Abstract
Lack of biomarkers specific to and either predictive or diagnostic of drug-induced vascular injury (DIVI) continues to be a major obstacle during drug development. Biomarkers derived from physiologic responses to vessel injury, such as inflammation and vascular remodeling, could make good candidates; however, they characteristically lack specificity for vasculature. We evaluated whether vascular remodeling–associated protease activity, as well as changes to vessel permeability resulting from DIVI, could be visualized ex vivo in affected vessels, thereby allowing for visual monitoring of the pathology to address specificity. We found that visualization of matrix metalloproteinase activation accompanied by increased vascular leakage in the mesentery of rats treated with agents known to induce vascular injury correlated well with incidence and severity of histopathological findings and associated inflammation as well as with circulating levels of tissue inhibitors of metalloproteinase 1 and neutrophil gelatinase–associated lipocalin. The weight of evidence approach reported here shows promise as a composite DIVI preclinical tool by means of complementing noninvasive monitoring of circulating biomarkers of inflammation with direct imaging of affected vasculature and thus lending specificity to its interpretation. These findings are supportive of a potential strategy that relies on translational imaging tools in conjunction with circulating biomarker data for high-specificity monitoring of VI both preclinically and clinically.
Keywords
Drug-induced vascular injury (DIVI) continues to be a major obstacle during drug development. Despite progress in characterizing the pharmacological, molecular, and biochemical events involved, there are still no widely accepted accessible and specific circulating biomarkers of DIVI (Kerns et al. 2005; Louden et al. 2006; Zhang et al. 2010; Zhang, Hanig, and De Felice 2012). Additionally, in the case of vasoactive compounds, attempts to associate the adverse observation in preclinical toxicity studies with physiologic biomarkers, such as excessive hemodynamic changes, have not been consistently successful (Louden et al. 2000). In order to determine the relevance of preclinical toxicity findings to human, there is a critical need to develop novel approaches that are both useful in preclinical DIVI assessment and have potential for clinical application. Currently, some of the more promising circulating biomarker candidates of DIVI in the rat are of limited utility since they are commonly associated with acute phase responses and have the potential to originate from nonvascular cell and tissue types and with injuries that are not vascular in origin (Ceciliani, Giordano, and Spagnolo 2002; Gruys et al. 2005; Hochepied et al. 2003).
A promising strategy to address monitoring of DIVI would be to complement such sensitive, but nonspecific, biomarker responses with targeted imaging of susceptible vasculature, thereby allowing direct monitoring of the pathology and lending specificity to sensitive biomarker interpretation. Recent advancements in targeted imaging tools now offer the opportunity to visualize remodeling, inflammation, and changes in vascular integrity (Lin et al. 2015). The value of imaging as a preclinical or clinical drug development tool would depend on imaging targets that reveal a common physiologic response to vascular injury regardless of the initiating insult. Likely targets may arise from the propensity of lesions to initiate inflammatory responses that typically lead to remodeling of the supporting vascular matrix. A logical preliminary approach would be investigating the applicability of known processes of vascular remodeling derived from disease models, such as protease activation and their circulating mediators. Such efforts may deliver meaningful targets for visualization and offer mechanistic insight into the progression of vascular injury as a consequence of either exaggerated hemodynamics or direct vascular toxicity.
There is now sufficient evidence indicating that both physiologic and pathologic stimuli, such as altered hemodynamics associated with vascular injury or vascular injury related to inflammation or oxidative stress, can trigger the degradation and reorganization of the vascular wall in a process termed as vascular remodeling (Renna, de Las Heras, and Miatello 2013). Remodeling, neointima formation, and reparative responses to vessel injury rely on structural changes brought about by a combination of cellular growth, death, migration, synthesis or degradation of the extracellular matrix, and infiltration of inflammatory cells. Notably, remodeling due to hemodynamic strains, such as hypertension, has been shown to involve degradation and vascular reorganization mediated by both serine and cysteine proteases and proteases such as metalloproteinases (Lemarie, Tharaux, and Lehoux 2010). In particular, the expression and activity of multiple matrix metalloproteinases (MMPs) are central to vascular remodeling, a process that is highly dependent on MMPs’ multifaceted balance of gene transcription, posttranslational proform activation, and interaction with the endogenous secreted tissue inhibitors of metalloproteinase (TIMPs; Brew and Nagase 2010). In normal vascular tissues, endothelial cells, medial smooth muscle cells, and adventitial connective tissue are major sources of MMPs, most of which are present in inactive form. However, macrophages and other types of infiltrating cells are also known to be important sources of MMPs and have a role in multiple inflammatory conditions including vasculitis (Piperi and Papavassiliou 2012). It is therefore not surprising that appreciation for the contribution of MMPs in the pathogenesis of vascular disorders has been increasing steadily in recent years, particularly with respect to atherosclerotic lesions and aortic and cerebral aneurysm (Carrell et al. 2002; Galis and Khatri 2002; Jayaraman et al. 2008; Newby 2007). Induction of MMPs in the vasculature generally involves a combination of growth factors and inflammatory cytokines derived from locally activated monocytes and macrophages (Huang et al. 2012). However, in contrast to macrophage-mediated proteolysis, intracellular signaling pathways sensitive to vascular wall mechanical stress have also been shown to influence MMP activation in vivo and in vitro following hemodynamic stress (Castier et al. 2009; Milkiewicz et al. 2007). Although our understanding of the highly complex regulation of MMPs remains limited, recent findings have uncovered stimuli-specific participation of MMP isotypes in vascular pathology. For instance, a marked and selective increase in expression and activity of MMP-3 was found in the vessel wall of coronary and abdominal aortic aneurysms (Carrell et al. 2002; Tengiz et al. 2004).
There has been much interest in assessing the potential value of MMPs and TIMPs as circulating diagnostic biomarkers in several clinical vascular disorders, given that regulation of protease activity involves secretion of MMPs and TIMPs by both inflammatory cells and constitutive cells of the vascular wall. Circulating levels of MMP-9, found to be highly expressed in atherosclerotic lesions, appear to be predictive of subsequent atherosclerotic events (Sundstrom and Vasan 2006). A recent study also found circulating levels of TIMP-1, MMP-9, and especially MMP-3 were able to distinguish between patients with active antineutrophil cytoplasmic antibody–associated vasculitis (AAV) and those in remission (Monach et al. 2011, 2013). These findings parallel an earlier observation in which elevations in circulating levels of both MMP-3 and MMP-9 correlated with patients diagnosed with active chronic vasculitis of large elastic arteries known as Takayasu arteritis (Matsuyama et al. 2003). Unfortunately, the lack of available assays capable of detecting circulating levels of MMPs in preclinical species has limited their potential usage as DIVI biomarkers in drug development.
In this study, we investigated whether vascular remodeling–associated protease activity resulting from well-characterized models of hemodynamic and nonhemodynamic-mediated DIVI could be visualized ex vivo in affected vessels, thereby providing a novel tool for preclinical drug development and establishing proof of principle to stimulate further development of less invasive imaging capability. These investigations sought to further confirm and expand on previous observations using the same optical imaging biomarkers to detect soluble guanylate cyclase activator (sGCA)-induced vascular injury in rats (Lin et al. 2015). Results were compared to circulating levels of TIMP-1 and neutrophil gelatinase–associated lipocalin (NGAL), both of which have been associated with vascular remodeling and inflammation preclinically and clinically (Cruz et al. 2012; Fabunmi et al. 1996; Galis, Muszynski, et al. 1994; Monach et al. 2011, 2013; Newby 2006; Yan et al. 2001), to support their utility as complementary accessible biomarkers of DIVI. Furthermore, MMP-3 and MMP-9 immunohistochemical staining was performed on the affected vessels from rat and dog models of DIVI to investigate conservation of MMP-isotype involvement in preclinical species and offer support for the expansion of translational imaging tools to aid in the screening of drugs associated with this liability.
Material and Method
Animal and Study Designs
Rats
Male Sprague–Dawley (SD) rats, strain Crl:CD(SD), approximately 8 to 10 weeks of age, were obtained from Charles River Laboratories (Raleigh, NC). Animals were acclimated for at least 4 days prior to randomization into treatment groups, housed individually in wire mesh cages (at 18–26°C, relative humidity of 50% + 20% on a 12-hr light/12-hr dark cycle), and fed American Institute of Nutrition (AIN)-93G purified rodent diet (DYET) chow from Research Diets Inc. (New Brunswick, NJ).
Fenoldopam mesylate
Hemodynamic-mediated DIVI was induced using fenoldopam mesylate (100 mg/kg) administered subcutaneously in saline (0.9% w/v sodium chloride in deionized water) at 5 ml/kg. Six rats in both treated and control groups were necropsied at 24- and 48-hr postsingle dose.
Allylamine
Allylamine was used to investigate the effects of a direct cardiovascular toxicant on rat mesenteric arteries in vivo. Groups of 6 male rats received either vehicle (deionized water) or allylamine mixed in deionized water once daily by oral gavage for 2 consecutive days at 50 or 75 mg/kg followed by necropsy 24 hr following the last dose.
Rats necropsied at 24 and 48 hr following fenoldopam administration were injected with AngioSense® 750EX and MMPSense™ 750 FAST (Perkin Elmer, Waltham, MA) via tail vein 24 hr prior to necropsy for imaging analysis. Allylamine-treated animals were injected with MMPSense 750FAST (Perkin Elmer) via tail vein 24 hr prior to necropsy following 2 consecutive doses for imaging analysis. Rats were sacrificed by exsanguination under isoflurane anesthesia, and blood was collected via vena cava for measurement of circulating biomarkers. Following exsanguination, the entire mesentery containing adventitial connective tissue and fat was excised from each rat and fanned out for image acquisition. Following imaging, to optimize the detection of vascular injury and characterize the distribution of lesions along longitudinal sections of vessels, the mesentery was prepared by the fan method: an approximately 1.5-cm wide section, including mesenteric lymph node, was taken from the center of the mesenteric fan, placed on a flat surface, and fixed in 10% neutral-buffered formalin. The fixed samples were trimmed, processed to paraffin blocks, sectioned, and stained with hematoxylin and eosin (H&E) using standard procedures for microscopic evaluation.
Dogs
Male beagle dogs were obtained from Marshall BioResources (North Rose, NY) aged approximately 15 weeks and housed in standard conditions for all studies. Dogs were administered minoxidil, a potassium channel opener (Sigma, St. Louise, MO; 10 mg/kg/day; n = 4) or the adenosine agonist, CI-947 (CarboSynth, San Diego, CA; 10 mg/kg/day; n = 4), compounds known to induce vascular injury in dog, in empty gelatin capsules along with controls (n = 3) for 3 consecutive days prior to necropsy. Selected tissues including coronary artery (right coronary artery including right ventricular branch; and left coronary artery including left ventricular, circumflex, and paraconal [interventricular] branches), renal artery, pancreatic artery, and hepatic artery were fixed in 10% neutral-buffered formalin; trimmed; processed to paraffin sections; and stained with H&E using standard techniques for microscopic evaluation. For both rat and dog, the histomorphologic arterial changes were qualitatively graded on a scale of 0 to 5 (0, normal; 1, very slight; 2, slight; 3, moderate; 4, marked; and 5, severe) based on the extent of vascular branches involved and the severity of the injury in the affected segment of the vessel. Severe vascular injury was characterized by diffuse or confluent segmental injury of the susceptible segment of the muscular branches of the artery.
All animal husbandry and experimental procedures were in accordance with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health Publication 25, No. 28, August 16, 1996) and were approved by the Institutional Animal Care and Use Committee.
Imaging
Ex vivo imaging of rat tissue for evaluation of fluorescent imaging probes in response to arterial injury was performed using an In Vivo Imaging System (IVIS) Spectrum (Perkin Elmer). The excised mesentery from treated rats were imaged with an excitation filter of 675 nm and emission filter of 720 nm for AngioSense 680EX (Perkin Elmer) detection and an excitation filter of 745 nm and emission filter of 800 nm for MMPSense 750FAST (Perkin Elmer). Fluorescence (FL) signal overlap between the 2 probes was determined to be less than 2% (data not shown). Exposure time was automatically determined by the instrument to exceed 10,000 max counts. One region of interest (ROI) was drawn to include the whole mesenteric artery area by using the vendor’s software (Living Image version 4.0) for each image. Average radiance (p/s/cm2/sr) was then calculated for each ROI.
Biomarker Evaluation
To assess accessible biomarkers in rats, circulating plasma levels of TIMP-1 and NGAL in rat were measured using a Meso Scale Discovery (MSD, Gaithersburg, MD) multiplex sandwich immunoassay kit for rat.
Biomarkers were measured simultaneously in duplicate aliquots of EDTA plasma (1:25 dilution), calibrated using an 8-point calibration curve from provided standards, and data were analyzed using the MSD Workbench 4.0 software.
Immunohistochemistry
For an immunohistochemical analysis of MMPs, polyclonal antisera against human MMP-3 (rabbit anti-MMP-3 ab52915; Abcam, Cambridge, MA) and human MMP-9 (rabbit anti-MMP-9 ab38898; Abcam) were used on formalin-fixed, paraffin-embedded tissue from rat and dog. Briefly, embedded tissues were sectioned into 4-μm thick sections and placed onto charged adhesive-coated (Fisher Scientific, Pittsburgh, PA) slides, deparaffinized and hydrated to phosphate-buffered saline (PBS). Antigen retrieval was performed with Diva™ (pH 6.2; Biocare, Concord, CA) by the use of a pressure cooker (Decoaking Chamber; Biocare) according to the manufacturer’s instructions. Endogenous peroxidases were blocked with H2O2 treatment followed by washing in PBS. Sections were treated for 15 min in a protein blocking solution (Sniper™; Biocare, Pacheco, CA) and then incubated with the optimal concentration of either anti-MMP-3 or anti-MMP-9 IgG for 1 hr at room temperature. After washed in PBS, sections were incubated with a horseradish peroxidase–conjugated antirabbit IgG polymer (Immunobioscience, Mukilteo, WA) followed by development with 3,3′-diaminobenzidine chromogen and finally counterstaining with hematoxylin. Adjacent sections were stained with H&E to demonstrate general morphology.
Statistics
Quantitative data are presented as mean ± standard error of the mean (SEM). For statistical analysis, we used t-test for 2 group comparison when the equal variance test was passed or Mann–Whitney rank sum test when the equal variance test failed. Differences were considered significant at p < .05.
Results
Rat DIVI Histopathology and Ex Vivo Imaging
Rat DIVI histopathology
To determine whether changes in vascular morphology were induced by fenoldopam or allylamine, we evaluated the mesenteric artery by histomorphology using standard techniques. Fenoldopam, the commercially available vasodilator and antihypertensive agent, was used to produce a hemodynamic-mediated DIVI model in the rat. The arterial changes in the mesenteric artery observed in this study were consistent with a response expected with hemodynamic stress associated with vasodilation and were similar to changes reported previously for fenoldopam and other vasodilators (Dalmas et al. 2011). In this study, fenoldopam-related histomorphologic arterial changes were observed in all treated rats at both 24- and 48-hr postdose. The changes in the mesenteric arteries at 24 and 48 hr were characterized by segmental medial degeneration/necrosis and medial hemorrhage associated with perivascular inflammation (Figures 1C and 2C). The vascular injury was limited to the large, medium, and small branches of the mesenteric artery and not observed in the major elastic branches, very small arterioles, or veins. Arterial medial injury and associated perivascular inflammation were more extensive in rats sacrificed at 48 hr compared to rats sacrificed at 24 hr. In addition, at 48 hr, arterial medial injury was associated with endothelial cell hypertrophy and rarely with medial inflammation. Arterial medial changes were consistently associated with perivascular inflammation (graded as moderate to marked at 24 hr and marked at 48 hr). At 24 hr, perivascular inflammation was acute, characterized by edema and neutrophilic cellular infiltration around affected arteries and small vessels. Inflammation was occasionally present scattered within the connective tissue of the mesentery unassociated with vascular structures. Perivascular inflammation at 48 hr was more intimately associated with the immediate perivascular tissue (adventitia) and was composed of mononuclear inflammatory cells, including immature fibroblasts and fewer neutrophils.
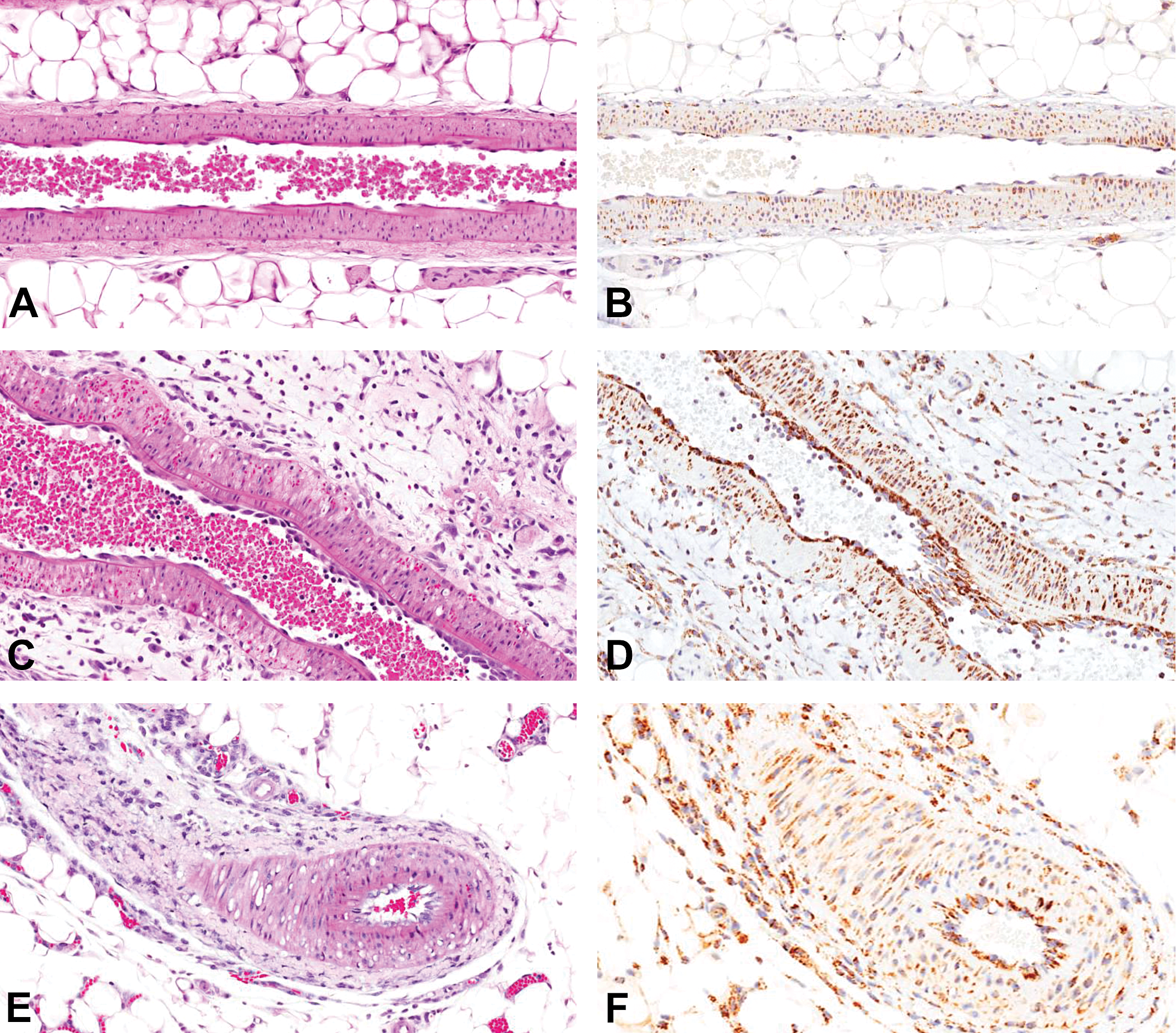
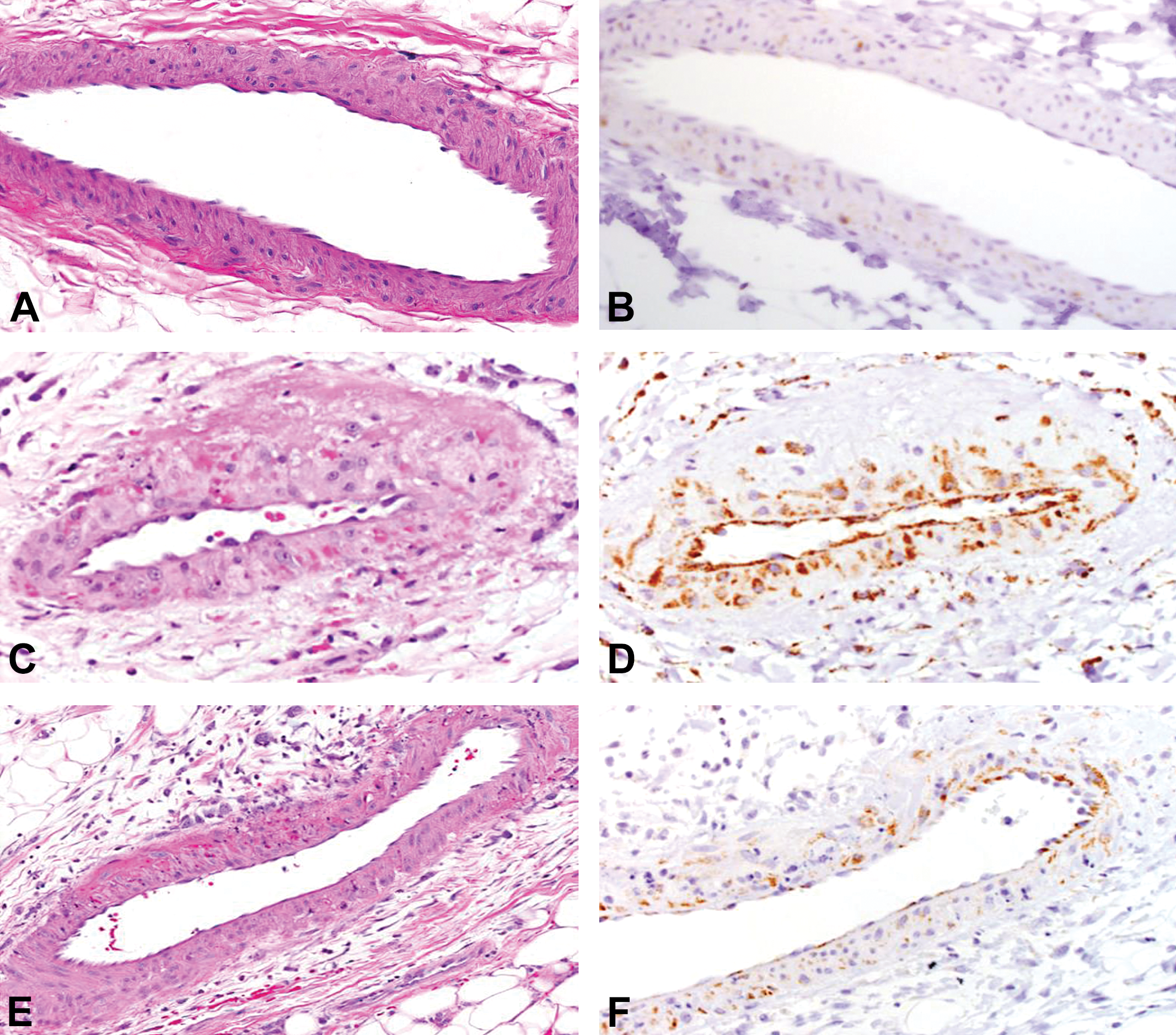
Representative images showing histopathology of drug-induced vascular injury in mesenteric artery of Sprague-Dawley rats (A, C, E) and matrix metalloproteinase 3 (MMP-3) immunohistochemistry (B, D, F). Unaffected vehicle control rat (A), segmental medial degeneration/necrosis with hemorrhage and perivascular inflammation 48 hr after treatment with fenoldopam (C), and mixed perivascular inflammation 24 hr after treatment with allylamine (E). Increased MMP-3 positivity of endothelial cells, arterial wall myocytes, and perivascular infiltrating cells 48 hr after treatment with fenoldopam (D) or 24 hr after treatment with allylamine (F) versus slight MMP-3 positivity of stain in arterial wall myocytes and occasional positive stain of endothelial cells and cells within the perivascular tissue of unaffected vehicle control tissue (B); anti-MMP-3 polyclonal antibody.
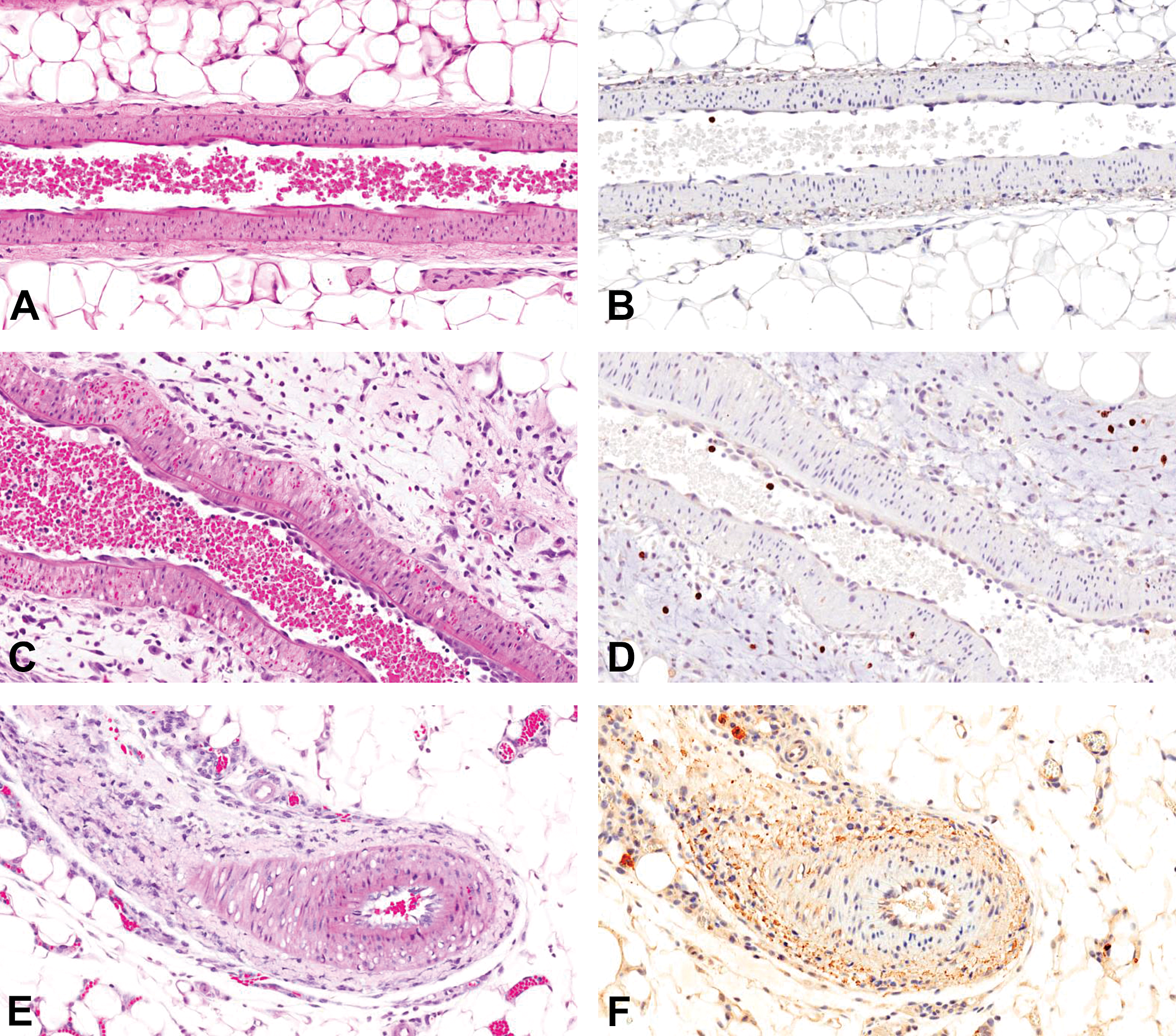
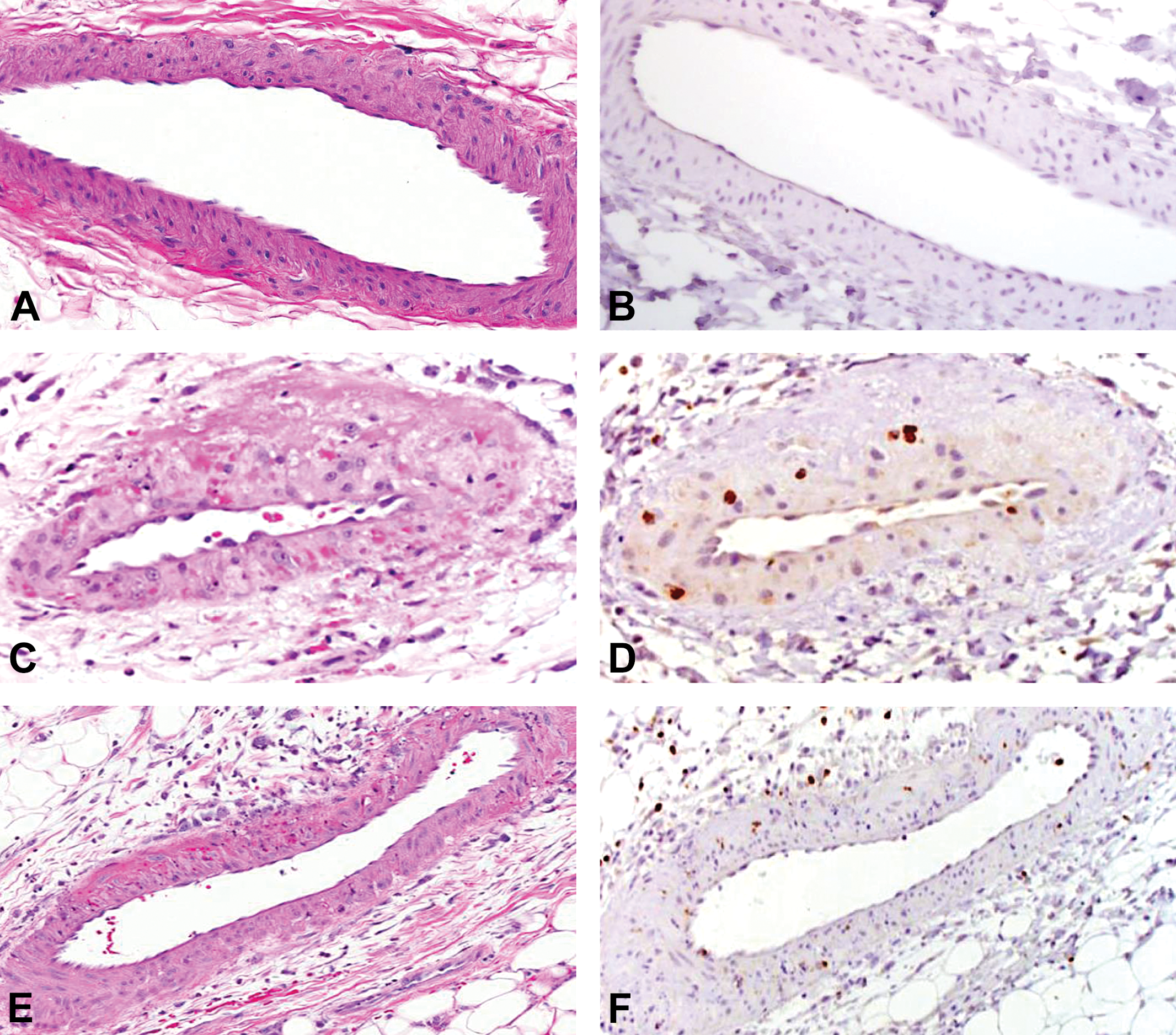
Representative images showing histopathology of drug-induced vascular injury (A, C, E) and matrix metalloproteinase 9 (MMP-9) immunohistochemistry (B, D, F) in mesenteric artery of Sprague-Dawley rats. Unaffected vehicle control rat (A), segmental medial degeneration and necrosis with hemorrhage and perivascular inflammation 48 hr after treatment with fenoldopam (C), and mixed perivascular inflammation 24 hr after treatment with allylamine (E). Occasional MMP-9 positively stained infiltrating inflammatory cells 48 hr after treatment with fenoldopam (D) or 24 hr after treatment with allylamine (F) versus rare positively stained mononuclear cells in perivascular tissue of unaffected vehicle control rat (B); anti-MMP-9 polyclonal antibody.
Allylamine was used to investigate the effects of a direct cardiovascular toxicant on rat mesenteric arteries in vivo not associated with hemodynamic alterations. Mesenteric artery was evaluated by histomorphology in rats following administration of 50 or 75 mg/kg of allylamine mixed in deionized water orally for 2 consecutive days. Histomorphological changes in the mesenteric artery of 50 and 75 mg/kg rats were qualititatively similar consisting of inflammation of the perivascular tissue (graded as very slight to slight) and characterized by periarterial/adventitial fibroplasia admixed with predominantly mononuclear inflammatory cells (Figures 1E and 2E). The mesenteric artery of 1 rat administered 75 mg/kg had focal, very slight injury of the tunica media characterized by hemorrhage and degeneration/necrosis of medial smooth muscle cells.
Rat ex vivo imaging
Gel zymography and degradation rates of known peptide substrates are commonly used methods for assessing the activity of functional MMPs in situ (Fields 2010; Kupai et al. 2010; Lombard, Saulnier, and Wallach 2005). The recent development of activatable fluorescent MMP peptide substrates allows for the visualization of active MMPs in vivo or ex vivo. Activation of MMPs has previously been shown to be increased in the mesenteric artery of rats treated with an sGCA vasodilator and is likely a critical component in the initiation of extracellular matrix (ECM) remodeling (Galis and Khatri 2002; Lin et al. 2015). Using the pan-MMP substrate probe MMPSense, we sought to visualize any functional changes to MMPs in response to arterial injury mediated by exaggerated hemodynamics. In addition, we attempted to investigate the association of any hemodynamic-mediated changes in MMP activity with alterations to vascular permeability by using the fluorescent blood pooling agent AngioSense™ in the same animal. Animals were injected with both optical imaging agents 24 hr prior to necropsy with excision of mesentery at 24 and 48 hr following a single dose of 100 mg/kg fenoldopam. Ex vivo images of mesentery from vehicle control animals following exsanguination showed a slight but detectable activated MMPSense signal diffusely distributed throughout the mesenteric vessels, adventitial connective tissue, and fat (Figure 3A). In contrast, a clear time-dependent increase in the intensity of activated MMPSense, from 24 to 48 hr, was observed in the ex vivo images of mesentery from fenoldopam-treated animals following exsanguination. Intense diffusion of AngioSense from the vasculature to the surrounding tissues was also observed in the mesenteric arteries of treated animals, in contrast to the mesentery of control animals, which showed relatively minimal distribution of the blood pooling probe representing the remaining circulating agent (Figure 4A).
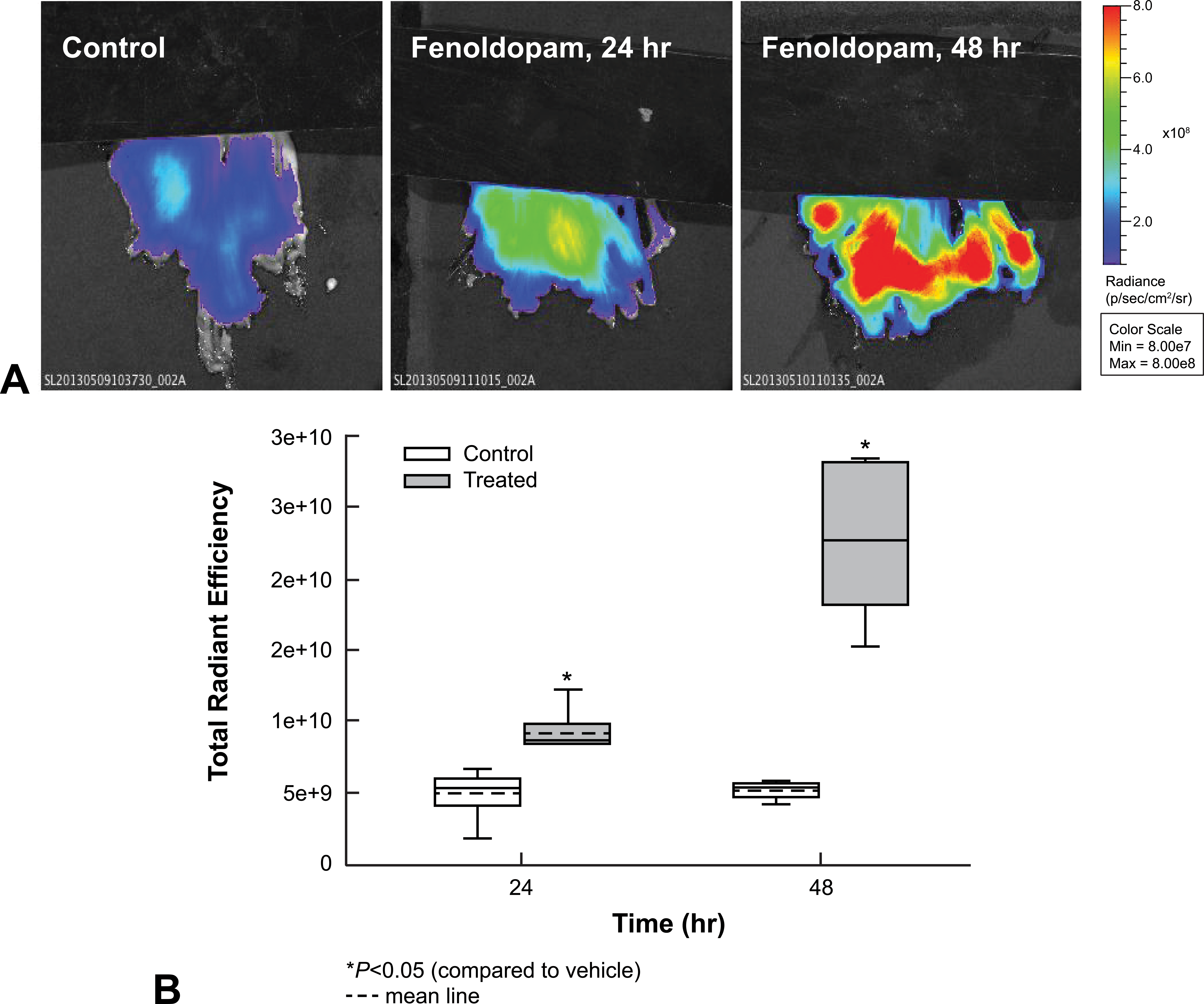
Representative fluorescence (FL) images using the optical imaging probe MMPSense™ 750FAST. Dissected mesentery from vehicle control (saline) animals and from animals at either 24 hr or 48 hr following a single fenoldopam treatment (100 mg/kg subcutaneous) are shown at the top (A). (B) The average radiance of FL from the mesentery was calculated for each treatment group at 24-hr and 48-hr posttreatment (n = 6/group). Data represent mean ± standard error of the mean (SEM).
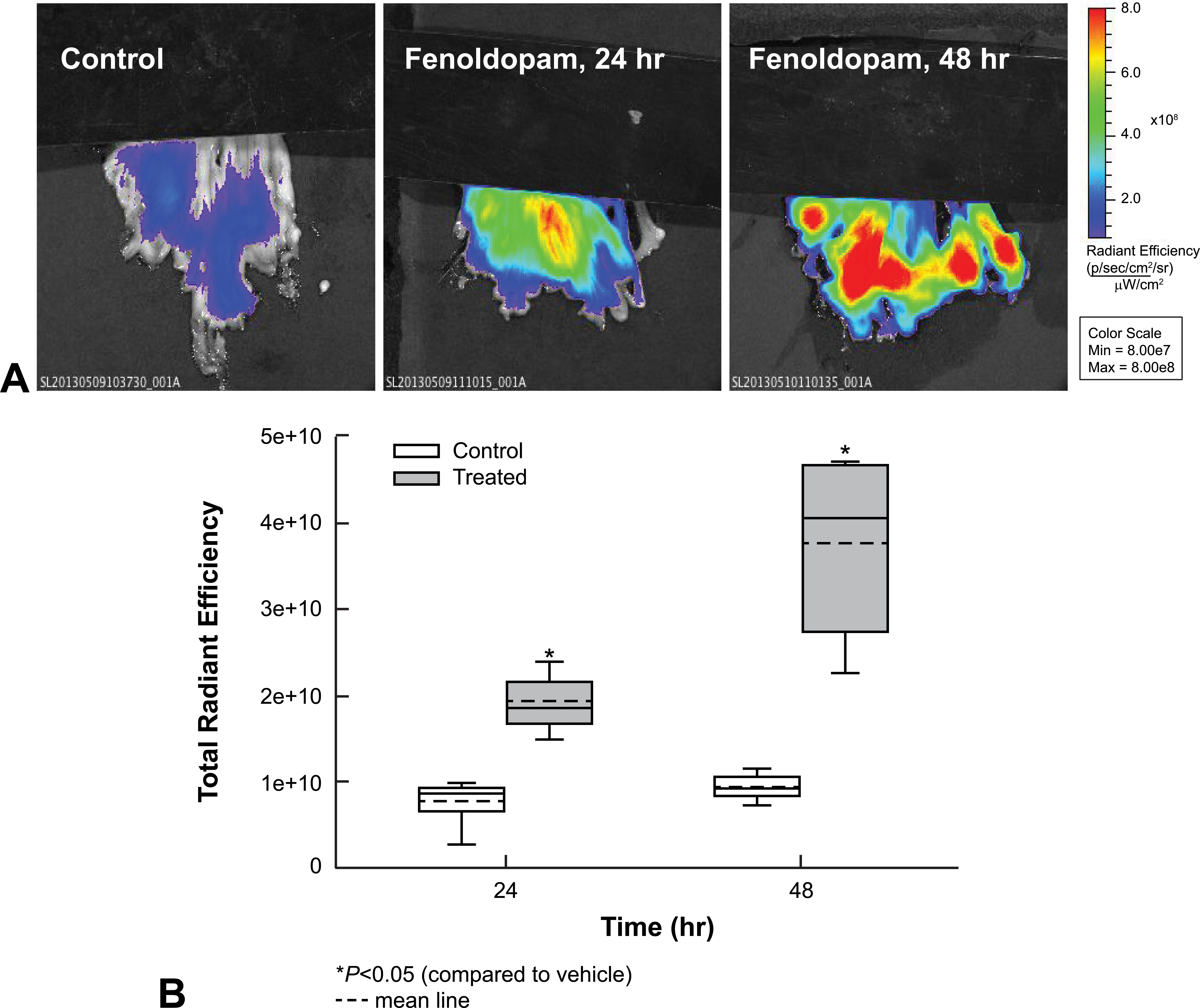
Hemorrhage induced by fenoldopam treatment is detected by AngioSense™ 680 EX. Representative fluorescence (FL) images of mesenteric arteries from control and treated animals are shown at the top (A). FL from mesenteric arteries of treated animals is significantly higher than that of control animals at both 24-hr and 48-hr posttreatment (B).
A statistically significant difference was observed in the FL intensity of both MMPSense (Figure 3B) and AngioSense (Figure 4B) probes between treated and control groups at both 24 and 48 hr posttreatment (p < .05). The FL intensity of MMPSense® in the mesentery of treated animals was approximately 1.9- and 4.3-fold greater than concurrent vehicle controls at 24 and 48 hr following fenoldopam treatment, respectively (Figure 3B). The FL intensity of AngioSense in the mesentery of treated animals was approximately 2.5- and 4-fold greater than concurrent vehicle controls at 24 and 48 hr following treatment, respectively (Figure 4B). Taken together, these results clearly demonstrate that both a significant increase to functional MMPs and increased vascular permeability in the rat mesentery, as early as 24 hr following fenoldopam administration, correlate well with the observed DIVI-associated histopathology following excessive vasodilation.
We next utilized allylamine, the reported cardiovascular toxicant, to investigate whether the observed changes to MMP activity induced by hemodynamic-mediated DIVI can also be expanded to include changes associated with direct vascular toxicity. Allylamine was administered at 75 mg/kg orally for 2 consecutive days prior to injection with the optical imaging probe MMPSense. The ex vivo images appeared to mirror our observations following fenoldopam treatment by having higher intensity and similar distribution of activated MMPSense in the mesenteric vessels, adventitial connective tissue, and fat of treated animals compared to vehicle controls (Figure 5A). The FL intensity in treated animals showed statistically significant elevations in signal intensity following administration of 75 mg/kg of allylamine (Figure 5B). The severity and incidence of the allylamine treatment–related histomorphologic changes of inflammation of the perivascular tissue and focal degeneration of the tunica media of the artery in 1 animal at 75 mg/kg correlated with the elevated FL intensity of MMP activity.
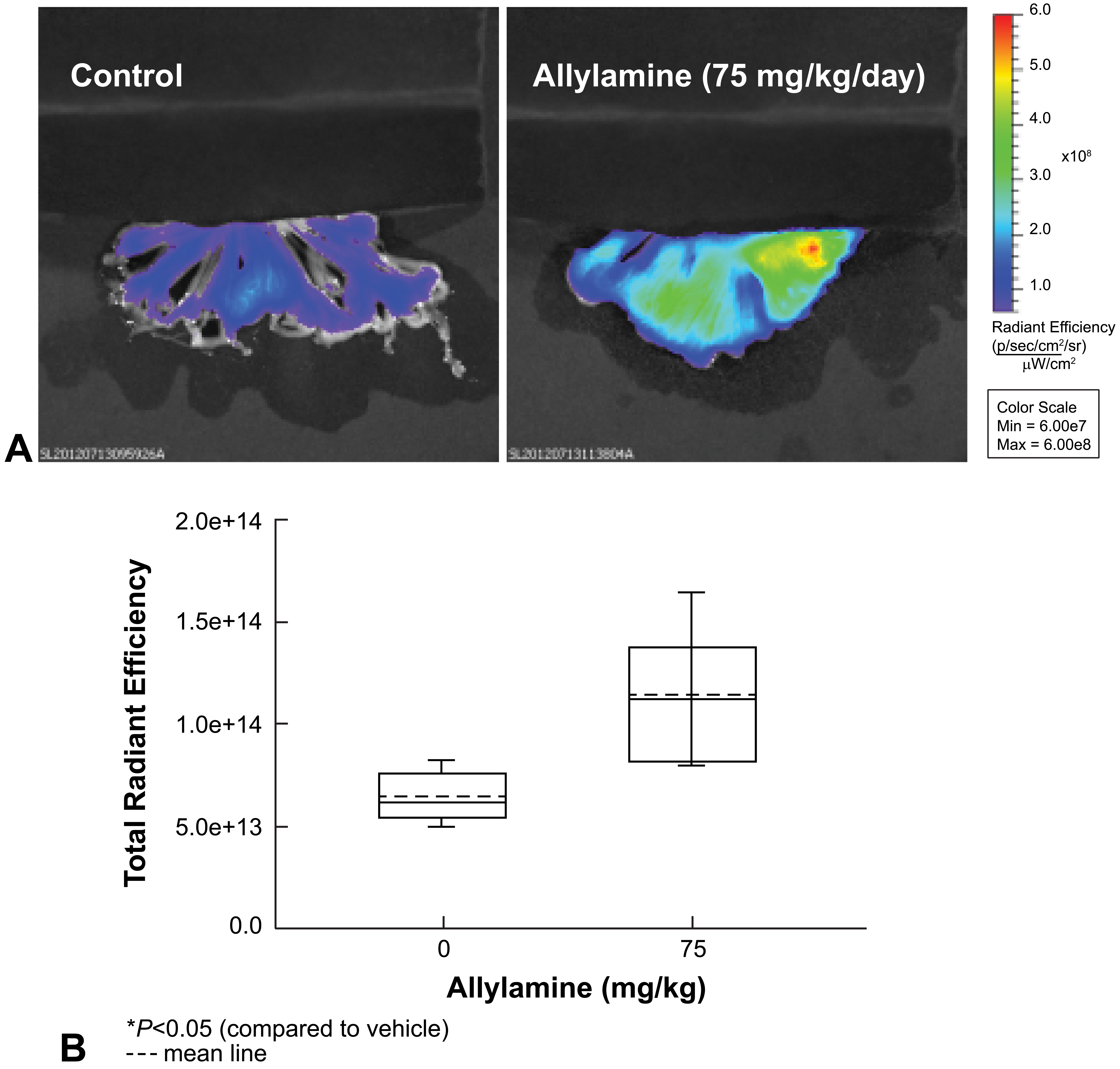
Inflammation induced by allylamine treatment is detected by MMPSense™ 750FAST. Representative fluorescence (FL) images of mesenteric arteries from control and treated animals are shown at the top (A). (B) FL from mesenteric arteries of treated (75 mpk) animals is significantly higher than that of control animals (p < .05).
Circulating Biomarkers
TIMP-1 and NGAL are both known to be associated with inflammation, vascular remodeling, and the modulation of MMP activation. Because of this association, we reasoned that relating changes to the circulating levels of TIMP-1 and NGAL with the ex vivo imaging data would serve to support their utility as accessible and informative biomarkers of DIVI. We used a commercially available immunoassay to compare biomarker responses to our imaging analysis and histomorphological findings following either fenoldopam or allylamine administration. Statistically significant increases were seen in plasma levels of TIMP-1 at both 24 and 48 hr following a single dose of 100 mg/kg fenoldopam. The mean vehicle control values of TIMP-1 were 8.0 ± 0.3 ng/ml and 9.8 ± 0.5 ng/ml at 24 and 48 hr and were increased to 14.5 ± 1.1 and 19.0 ± 2.3 ng/ml, respectively, following fenoldopam treatment (Figure 6A). Statistically significant increases in circulating NGAL were also seen at 48 hr following treatment. At 48-hr postdose, the mean vehicle control values of NGAL were 59.6 ± 4.9 ng/ml compared to 119.9 ± 9.6 ng/ml in fenoldopam-treated animals (Figure 6B).
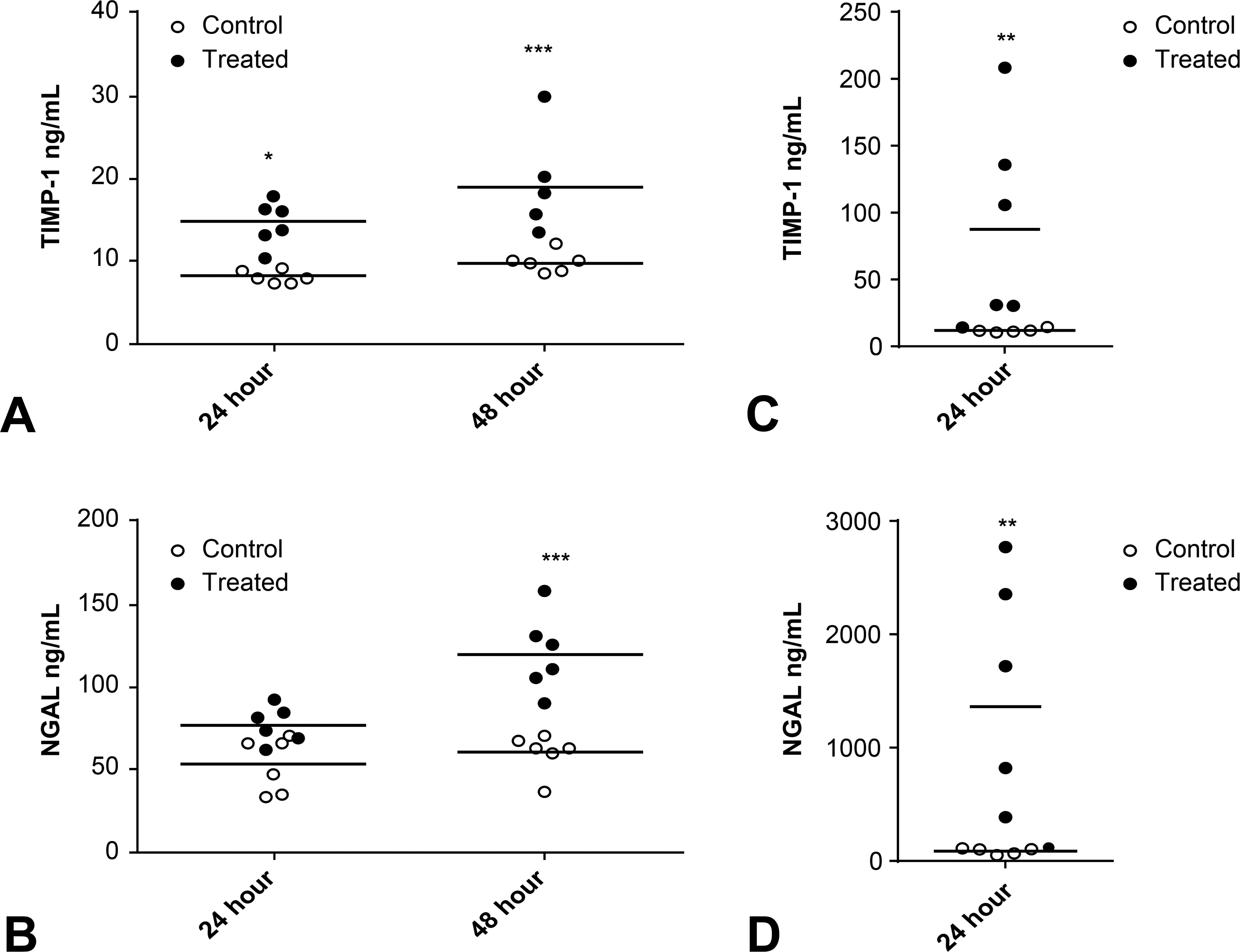
Circulating biomarkers of drug-induced vascular injury following treatment with fenoldopam (100 mg/kg subcutaneous; [A] tissue inhibitors of metalloproteinase 1 [TIMP-1] and [B] neutrophil gelatinase–associated lipocalin [NGAL]) or treatment with allylamine (75 mg/kg/day oral; TIMP-1 [C] and NGAL [D]). Bars represent mean values. An asterisk indicates significant difference of mean value relative to vehicle control mean. *p < .05; **p < .01; ***p < .001.
Statistically significant increases were also seen in plasma levels of TIMP-1 at 24 hr, following administration of 75 mg/kg of allylamine orally for 2 consecutive days. The mean vehicle control values of TIMP-1 were 11.9 ± 0.7 ng/ml and were increased to 87.5 ± 31.1, following allylamine treatment (Figure 6C). Statistically significant increases were also seen with circulating NGAL at 24-hr postdose, with mean vehicle control values of NGAL at 87.6 ± 11.8 ng/ml compared to 1,361 ± 443 ng/ml in allylamine-treated animals (Figure 6D). Given the inherent limitations of sampling vessels for histology to detect lesions that are focal in nature, group mean biomarker responses to injury were considered superior to individual animal responses for comparison purposes. These results demonstrate good correlation of the circulating biomarker levels with the observed increases in vascular MMP activity and the associated histopathology in the mesenteric artery.
Immunohistochemical Staining
Immunohistochemistry was used to determine the tissue expression and characterize the contribution of MMP-3 and MMP-9 to the treatment-related MMP activity demonstrated in ex vivo imaging of the mesenteric vasculature. Immunohistochemistry staining was performed on serial sections (adjacent to sections evaluated by histomorphology) from the affected sections of mesenteric artery from rats treated with fenoldopam or allylamine and on serial sections of mesenteric artery from vehicle control rats. Compared with (unremarkable) vehicle control rats, 24 hr after treatment with fenoldopam, variable MMP-3 positive staining was observed within the vascular endothelial cells and mesenteric connective tissue. The increased positive staining of the mesenteric connective tissue was localized to areas containing the inflammatory infiltrates. In contrast, 48 hr following fenoldopam treatment, vessels demonstrated variably increased MMP-3 positive staining of arterial myocytes and (hypertrophic) endothelial cells (Figure 1D). MMP-3 positive staining in mesenteric artery from rats treated with allylamine was increased in infiltrating inflammatory cells throughout the perivascular connective tissue and in the arterial wall (myocytes; Figure 1F). Comparatively, positive staining for MMP-9 in the vessels of fenoldopam-treated animals at 24 and 48 hr, following a single dose (Figure 2D) or following 2 consecutive doses of allylamine (Figure 2F), was restricted to occasional, individual (circulating) cells within the arterial lumen, vessel wall, or perivascular tissue despite evident arterial medial injury and associated perivascular inflammation noted by light microscopic evaluation.
Dog DIVI and MMP-3 and MMP-9
To determine whether expected changes in vascular morphology were induced by minoxidil (a potassium channel opener) or CI-947 (an adenosine agonist), coronary artery (including right ventricular branch and left circumflex, paraconal, and ventricular branches) was evaluated for histologic changes using standard techniques. Adjacent sections were evaluated by immunohistochemistry to characterize MMP-3 and MMP-9 expression in tissues with DIVI as evident by light microscopic examination.
The changes in the coronary arteries of dogs treated with minoxidil (Figures 7C and 8C) and CI-947 (Figures 7E and 8E) were characterized by medial necrosis, hemorrhage, and perivascular inflammation. The severity grades of the changes were very slight in the coronary artery of minoxidil-treated dogs and moderate to marked in the artery of CI-947-treated dogs. Widespread MMP-3 positive staining was present in most vessels of minoxidil-treated dogs, including arteries, arterioles, veins, and venules. These positive cells were present in the tunica media (myocytes) and endothelium (Figure 7E). Focal MMP-3 positive staining was present within the media (myocytes) and the endothelial cells of larger arteries as well as subepicardial arterioles of CI-947–treated dogs (Figure 7F). Individual, MMP-9 positive cells, whose morphology was consistent with infiltrating cells, were present perivascularly and focally within the media of the coronary arteries of both CI-947 and minoxidil-treated dogs (Figure 8D and F).
Representative images showing histopathology of drug-induced vascular injury and matrix metalloproteinase 3 (MMP-3) immunohistochemistry in the coronary artery of dogs (A, C, E). Unaffected vehicle control (A). Vascular injury characterized by medial degeneration and necrosis after 72 hr after treatment with minoxidil (C) or CI-947 (E). Increased MMP-3 positivity of arterial wall myocytes and endothelium of coronary artery 72 hr after treatment with minoxidil (D) or with CI-947 (F) versus minimal positivity of arterial wall myocytes of unaffected vehicle control dog (B); anti-MMP-3 polyclonal antibody IHC.
Representative images showing histopathology of drug-induced vascular injury (A, C, E) and matrix metalloproteinase 9 (MMP-9) immunohistochemistry (B, D, F) in the coronary artery of dogs. Unaffected vehicle control (A). Vascular injury characterized by medial degeneration and necrosis after 72 hr after treatment with minoxidil (C) or CI-947. Occasional positively stained inflammatory cells within arterial wall and periarterial tissue from dog after 72 hr after treatment with minoxidil (D) or CI-947 (F) versus minimal positive stain of perivascular mononuclear cells of unaffected dog administered vehicle (B); anti-MMP-9 polyclonal antibody.
Discussion
Efforts to discover accessible biomarkers that are predictive or diagnostic of DIVI have generally been met with limited success. Noted differences in species sensitivity further complicate efforts in identifying a broad, informative, and translational candidate. Moreover, noted differences in how DIVI is initiated, such as excessive hemodynamics, direct vascular toxicity, and immune-mediated vascular injury, are thought to progress by distinct pathological mechanisms. Biomarkers derived from universal physiologic responses to vascular injury, such as inflammation and remodeling, could make useful candidates. Unfortunately, biomarkers corresponding to these processes are typically not pursued as circulating DIVI biomarkers since they characteristically lack specificity.
Following a first study performed in a single model molecule, we investigated in this study whether vascular remodeling–associated protease activity and expected changes in vascular permeability from DIVI could be further visualized ex vivo in affected vessels using model molecules with different mode of actions in the rat and further evaluated them in dogs using histopathology and MMP immunohistochemistry (IHC). We found that visualization of MMP activation accompanied by increased vessel permeability in the mesentery of rats treated with agents known to induce vascular injury, through either exaggerated hemodynamics or direct cellular toxicity, correlated well with incidence and severity of DIVI histopathological findings and its associated inflammation. Additionally, increases in circulating levels of TIMP-1 and NGAL were found to be diagnostic of DIVI in rat, and the degree of response paralleled the intensity of MMP activation observed ex vivo by imaging and light microscopic examination. Immunostaining of endothelial cells, media, and adventitia from both DIVI-affected rat mesenteric and dog coronary vessels suggested that MMP-3 may serve as a marker of inflammation and consequent vascular remodeling. Supporting this possibility were observations that treatment-related MMP-3 positive staining either accompanied or, in the case of fenoldopam, followed histomorphological evidence of arterial injury, inflammation, and endothelial hypertrophy. The weight of evidence approach reported here shows promise collectively as a DIVI tool by means of complementing noninvasive monitoring of circulating biomarkers with direct imaging of affected vasculature and thus lending specificity to its interpretation.
As an initial assessment, we sought to image the vascular remodeling–associated protease response to DIVI, as this could represent a universal consequence of injury regardless of the initiating insult. The use of an activatable fluorescent pan-MMP substrate allowed for the visualization of changes in activity irrespective of the specific MMP isotype responsible. This more general approach not only captures changes in MMP activity that may correspond to constitutive cells of the affected vessels but also detects the contribution of inflammatory cells that typically infiltrate the media and adventitia following a vascular lesion. Although the expression of certain MMPs is more prominent in macrophages, basal expression for many other MMPs can also be found in vascular smooth muscle, lymphocytes, and endothelial cells (Galis, Muszynski, et al. 1994; Galis, Sukhova, et al. 1994; Herron, Banda, et al. 1986; Herron, Werb, et al. 1986; Yanagi et al. 1991). Additionally, measurement of vascular MMP activity can reflect proteases either distributed into the ECM, through secretion of freely soluble enzymes, or MMPs confined to the pericellular region by direct or indirect attachment to cellular surfaces (Kajita et al. 2001; Sternlicht and Werb 2001; Galis, Muszynski, et al. 1994; Lee et al. 1995; Sasaguri et al. 1998). Regardless of origin, inflammatory cytokines, such as interleukin-1 (IL-1), IL-4, and tumor necrosis factor-α, coordinately induce the activation of a broad range of MMPs (Galis, Muszynski, et al. 1994; Lee et al. 1995; Sasaguri et al. 1998) and can work synergistically with platelet-derived growth factor (PDGF) and fibroblast growth factor-2 (Bond et al. 2001; Fabunmi et al. 1996; Kennedy et al. 1995; Pickering et al. 1997; Yanagi et al. 1991), all of which are reported to be modulated in response to vessel injury and its associated inflammation. In this study, the increases in MMP activity as imaged in the ex vivo mesentery from rats treated with fenoldopam displayed a clear relationship with observed DIVI histomorphological changes that involved medial degeneration/necrosis and inflammation. Statistically significant increases to FL intensity were observed at 24 hr, when perivascular inflammation was acute and characterized by edema and neutrophilic cellular infiltration. Further elevation in FL intensity at 48 hr correlated with the more extensive arterial medial injury, endothelial cell hypertrophy, and perivascular inflammation associated with mixed inflammatory cells including fibroblasts. The temporal increases in protease activity also resembled the progressive increases to signal from extravasation of the AngioSense probe, indicative of changes to vascular permeability. The effects of excessive vasodilator-induced stretch or direct nonhemodynamically mediated mechanisms, including medial injury, hemorrhage, resultant inflammation, and endothelial activation, can each be seen as initiating events for protease response in vascular remodeling. The pathological effect of high doses of fenoldopam in the rat mesentery seems to deliver all of these outcomes, and as such was clearly able to demonstrate imaging evidence of MMP activation as a physiologic response to hemodynamic-mediated DIVI in the rat. In the absence of available reagents to detect circulating levels of MMPs directly in our rat studies, we examined NGAL and TIMP-1, in the same animals, for their value as circulating biomarkers known to be associated with vascular remodeling, inflammation, and MMP activation (Cruz et al. 2012; Fabunmi et al. 1996; Galis, Muszynski, et al. 1994; Yan et al. 2001). Although NGAL is best known as a biomarker measured in urine for renal injury/function, its potential prognostic value has been reported in a variety of clinical cardiovascular disorders (Cruz et al. 2012). The association of NGAL and MMP-9, specifically, has been found to result in increased MMP-mediated proangiogenic and proinvasive properties and preservation of enzymatic activity (Yan et al. 2001). The regulation of TIMP-1 response to vascular injury is a complex process, dependent on various cytokines and growth factors. Secretion of TIMP-1, in addition to being constitutively released, can be upregulated by fibrogenic cytokines, PDGF, and transforming growth factor-β (Fabunmi et al. 1996; Galis, Muszynski, et al. 1994; Yan et al. 2001). Thus, vascular stretch, injury, inflammation, and immune activation progressively move the MMP/TIMP balance in vasculature toward proteolysis (Newby 2006). Our interest in the circulating levels of TIMP-1 also stemmed from a recent report in which TIMP-1 elevation was found to be informative of clinical AAV and would therefore have the potential for clinical translation (Monach et al. 2011, 2013). Our results demonstrate that increases to circulating NGAL and TIMP-1 appear to be reflective of the observed increases in vascular MMP activity and histopathology in the mesenteric artery. Although both NGAL and TIMP-1 are known to be nonspecific, and elevated in various diseases and general organ damage, the single-dose (100 mg/kg) fenoldopam-induced injury, which resulted in DIVI in rat, can be used to infer association of biomarker responses to vascular changes in this study.
A similar degree of MMP-mediated FL was also observed following administration of the cardiovascular toxicant allylamine. Allylamine-induced toxicity has been characterized as producing vascular and myocardial lesions that closely resemble those found in atherosclerotic vessels and ischemic myocardium, respectively (Boor, Nelson, and Chieco 1980; Ramos, Grossman, and Cox 1988). The “atherosclerotic-like” lesions in aorta and other vessels in rat have been reported to occur after weeks to months of administration (Boor and Hysmith 1987). The vascular injury and inflammation induced by allylamine exposure is thought to occur by enzymatic metabolism to the toxic aldehyde, acrolein, by a semicarbazide-sensitive amine oxidase (SSAO; Boor and Nelson 1980). SSAO is an amine oxidase with prominent expression in vascular smooth muscle cells as well as distinct endothelial cell populations and adipocytes (El Hadri et al. 2002). In our acute toxicity study in rat using allylamine, treatment-related findings in the mesenteric artery were largely limited to perivascular inflammation following 2 consecutive doses. The limited histomorphologic evidence of substantial injury to the vessels was likely due to the relatively short duration of the study. However, the direct drug-induced inflammation of the vasculature was able to be detected by MMP activity imaging. Our results also demonstrate that increases in circulating NGAL and TIMP-1 levels appear to reflect the observed increases to vascular MMP activity and histopathology observed in the mesenteric artery of allylamine-treated rats. The markedly higher levels of these biomarkers following allylamine treatment compared to those observed with fenoldopam may reflect more widespread vascular and tissue injury that is not limited to the mesenteric artery as has been described for fenoldopam DIVI. Allylamine-induced arterial injury in the heart accompanied by myocardial degeneration has been observed in similar studies conducted in our laboratories (data not shown) as well as in several published reports (Awasthi and Boor 1998; Boor and Ferrans 1985; Kumar, Hysmith, and Boor 1990). Taken as a whole, these findings demonstrate utility in assessing MMP activity by vascular imaging, as MMP3 activity is able to clearly differentiate between the 2 distinct mechanisms underlying the pathobiology of vasculature. There have been several recent reports describing involvement of MMP isotypes in the pathogenesis of vascular disorders. In particular, MMP-3 or MMP-9 expression and activity have been implicated in coronary aneurysms, atherosclerosis, and hypertension, among other events (Galis and Khatri 2002; Galis, Sukhova, et al. 1994; Renna, de Las Heras, and Miatello 2013). As mentioned, circulating levels of MMP-3 and MMP-9 have also been shown to distinguish between patients with active AAV and those in remission (Monach et al. 2011, 2013).
Given this information, we examined DIVI-affected rat mesenteric arteries for changes to either MMP-3 or MMP-9 expression compared to controls using immunohistochemical staining. Overall, we found MMP-3 staining in the rat to be variable; however, there appeared to be strong MMP-3 staining of hypertrophic endothelial cells in vessels from fenoldopam-treated rats, 48 hr following a single dose. This response would be consistent with expected phenotypic changes to activated endothelial cells, and in agreement with observations in vitro, where induction of MMP-3 in endothelial cells required prolonged exposure to growth factors (Pintucci et al. 2003). Immunostaining of the allylamine-affected vessels displayed abundant treatment-related MMP-3 staining in cells throughout the perivascular connective tissue and in arterial wall myocytes compared to vehicle controls. The occurrence of MMP-3 staining is consistent with cell types known to metabolize allylamine to acrolein and may be indicative of cellular oxidative stress. Surprisingly, MMP-9 staining in both rat models was not remarkable and only limited to infiltrating (inflammatory) cells. Further investigation would be needed to identify the MMP isotypes responsible for driving the protease fluorescent images. Overall, the degree of change associated with MMP-3 staining in both studies would likely not solely account for the observed changes to MMP activity detected with vascular imaging.
While targeted ex vivo imaging of vascular changes could become a useful preclinical tool, it can also serve to prioritize probable imaging targets for further development of noninvasive imaging techniques for potential clinical application. Success of imaging targets for these efforts would rely on the translatability of the response across preclinical species and ultimately to the clinic. We sought to extend some of our findings in rat into dog, a species highly susceptible to hemodynamic DIVI mechanisms. Specifically, we looked at whether changes to vascular MMP-3 or MMP-9 could be detected in coronary artery from dogs treated with agents known to induce DIVI and confirmed through histopathological evaluation. We found remarkable treatment-related MMP-3 staining in the media and endothelium in coronary vessels from dogs treated with either minoxidil (Mesfin et al. 1995) or the adenosine agonist, CI-947 (Enerson et al. 2006). Overall, treatment-related MMP-9 staining appeared to be associated with infiltrating cells in the perivascular connective tissue as well as in the media. These findings suggest that MMP responses to vessel injury, especially those of MMP-3, are representative of a physiological process likely conserved across preclinical species. It is important to point out, however, that differences in the degree of change between the rat and dog were observed. Aside from the apparent possibility of a dissimilar physiologic response between species, differences may also be dictated by the particular vascular bed being examined. Additionally, the time of examination for a pathobiological event such as inflammation and remodeling is likely to have a substantial impact and may offer a partial explanation for some small differences in immunohistochemical staining between the studies presented here. These data also highlight the critical need to identify and develop surrogate circulating biomarkers of DIVI in nonrodent as evaluation of dog TIMP-1 and NGAL levels has not been successful.
In summary, we found that vascular remodeling–associated protease activity as well as expected changes to vessel permeability resulting from DIVI could be visualized ex vivo in affected vessels, thereby supporting the approach of developing a potential novel translational tool for drug development. Additionally, circulating levels of TIMP-1 and NGAL, both of which have been associated with vascular remodeling and inflammation preclinically and clinically, demonstrated utility as accessible biomarkers of DIVI in the rat in conjunction with MMP activity imaging. The role of MMPs in the pathophysiological response to injury was further confirmed by immunohistochemical staining for MMP-3 and MMP-9 and demonstrated conservation of MMP involvement in preclinical species. These findings offer support for the expansion of translational imaging tools to aid in the development of drugs associated with this liability. Ideally, a comprehensive strategy in which circulating biomarker data would be complemented with noninvasive imaging for confirmation of vascular damage could be adopted both preclinically and clinically.
Footnotes
Acknowledgments
The authors thank Donna Lynch for her support in designing and executing studies, Anjali Patel for her critical review of this article, and also colleagues at Merck who encouraged and supported these studies.
Author Contribution
Authors contributed to conception or design (RG, SL, BB, BC, LL, GM, TP, SP, TJ, FS, WG); data acquisition, analysis, or interpretation (RG, SL, BB, BC, LL, GM, TP, SP, TJ, FS, WG); drafting the manuscript (RG, SL, BC, LL, GM); and critically revising the manuscript (RG, SL, BB, BC, LL, GM, TP, SP, TJ, FS, WG). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
