Abstract
Preanalytical variables can have significant impacts on clinical pathology parameters evaluated during the conduct of a nonclinical safety or toxicity study. These preanalytical variables can be controlled by careful attention to factors such as animal dietary status (diet composition, fasted, and fed state), restraint and anesthesia, intercurrent procedures, timing of clinical pathology collections, and proficiency of animal technicians. The impact of preanalytical variables on test results can be significant enough to result in difficult interpretations and/or regulatory questions or can obfuscate the effects of a test article. Control of preanalytical variables starts with knowledge of what processes and procedures impact test results. Minimizing these effects improves the quality of results and maximizes the value of the study.
With the advent of precise and accurate modern analytical instruments and sophisticated laboratory information systems, the impact of preanalytical variables on laboratory results has become increasingly important. Although there are no data on the impact of preanalytical variability on laboratory data generated during toxicity studies, it is useful to examine statistics from human clinical laboratories as a reference point.
In human laboratory medicine, approximately 1% of test results are estimated to be either erroneous or impacted by preanalytical events. Of this 1%, preanalytical events introducing variability are responsible for the largest component (∼35% to 75%), followed by postanalytical errors (∼15% to 30%) with only a small number of analytical errors (Lippi et al. 2006; Simundic and Lippi 2012; Cemin and Daves 2015; Green 2013). In this era of sophisticated analytical laboratory instruments, analytical errors are mostly due to unidentified instrument issues or manually entered data (e.g., color of urine, manual differential), while examples of postanalytical error are generally due to editing or transmission errors. Preanalytical conditions contribute the most to variability of laboratory results; these include patient preparation, phlebotomy, and sample handling. For humans, patient preparation is problematic, as there is reliance on compliance of the subject. In addition, other patient-related variables such as diet, activity, exercise, alcohol intake, concurrent medications, and previous phlebotomies are uncontrolled. Several aspects of phlebotomy can contribute to errors and/or variability in human clinical laboratories. These include misidentification, insufficient volume, contamination, and venous stasis due to tourniquet, type of collection device, and mixing. The most common preanalytical issues in human laboratory medicine are clotting of hematology specimens (primarily pediatric tubes) and hemolysis (80% of clinical chemistry issues). Postcollection sample handling (e.g., delay to processing, ambient temperature, and inversion of tubes) is also problematic for human specimens. Depending on the size of the medical facility, some specimens may be delivered immediately to the laboratory, while others remain on the wards for a significant period of time.
The author’s experience suggests that the error rate for clinical pathology data collected in Good Laboratory Practice (GLP) laboratories during toxicology studies is low for the same reasons as for human clinical laboratories. Modern instrumentation with such capabilities as automated sample and reagent identification through barcoding and automated differentials is interfaced with laboratory information management systems with rigorous audit trails. These systems assure that samples are properly identified in the laboratory and that data are controlled and reported accurately. In laboratories processing samples from toxicity studies, personnel follow standard operating procedures that comply with good laboratory practices, which assure that each data point can be traced from its analysis to the corresponding report.
In terms of preanalytical errors or variability, many issues that plague human laboratory medicine are not present in nonclinical studies. For example, positive animal identification is performed using microchips or other mechanisms. Animals are fed the same diet. Collection devices, sample handling and transportation time and temperature, and centrifugation conditions are kept constant for a set of animals on a study. In the author’s experience, the primary sample quality issues affecting results in nonclinical studies are similar to those in human medicine: clotting of hematology specimens and hemolysis of clinical chemistry specimens. The advantage to controlling preanalytical errors and variability in nonclinical safety and toxicity studies is that there will be greater consistency of data, and changes that are present will be more likely to be truly related to the test article.
For animals, the biggest preanalytical confounders are related to husbandry and timing, intercurrent procedures, restraint and anesthesia, and venipuncture and processing. Understanding the procedures resulting in preanalytical variability is important so that steps can be taken to control and minimize their impact. This review will not cover preanalytical considerations due to species selection, study design, and protocol, as they are covered separately in this issue (Aulbach et al. in press).
Effects of Husbandry and Timing
Nonclinical safety and toxicity studies are conducted under controlled conditions with all animals similarly housed and fed. Variability may be introduced when animals are moved to a different cage or when animals are fasted. Most reports have shown little or no effect of caging on routine parameters. For example, there is no change in urine corticosterone (measured every 6 hr) or the corticosterone diurnal rhythm in rats moved from conventional polycarbonate cages to metabolic caging for 3 days (Eriksson et al. 2004). The type of diet fed to an animal can have effects on several parameters. Although the type of diet is generally constant within a toxicity study, fasting may not be consistent across time points (e.g., in life vs. terminal samples), or some groups may not receive nourishment due to effects of the test article (vomiting, anorexia). Aside from effects on glucose and other parameters related to energy metabolism, fasting status can also impact other results. For example, in a preliminary study conducted at Amgen, fed rats had higher concentrations of the novel muscle injury markers creatine kinase (CK) protein and parvalbumin, compared to overnight fasted rats.
The time of day for clinical pathology sample collection is a critical variable, in terms of both absolute time of day and timing relative to other procedures. It is well known that several clinical pathology parameters, most notably leukocytes and hormones, show a distinct diurnal pattern (Dhabhar et al. 1994). Other parameters may also show a diurnal rhythm, including triglycerides in mice (Pan et al. 2010). In general, the effects of diurnal rhythms on laboratory results are more evident in rodents compared to larger animals.
When possible, procedures such as dosing, intramuscular injection, restraint for other procedures, electrocardiograms, physical examinations, and other room disturbances should be conducted after the collection of clinical pathology samples. Some procedures must be conducted prior to clinical pathology collections, so for these, it’s important to understand the potential effects and mitigate their impact. Even transportation of cages from animal rooms to treatment areas has effects on laboratory results. For instance, moving mice in their cages on a rolling rack out of their room and onto an elevator for about 12 min caused a 12% and 15% decrease in mean thymus weights at 12 hr and 24 hr after transport, respectively, and a 36% decrease in lymphocytes at 4 hr after transport (Drozdowicz et al. 1990). Similarly, mice that went for a pushcart ride had a 30% to 40% increase in serum glucose compared to mice that were habituated to handling (Tabata, Kitamura, and Nagamatsu 1998).
The advanced cognition of nonhuman primates can have preanalytical impacts on clinical pathology results (Capitanio, Mendoza, and McChesney 1996). A study measuring lymphocytes and cortisol in rhesus monkeys was confounded by the fact that the monkeys on one side of the room could see the technicians preparing for blood collection procedures in the anteroom, while those on the other side of the room were blocked from this view. The result of this arrangement was that monkeys that had more time to anticipate blood collection (those viewing the anteroom) had higher cortisol and lower lymphocytes compared to those who were unaware of the impending collection.
Effects of Handling, Restraint, and Anesthesia
The effects of handling, restraint, and anesthesia can substantially affect clinical pathology results, especially in small animals and nonhuman primates. Standardization of techniques handling and restraint techniques is very important in collecting blood for clinical pathology measurements. Swaim evaluated the effect of holding mice by the tail compared to by the body while picking up for body weights (Swaim, Taylor, and Jersey 1985). Alanine aminotransferase (ALT) measured an hour after body weights was 3.7-fold higher in mice picked up by the body compared to the tail. Similarly, CK, but not ALT or aspartate aminotransferase (AST), was higher in rats handled under their front legs compared to the base of the tail (Yerroum, Braconnier, and Chariot 1999).
Longer periods of restraint in rodents (e.g., those that might be experienced during inhalation or infusion studies) can impact both hematology and clinical chemistry results (Pauluhn 2004). The effect of longer term restraint on leukocyte parameters are those associated with stress, such as decreased lymphocytes and eosinophils and increased neutrophils (Dhabhar et al. 1994). Serum enzymes such as AST, ALT, and CK can also be affected by restraint; these changes can be blocked by β-adrenergic blockers (Sanchez et al. 2002; Arakawa et al. 1997).
There are few effects of handling and restraint observed in dogs when handled in routine settings. No difference was observed in several hormonal parameters sensitive to stress regardless of whether blood was collected through a catheter or by venipuncture, or if animals were restrained in their cage or removed to an anteroom (Knol et al. 1992). There were no effects on the values for ultrasensitive cardiac troponin I (cTnI) in dogs administered vehicle repeatedly for 26 weeks (Casartelli et al. 2011).
The changes with handling and restraint in nonhuman primates are fairly minimal. Ways to decrease impact of handling and restraint in nonhuman primates include using positive reinforcement, voluntary blood collections, and paired housing. The effect of handling, restraint, and other procedures is perhaps best observed in nonhuman primates by the typical transient increases in AST, ALT, and sometimes neutrophils on the first few days of a study, a time when there are often many procedures being conducted (Figures 1 and 2). Other parameters may also be affected by procedures. For example, oral administration of vehicle does not affect the values for ultrasensitive cTnI in control cynomolgus monkeys, but procedures associated with intravenous dosing result in transient increases above baseline at 3 hr or less (Schultze et al. 2015; Reagan et al. 2016).
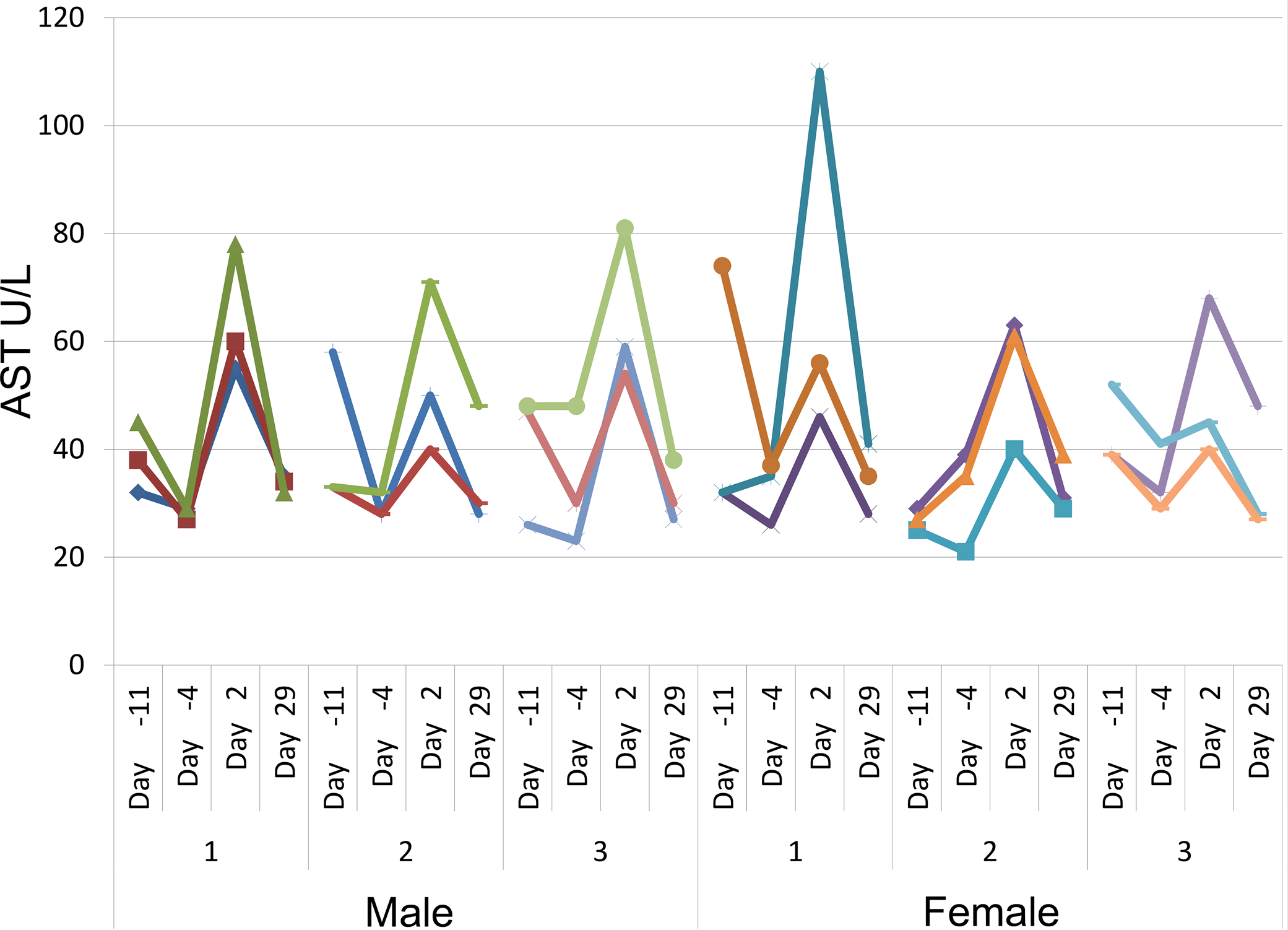
Serum aspartate aminotransferase (AST) activities (U/L) in cynomolgus monkeys during a 1-month toxicity study with weekly subcutaneous dosing. Each line represents data from a single animal. Each set of lines represents data from a single sex and group. AST activities were generally higher on day −11 (first prestudy time point) compared to day −4 (second prestudy time point). All animals underwent several procedures on day 1 prior to blood collection on day 2. The procedures resulted in minimal to mild increases in AST activities on day 2 in animals regardless of test article (group 1: vehicle; groups 2 to 3: low and high dose). AST activities at the end of study (day 29) were comparable to day −4 values.
Habituation has been shown to decrease variability of clinical pathology results. Animals that are habituated to procedures have lower glucose, CK, and fewer leukocyte changes than those that are exposed to a novel situation (Marquez, Nadal, and Armario 2004). For this reason, in large animals, clinical pathology values determined from the first prestudy collection are often more variable than those collected at the second prestudy time point, and there is less variability in parameters at later time points during the study (Figure 2). Habituation is most effective when procedures are predictable, rather than variable (Marin, Cruz, and Planeta 2007).
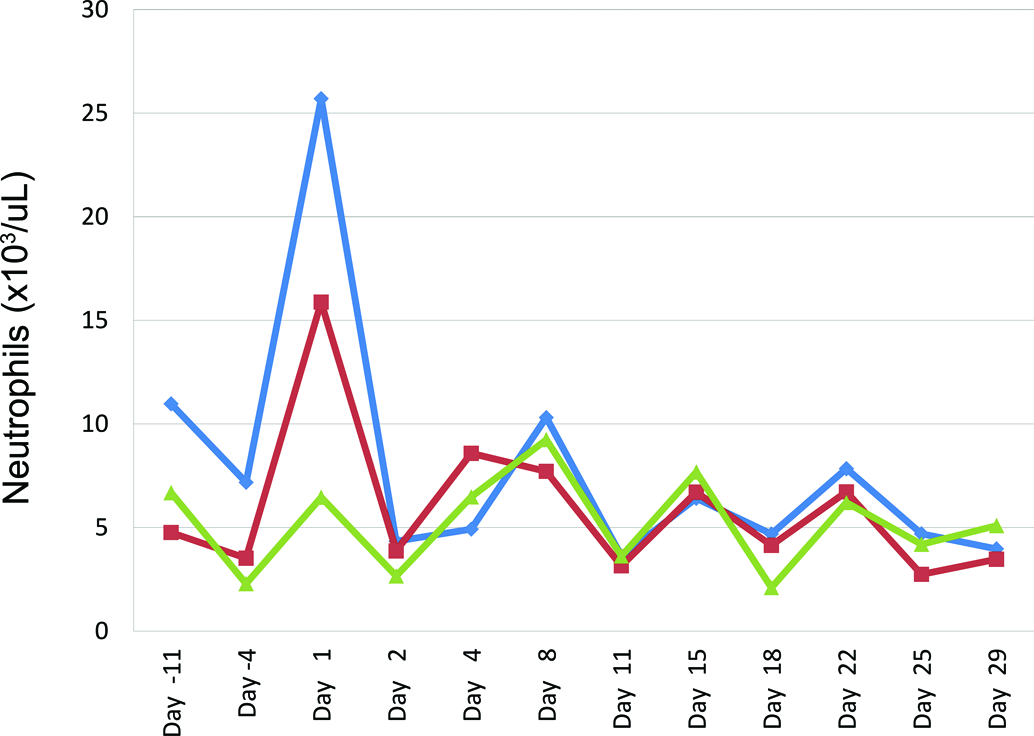
Neutrophil counts (×10e3/ul) from control cynomolgus monkeys on a 1-month study with intermittent dosing (days 1, 8, 15, and 22). Each line represents data from a single animal. Neutrophil counts on day 1 were 3- to 4.5-fold higher than those on day −4. The higher values on day 1 were considered to be secondary to the stress and excitement of the novel procedures conducted at the beginning of the study.
Under certain conditions, especially in rodents and nonhuman primates, animals are sedated either for blood collection or for other procedures adjacent to clinical pathology procedures. There are numerous papers comparing the effects of various anesthetics on clinical pathology results from samples collected from rodents (Traslavina et al. 2010; Deckardt et al. 2007; Walter 1999). Although these studies differ in terms of strain of rodent and site of collection, isoflurane effects on clinical pathology parameters are generally of a lower magnitude than those observed with carbon dioxide or barbiturates. However, isoflurane has been shown to have dramatic effects on cytokine measurements, especially in rodent models of inflammation. Cytokine concentrations are substantially lower (as low as 50% of those collected under carbon dioxide) when isoflurane is administered before or after endotoxin challenge or at necropsy in an animal with induced inflammation (Lawrance et al. 2009; Flondor et al. 2008; Hofstetter et al. 2005).
Nonhuman primates are often sedated using intramuscular ketamine injection for various study procedures. Only minor changes in clinical pathology have been reported in nonhuman primates while under ketamine anesthesia (Lugo-Roman et al. 2010; Kim et al. 2005; Bennett et al. 1992; Lynch et al. 2012). However, due to its muscle irritant properties, ketamine causes increases in parameters indicative of skeletal muscle injury (e.g., AST, ALT, and CK) starting by 24 hr and continuing up to 5 days after administration (Davy et al. 1987). For this reason, it is important to conduct procedures involving ketamine anesthesia more than 5 days prior to clinical pathology collections.
Effects of Venipuncture and Sample Handling
Preanalytical variables introduced at the time of blood collection include site, volume, method, and order of collection; skill of phlebotomist; and processing steps.
The effect of site of blood collection on clinical pathology parameters is greater in rodents compared to larger animals, and is greater on hematology parameters and cytokines compared to clinical chemistry parameters. In rodents, higher red blood cell (RBC) mass parameters (RBC count, hemoglobin, and hematocrit), leukocyte counts, and platelet counts are observed in blood collected from more peripheral vessels compared to more central vessels or the heart. For example, in mice, RBC, leukocyte, and platelet counts from cardiac collections were 80%, 30%, and 40%, respectively, of counts measured from blood collected from the tail vein (Doeing, Borowicz, and Crockett 2003; Nemzek et al. 2001). Higher cell counts in peripheral vessels are also observed in larger animals, but the differences are much smaller (Lynch et al. 2012). In a preliminary report that didn’t control for collection order or previous procedures, cytokines have been reported to be higher in blood collected from central vessels/heart compared to peripheral sites (Mella et al. 2014). In this study, the concentrations of cytokines (Interleukin-6 [IL-6], Macrophage Inflammatory Protein 2 [MIP-2], Chemokine [C-X-C motif] Ligand 1 [CXCL-1], Interleukin-1 Receptor Antagonist [IL-1RA], Interleukin-10 [IL-10], and Interleukin 1 beta [IL-1b]) were 1.5- to 3.0-fold higher in cardiac blood compared to facial vein blood.
The site of collection may also impact subsequent results. For control rats on a small investigative study, repeated collections from the tail vein resulted in increased muscle injury markers and corticosterone in subsequent samples, while collections from the jugular vein (anesthetized or unanesthetized) did not. Skeletal muscle injury parameters measured in rat blood from jugular collections were less variable, but also less sensitive, than those from tail vein collections (Miwa et al. 2015).
The volume of collection also impacts subsequent clinical pathology results. The primary effects are observed for RBC mass parameters (decreased), reticulocyte counts (increased), and RBC indices [increased mean cell volume (MCV) and red cell distribution width (RDW), decreased mean cell hemoglobin concentration]. It is important to note that Institutional Animal Care and Use Committee limits on volume of blood collections are limits rather than recommendations. Collecting the maximum amount of blood allowed will result in changes in clinical pathology that may impact the ability to detect toxicity or can impact the ability of the animal to adapt to the effect of a test article.
Controlling both the order and time of blood collection is important to obtaining interpretable results. It is very important to collect samples across group rather than by group. A random or stratified (e.g., round robin, 1 from each group, then a second from each group) order of collection limits bias in results due to order of collection (Hall and Everds 2008). Occasionally, a study design requires that animals are bled by group. In these cases, it is best to order groups so that effects on the control and high-dose groups are minimized. A collection scheme that works well to avoid bias is collecting samples by groups in this order: mid-dose, control, high-, and low-dose groups.
The skill of the phlebotomist is crucial to collection of quality samples and generation of high-quality results. Less skilled phlebotomists are particularly challenged by unthrifty or dehydrated animals, which can also be a challenge for even skilled phlebotomists. Difficult phlebotomy due to skill level or animal health can result in underfilled blood collection tubes, clots, hemolysis, or platelet clumps (rats and mice).
The type and brand of blood collection tubes should be consistent for a given analyte. Although technical documentation for assays often indicates that a variety of matrices can be used for analysis (e.g., serum, heparinized plasma), there can be small differences introduced by each matrix. Even tubes from different manufacturers that contain the same anticoagulant (e.g., pediatric microtainers for hematology) may result in slight biases in laboratory tests. Some gels in serum separator tubes have been shown to affect selected analytes in human serum (Schouwers et al. 2012; Bowen et al. 2007; Shi, van Rossum, and Bowen 2012). For these reasons, it is best to identify appropriate blood tubes and use those types of tubes consistently.
The processes involved in collection of blood, placing blood in a collection tube, and transporting blood to the lab can result in preanalytical issues. Best practices involve regular feedback to the phlebotomy team to improve performance and thus clinical pathology data. Underfilling of blood tubes can result in errors in hematology and coagulation parameters because of an excess of anticoagulant compared to the amount of blood. Excess ethylenediaminetetraacetic acid (EDTA) in hematology tubes can result in alterations in complete blood count results, particularly for RBC parameters (Anonymous 1993). Excessive EDTA may result in shrinkage of RBCs as well as morphologic changes such as echinocytes (Anonymous 1993). The dipotassium salt (K2) of EDTA is recommended over the tripotassium salt (K3) or the disodium salt due to fewer artifacts. EDTA also affects the morphology of bone marrow cells and may result in alterations that can be misinterpreted as dysplastic change (Lee et al. 2013). Underfilling of coagulation tubes results in excess citrate over the correct 1:9 ratio of anticoagulant to blood and prolongs coagulation times (Johnstone 1993; Peterson and Gottfried 1982).
Hematology samples that contain clots are invalid and cannot be evaluated in the laboratory. Clots and platelet clumps (particularly for rodents) in hematology specimens are prevented by agitating tubes sufficiently to mix blood with EDTA. For the pediatric tubes commonly used for collection of rodent blood, agitation is especially crucial because the surface tension in the small amount of blood restricts the ability to mix thoroughly. Although samples to replace clotted samples can sometimes be collected from large animals, doing so introduces variability, because the replacement collection occurs at a different time and under different circumstances (e.g., after feeding) compared to the original collection.
Platelet clumps are commonly observed in samples from rodents, even in nonclotted samples. On the Advia® hematology analyzers, platelet clumps can be observed as a streak heading from the origin diagonally upward to the right on the peroxidase channel cytogram. Microscopically, platelet clumps are best observed at the feathered edge of the blood smear. The number of platelet clumps at the feathered edge is not necessarily concordant with the “clump” count on the Advia hematology analyzer. For rodents, the Advia may count platelet clumps as eosinophils (Everds 2007). For this reason, any eosinophil count over 4% should be evaluated for potential interference by platelet clumps. In addition, a blood smear review should be conducted when platelet counts are unexpectedly low to determine if platelet clumps contributed to the low count.
Hemolysis is a common preanalytical confounder for clinical pathology tests. When hemolysis is observed as discoloration of serum, it is most commonly an ex vivo effect caused by phlebotomy procedures, rather than a result of the test article causing intravascular hemolysis. Hemolysis caused by procedures can be minimized by taking a number of steps during collection and handling. Shear forces are minimized by collecting blood smoothly using the largest bore needle appropriate for the species and vessel, and by gently placing blood into the collection tube. Blood tubes should not have contact with wet ice. In addition, some species (e.g., pig) are more sensitive to hemolysis compared to others (Thorne 2010). Although hemolysis can affect hematology results (especially RBC indices), its effects are more prominently observed for clinical chemistry parameters. Interference by hemolysis with clinical chemistry measurements can be method independent or method dependent. Hemolysis causes method-independent interference with the measurement of analytes normally found within RBCs (e.g., AST in all species, and K+ in most species except dog). Hemolysis can also cause interference that is specific to the methodology used for analysis (e.g., colorimetric assays).
Once collected, blood tubes should be taken to the laboratory for processing in a timely manner. In humans, it has been shown that inversion of lithium heparin tubes causes increased variability in commonly measured analytes such as AST and potassium (Lippi et al. 2014). How specimens are stored after collection and prior to analysis can impact values. For hematology, best practices are to prepare and fix or stain blood smears promptly. Hematology specimens should be analyzed on the hematology instrument within 6 hr if possible. Under some circumstances, whole blood cannot be analyzed on the day of collection. In these cases, blood should be refrigerated overnight until analysis. Coagulation and clinical chemistry specimens should be analyzed the same day or frozen for future analysis. Stability for most parameters is best at colder temperatures (in preference order: −80°C > −20°C > 4°C > room temperature). Multiple freeze thaw cycles should be avoided, although many parameters remain stable throughout several cycles.
Conclusion
Controlling preanalytical factors decreases the variability of clinical pathology data but requires careful attention to procedures from animal handling through sample analysis. Decreased variability in clinical pathology parameters results in increased ability to determine test article–related changes and decreases the time and effort evaluating and understanding spurious changes caused by preanalytical issues.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
