Abstract
The use of immunohistochemical (IHC) staining in determining and/or confirming the cellular origin of poorly differentiated sarcomas was evaluated in this study. Sarcomatous neoplasms were evaluated in a research study conducted in 2 strains of p53+/− haploinsufficient mice. The most common neoplasms were undifferentiated sarcomas, followed by osteosarcomas and rhabdomyosarcomas (RMSs). The RMSs were poorly differentiated and appeared similar to the pleomorphic, or adult type, RMS of humans. All sarcomas stained positive by IHC for the mesenchymal cell intermediate filament vimentin. The RMSs were identified by positive IHC staining for myogenin, a transcription factor specific to skeletal muscle. Osteosarcomas were easily identifiable on hematoxylin and eosin–stained slides; no generally accepted IHC stain specific for bone is presently available. Some of the undifferentiated sarcomas contained numerous macrophages that stained positive for F4/80, a macrophage marker; the positive-staining cells were considered to be infiltrating macrophages. One-third of the neoplasms observed in this study were associated with subcutaneous implanted electronic microchips used for animal identification. Based upon histopathologic evaluation and IHC staining, it was not possible to distinguish neoplasms associated with subcutaneous microchips from neoplasms not associated with microchips.
Genetically modified mice have been used as alternative models for carcinogenicity testing. Since neoplasms arise more quickly in these animals, carcinogenicity assays can be of shorter duration than the standard 2-yr carcinogenicity assay and may allow the use of fewer animals. In addition, selection of an appropriate animal model may lead to findings more applicable to humans and may provide information concerning the mechanisms of carcinogenicity (Pritchard et al. 2003). One useful model is the Trp53+/− heterozygous mouse, in which 1 allele of the Trp53 tumor suppressor gene has been inactivated. These mice develop a variety of spontaneous neoplasms, the most common of which are lymphomas and a variety of subcutaneous sarcomas (Storer et al. 2001). Histologically, the sarcomas have shown differentiation to fibrosarcoma, rhabdomyosarcoma (RMS), hemangiosarcoma, or histiocytic sarcoma or lacked differentiation toward any specific cell type and were considered undifferentiated sarcomas. However, these sarcomas can be poorly differentiated and pose a diagnostic challenge as to the cell of origin. Osteosarcomas may also develop in Trp53+/− mice, although generally at a lower frequency than subcutaneous sarcomas, and a few other neoplasms also occur sporadically (Floyd et al. 2002; Storer et al. 2001).
In a research study conducted in two Trp53+/− mouse strains, several poorly differentiated subcutaneous sarcomas were observed. Some were considered sufficiently differentiated to warrant a diagnosis of osteosarcoma or RMS based upon histopathologic examination of the hematoxylin and eosin (H&E)-stained slides alone, although there was a degree of uncertainty in a few of the RMS diagnoses. Others were poorly differentiated and could not be further classified based on examination of the H&E-stained slides and were diagnosed simply as sarcoma. In the current study, immunohistochemical (IHC) evaluations were performed on each of the sarcomas in an attempt to further determine the cell of origin. The IHC evaluations generally confirmed the diagnoses made from examination of the H&E-stained slides, thereby increasing the confidence in the diagnoses, and also allowed more accurate determination of the cell of origin of some of the poorly differentiated neoplasms. A finding of interest was that all the RMSs were poorly differentiated and were histomorphologically similar to the pleomorphic, or adult type, RMS of humans.
Some of the subcutaneous neoplasms examined in this current study had been found to be closely associated with subcutaneously implanted electronic microchips, commonly used for animal identification. There is evidence that the incidence of subcutaneous sarcomas in Trp53+/− mice increases with the use of implanted microchips, with some neoplasms arising at the implantation site (Blanchard et al. 1999; Floyd et al. 2002). The close association of neoplasm formation with microchip implantation is a potential confounding factor when attempting to determine the carcinogenicity of a test article. Comparison of the IHC staining results in this current study between those of microchip-related neoplasms and neoplasms not associated with microchips revealed no differences in the IHC staining for a given tumor type.
Material and Method
Animals
The tissues evaluated were from 2 strains of Trp53 haploinsufficient (Trp53+/−) male mice (Taconic Farms, Hudson, NY); the inbred B6.129-Trp53tm1Brd strain and the C3B6F1.129-Trp53tm1Brd, an outcross between C3H/HeNTac female (model C3H-F) × B6.129-Trp53tm1Brd (model P53N12-M) homozygous Trp53 null allele male to produce the haploinsufficient F1 progeny. All mice were identified by a microchip implanted subcutaneously, at 9 to 10 weeks of age, in the dorsal neck area. Animals were approximately 50 weeks of age when euthanized. This was a non-GLP (Good Laboratory Practice) study conducted under federal guidelines for the care and use of laboratory animals and was approved by the Alion Science and Technology and National Institute of Environmental Health Sciences Animal Care and Use Committees.
Necropsy and Gross Pathology
At study termination, all mice received a complete necropsy examination and selected tissues were collected and preserved in 10% neutral buffered formalin. Early death animals were also necropsied. Tissues collected for microscopic evaluation included nose; larynx; lung; liver and gallbladder; spleen; thymus; mesenteric, mediastinal, and bronchial lymph nodes; kidney; and gross lesions.
Histopathology
All tissues were processed for histology, embedded in paraffin, sectioned at a thickness of 5 µm, stained with H&E, for histopathological evaluation, and examined via light microscopy. Following the microscopic evaluation, a pathology peer-review panel was convened to review the H&E-stained slides of all diagnosed neoplasms.
Immunohistochemistry Staining
IHC stains performed on the sarcomas included vimentin (a marker for mesenchymal cells), smooth muscle actin (a marker for smooth muscle), myogenin (a specific marker for skeletal muscle), and F4/80 (a macrophage marker used to characterize histiocytic lesions). In addition, IHC staining for cytokeratin (an epithelial marker) and factor VIII (marker for endothelial cells) were each performed on a single neoplasm. Formalin-fixed, paraffin-embedded tissue sections were deparaffinized in xylene and rehydrated through graded alcohols. Heat-induced antigen retrieval in citrate buffer (pH 6.0, Biocare Medical, Concord, CA) was done in a Decloaker® pressure chamber for 5 min at 120°C, followed by 3% hydrogen peroxide for 15 min to quench endogenous peroxidase activity. Nonspecific sites were blocked by incubating slides for 20 min with 2.5% normal horse serum, 10% normal goat serum, 10% normal rabbit serum (Vector Laboratories, Burlingame, CA), or with a Dako protein–blocking reagent (DakoCytomation Corporation, Carpinteria, CA) at room temperature. Tissues were then incubated with each of the primary antibodies listed in Table 1 for 1 hr, or 30 min (cytokeratin) at room temperature, followed by incubation with the appropriate secondary antibody for 30 min at room temperature or with an antirabbit polymer detection kit (Vector Laboratories, Burlingame, CA). For negative control tissue sections, normal rabbit IgG or rat IgG (Calbiochem®, San Diego, CA) was diluted to match the protein concentration of the primary antibodies utilized. The antigen–antibody complexes were visualized using 3,3-diaminobenzidine (DakoCytomation Corporation). Slides were then counterstained with hematoxylin, dehydrated, and coverslipped. All slides were stained manually, with exception of the vimentin slides which were stained via a Biocare Intellipath™ FLX autostainer (Biocare Medical, Pachecho, CA).
Immunohistochemical Stains.
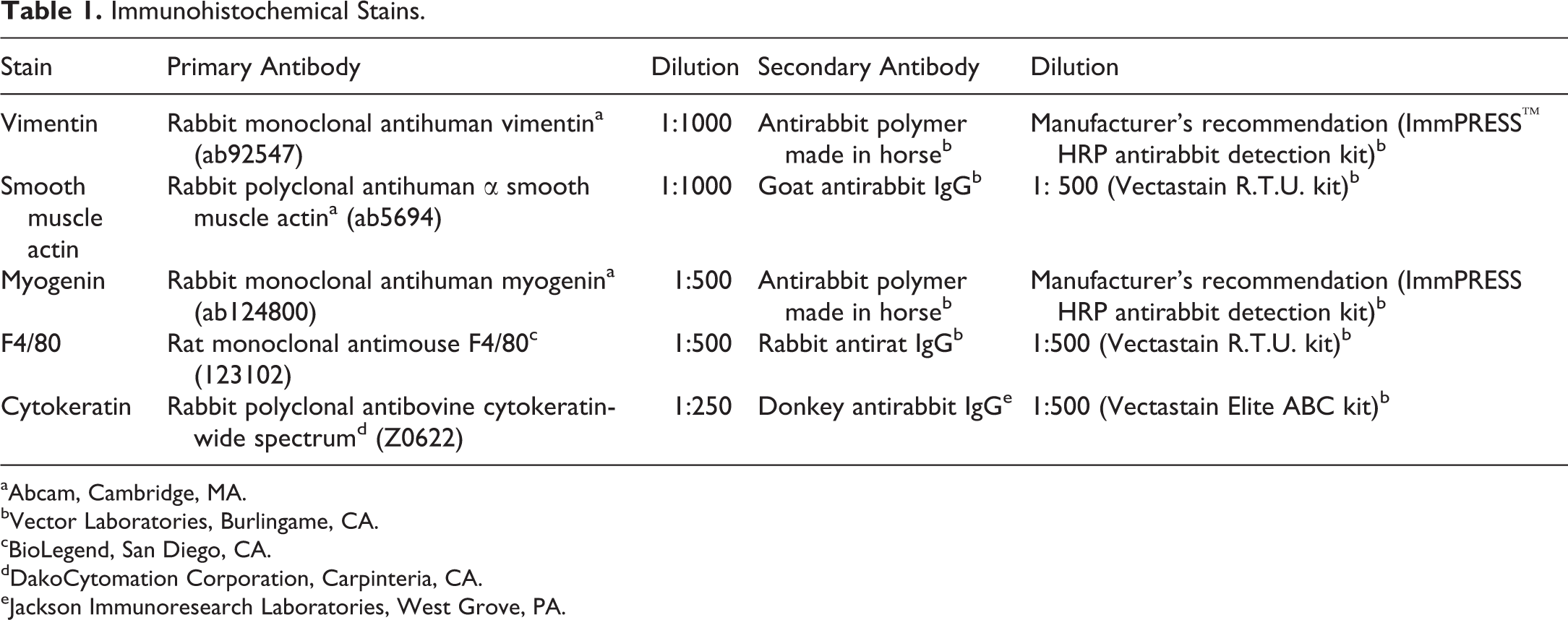
aAbcam, Cambridge, MA.
bVector Laboratories, Burlingame, CA.
cBioLegend, San Diego, CA.
dDakoCytomation Corporation, Carpinteria, CA.
eJackson Immunoresearch Laboratories, West Grove, PA.
Results
Gross Pathology
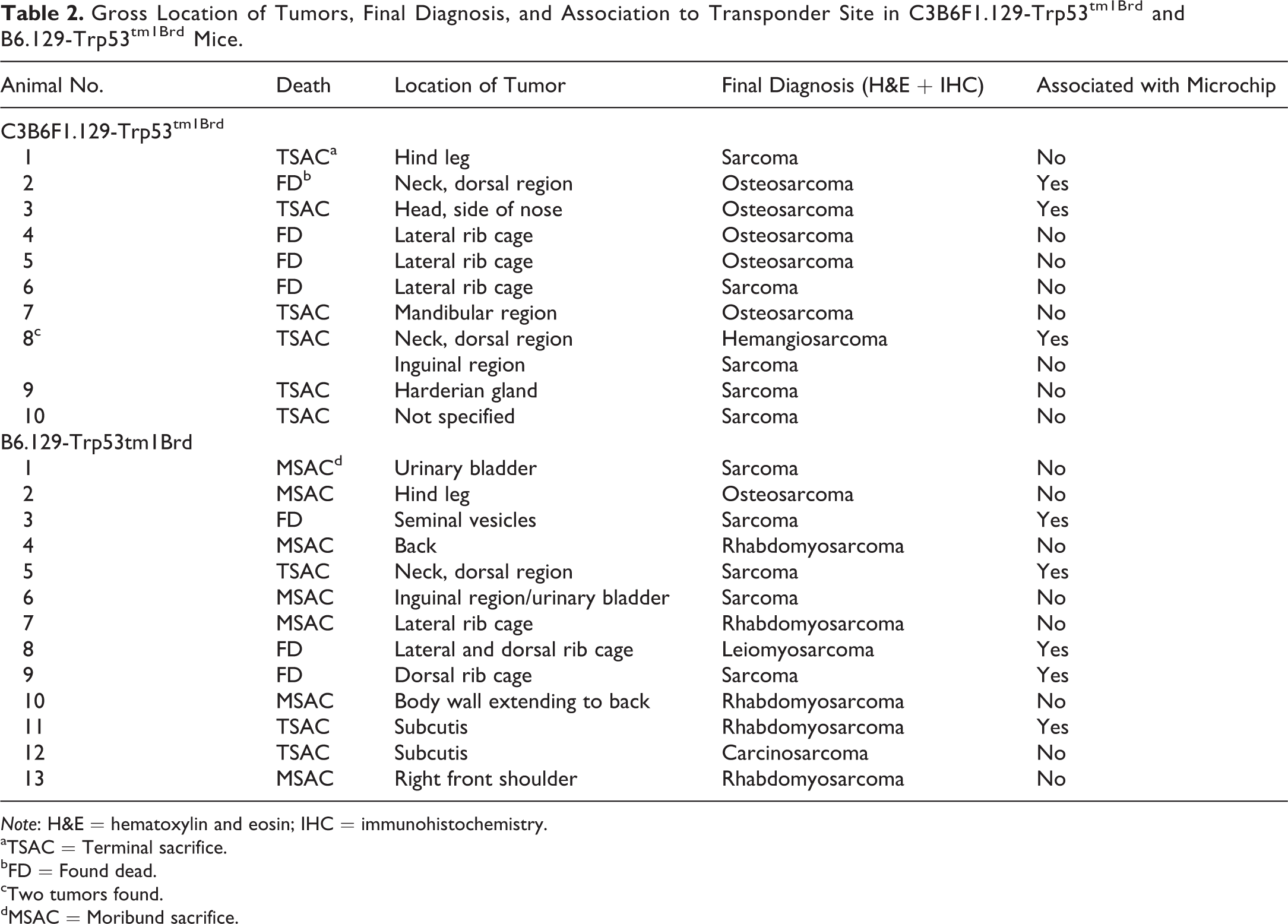
Several animals in each of the 2 Trp53+/− mouse strains had a single mass associated with the skin and subcutis; 1 C3B6F1.129-Trp53tm1Brd mouse had 2 masses. The masses were solid, discrete, and roughly spherical nodules ranging from 1 cm to 2.5 cm in diameter. Microchips were observed grossly within or adjacent to some of the neoplasms. A summary of the location of each neoplasm and whether it was associated with a microchip is listed in Table 2.
Gross Location of Tumors, Final Diagnosis, and Association to Transponder Site in C3B6F1.129-Trp53tm1Brd and B6.129-Trp53tm1Brd Mice.
Note: H&E = hematoxylin and eosin; IHC = immunohistochemistry.
aTSAC = Terminal sacrifice.
bFD = Found dead.
cTwo tumors found.
dMSAC = Moribund sacrifice.
Eleven masses were identified grossly in C3B6F1.129-Trp53tm1Brd mice. In 3 of the masses, the microchip was found within or directly adjacent to the mass. Masses in 3 animals were present on the lateral body wall over the rib cage. One of these 3 masses had invaded the thoracic cavity and involved the thymus, while another mass had invaded the abdominal cavity and involved the stomach. Invasion of the body wall by subcutaneous sarcomas in p53+/− mice was also reported by Mahler et al. (1998). Two animals had a mass on the neck, and 1 of these 2 animals also had a mass in the inguinal area. One animal had a mass on a hind leg, 1 had a mass on the side of the nose, 1 on the mandible, and 1 in a Harderian gland. The mass in 1 animal was observed at necropsy; however, its location was not recorded. Microscopically, there was no tissue around the mass to indicate the location from which it had been removed and, consequently, the tissue location was listed as tissue, not otherwise specified (NOS).
Thirteen masses were identified grossly in B6.129-Trp53tm1Brd mice. In 5 masses, the microchip was found within or directly adjacent to the mass. Masses were present on the back or on the lateral body wall over the ribcage in 7 animals; these were the most common sites of occurrence. Masses involved the urinary bladder in 2 mice and involved the urinary bladder and seminal vesicles in another animal; in all 3 mice, the masses appeared to have arisen from the body wall and invaded the abdominal cavity. One animal had a mass on a hind leg, 1 on a front shoulder, and 1 in the subcutis near the salivary gland.
Histopathology and Immunohistochemistry Findings
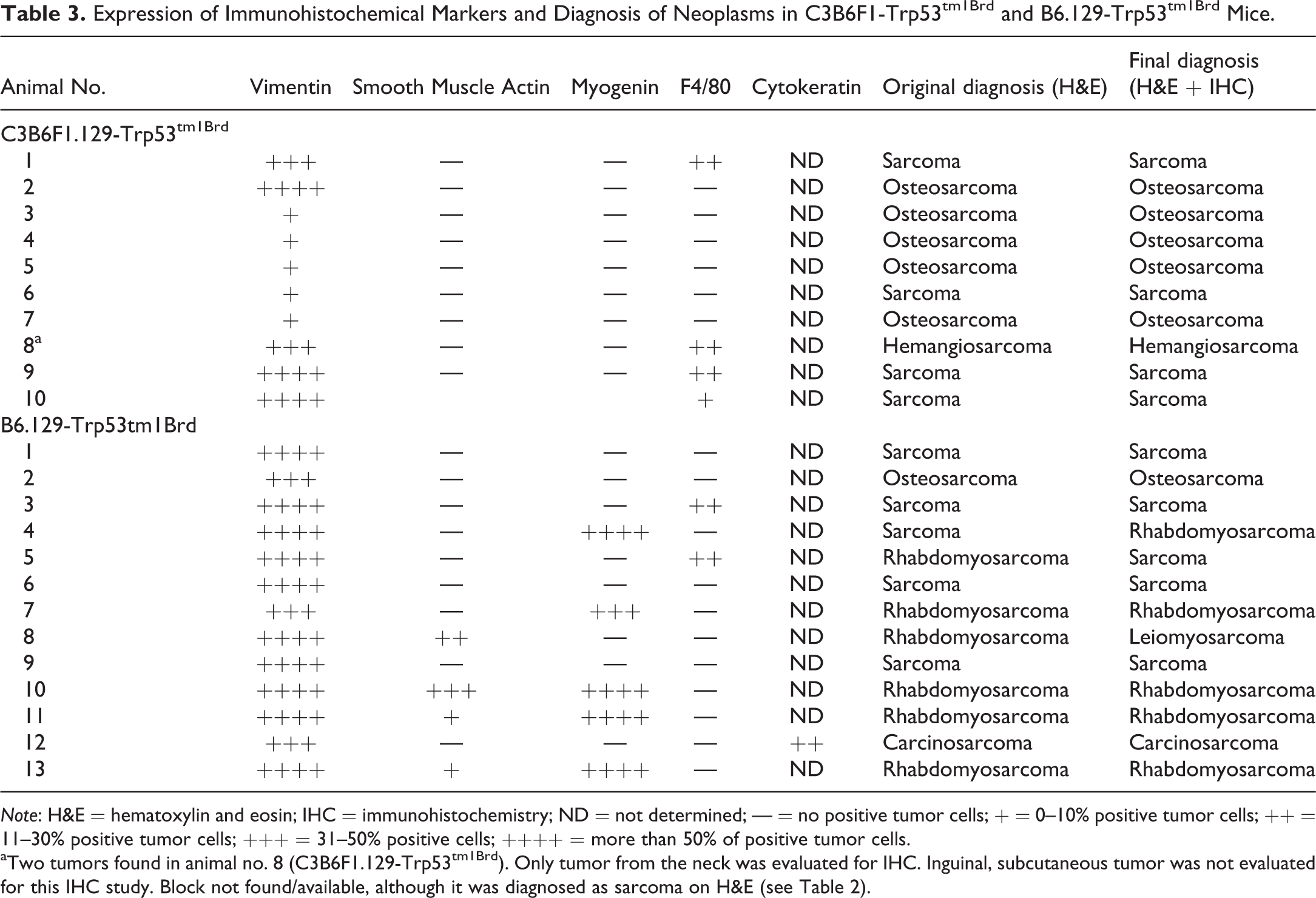
Results of the original and final histopathologic diagnoses of the neoplasms and the immunohistochemistry results are listed in Table 3. All the grossly observed masses in both strains were identified as various sarcomatous neoplasms. Osteosarcoma and sarcoma (also known as sarcoma, NOS) were the most common, followed by RMSs, with one each leiomyosarcoma, hemangiosarcoma, and carcinosarcoma. There were three cases, all occurring in B6.129-Trp53tm1Brd mice (Table 3), in which the diagnoses based on examination of the H&E slides were changed as a result of IHC staining.
Expression of Immunohistochemical Markers and Diagnosis of Neoplasms in C3B6F1-Trp53tm1Brd and B6.129-Trp53tm1Brd Mice.
Note: H&E = hematoxylin and eosin; IHC = immunohistochemistry; ND = not determined; — = no positive tumor cells; + = 0–10% positive tumor cells; ++ = 11–30% positive tumor cells; +++ = 31–50% positive cells; ++++ = more than 50% of positive tumor cells.
aTwo tumors found in animal no. 8 (C3B6F1.129-Trp53tm1Brd). Only tumor from the neck was evaluated for IHC. Inguinal, subcutaneous tumor was not evaluated for this IHC study. Block not found/available, although it was diagnosed as sarcoma on H&E (see Table 2).
Osteosarcoma
Osteosarcoma was a common subcutaneous neoplasm in C3B6F1.129-Trp53tm1Brd mice, accounting for 5 of the 11 subcutaneous masses observed grossly. In contrast, only a single osteosarcoma was present in the B6.129-Trp53tm1Brd mice. Grossly, the electronic microchip was seen within or adjacent to the osteosarcomas in 2 of the C3B6F1.129-Trp53tm1Brd mice. Microscopically, the osteosarcomas in the C3B6F1.129-Trp53tm1Brd mice were composed primarily of irregular intersecting trabeculae of eosinophilic osteoid, some of which was mineralized (Figure 1). Large polygonal cells resembling osteoblasts were sometimes seen within or directly adjacent to the osteoid. The trabeculae were separated by irregular bands of pleomorphic polygonal to fusiform cells, with moderately basophilic nuclei and varying amounts of cytoplasm generally with indistinct borders, sometimes mixed with varying amounts of fibrous tissue. The single osteosarcoma in a B6.129-Trp53tm1Brd mouse differed from the others by consisting predominantly of sheets and intersecting bundles of densely packed pleomorphic, polygonal to fusiform cells with lesser amounts of osteoid, some of which was mineralized (Figure 2). In each neoplasm, the presence of osteoid was considered diagnostic for osteosarcoma. Pleomorphic neoplastic cells comprising the osteosarcomas showed positive intracytoplasmic staining for vimentin, an intermediate filament typically present in cytoplasm of nonepithelial cells, although staining in some neoplasms was weak (Figure 3). No other IHC staining was performed on the osteosarcomas in this study, since, at present, there is no widely accepted IHC stain considered diagnostic for osteosarcoma.
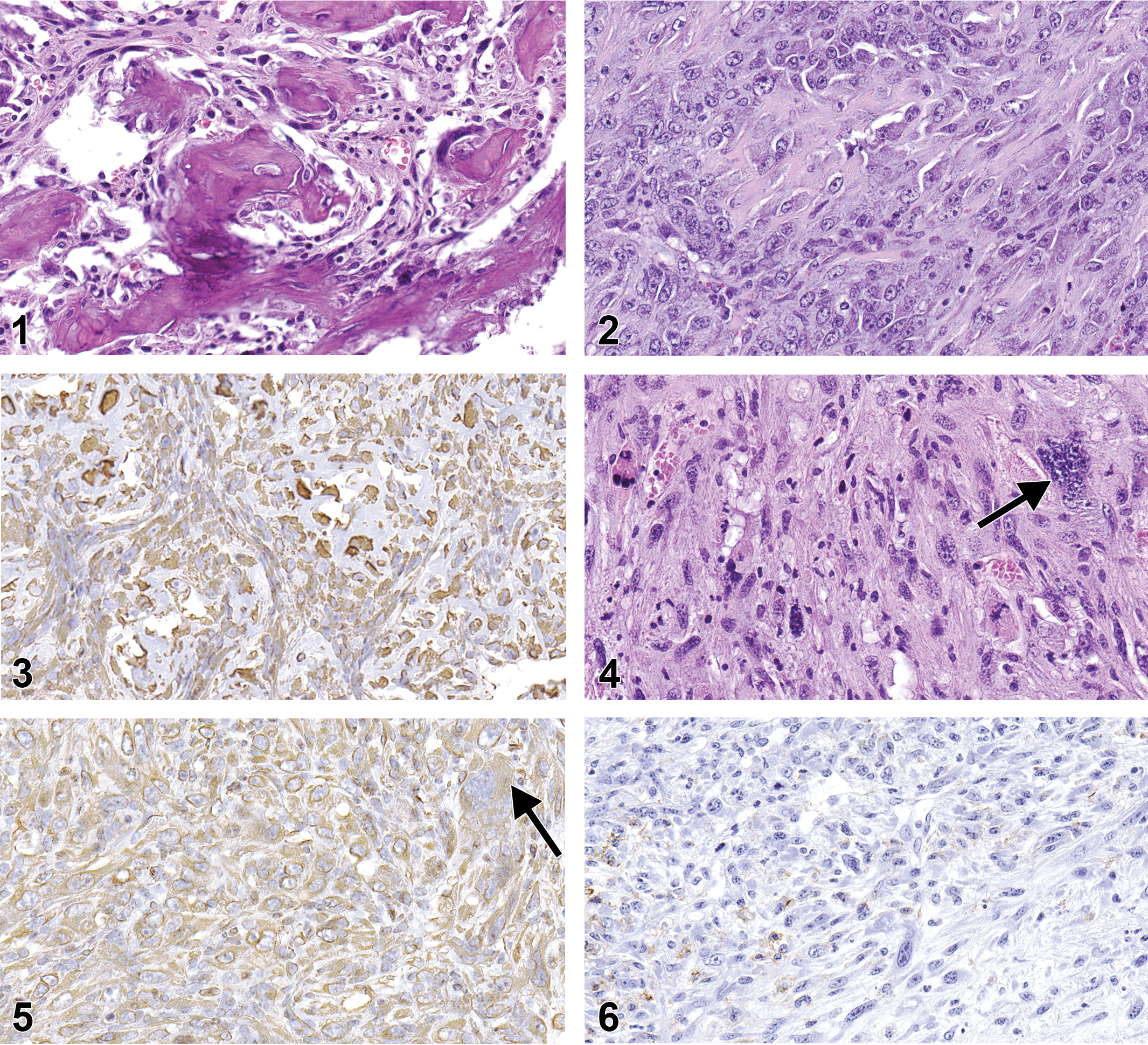
Osteosarcoma with mineralized osteoid and fibrous stroma with a few plump osteoblast-like cells. Hematoxylin and eosin stain. Original magnification 40×.
Sarcoma
Sarcoma was the most common neoplasm; 10 were observed, 5 in each of the 2 strains. One of the sarcomas was present within a Harderian gland and presumably had arisen from the glandular stroma. H&E-stained sarcomas showed no clear differentiation toward any cell type. However, based upon the results of IHC staining, the original diagnosis of sarcoma in 1 B6.129-Trp53tm1Brd mouse was changed to RMS, while the diagnosis of RMS in another B6.129-Trp53tm1Brd mouse was changed to sarcoma (Table 3). All other diagnoses of sarcoma were confirmed by IHC. Sarcomas stained positive for vimentin, although the staining was weak in some neoplasms. Sarcomas were negative for myogenin and smooth muscle actin staining. Five of the sarcomas contained varying numbers of cells that appeared on the H&E slides to be histiocytes suggestive of a possible histiocytic neoplasm. The cells stained positive by IHC for F4/80 (Table 3), a cell surface glycoprotein marker for mouse macrophages that can be used to diagnose histiocytic neoplasms (Hao et al. 2010), indicating the cells were of histiocytic origin. However, the F4/80 positive cells tended to be present near the outer surface of the neoplasm, rather than scattered diffusely within it, and in some of the neoplasms clusters of F4/80 positive cells were also seen in the adipose tissue adjacent to the neoplasm. Based upon the staining results, the F4/80 positive cells were considered to represent an inflammatory infiltrate of tumor-associated macrophages (TAMs) into the neoplasm and not to be indicative of a histiocytic neoplasm (Gazziola et al. 2003; Hatano et al. 1999).
The sarcomas had a morphologically similar appearance in both strains of mice. The neoplasms were solid cellular masses with variably sized areas of necrosis, often with hemorrhage, that in some cases involved most of the tumor. The neoplasms were composed primarily of densely packed interlacing bundles and occasional sheets of variably sized, moderately to highly pleomorphic, round to fusiform cells, with scant to large amounts of eosinophilic cytoplasm, generally with relatively distinct borders, and pleomorphic, round to elongated, and basophilic to vesicular nuclei with multiple nucleoli. Scattered throughout the neoplasms were few to numerous, moderately sized to very large, polygonal to ovoid cells with abundant eosinophilic cytoplasm, usually with distinct borders, and 1 or more nuclei. Some of the polygonal cells were very large and contained multiple nuclei or, occasionally, a single large, sometimes, bizarre nucleus. Numbers of mitotic figures within neoplasms varied considerably from being scant to relatively numerous. Some neoplasms also contained variable amounts of dense fibrous tissue, while others consisted entirely of neoplastic cells. One sarcoma from a C3B6F1.129-Trp53tm1Brd mouse differed in that it was comprised of a mixture of small round cells, with a single, deeply basophilic nucleus and a small amount of lightly eosinophilic cytoplasm and moderately large, round to polygonal cells with a single, moderately large vesicular nucleus and moderately abundant brightly eosinophilic cytoplasm. In addition, 1 sarcoma from a B6.129-Trp53 tm1Brd mouse contained substantial areas of abundant pale eosinophilic amorphous matrix substance.
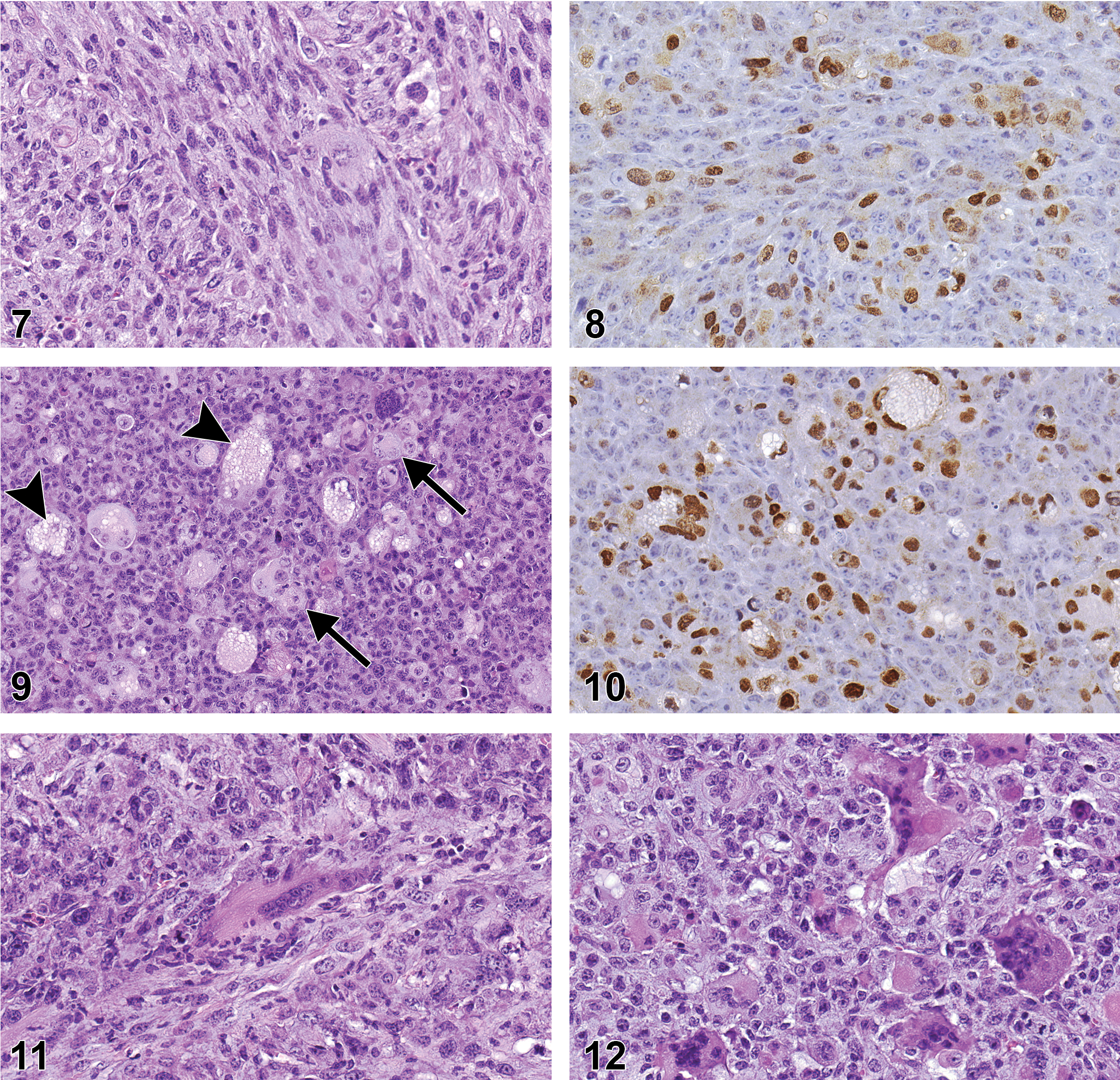
The sarcoma in a B6.129-Trp53tm1Brd animal that was originally diagnosed as an RMS based upon the H&E stain was of particular interest. The neoplasm appeared similar histomorphologically to other sarcomas but, scattered throughout the neoplasm were numerous large cells, with 1 or more large, sometimes bizarre nuclei and moderately abundant cytoplasm that appeared to be poorly differentiated rhabdomyoblasts (Figure 4). Consequently, the neoplasm was diagnosed as an RMS by the peer-review panel. The cytoplasm of the neoplastic cells, including the large rhabdomyoblast-like cells, stained positive by IHC for vimentin (Figure 5), but cell nuclei were negative for myogenin (not shown), an intranuclear skeletal muscle-specific transcription factor necessary for normal muscle differentiation and repair. Thus, the neoplasm was reclassified as a sarcoma. Many scattered cells were positive for F4/80, a marker for macrophages (Figure 6), and were considered to be indicative of an inflammatory cell infiltrate within the neoplasm.
RMS
Five RMSs were present, all of which occurred in B6.129-Trp53tm1Brd mice. The RMSs were histomorphologically similar to sarcomas in that they consisted mainly of undifferentiated cells but were distinguished from sarcomas based upon the presence of apparent attempts by the neoplasm to form skeletal muscle fibers and upon the results of IHC staining. The RMSs were further subclassified as pleomorphic RMSs based upon the histomorphologic appearance.
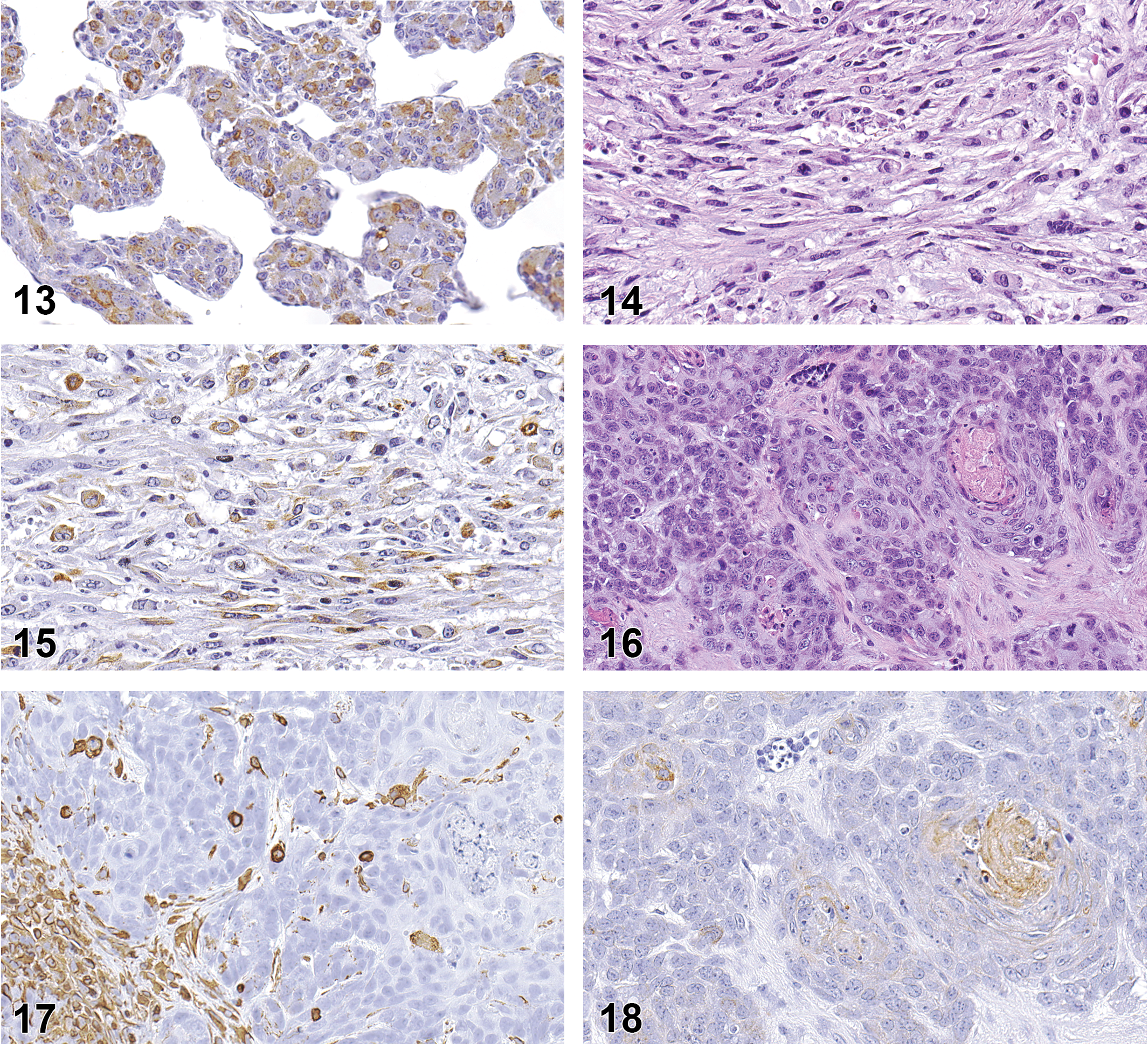
RMSs were composed of sheets of moderately to highly pleomorphic polygonal cells intermingled with varying numbers of bundles of atypical spindle cells (Figure 7). Each of the neoplasms contained areas of necrosis, which in some cases occupied much of the tumor. The cytoplasm of all neoplastic cells stained positive for vimentin, and varying numbers of nuclei of both polygonal and spindle cells stained positive for myogenin (Figure 8). The distinctive feature of these neoplasms was the presence of few to numerous cells resembling rhabdomyoblasts interspersed within the tumor. The rhabdomyoblast-like cells were round and small to moderate sized with a moderate amount of pale to brightly eosinophilic cytoplasm with distinct borders and a single nucleus (Figure 9). Occasionally, some of the rhabdomyoblast-like cells were large with multiple nuclei, and some of these cells also contained numerous clear intracytoplasmic vacuoles producing the so-called spiderweb cells (Figure 9). The nuclei of the rhabdomyoblast-like cells and multinucleated cells stained positive for myogenin (Figure 10). Small numbers of elongated ribbons of eosinophilic cytoplasm containing 1 to multiple nuclei, resembling “strap cells,” were sometimes present although no cross-striation characteristics of skeletal muscle were seen (Figure 11). One RMS contained large to very large cells with abundant eosinophilic cytoplasm and numerous nuclei that appeared to represent attempts to form muscle fibers (Figure 12). A few of these large cells had 1 or more ribbon-like projections of cytoplasm containing multiple nuclei. Another RMS also contained an unusual localized area of intersecting trabeculae of neoplastic cells with intervening spaces somewhat resembling the alveolar pattern associated with some human RMSs. The neoplasm showed positive intranuclear staining for myogenin but also expressed positive intracytoplasmic staining for smooth muscle actin (Figure 13). Positive staining for smooth muscle actin has been described to be expressed in pleomorphic RMSs in humans (Furlong, Mentzel, and Fanburg-Smith 2001; Stock et al. 2009) and is not specific for skeletal muscle.
Rhabdomyosarcoma with sheets of moderately to highly pleomorphic polygonal cells intermingled with varying numbers of bundles of atypical spindle cells.
Rhabdomyosarcoma stained for smooth muscle actin showing positive intracytoplasmic staining. The positive staining was considered not to be specific for skeletal muscle. Original magnification 40×.
Hemangiosarcoma
One neoplasm originating in the neck of a C3B6F1.129-Trp53tm1Brd mouse consisted of irregular variably sized sheets and clusters of anaplastic polygonal cells, separated by narrow bands of fibrous stroma, that formed multiple small to large, irregular blood-filled spaces lined by neoplastic cells. The neoplastic cells were large with round to ovoid vesicular nuclei with multiple nucleoli and moderately abundant eosinophilic cytoplasm with indistinct borders. The peer-review diagnosis had been sarcoma, NOS, but this was somewhat questionable due to the neoplastic cells producing vascular channels, characteristic of hemangiosarcoma. Consequently, a second review was conducted, and the neoplasm was diagnosed as a hemangiosarcoma. The neoplasm was stained for factor VIII, a generally accepted IHC stain for endothelial cells, and was negative. This was considered to be the result of the cells being highly undifferentiated and no longer expressing the marker and not proof that the neoplasm was not a hemangiosarcoma. In this case, the findings from the H&E stain were considered to take precedence over the IHC findings.
Leiomyosarcoma
One neoplasm originating on the dorsum of a B6.129-Trp53 tm1Brd mouse was diagnosed as RMS by the peer-review panel. It was morphologically similar to the other neoplasms and consisted of numerous interlacing bundles of highly anaplastic spindle cells mixed with varying numbers of anaplastic polygonal cells and scattered cells resembling rhabdomyoblasts (Figure 14). However, the neoplasm showed positive intracytoplasmic IHC staining for smooth muscle actin (Figure 15) and negative staining for myogenin consistent with the IHC staining characteristics of a leiomyosarcoma.
Carcinosarcoma
This neoplasm occurred in a B6.129-Trp53tm1Brd mouse and was located in the subcutaneous tissue adjacent to the salivary gland. The neoplasm surrounded a large central cavity containing some cell debris mixed with a few sloughed neoplastic cells and consisted of a combination of carcinomatous areas consisting of irregular nests and numerous irregular clusters of densely packed, pleomorphic polygonal cells separated by narrow bands of fibrous stroma, interspersed with sarcomatous areas composed of densely packed, pleomorphic spindle cells forming intersecting bundles (Figure 16). The polygonal cells were moderately large and had moderately abundant eosinophilic cytoplasm with indistinct borders, and round to ovoid, granular nuclei with 1 or more prominent nucleoli. The spindle cells had a small amount of eosinophilic, faintly fibrillar cytoplasm with indistinct borders, and flattened and elongated nuclei. Several thick, irregular bands of brightly eosinophilic, homogeneous matrix material containing a few nuclei were also present. The tumor spindle cells were positive by IHC for vimentin (Figure 17) and the polygonal cells were positive for cytokeratin (Figure 18), consistent with a neoplasm consisting of both mesenchymal and epithelial elements.
Discussion
A variety of spontaneous neoplasms have been reported in Trp53+/− mice, with the most common being subcutaneous sarcomas, lymphomas (usually of thymic origin), and, to a lesser extent, osteosarcomas (Floyd et al. 2002; Mahler et al. 1998; Storer et al. 2001). The subcutaneous sarcomas comprise a spectrum of neoplasms, including fibrosarcoma, RMS, hemangiosarcoma, histiocytic sarcoma, and sarcoma, and have been termed the “subcutaneous sarcoma complex.” Subcutaneous sarcomas can pose a diagnostic challenge, as some are poorly differentiated while others may show differentiation toward more than 1 cell type so that the cell of origin can be difficult to determine. IHC staining has been shown to be useful in arriving at accurate diagnoses and increasing diagnostic confidence.
Although osteosarcomas have been reported to occur somewhat less commonly than subcutaneous sarcomas in Trp53+/− mice (Floyd et al. 2002; Mahler et al. 1998), in this study half the neoplasms present in the C3B6F1.129-Trp53tm1Brd mice were osteosarcomas, whereas only a single osteosarcoma was observed in the B6.129-Trp53tm1Brd mice. Diagnosis of osteosarcoma in this study was straightforward with the H&E-stained slides due to the presence of osteoid, which is considered diagnostic for osteosarcoma. The histomorphologic features of the osteosarcomas observed in this study ranged from being composed primarily of large trabeculae of osteoid to being primarily cellular with small trabeculae and spicules of osteoid. All osteosarcomas showed some positive staining for vimentin, which is to be expected of a neoplasm of mesenchymal origin, and were negative for all the other stains. No other IHC staining was performed on the osteosarcomas in this study. Although IHC stains for a number of markers for osteosarcoma and have been described, at present, there is no widely accepted IHC stain for osteosarcoma and diagnosis is still dependent upon the identification of osteoid with an H&E stain (Rehg and Ward 2012). An IHC stain for sp7, also known as Osterix, a transcription factor that induces mesenchymal cells to differentiate into osteoblasts and subsequently into osteocytes, is currently available. IHC evaluations of 2 extraskeletal osteosarcomas, 1 each in a mouse and a rat, found good positive staining of neoplastic cells (Ito et al. 2016; Nagaike et al. 2011). This suggests sp7 may be a useful marker in the diagnosis of highly cellular osteosarcomas in which osteoid formation is minimal or absent. However, at this time, there is insufficient experience with this stain to completely evaluate its usefulness.
Eight of 9 sarcomas and 4 of 6 RMSs originally diagnosed from H&E-stained slides were confirmed by IHC. As shown in Table 3, there were three cases in B6.129-Trp53tm1Brd mice in which the diagnoses made from the H&E slides were changed based on IHC. All sarcomas stained positive by IHC for the mesenchymal cell intermediate filament vimentin. The RMSs were identified by positive IHC staining for myogenin, a transcription factor specific to skeletal muscle, while a single leiomyosarcoma was diagnosed based upon positive staining for smooth muscle actin but negative staining for myogenin.
The RMSs in B6.129-Trp53 tm1Brd mice were further subclassified as pleomorphic RMS. In humans, RMS is an uncommon neoplasm, occurring most commonly in children and adolescents and rarely in adults. RMS in humans has a highly variable morphology but is divided into basic 3 variants: embryonal, alveolar, and pleomorphic (Stock et al. 2009; Zanola et al. 2012). The embryonal variant is most common in children and consists of a mixture of round and spindle shaped cells resembling the embryonic stages of skeletal muscle development. The alveolar variant generally affects adolescents and consists of clusters of neoplastic cells separated by fibrous tissue septa; as neoplastic cells die and disappear, they leave cavities, giving the neoplasm a resemblance to pulmonary alveoli. The pleomorphic variant, also called adult-type RMS, is the least common, occurring mainly in adults. Pleomorphic RMSs can range from consisting primarily of either atypical polygonal cells and/or intersecting and interlacing bundles of atypical spindle cells, mixed with varying numbers of scattered rhabdomyoblast-like cells which identify the neoplasm as an RMS (Furlong, Mentzel, and Fanburg-Smith 2001; Stock et al. 2009). The histomorphologic structure of the pleomorphic RMSs observed in mice in the present study was consistent with that of pleomorphic RMS in humans.
Human pleomorphic RMS was at one time diagnosed based upon morphology and positive IHC staining for desmin, a muscle-specific intermediate filament, and/or myoglobin. However, desmin can be expressed in tumor desmoplasia and myofibroblasts of granulation tissue, as well as in some smooth muscle tumors (Le Calvez, Perron-Lepage, and Burnett 2006; Regh and Ward 2012), so is not a specific marker for skeletal muscle. In addition, numerous IHC studies of human pleomorphic RMS, many of which were conducted on a single neoplasm or only a small number of neoplasms, have demonstrated these neoplasms can show positive staining for a variety of markers (Furlong, Mentzel, and Fanburg-Smith 2001; Stock et al. 2009; Eyden 2010; Morotti et al. 2006; Rehg and Ward 2012; LeCalvez et al. 2006). In 2 of the larger of these studies (Furlong, Mentzel, and Fanburg-Smith 2001; Stock et al. 2009), it was found that each neoplasm stained for at least one skeletal muscle-specific marker including desmin, myosin, myoglobin, myogenin, or MyoD1, as well as nonspecific skeletal muscle markers including smooth muscle actin, pankeratin AE1/AE3, and CD34 protein, an endothelial marker. These findings demonstrated the need for a tissue-specific marker that was a reliable IHC indicator for skeletal muscle. Two markers investigated to fill this role were myogenin and MyoD1, nuclear transcription factors necessary for normal striated muscle differentiation and repair. In a large series of 956 human RMSs, primarily of the embryonal and alveolar types, Morotti et al. (2006) found that myogenin and MyoD1, both, were equally sensitive with positive staining in 97% of the neoplasms.
Based upon the results of IHC staining of RMSs, it is now well accepted that positive intranuclear IHC staining for myogenin or MyoD1 is necessary for a diagnosis of human RMS, although only a small number of positive cells may be present in some pleomorphic RMSs (Rehg and Ward 2012; Stock et al. 2009). Myogenin is preferable, as MyoD1 staining can be difficult to perform on paraffin-embedded tissue (Stock et al. 2009), and thus myogenin was the marker selected for use in the present study. Regenerative skeletal muscle will also be positive for myogenin, so it is important to differentiate normal skeletal muscle trapped within a neoplasm from neoplastic muscle cells. Staining for vimentin is positive but is of little value in diagnosing RMS, as it is simply an indicator of mesenchymal cells. Desmin staining is generally positive in RMS but sometimes may also be positive in leiomyosarcoma. Smooth muscle actin positivity can be observed in RMS and may lead to confusion in differentiating RMS with an abundance of spindle cells and few recognizable rhabdomyoblast-like cells from leiomyosarcoma. However, positivity for myogenin identifies the neoplasm as an RMS. Conversely, positive staining for smooth muscle actin without staining for myogenin is characteristic of leiomyosarcoma, although this must be interpreted with care due to the nonspecificity of smooth muscle actin. Lack of smooth muscle actin staining indicates the neoplasm is not of smooth muscle origin. H-caldesmon, a high-molecular-weight form of caldesmon, a muscle protein that combines with calmodulin and is thought to participate in regulating contraction, is a marker considered specific for smooth muscle. It has been reported in humans that positive staining for h-caldesmon occurs in smooth muscle neoplasms but not in RMSs (Coindre 2003). Although it was not necessary in this study to perform an h-caldesmon stain, the stain can be considered as an alternative to smooth muscle actin in suspected cases of leiomyosarcoma.
Pleomorphic RMSs have been reported to occur spontaneously in between 70% and 80% of A/J strain mice over 20 months of age (Sher et al. 2011). This strain has a null mutation of the gene for dysferlin, a protein in skeletal muscle that is thought to be involved in the repair of damage to the muscle fiber plasma membrane. Consequently, these mice develop a form of progressive muscular dystrophy resulting in abortive attempts at muscle regeneration with RMSs occurring at the site of the abnormal regeneration. Several mouse models of embryonic or pleomorphic RMS have been produced through deactivation of Trp53 or creation of mutant Trp53 alleles; in some models, the modified Trp53 works in concert with other genes, in particular KRas (Doyle et al. 2010; Tsumura et al. 2006; Zanola et al. 2012). Infection of Trp53+/− mice with cytomegalovirus has also been reported to increase the number of pleomorphic RMSs, leading the authors to hypothesize that cytomegalovirus may act in conjunction with modified Trp53 in RMS tumorigenesis (Price et al. 2012).
Rehg and Ward (2012) reported the findings of IHC evaluation of a large group of over 1,000 sarcomatous neoplasms in wild-type and genetically engineered mice. The group of neoplasms included a number of RMSs, including some pleomorphic RMSs. The RMSs stained positive for a variety of markers including myogenin, desmin, muscle-specific actin, smooth muscle actin, and keratin. Myoglobin, which has been used as a marker for RMS, has poor specificity, so it is not a good marker for RMS. Based upon the staining results, the authors concluded that definitive diagnosis of RMS often depends upon the demonstration of myogenin or MyoD1, in agreement with the findings in RMS of humans. A similar conclusion was reached in the present study based upon the results of the IHC staining.
One subcutaneous hemangiosarcoma, in a C3B6F1.129-Trp53tm1Brd mouse, was seen in this study. The diagnosis of hemangiosarcoma was based upon the formation of numerous large irregular, blood-filled channels lined by large, highly pleomorphic neoplastic cells consistent with hemangiosarcoma. The neoplasm stained by IHC for vimentin and F4/80. The positive F4/80 staining was considered to be due to an infiltration of histiocytes, which is discussed below. The neoplasm was stained for factor VIII, a specific marker for endothelial cells secreted by endothelial cells and appears by IHC as numerous small intracytoplasmic granules, and the staining was negative. However, factor VIII staining is variable and for a definitive diagnosis of hemangiosarcoma, it may be necessary to stain for a panel of endothelial markers (Rehg and Ward 2012).
Varying numbers of cells staining positive for F4/80, a marker for macrophages, were seen in some of the neoplasms in this study and were considered to be infiltrating macrophages reacting to the neoplasm rather than indicative of a neoplasm of histiocytic origin. Support for this conclusion comes from studies done with human malignant fibrous histiocytoma (MFH). At one time, MFH was thought to be a histiocytic neoplasm with a fibrous component. However, genetic analysis has shown the cell of origin of human MFH appears to be a mesenchymal progenitor cell (Gazziola et al. 2003). Moreover, the histiocyte-like cells in MFH were found to be an infiltrate of reactive monocyte/macrophage cells and not a part of the neoplasm, while the multinucleated cells, often seen as a part of these neoplasms, may be either reactive histiocytes or neoplastic cells (Hatano et al. 1999). There is evidence that this macrophage infiltrate, referred to as tumor associated macrophages (TAMs), is a feature of inflammation that may be related to carcinogenesis and may contribute to tumor development by releasing cytokines and growth factors and by promoting angiogenesis and fibrosis (Colotta et al. 2009; Okada 2014). Consequently, the presently preferred terminology for MFH in humans is undifferentiated pleomorphic sarcoma (UPS). Neoplastic cells in UPS will stain positively by IHC for vimentin but will be negative for desmin, myogenin, and smooth muscle actin (Rehg and Ward 2012).
One-third of the neoplasms in this study occurred at the site of microchip implantation, 3 of 11 sarcomas in the C3B6F1.129-Trp53tm1Brd mice, and 5 of 13 sarcomas in the B6.129-Trp53tm1Brd mice were at or near the site of microchip implantation. Neither H&E nor IHC staining revealed any differences between neoplasms associated with microchips and neoplasms of the same cellular origin but located distant from the microchips. This evaluation, however, may have been limited by the number of IHC markers used.
Le Calvez, Perron-Lepage, and Burnett (2006) examined 50 subcutaneous mesenchymal neoplasms arising at the site of implanted microchips in B6C3F1 mice. In each case, the microchip was within or adjacent to the neoplasm. Diagnoses made from examination of the morphology of the neoplasms on the H&E-stained slides included fibrosarcoma, RMS, MFH, leiomyosarcoma, and sarcoma, NOS. None of the neoplasms was positive for myogenin. Fifty percent of the neoplasms examined had positive IHC staining for desmin, and 87% were positive for smooth muscle actin (SMA). However, desmin may be expressed in tumor desmoplasia and myofibroblasts of granulation tissue as well as in some smooth muscle tumors and cannot be considered specific for skeletal muscle neoplasia. Moreover, smooth muscle actin may be present in nonmuscle neoplasms and in myofibroblasts, which can be a confusing factor since reactive myofibroblasts may be present in a neoplasm (Coindre 2003). Consequently, the authors considered the positive staining for desmin and SMA to be nonspecific for skeletal muscle. Furthermore, the authors considered the high prevalence of staining for smooth muscle actin in the group of neoplasms to be indicative of its nonspecificity for smooth muscle neoplasms. Interestingly, all tumors were positive for mouse macrophages, involving over 80% of the cells within the tumor in more than 95% of the tumors; sometimes, nontumor cells also stained positive for mouse macrophages, which was also a finding in the present study. The authors considered the high degree of staining for mouse macrophages indicated the nonspecificity of this marker. Although not stated, the positive staining for mouse macrophages may have represented infiltrates of TAMs. Based upon their findings, the authors concluded that all these neoplasms appeared to have a similar origin, although the exact origin could not be determined from histopathology or immunohistochemistry, and that the most suitable diagnosis was sarcoma, NOS, with a large myofibroblastic component.
While neoplasms arising at a microchip implantation site is a well-known occurrence in Trp53+/− mice, a clear cause and effect has not been demonstrated. Sarcomas occur in Trp53+/− mice without the presence of a microchip, and sarcomas associated with microchips may also occur in nongenetically modified mice (Blanchard et al. 1999; Johnson 1996; Le calvez, Perron-Lepage, and Burnett 2006; Palmer et al. 1998; Tillmann et al. 1997) and in rats (Elcock et al. 2001). However, sarcomas occurring in association with a microchip in nongenetically modified mice generally occur at a low incidence and with a latent period longer than in Trp53+/− mice. Thus, tumor formation at the site of microchip implantation is not totally dependent upon the loss of a Trp53 gene but may be enhanced by it.
The use of IHC in tumor diagnosis generally has focused on finding markers specific for a neoplasm arising from a specific cell type. The reason a neoplasm considered to be arising from a given cell type can stain positive for markers of a different cell type (such as positive staining for SMA in an RMS) remains unclear. However, Zanola et al. (2012), in a review of available literature, found reports of a large variety of genetic alterations in RMS, and other genetic abnormalities have been reported in a variety of soft tissue neoplasms (Coindre 2003; Regh and Ward 2012). Since the cellular markers identified by IHC represent products of activated genes, it is possible to speculate that alterations to gene function in a neoplastic cell may allow expression of genes and the consequent production of cellular markers not found in the normal tissue. Regardless of its origin, it is important to bear in mind the possible occurrence of nonspecific staining when diagnosing a neoplasm using IHC.
IHC staining was a valuable tool in establishing diagnoses of subcutaneous sarcomas in this study. In most cases, IHC confirmed the diagnoses made from the H&E stain; however, in a few cases in which there was some question concerning the diagnosis, in particular with regard to the pleomorphic RMSs, which can have a variable histomorphologic appearance and present a diagnostic challenge, IHC staining was extremely valuable. Conversely, in a few cases, the diagnosis was changed based upon the findings of IHC resulting in a more correct diagnosis, while in one case, IHC was noncontributory. When using IHC staining, it is important to use a stain that is known to be specific for a cell type, such as myogenin in the RMSs in this study. If a cell-specific IHC stain is unavailable, it may be necessary to use a battery of stains and look for positive staining for 2 or more markers known to be present in a certain cell, keeping in mind that nonspecific staining may occur. Thus, IHC stains must be evaluated with care, just as with H&E stains, but can yield valuable results that justify the effort.
Footnotes
Acknowledgments
The authors would like to acknowledge Ms. Beth Mahler and Elizabeth Ney for their expert assistance with formatting images and Natasha Clayton and Heather Jensen for their expertise with IHC staining. The authors also thank Drs. Amy Brix and Cynthia Willson for their critical review of this article.
Author Contribution
Authors contributed to conception or design (MJ, DM, HP, RH, TS, DD); data acquisition, analysis, or interpretation (MJ, DM, HP, RH, TS, DD); drafting the manuscript (MJ, DM, RH); and critically revising the manuscript (MJ, DM, HP, RH, TS, DD). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in part) by the Intramural Research Program of the National Institutes of Health (NIH) and the National Institute of Environmental Health Sciences (NIEHS).
