Abstract
A number of factors related to study design have the potential to impact clinical pathology test results during the conduct of nonclinical safety studies. A thorough understanding of these factors is paramount in drawing accurate conclusions from clinical pathology data generated during such studies, particularly when attempting to make the distinction between test article and nontest article–related effects. Study design and conduct variables with potential to impact clinical pathology data discussed in this overview include those related to species and test system, animal age, animal care and husbandry practices, fasting, acclimatization periods, effects of transportation and stressors, route of administration, effects of in-life and surgical procedures, influence of study length, timing of blood collections, impact of vehicle/formulation composition, and some general concepts related to drug class. The material presented here is a summary based on information presented at the 35th Annual Symposium of the Society of Toxicologic Pathology (June 2016), during Symposium Session 2 titled “Deciphering Sources of Variability in Clinical Pathology—It’s Not Just about the Numbers.”
Sound study design and study conduct practices are critical in generating accurate and relevant data during nonclinical toxicology studies. Due to their dynamic nature, clinical pathology end points in particular can be affected by a wide variety of factors related to how toxicology studies are designed and executed. The influence of these factors on clinical pathology data is completely independent of and in addition to direct test article–related effects which collectively can confound data interpretation and impair the ability to distinguish between test article–related and nontest article–related changes. Prior to analysis of clinical pathology data, it is important to become familiar with the study protocol and all study procedures/methods being utilized in order to draw accurate conclusions. In addition, efforts should be made during the study design phase to minimize the inclusion of study practices known to introduce sources of variability, which may limit the utility of clinical pathology data generated on toxicology studies.
Animals and Test System
Age-related Changes
Understanding differences between adult and young animals among clinical pathology end points, as well as changes occurring during growth, is essential to enable accurate interpretation of data generated on nonclinical toxicology studies. It is common to start with adolescent animals, particularly in dog and rodent studies in which a rapid growth phase is occurring. Growth causes physiological changes affecting clinical pathology end points that should not be confused with pathology or diseases. Specific effects at different stages of growth will occur in animals, as the maturation of organs and systems progresses (e.g., hematologic, musculoskeletal, lymphoid, etc.). These effects are generally only going to become discernibly prominent in studies that are of longer duration (i.e., >28 days) or in juvenile toxicology studies and are most notable among end points related to the hematologic system, bone growth, and increasing bodyweight/muscle mass (see Table 1).
Common Changesa among Clinical Pathology End Points Associated with Growth and Maturation in Dogs and Rats.
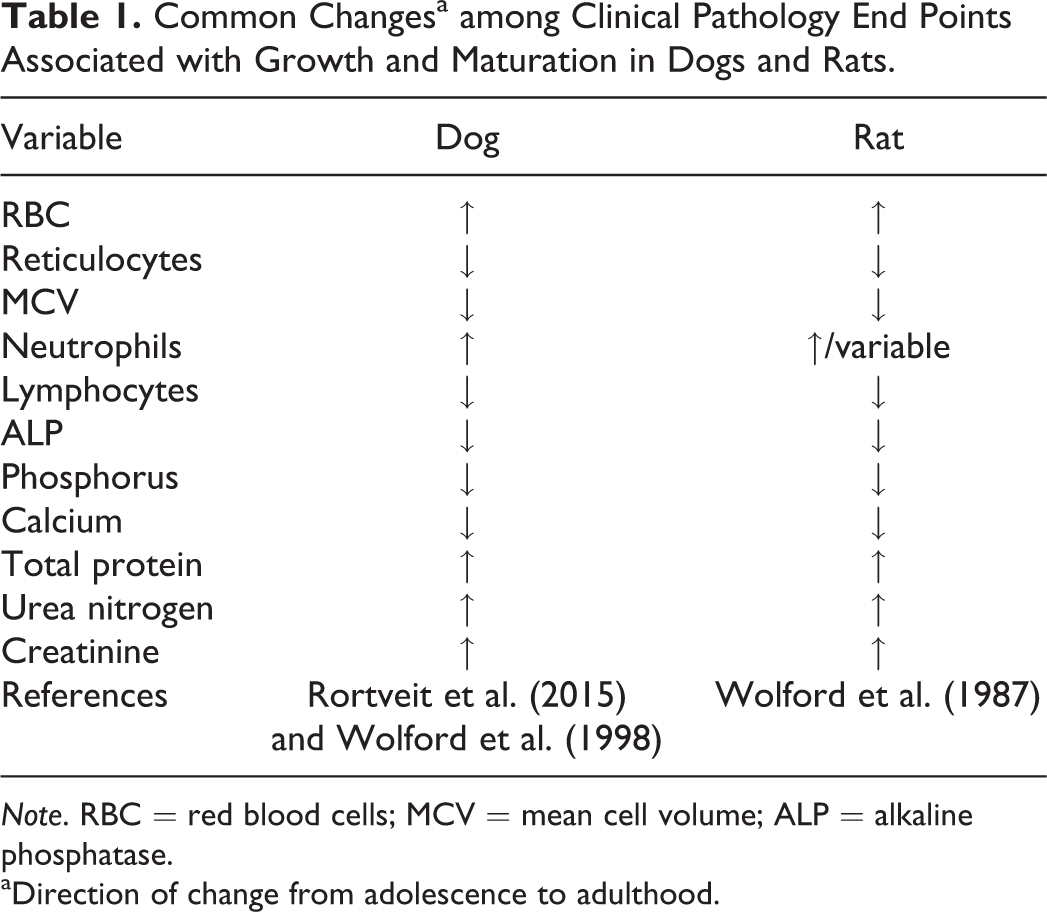
Note. RBC = red blood cells; MCV = mean cell volume; ALP = alkaline phosphatase.
aDirection of change from adolescence to adulthood.
In dogs, red blood cell (RBC) mass values (erythrocyte count, hemoglobin concentration, and/or hematocrit percentage) are lower in puppies and steadily increase with age (Rortveit et al. 2015; Wolford et al. 1998), while RBC mass values usually reach adult magnitudes around the time of sexual maturity in animals (Chandler 1992). Concurrent mild progressive decreases in reticulocyte counts as the RBC mass pool matures are also seen in maturing animals (i.e., dogs and rats) during this time (Wolford et al. 1987, 1998). Among white blood cells (WBC), younger animals tend to have higher lymphocyte and lower neutrophil counts early in life in comparison to adult animals (Wolford et al. 1987, 1998), although the overall proportion of neutrophils decreases until maturity (Provencher Bolliger and Everds 2010). Young dogs also have higher total WBC counts relative to adults, predominantly during the first 8 weeks of age (Harper et al. 2003). Platelet counts have been reported to be similar in newborn and adults (Palis and Segel 2010).
Changes in clinical chemistry variables depend on the developmental stage of organs and systems as well as the particular species, nutritional status, housing, and environmental conditions of the animals. The most common systems with developmental differences in clinical chemistry parameters are hepatic, renal, and skeletal. Hepatic maturity is reached quite early in life (i.e., 5 weeks to 8 weeks in dog), but young animals typically have lower serum albumin concentrations and lower glycogen stores that can make them susceptible to hypoglycemia (Johnston, Root Kustritz, and Olson 2001; Rosset et al. 2012). The development of the renal system may affect glomerular filtration rate (GFR) in young animals (decreased GFR in comparison to adults). In neonatal dogs, higher urea nitrogen concentrations at birth and 1 week of age can result from dehydration associated with renal immaturity because nephrogenesis is incomplete until 3 weeks of age. Decreased creatinine and total protein concentrations in young puppies may be attributed to their lower muscle mass (Rosset et al. 2012). Serum concentrations of calcium (total and unbound), phosphorus, magnesium, and alkaline phosphatase (ALP) activity are affected by skeletal system development. Progressive decreases in calcium and phosphorus concentrations and ALP activity will be seen over time as dogs and rats mature as related to slowing new bone growth/formation. Increased ALP activity in young animals can also be due to colostrum ingestion in dogs (Rosset et al. 2012).
Findings among clinical pathology end points in geriatric/mature animals attributable to old age are generally only applicable to rodents in longer term carcinogenicity studies. Given that only limited clinical pathology evaluations are conducted in most carcinogenicity studies, the effects of old age on the interpretation of these data are generally minimal (Young et al. 2011). Changes commonly observed among clinical pathology end points in older rodents (>12 months) are often related to naturally occurring or opportunistic disease (e.g., leukemia, sepsis, etc.) or associated general moribundity (increased liver enzyme activities, urea nitrogen/creatinine concentrations, etc.).
Animal Care and Husbandry
The influence of animal care and husbandry on clinical pathology data is often related to the degree, or lack thereof, of stressors introduced into the test system. Stressors have been shown to influence a wide variety of measured end points and organ systems (e.g., hematologic, lymphoid, endocrine, and reproductive) in nonclinical toxicology studies and should be taken into consideration during data interpretation (Everds et al. 2013). Sources of stress include those related to animal transportation and acclimatization, various in-life procedures, housing and fasting procedures as well as enrichment practices.
Transportation
Most laboratory animals used on preclinical studies will be transported from the supplier to the test facility where the study will take place. Transportation will inevitably cause stress and some discomfort, as the animals are exposed to unfamiliar environments for variable periods of time. Stress associated with transportation, although not long lived, has been shown to impact organ systems that can alter clinical pathology end points (Ochi et al. 2016), which highlights the importance of sufficient acclimatization periods following transportation. Typically, a minimum of 7 to 14 days (Obernier and Baldwin 2006; Ochi et al. 2016) is adequate in allowing a return to homeostasis for most species, although animals that are young, severely stressed, and/or have stress-sensitive genotype may require longer period to acclimatize.
The influence of stressors on clinical pathology results is often due to a superimposed combination of those caused by acute excitement or epinephrine-based effects and by cortisol/corticosterone release, which are typically longer acting or “chronic” in nature (Everds et al. 2013). Depending on the timing of the sample collections to the stressful event, the resulting effects on clinical pathology data may be varied. The most common clinical pathology end points affected by stress include total WBC (increased), neutrophil (increased), lymphocyte (decreased), and eosinophil (decreased) counts. Acute stress or excitement alone will generally cause transient increases in all leukocyte subtypes (as opposed to decreased lymphocyte and eosinophil counts) and may also transiently increase red cell mass and/or platelet counts as related to splenic contraction. Clinical chemistry end points are less commonly affected, although increases in total bilirubin concentrations have been reported as significantly increased in beagle dogs and cynomolgus monkeys following transportation, as well as increased aspartate aminotransferase (AST) activity in monkeys and decreased triglyceride concentrations in dogs (Ochi et al. 2016). Dogs also have a stress-induced isoenzyme of ALP which may be increased following stressful event (Stockham and Scott 2008).
Husbandry/Caging
Caging, food and water supply, air quality, humidity, temperature, and providing species recommended enrichment can all affect the well-being of laboratory animals and have the potential to induce stress. Although the effect of stress is usually transient, it is essential to take these factors into consideration when designing experimental protocols.
Housing of laboratory animals is an important animal welfare issue and has been extensively reviewed and transformed in recent years to allow animals to be housed in conditions more consistent with their own species natural preference. By creating more adapted housing environments, the animals are less stressed, reducing the potential impact on animal health and research data that is generated on study. Providing cages that allow for group instead of single housing has been beneficial for many laboratory animal species, based on the understanding that animals group housed have less “distress” than those housed individually (Everds et al. 2013). Based on this evidence, group/social housing of animals has been adopted by many modern laboratory facilities.
When comparing clinical pathology data in cynomolgus monkeys that were group or individually housed, only minor differences in a few clinical pathology end points were observed (Voyer and Provencher 2016). Pen housed animals, in comparison to singly housed animals, showed slight decreases in neutrophil count, with slight increases in absolute lymphocyte and eosinophil counts. Among clinical chemistry end points slight decreases in cholesterol, globulin and albumin concentrations, and slight increases in triglyceride and urea concentrations were observed in group- versus single-housed animals (Voyer and Provencher 2016). The overall magnitude of changes between pen and individually housed animals was not considered to be biologically significant or great enough to impact the overall interpretation of core toxicology measures in nonclinical studies using group-housed animals.
Enrichment
Similar to group housing, providing enrichment to laboratory animals is considered part of improving laboratory animals’ psychological and physiological “well-being” that consequently helps to improve the quality and reproducibility of results in research (Wirz et al. 2015) and will provide a state of eustress (“good stress”; Everds et al. 2013).
As an example, providing nesting materials to mice is an ethologically relevant environmental enrichment, which reduces cold stress, improves feed conversion, and increases litter size. In the field of toxicology, providing nesting material has not been a routine practice which raises fear of introducing new elements into study systems that may interfere with traditional toxicology study measures.
A recent study in mice sought to determine if providing nesting material would affect study results following administration of a known immunosuppressive agent (cyclophosphamide; Gariepy et al. 2015). It was hypothesized that nesting material would reduce study-related stress and could potentially impact immune function or immunological parameters. Results showed that clinical signs and observation scores associated with stress were reduced in mice provided with nesting material, control and treated, confirming the known positive effect on animal welfare measures. Expected changes in clinical pathology, immune function, and histopathology end points were observed in cyclophosphamide-treated animals, with and without nesting material with changes that were similar between animals with and without nesting material. These results, as well as those of another recent study in mice and rats (Churchill et al. 2016), showed that adopting measures to improve animal welfare did not have a meaningful impact on clinical pathology end points in mice and rats.
The implementation of exercise periods has been another recent advancement in providing a source of natural enrichment to laboratory animals participating in toxicology studies. Increased activity during exercise periods can occasionally be associated with mild to moderate sporadic increases in musculoskeletal enzyme signals including increases in the activities of AST, creatinine kinase (CK), alanine aminotransferase (ALT), and/or lactate dehydrogenase (LDH) related to exertion and mild muscle trauma.
Fasting
The fasting status of laboratory animals and the duration of fasting are important factors that may lead to variation in clinical pathology data collected during toxicology studies. The fasting of animals prior to blood collections is generally considered to reduce the overall variability in clinical pathology data and to reduce the incidence of lipemic samples (Hall 2001). Many regulatory guidelines and the Joint Scientific Committee for International Harmonization of Clinical Pathology Testing recommend fasting of animals before blood collection for hematology and clinical chemistry analysis (Food and Drug Administration 2003; Organization for Economic Cooperation and Development 1995; Weingand et al. 1992). In rats, serial blood sampling in fasted animals over 48 hr demonstrated progressive decreases in glucose, triglyceride, and cholesterol concentrations, relative to nonfasted animals (see Table 2; Waner and Nysak 1994). ALP activity was also decreased in fasted rats relative to nonfasted, presumably as a consequence of reduced contribution of the intestinal isoenzyme of ALP (Waner and Nysak 1994). Prolonged fasting or severe food restriction/anorexia (>48 hr) has been shown to cause bone marrow and lymphoid depletion; lower leukocyte, platelet, and reticulocyte counts; and increases in ALT and AST activities (Moriyama et al. 2008).
Effect of Fasting on Selected Clinical Pathology End Points in Control Rats.a
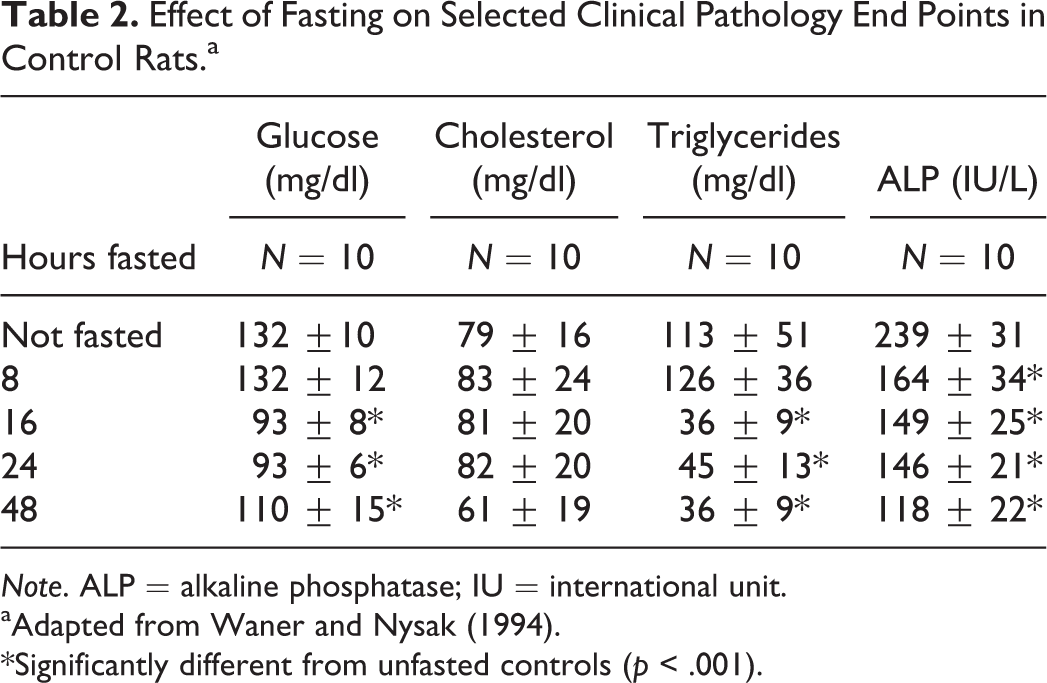
Note. ALP = alkaline phosphatase; IU = international unit.
aAdapted from Waner and Nysak (1994).
*Significantly different from unfasted controls (p < .001).
In-life Procedures
Room activities, clinical observations, special examinations (electrocardiogram, ophthalmology exams, etc.), and restraint all have the potential to impact clinical pathology test results either through stress induction or mild muscle trauma associated with study activities. Ketamine administration, which is commonly used for sedation during study examinations, has been associated with additional perturbations of muscle (CK or AST) and hepatic enzyme activities (ALT; Bennett et al. 1992). Care should be taken to avoid collecting blood for clinical pathology analyses in close proximity following these activities. Ideally, blood collection should occur prior to potential stressor or following sufficient amount of time for acclimatization (e.g., 2 days to 5 days).
Route of Administration
Intravenous (IV) administration of test compounds is a common dosing route utilized in routine toxicology studies in a range of laboratory animal species (nonhuman primate [NHP], dog, and rat). Several study-related procedures associated with IV dose administration have the potential to influence clinical pathology results including the use of indwelling or temporary catheters, restraint devices (e.g., slings), and/or concurrent IV fluid administration. Clinical pathology end points related to inflammation (e.g., acute phase proteins and cytokines), leukocyte kinetics, and/or minor muscle/tissue injury have the most potential to be impacted.
Similar to routine phlebotomy, a certain degree of tissue trauma at the infusion/injection site is inherent to IV dosing. Acute bolus dosing often utilizes a single needle stick in which the degree of tissue damage is minimal whereas indwelling or even temporary catheters are more likely to induce more pronounced changes among clinical pathology variables. Some of the more common tissue injury changes associated with IV dosing include mild elevations in AST, ALT, LDH, and/or CK activities attributable to mild localized skeletal muscle injury (Sheehan, Patten, and Mowat 2014). Prolonged animal restraint in studies with longer duration dose administration (2 hr to 24 hr) will also contribute to the effects on musculoskeletal markers but has also been shown to increase corticosterone concentrations in rats (Sheehan, Patten, and Mowat 2014). Indwelling or temporary catheters and IV dosing into the tails of rodents are often associated with a variety of inflammatory signals when compared to orally dosed animals. These effects may include mild to moderate increases in neutrophil count, globulin, acute phase reactants (e.g., C-reactive protein, serum amyloid A), and fibrinogen concentrations and/or reduced albumin concentration that are attributable to mild local inflammatory changes at the infusion site (Sheehan, Patten, and Mowat 2014). Transient increases in a variety of proinflammatory cytokines (e.g., interleukin-6) are also common following (2-hr to 6-hr postdose) acute bolus dosing with a single needle stick or with indwelling catheters (Sakai et al. 2016). Inflammatory effects may be further exacerbated by local extravasation of test material into the surrounding tissue causing additional tissue injury.
Another consideration related to effects on clinical pathology variables observed with IV dosing includes the potential to induce dilutional effects. Infusion studies often use larger dose volumes (up to 10 ml/kg/hr or more) or concurrent IV fluid administration (e.g., saline), which can have an acute dilutional effect on several routine concentration-based clinical pathology end points (e.g., electrolytes, total protein, and urea nitrogen concentrations) driving them lower. This phenomenon is most pronounced when samples for clinical pathology testing are drawn during, or shortly following (within 2 hr) the infusion procedure, or when samples for clinical pathology testing are drawn directly from the infusion catheter. A dilutional effect is most easily recognized when a number of individual, otherwise unrelated variables are decreased concurrently in all groups. Variables that are most commonly impacted by acute dilution include cell counts on hematology, electrolyte, protein, and urea nitrogen concentrations. Dilutional impacts on coagulation times can also be seen, as coagulation factors are diluted out resulting in prolongations in measured times (e.g., activated partial thromboplastin time [APTT], prothrombin time [PT]); this phenomenon is sometimes referred to as dilutional coagulopathy (Fries et al. 2005).
Intramuscular (IM) and subcutaneous (SQ) routes of administration are frequently used in vaccine studies and other small and large molecule test compounds. Single or repeated dose administration using IM or SQ routes are routinely associated with mild to moderate increases in the activities of muscle enzymes such as AST, CK, and/or ALT. In NHPs, IM ketamine is commonly used for chemical restraint during routine in-study procedures (e.g., physical and ophthalmic examinations). In addition to the local tissue trauma due to IM injection already described, ketamine has been associated with additional perturbations of muscle (CK or AST) and hepatic enzyme activities (ALT) through direct mechanisms (Bennett et al. 1992). Decreases in sodium, chloride, calcium, potassium, and glucose concentrations, and red cell mass values have also been reported with repeated IM ketamine administration (Bennett et al. 1992). These factors emphasize the importance of being familiar with all in-study procedures in order to accurately characterize fluctuations among these end points.
Study Length and Timing of Blood Draws
Factors related to study length also have the potential to influence clinical pathology data and interpretation of that data. Acute studies generally include any study ≤ 7 days in duration. Acute studies may involve a single or repeat dose administration and are often used for the purpose of dose range finding or broad hazard identification. Due to their short duration, acute studies often include a large number of blood draws in a relative short span of time making the potential for blood loss–related influences to be increased, particularly in large animal studies where serial blood sampling for toxicokinetic analysis are performed on main study animals. It is not uncommon for cumulative decreases in red cell mass to reach ≥5% to 15% that are attributable to study-related collections alone. In contrast, longer duration studies (i.e., >28 days) typically do not suffer from the acute blood losses seen in acute studies, but they may be more heavily influenced by age-related effects associated with growth and maturation (e.g., progressive increases in red cell mass, decreasing ALP activity). Given the great potential for age-related effects, it is often advisable to compare changes to controls in long duration large animal studies (vs. the traditional comparison to baseline) for end points with known age or procedure-related influences (e.g., red cell mass, ALP activity, urea nitrogen, and creatinine concentrations).
The timing of blood collection for clinical pathology testing in toxicology studies can dramatically impact the utility, value, and results of the clinical pathology data generated. Improper blood collection strategies can result in missing the positive signal window for a given end point, potentially leading to false negative test results. Factors to consider when designing collection schemes during the protocol generation phase include frequency of dose administration (single vs. repeat), dose route, known pharmacokinetic and pathokinetic profiles, kinetics of measured end points, analyte half-life, and relation of blood collection to other study activities. Given the nature of multiple interrelated factors to consider, one must often design specific blood collection time points to capture specific injury or pharmacodynamic signals of interest. For example, positive cytokine signals following IV administration of a pro-inflammatory compound are acute and generally short -lived (2-hr to 12-hr postdose) relative to a slow-acting oral hepatotoxin which may not illicit positive liver enzyme signals for several days to weeks following repeated dosing. Decreases in lymphocyte counts following administration of a known lymphotoxic compound may not be apparent for 7 days to 14 days or longer depending on the specific kinetics of the test compound. Performing clinical pathology testing on day 14 of a single-dose 14-day infusion study is less likely to yield positive signals, as compared to performing blood collections for clinical pathology testing on day 3 of the same study. These examples emphasize the importance of informed and strategic blood collection design for clinical pathology testing.
Surgery Studies
Studies involving surgical manipulation of the test system (e.g., stent placement and wound healing), or surgical administration of the test article (e.g., intracardiac or intrathecal stem cells, intrahepatic antineoplastic) inherently have great potential to impact clinical pathology test results. These studies are usually customized or unconventional in design, routinely utilize anesthetics, fluid support, and/or anticoagulative agents, and are not generally designed to prioritize clinical pathology end points. Signals associated with tissue injury and inflammation following surgical manipulation or dose administration are common and may include increases in neutrophil and lymphocyte counts, increases in acute phase reactants (e.g., fibrinogen concentration, C-reactive protein, etc.) or cytokines, and increases in skeletal muscle injury markers (e.g., ALT, AST, and CK). The duration, persistence, and severity of these signals will vary with the extent of the surgical procedures but may range from mild to severe and can persist for up to 10 days to 14 days. The effects of perioperative Coumadin or heparin therapy will (often intentionally) prolong coagulation times (e.g., APTT or PT) for up to several days following treatment. Additionally, fluid administration may falsely decrease a variety of concentration-based clinical pathology end points (e.g., cell counts and electrolytes) or may prolong coagulation times (i.e., dilutional coagulopathy), which are considerations when drawing blood samples for clinical pathology testing in close proximity to IV fluid support. Collectively, these factors will sometimes nearly preclude any utility of clinical pathology evaluations on surgery studies and emphasize the importance of protocol familiarization and sound study design.
Compound Class: Small versus Large Molecules
Small molecule test articles generally exhibit dose-dependent patterns among clinical pathology effects making interpretation and effect identification more predictable relative to large molecules. An exception may be when small molecules have a receptor-mediated mechanism where a “maximal” effect may be reached after receptor saturation (e.g., sodium–glucose cotransporter inhibitors for treatment of diabetes). Sodium–glucose transporter-2 inhibitors block glucose reuptake from kidney resulting in glucosuria with a modest lowering of serum glucose concentration, usually with a concurrent solute diuresis, and increased risk of urinary tract infection (Johnsson et al. 2013). In a majority of cases, the effects of small molecules tend to be similar, or more homogenous, within a group and between genders granted there are no differences in exposure or sensitivity. Small molecule effects can be progressive if accumulation of drug occurs with repeated dosing resulting in higher exposure over time, or if the target organ (e.g., liver, kidney, and gastrointestinal [GI]) becomes more vulnerable with repeated injury. In some cases, adaptation may occur resulting in less pronounced toxicity with induction of hepatic p450 enzymes resulting in enhanced metabolism and excretion of test article.
Large molecule/biologics, on the other hand, often lack a clear dose–response, as they generally involve receptor-mediated mechanisms. In many cases, the most severe effects are often seen at the mid-dose where the proportion of test article and its target are in optimal proportions, similar to a prozone phenomenon. Biologics tend to have more individualistic response and sometimes can exhibit idiosyncratic reactions depending on the difference in sensitivity or genetic makeup of individual animals. The biologics, especially large molecules, when administered in heterogeneous species are perceived as foreign proteins and frequently elicit an immunogenic response resulting in production of antidrug antibodies (ADAs). These ADAs (neutralizing Abs) can bind with the drug resulting in lower exposure and pharmacologic effect of the drug (Leach et al. 2014). ADAs can also bind with the drug creating immune complexes, which, depending on the size and charge of complexes and saturation of the clearance mechanism, may deposit in various tissues causing inflammation and subsequent tissue damage (Type III hypersensitivity reaction (HSR; Rojko et al. 2014). These immune complexes are frequently deposited in blood vessels causing vascular injury but can affect multiple tissues including the skin, GI tract, or lungs. Glomerulonephritis is commonly observed with immune complex deposition, which in severe cases can cause renal injury (increased urea nitrogen and creatinine concentrations and proteinuria). Type III HSRs are a commonly identified postdose immune reaction associated with large molecules (Heyen et al. 2014; Leach et al. 2014). Since prior sensitization of the immune system is required for the production of ADAs and immune complexes, Type III HSRs usually only occur after repeated exposure (2 to 3 or more doses) and can be progressive in severity with continued dosing (Doessegger and Banholzer 2015). The incidence may be higher with SQ route (compared with IV) because it allows longer exposure time, thus increasing the possibility of ADA and immune complex formation. Depending on the outcome and extent of the injury, these immune reactions can be associated with variety of clinical pathology changes, including inflammatory effects (increased leukocyte counts, cytokines, complement fractions, acute phase proteins, etc.), thrombocytopenia, and decreased albumin and/or increased globulin concentrations. Indications of immune stimulation may be present among other end points (presence of ADA, lack of exposure/effect; Leach et al. 2014) to use as supportive evidence, as it may be necessary to differentiate between signals related to immune stimulation, and those related to the presence of test article (i.e., increased globulin concentration) if the test article is an immunoglobulin.
Acute postdose reactions following administration of a large molecule may also involve complement, or complement activation-related pseudoallergy (CARPA), or a true Type I HSR, which involves an IgE-mediated anaphylactic reaction (Leach et al. 2014). As opposed to Type I or III HSRs, these acute reactions will often occur following the first dose with progressive diminishing of severity following subsequent dosing (Doessegger and Banholzer 2015; Szebeni 2005). Pretreatment with antihistamine drugs is usually helpful in preventing IgE-mediated anaphylaxis but may not prevent reactions caused by other mechanisms.
Clinical pathology effects can play an important role in monitoring the pharmacologic effects, especially for the drugs that have clinical pathology-related pharmacodynamic end points. A few examples may be diabetes drugs (insulin and glucose monitoring), cell proliferation factors (such as erythropoietin, granulocytic-colony stimulating factors with monitoring of erythropoiesis and granulopoiesis, respectively), or test article with known class effects such as glucocorticoids, opioids, and so on. Since these pharmacologic effects are intended for patient populations, the exaggerated pharmacology can result in unintended or adverse effects in healthy animals, such as severe hypoglycemia with insulin administration or a hypercoagulable state/disseminated coagulation when administering activators of coagulation factors. Easily monitored clinical pathology end points may allow selection of optimal dose levels. However, some of these responses may not follow a simple dose curve making it difficult to predict the unexpected outcomes. Regardless, a close monitoring of such clinical pathology end points is usually performed in toxicity studies (often with multiple collections for clinical pathology end points of interest) to assess pharmacologic effects and their reversal.
Exposure data and understanding of the pharmacokinetic profile are critical for proper evaluation of clinical pathology data, as development of ADA can result in reduced exposure. In some cases, prolonged exposure is expected based on the class/mechanism of the test article (e.g., bisphophonates) or slow clearance due to nature of the test article or specialized formulations. However, predicted half-life (based on the experimental model) may not always be true for the given species/study emphasizing the importance of using exposure data from the same study in which one is interpreting clinical pathology data.
Vehicle Effects
In most cases and ideally, vehicles are formulated using inert materials that do not cause toxicity. However, in some cases, vehicle formulations will occasionally cause toxicity confounding the ability to distinguish between test article–related and vehicle effects. Some examples include pseudoallergic reactions sometimes caused by radiocontrast media, liposomal drugs (Doxil, Ambisome, and DaunoXome), and micellar solvents containing amphiphilic lipids (e.g., Cremophor EL). These agents activate classical and the alternative pathways, giving rise to C3a and C5a anaphylatoxins that trigger mast cells and basophils resulting in HSRs sometimes referred to as CARPA (Szebeni 2005; Surapaneni, Das, and Das 2012). In addition to occasional HSRs, lipid-containing vehicles can cause increased lipids or triglycerides when administered at high concentrations.
Some vehicles have relatively safe profiles at recommended doses but can exhibit toxicity at much higher doses. One such example is Transcutol, diethylene glycol monoethyl ether, a highly purified ethylene oxide derivative that can contain significant levels of ethylene glycol and diethylene glycol as impurities. It has been safely used via various routes at low levels (daily exposure level of 10 mg/kg/day); however, toxicity may be seen at higher dose levels >1,500 to 2,000 mg/kg/day including renal effects, severe electrolyte imbalance, and even death (Sullivan, Gad, and Julien 2014).
Adjuvants may be considered another vehicle component that play an important role in enhancing immunogenicity of the antigenic component of vaccines. These adjuvants, because of their immunostimulatory nature, may cause local inflammatory reactions at the injection site and elicit relatively minor increases in systemic markers of inflammation (including leukocyte counts, cytokines, and acute phase proteins). During vaccine evaluation, inflammatory changes of greater severity than adjuvant groups alone can be attributed to the test article (antigenic component).
Some spurious effects can also be created by vehicles. For example, IV infusion preparations containing dextrose solution can cause high glucose concentration due to contamination if care is not taken during sample collection to avoid contamination. Vehicles containing bromide will result in artifactually high chloride concentration because the ion selective electrodes used for analysis cannot differentiate between chloride and bromide. Due to interference of polyethylene glycol with coagulation tests, certain assays have been reported to produce artifactually prolonged APTT results (Aulbach et al. 2012).
Bad Ideas in Study Design (for Clinical Pathology)
Not all studies are specifically designed or intended to emphasize clinical pathology results. That said, there are several fairly common practices in toxicology studies that severely limit the value of clinical pathology testing. Some of these practices include drawing more blood from treated animals relative to controls for toxicokinetic analysis, dose holidays or changing dose levels mid-study, or adding new animals in a treated group during an ongoing study. In some cases, these approaches may allow continued assessment of toxicity, reversibility, or give some information on tolerability of a dose level; however, they often result in the lack of controls, or an appropriate time-matched comparator group, and will generally limit the conclusions one can render from clinical pathology data generated on toxicology studies.
Conclusion
Study design and conduct variables have great potential to impact clinical pathology test results during toxicology studies. It is critical to have a complete understanding of these factors during data interpretation for accurate hazard identification. Efforts should also be made during the study design phase to minimize the inclusion of study practices known to introduce sources of variability which may limit the utility of clinical pathology data generated on toxicology studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
