markedly decreased viable chondrocytes in OCA preserved with a current standard of care protocol at day 28 after procurement. Risks of bone collapse and delayed union associated with large OCA grafts have been reduced in our center by optimizing graft thickness and geometry. Osseous integration potential for OCAs is enhanced, and untoward immune responses are mitigated by treating subchondral bone of OCAs with autogenous bone marrow aspirate concentrate. MOPS™ = Missouri Osteochondral Allograft Preservation System.
Discussion
Historically, fresh OCAs were kept in lactated Ringer’s solution and were transplanted within a week after procurement and were consistently associated with highly successful outcomes (Chu et al. 1999; Gross et al. 1983). However, currently, OCAs are typically stored in culture media at 4°C with approximately 2 weeks prior to implantation (LaPrade et al. 2009) in order to assure sterility. Therefore, OCAs in the United States are transplanted after 2–4 weeks of tissue bank storage, averaging 24 days and ranging between 15 and 43 days after procurement (Gortz and Bugbee 2006; LaPrade et al. 2009; McCulloch et al. 2007; R. J. Williams et al. 2007). Multiple studies have shown that human OACs stored using the standard method for U.S. tissue banks (i.e., stored in culture media at 4°C) undergo significant decreases in chondrocyte viability to below the 70% threshold that is considered essential for maximizing clinical success and transplant survivorship within 28 days or less after procurement. (Allen et al. 2005; Ball et al. 2004; A. M. Stoker, Stannard, et al. 2017; S. K. Williams et al. 2003). This narrow transplantation life window for effective surgical implantation severely limits clinical use (quantity) and success (quality) of OCAs.
Our translational research approach to current OCA transplantation limitations yielded a clinically applicable OCA preservation method, MOPS, which has been validated, patented, and meets regulatory requirements for use in patients. The MOPS protocol for preservation of OCAs in a proprietary solution and container at room temperature (∼25°C) maintained sufficient chondrocyte viability, articular cartilage extracellular matrix composition, and material properties for at least 60 days after procurement and resulted in functional outcomes in a preclinical canine model (Cook et al. 2014, 2016). In addition, client-owned clinical canine patients treated with MOPS-preserved OCAs have realized consistently successful long-term outcomes (Cook, Cook, and Kuroki 2010).
MOPS-preserved human femoral condyle OCAs maintained day-0 VCD levels for at least 56 days after procurement without media changes (A. M. Stoker, Stannard, et al. 2017). All human OCAs stored with MOPS at room temperature were negative for microbial growth and maintained extracellular matrix composition and biomechanical properties for more than 56 days after procurement as well. Initial clinical outcomes in human patients treated with MOPS-preserved allografts, BMC-treated donor bone, and treatment-specific postoperative rehabilitation showed low complication rates and morbidity and significant improvements in levels of pain and function at 1 year after surgery. However, continued assessment is necessary to determine whether these results can be sustained long term.
The potential adverse immune response to OCAs is an important safety consideration for clinical use of these allografts. Hyaline cartilage is considered to be relatively immunoprivileged due to chondrocytes being masked from host immune surveillance by their abundant extracellular matrix (Gortz and Bugbee 2006; Langer and Gross 1974; Langer et al. 1978), and the transplanted bone is irrigated to remove antigenic cells and proteins. The concept of OCA immunoprivilege has been supported by extensive evidence of good clinical outcomes after OCA transplantations despite no human leukocyte antigen (HLA) or ABO blood-type matching between donor and recipient (Hunt et al. 2014). In addition, patients do not require immunosuppressive treatments to avoid graft rejection after OCA implantation. However, leukocyte antigen sensitization of the recipient has been demonstrated in human patients (Hunt et al. 2014; Sirlin et al. 2001) and in a canine model (Stevenson 1987); yet, no clinical differences in outcomes were observed between 33 patients with positive anti-HLA antibody and 34 patients with negative anti-HLA antibody in a case-control study (Hunt et al. 2014). As such, the roles and clinical significance of subrejection immunologic responses to OCAs are not fully understood and warrant further investigation. The addition of autogenous BMC to the osseous portion of OCAs for all patients at our center is designed in part to mitigate any untoward immune responses, further optimizing outcomes after OCA transplantation (Oladeji et al. 2017).
This body of research has validated MOPS for significantly increasing effective storage duration of OCAs and significantly improving chondrocyte viability in stored OCAs at room temperature without changing a proprietary media in a proprietary container, which combined can profoundly increase the number of grafts that can be safely transplanted into eligible patients. These improvements will allow surgeons to have increased confidence in the quality of grafts they are transplanting, provide tissue banks and graft coordinators with a much longer period of time for matching and delivering OCAs, and offer patients higher chances for highly functional long-term outcomes and graft survivorship, resulting in decreased donor tissue waste and related financial costs. Although approximately 30,000 donors provide tissue for transplant in the United States in each year (www.aatb.org), the demand for osteochondral tissue will continue to far exceed supply, even with the use of MOPS. The number of total knee replacement surgeries alone in the United States is rapidly increasing from approximately 263,000 cases in 1999 to 616,000 cases in 2008 (Bernstein and Derman 2014). The demand for knee replacements is estimated to exponentially increase to 3.48 million cases by the year 2030 (Kurtz et al. 2007). So, although this body of research has provided important progress in addressing quantity, quality, and technique limitations for successful OCA transplantation, development and validation of effective preventative, preservation, and tissue engineering strategies for articular cartilage disorders in clinical canine and human patients are of critical importance. The authors suggest that a One Health–One Medicine comparative and translational approach to addressing this critical unmet need can prove successful.
Footnotes
Authors’ Contribution
All authors (KK, AS, JS, CB, CC, FP, JC) contributed to conception or design; data acquisition, analysis, or interpretation; drafted the manuscript; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: One or more of the authors (AMS and JCL) are patent holders for the Missouri Osteochondral Allograft Preservation System (MOPS) and receive associated royalties. One of the authors (JLC) is a board member of the Musculoskeletal Transplant Foundation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Portions of this work were funded by the Musculoskeletal Transplant Foundation, Synthes, the Thompson Laboratory for Regenerative Orthopaedics, and the University of Missouri Department of Orthopaedic Surgery.
