Abstract
This report describes the findings of preclinical testing of SCH 351591, a selective phosphodiesterase 4 inhibitor, in CD-1 mice over a wide range of doses, in which the heart and reproductive organs of both sexes demonstrated toxic effects. Repeat-dose toxicity studies assessed 5, 15, 50, 100, 200, 400, and 800 mg/kg/day, orally by gavage, for one or three months. Findings included higher testes and ovary weights and lower uterus weights (≥200 mg/kg), small ovaries/uterus (≥400 mg/kg), and histopathologic changes of large corpora lutea and ovarian atrophy at 200 and 800 mg/kg, respectively. In addition, chronic myocardial inflammation of the heart base occurred at 100 mg/kg. Vaginal staging of the estrous cycle revealed persistent diestrus. There was no histopathologic correlate or morphometric change to explain higher testes weights. A pilot fertility and early embryonic developmental toxicity study assessing doses of 100, 200, 400, and 800 mg/kg/day produced complementary results. Females had prolonged or abnormal estrous cycles, fewer successful pregnancies, increased ovarian corpora lutea, and decreased size of live litters owing to fetal resorptions. Male fertility was not affected. However, males had a 25% increase in testes weights at all doses. The pharmacology of specific PDE4 isoenzymes may explain both the reproductive and cardiac findings.
Keywords
Introduction
Cyclic nucleotide phosphodiesterases (PDEs) are enzymes that regulate the cellular levels of cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) by controlling their rate of degradation and thereby acting to modulate the various biologic processes controlled by these molecules. There are eleven recognized families of PDEs, of which isoforms of the phosphodiesterase 4 (PDE4) family appear to account for most of the cAMP-hydrolyzing activity of mammalian cells. From the characterization of PDE4 mRNAs and their corresponding protein products, it is known that there are at least sixteen to eighteen different PDE4 isoenzymes or variants expressed in mammalian cells that are likely to be involved in many different physiologic activities. Phosphodiesterase 4 isoenzymes are widely distributed in mammalian tissues. They can be found in blood vessels, smooth and cardiac muscle, the central nervous system (CNS), and most classes of immune cells (see Reviews in Beavo 1995; Bender and Beavo 2006; Matsumoto et al. 2003; Omori and Kotera 2007).
Phosphodiesterase 4 inhibitors are compounds of diverse chemical structure that have been extensively investigated as potential therapeutic agents, particularly for their anti-inflammatory properties (Giembycz 2000; Palacios et al. 1995). However, preclinical studies in various species including dogs (Heaslip and Evans 1995), rats (Larson et al. 1996; Zhang et al. 2008), pigs (Vogel et al. 1999), and monkeys (Losco et al. 2004) have demonstrated that the doses of PDE4 inhibitors that produce desired pharmacologic effects frequently overlap toxic doses. Although there are similarities in the nature of histopathologic findings, the target organs or systems affected tend to be characteristic of the species under study.
SCH 351591, a selective PDE4 inhibitor with approximately equal specificity for PDE4 isotypes A, B, C, and D, was previously evaluated in toxicity studies involving rats and monkeys. Rats administered 2 or 4 mg/kg/day developed gastrointestinal changes, including ileus and inflammation of the intestinal wall, and vascular effects that involved both inflammation of the muscular arteries, primarily of the mesentery and thymus, and apparent disturbance of the microcirculation, resulting in widespread interstitial edema of the soft tissues and hind limbs. The no-effect level for SCH 351591 in rats was 1 mg/kg/day (unpublished data, Schering-Plough Research Institute). Cynomolgus monkeys administered 12, 24, or 48 mg/kg/day developed inflammatory and degenerative changes of medium and large muscular arteries in various splanchnic organs, most notably the heart, kidneys, and stomach. In addition, monkeys experienced a significant mortality from sepsis, which was attributed to the immunomodulatory effects of the compound (Losco et al. 2004). The no-effect level for SCH 351591 in monkeys was determined to be 1.5 mg/kg/day (unpublished data, Schering-Plough Research Institute). Because mice proved to be singularly resistant to the toxic effects of SCH 351591, they were chosen for investigative toxicity studies, as well as for assessment of the reproductive effects of the compound. This paper describes the toxicologic findings in mice exposed to a wide range of doses of SCH 351591, in which a localized area of the myocardium and the reproductive tract of both sexes were determined to be target organs. The studies included two 3-month toxicity studies and one pilot fertility and early embryonic development study assessing the effects on both males and females.
Methods
Animals
Mice (Crl:CD-1[ICR]BR VAF/Plus) were purchased from Charles River Laboratories, Inc. Kingston, NY for both the toxicity and reproductive studies. For the toxicity studies, mice were six to eight weeks old at first dosing. They were eight weeks (males) or ten to eleven weeks (females) old at initiation of the reproductive study. Mice were housed individually in suspended stainless-steel cages, except during the cohabitation period for the reproductive study when each female mouse was housed with a male mouse, as specified in the study design. PMI Nutrition International, Inc. Richmond, IN. Certified Rodent LabDiet 5002 (meal) and tap water (automatic watering system) were provided ad libitum for all study animals. The animal rooms were maintained at a temperature of 72°F ± 4° F, relative humidity of 40%–70%, and a twelve-hour light/dark cycle. All studies were conducted in an AAALAC-accredited facility in accordance with the National Research Council’s Guide for the Care and Use of Laboratory Animals and the Animal Welfare Act.
Test Article
SCH 351591 is a potent (IC50 = 58nM) and highly selective inhibitor of PDE4 isoenzymes. SCH 365351 is the major active metabolite of SCH 351591 in rats, mice, and monkeys, and it is also a potent and highly selective PDE4 inhibitor ((IC50 = 20 nM). Both SCH 351591 and SCH 365351 inhibit all four PDE4 subtypes (A, B, C, and D) equally well (Billah et al. 2002). SCH 351591 was prepared in 0.4% (w/v) aqueous methylcellulose at concentrations ranging from 0.5 to 80 mg/mL, depending on the required dose to be administered to each group. All mice, including controls, were dosed at a volume of 10 mL/kg. The same 0.4% (w/v) aqueous methylcellulose was used as the vehicle control.
Study Designs
Three-Month Toxicity Studies
Two 3-month studies with similar design were performed in mice to cover the broad range of doses evaluated. The initial study involved the four lower doses investigated (5, 15, 50, and 100 mg/kg/day), which were chosen based on previous experience with this compound in rats. The second study was performed at the higher doses (200, 400, and 800 mg/kg/day) to demonstrate definitive toxicity after equivocal effects on the heart and reproductive organs were observed at the lower doses. For brevity, results from both studies are combined. Twenty mice/sex/group (a total of forty controls/sex from the two studies) were dosed once daily by gavage with 0 (4% methylcellulose vehicle), 5, 15, 50, 100, 200, 400, or 800 mg/kg of SCH 351591. Half of the study animals were sacrificed after one month of dosing, whereas the other half was sacrificed after three months of dosing. All mice were euthanized by exsanguination under isoflurane-induced anesthesia, and each underwent a complete necropsy. Organ weights were obtained from fresh tissue at necropsy for the heart, brain, kidneys, liver, spleen, lungs, thymus, ovaries, uterus, testes, and epididymides. A standard panel of thirty-nine tissues was preserved in 10% neutral buffered formalin (excepting testes), embedded in paraffin, stained with hematoxylin and eosin (H&E), and examined microscopically. Testes were fixed in Bouin’s solution for twenty-four hours and then transferred to 10% neutral buffered formalin prior to processing. Additionally, the uterus and vagina of females in the second (higher doses) toxicity study were assessed for staging of the estrous cycle, and the testes of all males were stained with periodic acid–Schiff (PAS) to assess spermatogenesis.
Because study results included higher testes weights in dosed males with no corresponding histopathologic correlate, a simple morphometric assessment was performed on the testes to investigate the possibility that changes in interstitial or seminiferous tubular fluid volume might be responsible for the higher testes weights. Morphometry was performed on the testes of ten males/group from the control, 400 mg/kg, and 800 mg/kg groups using a microscopic point-counting technique that compared the proportional volume fractions of the testes occupied by seminiferous tubular epithelial cells, tubular lumina, and interstitial tissue or space. Both testes from each animal were evaluated using a Weibel’s multipurpose grid (Reticle KR-821, Klarmann Rulings, Inc., Litchfield, NH, USA) with approximately 50% of the measurements taken from each testis. Care was taken to avoid areas of tissue artifact and to assess tubules cut in transverse section for uniformity. Sufficient measurements were collected from each animal (ten fields/mouse involving 420 points) to provide a relative standard error (RSE) of less than 5% (Ahern and Dunnill 1982).
A separate set of satellite animals was maintained at each dose level to obtain toxicokinetic parameters. Satellite group mice were sacrificed at multiple standard time points (0.5, one, two, four, nine, and twenty-four hours) post-dosing on days 0 (initial dose) and 59 for dose groups 0, 5, 15, 50, and 100 mg/kg, and on days 0 and 87 for dose groups 0, 200, 400, and 800 mg/kg. Pooled plasma samples from three satellite mice/time point were assayed by liquid chromatography for the presence of SCH 351591 and its primary metabolite SCH 365351.
Reproductive Toxicity Study
A pilot fertility and early embryonic developmental study was performed in which parental mice of both sexes were administered 0, 100, 200, 400, or 800 mg/kg SCH 351591. Dosed males were mated first with a cohort of dosed females followed by a second mating with a cohort of undosed females. For assessment of male fertility, the male mice were administered the test article as a single daily dose, orally by gavage, for three weeks prior to mating and throughout the cohabitation period until the day prior to sacrifice. For assessment of female fertility, the females were administered the test article in a single daily dose, orally by gavage, for two weeks prior to and throughout the cohabitation period, and on gestation days 0 through 6. Body weight and body weight gain were assessed twice weekly and on the day of sacrifice for both sexes. Both before and during cohabitation, vaginal smears of all females were collected daily to determine the stage of the estrous cycle. During cohabitation, each dosed female mouse was placed with a male mouse from the same group for a period of seven days or until positive evidence of mating (i.e., copulatory plug and/or a sperm-positive vaginal smear) was observed. Female mice were removed from cohabitation once mating was confirmed. If no mating occurred after seven days, up to two additional seven-day cohabitation intervals with different proven males were attempted. At the end of the twenty-one–day cohabitation period, remaining females were returned to their own cages whether or not positive evidence of mating was detected. (For females for which positive evidence of mating was not detected, dosing was continued for the twenty-one–day cohabitation period and for presumptive gestation days 0 through 6 that followed.)
Male mice were sacrificed at the end of their second cohabitation period (with undosed females). Female mice were sacrificed on gestation day 14, or, if mating was not confirmed, on the 14th day after the end of their cohabitation period. All mice were euthanized by CO2 inhalation. All mice were subjected to an external and visceral necropsy examination. For control and dosed males, the testes and epididymides were weighed, fixed in Bouin’s solution for twenty-four hours, and transferred to 10% neutral buffered formalin. For all females, the uterus and ovaries were exposed to collect reproduction data. The number of corpora lutea (CLs), implantation sites, fetuses (live and dead), and resorptions (early and late) were counted for all mice. The uteri of apparently nonpregnant mice were examined by ammonium sulfide staining for evidence of implantations. For both sexes, the following mating and fertility data were calculated:
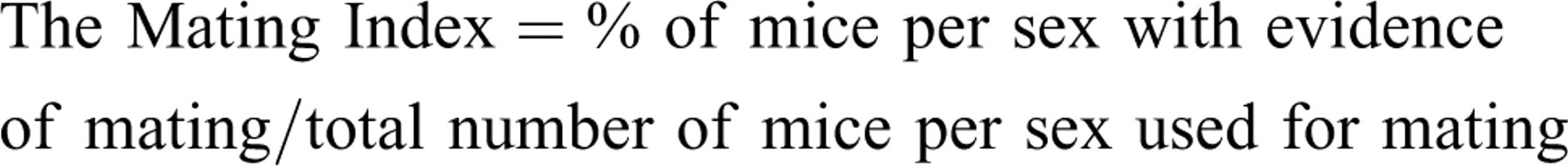

In addition, the precoital interval (number of days to mating) was recorded for all females.
Results
Three-Month Toxicity Studies
Pharmacokinetics
Plasma SCH 351591 concentrations, expressed as area under the curve (AUC), were sex independent; therefore, data for both sexes were combined. Plasma concentrations of SCH 351591 increased in a dose-related manner in the dose range of 5 to 100 mg/kg but appeared to plateau with marginal further increases at doses of 200 to 800 mg/kg (Figure 1a). Exposure to the metabolite SCH 365351 varied from 0.3% to 30% of the parent compound in various groups and at various time points. In general, the extent of metabolism of SCH 351591 to SCH 365351 increased with dose and was greater for female mice than for males (Figure 1b). However, it was noted that the percentage of drug metabolized decreased with multiple doses, particularly at doses >200 mg/kg, suggesting saturation of the metabolic processes with higher doses and over time (Figures 1c and 1d). At 100 mg/kg, which was the lowest dose at which myocardial and reproductive toxicity was defined, AUC values on day 59 were in the range of 153 (females) to 159 (males) for SCH 351591 and 12.3 (females) to 13.2 (males) for SCH 365351. Thus, metabolite exposure was approximately 8% that of the parent drug. Accumulation was negligible for both SCH 351591 (
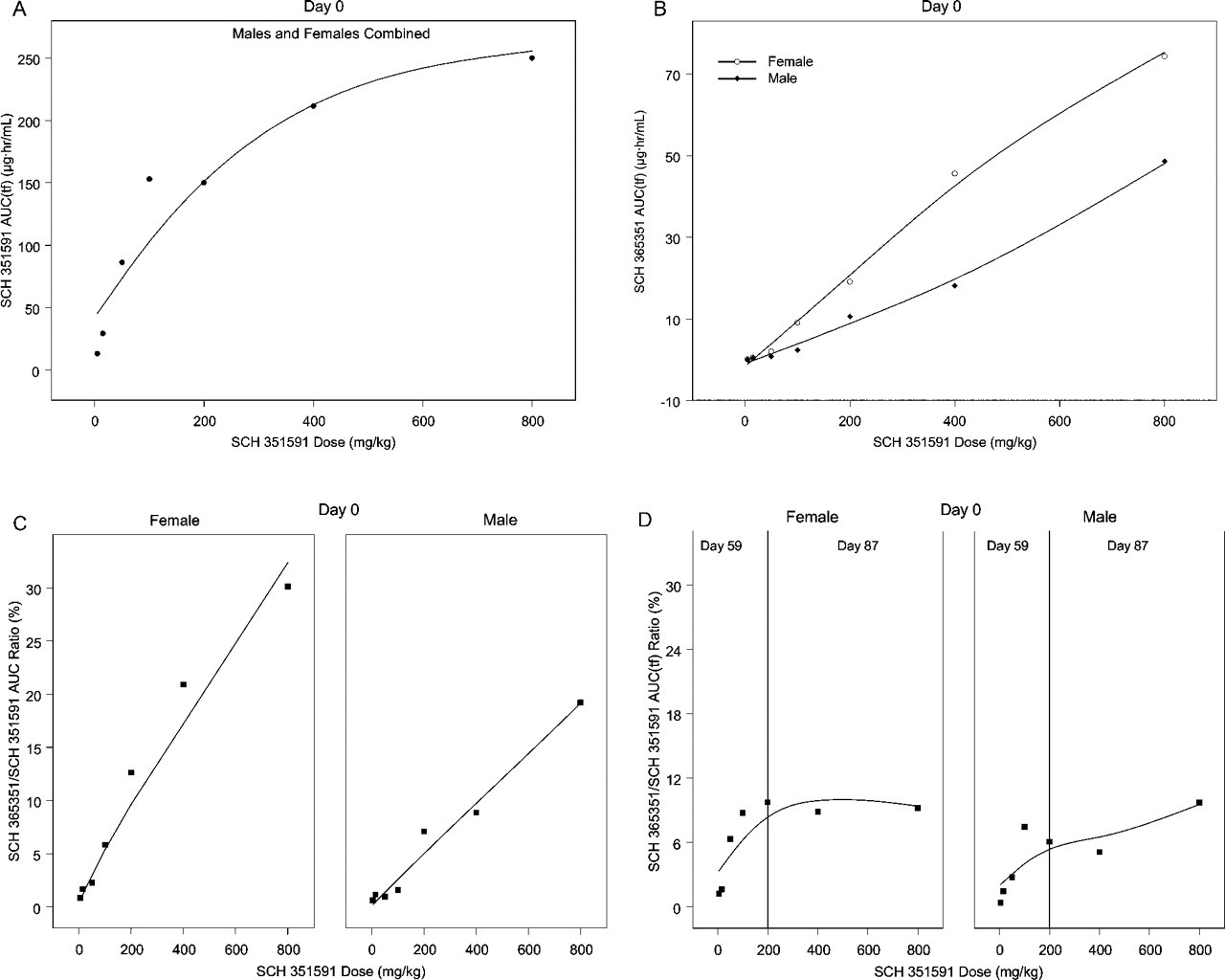
Toxicokinetic results of the two combined three-month toxicity studies expressed as mean area under the curve of SCH 351591 (a) and metabolite SCH 365351 (b) values on day 0 versus dose. c/d shows the area under the curve ratio values of metabolite to parent (SCH 365351/SCH 351591) versus dose.(c) shows the data for day 0, separated by sex, whereas (d) shows the ratios after repeated dosing. The percentage of drug metabolized decreased with repeated dosing and higher doses, suggesting saturation of the metabolic processes.
Organ Weights
Half of the toxicity study animals were sacrificed after one month of exposure, and the remaining mice were sacrificed after three months. Because the organ weight, necropsy, and histopathologic findings showed similar trends at both sacrifice intervals, the data are presented for the one-month and three-month sacrifices collectively. The mean absolute and relative (as a percentage of body weight) organ weight data for each group are expressed as the percentage difference from the concurrent control means (Table 1 ). Changes in the mean absolute and relative weights of male testes were equivocal (approximately +10%) for the 50 and 100 mg/kg dose groups, as compared with the concurrent controls, but rose to approximately +20%-25% for the remaining groups at ≥200 mg/kg. Testes weight changes were generally comparable at the one- and three-month sacrifices. For female mice sacrificed at one month, the changes in ovarian weights were highly variable and not dose related. However, for mice sacrificed at three months, ovarian weights were increased in non-dose–related fashion for all dose groups, with an approximate 30% increase above controls of mean absolute ovarian weights at 200 and 800 mg/kg/day. Higher ovarian weights correlated with the histopathologic finding of increased large CL in many females. There was a trend to minimally lower uterine weights at one or both sacrifices for doses of 100 mg/kg or higher that was not consistent in magnitude for the two sacrifices. In spite of the variability of the data for females, the ovarian and uterine weight changes were attributed to SCH 351591 administration owing to the histopathologic observations that correlated with individual animal organ weight variations.
Organ weight findings from the toxicity studies with SCH 351591 one-month and three-month necropsies. a
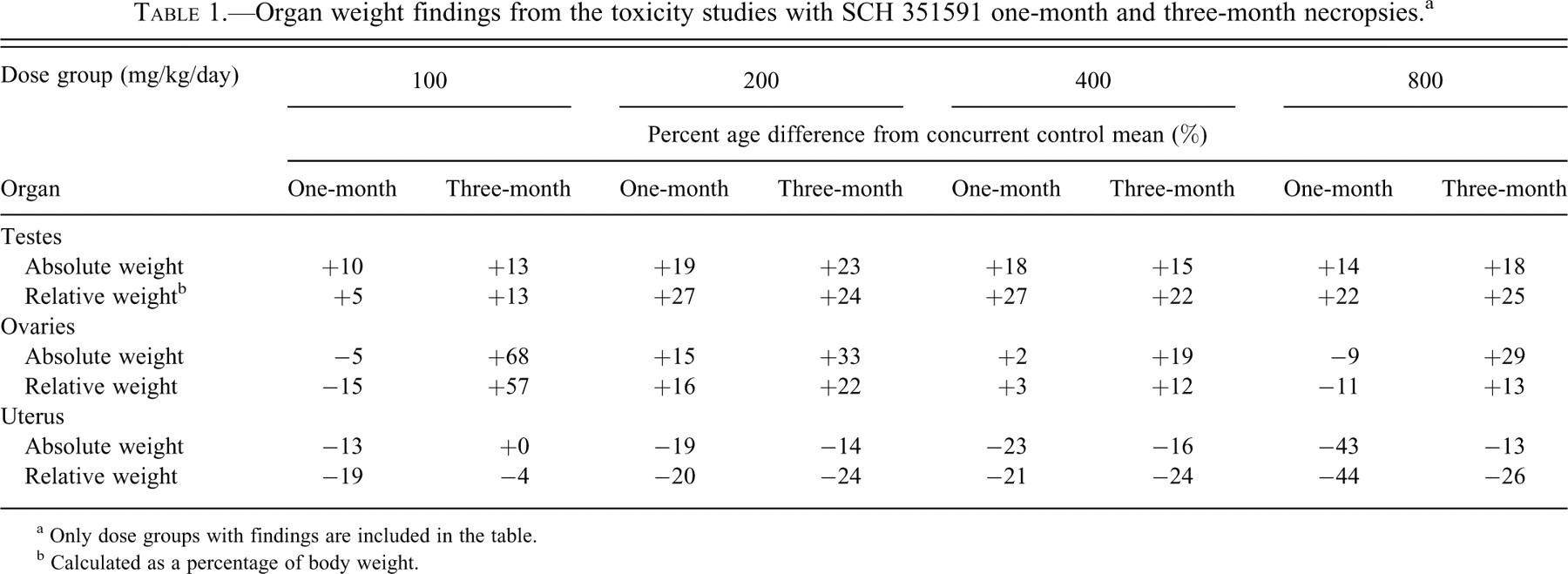
a Only dose groups with findings are included in the table.
b Calculated as a percentage of body weight.
Pathology Findings
There was no SCH 351591-related mortality. Pathologic results for the one-month and three-month sacrifices were consistent and similar at both intervals and are, therefore, presented together. SCH 351591-related macroscopic findings were limited to observations of small ovaries and/or uteri of a few females administered 400 or 800 mg/kg that correlated with the histopathologic finding of atrophy. No macroscopic change of the testes was appreciated at necropsy to correlate with higher organ weights.
Pertinent SCH 351591-related histopathologic findings occurred in the heart at ≥100 mg/kg and the female reproductive tract organs at ≥200 mg/kg. Minimal to moderate myocardial inflammation was characteristically located at the heart base, where it involved the root of the aorta and the emerging main coronary arteries. In mild cases, lesions appeared to initiate with edema and mixed neutrophil and/or mononuclear cell infiltrates localized to the proximity of the aortic root and coronary arteries (Figures 2a and 2b), but not within the arteries themselves. In more severe or chronic cases, the inflammation was characterized by interstitial edema along the periphery of lesions, foci of hemorrhage and necrosis with predominantly neutrophilic infiltrates, and extensive proliferation of fibroblasts and macrophages that destroyed the myocardium and the walls of medium and large arteries (Figures 2c–2e). Affected arteries, including the aorta, had changes ranging from segmental vacuolation of the medial smooth muscle, scattered neutrophilic infiltrates, and localized necrosis to chronic fibrosis with narrowing of vessel lumens. Coronary arteries in the area of inflammation often had proliferative changes, particularly of the media and intima, as well as hypertrophy of endothelial lining cells (Figure 2f). In some mice, inflammatory changes extended to involve the auricles and the epicardial surface of the heart or the endocardium, particularly of the left atria, with resultant thickening and malformation of the aortic or atrioventricular valves. Although the atria could be affected for both the left and right heart, the ventricles were uniformly spared. Chronic myocardial inflammation occurred in females at ≥200 mg/kg at the one-month sacrifice and in one or both sexes at ≥100 mg/kg at the three-month sacrifice. In general, the incidence and severity were not dose related, although females were affected slightly more often than males (Table 2).
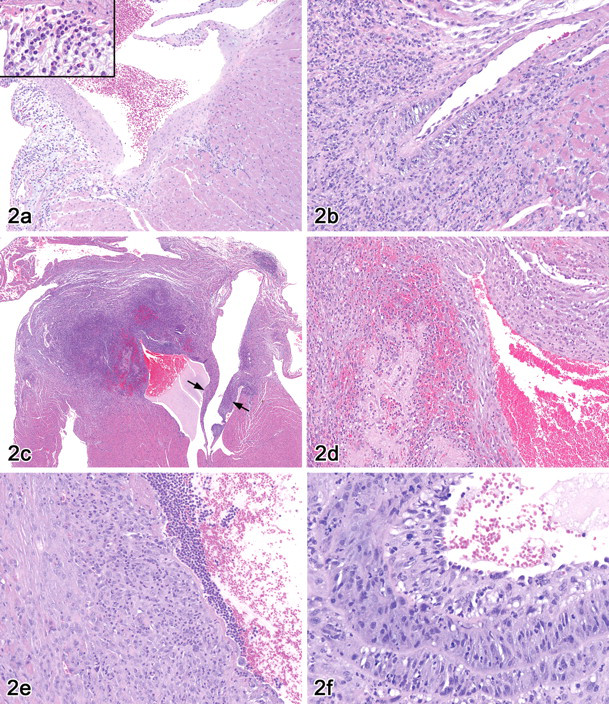
Histopathologic findings in the heart of SCH 351591-dosed mice. (a) Early inflammation at the base of the aorta showing predominantly neutrophilic infiltrates (H&E, 50×, Inset = 150×). (b) Early segmental lesion of an intramural coronary artery in the right heart with vacuolation of the media. Pathologic changes in the vessel are localized to areas with periarterial inflammation (H&E, 100×). (c) Severe inflammation affecting the atria and aortic outflow tract of an 800 mg/kg mouse. Note thickening and malformation of the atrioventricular valve leaflets (arrows) (H&E, 16×). (d) Higher magnification of (c) shows area of hemorrhage and necrosis with mixed cellular infiltrates (H&E, 100×). (e) Chronic lesion at the base of the aorta. Note neutrophilic exudate and fibrin along the luminal border and macrophages and fibroblast proliferation of the wall (H&E, 200×). (f) Chronic lesion of a left intramural coronary artery with endothelial hypertrophy and proliferative changes of all arterial layers (H&E, 200×).
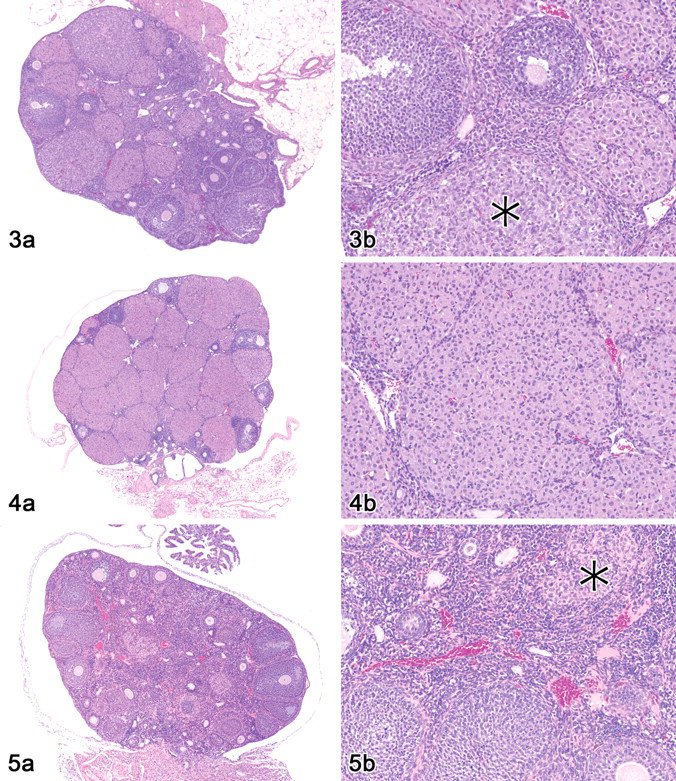
Figure 3. Ovary of a control group mouse in diestrus. (a) There are developing follicles of various stages and mature to regressing corpora lutea (H&E, 25×). (b) Higher magnification of (a) showing mature acidophilic and regressing basophilic (*) corpora lutea (H&E, 100×).
Figure 4. Ovary of an 800 mg/kg female. (a) Shows increased mature corpora lutea and no developing follicles (H&E 20×). (b) Mature corpora lutea are large and acidophilic with no sign of regression (H&E, 100×)
Figure 5. Ovary of a 400 mg/kg female. (a) Atrophy is characterized by lack of advanced developing follicles or active corpora lutea (H&E, 40×). (b) Same ovary with dense stroma and a corpus luteum remnant (*) (H&E, 100×).
SCH 351591-related histopathologic findings of the heart. a

a Only dose groups with findings are represented in the table.
b Data include findings for both the one-month sacrifice in parentheses and the three-month sacrifice.
Histopathologic findings of the female reproductive tract at ≥200 mg/kg included an increase in the numbers of large acidophilic-stained ovarian CLs that correlated with higher individual animal ovarian weights. In addition, there were rare cases of atrophy of the ovaries, uterus, and/or vagina (in ≥400 mg/kg females), alterations of the estrous cycle (established by staging of the uterus and vagina) as compared with control females, and infrequent findings of vaginal mucosal atypia. The latter finding was characterized by hypertrophy and mucification of the surface epithelium with or without atrophy of the stratum germinativum (Table 3 ). The incidence of findings in female reproductive organs was dose related between the 200 mg/kg group and the higher doses but was comparable for the 400 and 800 mg/kg groups. Almost all the mice appeared to be cycling because their ovaries contained a mixture of developing follicles, recent CLs, and abundant interstitial cells. However, approximately half of the dosed mice in the higher dose groups had increased numbers of very large CLs that lacked the vascularization or degeneration of regression, which would be expected in nonpregnant females. Figures 3–5 show a comparison of the ovaries of a control female in diestrus, a dosed female with increased CLs, and a dosed female with atrophy (mice dosed for three months).
SCH 351591-related histopathologic findings of the female reproductive organs. a

a Only dose groups with findings are represented in the table. Data includes findings for the 1-month and 3-month sacrifices combined.
b Refers to change of surface epithelial cells with or without thinning of the mucosal layers.
Figure 6. Tissues of a control female in diestrus. (a) The uterus has a thick myometrium and endometrium with abundant glandular development (H&E, 50×). (b) The vaginal mucosa shows multiple layers of cohesive mucified cells derived from the stratum germinativum (H&E, 200×).
Figure 7. Tissues of an 800 mg/kg mouse with increased ovarian CLs. (a) The uterus has normal glandular development, but thinning of the endometrial stroma and muscularis (H&E, 50×). (b) Shows mild vaginal atrophy with hypertrophy and mucification of the surface epithelium (H&E, 200×).
Figure 8. Tissues of an 800 mg/kg mouse with ovarian atrophy. (a) The uterus has marked thinning (atrophy) of the endometrium and myometrium (H&E, 100×). (b) Vagina, showing severe atrophy of the stratum germinativum with hypertrophy and mucification of the surface epithelium (H&E, 200×).
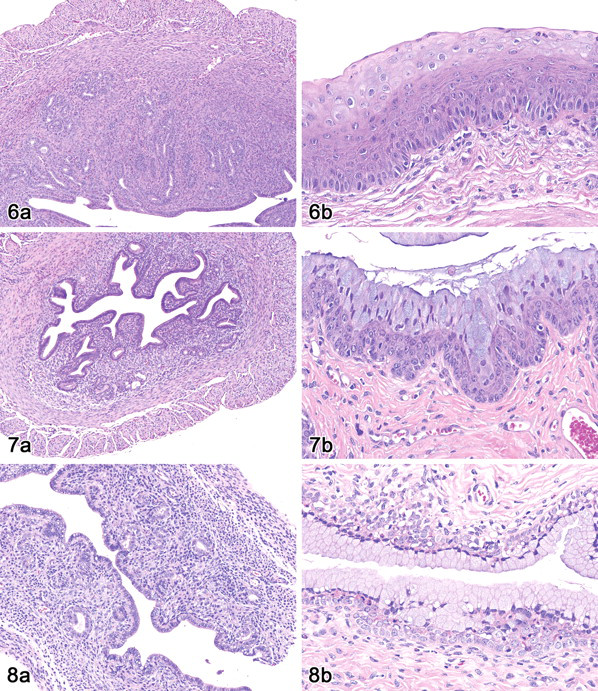
Staging of the estrous cycle of the uterus and vagina revealed that SCH 351591-administered mice were less likely to be in proestrus and more likely to be in diestrus than concurrent controls. Although most females had evidence of normal glandular development in the uterine endometrium, a number of mice with either increased CLs or ovarian atrophy had thinning of the endometrium and/or myometrium that correlated with lower uterine weights. In addition, a few mice, particularly those with ovarian abnormalities, had atypia of the vaginal mucosa that did not match the appearance of any normal estrous cycle stage. Such mice exhibited pronounced hypertrophy and mucification of the superficial vaginal mucosal layer. In addition, some females with mucification also had atrophy of the vaginal mucosa characterized by a thin (one to five cell layers) stratum germinativum. In general, mice with increased large CLs were in diestrus and accounted for most of the individuals with vaginal mucification. However, some mice with ovarian atrophy also exhibited similar vaginal changes and were more likely to have atrophy of the mucosa as well. Because of the normal physiologic variability, ovarian atrophy was diagnosed only in mice that lacked developing follicles and mature CLs and also exhibited atrophy of the uterus and atrophy with or without mucification of the vagina. Figures 6–8 compare the appearance of the uterus and vagina of a control female in diestrus, a dosed female with increased ovarian CLs, and a dosed female with ovarian atrophy.
In males, the testes had no test article–related histopathologic findings. Morphometric assessment showed no significant difference (variations < 10%) in the volume fraction of the testes occupied by seminiferous tubular cells, tubular lumina, or interstitial cells/space (Table 4).
Morphometric assessment of the testes a in a three-month toxicity study with SCH 351591.

a Data are the mean volume fraction (expressed as the percentage) occupied by the designated tissue component.
b Numbers in parentheses equal the percentage difference from controls.
Pilot Fertility and Early Embryonic Development Toxicity Study
Body/Organ Weight Effects
The only differences in body weight and body weight gain occurred in the females on gestation days 6 to 14. The mean weight gains for this interval in all dosed groups were lower than for concurrent controls. Mean weight gains were 10.7, 7.7, 9.8, and 7.5 g for the 100, 200, 400, and 800 mg/kg groups, respectively, as compared with 12.0 g for the control females. This variation was likely related to the increase in resorptions resulting in fewer live fetuses in the affected groups (see Table 5).
Summary of cesarean section data for SCH 351591-dosed males mated with dosed females.
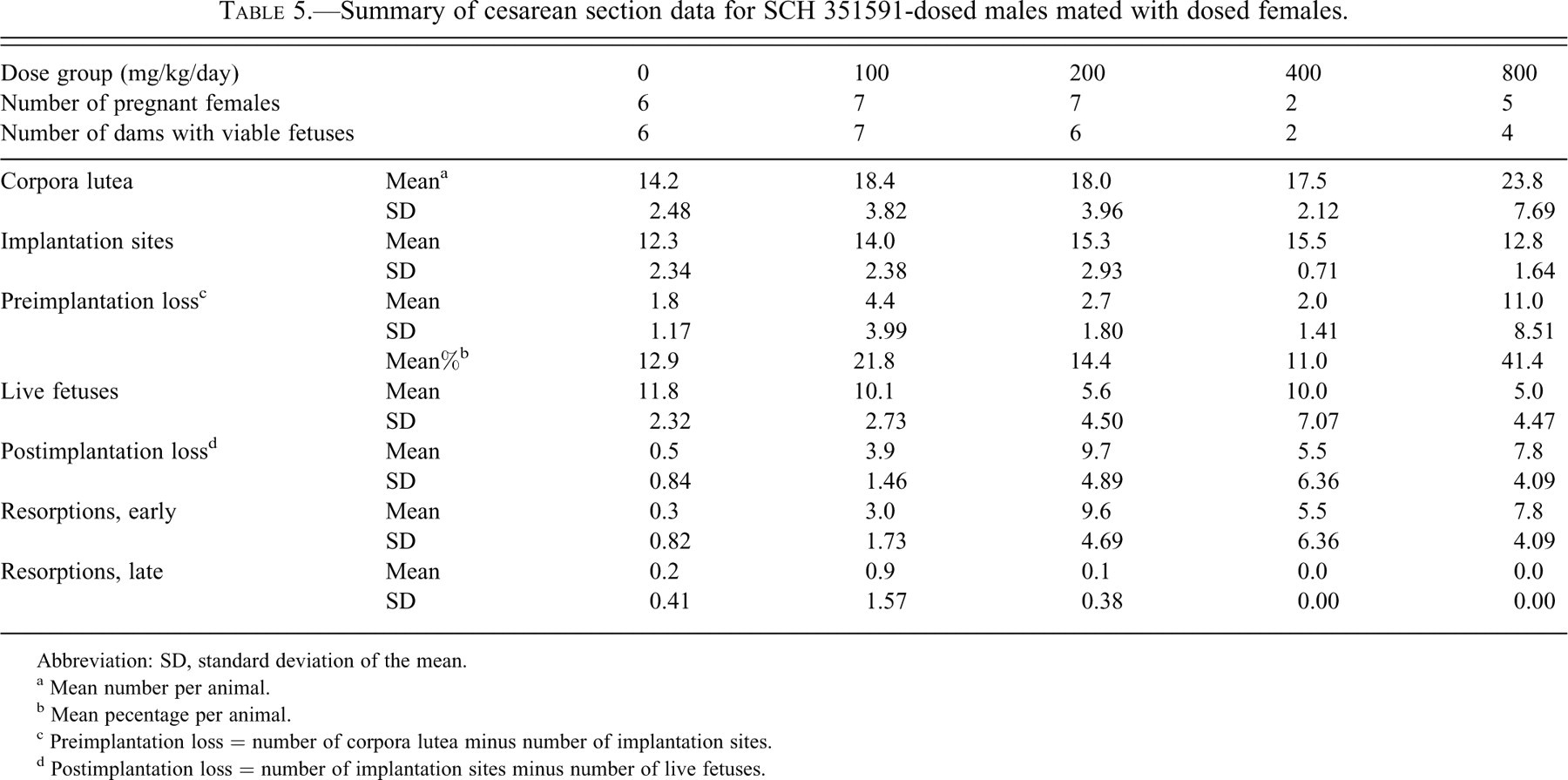
Abbreviation: SD, standard deviation of the mean.
a Mean number per animal.
b Mean pecentage per animal.
c Preimplantation loss = number of corpora lutea minus number of implantation sites.
d Postimplantation loss = number of implantation sites minus number of live fetuses.
The mean testes weights in each of the SCH 351591-dosed groups were approximately 25% greater than the concurrent control values. The mean weights of the epididymides were comparable with the controls (data not shown).
Estrous Cycle Effects
There was an increase in abnormal estrous cycles of females, including shortened or prolonged irregular cycles and periods of persistent diestrus or estrus in all SCH 351591-dosed groups. Abnormal cycles occurred in three of eight females in the 100 and 200 mg/kg groups and in four of eight and five of eight females in the 400 and 800 mg/kg groups, respectively. This increase in abnormal cycles coincided with an increase in the mean number of days to mating in all SCH 351591-dosed groups and with fewer females mating in the 400 and 800 mg/kg groups.
Mating of Dosed Males with Dosed Females
The summary of cohabitation data for dosed males mated with dosed females is presented in Table 6 . All females in the control, 100, and 200 mg/kg groups mated, but only five of eight (62.5%) of the females mated in the 400 and 800 mg/kg groups. Although this finding resulted in lower male mating indices for the latter two dose groups, the decrease in mated pairs was attributed to abnormal cycles in the females, because a second cohabitation of treated males with untreated females resulted in 100% successful mating for all dose groups. The precoital interval (the mean number of days to mating) was increased in all SCH 351591-dosed groups. The mean time to mating was 10.8, 6.1, 8.0, and 10.2 days in the 100, 200, 400, and 800 mg/kg groups, respectively, as compared to 3.9 days in the control group. Again, this finding was attributed to abnormal estrous cycles in the females.
Summary of cohabitation data for SCH 351591-dosed males mated with dosed females.
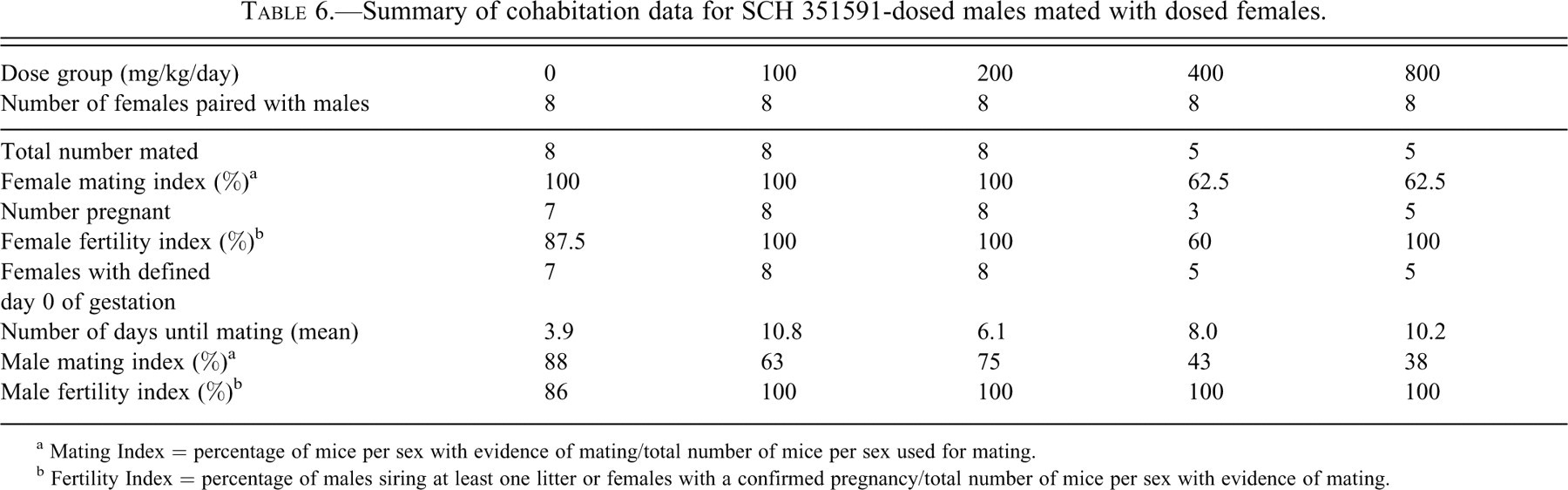
a Mating Index = percentage of mice per sex with evidence of mating/total number of mice per sex used for mating.
b Fertility Index = percentage of males siring at least one litter or females with a confirmed pregnancy/total number of mice per sex with evidence of mating.
Of the females in the 400 and 800 mg/kg groups that mated, three of five and five of five became pregnant, respectively, thus representing a female fertility index of 60% and 100%, respectively. The fertility index in the 100 and 200 mg/kg groups was 100% as compared to 87.5% in the control group, in which one of eight females did not become pregnant. This finding indicated that there was a SCH 351591-related effect on mating, but not on fertility in females (number pregnant/number mated × 100). There was no effect on male fertility. The male fertility index was 100% in all SCH 351591-dosed groups compared to 86% in controls.
Mating of Dosed Males with Untreated Females
The female mating index of the second cohabitation period (dosed males with untreated females) was 100% in each group, and the mean days to mating was comparable among all groups, varying from 2.8 to 4.6 days in the treated groups as compared to 3.3 days for control males (data not shown). The male mating indices were considered normal, varying between 75% and 88% in the SCH 351591-dosed groups as compared to 100% in controls. There was no effect on fertility. The male fertility indices ranged from 100% in the 100 mg/kg group to 83% in the 800 mg/kg group as compared to 62.5% in control males. The female fertility index followed a pattern comparable to that in males (data not shown).
Reproductive Parameters
Reproductive parameters for dosed females mated with dosed males are presented in Table 5. In the dosed females that were mated with dosed males, the number of implantations was comparable among all groups, and there were no dead fetuses in any of the groups. The mean number of CLs was increased in all SCH 351591-dosed groups relative to controls. The means were approximately eighteen in the 100, 200, and 400 mg/kg groups and twenty-four in the 800 mg/kg group as compared to fourteen in the controls. Two females in each of the 100 and 800 mg/kg groups and one female in the 200 mg/kg group had an exceptionally higher number of CLs (23–36 CLs/animal). This finding correlated with higher preimplantation losses (number of CLs minus number of implantation sites) in the 100 and 800 mg/kg groups than in the other dosed groups and controls.
There also appeared to be a test article–related but non-dose–related increase in fetal resorptions in all SCH 351591-dosed groups. Most resorptions occurred in early gestation. As a consequence, the mean numbers of live fetuses were lower relative to controls in all groups. Mean numbers of live fetuses were 10.1, 5.6, 10.0, and 5.0 in the 100, 200, 400, and 800 mg/kg groups, respectively, as compared to 11.8 in controls. There were only two pregnant females in the 400 mg/kg group, which may have contributed to a non-dose–related outcome in this parameter. The postimplantation losses reflected the increase in resorptions in all groups and varied from approximately 28% to 63% in the SCH 351591-dosed groups compared to 3.9% in controls. In the second cohabitation period (untreated females mated with dosed males), there were no adverse effects on the reproductive parameters (data not shown).
Discussion
Myocardial Toxicity
SCH 351591-dosed mice, which were otherwise highly resistant to the vascular toxicity of the drug as compared with rats and monkeys, developed a characteristically located cardiovascular lesion at the base of the major vessels of the heart. Pathologic changes affecting the cardiovascular system, although species-specific in location, have been a recurring issue with PDE inhibitors and a major deterrent to the development of this class of drugs. Theories have been proposed for the mechanism of toxicity, including mechanical injury from extreme vasodilatation of arteries and apoptosis and nitrative stress, either as a primary mechanism or secondary to the release of inflammatory chemokines from activated neutrophils, mast cells, and/or endothelial cells (Joseph et al. 1996; Lugnier and Komas 1993; Slim et al. 2003; Zhang et al. 2002; Zhang et al. 2008). Most of the proposed mechanisms were attributed to exaggerated pharmacology of the drugs, which resulted in local accumulation of cAMP and activation of inappropriate molecular signaling.
In other species, most notably rats (Zhang et al. 2008) and monkeys (Losco et al. 2004), the toxicity of PDE4 inhibitors has been closely linked to a primary vasculitis involving small to medium-sized muscular arteries, and any surrounding tissue involvement is considered secondary. Similar lesions, originating in the coronary arteries, have been reported for various vasoactive drugs in which the toxicity was attributed either to the pharmacologic effects of the compounds or to the local production of nitric oxide and pro-inflammatory chemokines (Jones et al. 2003; McDuffie et al. 2006; Mesfin et al. 1989). The characteristic lesions reported in time-sequenced investigative studies included medial hemorrhage and necrosis followed by inflammation, smooth muscle proliferation, and fibrosis (Mesfin et al. 1989).
The findings in mice have similarities, but they cannot be ascribed to a primary arteritis alone, even though a few mice did have localized coronary artery involvement. The most common site of toxicity in all affected mice—and the only site affected in animals with mild, presumably early, lesions—was the peri-aortic myocardium (Figure 2a). Heart lesions in mice, even if they initiated within the aorta, would be atypical as compared with those described for other drug-induced vascular toxicities because the aorta lacks a muscular media, often the primary site of drug-induced injury (Joseph et al. 1996; Mesfin et al. 1989). The aorta also contains abundant elastic fibers within a thick, resilient wall, making it less susceptible to damage from hemodynamic stress. Because the mildest and presumably, earliest finding was inflammatory cell infiltrates in the tissues adjacent to the aortic root, drug-induced local release of pro-inflammatory chemokines may be the initiator of the myocardial lesion in mice. Owing to the small sample size and the inability to observe the development of the lesions over time, it was not possible to classify the heart lesions in mice as either primary arteritis or myocarditis. The myocardial component is likely to be important, because there were no lesions of the aorta or pulmonary arteries at any distance from the myocardial outlet.
Current research that may be germane to the unique location of myocardial findings of mice centers around the mechanism by which cAMP, along with its substrates and degrading PDE enzymes, is localized to specific tissues and even to specific subcellular compartments. Cyclic adenosine monophosphate is a freely diffusible molecule that is the second messenger for many biologic processes, but it produces widely different molecular and cellular effects in various tissues. In the heart, cAMP, generated by β-adrenergic receptor stimulation, determines the strength, frequency, and duration of myocyte contraction. The localization of specific cAMP-mediated signaling occurs when the cAMP target enzyme, protein kinase-A (PKA) is compartmentalized within the cell via binding to A-kinase anchoring proteins (AKAPs) specific to subcellular microdomains (Zaccolo and Pozzan 2002). In cardiac myocytes, such anchoring proteins have been found discretely distributed throughout the T tubular system, where they are linked to β-adrenergic receptors in the plasma membranes (Conti et al. 2003; Jurevicius and Fischmeister 1996).
Equally important to modulating the localized cAMP concentration is the necessity of controlling the rate of its degradation by concentrating appropriate PDE enzymes in the microenvironment. Specific PDE isoforms have been selectively recruited and targeted to the plasma membrane sites of cAMP production via the same A-kinase anchoring proteins that are involved in localizing cAMP production (Feishmeister et al. 2006; Perry et al. 2002; Zaccolo and Pozzan 2002). For example, PDE4D has nine isoform variants with identical catalytic domains and carboxyl termini, but unique amino termini that are important for subcellular localization. One of these variants, PDE4D3, is of particular importance in the heart because it binds to the targeting protein mAKAP via its unique terminal portion, creating a signaling molecule that co-localizes with the ryanodine receptor (Ry/R2)/calcium release channel in cardiac muscle. PDE4D knockout mice develop progressive cardiomyopathy and arrhythmias that have been attributed to reduced PDE4D3, resulting in “leaky” RyR2-calcium channels that alter the strength and timing of myocyte contraction (Fischmeister et al. 2006; Lehnart et al. 2005).
It is not surprising that SCH 351591, which has equal specificity for all four PDE4 subtypes and presumably, for their multiple variants, would cause toxic changes in the heart. The localization of the lesions to the heart base and root of the major vessels is likely to be related to the specific PDE4 isoform variants that normally function at this site. The identification of the specific isoforms responsible for the heart lesion in mice has not been established, nor has the mechanism of toxicity for the lesion.
The differences in species sensitivity, whereby mice are up to 100 times less sensitive to the toxicity of SCH 351591 than rats or monkeys, is a point of interest (see Introduction: No-effect levels of 1 and 1.5 mg/kg/day for rats and monkeys, respectively, unpublished data, Schering Plough Research Institute). Researchers have studied the interspecies differences in PDE4 inhibitor toxicity (although mice were not included in the study). Bian et al. (2004) compared the mRNA expression of PDE4A, B, C, and D in blood leukocytes of rats, monkeys, and humans and noted significant differences demonstrating that rats > monkeys > humans. The differences corresponded to the potencies of several PDE4 inhibitors, including SCH 351591, at inhibiting tumor necrosis factor production by leukocytes. The PDE4 inhibitors evaluated were highly selective for the PDE4 class as opposed to other PDEs and were selected because they inhibited rat and human PDE4s with equal potency. It was concluded that, at least for rats and humans, the concentration of PDE4 in the tissues was the critical factor responsible for different effects between species. The low sensitivity of mice to the cardiac toxicity of SCH 351591, in comparison with other species, suggests that they are not the best choice of species for preclinical assessment of this drug class. However, the differences that make their toxicity unique are worthy of further investigation.
Reproductive System Toxicity
For female mice, SCH 351591 produced histopathologic and functional changes in the reproductive system that were consistent with the effects of a weak endocrine disrupter. Normal fertility was compromised for some animals, but successful pregnancies occurred in others. Most females appeared to be cycling, as determined from the histopathologic observation of developing follicles and CLs in the ovaries of the toxicity study mice as well as by vaginal swab assessments in the reproductive study. However, there was a dose-related incidence of altered estrous cycles that resulted in increased time to mating, fewer successful matings, and reduced fertility in treated females. In addition, females that became pregnant had smaller litters owing as a result of both preimplantation loss and increased early resorptions. Reduced fertility in the reproductive study was attributed solely to effects on females, as the mating of SCH 351591-dosed males to untreated females resulted in fertility and reproductive assessments comparable with the concurrent controls. The morphologic changes in females in the three-month study that correlated with reduced fertility in the reproductive toxicity study included a few cases of ovarian, uterine, and/or vaginal atrophy suggestive of insufficient hormonal stimulation and the more frequent changes of large, persistent CLs and prolonged or atypical diestrus, which is consistent with excessive or prolonged progesterone stimulation.
When regular estrous cycles are disrupted or prolonged by compounds affecting the hypothalamic-pituitary-ovarian (HPO) axis, the reproductive tissues can appear asynchronous. In their review of compounds such as endocrine disrupters that affect the HPO axis, Li and Davis (2007) described a variety of possible changes in the female reproductive tract. The uterus and vagina show the effects of the current dominating hormones, ranging from estrogen-stimulated hypertrophy to progesterone-dominated prolonged diestrus. Ovaries may contain follicular cysts or increased or hypertrophic CLs, or they may appear atrophied or inactive. The vagina may exhibit comparable changes of hyperkeratinization, increased mucification, or atrophy. The finding of vaginal atrophy associated with hypertrophy with mucification of the surface epithelium, as seen in some mice in this study, is characteristic of endocrine disruption in which progesterone stimulation dominates (Yuan 1991). Variability of findings among animals in the same treatment group is not unusual because the tissue appearance depends on the stage of cycling and dominant hormonal status of individual animals.
The mechanism by which a PDE4 inhibitor might produce hormonal perturbation in females is likely to be multifactorial because PDE4s are important players in molecular signaling in both the central nervous system (CNS) and in the ovary. Research involving the role of PDE4s in the CNS has demonstrated that cAMP stimulates release of gonadotropin-releasing hormone (GnRH) in the hypothalamus, which subsequently regulates the pituitary’s secretion of follicle-stimulating hormone (FSH) and luteinizing hormone (LH). Studies with isolated GnRH-secreting neuronal cell lines in culture have demonstrated the presence of multiple isoforms of PDE4B and D that act to reduce levels of cAMP and provide negative feedback to the secretory neurons (Sakakibara et al. 1998; Weiner and Charles 2001). Additionally, researchers have demonstrated reduced female fertility associated with the inhibition of LH secretion in transgenic rats that overexpress a specific PDE4 isoenzyme (PDE4D1) known to be active in the negative feedback of GnRH secretion (Paruthiyil et al. 2002).
PDE4s are also important players in the molecular signaling that controls ovarian follicle maturation and ovulation. Modulation of the concentration of cAMP within functional compartments is controlled, in part, by the rate of cAMP production and, in part, by its rate of degradation by PDEs (reviewed in Conti 2002). Specific PDEs, restricted to cell types or compartments of the ovary, control the steps that lead to successful ovum development and release. For example, PDE4D is expressed in the follicular mural granulosa cells. Its concentration increases and decreases in a specific cyclic pattern in response to gonadotropic stimulation. It is induced by FSH in preantral follicles, where it is associated with the proliferation of granulosa cells, and by LH in preovulatory follicles, where it is associated with the changes leading up to ovulation. The timing and intensity of the increase is directly correlated with hormonal levels and the functional results (Conti 2002). Mice deficient in PDE4D experienced a 70%–90% reduction in the rate of ovulation in response to human chorionic gonadotropin (HCG) stimulation (Jin et al. 1999).
As with the heart, the specificity of a physiologic response in reproductive tissues appears to be the consequence of controlling the local levels of cAMP. This control is accomplished, at least, in part, by compartmentalization of specific PDE4 isoforms. Because SCH 351591 is equally potent in inhibiting all four isoforms of PDE4s, it is likely that the hormonal perturbations observed in these studies are the result of altered cAMP levels at critical functional sites that interfered with the complicated molecular signaling that controls the estrous cycle, ovum maturation and release, and, ultimately, normal fertility.
As compared with females, the effect on the male reproductive system of SCH 351591 administration was less dramatic and not biologically significant. Male fertility was not impaired, and there were no discernable changes in testicular morphology. In both the three-month toxicity and the reproductive studies, findings were limited to an approximately 20%–25% increase in mean testes weights in SCH 351591-dosed mice as compared to controls. Because there were no apparent changes in cellularity of the testes, the most likely cause of the weight variation was believed to be an increase in fluid volume within the seminiferous tubules and/or the interstitium. A review of the literature supports the theory that inhibition of PDE4s might produce testicular fluid disturbances either via a CNS or local mechanism. SCH 351591-related effects at the level of the CNS would be similar to those discussed for females. PDE4-modulated cAMP stimulation of the hypothalamus is the driver, via GnRH, of both FSH and LH secretion by the pituitary. In the testes, LH stimulates the Leydig cells to secrete testosterone, and both testosterone and FSH act to promote spermatogenesis through direct stimulation of the Sertoli cells (O’Shaughnessy et al. 2009). Sertoli cells are the source of intratubular fluid that facilitates the movement of newly released sperm into the epididymis (Russell et al. 1990). They may also be involved in the maintenance of balance between intratubular and interstitial fluid. At the molecular level, Sertoli cells appear to function in fluid transport between the tubular lumina and interstitial space under the control of cAMP-modulated aquaporins (water ion channels) that are sensitive to FSH stimulation (Badran and Hermo 2002; Grover et al. 2004; Yang et al. 2003). Both PDE4B and PDE4D mRNA are present in Sertoli cells and are the likely modulators of cAMP degradation. Studies with PDE-specific antibody probes have demonstrated that PDE4D is the main form that is induced by FSH stimulation (Conti et al. 1995). Interstitial fluid volume is also affected by local capillary permeability, which may be under hormonal control and thus mediated by levels of cAMP. Injection of HCG into rats caused appreciable rises in capillary permeability and lymph flow in the testes, with resultant increases in interstitial fluid (Setchell and Sharpe 1981).
There were no significant microscopic differences in SCH 351591-administered mice as compared with controls in either seminiferous tubular luminal diameter or interstitial compartments in the toxicity study. Morphometric measurements of the volume fractions of the tissue failed to reveal subtle differences in proportion of the compartments. A difference in fluid content as compared with controls could still be the cause of the higher testes weights, but it would not be discernable if the excess fluid were evenly distributed between tissue compartments. Because the mechanisms associated with maintenance of both interstitial and tubular fluid are under the same influence of cAMP levels, a new equilibrium could have been reached in treated mice that involved slightly increased fluid volumes in both compartments. Because the morphometric evaluations were performed retrospectively on histologic slides, it was not possible to measure changes in actual tissue volume of the testes of treated and control mice.
The above discussion relates mainly to a presumptive increase in fluid production or compartmentalization within the testes as a cause of higher weights, but obstructed outflow of the efferent ducts and impaired resorption or obstruction in the epididymis were also considered as possibilities. The efferent ducts were not collected or examined in these studies, but the epididymides were weighed and examined histologically. There were no changes in organ weights or histologic appearance for this tissue, making it less likely that altered resorption or obstruction was at fault.
In summary, unpredictable toxicity at clinical doses has impaired the use of PDE4 inhibitors as therapeutic agents. The studies reported herein describe a unique variant in mice of the cardiovascular toxicity already attributed to this class of drugs in other laboratory species.
Additionally, we described effects on the reproductive system, particularly of females, that reveal the intricate role of PDE4 enzymes in modulating the estrous cycle. The molecular mechanisms by which PDE4 inhibition produces toxicity are yet to be determined. However, it is highly likely that the toxicities observed in different species will eventually be linked to molecular changes localized to specific cell and subcellular compartments. Current research has shown that the specificity of PDE function in nature is controlled in part by the evolution of many site- or tissue-specific isoforms, and in part by compartmentalization of active isoforms to certain subcellular locations via anchoring proteins. It is possible that further research into the natural mechanisms of PDE functional localization may yet reveal the means of limiting adverse tissue exposure and make this class of compounds beneficial as therapeutic agents.
Footnotes
Acknowledgments
The authors would like to acknowledge the technical assistance of K. Meredith for document preparation and K. B. Lee for photographic images.
