Abstract
New medullary bone formation has been observed in rats administered a variety of antineoplastic compounds. Similar effects reported in rats administered granulocyte colony-stimulating factor (G-CSF) were attributed to exaggerated pharmacology of G-CSF as a cytokine and growth factor, resulting in stromal proliferation in addition to the intended hematopoietic effects. Similar phenomena of marrow stromal change are reported among other species in association with various growth factors. Case study summaries of test item–related histopathologic changes in bone marrow, reflecting trabecular and/or endosteal new bone formation, are presented. In each of these cases, it was concluded that the new medullary bone and stromal proliferation did not reflect a primary target–related toxicity; rather, the mesenchymal changes were attributed to nonspecific, secondary effects of cytokines elaborated in response to primary cytotoxic effects on hematopoietic cells with subsequent impact on circulating blood cells. The common features associated with marrow stromal changes in the case studies, as well as with a variety of pharmacologic compounds across several species described in the literature, are hematologic effects and/or changes in growth factor levels and cytokine expression.
In rodent preclinical studies with pronounced effects on the hematopoietic system, bone marrow stromal proliferative alterations have occasionally been observed. These changes have necessitated further consideration in risk assessments of the associated test compounds. Herein, illustrative case studies are presented with a literature-based rationale for considering that these stromal effects are attributable to growth factor and/or cytokine expression as a secondary manifestation of injury to hematopoietic cells. These case examples were extracted from general toxicity studies conducted to support drug development, in which the primary objective for each of the studies was to characterize the overall toxicity profile of the test compound and not to provide investigative end points into mechanisms of toxicity. In each of these cases, it was concluded that the new medullary bone and stromal proliferation did not reflect a primary target–related toxicity; rather, the mesenchymal changes were attributed to nonspecific, secondary effects elaborated in response to primary cytotoxic effects on hematopoietic cells. The common features associated with marrow stromal changes in the case examples, as well as with a variety of pharmacologic compounds across several species, are hematologic effects and/or changes in growth factor levels and cytokine expression.
Case Example #1: New Bone Deposition and Bone Marrow Stromal Changes in Rats
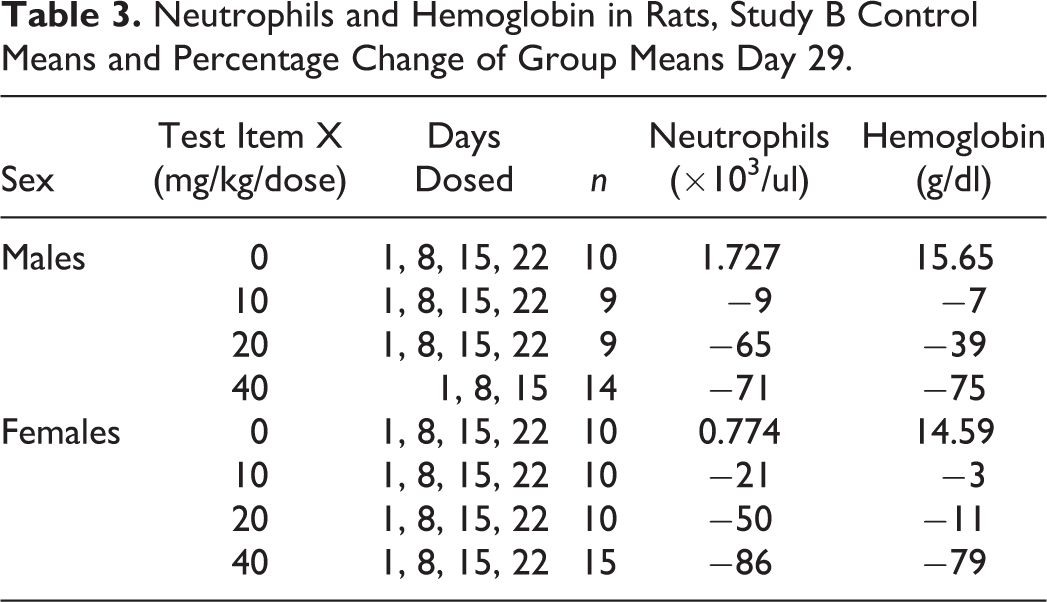
In studies A and B, rats that were administered an intravenous protein–based biotherapeutic test item (compound X) for up to 4 weekly doses of 40 mg/kg or higher had variable occurrences of endosteal new bone deposition (Figure 1). These dose levels were also associated with bone marrow hypocellularity and pronounced effects on numbers of circulating neutrophils (mean values decreased up to 97% compared to control mean values; individual value as low as 0.05 × 103/μl) and/or red blood cell mass (hemoglobin decreased up to 79%; as low as 1.5 g/dl) (Tables 1 –4).
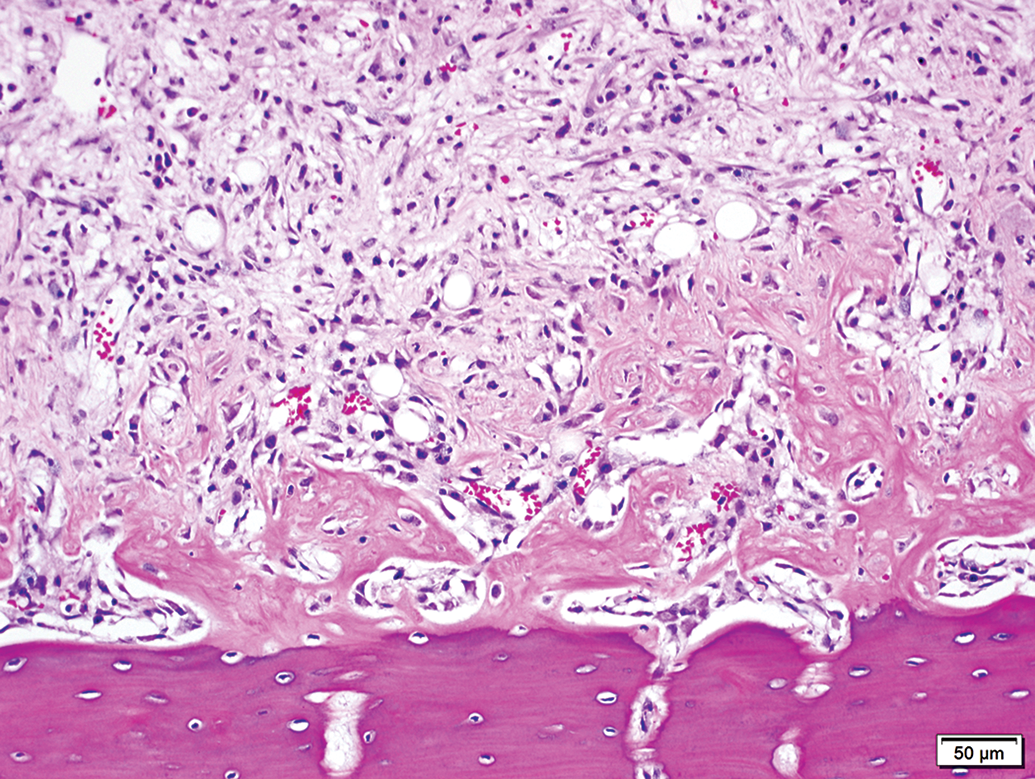
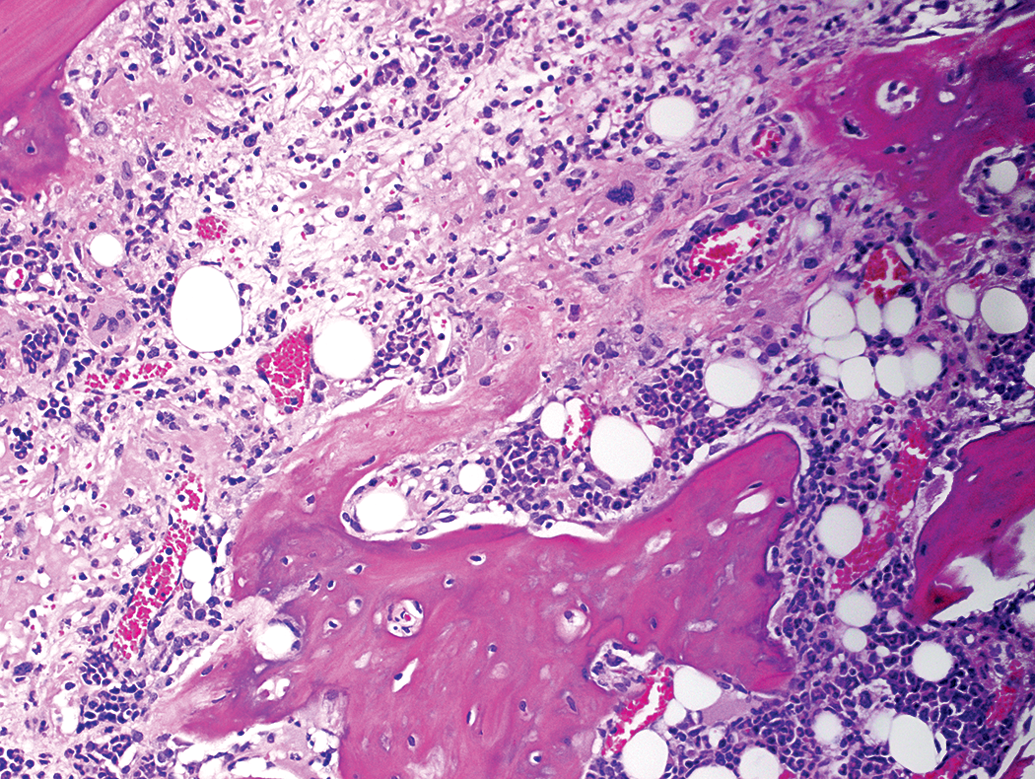
Femoral diaphysis from a male rat (study B) administered 3 weekly intravenous doses of compound X at 40 mg/kg. Endosteal new bone deposition (middle portion of the image) is characterized by loose, pale osteoid surrounding numerous osteocytes and lined by osteoblasts, contrasting with mature cortical bone (below). Medullary adipocytes are largely replaced by stromal tissue having pale basophilic matrix and increased cellularity (upper portion of the image). Marked hematopoietic hypocellularity. Concurrent hematology: neutrophils 0.27 × 103/μl; hemoglobin 4.7 g/dl. Hematoxylin and eosin stain.
Neutrophils in Rats, Study A Control Means and Percentage Change of Group Means Day 6, Following a Single Dose.
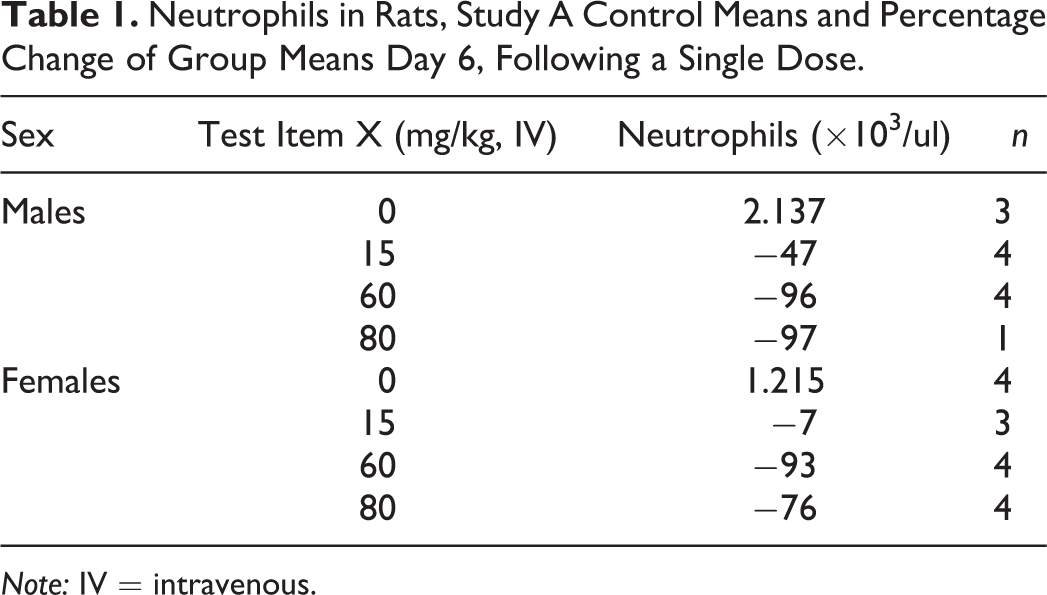
Note: IV = intravenous.
Incidences of Bone Marrow Histopathology in Rats, Day 29, Study A.
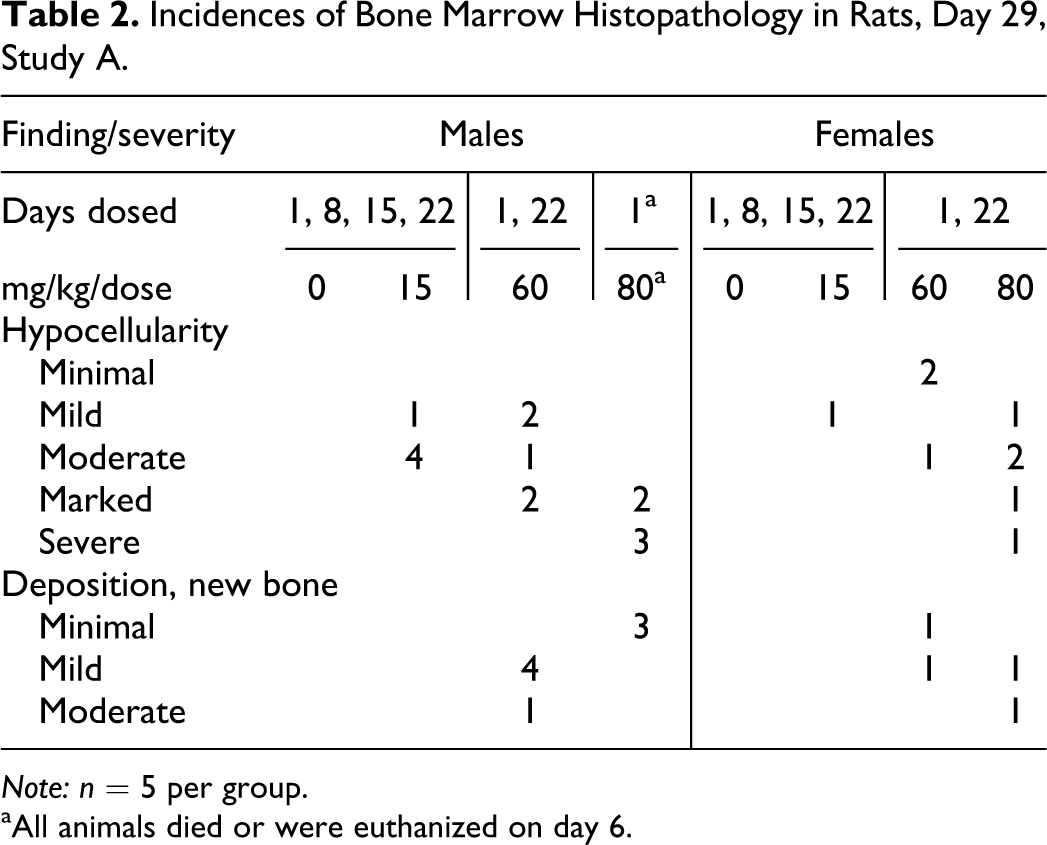
Note: n = 5 per group.
aAll animals died or were euthanized on day 6.
Neutrophils and Hemoglobin in Rats, Study B Control Means and Percentage Change of Group Means Day 29.
Incidences of Bone Marrow Histopathology in Rats, Day 29, Study B.
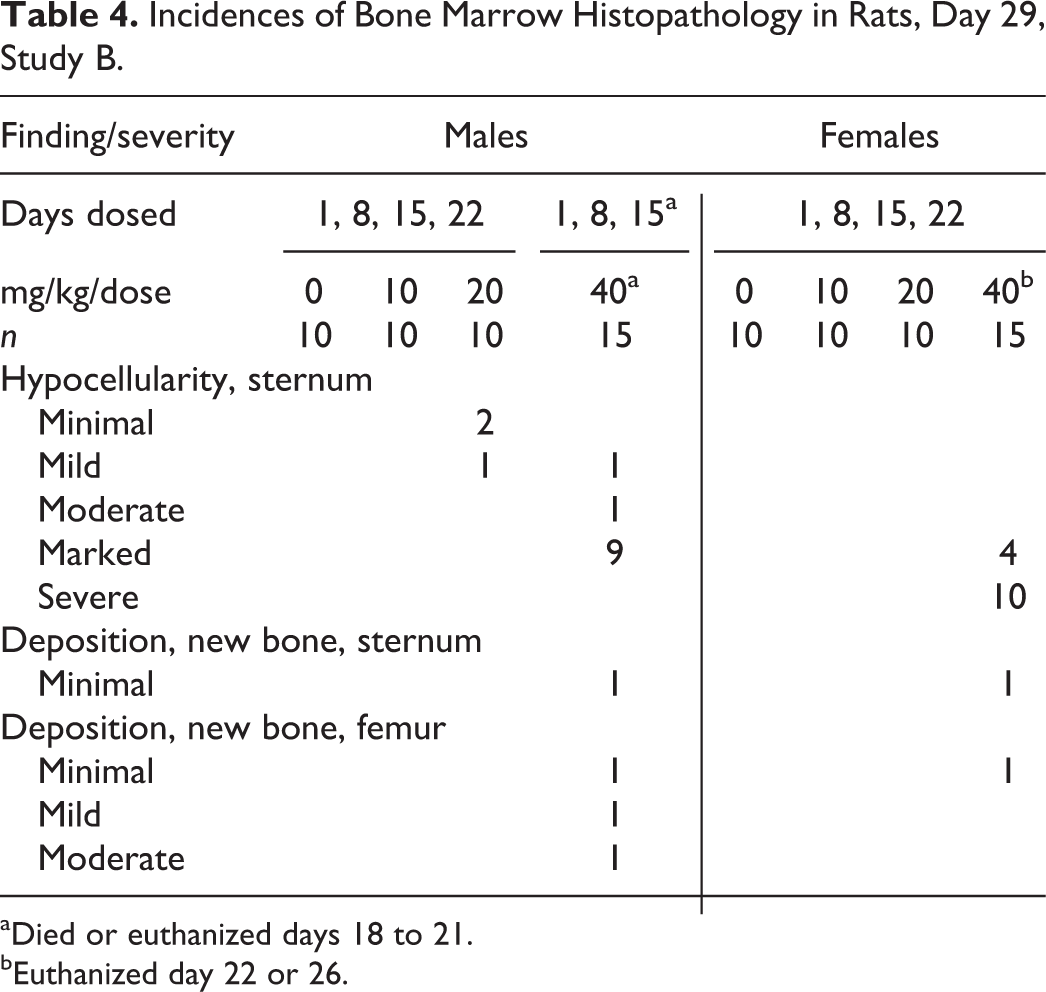
aDied or euthanized days 18 to 21.
bEuthanized day 22 or 26.
Case Example #2: New Bone Deposition and Bone Marrow Stromal Changes in Mice
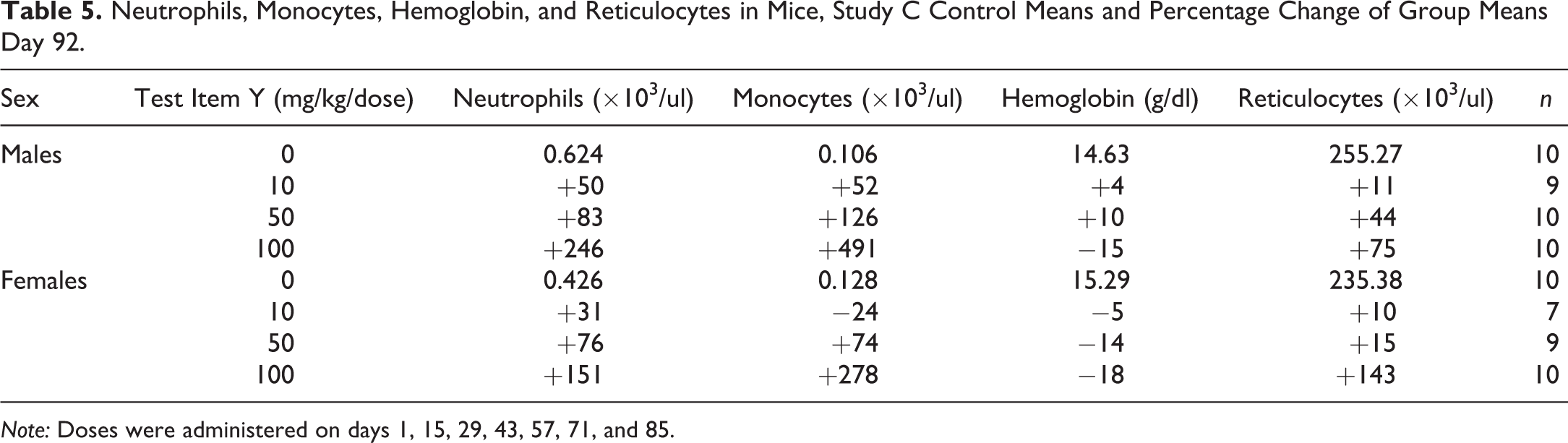
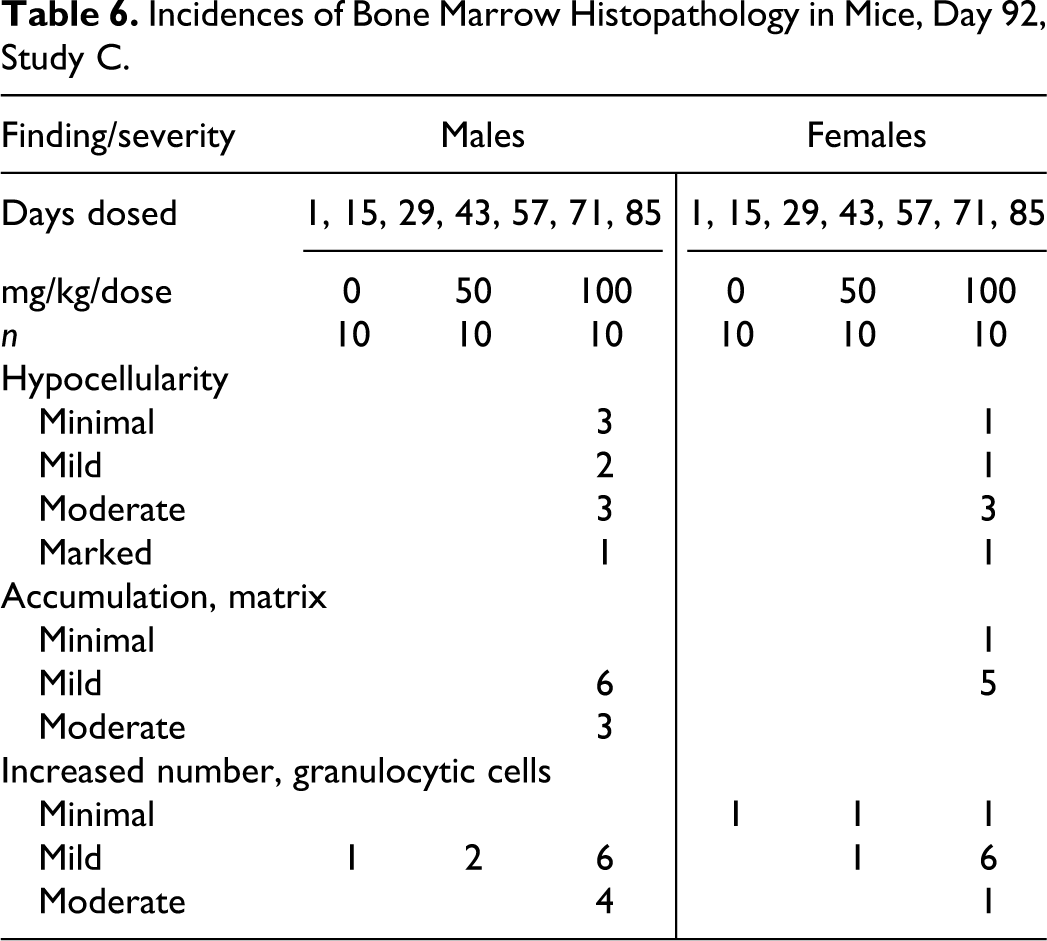
A study (C) on mice with 7 biweekly intravenous administrations of another protein-based biotherapeutic test item (compound Y) had accumulation of eosinophilic matrix (Figure 2) within bone marrow at the highest dose of 100 mg/kg, accompanied by pronounced increases in circulating neutrophils, monocytes, and reticulocytes. Reticulocyte increases were in response to decreased hemoglobin levels (Table 5). Animals having marrow matrix accumulation also had hematopoietic hypocellularity with variably increased representation of granulocytic precursors (Table 6).
Distal femur from a female mouse (study C) administered 7 biweekly intravenous doses of compound Y at 100 mg/kg/dose. Accumulation of eosinophilic matrix and new bone production associated with variable granulocytic cellularity.
Neutrophils, Monocytes, Hemoglobin, and Reticulocytes in Mice, Study C Control Means and Percentage Change of Group Means Day 92.
Note: Doses were administered on days 1, 15, 29, 43, 57, 71, and 85.
Incidences of Bone Marrow Histopathology in Mice, Day 92, Study C.
Bone and/or Stromal Changes Have Been Observed in a Variety of Species Administered Pharmacologic Compounds or Having Altered Cytokine Expression
New bone deposition (endosteal proliferation, osteosclerosis) has been observed among rats administered a variety of chemically and pharmacologically distinct small molecule antineoplastic compounds (Courtney et al. 1991; Norbury and Allen 1981). Findings are variably described as woven bone with myelostromal proliferation or osteopetrosis. Additionally, rats administered recombinant human granulocyte colony-stimulating factor (rhG-CSF) manifest medullary bone changes including trabecular and endosteal new bone deposition (Suzuki et al. 1997). However, mice treated with rhG-CSF do not manifest similar effects in bone (Suzuki et al. 2008); rather, bone marrow stromal changes in mice are often associated with increased thrombopoietin and abnormal megakaryocytes and/or platelet production. Mice overexpressing thrombopoietin develop megakaryocytic hyperplasia and myelofibrosis and osteosclerosis of the marrow cavity (Villeval et al. 1997; Yan at al. 1996). Cytokines, most notably transforming growth factor-beta (TGF-β), have been implicated in the pathogenesis of the marrow stromal effects (Chagraoui et al. 2002; Zingariello et al. 2013; Yanigida et al. 1997). Similar phenomena of marrow stromal change are reported among nonrodent species but associated with different hematologic phenomena or growth factors compared to mice or rats. Dogs administered recombinant human erythropoietin develop myelofibrosis (Bader et al. 1992). Osteosclerosis and/or myelofibrosis are likewise observed in dogs having spontaneous disease associated with anemia (Reagan 1993) including congenital hemolytic anemia (Searcy, Tasker, and Miller 1979; Prasse et al. 1975) and immune-mediated anemia (Stokol, Blue, and French 2000).
In humans, myelofibrosis (bone marrow deposition of reticulin and/or collagen) and bone marrow osteogenesis are associated with abnormal megakaryocyte development (Ciurea et al. 2007) and increased release of TGF-β as well as other cytokines (platelet-derived growth factor, calmodulin, vascular endothelial growth factor, and basic fibroblastic growth factor; Tefferi, 2000; Papadantonakis, Matsuura, and Ravid 2012; Martyre et al. 1997).
Common Feature Is Resemblance to Cytokine and Growth Factor–induced Proliferative Bone Marrow Lesions
In the case studies presented above, the effects on bone within the marrow cavity were considered secondary to test item–related effects on hematopoietic cells in the bone marrow. Although hematopoietic growth factors and cytokine levels were not evaluated in these studies, hematology and bone marrow histopathology provided indirect evidence for these influences. These secondary effects on the mesenchymal compartment of the marrow cavity are considered nonspecific, as they occur in association with a variety of pharmacologic compounds and across several species. The common features associated with marrow stromal changes in the case examples and literature citations, across species, are hematologic effects (pronounced hematopoietic stimulus) and/or changes in growth factor levels and cytokine expression. Due to the dynamic nature of hematopoietic tissue, hematologic effects may manifest as increases or decreases in circulating cell numbers at a given time point. There appear to be species differences in the hematopoietic lineage and hematopoietic growth factor most associated with a marrow stromal response and the precise pathogenesis of the increased stromal cell and matrix development is not certain, however, profibrotic and pro-osteogenic cytokines are frequently implicated.
Pharmacologic compounds associated with induction of marrow stromal changes in preclinical studies are not expected to have similar manifestations in clinical applications. The underlying hematologic circumstances for the marrow stromal phenomenon are readily monitorable in the clinical setting where the extreme hematologic effects observed in preclinical studies would not be permitted to develop. Species sensitivities to particular hematologic and cytokine associations further diminish direct translatability of preclinical observations to human relevance.
In light of the limited data available in the cases presented, this brief communication is not intended as a rigorous demonstration of the pathogeneses of the observed morphologic effects in bone marrow; rather, the intent is to summarize select case observations in the context of pertinent literature. Influences of hematopoietic growth factors and cytokines on the complex bone marrow stromal environment and its interactions with hematopoietic precursor cells provide a plausible explanation for the observed mesenchymal cellular and matrix alterations associated with administration of certain pharmacologic compounds. In our experience, these arguments have been accepted by regulatory reviewers in risk assessment evaluations.
Footnotes
Authors’ Note
All authors are employees of AbbVie. The design and study conduct were provided by AbbVie. AbbVie participated in the interpretation of data, review, and approval of the publication. All animal studies were reviewed and approved by the AbbVie’s Lake County Institutional Animal Care and Use Committee. Animal studies were conducted in an American Association for Laboratory Animal Care (AALAC)-accredited program, and veterinary support was available to ensure appropriate animal care.
Acknowledgments
The authors would like to acknowledge the contributions of Karen Bodie, Donna Davila, Tracy Henriques, Helga Lorenz, and Ronnie Yeager for the conduct of the case example studies presented herein.
Author Contribution
Authors contributed to conception or design (LL, ML, KB, SF, KW); data acquisition, analysis, or interpretation (ML, KB, SF, KW); drafting the manuscript (KW); and critically revising the manuscript (LL, ML, KB, SF). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this research was provided by AbbVie.
