Abstract
Hartley guinea pigs are widely used animal models of disease, particularly in studies of osteoarthritis. The purpose of this study was to investigate lesions in the costal cartilage from 16 male, 5- to 6-month-old Hartley guinea pigs. Routine histological sections from the costal cartilage and costochondral junction (longitudinal and cross sections) and sternum (for evaluation of bone marrow) were examined. All 16 (100%) animals had histological lesions involving the costal cartilage that included matrix degeneration and mineralization, reduced cellularity, and evidence of chondrocyte necrosis. Of the 16, 4 (25%) of the lesions contained blood vessels and 3 (19%) contained central osseous metaplasia. The cartilage lesions were accompanied by degeneration (sometimes with regeneration and/or fibrosis) in adjacent skeletal muscle in 15 of the 16 (94%) animals. The lesions in the costal cartilage were interpreted as dystrophic mineralization of unknown cause and appear to be incidental findings, although they bear some resemblance to lesions occurring in Tietze’s disease in humans. The significance of the lesions in skeletal muscle is unclear. Histological lesions of cartilage matrix degeneration and mineralization in these sites have not, to our knowledge, been reported previously.
Keywords
Hartley guinea pigs are widely used as animal models of human disease, particularly in studies of osteoarthritis (Flahiff et al. 2004). Here, we report the results of examination of incidental lesions discovered in the costal cartilage of 16 male Hartley guinea pigs that were euthanized for colony surveillance purposes. The Society of Toxicologic Pathology recommends examination of the sternum and rib for general toxicity studies to determine the health of bone (Haschek, Rousseuax, and Wallig 2013). Therefore, it is important to report novel lesions involving the costal cartilage and costochondral junction and to attempt to determine their biological significance. This site is also commonly examined to evaluate the endochondral ossification process in young animals because it is easy to collect and analyze. Our objective is to characterize previously unreported lesions consistently observed in the costal cartilage in male Hartley guinea pigs.
Materials and Methods
The costal cartilage and costochondral junctions, with adjacent attached skeletal muscle, and sternums from 16 five- to six-month-old male Hartley guinea pigs were collected after euthanasia. The costochondral junction and sternum (for evaluation of bone marrow) were fixed in 10% neutral buffered formalin and decalcified in 10% EDTA. After decalcification, the ribs were cut into longitudinal and transverse planes, processed routinely, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin for histological examination. Two ribs (1 for each plane) from each animal were examined. No other sites were examined.
Animals were group housed in an Association for Assessment and Accreditation of Laboratory Animal Care International accredited facility under approved Animal Care and Use Committee guidelines. The study was conducted in compliance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare. Animals were fed ad libitum Purina 5026 guinea pig diet (Purina LabDiet, St. Louis, MO), were administered no systemic treatments, and exhibited no clinical signs. Animals originated from two different vendors (vendor A, n = 10; vendor B, n = 6).
Lesions in the costal cartilage were characterized by grading the severity of matrix mineralization and necrosis on a scale of 0 to 3 (0, absent; 1, mild; 2, moderate; 3, severe). The presence of altered rib contour, chondral blood vessels, osseous metaplasia, and accompanying lesions in skeletal muscle adjacent to costal cartilage was reported as present (+) or absent (−; Table 1).
Lesions in the Costal Cartilages of Hartley Guinea Pigs.
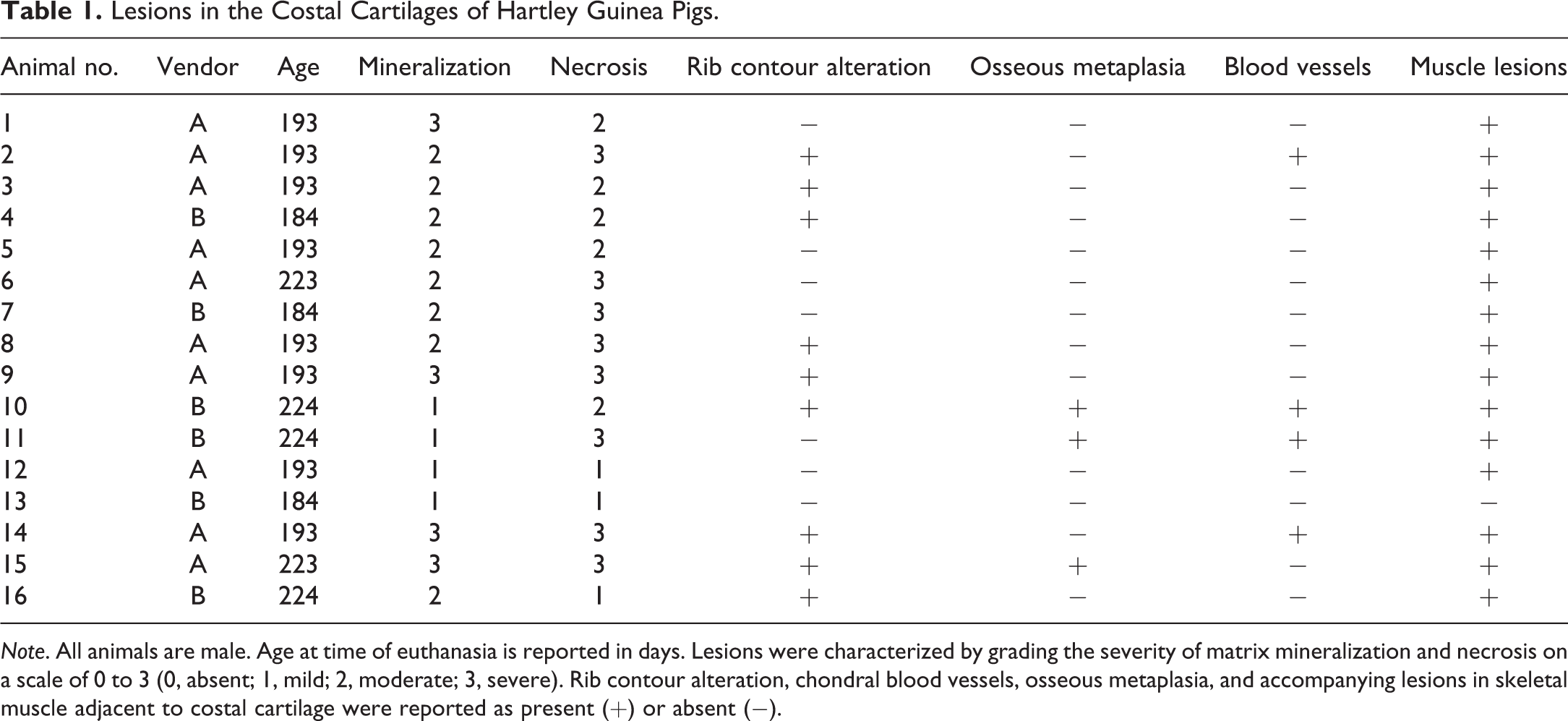
Note. All animals are male. Age at time of euthanasia is reported in days. Lesions were characterized by grading the severity of matrix mineralization and necrosis on a scale of 0 to 3 (0, absent; 1, mild; 2, moderate; 3, severe). Rib contour alteration, chondral blood vessels, osseous metaplasia, and accompanying lesions in skeletal muscle adjacent to costal cartilage were reported as present (+) or absent (−).
Results
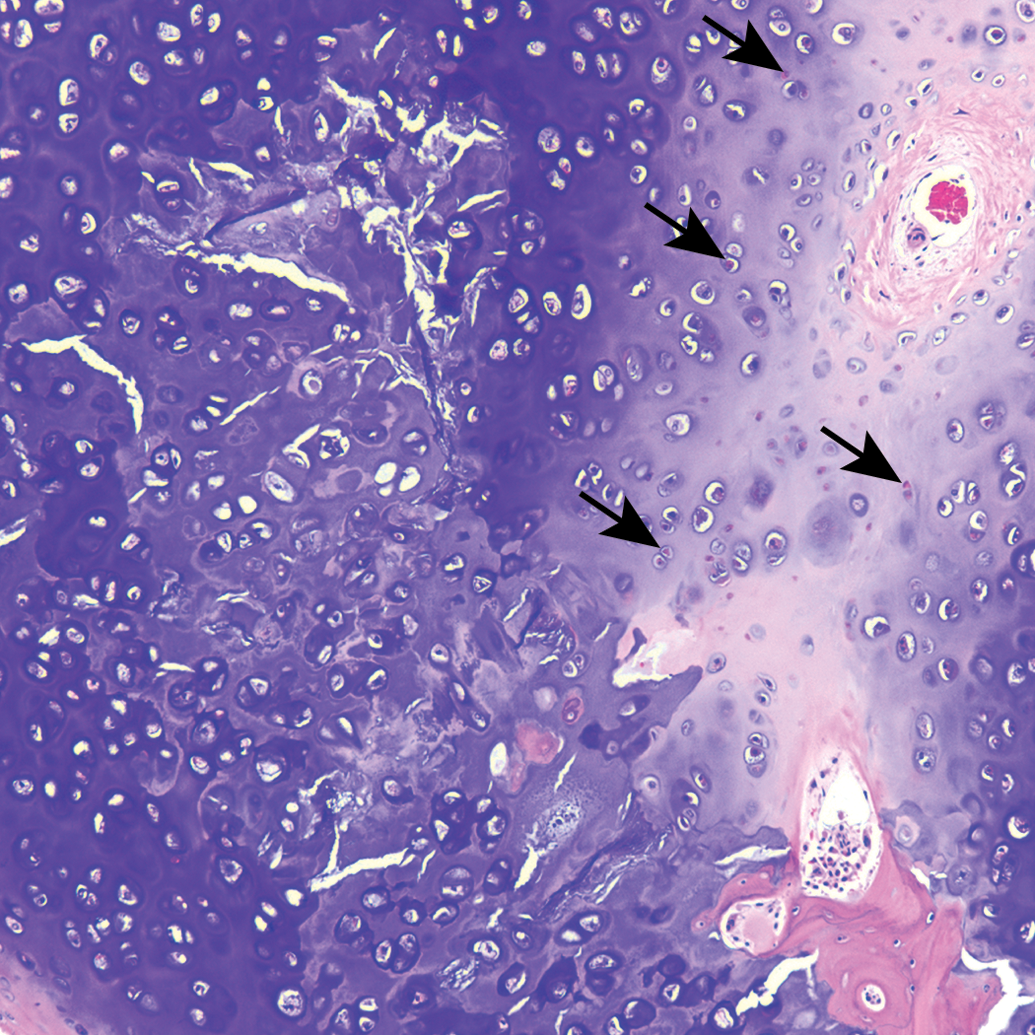
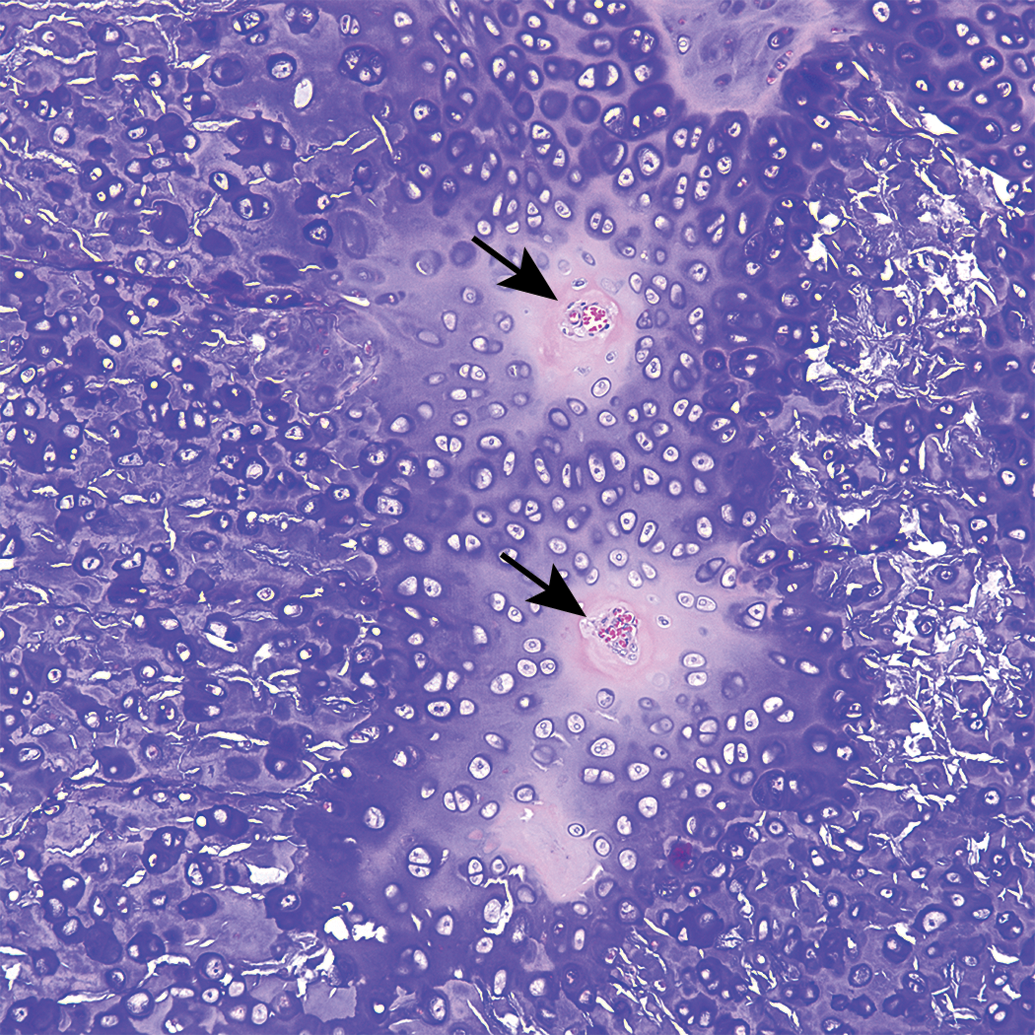
All sections of costal cartilages examined contained histological lesions of cartilage matrix degeneration and mineralization involving the central area of the costal cartilage (Table 1). These areas contained reduced numbers of chondrocytes compared to adjacent normal cartilage and variable numbers of necrotic chondrocytes (Figure 1). The lesions tended to be well demarcated and centrally located and were surrounded by a complete peripheral rim of unaffected cartilage (Figure 2). The contour of the costal cartilage was altered in seven cases at the site in which the lesion was most severe (Figure 2). In many of the sections, fragmentation of the matrix was associated with matrix mineralization, and presumably occurred at the time of sectioning. Three sites contained blood vessels in the interior of the lesion (Figure 3) and three sites contained areas of central osseous metaplasia (Figure 4). In two cases, blood vessels and central osseous metaplasia were concurrently observed. In all but one site, the cartilage lesion was accompanied by adjacent areas of degeneration and regeneration of skeletal muscle characterized by vacuolation/fragmentation of myofibers, variation in fiber diameter, the presence of internal nuclei, and, in some cases, the presence of fibrous connective tissue between adjacent myofibers (Figures 5–6). In most cases, these changes were mild; however, marked changes were noted in two of the animals (animal no. 8 and 14). No significant lesions were identified in the growth plates, costochondral junctions, or costal or sternal bone or bone marrow in any of the sections. There was no difference observed in lesions between vendors.
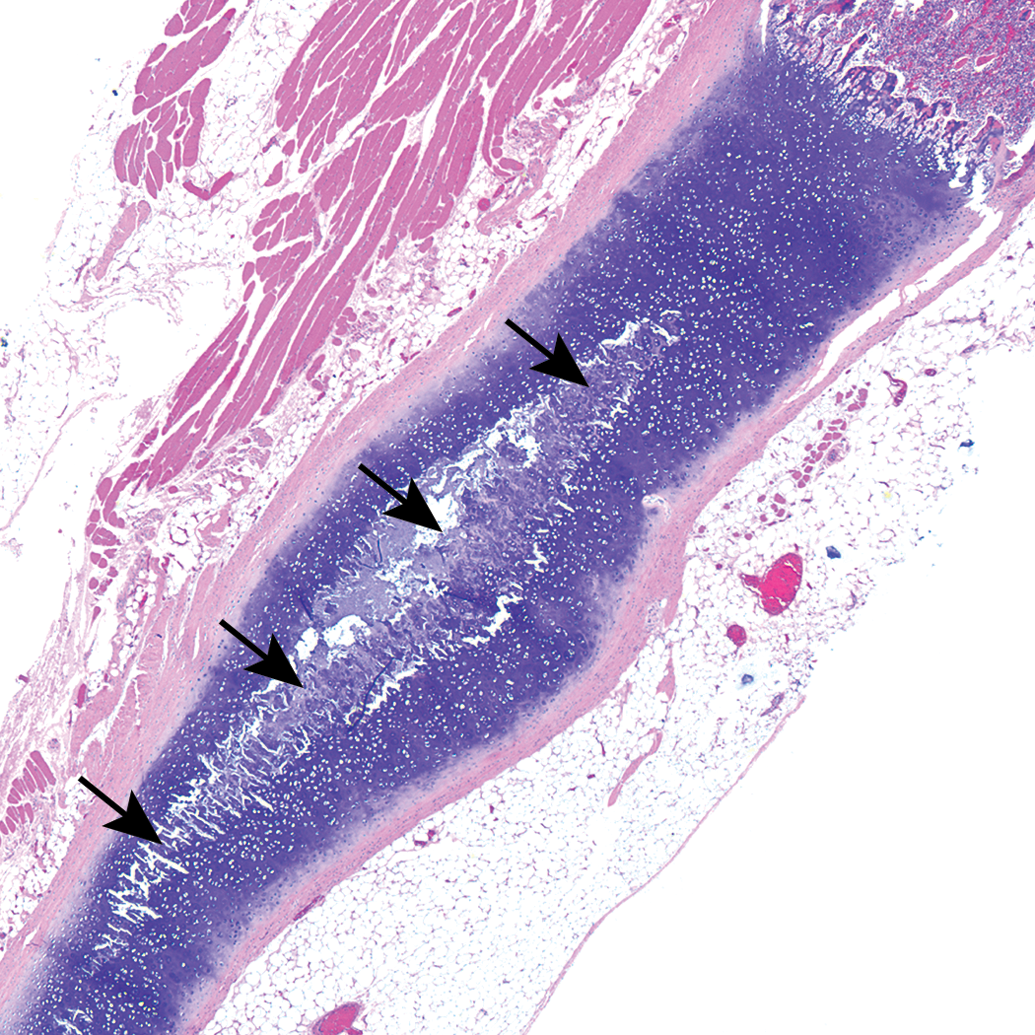
Guinea pig costal cartilage. Central mineralization of costal cartilage (arrows) accompanied by altered rib contour.
Guinea pig costal cartilage. Extensive matrix degeneration accompanied by chondrocyte necrosis (arrows) characterized by loss of basophilic nuclei and replacement with an eosinophilic amorphous structure and/or the presence of empty lacunae.
Guinea pig costal cartilage. Blood vessels (arrows) present in central area of mineralized costal cartilage.
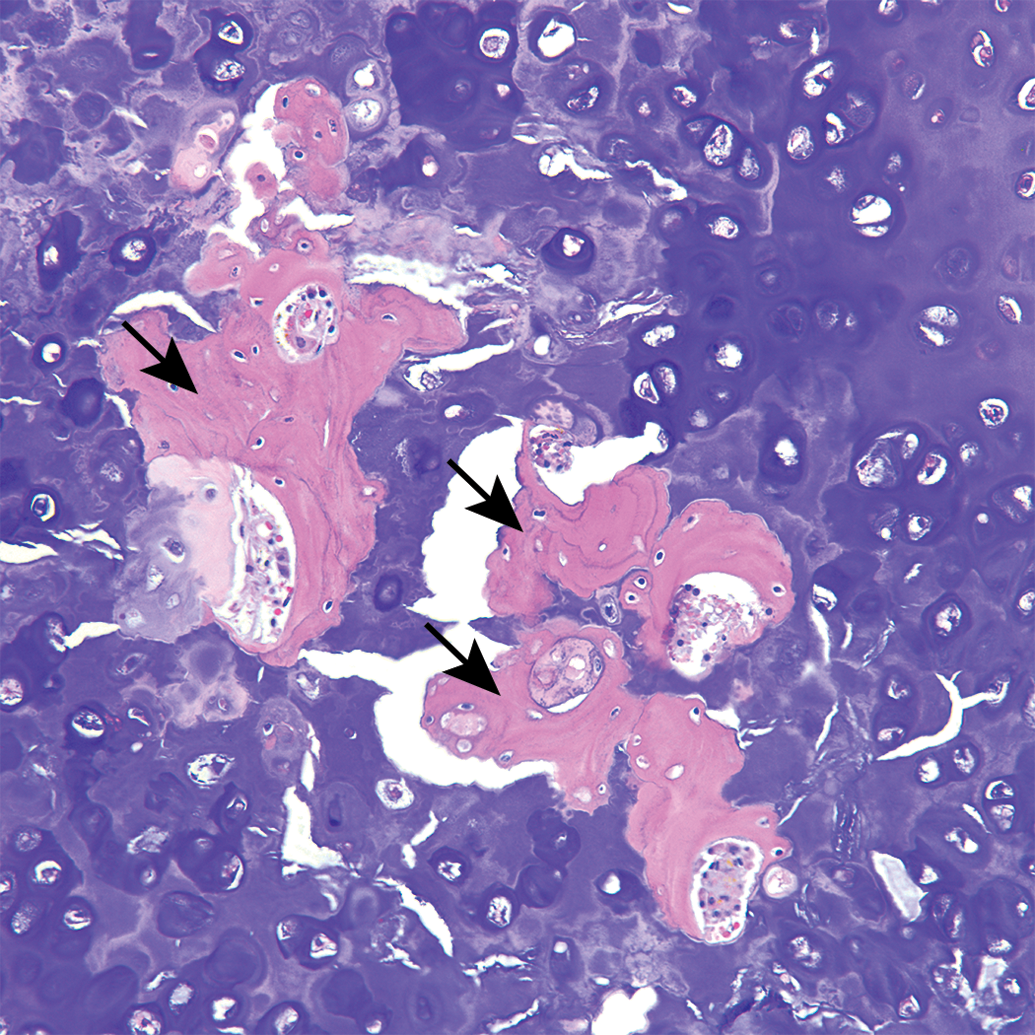
Guinea pig costal cartilage. Osseous metaplasia (arrows) surrounded by mineralized cartilage matrix.
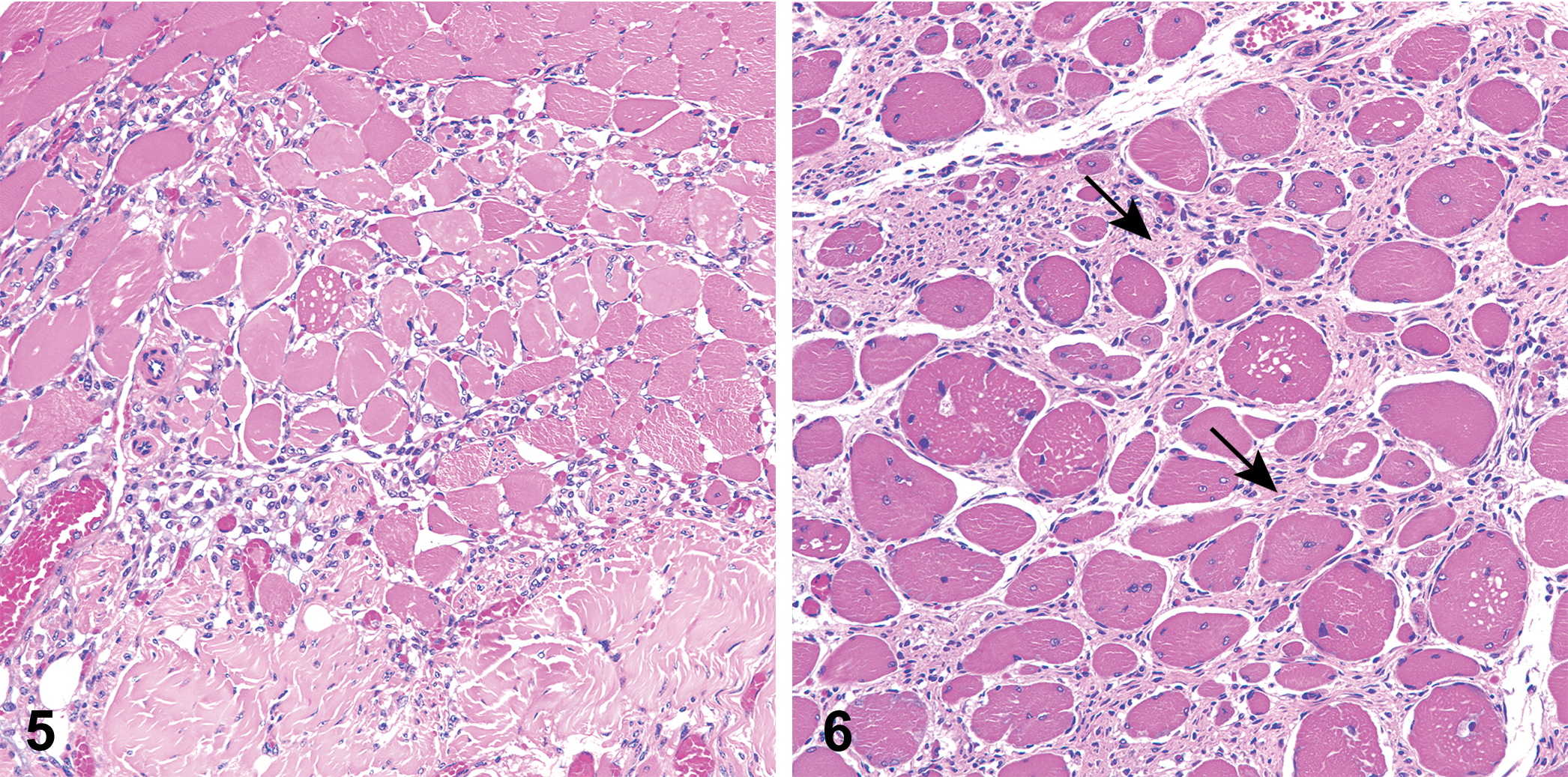
Skeletal muscle adjacent to costal cartilage. Muscular degeneration, characterized by vacuolization and fragmentation of muscle fibers, accompanied by regeneration (variation in fiber diameter, presence of internal myocyte nuclei, and hypercellularity), and the presence of fibrous connective tissue surrounding myofibers (arrows).
Discussion
The lesions present in the costal cartilage of 16/16 (100%) of the examined guinea pigs were interpreted as dystrophic mineralization of unknown cause and appear to be incidental findings, as no clinical signs were reported. The altered contour of the affected ribs, however, raises the possibility that these lesions could have potential clinical significance. A previous electron microscopy study reported “regular calcification at the central zone of cartilage in young guinea pig ribs” at the ultrastructural level, but histological lesions of matrix degeneration and mineralization in these sites have not, to our knowledge, been reported previously (Thyberg, Nilsson, and Friberg 1973). It is possible that ultrastructural calcification, observed in the costal cartilage in very young guinea pigs (0 to 65 days), preceded the dystrophic mineralization that was seen in the present study in animals that were 193 to 224 days of age (Thyberg, Nilsson, and Friberg 1973).
A background lesion identified in Fischer rats, termed mucinous degeneration or chondromucinous cystic degeneration, also affects cartilage (usually sternum but also articular and growth plate cartilage) and is associated with aging. The lesions are present in both sexes, are first seen at 130 to 180 days of age (i.e., older rats), and primarily involve the caudal 4 intersternebral cartilages. Although degeneration and necrosis of chondrocytes are features described in the rat lesions that are similar to those described in the present study in guinea pigs, they are also accompanied by chondrocyte clones, formation of cleft-like fractures and cysts, and cartilage proliferation, which were not observed in the present study. Importantly, mineralization of cartilage matrix is not described as a feature of the lesions in rats (Fossey et al. 2016; McInnes and Scudamore 2014).
Costal cartilage calcification (CCC) is a common finding associated with aging in humans but is not typically seen prior to 30 years of age (Ontell et al. 1997). Current literature does not, to our knowledge, report whether calcification of costal cartilage is associated with aging in guinea pigs as it is in humans. Nineteen cases of premature CCC in humans aged 17 to 31 were reported to occur with a variety of concomitant metabolic disorders (Rhomberg and Schuster 2014). However, because the animals in the present study were clinically normal, it is less likely that the costal cartilage mineralization we describe can be attributed to comorbid metabolic disorders. Of the 19 patients in the human report affected by premature CCC, 17 were female, and the report does not mention whether a female predominance has been reported previously (Rhomberg and Schuster 2014).
Literature in human medicine describes lesions similar to those seen in the present report associated with Tietze’s disease or Tietze syndrome, a condition first described by Tietze as a “nonsuppurative” painful swelling of the upper costal cartilages that spontaneously regresses (Kayser 1956). The swelling of the costal cartilage associated with Tietze’s disease may be associated with heat, erythema, and mild to severe pain (Rabey 2008). Because costochondritis shows similar clinical signs of tenderness and pain, it is commonly confused with Tietze’s disease. However, costochondritis is distinguished from Tietze’s disease by location: costochondritis occurs at the costosternal joint, whereas Tietze’s disease occurs within the costal cartilages (Gijsbers and Knapp 2010; Kayser 1956; Rabey 2008). In addition, costochondritis has no noticeable swelling, whereas Tietze’s disease exhibits chondral swelling. Due to both the altered rib contour noted in several of our animals and the location of the lesions, the lesions present in the guinea pigs in this report appear most similar to those identified in Tietze’s disease.
The underlying pathophysiology of Tietze’s disease in humans is not well understood, and documentation of the histological lesions is rare. It usually affects older children and young adults, has an abrupt onset, and may be associated with chest pain radiating to the arms or shoulders. Although it is sometimes described as an inflammatory disorder, those few cases that include histological evaluation exhibit no evidence of inflammation. In fact, a “complete absence of inflammatory changes in the cartilage or overlying soft tissue is striking” (Levey and Calabro 1962, 265). Laboratory studies, including white blood cell and differential, hematocrit, and erythrocyte sedimentation rate are also reported to be normal. Radiographic studies sometimes report increased calcification at affected sites. The condition is primarily considered to be benign and self-limiting (Gijsbers and Knapp 2010; Rabey 2008). The majority of Tietze’s disease cases also present unilaterally and the disease is considered to be rare (Gijsbers and Knapp 2010). In contrast, the lesions identified in the guinea pigs that were examined in the present study were present in every rib that was examined, suggesting that additional sites likely were affected.
The histologic lesions of Tietze’s disease are reported to include “increased vascularity” (although costal cartilages normally are avascular tissues) and degenerative matrix changes with the formation of clefts that may undergo calcification and patchy loss of ground substance, leading to a fibrillar appearance to the costal cartilage matrix (Cameron and Fornaiser 1974; Levey and Calabro 1962). Tietze’s disease also has been described as “dystrophy” of the costal cartilage, and concurrent osseous metaplasia, as was identified in the present study, has been reported (Levey and Calabro 1962). Osteogenesis is preceded by infiltration of cartilaginous tissue with blood vessels. Therefore, the blood vessels seen in 4 of the lesions may represent the preliminary stages of the osseous metaplasia present in three of the lesions. In fact, in two animals, blood vessels and osseous metaplasia were concurrently present within the same lesion.
The pathogenesis of the skeletal muscle lesions that was identified in the present study, and their potential association with the costal cartilage lesions, currently are unexplained. Interestingly, adjacent skeletal muscle degeneration and regeneration were also reported in one case of Tietze’s disease (Kayser 1956).
Future work remains to determine if these lesions are also present in female Hartley guinea pigs, and are present in other cartilaginous tissues, and to determine the age of onset and whether or not severity progresses with age. Tietze’s disease in humans is poorly understood but is described to occur equally in males and females and is not associated with concurrent systemic disease (Kayser 1956; Levey and Calabro 1962). Further characterization of the lesions occurring in guinea pigs and determination of their pathogenesis may provide insights into the disease in humans.
Conclusions
In summary, we report the presence of costal cartilage degeneration and mineralization in young male guinea pigs that has some similarities to Tietze’s disease in humans.
Despite the lack of observed clinical signs in these animals, the lesion may confer pain due to local deformity similar to Tietze’s disease. Since costochondral junctions are a common site analyzed for general toxicity studies, it is important to report this lesion that was present in all of the animals that were examined. Future work remains to determine biological significance of these lesions and may lend insights into the disease in humans.
Footnotes
Acknowledgments
Tissues and slides in this study were prepared and provided by American Preclinical Services, Inc. Josh Parker and Lindsey Harper from the Carlson Lab at the University of Minnesota, College of Veterinary Medicine assisted with image processing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
