Abstract
The immune system is a critical component in defense against viral, bacterial, parasitic, and fungal diseases. Immunological mechanisms, including immunological mediators, innate immunity, cell-mediated immunity, and humoral-mediated immunity, serve to maintain homeostasis and protect the host from disease. Immunological variation can impact defense mechanisms, however. Two factors in particular that can influence immune function are the single nucleotide polymorphisms (SNPs) and aging. SNPs affecting inflammatory cytokines are an important modifier involved in a number of diseases such as asthma, periodontal disease, atherosclerosis, diabetic retinopathy, psoriasis, and osteoporosis. Age-related alterations to the immune system have also been studied and documented. The genetic makeup of different strains of mice and the age of these different strains cause large differences in susceptibility to infection, with influenza virus infection among the most widely studied. The mechanism of these differences due to either genetics or age is not known but can be investigated in strain- and age-specific infectious disease models.
Keywords
The immune system is crucial for defense against opportunistic infections, prevention, and clearance of viral, bacterial, parasitic, and fungal diseases, as well as preventing the exacerbation of latent viral infections. Immunological mechanisms that support health and prevent disease include immunological mediators, such as cytokines and chemokines, that call in and activate cells of the immune response; cells of innate immunity, including natural killer, macrophages, and dendritic cells; cell-mediated immunity; and humoral-mediated immunity, including IgM and IgG. Immunological variation can occur in any one or more of these defense mechanisms as well as microbiome or diet. Two factors in particular that can influence immunological variation with regard to immune function are the genetics of inflammatory single nucleotide polymorphisms (SNPs) and aging. SNPs affecting inflammatory cytokines are an important modifier involved in a number of diseases. Large differences in inflammatory responses have been reported for SNPs that are associated with inflammatory cytokines and are associated with disease conditions such as asthma, periodontal disease, atherosclerosis, diabetic retinopathy, psoriasis, and osteoporosis. Additionally, there is an abundance of studies reflecting the effect of age on the immune system. Well-characterized strains of mice and rats are available and important in better understanding the impact of SNPs and aging on the immune response using animal models of infectious disease. The mechanisms underlying these differences due to either genetics or age are not yet fully elucidated, though much progress has been made in these fields. The importance of continued progress toward the betterment of human health and the quality of life cannot be underestimated.
SNPs
SNPs have been studied as contributors to physiological variation. The number of known SNPs has increased exponentially since their discovery and is reported to be greater than 9 million (Sherry et al. 2001). The roles and impact for SNPs and the association with disease are becoming more prominent at the present time.
Many diseases affecting man, such as influenza, have an inflammatory component. SNPs occurring in the inflammatory cytokine region are believed to have a significant impact on inflammatory disease treatment and outcome. Studies on the effect of SNPs in the inflammatory cytokine region and influenza disease have not been investigated; however, these studies would be important in gaining further knowledge toward treatment options, as yearly epidemics and sporadic pandemics (1918, 1957, 1968, and 2009) have the potential to affect a large percentage of the population. The influenza pandemic in 1918, for example, caused by an H1N1 virus, resulted in the death of at least 50 million people worldwide (Johnson and Mueller 2002). The pandemic in 1957, due to an H2N2 virus, and the pandemic in 1968, due to an H3N2 virus, caused approximately 2 million deaths (Nicholson, Wood, and Zambon 2003; Webby and Webster 2003). The pandemic in 2009, due to an H1N1 virus, resulted in 18,631 laboratory-confirmed deaths, though studies suggest that the death toll may have been 10-fold higher (Simonsen et al. 2013).
Large differences in inflammatory responses have been reported for SNPs for the inflammatory cytokines interleukin 1 (IL-1) and TNFα that are associated with numerous disease conditions as modifiers not the cause. IL-1 and TNFα SNPs result in increased production and/or longer periods of production (more stable RNA) for these inflammatory cytokines and are associated with asthma, periodontal disease, atherosclerosis as a cause of coronary artery disease (CAD), diabetic retinopathy, psoriasis, and osteoporosis (Kornman and Polverini 2014; Tsimikas et al. 2014; Kornman and di Giovine 1998). IL-1 (+) gene variants, defined by 3 SNPs in the IL-1 gene cluster, are associated with higher levels of proinflammatory cytokines and/or longer periods of production due to more stable RNA. IL-1 (+) gene variants, smoking, and diabetes are associated with chronic periodontitis (CP) progression. All other genotypes are classified IL-1 (−). Elevated IL-1β levels are associated with CP as well (Tsimikas et al. 2014; Kornman and di Giovine 1998). Additionally, a clinically relevant biological link between proinflammatory IL-1 genotype and coronary artery disease has been observed, as defined in a study by Tsimikas et al. (2014) and Ray (2014).
SNPs have evolved into new areas of research and interest, including pharmacogenetics (study of genetic variation in drug efficacy) and toxicogenomics (study of genetic variation in toxicity). Thus, genetic polymorphisms in the human genome may result in better pharmaceuticals using personalized, stratified, or precision medicine approaches including stratified trials, especially for inflammatory diseases (Kornman and Polverini 2014; Tsimikas et al. 2014; Braun et al. 2015; Ray 2014). Luster and colleagues have reported on numerous studies showing an association between cytokine SNPs and increased susceptibility to disease. The odds ratios of disease for IL-1RA (+2018) or TNFα (−308) were elevated and suggested that certain cytokine polymorphisms play a significant role in silicosis by modifying the extent and susceptibility to disease (Yucesoy et al. 2001). Yucesoy et al. also reported an association between the IL-1RA (+2018) polymorphism and silicosis and suggested that this SNP may confer increased risk for the development of disease.
Aging
It is well known that decreases in immunological responses have been observed in older individuals. This decline in immune function is referred to as “immunosenescence.” The processes underlying immunosenescence are complex; however, as some studies have shown that while certain functions and mechanisms decline with age, others remain unaffected or even become elevated. Thymic involution, inflammation, oxidative stress, DNA damage, and genetic factors, among others, likely contribute to alterations in cell numbers and/or function (Ponnappan and Ponnappan 2011).
The clinical evidence for immunologic dysfunction in the elderly include increased incidence of infections (bacterial and viral), increased incidence of cancer, increased prevalence of autoimmune diseases, decreased responses to vaccination and secondary vaccine challenges, and increased rate of graft-versus-host disease among older patients (Longo 2003). There was reported a 25% increase in deaths attributable to infectious disease that occurred between 1980 and 1992 for persons aged 65 years and older (Longo 2003). In addition, influenza and pneumococcal pneumonia have a higher fatality rate in older people than in young adults, and in Western countries there is an increase in the mortality rate by 89-fold for pneumonia and influenza in people over 65 years compared to those aged 25–44 years old (Troen 2003). Age-associated decreases in naive T and B cell numbers, with relative increased proportions of memory cells in each compartment, respectively; decreases in neutrophil and monocyte/macrophage function; alterations in cell-signaling; and general increases in inflammation may contribute to these outcomes (Ponnappan and Ponnappan 2011).
Age can be perplexing, however, as it relates to influenza disease in humans. Interestingly, there was an atypical high mortality rate among nonelderly adults while the elderly were relatively spared and young children had milder disease during the 2009 H1N1 pandemic. The presence of cross-reactive antibody does not explain the mortality trend, as children did not have cross-reactive antibody to the new pandemic H1N1 virus of 2009, while 6% to 9% of adults (18–64 years of age) had cross-reactive antibody and 33% of older adults (over 64 years of age) had cross-reactive antibody. Numerous attempts have failed to explain this discrepancy (Katz et al. 2009).
Studies in mice indicate that both genetics and age determine the susceptibility and resistance to pandemic influenza virus (H1N1). The effect of age in genetically identical C57BL/6 mice is demonstrated in Figure 1. Adult C57BL/6 mice (8 weeks of age) were more resistant to influenza H1N1 virus infection than either very young (4-week-old mice) or elderly mice (6- to 9-month-old mice). Mouse models are available to better understand the role of cytokines using C57BL/6 TH1 mice that produce predominantly interferon gamma (IFNγ) and a strong cell-mediated immune response or BALB/c TH2 mice that produce predominantly humoral-mediated response as reviewed by Netea et al. (2005). In humans, it is most likely a combination of age, genetics/SNPs, and prior exposure to influenza strains which plays a role in susceptibility and resistance. It is important to understand the effects of immunological variations in order to better guide decisions regarding patient care and treatment.
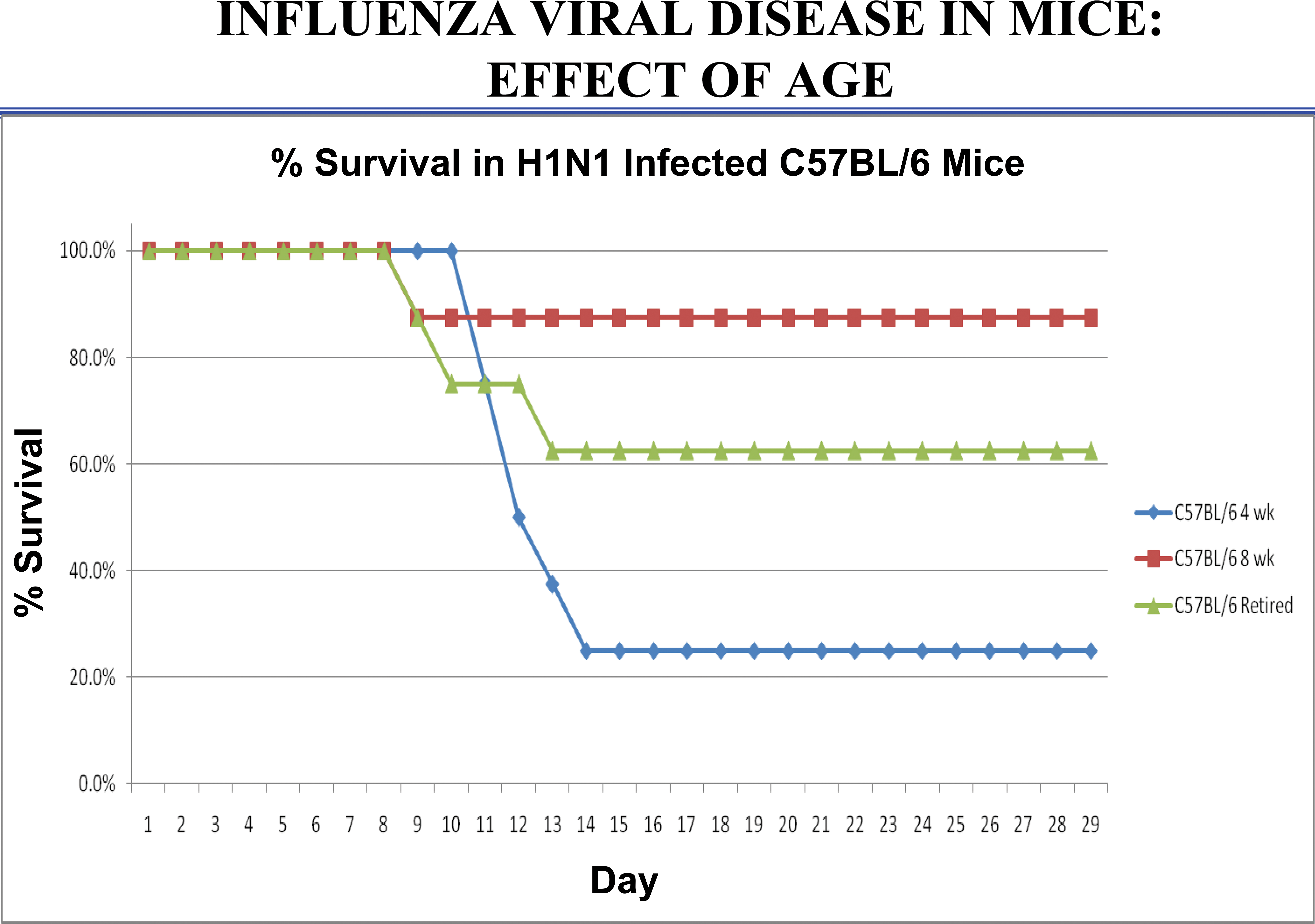
Effect of age of female C57BL/6 mice when infected with pandemic influenza A/California/04/09 (H1N1) virus obtained from Centers for Disease Control by Alphavax and passaged in Madin Darby Canine Kidney (MDCK) cells one time before providing to Burleson Research Technologies. Influenza A/California/04/09 virus stock was used to infect mice intranasally with 50-µl volume (6 × 105 plaque forming units [PFU]/mouse). Juvenile mice (4 weeks old at infection), adult mice (8 weeks old at infection), and elderly mice (retired breeders; 6–9 months). Study design was approved by the Institutional Animal Care and Use Committee (IACUC) committee in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited vivarium.
Conclusion
The immunological defense processes that provide health and prevent disease are multifactorial, encompassing a variety of mechanisms. Immunological variation can occur in any one or more of these defense mechanisms, accounting for differences in individual immune responses to antigenic challenge. SNPs and the immunosenescence are two factors that may contribute to such variation. Further study in this important area of immunology will lead to better patient care and treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
