Abstract
Oncoimmunology (or immunooncology) is a burgeoning specialty of precision (“personalized”) medicine designed to heighten the antitumor response of the immune system against molecules expressed excessively or only by tumor cells. This focus is necessary, as cancers are polyclonal tissues comprised of antigenically heterogeneous cells, the exact composition of which is shaped by the balance between antitumor immunity and tumor-promoting inflammation. Key targets include enhancing immune system (especially T cell) reactivity, inhibiting immune checkpoints, and promoting tumor cytolysis. Therapeutic modalities to address these targets include administering antibodies, cytokines, or small molecules that directly stimulate the immune system, attack tumor-associated antigens, or interfere with tumor–stroma interactions; adoptive transfer of autologous T cells following ex vivo selection/expansion/activation (typically after lymphoid-depleting regimens and in conjunction with immunostimulatory therapy); and vaccination (against tumor antigens). Pathology involvement in oncoimmunology product development is critical to assess expression of target molecules in tumor cells, stromal cells, and tumor-infiltrating leukocytes.
Keywords
Neoplasia is a complex disease in which divergent molecular anomalies permit cells to escape normal growth-limiting mechanisms. Traditional treatment regimens stress tumor annihilation by surgical removal in conjunction with cytotoxic (chemical and/or radiological) therapy. A major disadvantage of this strategy is that cytotoxicity is not limited to neoplastic cells but also impacts other rapidly proliferating populations of normal cells. To limit off-target damage, oncologic practice increasingly has sought to harness the immune system to selectively attack tumor cells. Successes achieved in this arena have led to the establishment of oncoimmunology (or immunooncology) as a major medical discipline (Palucka and Coussens 2016). Indeed, as of 2017, the number of antineoplastic immunotherapies in development exceeds 1,100 test articles being studied in nearly 1,500 clinical trials based on predictions that worldwide sales for such products will increase 7-fold from current levels (about USD$6 billion yearly) by 2025 (Dance 2017; Loftus 2017).
Oncoimmunology products regulate the balance of the immune system to either enhance antitumor immunoreactivity or reduce tumor-promoting influences (Palucka and Coussens 2016; Pol et al. 2015). This dual approach is warranted because most neoplasms are heterogeneous, where each tumor cell clone features a different molecular signature—subtly so in many benign lesions but often markedly so for more anaplastic malignancies. Oncoimmunologic therapies that enhance antitumor responsiveness attack molecules that are overexpressed or overactive, and ideally that are only expressed and active in neoplastic cells, thus limiting their growth or causing their death (Table 1; Palucka and Coussens 2016). Oncoimmunologic treatments that decrease tumor-promoting stimuli seek to diminish the numbers of immunosuppressive regulatory T cells (Treg) or lessen the production of stromal cell–derived growth factors within the tumor microenvironment (Palucka and Coussens 2016). So far, oncoimmunologic therapy has been shown to work best in patients whose immune systems have already begun mounting a neoplastic tissue-specific response by producing tumor-targeted circulating and tumor-infiltrating lymphocytes.
Selected Targets for Oncoimmunologic Product Development.
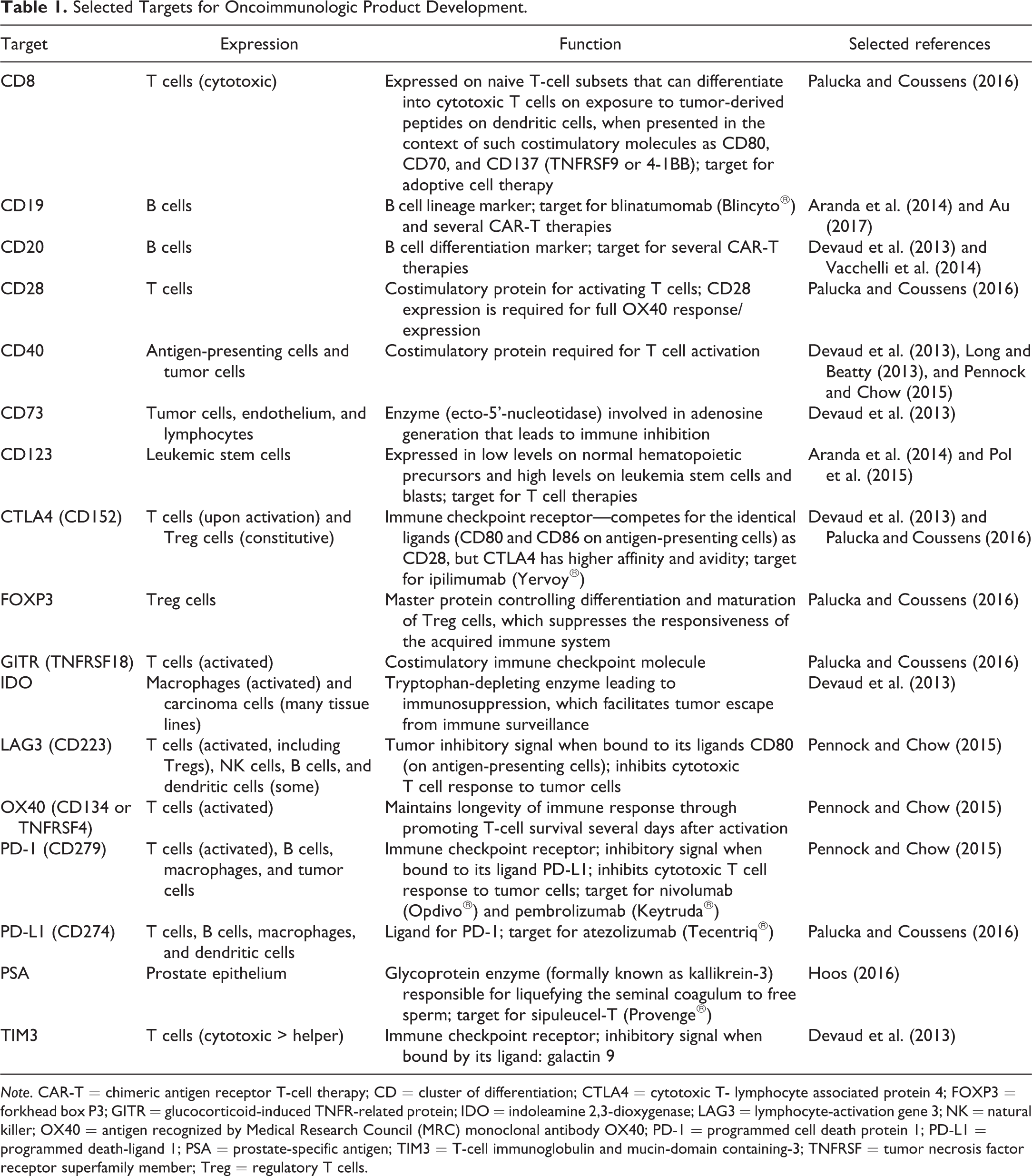
Note. CAR-T = chimeric antigen receptor T-cell therapy; CD = cluster of differentiation; CTLA4 = cytotoxic T- lymphocyte associated protein 4; FOXP3 = forkhead box P3; GITR = glucocorticoid-induced TNFR-related protein; IDO = indoleamine 2,3-dioxygenase; LAG3 = lymphocyte-activation gene 3; NK = natural killer; OX40 = antigen recognized by Medical Research Council (MRC) monoclonal antibody OX40; PD-1 = programmed cell death protein 1; PD-L1 = programmed death-ligand 1; PSA = prostate-specific antigen; TIM3 = T-cell immunoglobulin and mucin-domain containing-3; TNFRSF = tumor necrosis factor receptor superfamily member; Treg = regulatory T cells.
Many molecular targets are found in multiple tumor types, thereby allowing oncoimmunologic products to be deployed against a broad spectrum of neoplasms (Hoos 2016; Palucka and Coussens 2016; Pol et al. 2015). To date, investigation has concentrated on aggressively invasive or metastatic indications such as epithelial (e.g., breast, lung [non-small cell], and prostate carcinomas), hematologic (leukemias, lymphomas, and multiple myelomas), and neural (glioblastomas and melanomas) malignancies. In addition, such targets may serve as disease biomarkers and can be used as the foundation for developing genomic- or proteomic-based in vitro companion and complementary diagnostic tests (where companion kits comprise an “essential” part of an approved oncoimmunologic product, used to select patients who are most likely to respond to a therapeutic agent, while complementary tests may be utilized as an aid but are not required to prescribe a therapeutic agent; Hoos 2016). Oncoimmunologic therapies are registered as biologics, cell therapies, or drugs (whichever is appropriate), while companion and complementary diagnostic tests are registered as medical devices.
Many modalities involving administration of biomolecules or small molecules have been utilized to rebalance the oncoimmunologic response to favor tumoricidal activity. A very successful strategy, effective in approximately 20% of all cancers (Dance 2017), is to inhibit immune checkpoints to prevent tumor-mediated inactivation of T cells (Pennock and Chow 2015). Indeed, the four most highly prescribed oncoimmunologic products currently approved for use are all checkpoint-inhibiting antibodies: atezolizumab (Tecentriq®) against programmed death-ligand 1 (PD-L1), nivolumab (Opdivo®) and pembrolizumab (Keytruda®) against programmed cell death protein 1 (PD-1), and ipilimumab (Yervoy®) against cytotoxic T lymphocyte associated protein 4 (CTLA4). The choice of agent in the clinic may be decided by the nature of the associated diagnostic test; for example, Keytruda is approved for use only with a companion test (PD-L1 immunohistochemical [IHC] 22C3 pharmDx), while Opdivo may be deployed with the aid of a complimentary test (PD-L1 IHC 28-8 pharmDx) but also may be used in the absence of the diagnostic test. Checkpoint inhibitors may be used in combination to attack separate pathways (e.g., Opdivo and Yervoy), thus achieving greater antineoplastic efficacy. Another common tactic is to inject immunostimulatory cytokines or cytokine-bearing gene therapy vectors that bolster immune cell responsiveness (usually directed to T cells or macrophages) or alter the chemokine milieu in the tumor microenvironment (Devaud et al. 2013; Long and Beatty 2013; Hoos 2016). A third approach is to administer monoclonal antibodies that block tumor-promoting signaling pathways in neoplastic cells that are dependent on intercellular signaling between tumor cells and other intraneoplastic cell types (e.g., leukocytes, stromal cells) or stroma (Vacchelli et al. 2014). A fourth option is to vaccinate individuals with purified aliquots of tumor-specific antigens or peptides to generate active immune responses for tumor cytolysis and persistent surveillance (Wolf, Heine, and Brossart 2014; Pol et al. 2015). The vaccine composition typically is personalized to address the molecular profile determined for their specific tumor. The vaccination may be performed ex vivo by exposing isolated leukocytes with a purified tumor-specific antigen; an example of this tactic is sipuleucel-T (Provenge®), in which a patient’s antigen-presenting cells (APCs) are incubated in the presence of prostate-specific antigen and granulocyte–macrophage colony-stimulating factor (GM-CSF), which aids in maturation of the antitumor APCs (Hoos 2016). A fifth option is intratumoral delivery of oncolytic viruses (Hoos 2016). Direct injection into the neoplasm avoids the host immune response, while disintegration of infected tumor cells releases new virions to infect and destroy the remaining neoplasm. An example of this approach is talimogene laherparepvec (Imlygic®), a genetically modified version of human herpes simplex virus (HSV) type 1 designed to infect and kill tumor cells (Hoos 2016). The oncolytic virus can be modified so that it simultaneously delivers an immunomodulatory chemokine (Devaud et al. 2013); for instance, Imlygic hijacks the machinery within tumor cells to elicit production of GM-CSF. In addition, necrotic debris may launch a useful and possibly long-lasting immune reaction against tumor antigens. Finally, oncoimmunologic agents may be engineered, which directly promote interaction of tumor-targeted lymphocytes with neoplastic cells. An example of this strategy is the bispecific monoclonal antibody blinatumomab (Blincyto®), where the 2 arms recognize cluster of differentiation 3 (CD3; a T cell lineage marker) and CD19 (a B cell lineage marker) and thus serve to form a bridge between adjacent CD3-expressing effector lymphocytes and CD19-expressing neoplastic B cells (Hoos 2016).
In many cases, oncoimmunologic treatments of different types are likely to prove more effective if used in combination (Wolf, Heine, and Brossart 2014). For example, administration of an immune checkpoint inhibitor to provide immediate antitumor activity may be paired with repeated immunization to provide persistent immunosurveillance against residual tumor cells (Wolf, Heine, and Brossart 2014). Similarly, an increasingly vital strategy is adoptive T-cell transfer (Aranda et al. 2014). This procedure involves isolation of autologous T cells from cancer patients, after which antitumor T cells are selected, clonally expanded, and then activated in vitro using immunostimulatory cocktails of cytokines. In the meantime, the patient’s immune system (particularly lymphoid elements) is ablated using chemotherapy or whole-body radiation. Once immune system depletion is confirmed, the modified T cells are reintroduced into the patient where they reestablish the acquired immune system and foment an aggressive antitumor response (Aranda et al. 2014). Certain oncoimmunologic cell therapies may be used as stand-alone antineoplastic agents. A variant of autologous T-cell transfer is chimeric antigen receptor T-cell (CAR-T) therapy, in which immune effector cells are modified using a viral gene vector with a genetically engineered receptor complement that is designed to detect cells expressing tumor-specific antigens and then destroy them (Au 2017). A variant of combinatorial oncoimmunologic therapy is to administer a single agent that has the capacity to generate two (or more) distinct modes of antineoplastic immunoreactivity. As noted above, one example is direct intratumoral injection of an oncolytic virus genetically modified to deliver a chemokine or cytokine (Devaud et al. 2013). Another instance would be the administration of a bispecific monoclonal antibody (i.e., a bi-specific T-cell engager [BiTE]) capable of both activating T cells and then connecting them to neoplastic cells expressing the antigens they are designed to attack (Hoos 2016).
Pathology evaluation plays a prominent role in the discovery and development of oncoimmunologic products. Three primary duties may be provided by pathologists in this regard. First, an important responsibility of comparative pathologists is evaluating immunological aspects of antitumor immunology using animal models. The highly conserved motifs utilized by mammalian species provide the basis for translational medicine research (Li et al. 2017). Common animal models for oncoimmunology research include genetically engineered mice (GEM) with induced mutations in tumor-associated genes (e.g., ablation of an immune checkpoint molecule) as well as immunocompromised mouse strains or GEM with “humanized” immune systems that have been engrafted with human tumor tissue (frequently immortal cell lines or patient-derived samples [i.e., “avatars”]). Appropriate phenotypic assessment of both intended (“efficacious”) and adverse (“toxic”) oncoimmunologic responses in animal models requires active and early participation by properly trained and highly experienced pathologists; these individuals commonly have a veterinary medical (DVM or equivalent) background, but personnel with a medical education (MD or equivalent) or graduate training in comparative pathology (PhD or DSc) are employed for this purpose in some institutions. Such special investigations typically include pathologists as one member of the research team. This pathology function tends to be undertaken in the academic and bio/pharmaceutical settings by experimental pathologists.
Second, pathologists in diagnostic medical practices (i.e., MD background or equivalent) are instrumental in deploying oncoimmunologic therapies. One aspect of the diagnostic pathology role is monitoring patients for potential adverse reactions associated with oncoimmunologic therapies. Principal considerations are early detection of excessive immune system responsiveness (e.g., high levels of circulating cytokines or tumor lysis markers) or unexpected consequences (e.g., potential neoplastic transformation of adoptively transferred autologous T cells following ex vivo genetic modification). A second element of this diagnostic pathology role is interpretation of companion and complementary diagnostic tests used to define whether or not a patient might be a good candidate for a particular oncoimmunologic agent. These functions can be done using conventional pathology tools (e.g., bright-field microscopy of tissue sections processed for routine histochemical stains and IHC methods), in some cases supplemented by special techniques that require advanced equipment (flow cytometric or laser scanning cytometric analysis of lymphocyte subpopulations, protein–protein interactions using Biacore™ analysis, serum chemistry measurements of biomarkers, etc.).
The third and unique task for pathologists in oncoimmunologic product development is to validate molecular tumor targets (Aeffner et al. 2016). For example, pathologists are often asked to evaluate the expression, as defined by IHC localization, of one or several molecular markers, for immune effector cells or tumor cells, in batteries of human tumor specimens (Figure 1; Table 1). Using bright-field or fluorescent microscopy, the analysis may yield a straightforward qualitative assessment (i.e., “Is a molecule expressed, or not?” or “Are 2 molecules expressed in relation to one another?”) or a semiquantitative examination (i.e., “In what percentage of cells, and at what intensity, is a molecule expressed?”; Gibson-Corley, Olivier, and Meyerholz 2013). An increasingly common approach is to perform a genuinely quantitative analysis of tumor cell–related molecular expression (i.e., percentages of labeled cells having a given labeling intensity for a given marker) using digital imaging software. Such digital pathology solutions can utilize cell type–specific markers (for tumor cells, stromal elements, and/or different leukocyte lineages) coupled with suitable gating (i.e., cutoffs for signal intensities that define cells with different functional states) to simultaneously examine multiple tumor compartments (Aeffner et al. 2016). Assessments of this kind can help direct future development efforts for oncoimmunologic programs toward or away from certain tumor types, identify potential subclasses of tumors that are likely to be more susceptible to a given molecular-based intervention, and provide guidance regarding which molecules might serve as useful markers for use in companion diagnostic kits. Pathologists who interpret these data sets generally have a medical or veterinary medical background (since the assessment involves a research rather than a diagnostic function), but they often work closely with technical staff (usually BS- or MS-level staff with science degrees) trained to efficiently annotate tumor images and perform digital analyses (Aeffner et al. 2016). This pathology role generally is undertaken in high-throughput laboratories in bio/pharmaceutical firms and contract research organizations. Scientists with both experimental pathology and toxicologic pathology expertise are important members of the nonclinical research team for this application.
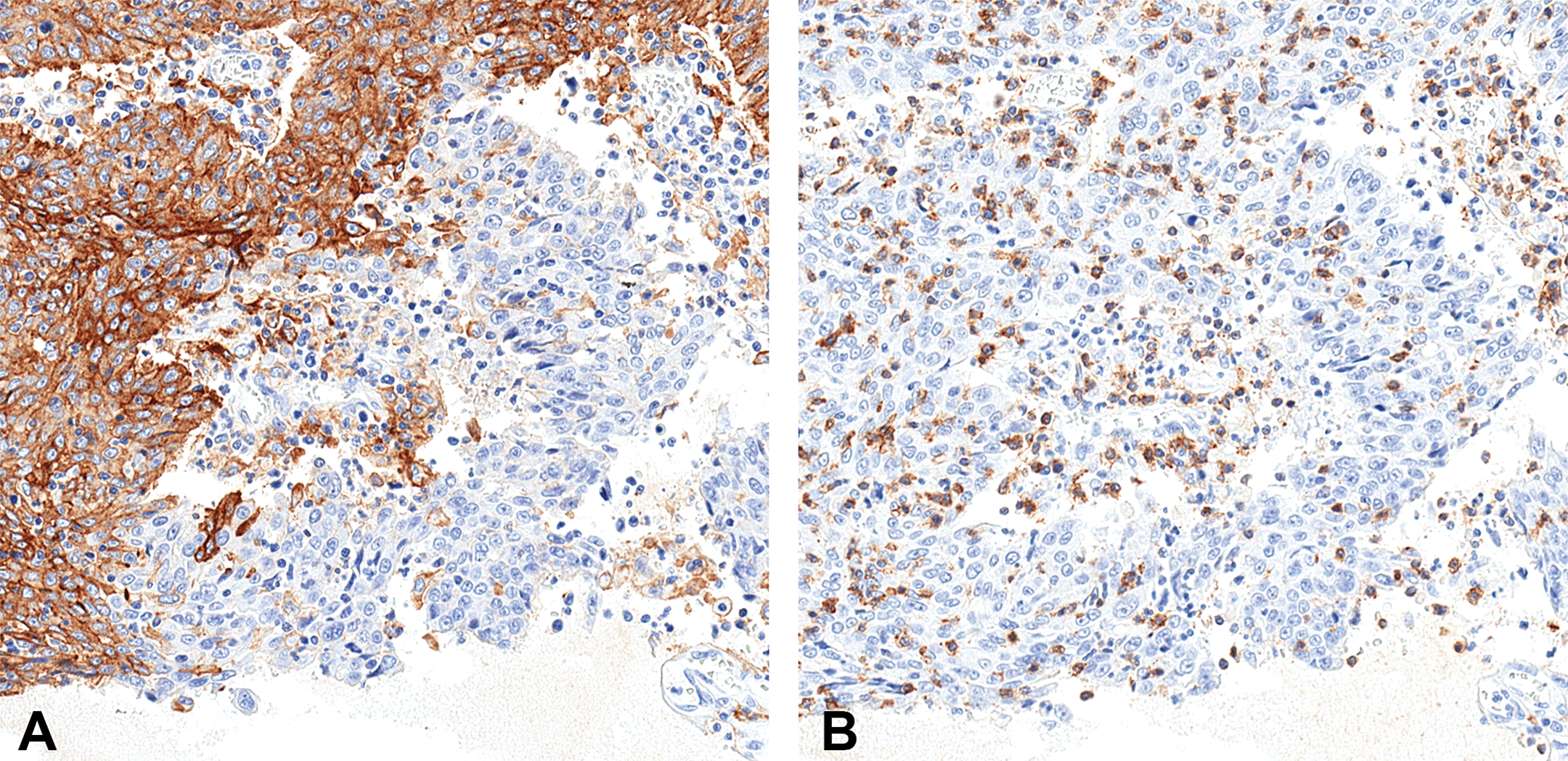
Pulmonary carcinoma (non-small cell lung cancer [NSCLC]) from a human patient demonstrating extensive but not universal expression of programmed death-ligand 1 (PD-L1), the molecule that binds the immune checkpoint receptor programmed cell death protein 1 (PD-1) in neoplastic cells (panel A). Cytotoxic (CD8+) T lymphocytes are scattered in the NSCLC regions expressing PD-L1 (panel B). Indirect immunohistochemical methods on serial paraffin sections to detect rabbit polyclonal antihuman PD-L1 (Cat# M4420; Spring Biosciences, Pleasanton, CA) and mouse monoclonal antihuman CD8 (Cat# M7103; Dako, Agilent Technologies, Santa Clara, CA), using 3,3’-diaminobenzidine as the brown chromogen and hematoxylin as the counterstain. Original objective magnification: 20×.
This minireview should serve as a brief introduction to primary issues of importance in the oncoimmunology (or immunooncology) arena, and in particular, the key roles that pathologists contribute to this endeavor. Scientists who wish to contribute to this venture will need broad foundational knowledge in cell and molecular biology for both normal and neoplastic tissues as well as immunology. In addition, individuals will be best served, if they have real-world experience in conventional scientific and regulatory practices utilized in industrial product discovery and development. The intense economic interest in oncoimmunology as a source for innovative and life-changing anticancer therapies guarantees the need for skilled comparative pathologists in this field for decades to come.
Footnotes
Author Contribution
Authors contributed to conception or design (BB, FA); drafting the manuscript (BB); and critically revising the manuscript (BB, FA). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
Since the time this research was completed, the second author’s affiliation has changed. Famke Aeffner is now with Amgen, Inc., South San Francisco, CA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
