Abstract
Veterinary clinical pathologists are well positioned via education and training to assist in investigations of unexpected results or increased variation in clinical pathology data. Errors in testing and unexpected variability in clinical pathology data are sometimes referred to as “laboratory errors.” These alterations may occur in the preanalytical, analytical, or postanalytical phases of studies. Most of the errors or variability in clinical pathology data occur in the preanalytical or postanalytical phases. True analytical errors occur within the laboratory and are usually the result of operator or instrument error. Analytical errors are often ≤10% of all errors in diagnostic testing, and the frequency of these types of errors has decreased in the last decade. Analytical errors and increased data variability may result from instrument malfunctions, inability to follow proper procedures, undetected failures in quality control, sample misidentification, and/or test interference. This article (1) illustrates several different types of analytical errors and situations within laboratories that may result in increased variability in data, (2) provides recommendations regarding prevention of testing errors and techniques to control variation, and (3) provides a list of references that describe and advise how to deal with increased data variability.
Keywords
Veterinary clinical pathologists who work in the biopharmaceutical industry often have primary responsibilities that include description and interpretation of clinical and morphologic pathology data from animals used in drug safety studies, hazard identification, and risk assessment. These responsibilities may extend from early drug discovery studies to human clinical trials. In addition, veterinary clinical pathologists may serve as scientific resources within a company, provide diagnostic support, manage laboratories, and assist in evaluation of innovative biomarkers and new technologies in drug development (Schultze, Bounous, and Bolliger 2008). Veterinary clinical pathologists often work very closely with laboratory staff members to counsel colleagues and clients regarding unexpected laboratory test results. These discussions can be challenging and may involve exploration of causes for increased or undesirable variability and irregularities in the data used in drug development studies.
Suspected increased variability in clinical pathology data and diagnostic testing errors are often referred to using the general term of “laboratory error.” Diagnostic testing errors and practices that cause increased variability in clinical pathology data may occur during all stages of testing (preanalytical phase, analytical phase, and postanalytical phase; Moore and Barger 2016). Errors in the preanalytical phase occur prior to sample analysis and are generally external to the clinical pathology laboratory. In preanalytical errors, the analysis of the sample is appropriate, but the sample itself is not representative of the patient. Errors in the analytical phase occur within the laboratory during the testing process and are usually attributed to operator or instrument error. Errors in the analytical phase lead to inaccurate test results. Errors in the postanalytical phase of testing occur after the test is completed. In this instance, accurate test results may not trigger the appropriate patient management. Regardless of the phase of testing in which errors occur or practices result in increased data variability, they are undesirable and may have negative consequences such as harm to patients and increased costs of drug development (Moore and Barger 2016).
Results/Discussion
There have been numerous studies of the frequency of diagnostic testing errors in human medicine (Table 1). In general, the frequency of errors in the preanalytical phase and postanalytical phase comprise the vast majority (Bonini et al. 2002; Sonntag 2009). Errors within the analytical phase are usually few, and results indicated that the number of errors within the analytical phase has continued to decrease over the last decade (Plebani 2006, 2010; Sakyi et al. 2015).
Frequency of Diagnostic Testing Errors.
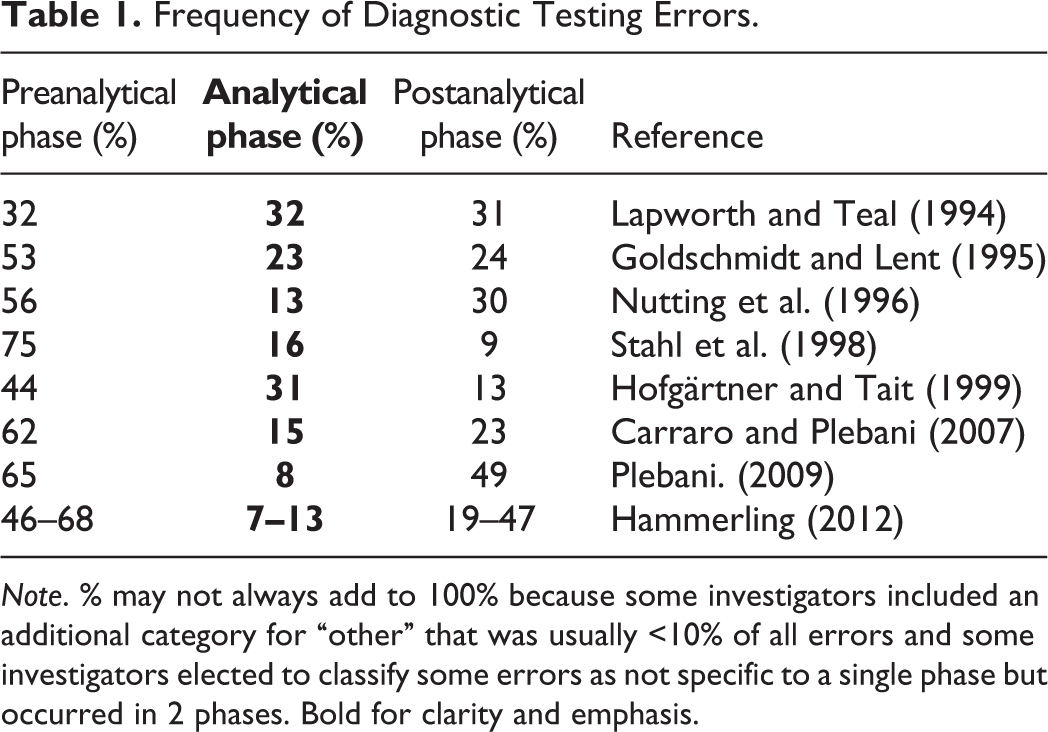
In contrast, studies of the frequency of diagnostic testing errors in veterinary medicine are few. Hooijberg, Leidinger, and Freeman (2012) evaluated a commercial veterinary clinical pathology laboratory during an 8-year period and determined that the annual error rate/total number of samples ranged from 0.7 to 1.3%. Error results were categorized as preanalytical (52 to 77%), analytical (4 to 14%), postanalytical (9 to 21%), and others (6 to 19%). As with studies in human medicine, Hooijberg and associates found the analytical category had the fewest errors.
The two major causes of analytical errors are instrument malfunction and operator error. Some examples of analytical errors include equipment malfunction, procedures (i.e., standard operating procedures and assay instructions) not followed, undetected failure of quality control, sample mixups, and test interference. In many studies of total testing errors, analytical errors are often <10% and their frequency is decreasing due to standardization of laboratory procedures and assays, automation within the clinical pathology laboratory, and advances in technology (Plebani 2006, 2010; Sakyi et al. 2015). However, in human medicine, immunoassays remain a prime concern for analytical errors due to the presence of heterophilic antibodies, antianimal antibodies, and anti-idiotype antibodies within patient sera. Analytical errors in immunoassays are a serious concern because they are often difficult to identify, produce serious errors, and may adversely affect patient care. (Plebani 2006, 2007, 2009). Comprehensive reviews of serious concerns with the use of immunoassays in veterinary medicine were not found. However, given the growing application of these types of assays within the clinical pathology laboratory and the numerous species used in safety assessment studies, one could hypothesize the potential for challenges of similar or greater magnitude when using this type of assay on various species used in drug development.
Case Examples
Example 1: Inability to Follow Proper Laboratory Procedures—Unexpected Delay in Sample Processing
The analytical phase of studies begins upon the receipt of the sample within the clinical pathology laboratory. Appropriate processing of the submitted sample in a timely manner is the responsibility of the laboratory staff. Blood collected into tubes without anticoagulant is allowed to clot and then spun in a centrifuge to obtain serum for clinical chemistry analysis. Delays in centrifugation and/or removal of the serum from the cells can result in alterations in the concentration of several analytes that are included in the minimal database used to assess the health of animals used in discovery, lead optimization, and good laboratory practice/regulatory safety assessment studies.
Blood from 3 healthy, sexually intact, male (2), and female (1) Collie dogs was collected without anticoagulant and submitted to the clinical pathology laboratory where it was allowed to remain unprocessed at room temperature for various times. Aliquots of blood were spun in a centrifuge, and serum was removed at 0.5, 1, 2, 4, 8, 16, or 24 hr after phlebotomy. Serum glucose concentrations were determined (Table 2). Notice the effect of glucose metabolism (glycolysis) by the erythrocytes, as the time prior to separation of the erythrocytes and serum increases. A common estimation is that erythrocytes will metabolize ∼10% of the glucose/hour if the sample is maintained at room temperature (Evans and Duncan 2003). Delayed processing of properly collected samples submitted to the clinical pathology laboratory for analysis due to negligence, overscheduling of laboratory personnel, or understaffing may lead to potential analytical error that results in inaccurate data and is preventable.
Serum Glucose Concentrations (mg/dl).

Example 2: Test Interference
A xenobiotic caused poorly dose-responsive increases in creatinine concentrations in rat and dog toxicity screening studies. The increases in creatinine concentration, which were reported in several studies of molecules within this chemical platform, occurred without increases urea nitrogen concentration or evidence of dehydration. The clinical pathology laboratory staff continued to report results of the creatinine assay and did not question the data. Results suggested test article–related positive interference with the creatinine assay to the client who approached the laboratory staff and inquired about methods to test for assay interference.
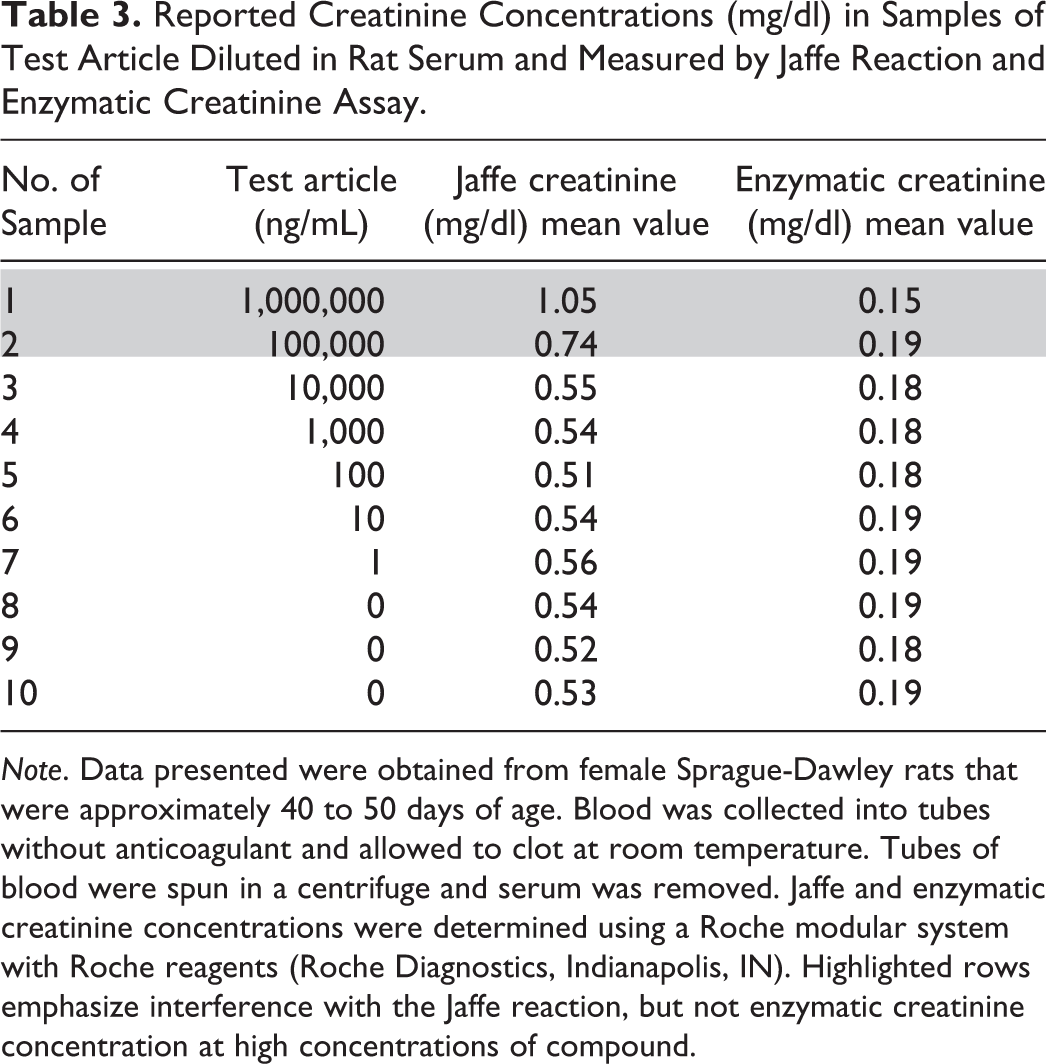
To investigate the possibility of test article–related positive interference with the creatinine concentration, clinical pathology laboratory staff created stock solutions of compound and made serial dilutions in rat serum. Creatinine concentrations were determined using two different methods: the Jaffe reaction and by an enzymatic creatinine reaction. Results indicated that at high concentrations, the test article had functional groups that reacted in the Jaffe reaction but not in the enzymatic creatinine assay. Reporting increases in creatinine concentration in the absence of other supportive data for azotemia or decreased renal function caused toxicologists to question the validity of the Jaffe reaction for this study.
Values for creatinine concentration highlighted in gray in Table 3 illustrate that at high concentrations, the compound caused positive interference using the Jaffe reaction. Creatinine concentrations measured using the enzymatic method did not show the positive interference and could be monitored in several preclinical species and in human clinical trials.
Reported Creatinine Concentrations (mg/dl) in Samples of Test Article Diluted in Rat Serum and Measured by Jaffe Reaction and Enzymatic Creatinine Assay.
In this example, the analytical error occurred when the clinical pathology laboratory staff members repeatedly noticed the increased creatinine concentrations (Jaffe method) in several studies but failed to question the creatinine assay results or interact with the client to investigate the lack of correlation of the creatinine value with the urea nitrogen concentration or clinical condition of the animals. Proactive communication with the client regarding the Jaffe method for creatinine measurement and its lack of specificity may have alleviated numerous questions and concerns. However, due to good postanalytical practice, the error was eventually detected, explored, and addressed sufficiently to provide accurate assessment of creatinine concentrations in study animals and to provide a path forward for continued drug development.
Example 3: Instrument Malfunction/Operator Error
Technicians in a clinical pathology laboratory at a contract research organization (CRO) analyzed blood samples from adult Sprague-Dawley rats using an ADVIA 120 hematology instrument (Siemens Medical Solutions, Malvern, PA). The xenobiotic in the study had no history of causing alterations in platelet and erythrocyte counts in any species evaluated in discovery and safety assessment studies. The sponsor questioned the results of the study due to perceived irregularities in the platelet and erythrocyte counts. The sponsor noted that the platelet counts varied >10-fold within some treatment groups and that numerous platelet counts were inconsistent with life (Table 4). There was also a lack of dose-responsive change in platelet and erythrocyte counts.
Platelet Counts in Male Rats Treated with a Xenobiotic for 4 Weeks.

In discussions, the sponsor requested complete hematology data review including evaluation of the ADVIA 120 erythrocyte and platelet cytograms from all rats in the study (Figure 1). A thorough investigation revealed abnormal platelet and erythrocyte cytograms that were correlated with a blood clot within the hematology instrument that resulted in abnormal values. The instrument operator was inexperienced and failed to notice the abnormal cytograms and continued with the analysis of blood samples to complete the study. Many erroneous test results were generated, as the clot moved in and out of the red cell channel within the ADVIA 120.
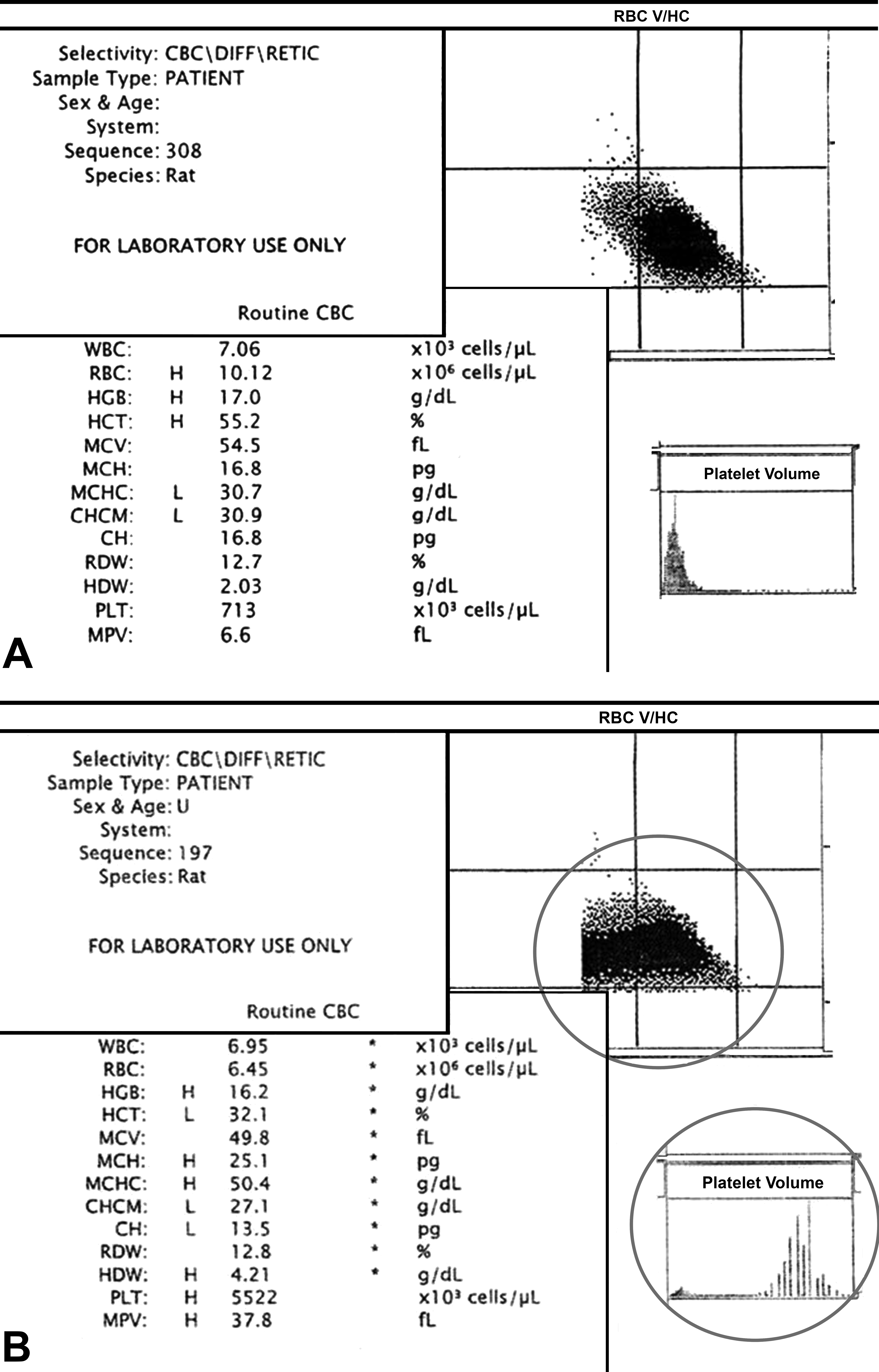
ADVIA 120 erythrocyte and platelet cytograms from a rat (A—normal cytogram) compared to those from a rat given a xenobiotic for 4 weeks (B—abnormal cytogram). Notice the clipped margins of the erythrocyte cytogram (upper red circle) and markedly altered platelet cytogram (lower red circle) that occurred secondary to a blood clot within the instrument.
Example 4—Clinical Pathology Staffing Qualifications
Prevention of diagnostic testing errors, regardless of the phase of study, is highly desirable and should be the goal of all. Quality begins with appropriate training of all laboratory staff members. Certification/accreditation by professional societies of pathology or laboratory medicine is highly desirable for employees. Consider the employees of 2 CROs (Table 5). The CROs are very similar. Each has been in business for the same period of time and each company has approximately the same number of employees. The clinical pathology laboratory at CRO A and CRO B are both charged with providing routine assessments in hematology, hemostasis, clinical chemistry, and urinalysis for rodents, dogs, and nonhuman primates used in safety assessment studies. The CROs are also asked to process a large volume of samples for drug adsorption and metabolism studies. The clinical pathology laboratories at the CROs operate for the same period of time daily and have approximately the same number of employees.
Clinical Pathology Employment Rosters for 2 Contract Research Organizations.
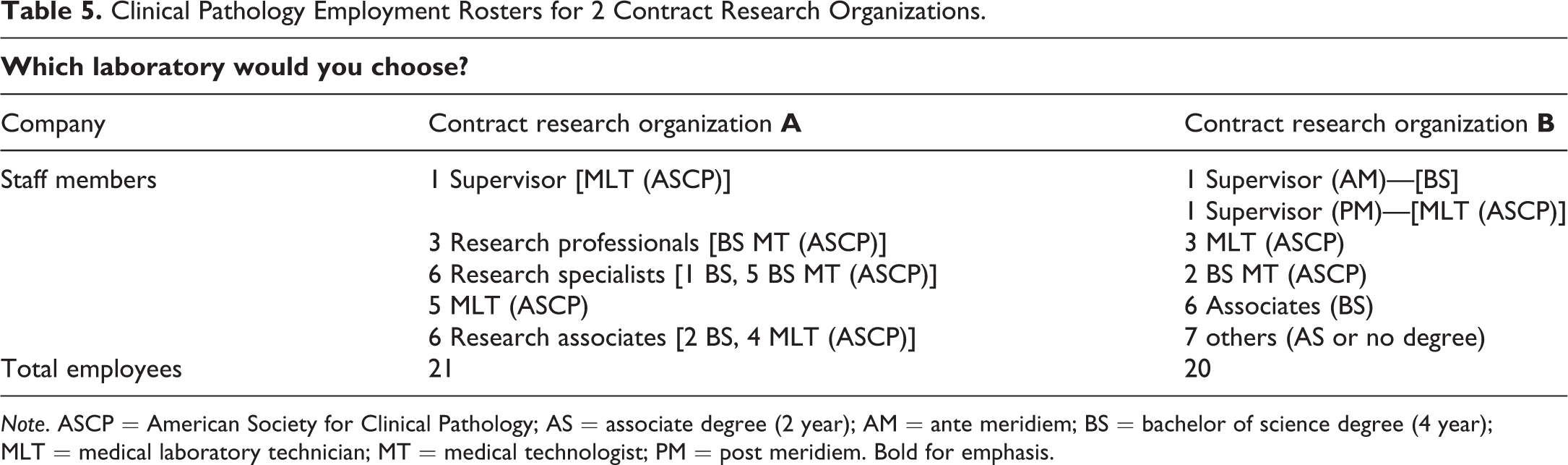
Employment titles for laboratory personnel vary widely between companies. Job titles such as research professional, research specialist, research associate, and associate can be confusing; are difficult to compare; and are often defined by different criteria at various companies. However, upon closer inspection, it becomes clear that the academic credentials for education and training of employees and the certification of laboratorians by the American Society for Clinical Pathology of the employees at CRO A are superior to that of employees at CRO B. Of the 21 clinical pathology laboratory personnel at CRO A, 18 have obtained certification as medical laboratory technician (MLT) or medical technologist (MT) by the American Society for Clinical Pathology (ASCP). In contrast, only 6 of the 20 clinical pathology laboratory employees at CRO B have been certified by the ASCP as MLT or MT. Several employees at CRO B have completed no college degree. Which clinical pathology laboratory would you choose?
Well-written procedures and study protocols are tools that will help to decrease the number of analytical errors and reduce unnecessary variation in clinical pathology data. Appropriate validation of laboratory instruments and procedures is essential for smooth operation of the laboratory and the generation of accurate data. Regular use of commercially available quality control measures in all areas of the clinical pathology laboratory is recommended. A program of regular instrument maintenance, service, and documentation is needed to assure quality service and continuous workflow. Most importantly, an awareness and willingness of the laboratory staff to monitor results for sources of error should be encouraged (Moore and Barger 2016). Guidance documents and informative websites for employees who work in laboratories that analyze animal samples can be difficult to locate. Most of the literature, guidance documents, and laws refer to laboratory practice for the analysis of human tissues and body fluids. The references listed in Table 6 can provide helpful information for those working in laboratories in which animal samples are analyzed.
Guidance Documents/Websites for Veterinary Laboratories.

It is the responsibility of all clinical pathology laboratory personnel to engage in behaviors and actions that decrease unnecessary data variability and to prevent or reduce analytical errors. McConnell, Nunnally, and McGarvey (2009) commented that laboratorians working in highly regulated industries at times confuse compliance with good quality laboratory work. Although validation studies and laboratory quality control programs are essential to daily work activities in the clinical pathology laboratory, these practices may underestimate analytical error. Validation studies are often conducted under pristine laboratory conditions, use the most highly qualified and/or experienced laboratory personnel, and involve a limited number of well-maintained instruments. Following completion of the validation study, there is the possibility for additional variation to occur in testing procedures due to a variety of events that may occur in most laboratories. Degradation of optimal validation conditions may occur as laboratories utilize analysts with lower skill levels, staff turnover occurs, batch changes of reagents occur, or equipment undergoes maintenance procedures. McConnell, Nunnally, and McGarvey (2009) recommended four practices to estimate analytical variation within the laboratory. Use of thorough and well-documented validation packages is a prime requirement of a quality laboratory. Specimen stability data are necessary to provide assurance of accurate results that are representative of the patient’s sample being analyzed. Routine use of commercially available quality control samples in all assays is an expectation. Use of “blind controls” (commercially available controls of known value that are submitted to the laboratory identified as a patient specimen) was suggested but acknowledged as a potentially helpful practice that is not in wide use.
In addition, safety assessment studies may pose some special problems to the clinical pathology laboratory, as these studies may involve multiple sample collections spread across lengthy periods of time (days, weeks, and months) as opposed to single finite determinations that occur with many diagnostic specimens. For best results in safety assessment studies, laboratorians are encouraged to avoid mid-study changes in reagent lots or test kits, delayed sample analysis, instrument recalibrations or complete instrument changes during a study, and technician shift changes mid sample batch analysis.
Conclusion
Diagnostic testing errors and increased data variability may occur in the preanalytical, analytical, or postanalytical phases of studies. True analytical errors are infrequent, occur within the clinical pathology laboratory, and are usually the result of operator or instrument error. Errors in the analytical phase are very important because they lead to inaccurate test results that may harm patients as well as increase the cost of business. The frequency of these types of errors has decreased due to automation, standardization of laboratory testing procedures, more strenuous educational and training requirements for laboratorians, and implementation of quality control practices. Instrument malfunctions, inability of employees to follow proper procedures, undetected failures in quality control procedures, sample misidentification within the laboratory, and/or test interference may cause analytical errors. All laboratory employees should strive to decrease unnecessary data variability and to prevent or reduce analytical errors. Well-written laboratory testing procedures, validation of laboratory instruments and assays, strong quality control programs, and proper education and training of laboratory professionals are practices that will decrease analytical errors and reduce data variability.
Footnotes
Authors’ Note
This article summarizes data from an oral presentation entitled “Sources of Variation in Clinical Pathology Data—Analytical Considerations” that was given at the Society of Toxicologic Pathology 35th Annual Scientific Symposium held in June 2016, in San Diego, CA. The oral presentation by Drs. Schultze and Irizarry was one of the four given in a special session entitled “Deciphering Sources of Variability in Clinical Pathology—It’s Not Just about the Numbers.”
Authors' Contribution
All authors (AS, AI) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
