Abstract
Evaluation of the female reproductive system in a general toxicity setting can be challenging for the toxicologic pathologist due to the cyclic nature of the estrous and menstrual cycles, timing of puberty and reproductive senescence, and species differences. Age in particular can have a significant impact on the histologic appearance of the female reproductive system and create challenges when trying to distinguish test article–related findings from normal developmental or senescent changes. This review describes the key physiologic and histologic features of immaturity, the transition through puberty, sexual maturity, and reproductive senescence in the female reproductive system, with an emphasis on practical applications for the toxicologic pathologist, and includes recommendations for distinguishing and documenting these developmental periods. Rats and cynomolgus monkeys are used as examples throughout with correlations to clinically observed end points to better aid the toxicologic pathologist in understanding how age may impact study interpretation.
Evaluation of the female reproductive system in a general toxicology setting can be difficult and at times frustrating for the toxicologic pathologist. The cyclic nature of the estrous and menstrual cycles drives the marked variability in the size, shape, and appearance of the reproductive organs in normal animals. The impact and timing of puberty and reproductive senescence further complicate the picture. In addition, the reproductive strategies of our commonly used laboratory animals are wildly different. As a result, the toxicologic pathologist is required to have a thorough understanding of the normal anatomy, physiology, and histology of each of these species prior to deciding what is abnormal. The normal estrous and menstrual cycles have been reviewed extensively (Long and Evans 1922; Bartelmez 1951; Koering 1969; Yuan and Carlson 1985; Brenner and Slayden 1994; Poonia et al. 2006; Watanabe et al. 2006; Li and Davis 2007; Buse, Zöller, and van Esch 2008; van Esch et al. 2008; Westwood 2008; Yoshida et al. 2009; Vidal et al. 2013; Dixon et al. 2014; Sato, Nasu, and Tsuchitani 2016), but in the toxicologic pathology literature, less attention has been given to the impact of age on the interpretation of potential toxicologic findings in the female reproductive system (Picut et al. 2014, 2015; Shirai, Houle, and Mirsky 2015). While each of the commonly used nonclinical species presents its own unique set of potential issues related to age, in the author’s experience the rat and nonhuman primate (NHP) are the most likely to be encountered. Rats are the most commonly used rodent in nonclinical toxicity testing and a wide range of ages will be encountered from young animals in juvenile toxicity testing to aged animals in carcinogenicity studies. With the increasing numbers of biopharmaceuticals and the relatively recent update to biopharmaceutical regulatory guidance documents (International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use [ICH] 2011), toxicologic pathologists are being asked to evaluate the reproductive system from mature NHPs as a surrogate for more typical fertility-based reproductive toxicity studies as well as evaluate very young animals in enhanced pre- and postnatal development studies. As a result, the rat and NHP (cynomolgus monkey unless otherwise stated) will be the focus of this review.
Definitions and Background
In order to understand how age can impact nonclinical toxicology results, a common definition of age and developmental periods needs to be established. A commonly used age classification scheme is defined in ICH E11 Clinical Investigation of Medicinal Products in the Pediatric Population (ICH 2000) and serves as a helpful tool to understand basic periods of development (Table 1). Various references have extrapolated this scheme to include similar developmental periods in the commonly encountered nonclinical toxicology species (Beck et al. 2006; Anderson et al. 2009; Barrow, Barbellion, and Stadler 2011). While this serves as a useful tool in designing juvenile toxicity studies, it can create confusion and challenges when dealing with the reproductive system. This is in a large part due to the fact that the onset of reproductive cycling and development of full reproductive potential occur over a relatively long period of time and can span several of the categories as defined in ICH E11. For example, a young girl or NHP may show the first signs of menstrual bleeding at the end of the “child” period but may not have normal cycles or reach full sexual maturity until the end of the “adolescent” period or beginning of the “adult” period. As a result, these terms for defining developmental periods of the reproductive system are not typically used and when used may erroneously confer specific ages rather than reproductive developmental end points.
Developmental Stages in Humans.

aDefinition of age varies with region.
When categorizing or documenting developmental stages of the reproductive system, toxicologic pathologists most often use the terms immature and mature, with some also including pubertal or peripubertal, to describe animals in transition to maturity (Halpern et al. 2016). A forth category of senescent/postmenopausal is an important aspect of the life span of the female reproductive system but is not commonly documented in nonclinical toxicity testing. In the author’s experience, there seems to be considerable confusion, in the literature and among pathologists, as to how to define and differentiate “puberty” and “sexually mature.” The dictionary definitions are as follows:
Review of established dictionary definitions only furthers the confusion, and it is difficult to separate where puberty ends and sexual maturity begins. This is not surprising with a complex biologic process that occurs over an extended period of time, but toxicologists and pathologists are often asked to draw a line in this proverbial sand in order to interpret nonclinical toxicity studies. As a result, it is common to see the use of specific and easily defined milestones or landmarks of sexual development. These are most often readily observable clinical signs, which in the rat is vaginal opening and in the NHP is menarche. In the rat, vaginal opening occurs at approximately 32 to 34 days of age (Lewis et al. 2002) and is a common end point in reproductive and juvenile toxicity testing. Vaginal opening serves as a marker for the onset of puberty and in the rat it is generally linked to the onset of estrous cyclicity and first ovulation (Figure 1). This is not the case in the mouse, which shows strain-dependent differences in the timing of estrous cyclicity following vaginal opening (Nelson et al. 1990). In the NHP, the disconnect between the externally observed landmark of sexual development, menarche, and the onset of cyclicity is even more dramatic with menarche occurring at approximately 2 to 3 years of age (Weinbauer et al. 2008), while the first ovulation and regular cyclicity often occur many months later. Despite these challenges, many of these features can be observed histologically and in an attempt to better characterize the stages that a toxicologic pathologist might observe in practice, each of these developmental periods will be reviewed based on the histologic features with representative examples and correlations to clinically observed end points where applicable.
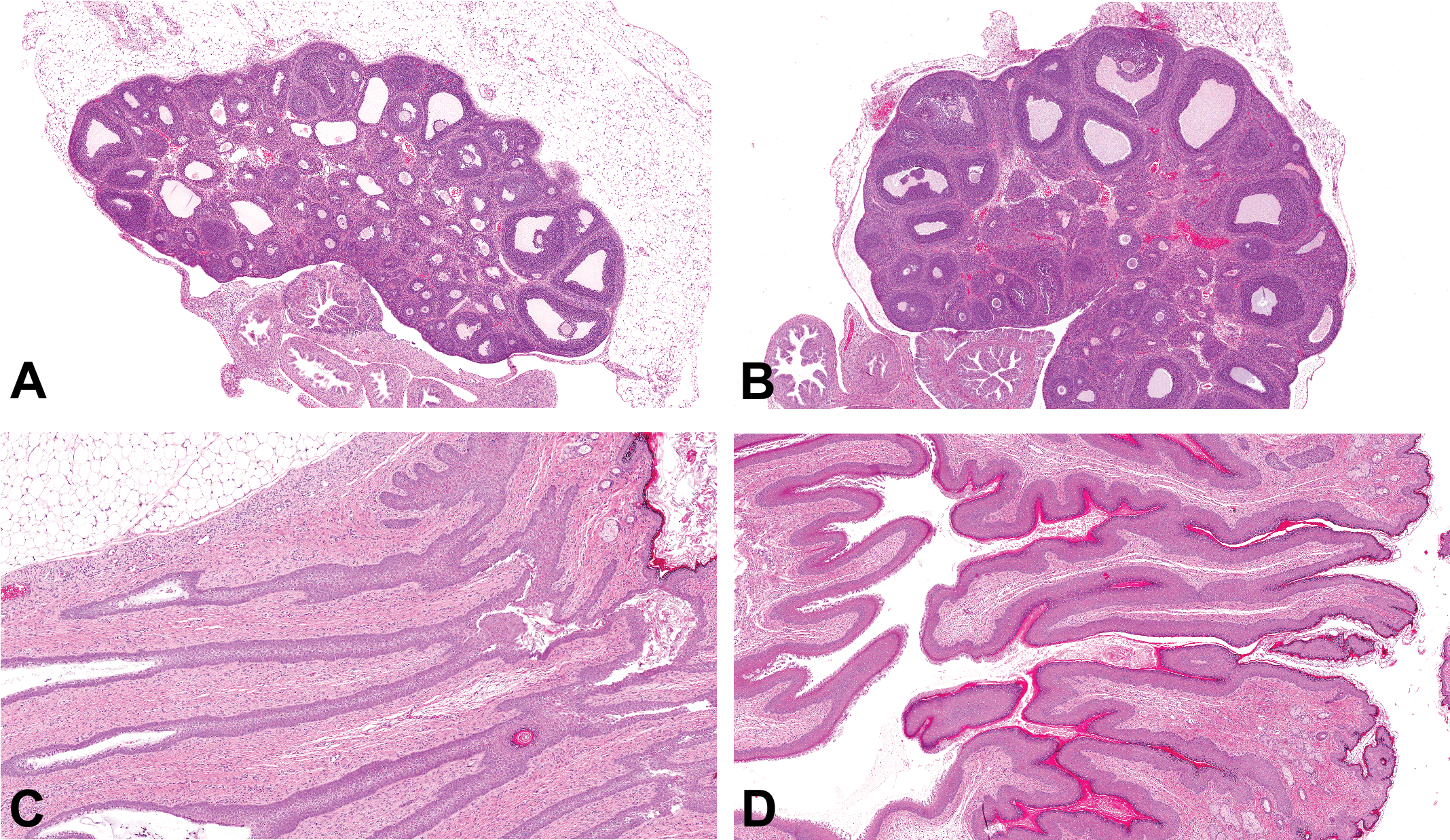
Photomicrographs (H&E) of the ovary and distal vagina from an immature rat on postnatal days (PND) 24 (A and C) and a rat entering the first proestrus on PND 34 (B and D). On PND 24, the ovary displays waves of follicular development along with follicular atresia and no corpora lutea are present (A). The distal vagina is closed and consists of a nonkeratinized stratified squamous epithelium without a lumen (C). On PND 34, the ovary displays multiple large antral follicles that are ready to ovulate and lacks the follicular atresia present in A (B). The proximal vagina has typical mucification consistent with proestrus, and the distal vagina has increased keratinization and the development of a lumen (D).
Immature
In order to understand the histologic features of the immature ovary, a basic understanding of ovarian development is required. During embryofetal development, primordial germ cells migrate from the yolk sac to the gonadal ridge and populate the gonad. These primordial germ cells become oogonia and begin to arrange in aggregates called germ cell nests (Wear, McPike, and Watanabe 2016). Following multiple rounds of mitotic division, oogonia enter meiosis and the primary oocytes arrest at the diplotene stage of development. The germ cell nests ultimately breakdown and individualize into primordial follicles (Pepling 2012).
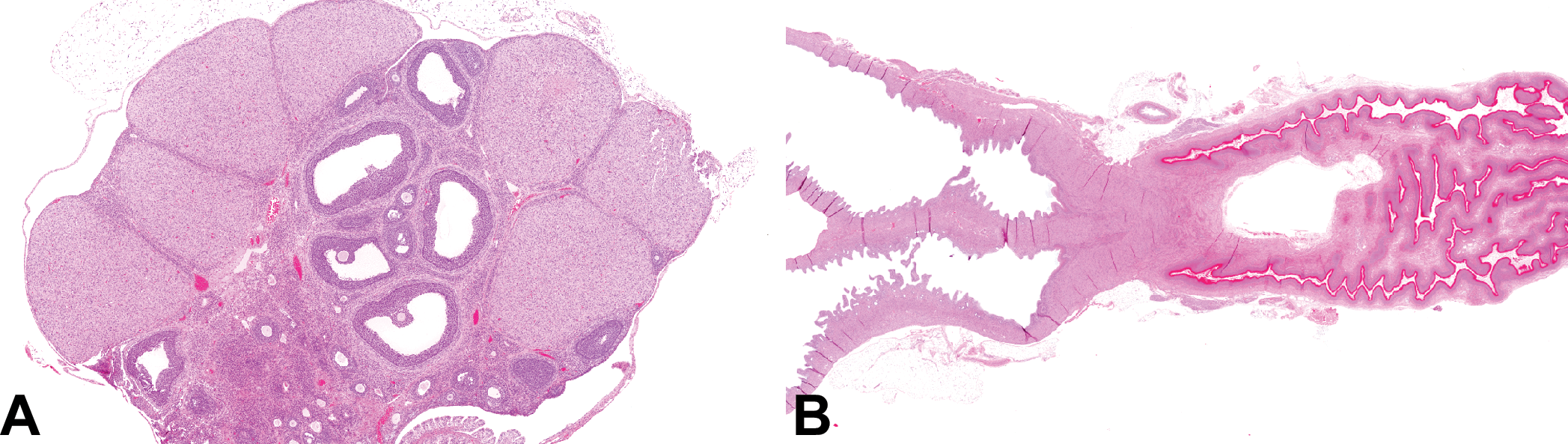
In the rat, remnants of the germ cell nests can still be observed in the first few postnatal days (PND; Rajah, Glaser, and Hirshfield 1992). By PND 6, most follicles have individualized to form primordial follicles, but remnants of germ cell nests can still be present. Follicular development continues slowly over the following weeks and has been previously reviewed (Picut et al. 2014, 2015). By approximately PND 15, early antral follicles begin to form, and over the following few weeks waves of tertiary follicles will form, but then undergo atresia (Figure 1A). The development of antral follicles should not be confused with maturity, as there is no ovulation at this point. During this period, the external aspect of the vagina is closed when viewed clinically. The histologic features of the immature closed vagina are detailed in Figures 1 and 2 and have been previously described (Forsberg, Jacobsohn, and Norgren 1966, 1968). It is not common to evaluate the distal aspect of the vagina in nonclinical toxicity studies, but it can provide useful information and additional characterization when presented with early or delayed vaginal opening. In one recent example, early onset of vaginal opening in rats given dabrafenib (an inhibitor of BRAF kinase) was observed; however, the histologic features of the ovary and distal vagina demonstrate that there was no evidence of precocious puberty despite the early vaginal opening (Posobiec et al. 2015). The ovaries lacked evidence of ovulation, there was no evidence of estrous cyclicity, and the vaginal opening was due to a direct effect of dabrafenib on the distal vagina and vulva (hyperplasia and increased keratinization).
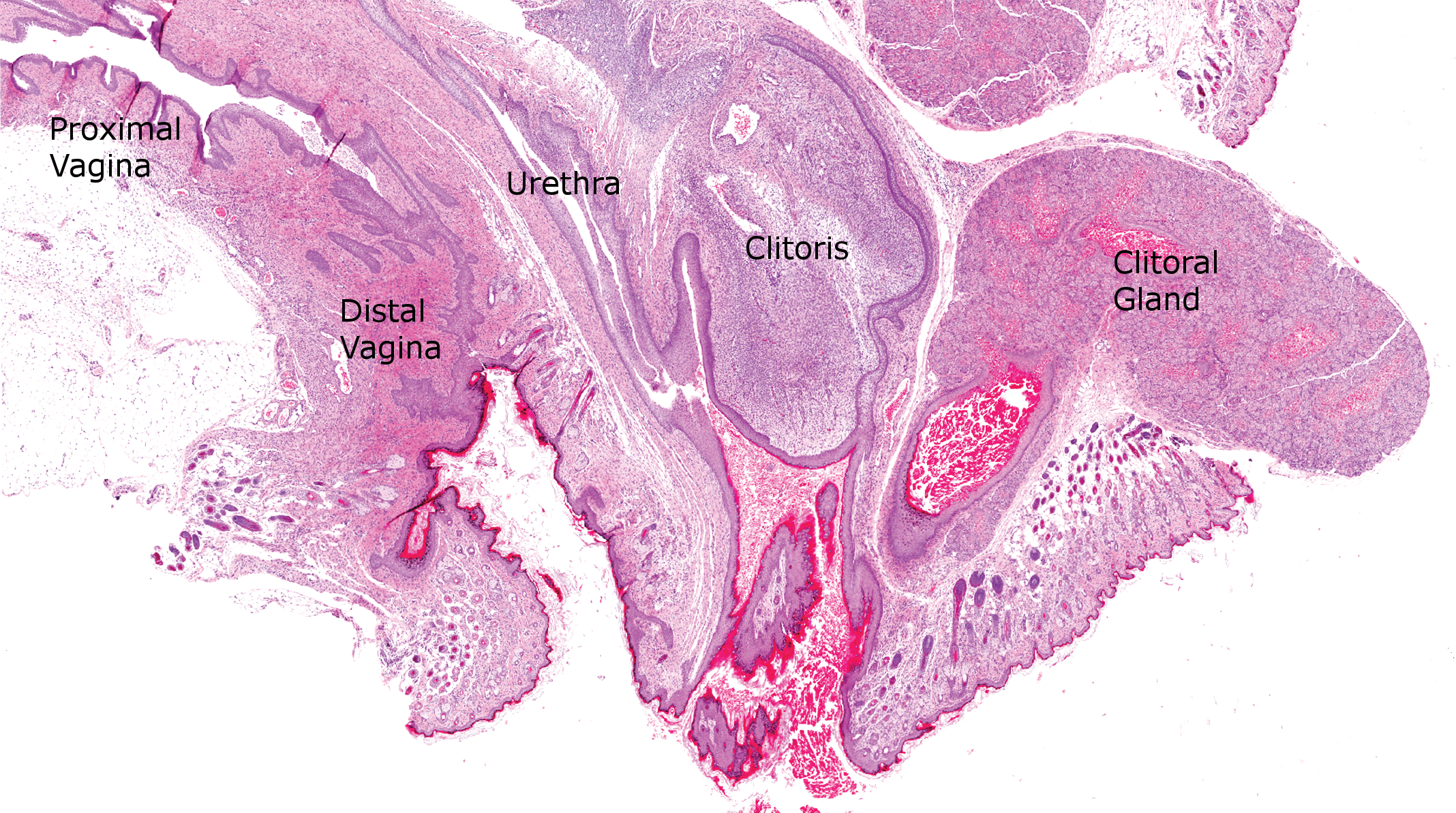
Photomicrograph (H&E) demonstrating the histologic features of the distal urogenital tract of a postnatal day 22 rat.
In the newborn NHP, the ovary consists of a dense band of primordial follicles within the cortex (Van Wagenen and Simpson 1973; Buse, Zöller, and van Esch 2008). Interestingly, upon close inspection, oogonia are still evident and can be observed undergoing mitotic division (Buse, Zöller, and van Esch 2008). Oogonia can be seen individually or as a part of germ cell nests (Figure 3; Van Wagenen and Simpson 1973). Oogonial mitoses can still be observed at 6 months of age in the cynomolgus monkey (Figure 3; personal observation) and have been reported in rhesus macaques as old as 10 months of age (Van Wagenen and Simpson 1973). Follicular development begins slowly, and by 6 months antral follicles are often present (Van Wagenen and Simpson 1973). Since many NHPs are immature in nonclinical toxicity studies, it is common to see numerous small to medium-sized follicles with varying degrees of atresia within the ovary along with an inactive endometrium (Figure 4A). Awareness of these normal developmental features is critical when assessing NHPs of this age. In addition, numerous polyovular follicles can often be seen in young macaques (Van Wagenen and Simpson 1973; Cline et al. 2008).
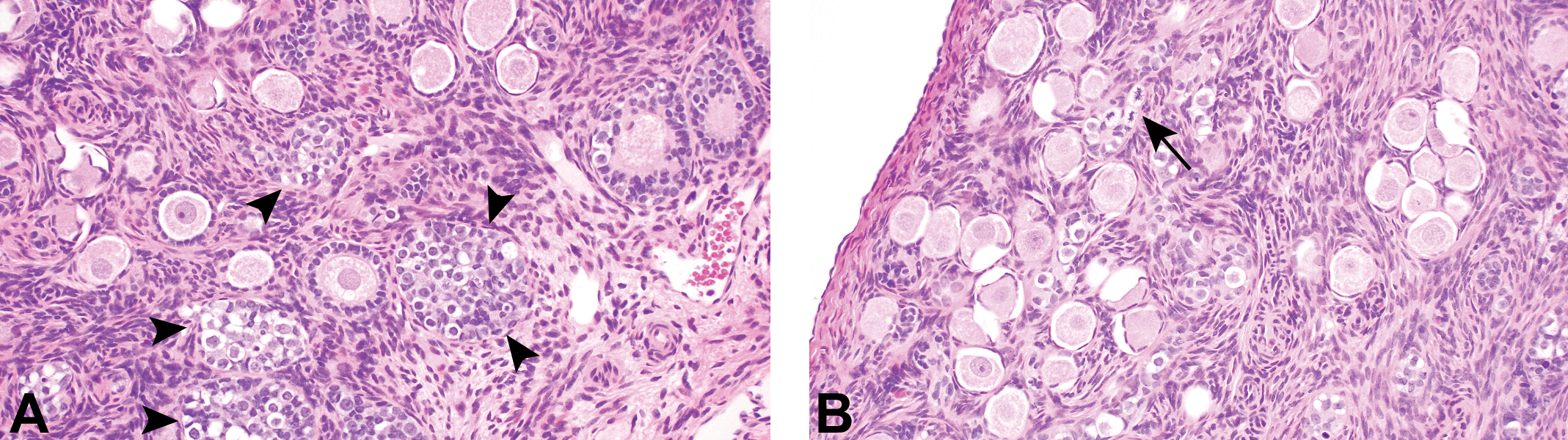
Photomicrographs (H&E) of the ovary from a 6-month-old cynomolgus monkey. Numerous germ cell nests (arrowheads) are still present (A) as are oogonia and oogonial mitotic figures (arrow; B).
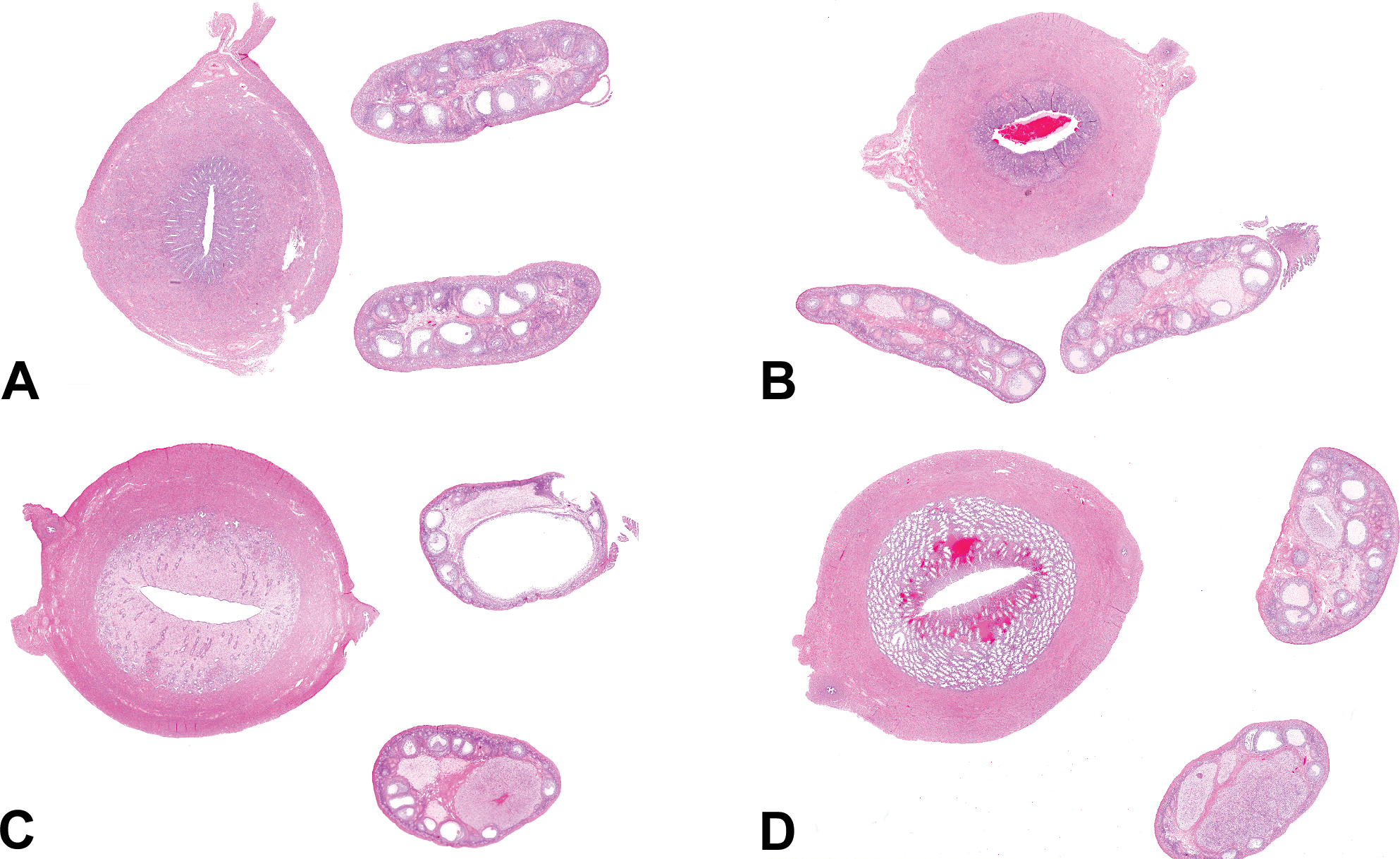
Photomicrographs (H&E) of the ovaries and uterus from an immature nonhuman primate (NHP; A), a mature NHP with an anovulatory cycle (B), a mature NHP with a large follicle and late follicular phase endometrium (C), and a mature NHP with a regressing corpus luteum and menstrual phase endometrium (D). Note the lack of corpora lutea in A and the presence of multiple corpora lutea in varying stages of regression in B to D.
The key histologic features to establishing that a given animal is immature include the following: no evidence of ovulation and no evidence of onset of cyclicity.
Pubertal
As rats enter their first proestrus at approximately PND 32 to 33, large antral follicles are present and are ready to ovulate (Figure 1B). Corpora lutea are not yet present, and distinguishing this pattern from a less mature ovary can be challenging (Figure 1A). The key feature during this stage of development is the beginning of vaginal opening. As estradiol levels rise during the first proestrus, the distal vagina undergoes physiologic hyperplasia and increased keratinization leading to development of a lumen (Figure 1D; Forsberg, Jacobsohn, and Norgren 1968). Following ovulation, multiple corpora lutea will be present within the ovary and additional keratinization will be present throughout the entire vagina, consistent with the first estrus. Rats at this age lack the classical pattern of regressing corpora lutea from multiple cycles as would be expected in an older animal, and it is common to see only 1 or 2 generations of corpora lutea at this age.
In a group of rats, the onset of cyclicity typically occurs over a 4- to 5-day period. It is common to see irregular cycles in the following few weeks, as the rats move toward full sexual maturity and more regular cyclicity. Cycling irregularity in young rats can include longer periods of diestrus, which can last 4 or 5 days as opposed to the more typical 2 days. This is different from the classical persistent diestrus/pseudopregnancy-like state where diestrus lasts 12 to 13 days. Another commonly observed cycle irregularity is 2 consecutive days of vaginal estrus (often termed “double estrus”; Goldman, Murr, and Cooper 2007). This occurs commonly in both rats and mice and is considered to be a normal variant. An important point for toxicologic pathologists evaluating young rats and mice is that there are distinct differences in the histology of the female reproductive system when compared to the classic 4-day estrous cycle depicted in most reviews. In order to understand the histologic pattern observed with 2 consecutive days of vaginal estrus, a brief review of the physiologic differences is warranted. In the classic 4-day cycle, the gonadotropin surge occurs during proestrus with ovulation during estrus. This leads to the expected pattern of large antral follicles within the ovary during proestrus and new basophilic corpora lutea during estrus. Vaginal keratinization occurs during estrus along with the presence of the newly formed corpora lutea. In cycles with 2 days of vaginal estrus, the gonadotropin surge does not occur during the period of vaginal proestrus, but rather occurs during the first day of vaginal estrus with ovulation following in the early morning of the second day of vaginal estrus (Goldman, Murr, and Cooper 2007). This leads to the presence of large antral follicles within the ovary with distinct keratinization within the vagina on the first day of a “double estrus” (Figure 5). This gives the histologic impression of a “proestrus” ovary with an “estrus” vagina. In addition, it is common to see the uterus distended with fluid at this time, which would typically be consistent with “proestrus” in the classic 4-day cycle descriptions, but often occurs here with vaginal keratinization (Figure 5). It is important to recognize this poorly described histologic pattern and not consider it to be a “mismatch” and diagnose an abnormality. On the second day of a “double estrus,” ovulation has occurred, so the expected pattern of new corpora lutea within the ovary along with vaginal keratinization is similar to the classic features of “estrus” histologically. This highlights the fact that estrous cycle stages are clinical entities and are not suitable morphologic diagnoses for histologic patterns or findings (Dixon et al. 2014).
Photomicrographs (H&E) of an ovary, uterus, and vagina from a postnatal day 38 rat with features consistent with the first day of 2 consecutive days of vaginal estrus. The ovary has multiple large follicles with one cohort of regressing corpora lutea from the previous cycle (A). These features are typical of what is expected during proestrus but occurs with a heavily keratinized vagina (B). This is a normal variant of the rat estrous cycle and should not be diagnosed as a “mismatch.” It often occurs with a dilated, fluid filled uterus (B).
Defining the onset of puberty and attainment of sexual maturity in the NHP primate is challenging. Clinically, the onset of menstrual cyclicity is determined by the observation of menstrual bleeding events. The first evidence of bleeding in species with a menstrual cycle is termed menarche. The dictionary definition of menarche is as follows:
In the author’s experience, this definition can lead to confusion as many often equate “the beginning of the menstrual function” with onset of regular cyclicity. In young NHPs, it is normal to see long anovulatory periods following the first evidence of menstrual bleeding. Resko et al. (1982) have studied this in detail in rhesus macaques and found that the initial intermenstrual intervals were >90 days and steadily decreased in length to approximately 40 days over a 1.5-year period following menarche. Along with these irregular cycle lengths, only 15% of animals ovulated in the first 5 intermenstrual intervals, but with time, ovulation rates increased slowly with successive intervals. This is important to recognize, as more nonclinical studies attempt to use mature NHPs to assess the reproductive system, and although menarche is reported at approximately 2.5 to 3 years of age in NHPs (Weinbauer et al. 2008), attainment of a more regular cycle may not occur until closer to 4 years of age. This highlights the point that evidence of a bleeding event or use of NHP age in isolation does not guarantee maturity or a regular cycle.
The toxicologic pathologist needs to be aware of these challenges, as these physiologic events are regularly seen histologically. As NHPs approach puberty, there are continual waves of follicular development and since these follicles produce estrogens, it is not uncommon to see endometrial glandular and stromal breakdown with hemorrhage, endocervical squamous metaplasia, and/or vaginal keratinization in these peripubertal animals (Cline et al. 2008; Vidal et al. 2013). It is also common to see a similar pattern of changes along with a single remnant of a corpus luteum. While this demonstrates evidence of a prior ovulation, it does not demonstrate sexual maturity and more likely reflects the presence of long anovulatory periods in young NHPs. Further, since there can be endometrial glandular and stromal breakdown with hemorrhage in these anovulatory NHPs, it is important to not confuse the presence of blood in the endometrium with a menstrual phase endometrium. Anovulatory cycles will have an inactive endometrium with simple, straight glands or a weak follicular phase pattern and may have variable amounts of superficial hemorrhage and no recent corpus luteum. In contrast, the menstrual phase endometrium will be considerably thicker with prominent complex, sacculated glands, more prominent hemorrhage, and stromal breakdown along with a recent corpus luteum that will likely be showing regressive changes (Figure 4; Vidal et al. 2013). Since bleeding can be a part of anovulatory cycles, the presence of clinical bleeding events does not necessarily mean that ovulation has occurred (Figure 4B).
The histologic features in a pubertal animal are not easily defined and can have overlap with both immature and mature animals. The histologic features in a pubertal animal typically include the following: evidence of ovulation and/or evidence of onset of cyclicity. Due to the variability in this age-group coupled with the disconnect between clinical landmarks of puberty and onset of cyclicity (as described in mice and NHPs above), the author and others often use the term “peripubertal” to avoid confusion around strict definitions. For example, an NHP might have clinical evidence of bleeding and be considered pubertal or even mature clinically but might not have evidence of ovulation histologically. In these cases, peripubertal seems to be a more acceptable term.
Mature
Female rats reach sexual maturity at a young age (typically by 8 weeks) and the peripubertal window is quite short, so in many cases toxicologic pathologists will be presented with mature rats. The histologic features of the normal rat estrous cycle have been extensively reviewed (Long and Evans 1922; Yuan and Carlson 1985; Peluso 1992; Li and Davis 2007; Westwood 2008; Yoshida et al. 2009; Vidal et al. 2013; Dixon et al. 2014; Sato, Nasu, and Tsuchitani 2016). The mature cycling rat has multiple generations of corpora lutea, which reflect the recent 3 to 4 cycles.
In NHPs, the peripubertal window is long and may last years (Resko et al. 1982). As NHPs reach sexual maturity and menstrual cyclicity becomes more regular, an additional factor must be considered. NHPs have a strong social influence on reproductive function. In established social hierarchies, the dominant animals cycle regularly, while the subordinate animals have a high incidence of anovulatory cycles and/or luteal phase defects (Adams, Kaplan, and Koritnik 1985). Subordinate NHPs may only cycle approximately half of the time due to this social influence. As a result, when tracking menstrual cycles in NHPs, it is common to observe abnormal cycles. This is the best-case scenario, and Weinbauer et al. (2008) have described the impact of changing the social hierarchy when animals are moved from single housing to new group housing. Mean cycle length increased from approximately 31 to approximately 46 days in the 6 months following transfer (Weinbauer et al. 2008). This is important to understand for nonclinical toxicity testing, as randomization at the outset of a study will set up a new social hierarchy that can have profound effects on the female reproductive system. Histologically, anovulatory cycles in mature NHPs look similar to those described for peripubertal animals, with the only clear distinction being the presence of multiple remnants of corpora lutea (Figure 4B).
The key histologic features to establishing that a given animal is sexually mature include the following: evidence of ovulation from multiple cycles and evidence of current cyclicity (Figure 4C and D). As NHPs have a relatively high incidence of anovulatory cycles in mature animals, evidence of current cyclicity is not always possible and the presence of multiple remnants of corpora lutea is often the only histologic indicator of maturity in these animals (Figure 4B). An analogous situation occurs in dogs due to the long anestrus period. While age alone is never recommended as a stand-alone determinate of sexual maturity, in these anovulatory NHPs age may help as a part of the weight of evidence when deciding to consider a given animal mature versus peripubertal.
Senescent/Postmenopausal
Reproductive senescence is a series of estrous cycle irregularities that occur as normal age-related changes in rats and mice, which marks the end of the functional reproductive life span in these species (vom Saal, Finch, and Nelson 1994). Reproductive senescence differs from menopause in that the initial changes are centrally mediated with alterations in hypothalamic function (Gore et al. 2000; Neal-Perry and Santoro 2006; Lederman et al. 2010). The sequence of events varies with both species and strain, but periods of increased cycle length, persistent estrus (also called persistent vaginal cornification), repeated pseudopregnancy (also called persistent diestrus), and/or eventually, persistent anestrus late in life can all be observed (vom Saal, Finch, and Nelson 1994). In rats, the timing of reproductive senescence can vary with different husbandry practices. The onset of reproductive senescence often occurs at an earlier age in rats housed singly when compared to group housed rats (LeFevre and McClintock 1988, 1991). It is believed that the social stress of isolation plays a role. In contrast, feed restriction and the associated decrease in body weight gain can prolong functional reproductive life span and delay the onset of reproductive senescence (Merry and Holehan 1979; McShane and Wise 1996). In addition, there are strain differences in the pattern of changes and timing in the onset of reproductive senescence (vom Saal, Finch, and Nelson 1994). Sprague-Dawley rats can begin to be observed in persistent estrus at 6 to 7 months of age and in the author’s experience as early as 4 to 5 months, especially when singly housed (vom Saal, Finch, and Nelson 1994; Shirai, Houle, and Mirsky 2015). During persistent estrus, there is a characteristic pattern of changes observed histologically including numerous follicular cysts with an absence of corpora lutea and secondary changes in the uterus, cervix, and vagina (Shirai, Houle, and Mirsky 2015). With prolonged periods of increased circulating estradiol, pituitary lactotrophs are stimulated leading to an increased production of prolactin (Pady, McComb, and Shull 1999). Increases in prolactin drive the proliferative changes commonly seen in the mammary gland (Welsch, Nagasawa, and Meites 1970). This is in contrast to Han Wistar rats, which are reported to begin having cycle irregularities after 6 months of age and move into persistent diestrus (Mitchard and Klein 2016). These factors must be carefully considered when designing and evaluating nonclinical toxicity studies. Any study with an in-life period ≥3 months has the potential to be impacted by reproductive senescence. Special attention needs to be paid to studies with necropsies occurring at different time points as it is not uncommon to see normal appearing rats at earlier time points and features of reproductive senescence at later time points. This can make evaluation of recovery/reversibility challenging. In addition, changes in the timing of reproductive senescence and related changes can occur as a test article–related effect (Wetzel et al. 1994; Nishiyama, Okudaira, and Saito 2006). This requires longitudinal assessment to definitively diagnose but can be observed in 3- to 6-month general toxicology studies with a very different histologic presentation of the treated animals. In these cases, the animals given the test article look relatively normal for animals going through reproductive senescence, but the age and timing do not match the concurrent controls. This can be seen with test article–related follicular cysts, as rats enter a persistent estrus phase much earlier than normal and the pattern of secondary changes is similar to that observed during the persistent estrus phase of reproductive senescence. It is suggested, however, to exercise some caution and not overdiagnose as a slight difference in the incidence is not likely to be significant due to the high degree of interanimal variability (LeFevre and McClintock 1988).
Most toxicologic pathologists are unlikely to encounter postmenopausal NHPs unless specifically working on animal models of aging. NHPs typically reach menopause after 20 years of age and the pattern of changes related to cessation of menstrual cyclicity have been previously described (Shideler et al. 2001; Kavanagh, Williams, and Wagner 2005; Buse, Zöller, and van Esch 2008). Unlike rodents, the primary underlying cause is reduced follicular reserves that lead to ovarian atrophy (Buse, Zöller, and van Esch 2008). While a detailed discussion of aging and age-related changes in NHPs is out of scope for this review, one finding worth noting is endometrial polyps. Endometrial polyps can occur as a spontaneous finding in both rhesus macaques and cynomolgus monkeys or occur as a test article–related change (Baskin, Smith, and Marx 2002; Kaspareit et al. 2007; Cline et al. 2008; Bennett et al. 2009). In rhesus macaques, endometrial polyps typically occur in older postmenopausal animals, but in cynomolgus monkeys, endometrial polyps have been observed in relatively younger animals (approximately 9 to 10 years old, with a range of 6 to 17 years). Awareness of this finding is important if attempting to start with mature cynomolgus monkeys in longer chronic toxicity studies.
The histologic features in an aging animal are not easily defined and are subject to species and strain differences.
Documentation of Sexual Maturity
Despite the challenges and considerable variability in the histologic features across the different stages of reproductive development, the toxicologic pathologist is often required to summarize the pattern of changes observed and document whether the animal is considered sexually mature in the context of the study. This is more typically encountered when working with nonrodents, as age alone is not an effective way to determine if animals are sexually mature; although, documentation of reproductive maturity in rodents can be helpful with juvenile toxicity studies and/or early deaths on studies. Labs will often document when an animal is clearly immature and then assume that if not stated otherwise, the animal is mature (Halpern et al. 2016). This can create problems as a study with all mature animals will have nothing documented, and unless this assumption is clearly included in the materials and methods in the study report, downstream readers will have no way to know if the animals were mature or not. Another approach and the author’s preferred approach is to document each animal as immature, pubertal (or peripubertal), or mature. This clearly documents reproductive status in all animals and gives the reader a complete picture of the potential variability across the study. Recognizing the challenges with NHPs around the time of puberty, some labs will forgo the pubertal designation and document only immature and mature or alternatively, simply document the presence or absence of corpora lutea and not make an assessment of maturity. When using this approach, it should be recognized that many of the NHPs that are in the year or 2 postmenarche may have evidence of prior ovulation and be erroneously considered fully sexually mature (see discussion above). Regardless of the terms used, another challenge is where to document this in the data entry system. Some labs will include immature, pubertal, and mature or the presence/absence of corpora lutea as a morphologic diagnosis under “ovary.” This has the advantage of easily being pulled into tables and historical control data, but once maturity status or presence/absence of corpora lutea is used as a morphologic diagnosis, it is no longer possible to also call the ovary normal. Some will even view “immature” as meaning the reproductive tract is not suitable for evaluation even though assessment at a younger age may be part of the intended study design. In an attempt to circumvent this issue, some labs will place the maturity status or presence/absence of corpora lutea as a tissue comment under “ovary.” This allows the pathologist to then clearly diagnose the ovary as normal, but the ability to easily generate tabular and historical control data is lost. An additional challenge with placing these data as morphology or comment under “ovary” is that there are inevitable comments about why the ovary is considered immature, but there is nothing noted for other reproductive organs. Another approach is to create an “organ” in the data entry system called “Sexual Maturity” or “Reproductive Status” and place maturity status and/or presence/absence of corpora lutea here. This would include a full assessment of all organs in the reproductive system, allow for individual diagnoses under “ovary,” and still be pulled easily into tabular summaries and historical control data sets. This is the author’s preferred approach when documentation of reproductive status is required, although it is recognized that with changes in the industry related to Standardization for Exchange of Nonclinical Data (SEND), additional work may be required in using this approach.
Conclusion
The age and reproductive status of animals in nonclinical toxicology studies can have a significant impact on the assessment of the reproductive system, interpretation of potential test article–related findings, and ultimately the risk assessment. This requires the toxicologic pathologist to have a thorough understanding of the normal anatomy, physiology, and histology of a variety of different age ranges with an understanding of strain and species differences. Careful review and documentation of sexual maturity within the pathology data will allow toxicologists and downstream readers to better understand how age as a variable may have impacted the results and interpretation.
Footnotes
Author Contribution
All authors (JV) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
