Abstract
Inhibition of the mitogen-activated protein kinase/extracellular signal-regulated (MAPK/ERK) pathway is an attractive therapeutic approach for human cancer therapy. In the course of evaluating structurally distinct small molecule inhibitors that target mitogen-activated protein kinase kinase (MEK) and ERK kinases in this pathway, we observed an unusual, dose-related increase in the incidence of green serum in preclinical safety studies in rats. Having ruled out changes in bilirubin metabolism, we demonstrated a 2- to 3-fold increase in serum ceruloplasmin levels, likely accounting for the observed green color. This was not associated with an increase in α-2-macroglobulin, the major acute phase protein in rats, indicating that ceruloplasmin levels increased independently of an inflammatory response. Elevated serum ceruloplasmin was also not correlated with changes in total hepatic copper, adverse clinical signs, or pathology findings indicative of copper toxicity, therefore discounting copper overload as the etiology. Both ERK and MEK inhibitors led to increased ceruloplasmin secretion in rat primary hepatocyte cultures in vitro, and this increase was associated with activation of the Forkhead box, class O1 (FOXO1) transcription factor. In conclusion, increased serum ceruloplasmin induced by MEK and ERK inhibition is due to increased synthesis by hepatocytes from FOXO1 activation and results in the nonadverse development of green serum in rats.
Keywords
Introduction
Drug development has evolved from an era of untargeted drugs, characterized by uncertain mechanism of action and compound-related off-target effects to targeted therapies with selective mechanisms of action. Elucidation of structure-activity and structure-toxicity relationships provides an avenue to define and constrain compound-related effects to the pathway of interest and improve safety profiles. Much effort in early-stage drug development is now expended on achieving a more complete mechanistic understanding of on-target toxicity liabilities to improve risk assessment and better predict species translatability in terms of toxicity and patient safety.
Hyperactivity of the mitogen-activated protein kinase/extracellular signal-regulated (MAPK/ERK) pathway is highly prevalent in human cancer. Pro-oncogenic driver mutations in the upstream membrane receptor tyrosine kinases (e.g., epithelial growth factor receptor) and the signaling molecules, Kirsten Rat Sarcoma Viral Oncogene Homolog (KRAS) and B-Raf Proto-Oncogene, Serine/Threonine Kinase (BRAF), are responsible for dysregulation of the MAPK pathway leading to tumor induction and progression (Burotto et al. 2014). MEK1/2 and ERK1/2 are positioned distally on the MAPK pathway and represent attractive molecular targets for cancer therapy. In early preclinical toxicity studies of small molecule ERK inhibitors in rats, we made the disconcerting observation that higher doses were associated with green serum. Initial investigations indicated this observation was related to an increase in serum ceruloplasmin concentration. Ceruloplasmin is the major copper-carrying protein in the blood, which translates to “a blue substance from plasma” (Vashchenko and MacGillivray 2013). Holoceruloplasmin (the copper-loaded form) contains 6 copper centers and thus acquires the blue color of copper. Approximately 95% of the copper in circulation is bound by ceruloplasmin and 5% by albumin and di-histidine containing complexes (including transcuprein and coagulation factors V and VIII; Danzeisen et al. 2007). High concentration of ceruloplasmin in solution with straw-colored serum may therefore impart a green hue to the serum.
Ceruloplasmin (EC 1.16.3.1) is a member of the multicopper oxidase family, which includes hephaestin and zyklopen. A beneficial consequence of ceruloplasmin oxidase activity is the detoxification of reactive ions aiding in cytoprotection. Ceruloplasmin is found to oxidize the cupric ion (Cu2+) and also transport copper in the blood, although the physiological importance of ceruloplasmin in copper transport is still the subject of conjecture. Ceruloplasmin is also a ferrioxidase, converting the toxic ferrous (Fe2+) to nontoxic ferric ion (Fe3+) for subsequent export in transferrin. By forming macromolecular complexes with the iron-containing inflammatory proteins lactoferrin and myeloperoxidase, ceruloplasmin ameliorates potentially deleterious iron-related oxidation reactions. In addition to oxidizing copper and iron to less toxic forms, ceruloplasmin enhances the oxidation of nitric oxide to nitrite, a protective mechanism against ischemic stress (Shiva et al. 2006). Ceruloplasmin is also a component of innate immunity, and the liver increases synthesis of ceruloplasmin as part of the interferon-γ and interleukin 1 (IL1)-β-induced acute phase response to inflammation and infection (Vashchenko and MacGillivray 2013).
Rat ceruloplasmin has 93% amino acid identity to the human protein, with concordant tissue expression patterns. The liver is the major site of ceruloplasmin synthesis, with minor expression noted in spleen, lung, testis, and brain (Fleming and Gitlin 1990; Hellman and Gitlin 2002). The majority of ceruloplasmin in circulation is the holoenzyme form (i.e., copper loaded; Hellman and Gitlin 2002). In humans, serum ceruloplasmin levels are decreased in aceruloplasminemia and Wilson’s disease (a genetic copper storage disease of the liver), and hepatic synthesis is increased in conditions of iron deficiency, oxidative stress, acute phase response, and hypoxia (Vashchenko and MacGillivray 2013). Ceruloplasmin is also increased by estrogenic drugs and oral contraceptives and during pregnancy (Tovey and Lathe 1968; Carruthers, Hobbs, and Warren 1966).
Despite being the major copper carrier in circulation, ceruloplasmin blood concentration is not an indicator of increased whole body copper status (Danzeisen et al. 2007), and blood copper levels are more reflective of ceruloplasmin regulation than of whole body copper stores.
The following describes mechanistic studies carried out to characterize the relationship of the MEK-ERK pathway with increased ceruloplasmin synthesis and the risk assessment of these findings.
Materials and Methods
Animal Studies
Rat studies with ERKi GNE-470
Study 1 in male rats
This study was a pilot toxicity study to determine the toxicity and toxicokinetics of a lead ERK inhibitor in male rats. Six male Sprague-Dawley rats per dose group were dosed by oral gavage once daily for 14 days, with a small molecule inhibitor of ERK (ERKi) at 0, 50-, 150-, or 450-mg/kg GNE-470, formulated as an aqueous suspension in 1.25% cab-o-sil/0.25% methyl cellulose/0.01% Tween 80 aqueous suspension in a dosing volume of 10 ml/kg. Two rats in the 450-mg/kg dosage group were found dead on study day 8 or 9, and the samples could not be obtained. The remaining 4 rats from the same group were necropsied on day 9 due to poor clinical condition. All other animals were fasted on day 14 and underwent terminal necropsy 24 hr after the last dose on day 15. Blood was collected into serum separator tubes at necropsy via retro-orbital bleed under isoflurane anesthesia for routine clinical pathology, ceruloplasmin analysis, and α-2 macroglobulin analysis. Tissues (bone sternum, brain, heart, stomach, small and large intestines, kidneys, liver, lungs, skin, spleen, and thymus) were collected and histopathologically evaluated by an American College of Veterinary Pathology-boarded pathologist. Liver tissues were also collected for liver copper analysis.
Study 2 in male and female rats
This study was an investigative study to determine the kinetics and gender pattern of ceruloplasmin increase for a lead ERK inhibitor in male and female rats. Four male and 4 female Sprague-Dawley rats were dosed by oral gavage once daily for 14 days with 150 mg/kg of GNE-470 formulated as an aqueous suspension in 1.25% cab-o-sil/0.25% methyl cellulose/0.01% Tween 80 aqueous suspension in a dosing volume of 10 ml/kg. Blood for ceruloplasmin analysis was collected into serum separator tubes via tail-vein bleed prior to dosing on day 1 (baseline), day 8 (after 7 daily doses), and day 15 (14 daily doses).
Rat studies with MEKi GNE-573
This study was an investigative study to determine whether an MEK inhibitor would increase ceruloplasmin levels in male rats, to demonstrate pathway-related effects. Four or five male Sprague-Dawley rats per group were dosed by oral gavage once daily for 7 days at 0, 20, or 60 mg/kg of GNE-573, a small molecule inhibitor of MEK (MEKi), formulated in hydroxypropyl-methylcellulose, in a dosing volume of 10 ml/kg.
Blood was collected into serum-separator tubes on study day 0 (predose) via the tail vein and on study days 4 and 8 from unfasted animals via the retro-orbital sinus under isoflurane anesthesia for routine serum chemistry and ceruloplasmin analysis. Histopathological examination was conducted on the kidneys only.
All studies were conducted according to a written study protocol and approved by an Institutional Animal Care and Use Committee in compliance with national regulations on animal welfare standards.
Serum Ceruloplasmin and α-2 Macroglobulin Analysis
Serum samples were collected from rats treated orally with vehicle or GNE-470 at 50 or 150 mg/kg for 14 days or 450 mg/kg for 9 days (n = 6, 6, 5, and 4, respectively) and from rats treated orally with vehicle or GNE-573 at 20 or 60 mg/kg for 7 days (n = 4 and 5, respectively) and were analyzed for ceruloplasmin and/or α-2 macroglobulin using rat-specific ELISA (Kamiya Biomedical, Seattle, Washington) kits. Serum samples were analyzed in duplicate following the manufacturer’s instructions. Values are expressed as mean ± SD.
Clinical pathology
Serum for clinical chemistry analysis was analyzed on a Cobas Integra 400 (Roche diagnostics, Indianapolis, IN), and blood for hematological analysis was analyzed on a Sysmex XT 2000iV (Sysmex America, Inc. Mundelein, IL).
Plasma and Liver Copper Analysis
Liver tissues were weighed on a 4-place balance into a clean Teflon microwave digestion vessel, and a small amount of purified water was used to rinse down the interior of the vessel. Approximately 3 ml of ultrapure nitric acid was added to the vessels, which were then capped and placed in the microwave for complete tissue digestion using a combination of heat and pressure. The vessels were cooled, and the contents were transferred into a final volume of 50 ml. The diluted digests and plasma samples were then analyzed by inductively coupled plasma mass spectrometry against standards of known concentration.
Western Blot of Liver for Adenosine Triphosphate7B (ATP7B) Expression
Western blot analysis of ATP7B was performed as described previously (Pai et al. 2008). In brief, snap frozen liver tissues from rats (n = 4) treated orally for 14 days either with vehicle or GNE-470 at 50 and 150 mg/kg were homogenized in modified Radioimmunoprecipitation assay buffer (RIPA) containing protease and phosphatase inhibitors (Roche Diagnostics, Sigma-Aldrich, Thermo Scientifics, IL) and clarified by centrifugation. Total protein concentrations of the lysates were determined using bicinchoninic acid assay (Pierce, Rockford, IL) and equal amounts of protein (50 μg) were resolved on sodium dodecyl sulfate polyacrylamide gel electrophoresis transferred to nitrocellulose membranes and incubated with ATP7B-specific primary antibody (Novus Biologicals, Littleton, CO). After washing and incubating with secondary antibody, immunoreactive protein was visualized by the Enhanced Chemiluminescence (ECL) detection system (GE Healthcare, Buckinghamshire, United Kingdom) and imaged using GeneGnome XRQ imaging system (Syngene, Frederick, MD).
In Vitro Ceruloplasmin Analysis
Freshly isolated and plated rat hepatocytes (cat No. RTFN-96) were obtained from CellzDirect (Carlsbad, CA) and treated in triplicate with either vehicle (dimethyl sulfoxide [DMSO]) or varying concentrations of GNE-470 and GNE-573 (3.125-100 µM). Culture supernatants were collected after 24 and 48 hr of incubation and utilized for ceruloplasmin analysis. Cytotoxicity was assessed after 48 hr of incubation using the Cell TiterGlo assay (Promega, Madison, WI). Values are presented as mean percentage of control ± SD. Undiluted ceruloplasmin levels in cell supernatants were assessed in duplicate, using a commercially available ELISA kit (cat # ERC4101-1, lot # 011131110; AssayPro, St. Charles, MO, USA) following the manufacturer’s instructions. Values are presented as mean percentage of control ± SD from experiments performed with 4 replicates.
Forkhead Box, Class O1 (FOXO1) Reporter Assays
FOXO1 transcriptional activity was evaluated using a commercially available dual luciferase reporter assay kit following the manufacturer’s instruction (SA Biosciences, Valencia, CA, USA). In brief, freshly isolated and plated rat primary hepatocytes were obtained from CellzDirect (USA) and transiently transfected with (1) FOXO1 reporter containing a mixture of inducible FOXO1 transcription-factor-responsive construct and constitutively expressing Renilla luciferase construct (40:1), (2) negative control containing a mixture of noninducible firefly luciferase reporter and constitutively expressing Renilla luciferase construct (40:1), and (3) positive control containing a mixture of constitutively expressing green fluorescent protein (GFP) construct, constitutively expressing firefly luciferase construct, and constitutively expressing Renilla luciferase construct (40:1:1), using LipofectAMINE-2000 reagent according to the manufacturer’s instructions (Invitrogen, Grand Island, NY). After 24 hr transfection, cells were treated either with GNE-470 or GNE-573 for 4 and 24 hr. Luciferase activity was measured using the dual luciferase assay system (Promega, Madison, WI). Luciferase counts were normalized using Renilla luciferase transfection control, and mean relative activity was calculated against vehicle control. Values are presented as mean percentage of control ± standard error of the mean (SEM) from two separate experiments performed with 4 replicates for each condition.
Statistical Analysis
For in vitro tests and copper analyses, the Student’s two-tailed t-test with Welch’s correction was used to compare data between two groups. p Values of <.05 were considered statistically significant.
For clinical pathology analyses, treated groups were compared to the control group at the same time point using a one-way analysis of variance and Dunnett’s test as the post hoc test for significance at p <.05.
Results
MEK and ERK Inhibitors Increased Serum Ceruloplasmin in Rats
Treatment with oral ERKi GNE-470 (Figure 1A) was associated with the development of green serum in male rats (Figure 2). This observation was made in 4 of the 5 samples from rats given 150 mg/kg for 14 days and in 2 of the 4 rats given 450 mg/kg for 9 days (the limit of tolerability). Serum chemistry analysis of rats dosed with GNE-470 included a dose-dependent increase in inorganic phosphorus concentration consistent with the on-target inhibition of Fibroblast Growth Factor (FGF)-23 signaling in the kidney which results in an increase in activated Vitamin D and enhanced phosphorus absorption in the intestine (Diaz et al. 2012). There was also increased alanine aminotransferase (ALT) activity at ≥150 mg/kg. The absence of any change in total bilirubin concentration (Tables 1 and 2) made a differential diagnosis of hyperbiliverdininemia (green jaundice) as a potential cause for green serum, unlikely. An alternative consideration for development of green serum is elevated serum concentration of ceruloplasmin, a copper-carrying acute phase protein. GNE-470 caused an approximately 3-fold elevation of serum ceruloplasmin concentration in male rats dosed with 150 mg/kg for 14 days and an approximately 4-fold elevation in rats dosed with 450 mg/kg for 9 days (Figure 3A). Ceruloplasmin was also elevated in serum that was not visibly green, indicating that serum color was a less sensitive indicator than direct measurement of ceruloplasmin (Supplemental Figure 1). Ceruloplasmin is an acute phase protein and could therefore be elevated secondary to inflammation. α-2-macroglobulin, the major acute phase protein in rats, was measured to determine whether an acute phase response alone was driving the increase in ceruloplasmin. α-2-macroglobulin was increased only at 450 mg/kg (Figure 3B) and also did not correspond with the presence of green serum in individual samples (Supplemental Figure 1). Serum albumin and globulin are also acute phase proteins (reduced and increased, respectively, during an acute phase response). We measured a marked dose-dependent decrease in albumin and a mild increase in serum globulins at 150 to 450 mg/kg (Table 1), which supported an acute phase response at these dose levels.
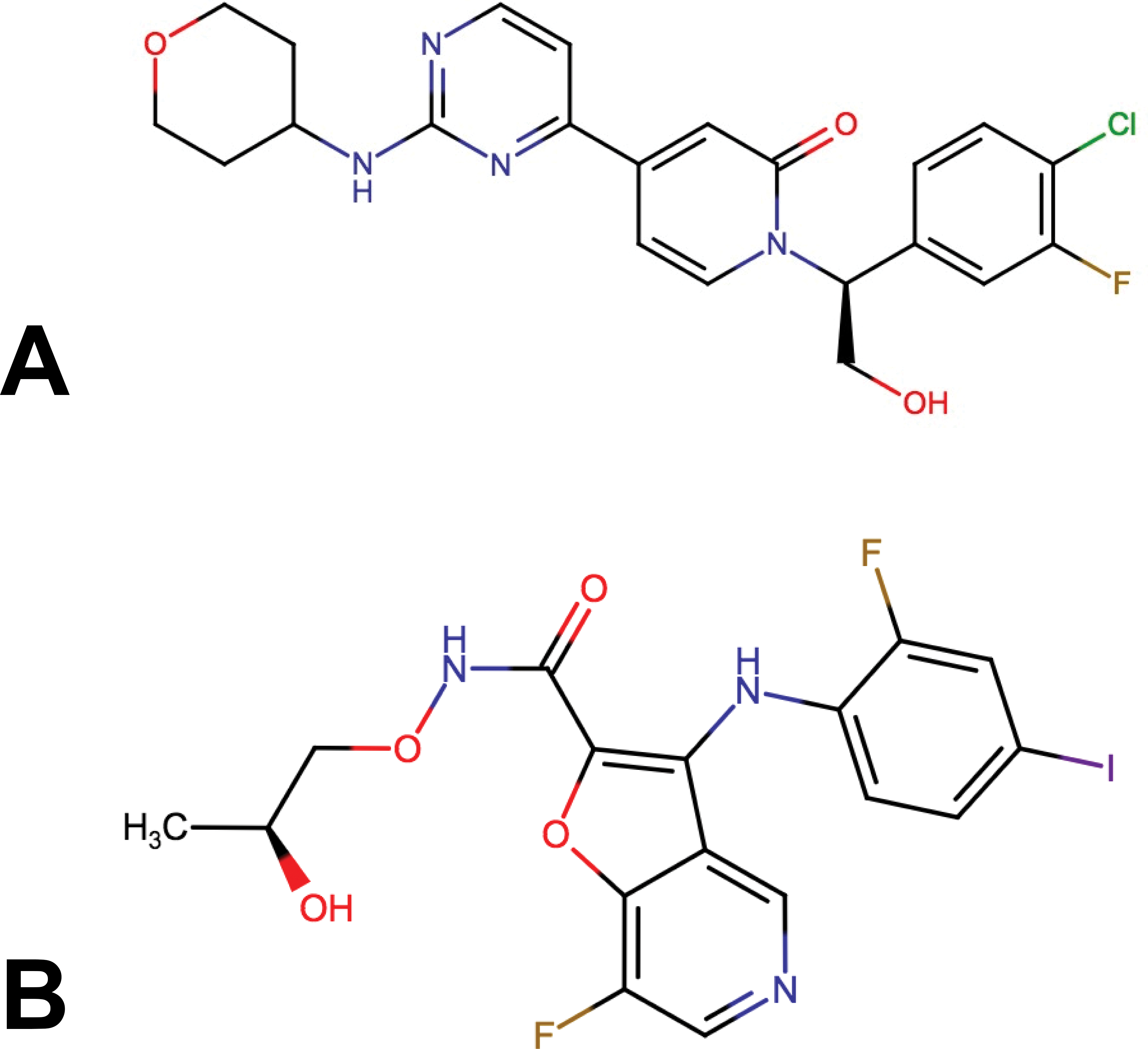
Chemical structures of ERKi GNE-470 (A) and MEKi GNE-573 (B).

Green serum (on right) caused by ERKi GNE-470 administration in rats. (Samples were frozen; therefore, the normal colored serum on the left appears opaque.)
Clinical Pathology Panel for GNE-470.
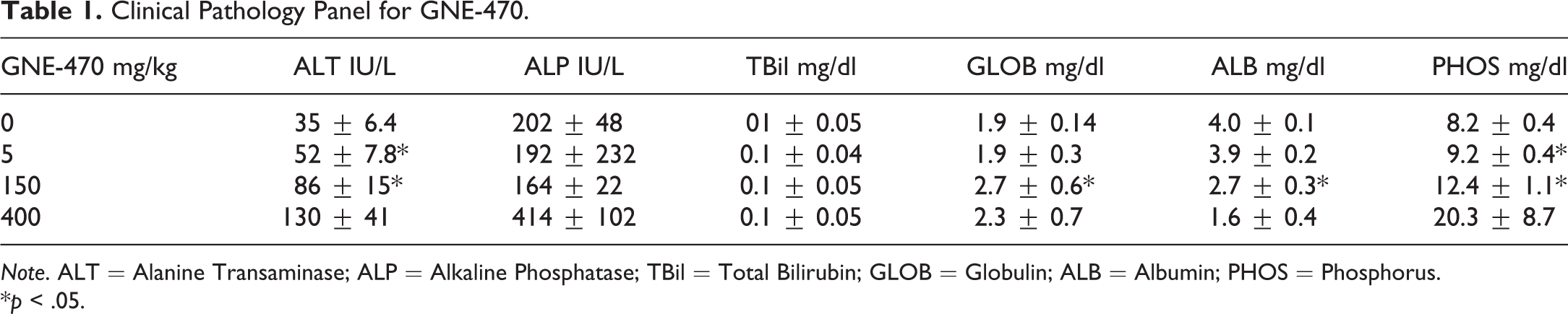
Note. ALT = Alanine Transaminase; ALP = Alkaline Phosphatase; TBil = Total Bilirubin; GLOB = Globulin; ALB = Albumin; PHOS = Phosphorus.
*p < .05.
Clinical Pathology Panel for GNE-573.
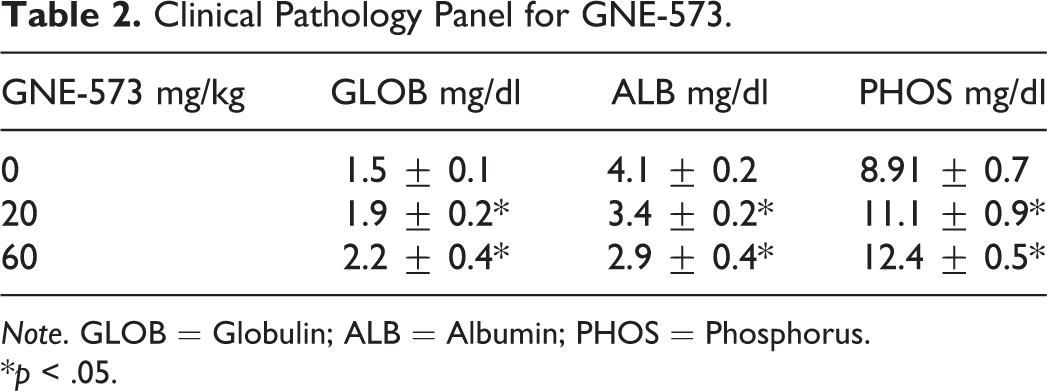
Note. GLOB = Globulin; ALB = Albumin; PHOS = Phosphorus.
*p < .05.
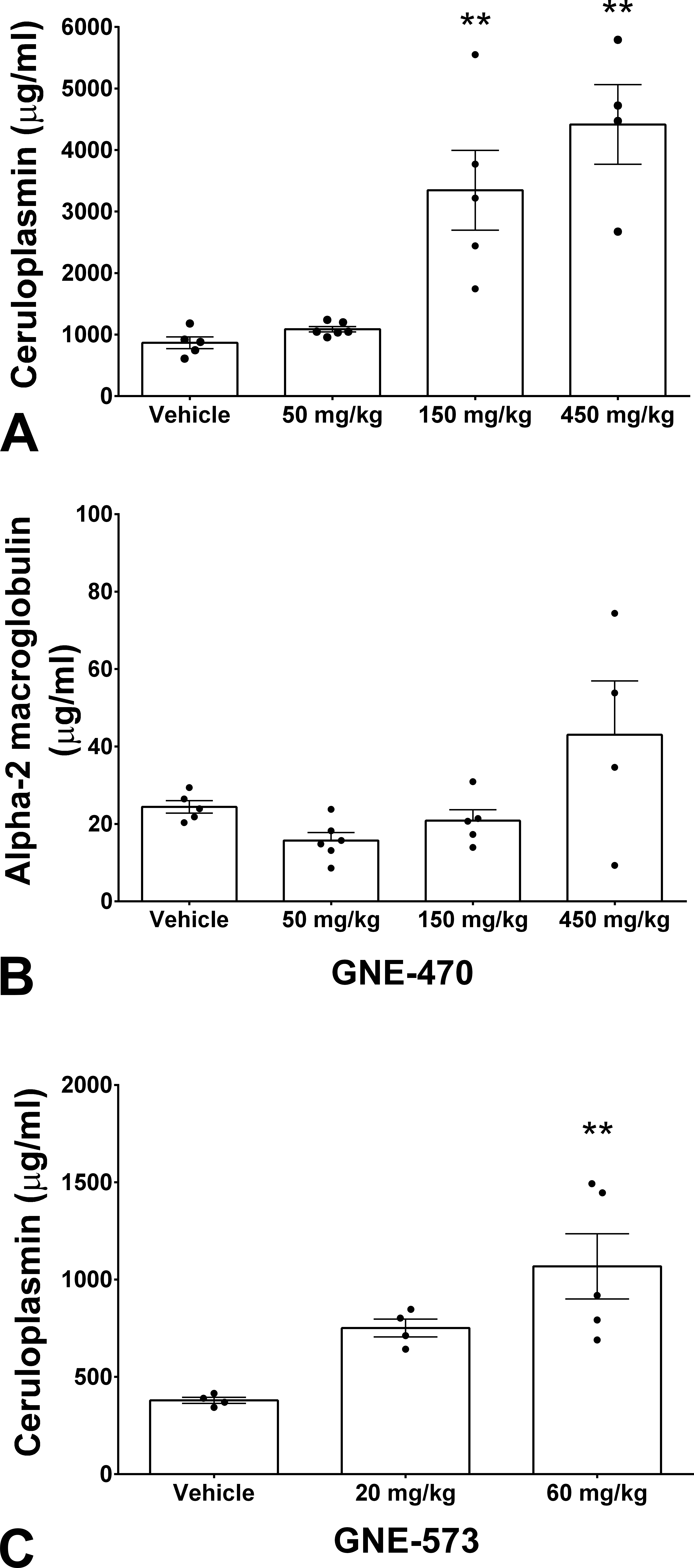
ERKi GNE-470 caused increased serum ceruloplasmin levels when dosed daily to male rats for 8 days at 450 mg/kg or 14 days at 150 mg/kg (A). Elevation in α-2-macroglobulin levels was not observed at 150 mg/kg but only at 450 mg/kg (B). MEKi GNE-573 also caused elevated ceruloplasmin levels when dosed daily to male rats at 20 and 60 mg/kg for 7 days (C).
Widespread, mild to moderate-grade inflammatory histopathologic changes were present in the gastrointestinal tract of rats only at the untolerated dose of 450-mg/kg/day GNE-470. The stomach and intestines displayed epithelial necrosis/apoptosis and areas of mucosal erosion and ulceration with accompanying inflammatory cell infiltrate. There was a low incidence of inflammation in the skin (2 rats at 150 and 1 rat at 450 mg/kg/day of GNE-470). Skin findings included areas of ulceration and attendant mixed inflammatory cell infiltrate, with a dose-dependent, minimal-moderate grade severity. Two rats in the 450-mg/kg group had hepatic periportal inflammation or necrosis, likely secondary to bacterial access to portal circulation as a result of gastrointestinal epithelial necrosis and breakdown of the mucosal barrier.
The MEKi GNE-573 (Figure 1B) also caused an increase in serum ceruloplasmin concentration in male rats dosed at 20 mg/kg and 60 mg/kg for 7 days (1.5-fold and 2-fold, respectively, Figure 3C). A clinical pathology panel showed a dose-dependent marked increase in phosphorus, reflecting on-target pharmacology, minimally increased serum globulin, and moderately decreased serum albumin (Tables 1 and 2). Green serum was not observed in this study, likely due to lower ceruloplasmin levels.
A longitudinal study was undertaken to explore the effects of the ERKi GNE-470 on serum ceruloplasmin and extended to male and female rats. GNE-470 caused a time-related elevation of serum ceruloplasmin in both male and female rats dosed daily at 150 mg/kg, increasing from 2-fold at day 7 to 3-fold by day 14 of daily oral dosing (Figure 4). Interestingly, female rats had a ceruloplasmin baseline approximately 2-fold higher than male rats, but the relative elevation caused by GNE-470 was of similar magnitude in both sexes.
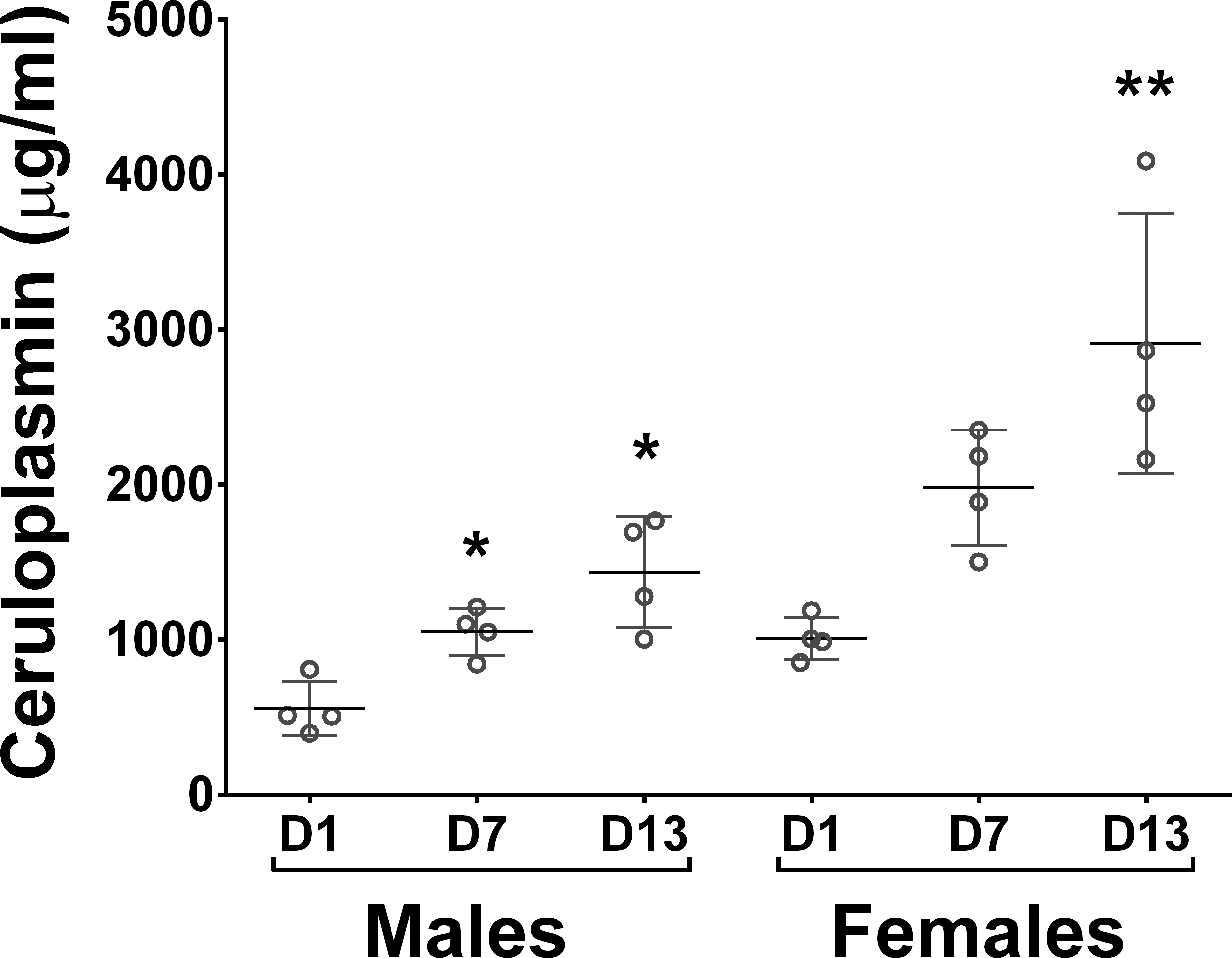
Time- and gender-related effects of GNE-470 on serum ceruloplasmin levels when dosed daily to male and female rats at 150 mg/kg for 14 days.
Total plasma copper concentration (ceruloplasmin and nonceruloplasmin-bound copper) was elevated in male rats treated with GNE-470 (Figure 5A). Total copper levels shared a strong correlation (R 2 = .89)) with serum ceruloplasmin levels (Figure 5B), indicating that the elevation in plasma copper levels was strongly associated with ceruloplasmin and therefore likely to represent ceruloplasmin-bound copper rather than nonceruloplasmin-bound copper (i.e., bound to albumin). Liver copper concentration was not elevated in rats dosed with GNE-470 (Figure 5C), indicating normal copper homeostasis in the liver. This was consistent with the absence of histopathological findings suggestive of hepatic copper dysregulation and toxicity (such as widespread hepatic inflammation and associated hepatocellular degeneration) in rats at tolerated doses with green serum. Liver copper concentration did not correlate with plasma copper (Supplemental Figure 2), signifying, as expected, that plasma copper is a poor indicator of total body copper levels.
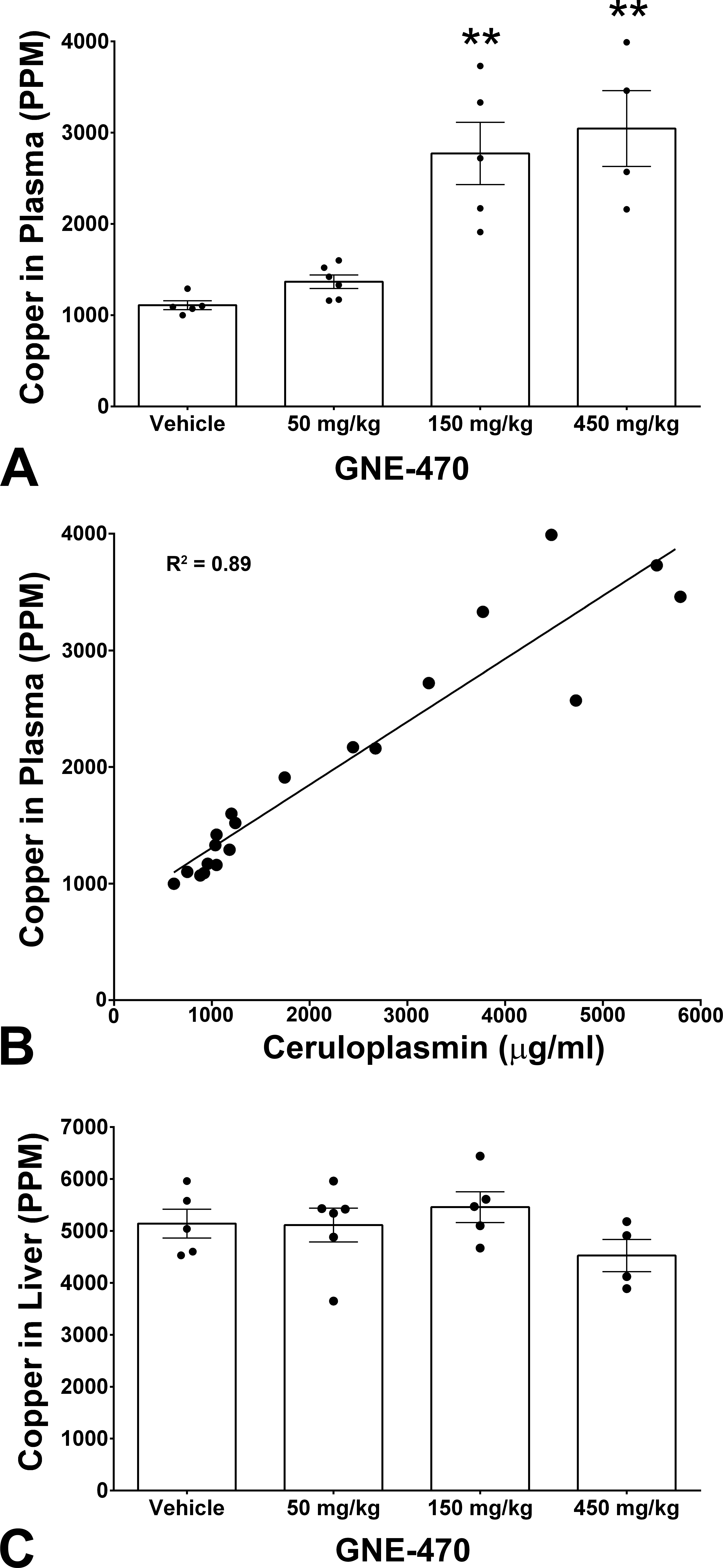
Total copper plasma levels were elevated in serum of rats dosed daily with GNE-470 for 8 days at 450 mg/kg or 14 days at 150 mg/kg (A); data represent mean ± SD of n = 6 male rats (150 mg/kg) or n = 4 male rats (450 mg/kg). Serum levels of total copper were strongly correlated (R 2 = .89) with serum ceruloplasmin levels in the same study (B). Liver copper levels were not detectably elevated (C).
PKD Off-target Activity Is Not Related to Ceruloplasmin Elevation
GNE-470 had off-target activity on protein kinase D (PKD) in an in vitro kinase-binding panel with 66.2% binding at 0.1 µM (data not shown). This off-target activity could be biologically relevant, given that plasma-free drug levels achieved in rats at 150 mg/kg was approximately 0.4 µM over 12 hr. PKD phosphorylates and stabilizes ATP7B, also known as the Wilson’s copper ATPase, an enzyme that incorporates copper into ceruloplasmin. Dysfunction of this enzyme in humans can lead to copper accumulation in the body as observed in Wilson’s disease (Lutsenko et al. 2007; Pilankatta et al. 2011). PKD enzyme inhibition leads to destabilization of ATP7B and therefore could reduce levels of the ATP7B protein and decreased ceruloplasmin levels. However, we reasoned that this pathway could be relevant due to demonstrated drug activity in vitro and the potential for feedback mechanisms to reverse the anticipated pharmacodynamic effects. Western blot analysis of liver samples from rats dosed with GNE-470 at 50 and 150 mg/kg showed no effects on the levels of ATP7B (Supplemental Figure 3), indicating that this protein was not modulated by the drug and an off-target hit was unlikely to account for the observed effects on ceruloplasmin.
MEKi and ERKi Increase Ceruloplasmin Levels and FOXO1 Transcriptional Activity in Primary Rat Hepatocytes In Vitro
To explore the molecular mechanism leading to an MAP kinase inhibition–related increase in ceruloplasmin, we treated primary rat hepatocytes with GNE-470 and GNE-573 for 24 or 48 hr. Both ERKi and MEKi caused an increase in ceruloplasmin levels in rat hepatocytes after 24 (Figure 6A) and 48 hr of treatment (data not shown). This increase in ceruloplasmin was not associated with cytotoxicity as determined by ATP release (data not shown).
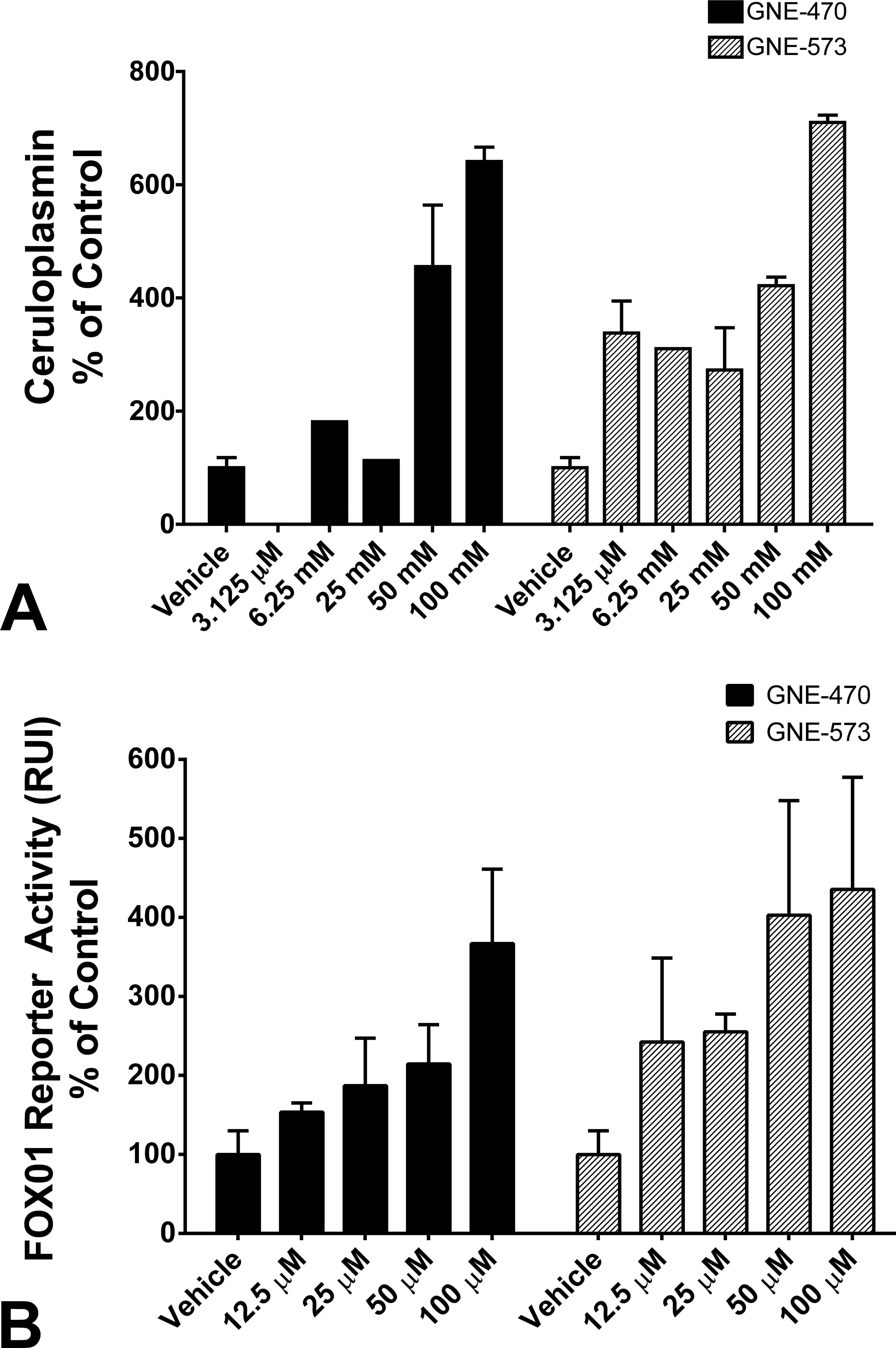
ERKi GNE-470 and MEKi GNE-573 caused an elevation in secreted ceruloplasmin in rat primary hepatocytes treated for 24 h (A). Presented is the mean ± SD of n = 3 replicate wells. ERKi GNE-470 and MEKi GNE-573 caused elevations in Forkhead box, class O1 (FOXO1) transcriptional activity in primary rat hepatocytes treated for 4 hr, as measured by luciferase counts in a relevant FOXO1 reporter system. Presented is the mean ± SD of n = 4 replicate wells.
We investigated whether the ceruloplasmin effect was related to the upstream activity of FOX01, which has been demonstrated to act on the ceruloplasmin promoter in MAPK and IL-6 signaling pathways (Leyendecker et al. 2011; Sidhu, Miller, and Hollenbach 2011). GNE-470 and GNE-573 caused a dose-dependent increase in FOXO1 reporter activity in primary rat hepatocytes after 4 hr of treatment (Figure 6B).
These experiments on isolated hepatocytes in vitro support the direct effect of inhibiting the MEK-ERK pathway on ceruloplasmin expression by FOXO1. This experimental system, comprising a simple monoculture of primary hepatocytes, also establishes that inflammation and the acute phase response are not necessary for this MEK-ERK induction of ceruloplasmin.
Discussion
In this report, we show that pharmacological inhibition of the MEK-ERK pathway in rats leads to increased synthesis and serum concentration of ceruloplasmin with the consequence of imparting a green color to serum. Structurally distinct ERKi and MEKi were associated with elevated serum ceruloplasmin and green serum at high concentrations in repeat dose rat studies appearing within 1 week of dosing. Furthermore, in vitro experiments in rat hepatocytes treated with both MEKi and ERKi resulted in a dose-dependent increase in ceruloplasmin expression, demonstrating a direct pharmacologic effect of the MEK-ERK pathway on synthesis of ceruloplasmin by the hepatocyte.
Ceruloplasmin is a moderate acute phase protein in rodents, dogs, and humans. Elevation of ceruloplasmin is lower and often delayed in comparison to the changes in major acute phase proteins, which is α-2-macroglobulin in rats (Cray 2012). As ceruloplasmin could be secondarily increased due to inflammation rather than a direct MEK-ERK pathway effect, we investigated levels of several acute phase proteins (α-2-macroglobulin, albumin, and globulin) and conducted histopathology in our rat studies. We demonstrated that ceruloplasmin elevations were dose related but inordinately increased compared to the mild increase in globulins (a positive acute phase protein) and α-2-macroglobulin, the latter occurring only at a nontolerated dose of our ERKi. Of note, other factors can influence the levels of serum albumin and globulin. Immunoglobulins are produced by lymphocytes and contribute to the globulin fraction of the serum. Furthermore, reduced serum albumin levels are characteristic of MAPK inhibition by an undetermined mechanism (Diaz et al. 2012). Widespread inflammation was present in an ERKi rat study only at an untolerated dose; however, rats at tolerated doses with green serum and/or elevated serum ceruloplasmin had no histologic evidence of inflammation. We therefore conclude that although inflammation may contribute to elevated ceruloplasmin, it is unlikely to be the primary cause of this effect in these studies. In addition, the observation of green serum is extremely rare in animal studies, and inflammation, which is quite commonly observed, is seldom associated with green serum.
The other major differential diagnosis for green serum is an increase in biliverdin concentration. Biliverdin is a breakdown product of heme catabolism that is almost completely converted to bilirubin by biliverdin reductase (Nytofte et al. 2011). In rare clinical cases of both natural and drug-induced cholestatic liver disease, elevated serum biliverdin together with an increase in total and conjugated bilirubin has resulted in green serum and so-named “green jaundice” (Fenech, Bannister, and Grech 1967; Greenberg, Bossenmaier, and Schwartz 1971). Genetic mutations of biliverdin reductase in people have also resulted in green jaundice and green serum but only in concert with conditions of cirrhotic liver dysfunction or hyperbilirubinemia from cholestasis (Gafvels et al. 2009; Nytofte et al. 2011). Since we did not observe elevated levels of serum total bilirubin, and there was no microscopic evidence of cholestasis and biliary injury, we excluded biliverdinemia as a potential cause of the green serum.
The biology of copper handling and ceruloplasmin has previously been the subject of some conjecture, although current thought is that ceruloplasmin plays a role in copper transport to tissues (Bielli and Calabrese 2002; Healy and Tipton 2007). We therefore explored the effects of MEKi and ERKi on liver and serum copper levels in rats. There was no difference in liver copper content and there was no correlation between liver copper and serum copper or serum ceruloplasmin concentration. This is not unexpected, since serum ceruloplasmin does not correlate with liver copper levels in humans (de Romana et al. 2011). We did observe a high correlation of plasma copper and ceruloplasmin, indicating that MEK-ERK inhibition resulted in increased synthesis and release of ceruloplasmin with apparently normal incorporation of copper into the molecule and proportionately increased circulating copper levels. Several findings suggest that the elevation in ceruloplasmin is nonadverse. Copper is oxidized and detoxified by the ceruloplasmin carrier. We saw no evidence of abnormal copper deposition in organs or any findings consistent with copper-induced toxicity; for example, no evidence of hepatic inflammation in rats at tolerated doses with green serum, absence of increased cytoplasmic eosinophilic globules in renal proximal tubular epithelial cells, and no evidence of hyperplastic/hyperkeratotic areas of gastric mucosa (Hebert et al. 1993). In addition, we did not observe heart-related microscopic changes or neurological-related adverse clinical signs in our rat studies (which have been postulated as potential risks for humans with increased circulating ceruloplasmin; Dadu et al. 2013; Tombini et al. 2013). In sum, the increased circulating copper observed in rat studies has no adverse toxicological consequences.
The molecular mechanism of these observations was suspected to involve the FOX01 transcription factor, which has been demonstrated in vitro to promote ceruloplasmin expression. In rat hepatoma cell lines and primary rat hepatocytes, inhibition of the MAPK pathway downstream of insulin receptor increased ceruloplasmin transcription via upregulation of FOX01 activity (Leyendecker et al. 2011). In human hepatoma cell lines, activating FOX01 with IL-6 also resulted in increased ceruloplasmin expression (Sidhu, Miller, and Hollenbach 2011). Our studies are the first to demonstrate the relevance of this molecular pathway in vivo. We also show that inhibitors of both MEK and ERK increased ceruloplasmin production in primary rat hepatocytes in vitro, confirming that the liver is the target organ for this effect and no systemic factors are required. In addition, this elevation in ceruloplasmin correlates with and is preceded by elevated FOXO1 levels, which is consistent with this molecular mechanism as a driver of the observed effects.
There are numerous MEK inhibitors that are in clinical development or marketed with no reports of inducing green serum (Luke, Ott, and Shapiro 2014). It is possible that humans are less sensitive to this effect or that the effect is related to super pharmacological activities, which would be consistent with the lack of elevated ceruloplasmin levels in rats dosed at 50 mg/kg (covering approximately 1× of projected human efficacious exposures), and its presence only in rats dosed at or above 150 mg/kg (covering approximately 6× of projected human efficacious exposures). In the clinic, MAPK inhibitors are known to cause dose-limiting adverse events, mainly rash and gastrointestinal effects, at pharmacological doses, which limits exploration of potential super pharmacological effects. A few recent reports suggest that increased circulating ceruloplasmin may be involved in cardiovascular and neurological disease. High ceruloplasmin was associated with heart failure, cardiovascular disease, and all-cause mortality despite adjustment for N-terminal prohormone of brain natriuretic peptide, cardiac troponin T, and C-reactive protein. Causal pathophysiology of this association is not understood, although one hypothesis is that increased ceruloplasmin can increase NO oxidase activity, leading to decreased nitric oxide in the heart and greater oxidative stress (Dadu et al. 2013). Ceruloplasmin has also been related to epileptic seizures in humans and tied to states of inflammation and iron dysregulation (Tombini et al. 2013).
Even so, if elevated ceruloplasmin were to manifest in humans dosed with MAPK inhibitors, this elevation would likely be considered nonadverse based on the weight of published evidence. Ceruloplasmin elevation was considered nonadverse in rats, and there is a body of literature supporting that in general, ceruloplasmin elevation in humans is nonadverse. Ceruloplasmin elevation in women has been associated with oral contraceptives and pregnancy and has not been directly related to adverse effects (Tovey and Lathe 1968; Carruthers, Hobbs, and Warren 1966). In addition, blood with elevated ceruloplasmin is considered suitable for transfusion (Elkassabany et al. 2008; Wolf et al. 1969). Our perspective is that in the context of cancer therapy, elevated serum ceruloplasmin is unlikely to represent a safety liability, and this would also be the case in non-oncology indications, as serum ceruloplasmin can be readily monitored.
In conclusion, we demonstrate that chemically distinct ERK and MEK small molecule inhibitors caused a pathway-related, pharmacological increase in ceruloplasmin that was nonadverse in acute rat toxicity studies. Rats did not display signs of copper-associated toxicity or elevated liver copper levels. Therefore, the MEK-ERK inhibitor–related increase in serum ceruloplasmin in the rat is not expected to represent a serious safety liability, even if it were to translate to humans.
Footnotes
Author Contribution
Authors contributed to conception or design (DD, DMD, JT); data acquisition, analysis, or interpretation (DD, RP, GC, NL, JS, JT); drafting the manuscript (DD, RP, GC, NL, JT); and critically revising the manuscript (DD, GC, DMD, JS, JT). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors are employees of Genentech, Inc., and stockholders of the Roche group.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
