Abstract
There are currently no reports describing mammary gland development in the Harlan Sprague-Dawley (HSD) rat, the current strain of choice for National Toxicology Program (NTP) testing. Our goals were to empower the NTP, contract labs, and other researchers in understanding and interpreting chemical effects in this rat strain. To delineate similarities/differences between the female and male mammary gland, data were compiled starting on embryonic day 15.5 through postnatal day 70. Mammary gland whole mounts, histology sections, and immunohistochemically stained tissues for estrogen, progesterone, and androgen receptors were evaluated in both sexes; qualitative and quantitative differences are highlighted using a comprehensive visual timeline. Research on endocrine disrupting chemicals in animal models has highlighted chemically induced mammary gland anomalies that may potentially impact human health. In order to investigate these effects within the HSD strain, 2,3,7,8-tetrachlorodibenzo-p-dioxin, diethylstilbestrol, or vehicle control was gavage dosed on gestation day 15 and 18 to demonstrate delayed, accelerated, and control mammary gland growth in offspring, respectively. We provide illustrations of normal and chemically altered mammary gland development in HSD male and female rats to help inform researchers unfamiliar with the tissue and may facilitate enhanced evaluation of both male and female mammary glands in juvenile toxicity studies.
Introduction
Rapid mammary gland development occurs during late embryogenesis, puberty, pregnancy, and lactation. Unlike most organs, the majority of mammary gland development occurs after birth. Female human and rodent mammary glands undergo a similar developmental process, although there are specific differences in timing (Fenton 2006; Interagency Breast Cancer and Environmental Research Coordinating Committee [IBCERCC] 2013). During embryogenesis, the mammary bud and the resulting primary duct of both humans and rodents extend into the developing mammary fat pad. Secondary side branching of the primary duct occurs and a small ductal tree is present at the time of birth. Prior to puberty, the female gland undergoes isometric growth, and as puberty progresses, the ductal growth occurs exponentially. In adulthood, the female mammary gland enters a resting static state, with slight hormone-induced changes in the extent of lobular development in response to phases of the menstrual/estrous cycle (Russo and Russo 2004). At this point, the overall structure of the adult female mammary gland is tubuloalveolar, meaning that the gland is composed primarily of ducts with lumens. Starting at pregnancy and continuing through lactation, the overall structure of the mammary gland becomes lobuloalveolar (LA), where alveolar lobules, surrounded by the myoepithelium, are the predominant structures serving the function of milk production.
Improper mammary gland development at different life stages has important health consequences. There is accumulating evidence in rodents that fetal exposure to certain chemicals may lead to inappropriate development of the mammary gland in offspring, which may result in nipple/areola retention in males and enhanced or delayed ductal development in females (Enoch et al. 2007; Axelstad et al. 2014; Mandrup et al. 2012; White et al. 2011; Fenton et al. 2002). During puberty, endocrine disruption can result in early mammary gland development, a common indicator of precocious puberty in girls, or gynecomastia, which is abnormal breast development in males (Colon et al. 2000; Durmaz et al. 2010; Basaria 2010; Macon and Fenton 2013). Both of these conditions are associated with an increase in the risk of certain cancers developing later in life. Chemically induced abnormal mammary gland development during lactation can lead to an inadequate production of milk in rodents (Vorderstrasse et al. 2004; White et al. 2007; Manservisi et al. 2014).
The most important potential disease outcome involving the mammary gland in humans is breast cancer. According to the American Cancer Society (ACS), in the United States, breast cancer develops in about 1 in 8 women and 1 in 1,000 men (ACS 2014). In women, breast cancer is the most commonly diagnosed form of cancer (122 in 100,000 women annually), and breast cancer kills more women (21.5 in 100,000 women annually) compared to any other type of reproductive or endocrine-related cancer, in the United States (Center for Disease Control and Prevention 2014; Surveillance, Epidemiology, and End Results Program 2014). Approximately 85% of breast cancers occur in women who have no family history of breast cancer, suggesting the potential importance of environmental factors (IBCERCC 2013).
Rodent studies have proven that the mammary gland is a tissue sensitive to the effects of endocrine disrupting chemicals (EDCs), which are exogenous chemicals that, individually or in mixtures, may interfere with any aspect of hormone action (Zoeller et al. 2012). There are certain stages of development in which the mammary gland is more susceptible to the effects of EDCs. These “windows of susceptibility” for the mammary gland are during embryogenesis, puberty, and pregnancy/lactation, when the mammary gland is undergoing extensive proliferation. Terminal end buds (TEBs) are highly proliferative epithelial structures present during puberty in the rodent mammary gland. Since TEBs are the precursors of future branching ducts, damage or altered programming of these mammary gland structures can have lasting consequences within the mammary gland (Russo et al. 1983). EDCs have been reported to change mammary gland development following prenatal, neonatal, and peripubertal exposure (reviewed in Rudel et al. 2011). Chemical disruption of mammary gland development during the early windows of susceptibility (prenatal) can result in impaired lactation and increased susceptibility to cancer as adults.
In addition to the temporal similarities during mammary gland development, the cellular composition of human and rodent mammary glands is similar. In humans, the functional portion of the mammary gland is called the terminal ductal lobular unit (TDLU), and in rodent species, it is called the LA unit or the terminal ends, not to be confused with TEBs. Both the TDLU and LA unit are composed of similar luminal cuboidal epithelium and surrounding myoepithelial cells, with a tubuloalveolar morphology in female rodents (abundant ducts and few alveoli; Cardiff and Wellings 1999; Lucas et al. 2007). During pregnancy and lactation, the mammary epithelium shifts to an LA morphology, with abundant alveolar lobules and fewer ducts.
The structure of mammary glands in men is considered a rudimentary form of the female gland (Howard and Gusterson 2000). On the other hand, mammary glands of rodents exhibit several sex-associated structural differences. Male rats have branched mammary epithelium present at birth, though they have been reported to be smaller in size than those of females, and the mammary epithelium of sexually mature males have a permanent LA structure (Cardy 1991; Rudmann et al. 2005). Male mice undergo an embryonic regression of the mammary gland buds on embryonic day (E) 14 due to an androgen surge. The epithelium connecting the mammary bud detaches and often completely regresses, although male mice may occasionally possess a residual stalk of epithelial tissue that is structurally similar to the female primary duct (Richert et al. 2000). Recent data suggest these epithelial stalks in male mice may respond to EDC exposure (Vandenberg et al. 2013).
The rat mammary gland is an excellent model to study the effect of environmental chemicals that may impact breast cancer risk in humans. Altered rodent mammary gland development during windows of susceptibility can be evaluated through the use of mammary gland whole mounts. Whole-mount preparations of the mammary gland are fixed, defatted, and stained on a charged microscope slide. The glandular epithelial and stromal cells are stained with carmine and the outlines of adipocytes are intact, as they were in the rodent. Whole mounts allow for evaluation of the total number and relative abundance of mammary gland ductal structures, including TEBs, overall growth of the epithelial cells, and branching density (Schwarzman et al. 2015).
There is a great need for a thorough description of normal female and male mammary development in rat strains that are used in chemical testing. The National Toxicology Program (NTP) has moved away from the use of the inbred Fisher 344/N rat due to decreased fecundity, sporadic seizures, and high rates of mononuclear cell leukemia and testicular neoplasia (King-Herbert and Thayer 2006). The majority of current studies in the literature were based on the Charles River Sprague-Dawley strain, but this strain forms galactoceles in mammary tissue spontaneously, complicating interpretation of results. One of the preferred testing strains in the NTP is now the Harlan Sprague-Dawley (HSD) rat because the litter size, sex ratio, and body weights (BWs) were favorable (King-Herbert, Sills, and Bucher 2010). NTP and European-based guideline studies (modified one-generation reproduction studies, reproductive assessment by continuous breeding studies, and 2-year bioassays) have recently been modified to include assessing mammary gland development in their standard testing protocols (NTP 2011; Organisation for Economic Co-operation and Development [OECD] 2011). Strain differences in mammary gland development are reported (Stanko et al. in press), necessitating a pictorial of normal HSD rat mammary gland development.
The current studies present a qualitative pictorial characterization of mammary gland development in male and female HSD rats from the time the mammary bud is first evident until young adulthood. The study focuses on HSD rats, as this strain is currently being used in some NTP test guideline studies, and specific information on mammary development in this strain is lacking in the literature. Bud development was characterized histologically using fetal transverse hematoxylin and eosin (H&E)–stained sections starting on E15.5 and continuing through E21.5. Postnatal development was evaluated using mammary gland whole mounts, coronal histology sections, and sex hormone receptor immunohistochemically (IHC) stained sections prepared from the 4th and 5th inguinal gland starting at postnatal day (PND) 1 and continuing through PND 70.
To provide representative examples of accelerated or delayed mammary gland growth and differentiation in males and females, diethylstilbestrol (DES) or 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), respectively, was administered to a subset of pregnant dams during embryonic mammary gland organogenesis. DES and TCDD were selected because they have been previously shown in the literature to either accelerate or delay mammary gland growth in rats or other species, respectively, following early life exposures (Brown and Lamartiniere 1995; Fenton et al. 2002; Hovey et al. 2005). DES is a synthetic, estrogen-like endocrine-disrupting chemical that has been associated with an increased risk of breast cancer in women over 40 years of age, exposed in utero (Hoover et al. 2011). TCDD has been shown to reduce estradiol production, delay vaginal opening, and disrupt normal estrous cyclicity, and it was chosen as a model chemical to demonstrate stunted mammary gland growth, as it has already been shown to do this in 3 strains of rats, at a 1 μg/kg single oral dose delivered on gestational day (GD) 15 (Fenton et al. 2002; Brown et al. 1998; Lewis et al. 2001).
Effects of chemicals on male rat mammary gland development are currently understudied or ignored, and our work identified male- and female-associated sex differences and chemically altered mammary development of HSD rats. The data presented herein will benefit future studies assessing morphological changes in mammary gland end points in the developing HSD rat and other related strains and will serve as a point of reference, as chemical-induced effects in mammary tissue of male and female rats continue to be identified. It is our hope that this pictorial description will assist contract pathologists and individual investigators to accurately identify characteristics of altered mammary gland development.
Material and Method
Animals
Three blocks of timed-pregnant Sprague-Dawley rats were purchased from Harlan Laboratories (Indianapolis, IN). Upon arrival, pregnant dams (GD 10 or 13) were housed individually in polypropylene cages with bedding and nestlets and were provided 5412-01 phytoestrogen-reduced diet (Zeigler; Madison, WI). Water and chow were supplied ad libitum. The animals were maintained in a room with a 12:12-hr light/dark cycle, at 20°C to 24°C, and 40% to 50% relative humidity. All animals were treated humanely and with regard to alleviation of potential suffering, as approved by the National Institute of Environmental Health Sciences Animal Care and Use Committee.
Pictorial Characterization
Block 1
Timed-pregnant dams were randomly distributed among 3 treatment groups (n = 9/group), with mean BW being equivalent between the groups. Dams in the 3 groups were treated by oral gavage on GD15 and GD18 with vehicle control (5 ml sesame oil/kg BW), TCDD (0.5 µg TCDD/kg BW), or DES (10 µg DES/kg BW). Dam BW was recorded daily throughout gestation. TCDD was purchased from Radian Corporation (Austin, TX), and DES was purchased from Sigma (St. Louis, MO). Dosing solutions were prepared fresh on the days of treatment. The dose of DES was determined from previous studies (Sharpe et al. 1998; Goyal et al. 2003) to ensure there was no prenatal loss or delayed parturition and was similar to doses previously shown to induce persistent mammary changes in mice, albeit their exposure window was longer (Hovey et al. 2005). The TCDD dose was derived from a previous study (same dose, but split between 2 days in this study) in rats where mammary development was persistently delayed (Fenton et al. 2002). GD15 and GD18 were chosen as exposure days to target the reported sensitive period of female mammary bud development (Fenton et al. 2002) and the period during which the male nipple undergoes sexual dimorphism (Foley et al. 2001). Treated and control dams were allowed to give birth and neonates were equalized to 10 pups per litter (5 male, 5 female) to equalize the growth potential across litters. On PNDs 1, 4, 8, 12, 15, and 21, 1 dam and litter per treatment group were euthanized and mammary glands were collected from the dam and pups for whole mounts and histology. At PND 21, all remaining litters were weaned. Offspring (3 males and 3 females per litter) were sacrificed on PNDs 33, 46, and 70 to continue to monitor postpubertal mammary gland development.
Block 2
Untreated timed-pregnant dams (n = 14) were utilized for fetal mammary gland developmental assessment. Two dams were sacrificed, and male and female fetuses were collected (n = 3 fetuses/sex collected) for mammary gland evaluation on E15.5, 16.5, 17.5, 18.5, 19.5, 20.5, and 21.5.
Block 3
Because of a problem in the quality of fixation in late fetal developmental samples from block 2, untreated timed-pregnant dams (n = 8) were utilized for fetal mammary gland developmental assessment. Two dams were sacrificed, and male and female fetuses were collected (n = 3 fetuses/sex collected), for mammary gland evaluation on E18.5, 19.5, 20.5, and 21.5. The only difference from block 2 was the method of fixation (described later) and the more limited life stages collected.
Mammary Gland Samples
The 4th and 5th inguinal mammary glands are the easiest and largest glands to assess and were removed from one side of the body in all dams and PND 1 to PND 70 pups (both sexes) from block 1. Glands were transferred to a charged microscope slide and stretched to normal size (the approximate size they were in the body). They were briefly sandwiched under the weight of a 50-ml conical tube filled with water, then fixed in Carnoy’s solution and stained in carmine as a whole mount as previously described (Davis and Fenton 2013; Fenton et al. 2002). Using the evaluation criteria specified in Davis and Fenton (2013), a qualitative developmental score was given, determined by comparison of longitudinal growth, lateral growth, branching density, number of TEBs, number of terminal ends, and budding. Each characteristic has varying importance on the overall score depending on the time point captured. For example, there is very little budding in early life stages, but this criteria becomes very important by PND 21. Slides were evaluated by 2 individuals blinded to the respective treatment group. If differences in developmental scores were greater than 1.0, then both scorers reevaluated all slides in contention, blindly. Discussions on exact criteria related to the scores may have been needed until both scorers agreed on scores less than or equal to one score difference. Less than 5% of all glands required reevaluation. A representative mammary gland was selected for pictorial representation from each age-group, for each sex. Using the whole-mount samples, TEBs were counted as the number of ends that measured 100 μm across the widest portion of the structure in each gland.
The 4th (and often 5th) gland contralateral to the whole-mounted gland was lifted from the skin of the rat, laid flat, skin side down in the histocassette, fixed in 10% neutral-buffered formalin for 24 hr, transferred to 70% ethanol, and processed and embedded in paraffin within 48 hr. Each block was step sectioned until the lymph node was visible. This was done to ensure that the majority of the epithelium present was in the same plane of view for evaluation. After reaching the lymph node, serial 5-µm sections were collected for H&E and IHC staining. The following nuclear receptors were assessed by IHC staining: estrogen receptor alpha (ERα), progesterone receptor (PR), and androgen receptor (AR).
Fetuses in blocks 2 and 3 were euthanized with wet ice and decapitation (E15.5–E17.5) or by decapitation (E18.5–E21.5). In block 2 (n = 14), fetuses were fixed in Bouin’s fixative (Poly Scientific R&D Corp. Bay Shore, NY) for 48 hr. However, late gestation fetuses demonstrated poor fixation. Therefore, in block 3 (n = 8), Bouin’s fixative was injected into the abdominal cavity and fetuses were further fixed in Bouin’s fixative for 48 hr. This protocol resulted in better fixation and is recommended for future fetal mammary gland analysis. Following fixation, a transverse cut was made at the level of the umbilicus. The upper half of the embryos was discarded while the bottom half was rinsed 3 times in 70% ethanol to remove the Bouin’s solution and placed in 1× phosphate-buffered saline at 4°C. Samples were then processed and embedded in paraffin. For optimal histopathological evaluation of the fetal mammary glands (E15.5–E21.5), the fetus was embedded in the transverse plane. The fetus was exhaustively sectioned (6 μm) through in the region of the 4/5th mammary gland. Fetal cross sections that potentially contained mammary tissue were identified by light microscopy and were then H&E stained.
ERα and PR Immunohistochemistry
Unstained tissue sections were deparaffinized and rehydrated through a series of alcohols. All incubations were performed at room temperature. Endogenous peroxidase was blocked with 3% hydrogen peroxide for 15 min. Heat-induced epitope retrieval (HIER) was performed using 0.01 M citrate buffer, pH 6.0 (Biocare Medical, Concord, CA) at 110°C for 15 min in a decloaking chamber (Biocare Medical). The sections were incubated with 10% normal horse serum (Jackson ImmunoResearch Laboratories, Inc., West Grove, PA) for 20 min. Endogenous biotin was quenched with the Avidin–Biotin Blocking Kit (Vector Laboratories, Burlingame, CA). Sections were incubated with either mouse monoclonal anti-ERα antibody (ER1D5, stock concentration of 1 mg/ml) at a 1:200 dilution (catalog # IM1545; Beckman Coulter, Brea, CA) or mouse monoclonal anti-PR antibody (PR10A9, stock concentration of 1 mg/ml) at a 1:150 dilution for 1 hr (catalog # GTX39501; GeneTex, Irvine, CA). For a negative ERα and PR control, sections were incubated with either concentration-matched mouse IgG1 or IgG2a isotype control serum, respectively, in place of the primary antibody (BD Biosciences, San Diego, CA). Sections then were incubated with biotinylated horse antimouse IgG (Vector Laboratories) for 30 min at either 1:500 or 1:1000 dilution, respectively, and then labeled with either Streptavidin Super Sensitive Horseradish Peroxidase Label (Biogenex Laboratories, San Ramon, CA) or Vectastain Elite ABC reagent, R.T.U. (Vector Laboratories) for 30 min, respectively. The antigen–antibody complex was visualized following a 6-min incubation with 3, 3′-diaminobenzidine (Dako Corporation, Carpinteria, CA). The sections were counterstained on automated equipment with modified Harris hematoxylin, dehydrated, cleared, and coverslipped with Permount.
AR Immunohistochemistry
Slides for AR IHC underwent deparaffinization, HIER, and were blocked for endogenous peroxidase as described above. The sections were incubated with 10% normal donkey serum (Jackson Immunoresearch) for 20 min, followed by the Avidin–Biotin Blocking kit (Vector Laboratories) to quench endogenous biotin. Rabbit polyclonal anti-AR antibody (catalog # sc-816, stock concentration of 0.2 mg/ml; Santa Cruz Biotechnology, Dallas, TX) was applied to the sections at a dilution of 1:100 for 1 hr. Negative control slides received normal rabbit IgG (Calbiochem, Billerica, MA) at the same concentration as the primary antibody. A donkey antirabbit biotin conjugated secondary antibody was applied for 30 min at a 1:500 dilution, followed by Vectastain Elite ABC reagent, R.T.U. (Vector Laboratories) for 30 min. Chromogen incubation, counterstaining, and coverslipping were identical to the ERα and PR protocol described above.
ERα/PR/AR Quantification
A modified Quick Score method (Detre, Saclani Jotti, and Dowsett 1995) was used to quantitate IHC nuclear staining for ERα, PR, and AR in the mammary epithelium. This common method has been validated in the quantification of ER staining in breast carcinomas and was found to produce similar results to the common H-score method, which takes into consideration the staining intensity in conjunction with the percentage of cells staining positively (Detre, Saclani Jotti, and Dowsett 1995). The overall intensity of the staining is scored on a scale of 0 to 3; where 0 = no staining/negative, 1 = weak staining, 2 = intermediate staining, and 3 = strong staining. The estimated percentage of cells that are stained throughout the entire gland is scored on a scale of 1 to 6, where 1 = 0% to 4% staining, 2 = 5% to 19% staining, 3 = 20% to 39% staining, 4 = 40% to 59% staining, 5 = 60% to 79% staining, and 6 = 80% to 100% staining. The Quick Score is then calculated by multiplying the average intensity (0–3) by percentage of cells stained (1–6), resulting in Quick Scores ranging from 0 to 18 for nuclear localized proteins. Nuclear staining was quantified by 2 reviewers unaware of the sample age, sex, or treatment group. If discrepancies in score were greater than 5, then slides in question were reevaluated blindly.
Bright-Field Slide Scanning
Bright-field digital images were taken from H&E or IHC slides scanned on the Leica ScanScope XT™ instrument (Vista, CA) using ImageScope™ software (v12.3.0.5056; Leica Biosystems, Buffalo Grove, IL). The extraction feature in ImageScope was used if a digitally scanned image required rotation. Image enhancement (imaging, resizing, and white balance correction) was done using Adobe Photoshop™ Creative Cloud (Adobe Photoshop, 2014.0.0, San Jose, CA). Automated quantitative analyses of epithelial nuclear density and epithelial percentage area in H&E-stained sections were made according to previously published protocols (Sandhu et al. 2016). Digital images were uploaded to eSlide Manager™ and visualized with ImageScope v 12.2 (Leica Biosystems). A technician was blinded to treatment and placed regions of fixed size on each image (784996.03 µm2), avoiding areas of tissue folding. The annotated images were imported to Definiens Architext XD 2.4 for analysis with Tissue Studio™ version 4.1. A Definiens Composer algorithm was used to identify epithelial regions and to calculate the percentage of total area composed of epithelium and to compute the density of nuclei per square millimeter of epithelium. The algorithm outcomes were reviewed by a veterinary pathologist for accuracy (see Supplementary Figure 1, e.g., of annotation). The algorithm was not optimized for male or older rats (older than PND 70), both of which have higher prevalence of lobular, vacuolized epithelium, so the current analyses included only female animals prior to PND70.
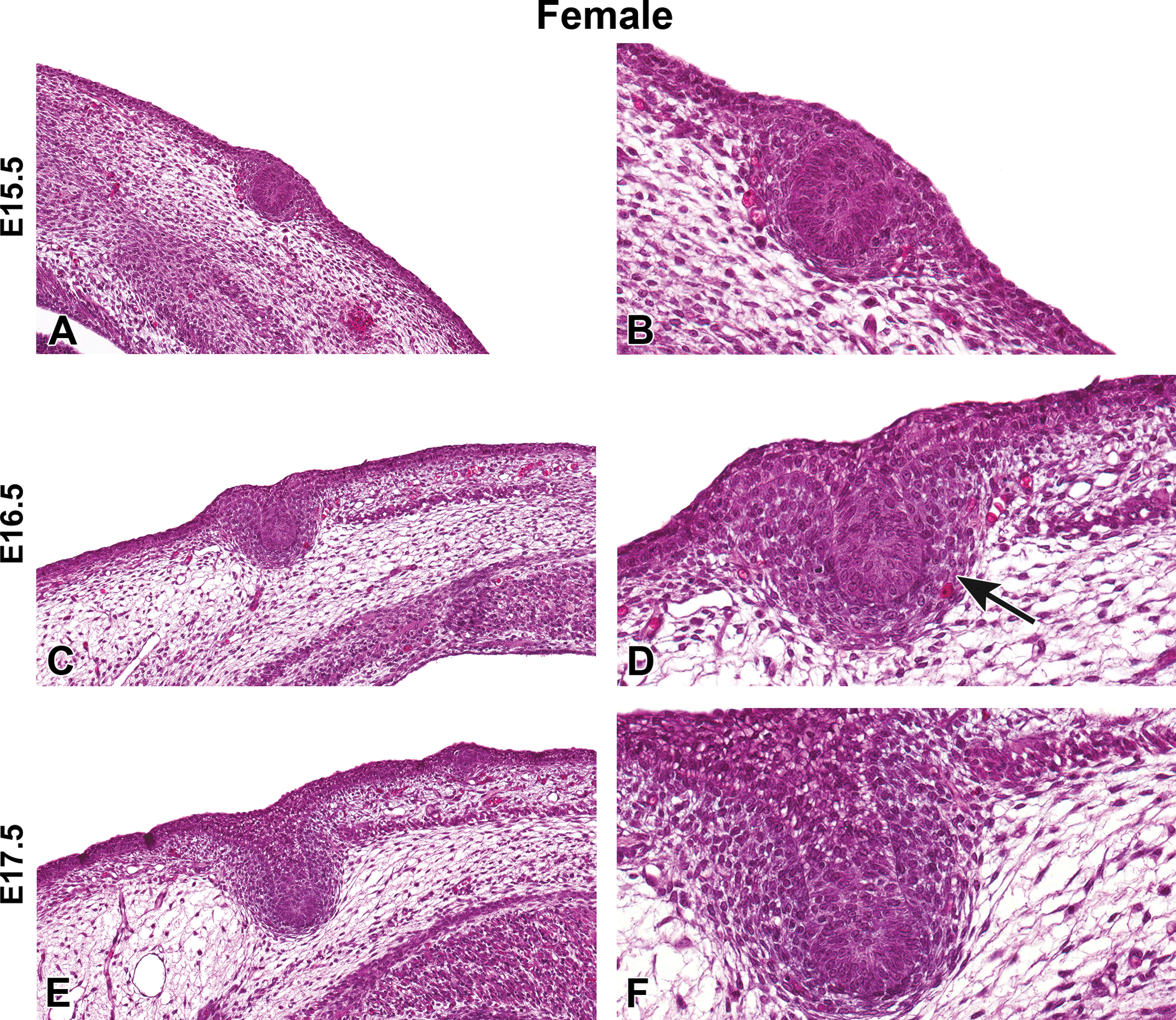
Representative hematoxylin and eosin images of female Harlan Sprague-Dawley rat fetal mammary gland development from embryonic day (E) 15.5 to E17.5. (A and B) Mammary bud formation, E15.5. (C and D) Mammary stalk formation, E16.5. (D) Mammary mesenchyme formation (arrow). (E and F) Elongation of mammary stalk, E17.5. (A, C, E) Original magnification 16×. (B, D, F) Original magnification 40×.
Statistics
This study was not powered for statistical analysis of quantifiable characteristics. Instead, pictorial representations are shown of examples of mammary glands throughout development and that were thought to have been altered by chemical exposure. Epithelial percentage and epithelial nuclear density were analyzed by treatment group using linear regression adjusted for continuous age (PND). Statistical significance was defined as p < .05, and analyses were performed using SAS software, version 9.3 (SAS Institute, Cary, NC).
Results
HSD Rat Mammary Gland Development
Fetal mammary bud
H&E-stained sections of E15.5 to E21.5 fetuses were collected from untreated dams to demonstrate the progression of female and male mammary bud development (Figures 1 –4). At E15.5, the early mammary bud of both sexes consisted of a sphere of concentrically arranged epithelial cells subjacent to the plane of the surrounding epidermis (Figures 1A and B, 3A and B). Cells surrounding the bud continued to elongate, condense, and later connect to the skin surface by a stalk of epidermal-like cells at E16.5. During this gestational time point, the mature mammary bud was surrounded by 3 to 5 layers of condensed fibroblasts referred to as the primary mammary mesenchyme. Developing from the mesenchyme, along with the formation of the ectodermally derived mammary buds, was the support structures including the adipose tissue, which later formed the fat pad containing blood vessels, lymphatics, and connective tissue (Figures 1C and D, 3C and D). The bud remained quiescent from E16.5 to E17.5 and was relatively unchanged except for further progression into the underlying adipose tissue. The first apparent sex-specific difference appeared thereafter; in the E18.5 male mammary gland, there was a destruction of the developing mammary bud (Figure 4). In the female, the mammary gland began to invaginate where the nipple will eventually develop (Figure 2A). Invagination continued in the female through E19.5 when the outer structure of the nipple formed (Figure 2C and D) and the epidermis covered the stalk. At E20.5, the nipple sheath formed in the female fetus (Figure 2E and F). This was a modification of the skin present in the immediate vicinity of the mammary duct. The epidermis thickened and projected down into the dermis in an umbrella-like fashion, forming a ridge that surrounded the origin of the primary epithelial duct. In the male, epithelial elongation occurred yet the mammary gland did not invaginate or undergo nipple formation (Figure 4A–F). At E21.5, solid epidermal cords extended from the mammary bud and grew through the mammary mesenchyme into the secondary mammary mesenchyme, or fat pad precursor tissue, forming mammary sprouts in both the male and female (Figures 2G and H, 4G and H). Once the mammary sprout reached the fat pad, it began a process of ductal branching morphogenesis that gave rise to the rudimentary ductal tree, consisting of a primary duct, several secondary branches, and numerous tertiary branches, which was present at birth in both males and females.
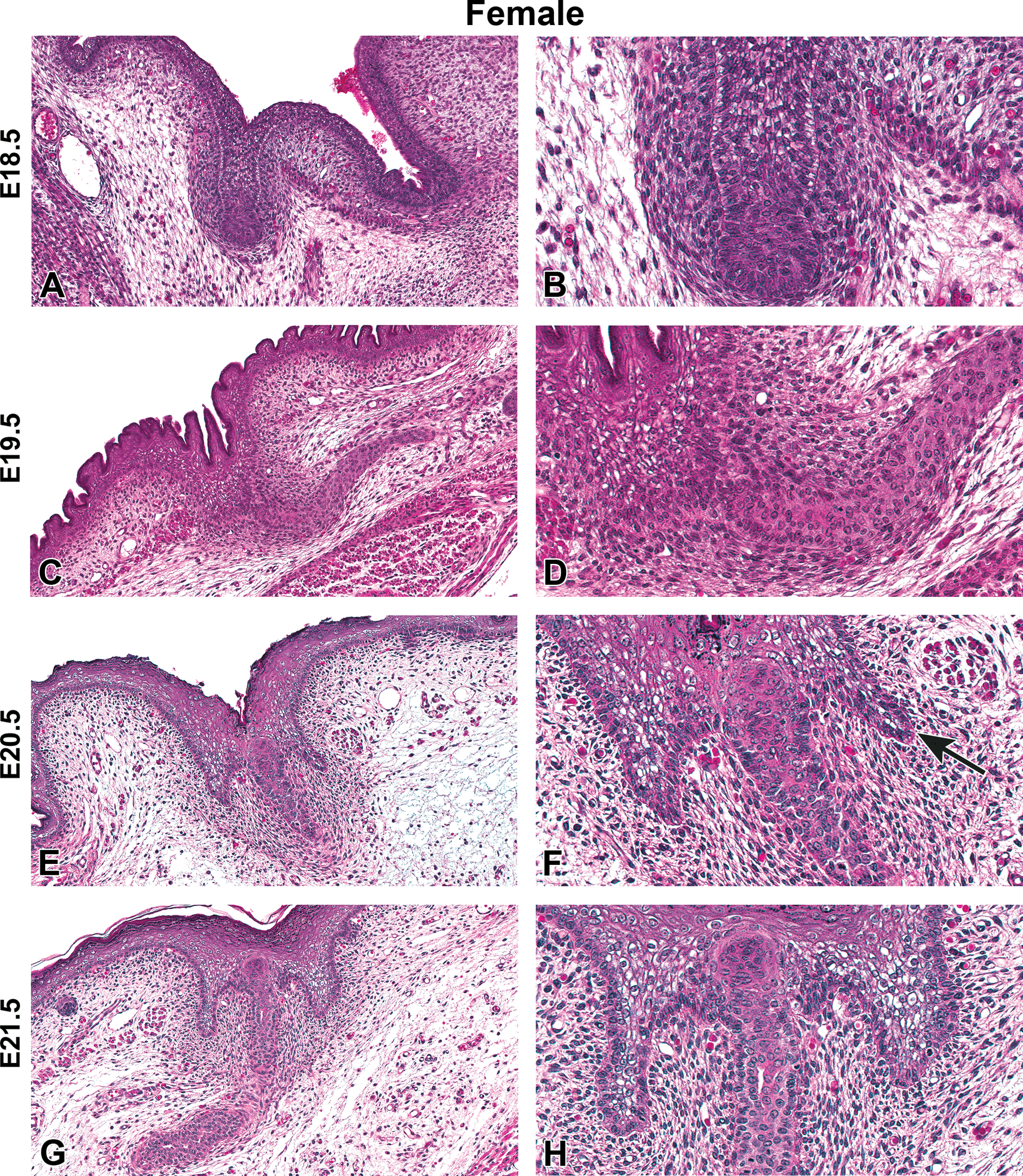
Representative hematoxylin and eosin images of female Harlan Sprague-Dawley rat fetal mammary gland development from embryonic day (E) 18.5 to E21.5. (A and B) Invagination of the skin above the mammary gland, E18.5. (C and D) Elongation of the mammary gland stalk, E19.5. (E and F) Formation of the nipple sheath, E20.5. (F) Nipple sheath forms (arrow). (G and H) Primary duct begins lumen formation, E21.5 (A, C, E, G) Original magnification 16×. (B, D, F, H) Original magnification 40×.
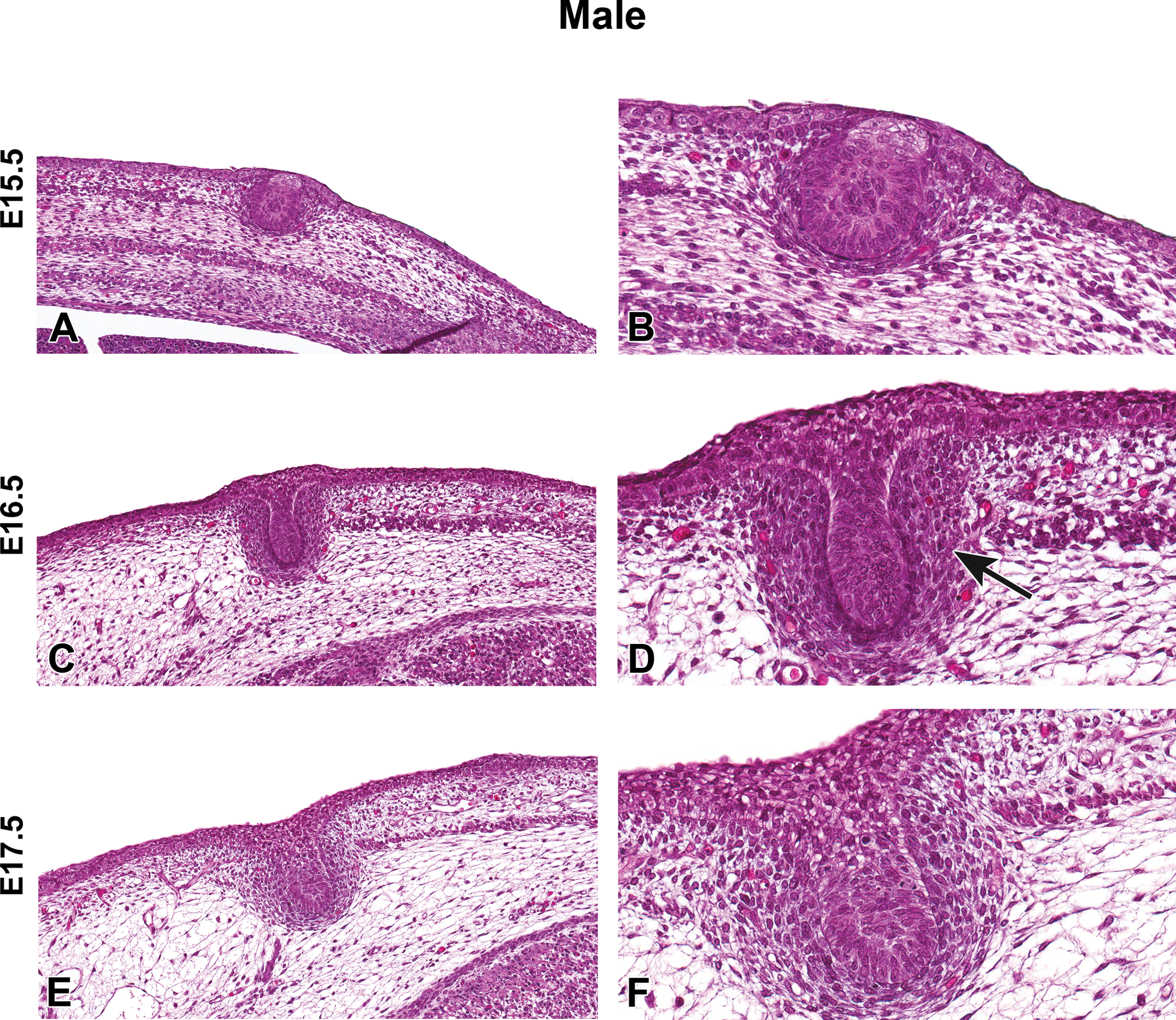
Representative hematoxylin and eosin images of male Harlan Sprague-Dawley rat fetal mammary gland development from embryonic day (E) 15.5 to E17.5. (A and B) Mammary bud formation, E15.5. (C and D) Mammary stalk formation, E16.5. (D) Mammary mesenchyme formation (arrow). (E and F) Elongation of mammary stalk, E17.5. (A, C, E) Original magnification 16×. (B, D, F) Original magnification 40×.
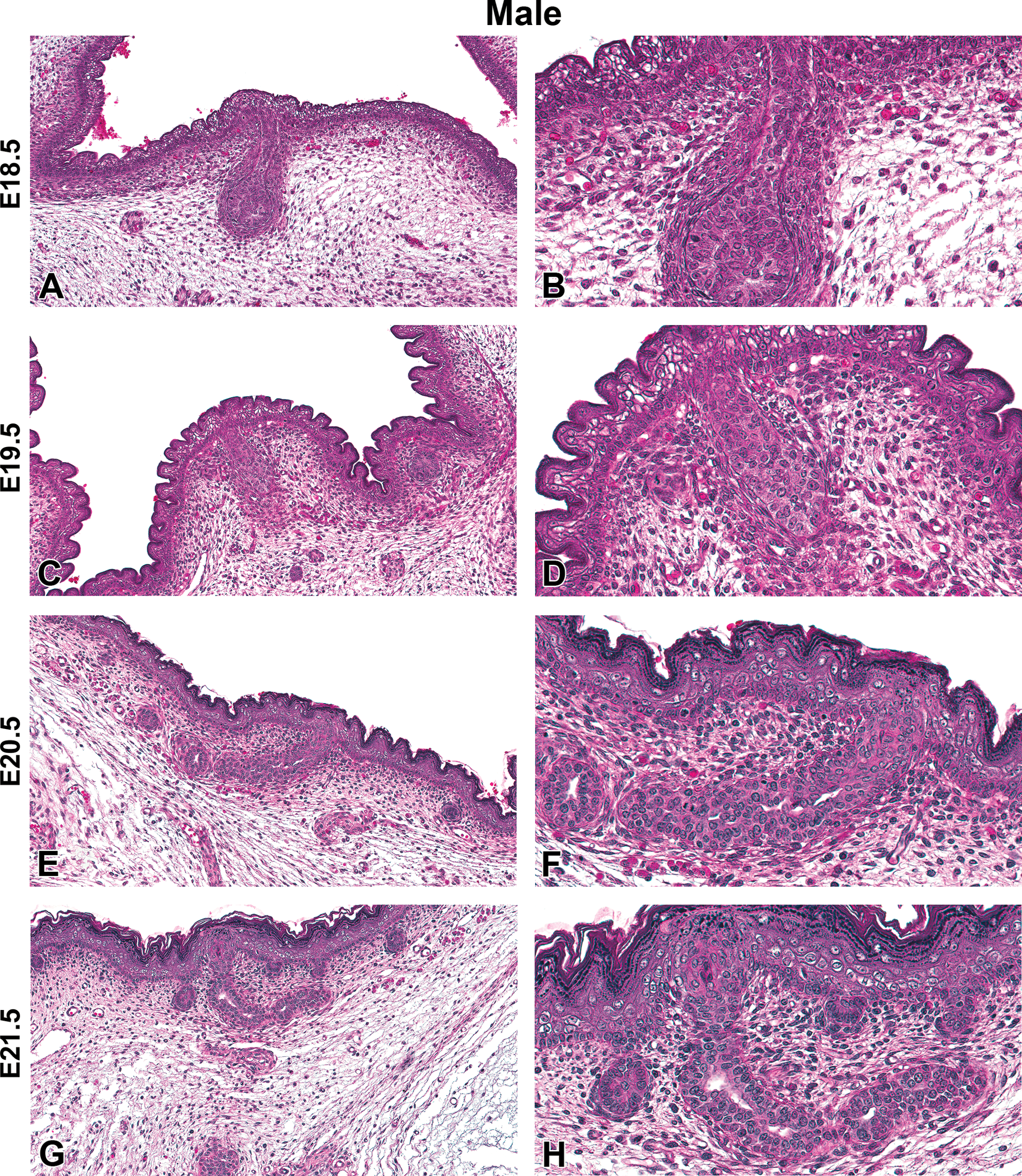
Representative hematoxylin and eosin images of male Harlan Sprague-Dawley rat fetal mammary gland development from embryonic day (E) 18.5 to E21.5. (A and B) Atrophy of the mammary gland stalk, E18.5. (C and D) Further atrophy of the mammary gland stalk, E19.5. (E and F) Primary duct begins lumen formation, E20.5. (G and H) Elongation of the primary duct with apparent lumen, E21.5. (A, C, E, G) Original magnification 16×. (B, D, F, H) Original magnification 40×.
Cellular morphology in control pups and pregnant/lactating dam
Cellular morphology of the female and male mammary gland was compared/contrasted using H&E-stained longitudinal coronal sections (Figures 5 and 6, respectively). In these studies, samples were collected at 7 ages, but only 4 age-groups are presented in Figures 5 and 6. At PND 4, both females and males displayed a simple mammary gland primarily composed of branching ducts and a few buds (Figures 5A and 6A). Histologically, the ducts of the prepubertal female and male rat mammary gland are characterized by a single or double layer of cuboidal epithelium, which after branching a number of times and causing extension toward the lymph node become multilayered at the duct end to form the TEBs (i.e., Figure 6B). These similarities between sexes in cellular structure continued through weaning (Figures 5B and 6B). Around PND 21, the TEBs begin to cleave into new ducts that form clusters of 3 to 5 alveolar buds each with a centrally located lumen surrounded by a layer of cuboidal epithelial cells. Between PND 21 and PND 46, the females will have undergone vaginal opening, began regular hormone surges, and have an established estrous cycle (Miousse et al. 2013; Stanko et al., in press). The males are peripubertal at PND 46, likely progressing through preputial separation a few days prior to this time point (Rocca and Pepperl 2000). By PND 46, one of the most prominent changes was an increase in the lobular architecture of the male gland. Luminal epithelial cells contained enlarged vacuoles within the cytoplasm, and by PND 70, the male mammary gland was a sexually mature gland. The glandular tissue was greater in volume and was organized into complex lobules/alveoli that consisted of large, pale staining, foamy, and vacuolated cells arranged around indistinct lumen that occasionally contained small amounts of eosinophilic secretory material. Ducts were lined by multiple layers of epithelium (pseudostratified or striated epithelium) and also had indistinct lumen, resembling alveoli. The mammary structure in the mature male is termed LA (Figure 6D). At PND 70, the mammary gland of the mature, virgin female rat varies little histologically from that of a prepubertal female except for more abundant branching, narrower ducts, and more numerous buds and lobules. The female gland at this time point is characterized by branching tubular ducts, terminal end and alveolar buds, and clusters of patent alveoli; the mature female gland is tubuloalveolar (Figure 5D).
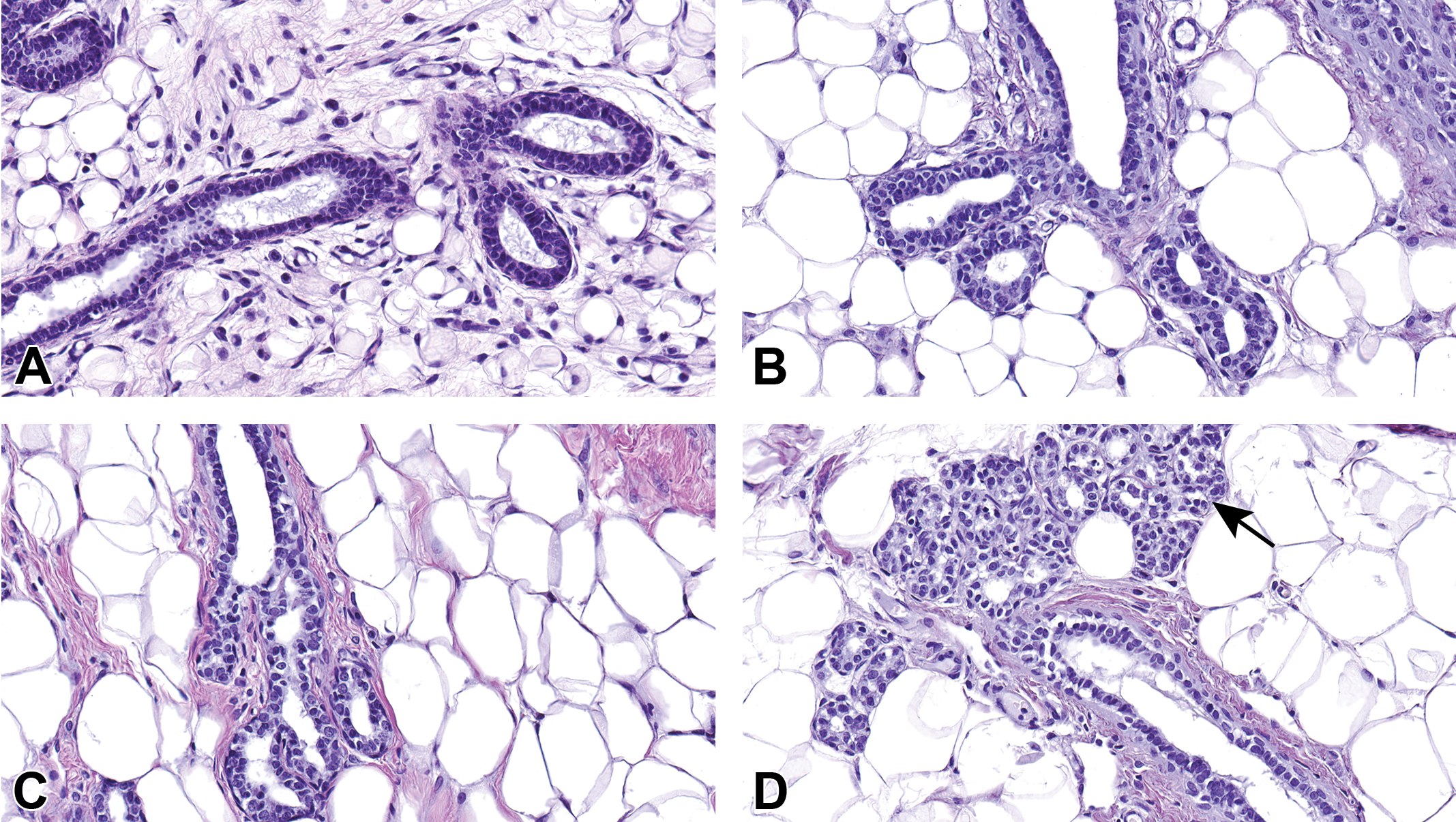
Hematoxylin and eosin images of female mammary gland development in Harlan Sprague-Dawley rat offspring from birth to sexual maturity. (A) At postnatal day (PND) 4, mammary gland ducts are lined by a single or double layer of cuboidal epithelial cells. (B) At PND 21, there is early branching of the mammary epithelium. (C) By PND 46, the terminal end buds have cleaved to form new ducts. (D) By PND 70, female mammary gland is tubuloalveolar. The lobule is composed primarily of ducts and clusters of 3 to 5 alveolar buds, each with a centrally located lumen surrounded by a layer of cuboidal epithelial cells. Terminal ductal lobular unit (TDLU) forming (arrow), (A–D) original magnification 40×.
Hematoxylin and eosin images of male mammary gland development in Harlan Sprague-Dawley rat offspring from birth to sexual maturity. (A) At postnatal day (PND) 4, the mammary gland ducts are lined by simple cuboidal epithelial cells. (B) At PND 21, there is early branching of the mammary gland epithelium. (C) At PND 46, the terminal end buds have cleaved to form new ducts. (D) By PND 70, the male mammary gland is predominantly lobuloalveolar. Alveoli are prominent and ducts are infrequent. Alveoli and ducts are lined by stratified epithelium that consists of tall vacuolated cuboidal to short columnar epithelial cells, (A–D) original magnification 40×.
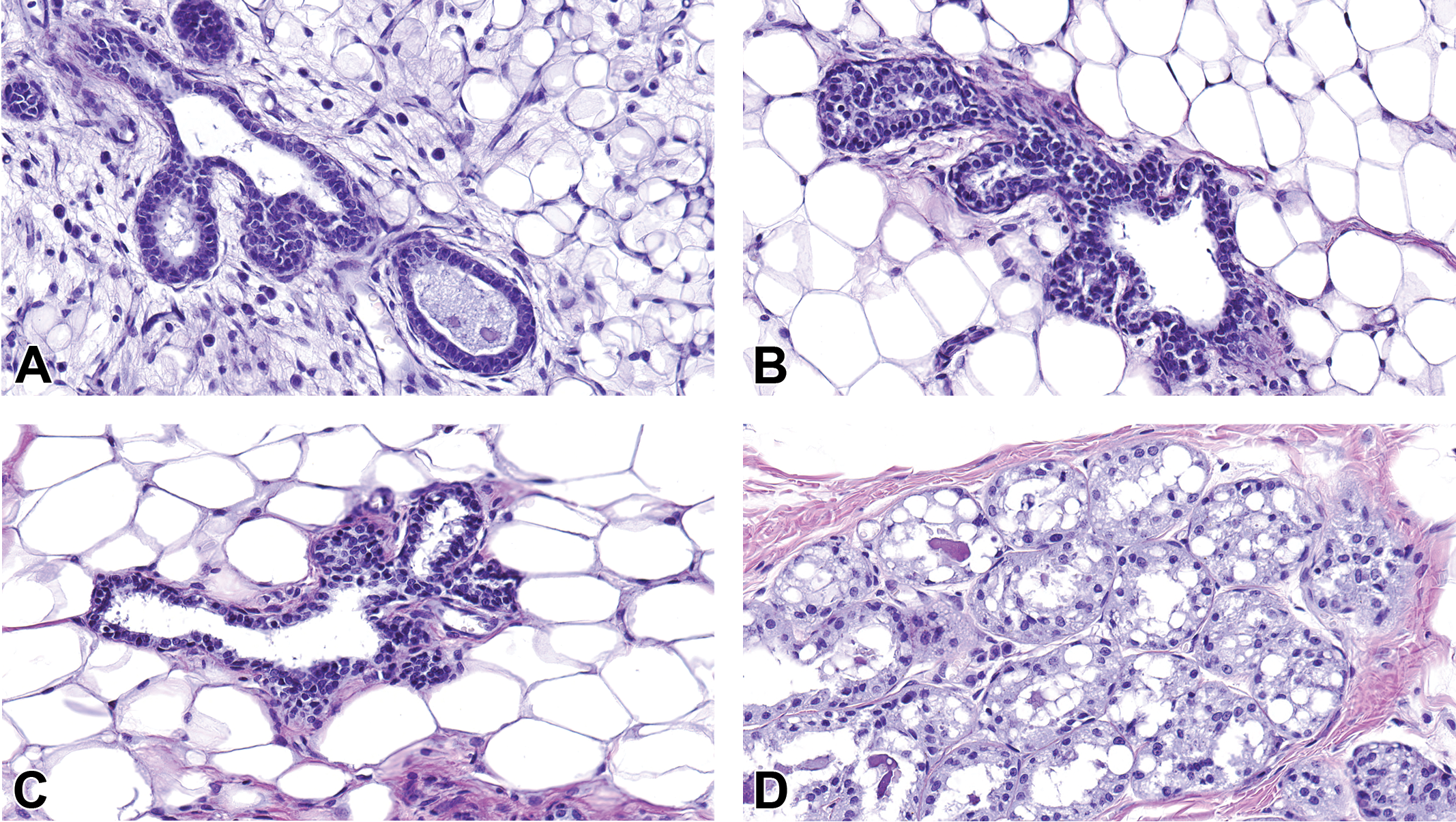
Sections of H&E-stained mammary glands from pregnant and lactating control dams are shown in Figure 7. At GD15, the mammary gland already had acquired a LA structure (large, vacuolated cytoplasm with a diminished lumen) with abundant adipose tissue and tubular ducts (Figure 7A). Between GD15 and GD21, extensive ductal growth, lobular development, and alveolar maturation occurred to form a milk-producing gland. With differentiation of the gland, there was a concurrent reduction in the amount of adipose tissue and ducts were less prominent (Figure 7B and C). The secretory alveoli became lined by low cuboidal epithelium, surrounded by a layer of myoepithelial cells, a basement membrane, and a network of capillaries and lymphatics. The continued growth of the mammary gland during pregnancy was due to increased height of the epithelial cells and expansion of the alveolar lumen. Lactation begins after the dam gives birth. Alveolar lumens were markedly expanded and distended with milk by PND 4 (Figure 7D). Since the mammary epithelial growth is quite extensive from late pregnancy to early lactation, it is difficult to distinguish ducts and adipose tissue on the mammary gland whole mounts due to the dramatic uptake of stain by the LA epithelium (data not shown). H&E-stained longitudinal mammary sections are therefore the best way to evaluate the late pregnant and lactating rat mammary gland.
Hematoxylin and eosin-stained sections of Harlan Sprague-Dawley dam mammary gland during gestation and lactation. Control glands from (A) gestational day (GD) 15, (B) GD 17, (C) GD 21 and (D) postnatal day 4. Original magnification 40×.
Male and female ERα-, PR-, and AR-stained sections
Nuclear ERα and PR in control male and female HSD rat mammary epithelia were not detectable at birth (not shown). In females at PND 8, no nuclear epithelial ERα staining was detected. However, faint cytoplasmic staining was present, which increased in amount and intensity by PND 33 (Figure 8A and B). At PND 70, the cytoplasmic staining was diminished in females (Figure 8C) but remained evident in males (Figure 9C). ERα was detected in epithelial nuclei as early as PND 21 in control females (not shown) but not until PND 33 in males (Figure 9B). PR in the nuclei of control female mammary epithelium was first detected at PND 33 and continued through PND 70 (Figure 8E and F). PR was not present in male mammary epithelium at any life stage examined (Figure 9D–F). AR positive staining was detected in the nuclei of control male mammary epithelium as early as PND 4. Presence of AR was variable but continued to increase in both staining intensity and number of positively stained cells, reaching a peak at about PND 46. By PND 70, AR nuclear receptor staining diminished to low levels in control males (Figure 9G–I). In females, positive nuclear AR-stained mammary epithelium was variable, appearing at about PND 15, and staining was very low throughout development when present (Figure 8G–I).
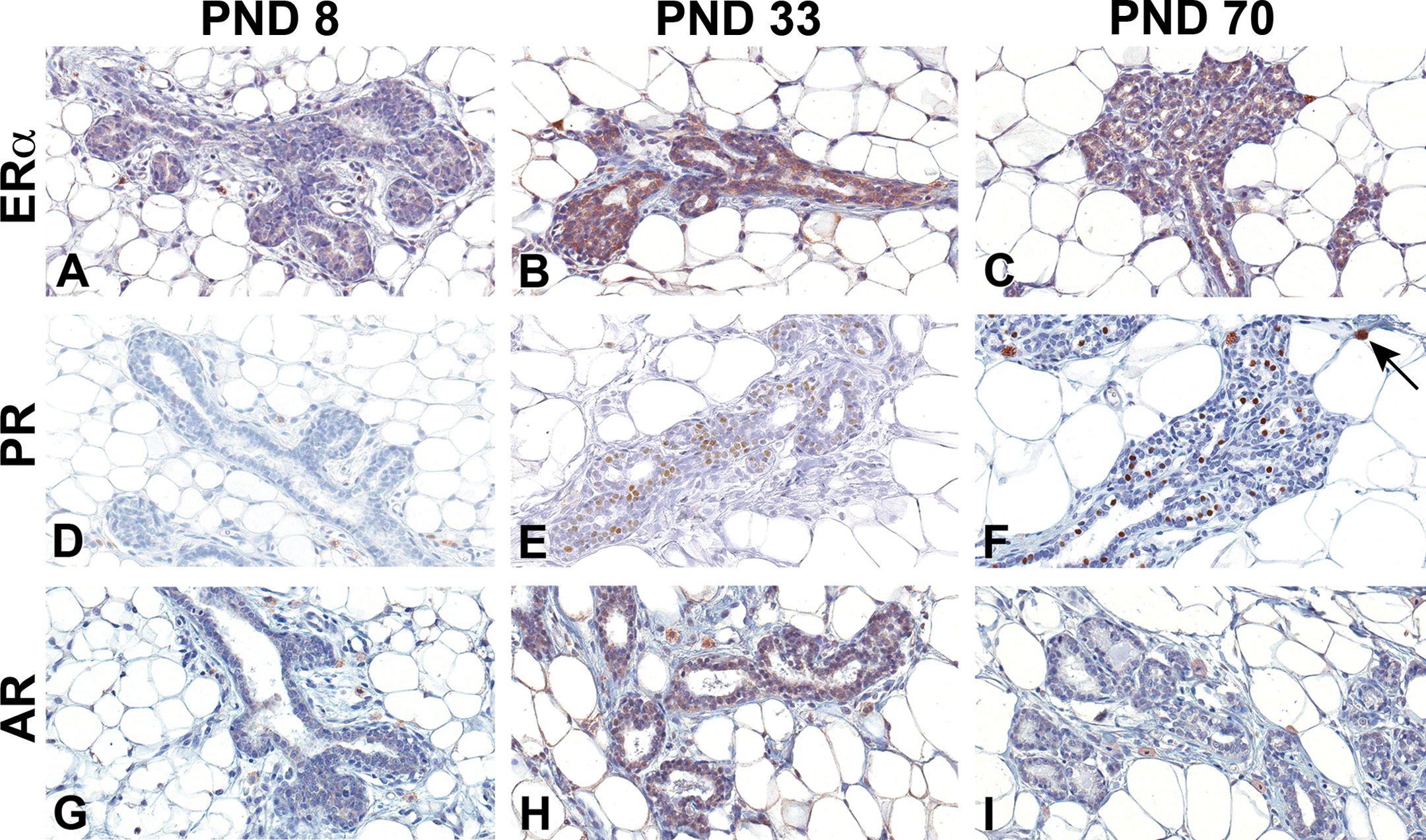
Estrogen receptor alpha (ERα), progesterone receptor (PR), and androgen receptor (AR) immunohistochemistry staining of Harlan Sprague-Dawley female mammary gland epithelium. (A–C) ERα, (D–F) PR, and (G–I) AR. (A, D, and G) Postnatal day (PND) 8, (B, E, and H) PND 33, and (C, F, and I) PND 70. Original magnification 40×. Nuclear staining of ERα became apparent at PND 21, peaked by PND 33, and slightly decreased by PND 70. Nuclear epithelial staining of PR was evident by PND 33 and peaked by PND 46, while AR was consistently low and variable over time in the female mammary gland. There was nonspecific staining of the mast cells in the surrounding stroma (arrow).
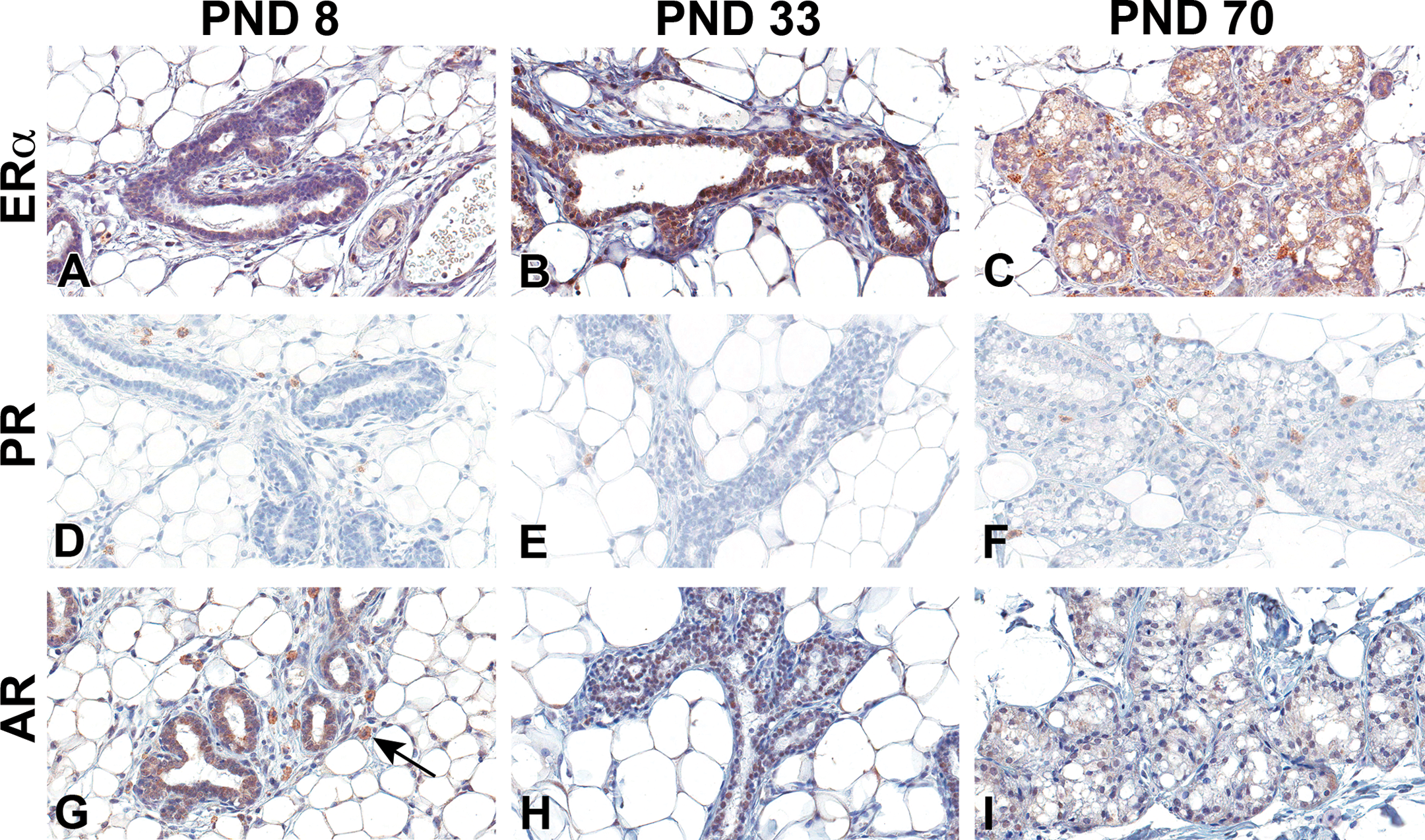
Estrogen receptor alpha (ERα), progesterone receptor (PR), and androgen receptor (AR) immunohistochemistry staining of Harlan Sprague-Dawley male mammary gland epithelium. (A–C) ERα, (D–F) PR, and (G–I) AR. (A, D, and G) Postnatal day (PND) 8, (B, E, and H) PND 33, and (C, F, and I) PND 70. Original magnification 40×. Male mammary gland sections contained low numbers of weakly staining epithelia for ERα. It was evident at PND 33 and remained low over time. AR immunohistochemical staining was evident in the male early, peaked by PND 46, and was found at lower levels in the differentiated adult gland. There was no nuclear staining of PR at any time point. There was nonspecific staining of the mast cells, not a part of the mammary gland epithelium (arrow).
Pup mammary gland whole-mount analysis
Normal mammary gland development in both females and males was demonstrated with whole mounts (Figures 10 and 11, respectively). At PND 1, the mammary gland of female and male HSD rats is composed of a primary duct and several branched secondary and tertiary ducts and are fairly indistinguishable by sex. Mammary epithelium of the 4th gland had extended longitudinally to the lymph node and there were few alveolar buds present at birth (Figures 10A and 11A). From about PND 4 to PND 21, alveolar buds increase in number until they have densely covered most of the ducts. Longitudinal epithelial growth keeps pace with the increased fat pad size, and epithelia has not extended past the lymph nodes in most cases (Figures 10B, C and 11B, C). Very few TEBs have formed by weaning (see TEB counts in Supplementary Table 1). TEBs are the highly proliferative terminal ductal structures primarily responsible for epithelial extension through the fad pad. Between PND 21 and PND 33, there is a 4- to 7-fold increase in the number of TEBs in both males and females, respectively (Supplementary Table 1). Longitudinal growth is rapid, and over this time period, the epithelium has grown past the lymph nodes; lateral growth has also increased and alveolar buds have elongated and branched to form extensive tertiary ducts (Figures 10D and 11D). Also by PND 33, the 4th and 5th glands may have grown together (Figures 10D and 11D; this is consistently apparent in females at this age). As vaginal opening is detected in the female HSD between PND 34–38 (Miousse et al. 2013; Stanko et al., in press), mammary epithelium proliferation accelerates quickly and by PND 46 and PND 70, ductal growth continues to extend to the outer edges of the fat pad (Figure 10E). TEBs have differentiated into terminal ends and the alveolar buds within the gland have become mature structures demonstrating dense branching density (Figure 10F).
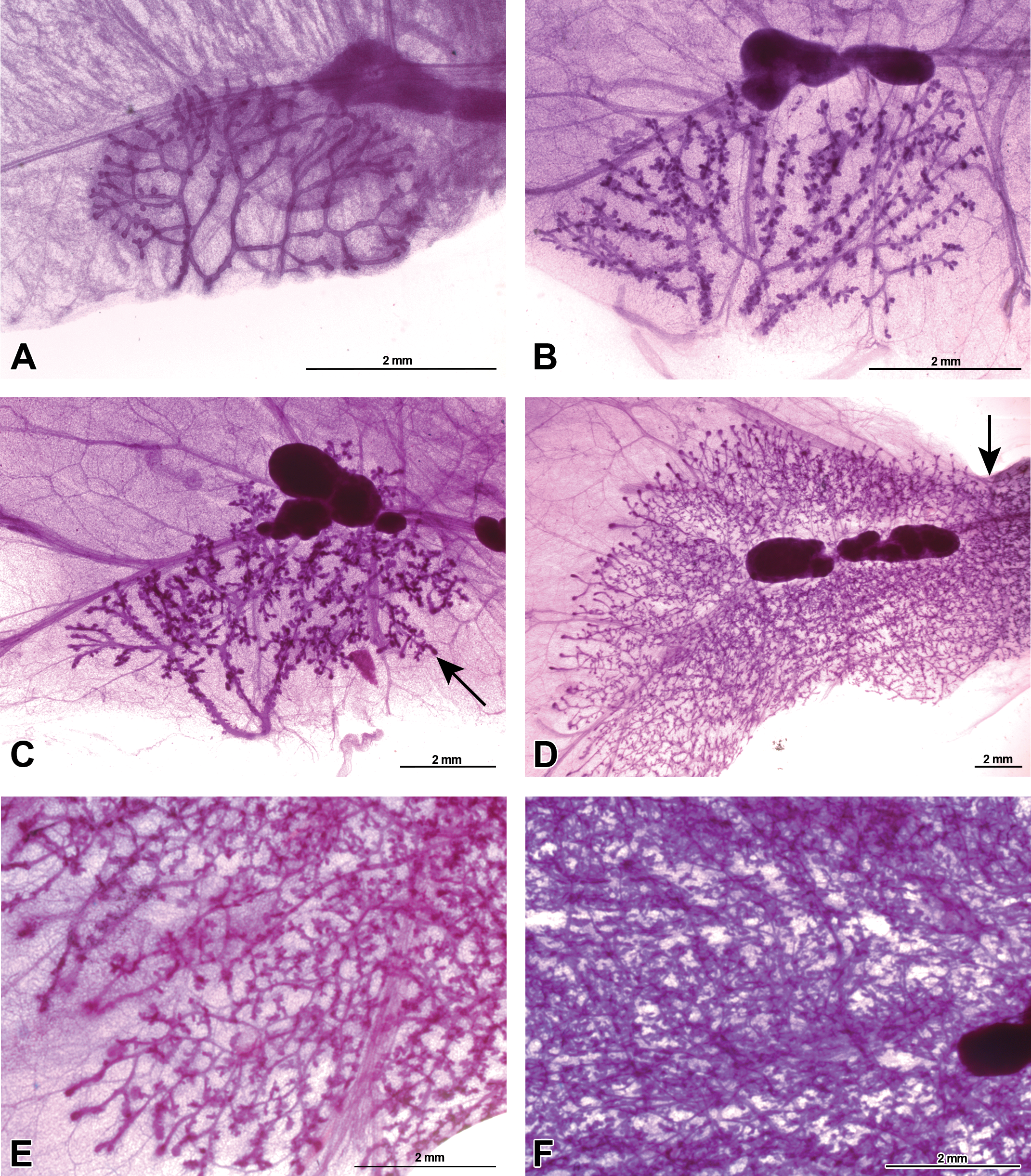
Whole-mounted female mammary gland developmental progression from birth to sexual maturity. Representative carmine stained 4th mammary gland whole-mount images of Harlan Sprague-Dawley rat, picturing the mammary epithelium, inguinal lymph node, and surrounding stroma and fat pad. (A) Mammary gland present at birth on postnatal day (PND) 1. (B) Mammary gland on PND 8 showing increased budding on ducts. (C) Prepubertal mammary gland on PND 21; beginning of terminal end bud (TEB) presence (arrow). (D) Peripubertal mammary gland on PND 33 demonstrating abundant TEB presence, exponential mammary epithelium growth, and convergence of the 4th and 5th gland (arrow). (E) Continued postpubertal mammary gland growth on PND 46 with terminal ends beginning to differentiate at the edges of the fat pad. (F) The mammary gland has filled the fat pad and image shows density of mammary gland epithelium yet ducts are still visible on PND 70. Sizes are shown in figures.
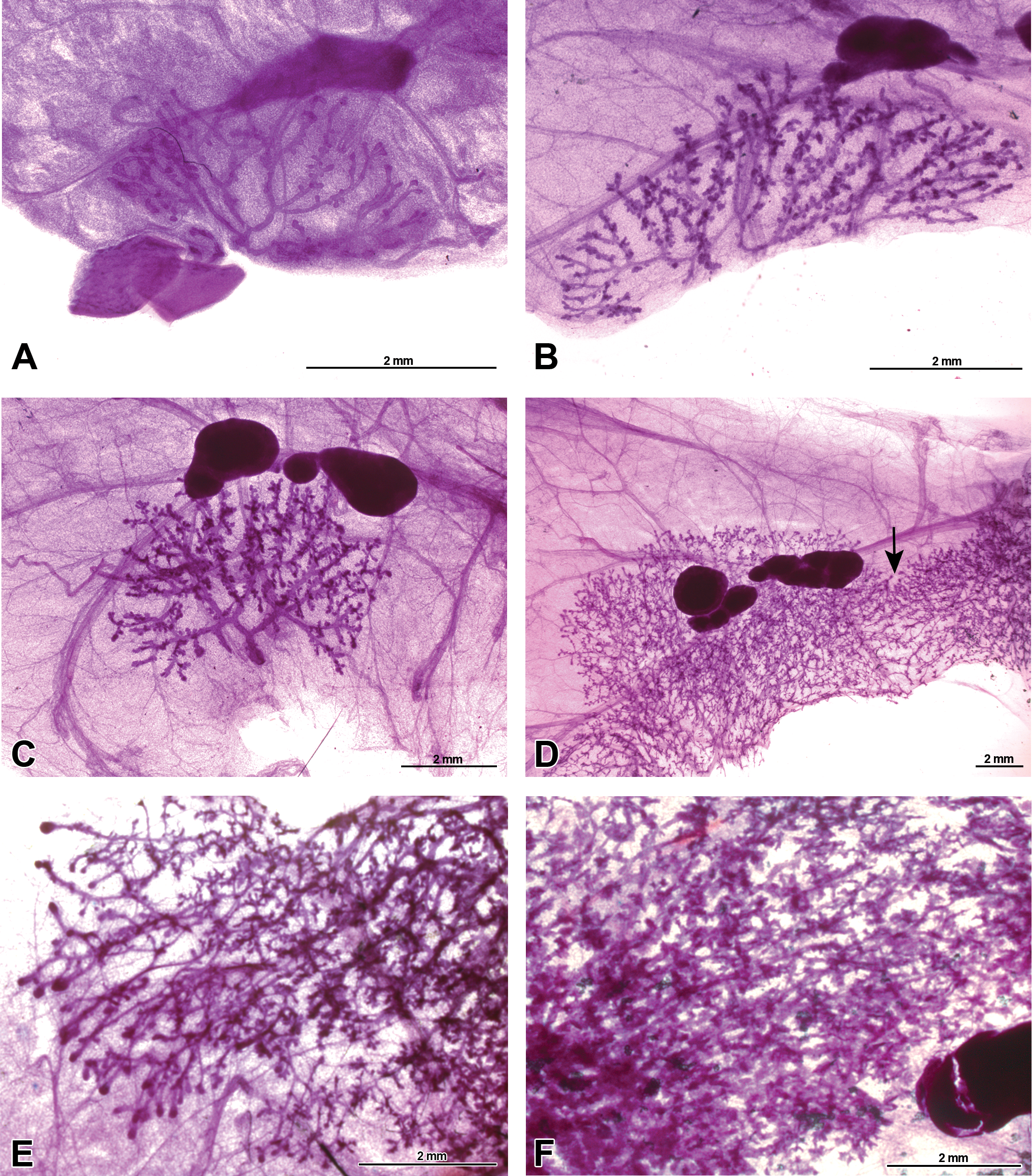
Whole-mounted male mammary gland developmental progression from birth to sexual maturity. Representative carmine stained 4th mammary gland whole-mount images of Harlan Sprague-Dawley rat, picturing the mammary epithelium, inguinal lymph node, and surrounding stroma and fat pad. (A) Mammary gland present at birth on postnatal day (PND). (B) Mammary gland on PND 8 showing increased budding on ducts. (C) Mammary gland on PND 21 demonstrating the beginning of terminal end bud (TEB) presence. (D) Peripubertal mammary gland on PND 33 demonstrating mammary epithelium growth and connection of the 4th and 5th gland (arrow). (E) Continued postpubertal mammary gland growth on PND 46 with terminal ends beginning to differentiate before reaching the edges of the fat pad. (F) The mammary gland fails to fill the fat pad and image shows the enlarged mammary gland lobules and decreased visibility of mammary gland ducts indicative of the sexually dimorphic lobuloalveolar mammary epithelium on PND 70.
Mammary glands developed similarly in both sexes before puberty. About the time that females undergo vaginal opening (PND 34–38), sex-specific differences became apparent. An unusual phenomenon was observed in males in that the 5th gland was not always present in this rat strain. The 5th mammary gland was missing in 50% of males examined (data not shown). Differences between the 4th mammary gland in male and female rats became apparent after PND 21 (Figures 10D–F and 11D–F). The number of TEBs in male mammary glands was consistently lower than in the female mammary gland (Supplementary Table 1). Approaching their first estrous cycle, the female mammary gland grew at a much faster and more uniform rate compared to those of males. By PND 33 in females, the mammary epithelium had grown past the lymph node, the 4th and 5th glands had grown together, and there was lateral growth to the edge of the fat pad. The number of TEBs had reached a maximum, and terminal ends developed where the gland had reached the edge of the fat pad. Compared to female glands, the male glands had reduced longitudinal and lateral growth and fewer TEBs (Figure 11D and E; Supplementary Table 4). There was a greater degree of variability in mammary gland development among the male mammary glands after PND 21. Of the animals that had a 5th gland, not all had merged with the 4th gland at PND 33. These factors made selecting a representative image much more difficult. The epithelial area of male mammary glands changed little after PND 33, and in most the glands did not ever reach the edges of the fat pad. In female mammary glands, TEBs began to diminish as the gland reached the end of the fat pad at PND 46, while in males, TEBs began to differentiate into terminal ends prior to reaching the end of the fat pad (Figure 11E). By PND 70, very few TEBs were found in the female mammary gland (Supplement Table 1). The male mammary gland became very dense and exhibited the appearance of a sexually mature male mammary gland by PND 70, with prominent alveolar sacs and ducts that were barely visible (Figure 11F).
Chemical-induced changes in mammary gland development
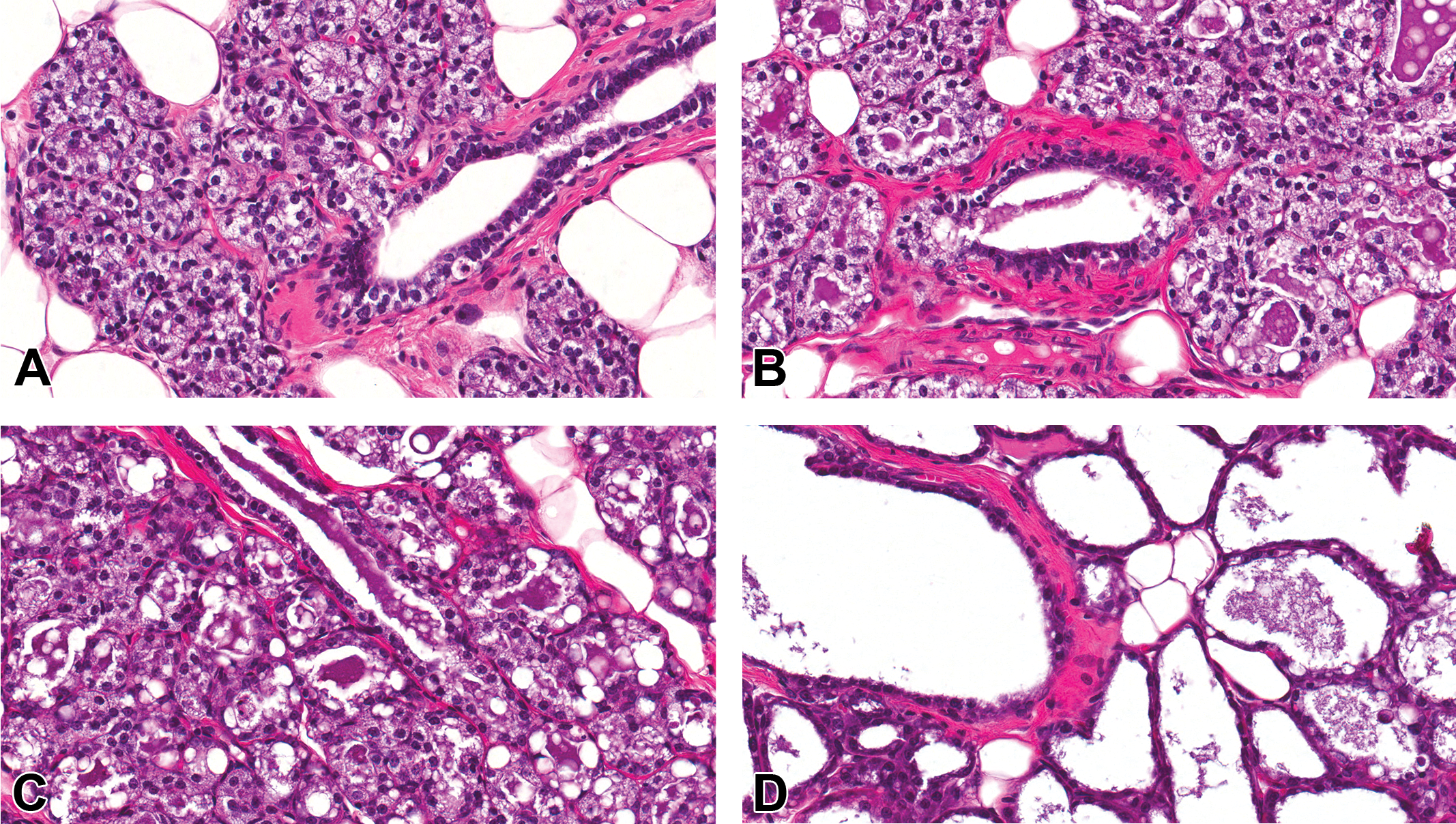
DES was administered to a subset of pregnant dams during embryonic mammary gland organogenesis. TCDD, a ligand for the aryl hydrocarbon receptor, was administered to a different subset of pregnant dams. Whole-mount evaluations proved to be a powerful tool for detecting effects of these chemicals on the mammary gland. Mammary tissue from TCDD-exposed males visually contained fewer terminal branches and lobules when compared to control animals (PND 33 shown; Figure 12A vs. C). TCDD induced stunted epithelial outgrowth, fewer branches, and decreased bud development (i.e., delayed mammary gland development), which persisted from PND 4 to 46. At PND 70, the mammary epithelium of TCDD-exposed males was more similar to age-matched control males, and delays in branching were less obvious. The reduced branching was confirmed by H&E-stained sections (Figure 12B and D).
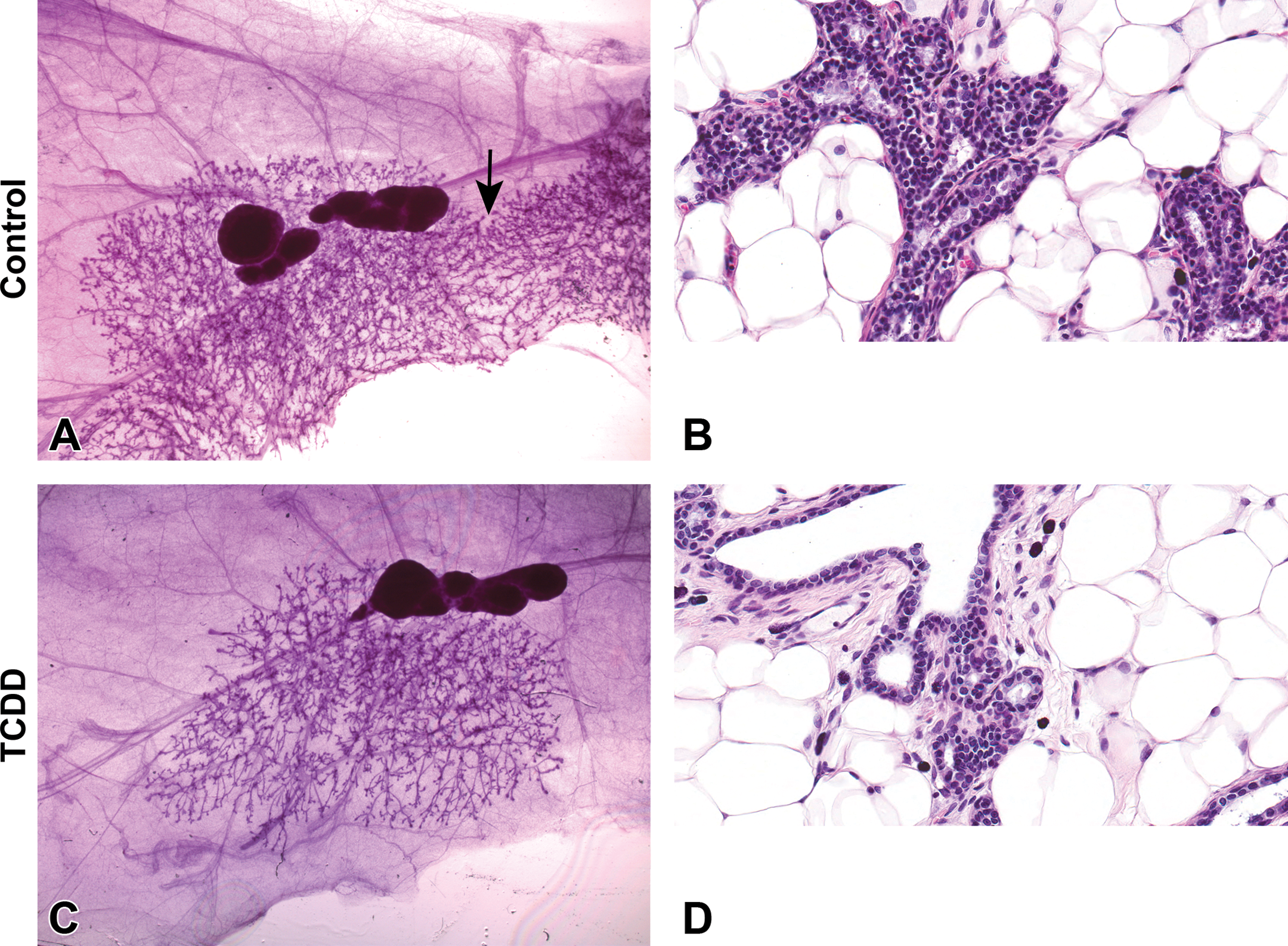
Whole-mount and histology comparison of mammary gland development of control and prenatally 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)-exposed Harlan Sprague-Dawley (HSD) males at postnatal day 33. Representative carmine stained 4th mammary gland whole-mount images (A and C) and H&E-stained histology sections (B and D) of control (A and B) and TCDD (C and D) prenatally exposed offspring. (A) Mammary whole mount of control HSD male demonstrating normal lateral growth of the 4th gland past the lymph node and the presences of the 5th gland and its connection to the 4th gland (arrow). (B) H&E section has normal tubuloalveolar morphology characterized by numerous branching from the major duct. (C) Mammary whole mount of HSD male gestationally treated with TCDD demonstrating stunted lateral mammary gland branching and absence of the 5th gland, which was not specific to TCDD treatment, (A, C) original magnification 0.8×. (D) H&E section has normal tubuloalveolar morphology but with reduced side branching of ducts, (B, D) original magnification 40×.
Similar to males, the mammary glands of TCDD-exposed females exhibited delays in mammary gland development, characterized by fewer terminal branches and TEBs when compared to control animals (PND 21 weanlings shown; Figure 13A vs. C; Supplemental Table 1). Figure 13 also highlights the advantages of the whole mount compared to histology sections for detecting mammary gland developmental delays; even though the epithelial tree of TCDD-exposed animals is correct in structure, it lacked the branching and budding that was evident in untreated controls. TEBs were also evident in controls at PND 21, whereas they were absent in TCDD-treated animals, a finding that was confirmed in H&E-stained sections (Figure 13B and D). At PND 33, the control animals exhibited convergence of the 4th and 5th mammary glands, an event that was delayed in TCDD-treated females (not shown). The growth of the mammary epithelium in TCDD-exposed females had not caught up to that of the controls by PND 70 and failed to reach the ends of the fat pad.
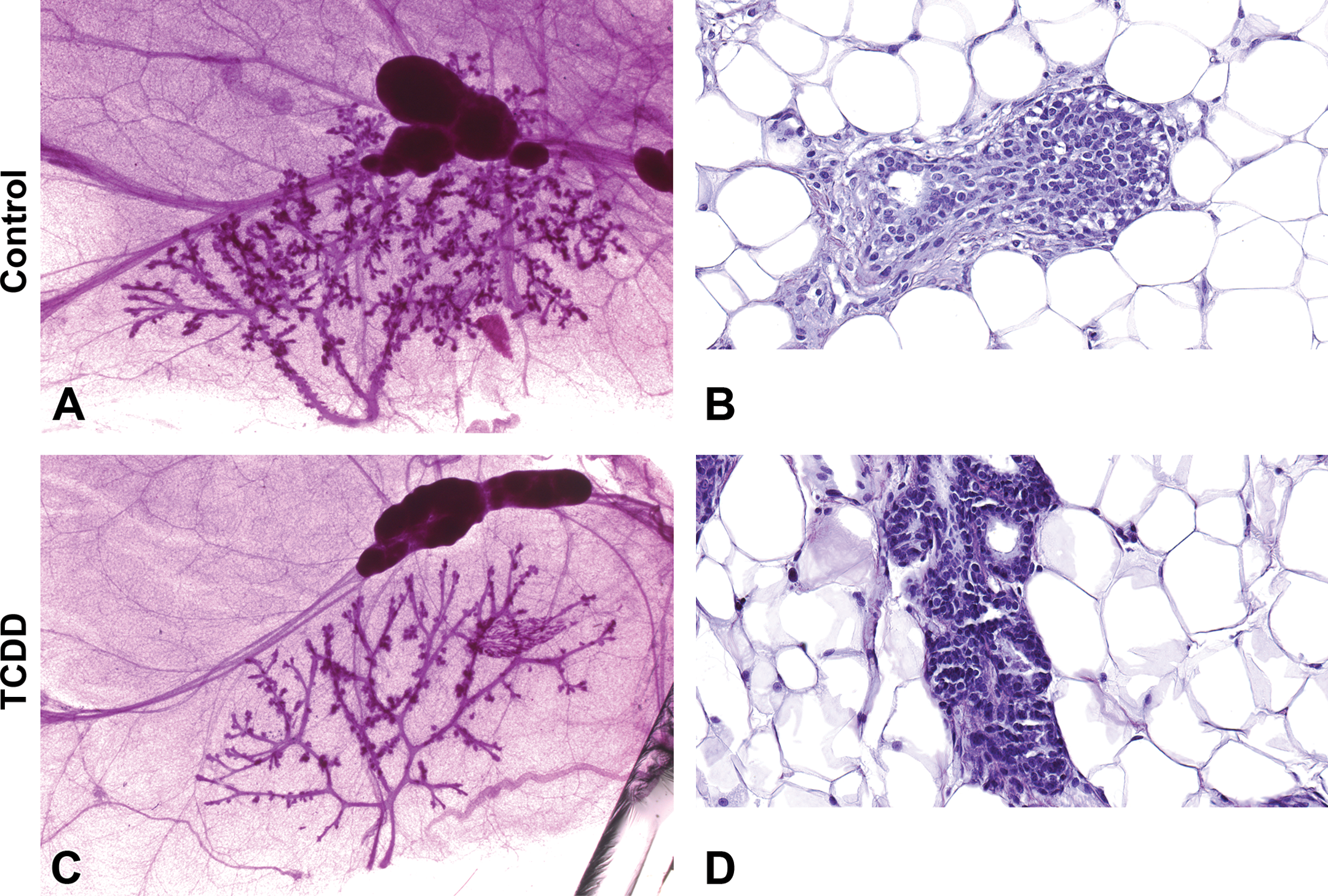
Whole-mount and histology comparison of mammary gland development of control and prenatally 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)-exposed Harlan Sprague-Dawley (HSD) females at postnatal day 21. Representative carmine-stained 4th mammary gland whole-mount images (A and C) and H&E-stained histology sections (B and D) of control (A and B) and TCDD (C and D) prenatally exposed offspring. (A) Mammary whole mount of the 4th gland of the control HSD female demonstrating normal lateral growth just past the lymph node and extensive side budding of the ducts. (B) H&E section of normal tubuloalveolar morphology and terminal end bud (TEB) development. (C) Mammary whole mount of the 4th gland of the HSD female gestationally treated with TCDD demonstrating stunted mammary gland growth, less extensive side budding of the ducts, and failure to form complex ends, (A, C) original magnification 2×. (D) H&E section characterized by normal tubuloalveolar morphology with small ductal buds, (B, D) original magnification 40×.
Quantitative digital image analysis was conducted on all H&E-stained sections from control and treated females (7 age-groups). This analysis agrees with data from whole-mount analyses; it further confirmed that epithelial area is significantly reduced following TCDD treatment compared to controls (Table 1). Furthermore, there is a suggestion that epithelial nuclear density, an additional measure of epithelial content, may also be reduced, though not significantly. These digital analyses were performed blinded to treatment status and therefore provide an objective, quantitative measure of epithelial changes induced by treatment.
Age-adjusted Treatment Associations with Digitally Quantified Epithelial Measures, among Female Rats Less than 70 Days.
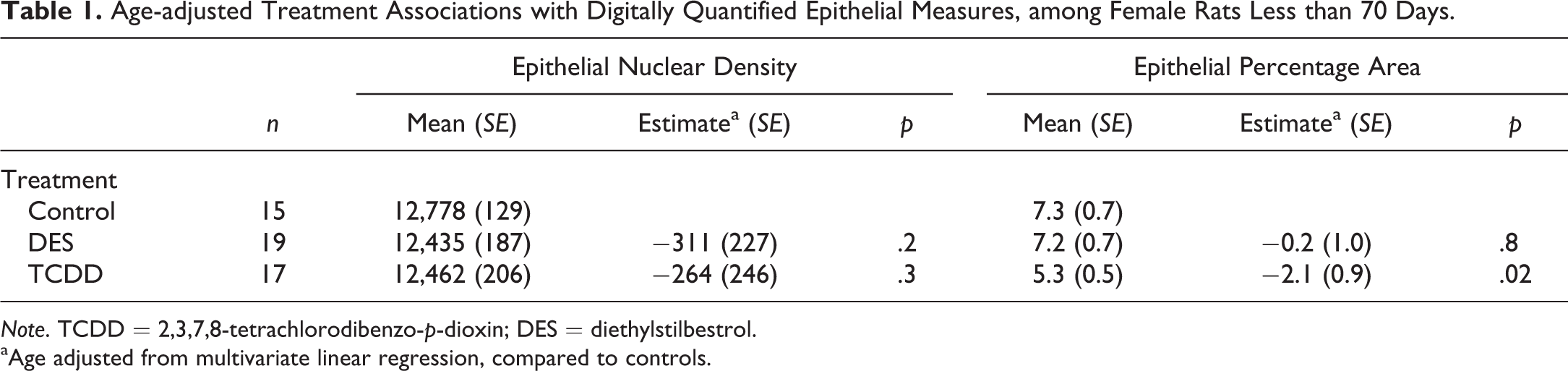
Note. TCDD = 2,3,7,8-tetrachlorodibenzo-p-dioxin; DES = diethylstilbestrol.
aAge adjusted from multivariate linear regression, compared to controls.
The dose of DES chosen in our studies had little effect on the mammary epithelium of female offspring. In making qualitative comparisons between the whole mounts from the DES-exposed females and controls, the DES-treated prepubescent female glands (PND 8–21) appeared more dense and exhibited more alveolar budding on the ducts, and the 4th and 5th glands of both control and DES-exposed females had grown together by PND 33 (data not shown). A tubuloalveolar structure was present in H&E-stained section with no appreciable difference in cellular structure or organization between the treatment groups (data not shown). Similarly, the mammary epithelium of DES-exposed and control males was indistinguishable from each other from PND 4 to PND 46 (data not shown). However, TEBs were more abundant in DES-treated males compared to controls at PND 33 (Supplementary Table 1). At PND 70, when the control mammary glands became sexually mature, mammary tissue from DES-exposed animals contained typical LA structures centrally, matching the appearance of the control; however, the outer edges of the mammary gland displayed a tubuloalveolar structure characterized by a predominance of ducts and fewer alveoli (Figure 14A, B vs. C, D). These findings may suggest that the DES-exposed males were feminized, or the mammary epithelium exhibited a delay in maturation. Quantitative analyses of the epithelial area on mammary section from control and DES-treated females showed no differences between the groups (not shown).
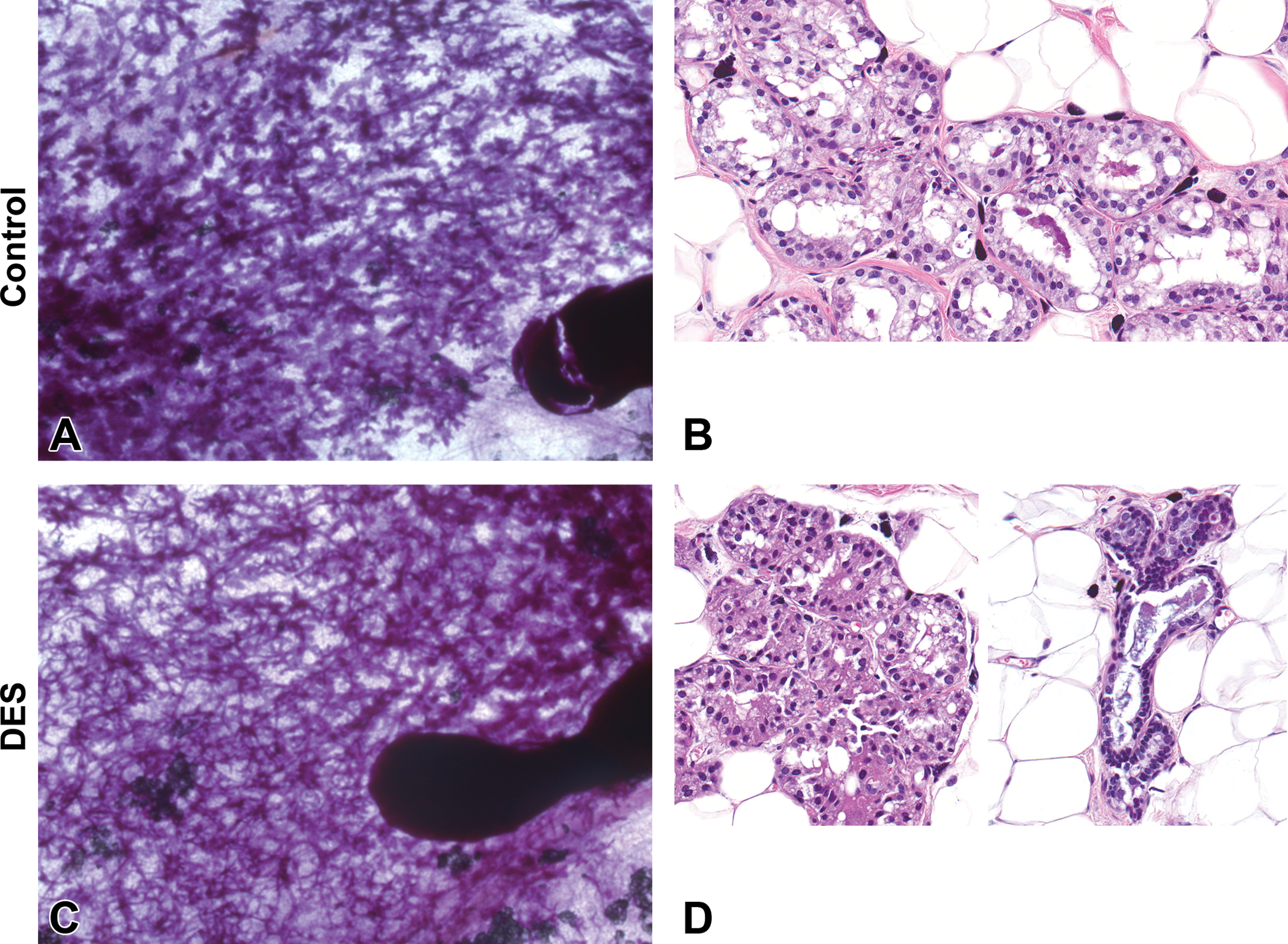
Whole-mount and histology comparison of mammary gland development of control and prenatally diethylstilbestrol-exposed males at postnatal day 70. Representative carmine-stained mammary gland whole-mount images (A and C) and H&E-stained histology sections (B and D) of control (A and B) and 2,3,7,8-tetrachlorodibenzo-p-dioxin (C and D) prenatally exposed offspring. (A) Mammary whole mount of control Harlan Sprague-Dawley (HSD) male demonstrating increased density of the mammary epithelium with indiscernible ducts. (B) H&E section of normal lobuloalveolar morphology. (C) Mammary whole mount of control HSD male demonstrating dense mammary epithelium with discernable ducts, (A, C) original magnification 2.5×. (D) H&E section of normal lobuloalveolar morphology of the mammary epithelium located at the center of the mammary gland (left) and ducts and tubules with morphologic characteristics similar to those observed in female rats (tubuloalveolar morphology) at the periphery of the mammary gland (right), (B, D) original magnification 40×.
Although DES appeared to have limited effects on mammary morphology in male and female pups, its effects on nuclear receptor staining were evident in both sexes (Figure 15). Quick Scores of nuclear receptors from control and treated gland sections demonstrated that ERα-positive nuclei were observed in mammary glands of DES-exposed females on PND 15, while labeled nuclei were not detected in control mammary glands until PND 21 (Figure 15A). There was an overall decrease in AR staining in males (Figure 15D). Some, but not all, TCDD-exposed animals expressed ERα at PND 21 (Figure 15A). ERα was observed for the first time in mammary glands of the control, DES-exposed, and TCDD-exposed males at PND 33. After PND 33, ERα in male mammary glands was very low, if present at all (Figure 15B). PR in mammary epithelium of control and DES-exposed female rats was first observed at PND 33 but was delayed in appearance in mammary epithelium of TCDD-exposed females until PND 46 (Figure 15E). While maximum AR-stained mammary epithelium was observed at PND 46 in all males, the Quick Score was lower in glands of DES-exposed males compared to those of other males. Mammary glands of control males typically exhibited slightly higher Quick Scores and more consistent AR-stained mammary epithelium than glands of chemically exposed males (Figure 15D). The Quick Score results are summarized over time, by sex, in Figure 15, and raw data are reported in Supplementary Tables 2, 3, and 4. Although interesting results are shown here, these studies were not powered to perform statistical analyses and should be considered preliminary and followed up in future studies.
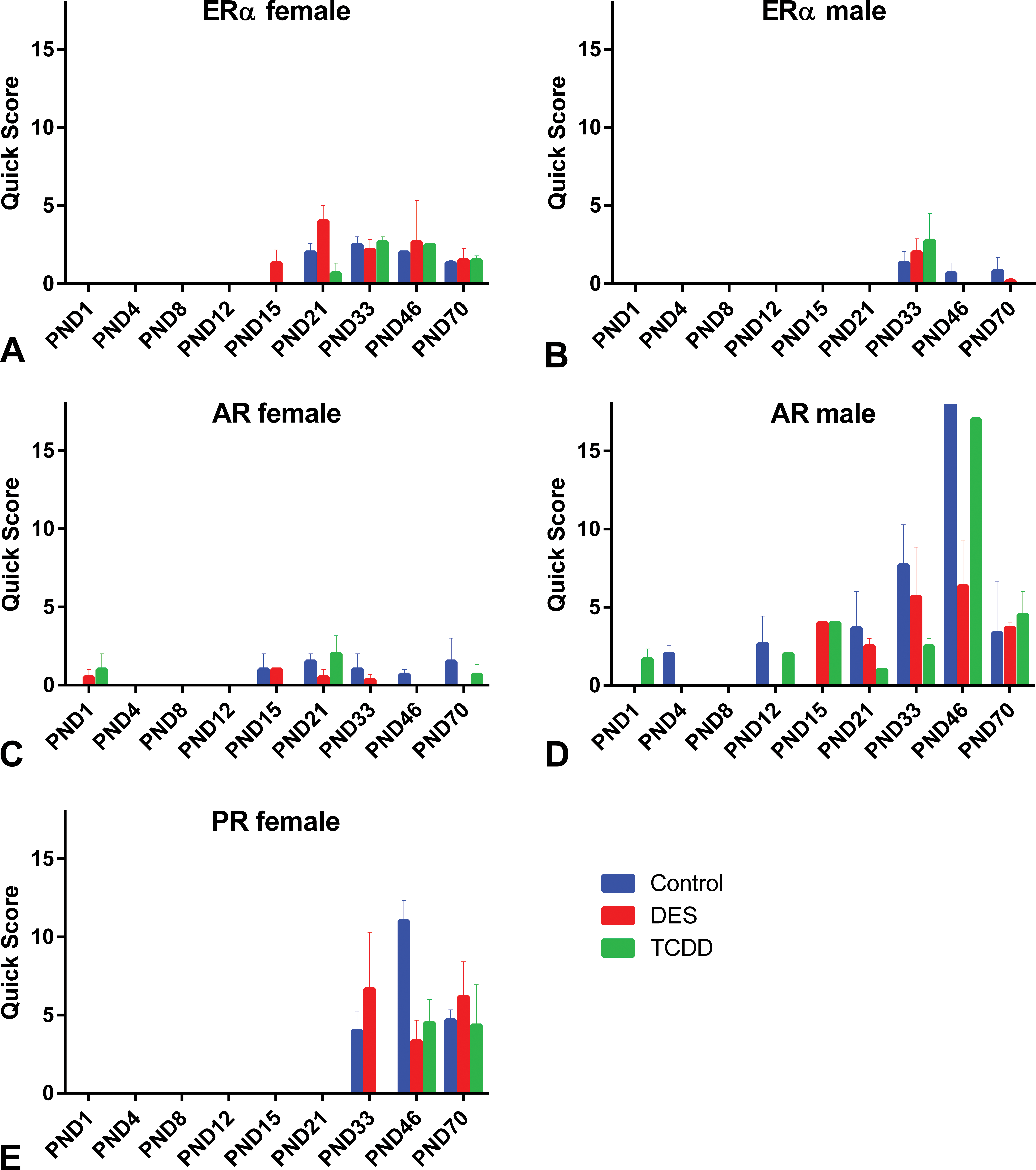
Estrogen receptor alpha (ERα), progesterone receptor (PR), and androgen receptor (AR) quantification in female and male offspring mammary glands. Quick Scores were determined for the nuclear receptor staining. (A) Nuclear ERα first appeared at postnatal day (PND) 15 in the diethylstilbestrol (DES)-exposed females and PND 21 in control and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)-exposed females, peaking at PND21 in DES-treated females and PND33 in control females. (B) Nuclear ERα first appeared at PND 33 in the males. (C) There was very little AR staining in the females and was sporadic in the chemically exposed animals and more consistent in the control females. (D) AR-stained male mammary epithelia at low levels until puberty in control and TCDD-exposed animals. DES had a less intense nuclear staining. (E) PR was only expressed in the females and first appeared at PND 33 in control and DES-exposed animals and PND 46 in TCDD-exposed animals. Values from this figure are shown in Supplementary Tables 2, 3 and 4.
Compiled literature on the progression of mammary gland development and hormones levels in female rats
Figure 16 is a compiled literature search on what is currently known about female rat mammary gland development with regard to receptors present in the mammary gland and sex hormone levels throughout the life span (mostly in Charles River Sprague-Dawley rat). Male and female mammary gland development and hormone receptor data from our studies have been added to the figure to fill in data gaps. Specifically, we have added information on the ages at which steroid receptors are expressed in epithelial cells during development, the branching activity in male glands, and details of male gland development in general. Hormone levels are presented as a range, as all studies did not collect the hormone information in the same stage of the estrous cycle, eliminating the opportunity for us to plot as curves or provide standard errors.
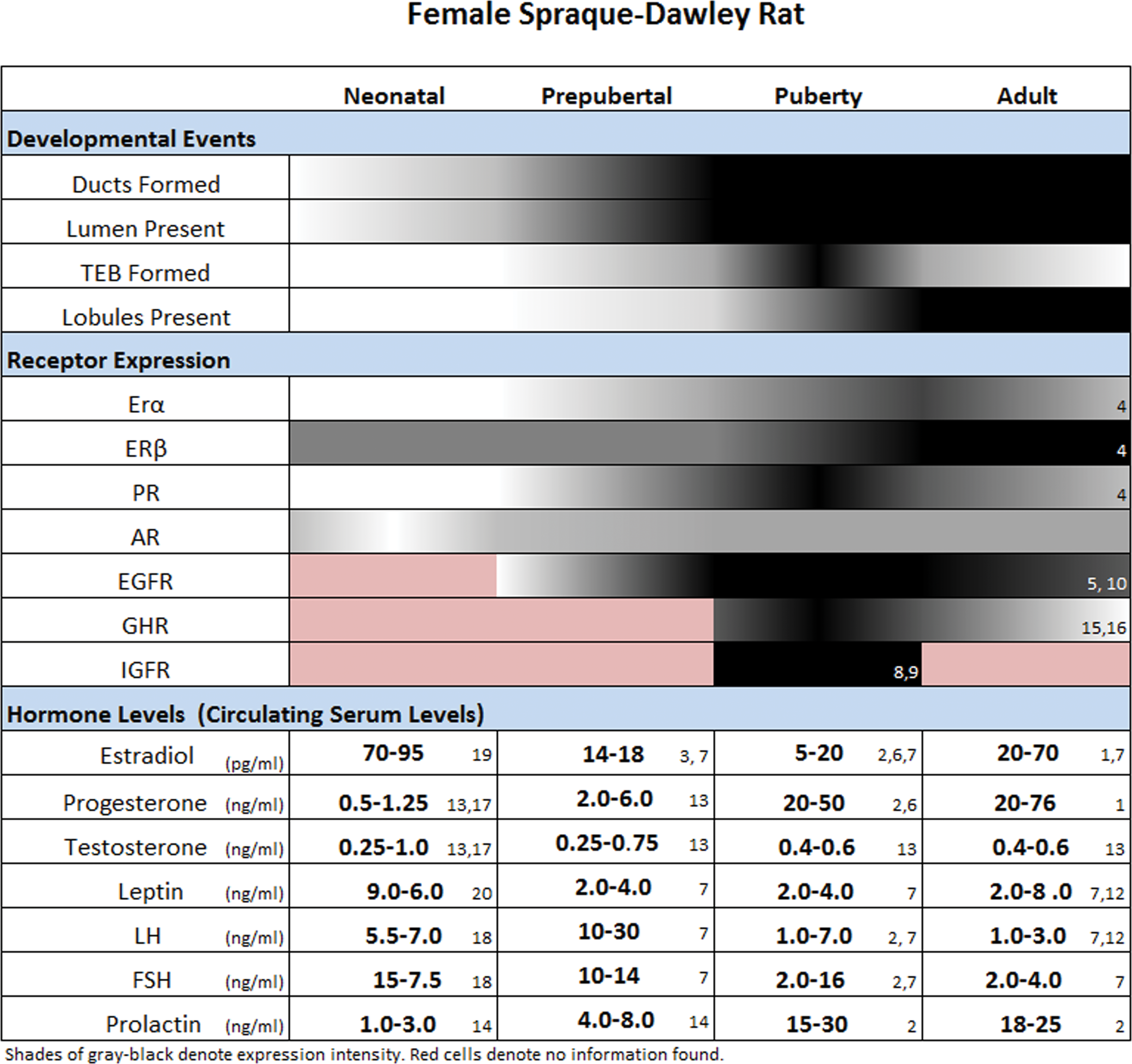
Compiled literary data on the female rat. The developmental events, receptor expression of the mammary gland, and the range of hormone levels in the serum of female rats (largely Charles River CD1 origin), from neonates to adults. Darkest bar intensity represents strongest expression/presence. 1 (Emanuele et al. 2001), 2 (Biegel et al. 1998), 3 (Kim et al. 2002), 4 (Saji et al. 2000), 5 (Kariagina, Aupperlee, and Haslam 2008), 6 (Miousse et al. 2013), 7 (Cheung et al. 2001), 8 (Cotroneo et al. 2002), 9 (Tan et al. 2004), 10 (Thordarson et al. 1995), 11 (Hvid et al. 2011), 12 (Kao, Hiipakka, and Liao 2000), 13 (Dohler and Wuttke 1975), 14 (Darcy et al. 1999), 15 (Weisz and Ward 1980), 16 (Wilson and Handa 1997), and 17 (Frawley and Henricks 1979).
Discussion
This study has generated previously unknown information on the HSD mammary gland development. First, we report that the 5th gland was missing in about 50% of the HSD males in our study, a phenomenon previously reported only in the male Wistar Han rat (Mandrup et al. 2012). Second, we detailed male mammary gland development in the HSD rat (which has not been extensively studied), and we have shown that the HSD male mammary gland is structurally similar to the females until the females undergo vaginal opening. Third, the male mammary gland grows at a slower rate than the females beginning about PND33 and stopped growing in overall area after about PND 45, which coincides with the timing of male puberty and increased serum testosterone levels (Stoker et al. 2000). The male mammary gland stopped growing before reaching the end of the fat pad, while the female mammary epithelium continued to extend until it reaches the end of the fat pad. Fourth, we report the presence/absence of steroid hormone receptors during mammary development, which has not been extensively studied during early prepubertal time points, especially in the male. We demonstrate neonatal and prepubertal AR presence in the female HSD mammary glands and neonatal through adult ERα and AR presence in male HSD mammary glands, along with the complete absence of PR in the male gland. Finally, we report for the first time the effects of TCDD and DES on developmental patterns in the male mammary gland.
Our data also confirm that the HSD mammary gland undergoes similar stages of development, as has been previously shown in other rat strains, particularly in the more extensively studied female rat (Fenton et al. 2002; Rayner, Enoch, and Fenton 2005; Moral et al. 2011; Mandrup et al. 2012). However, the pace of development is accelerated in this strain, as has been recently reported in a 3-strain comparison of rats (Stanko et al. in press). By PND 33, 100% of the 4th and 5th mammary glands in the control females had already grown together (data not shown). This event does not occur until around PND 45 in other strains, such as Charles River Sprague-Dawley (Stanko et al. in press) and Long Evans rats (Rayner, Enoch, and Fenton 2005). This information is vital for future carcinogen-induced mammary tumor model studies in rats, as the HSD female rat may have a smaller window of susceptibility to mammary carcinogens or EDCs, compared to other rat strains.
The adult female rat mammary gland has been extensively studied but developmental time points are poorly understood. The pictorial atlas developed within these studies confirms important information on the development and timing of the mammary glands of male and female HSD rats. The female mammary gland that is undergoing lactation or involution has been well documented (Figure 7), but embryogenesis and puberty are other critical windows of susceptibility that deserve more attention. The developmental events in Figure 16 were characterized in the female HSD rat in these studies. Once the male reached sexual maturity, there was a structural change of the mammary gland from a tubuloalveolar to a LA structure, which has been previously described in other adult rat strains, but the precise mechanism(s) for the slowed growth and shift in morphology is unknown (Cardy 1991; Rudmann et al. 2005; Lucas et al. 2007).
The mammary gland bud developed by E15.5 in males and females, and on E18.5, the male mammary glands became sexually dimorphic due to the first male androgen surge (Ward et al. 2003). Androgens interact with the dense mammary mesenchyme and triggered the destruction of the mammary bud in what appeared to be 2 main actions. First, the mammary mesenchyme condensed around the neck of the mammary bud and severed its connection to the epidermis. Second, there was a massive wave of apoptosis that eliminated the mammary mesenchymal cells (Abdalkhani et al. 2002). In both males and females, the epithelial ducts with a lumen were present at birth and continued to rapidly expand through to adulthood. TEBs began to form just before puberty, reaching a peak around PND 33, and differentiated into mature lobules once reaching the end of the fat pad or other mammary glands at the beginning of adulthood.
Chemically exposed HSD female rats showed expected and novel results. The TCDD-treated HSD rat also demonstrated smaller and developmentally delayed mammary glands starting at PND 8 that remained underdeveloped by PND 70 in females and until PND 46 in males. By implementing a quantitative digital analysis algorithm, we recapitulated the qualitative scoring in a treatment-blinded, objective method. Furthermore, this method allows assessment of parameters that are also relevant in human tissues (Sandhu et al. 2016), facilitating the animal-to-human approach advocated by the IBCERCC (2013).
This is the first demonstration of TCDD-induced delays in male mammary gland development in any strain of rat. The model chemical we chose for accelerated growth, DES, did not result in noticeably accelerated mammary gland development in the female, although changes in TEB counts and nuclear receptor staining were evident in males and females. Feminization of the mammary epithelium may have occurred in the males exposed to DES in utero. However, it may be normal for the outer edges of male rat mammary glands to display a tubuloalveolar structure while proliferating TEBs are still present (OECD 2008). We could not determine the persistence of these differences, as PND 70 was the oldest age evaluated. This strain of rat already exhibits accelerated mammary gland development compared to other strains (Stanko et al. in press), therefore it may be more difficult to quantitate accelerated development in females without the use of biomarkers such as receptor staining status or TEB counts. Further, the DES dose that was used in our study may have been too low for the effects we anticipated. Other studies have used injected route of exposure, higher doses, and longer exposure periods than those used here (Ninomiya et al. 2007; Hovey et al. 2005; Fielden et al. 2002).
The steroid hormone receptors, estrogen and progesterone, have been extensively studied in the adult rodent mammary gland (Dohler and Wuttke 1975; Biegel et al. 1998; Emanuele et al. 2001; Kim et al. 2002), and their presence or absence in the female gland over time is shown in Figure 16. The androgen and protein receptor categories were less well defined in the mammary gland literature and there are currently data gaps in the neonatal and peripubertal time points for some of these receptors. The ERα, PR, and AR staining performed in these studies added information to Figure 16, as staining during developmental periods were seldom reported. The findings in this study agree with previously published ERα data (Saji et al. 2000) in female Sprague-Dawley rats in that no nuclear staining was observed until PND 21 in the controls. PR was not found in neonatal females, but as previously reported in other strains (Kariagina, Aupperlee, and Haslam 2008), it is present in prepubertal and adult females. AR-staining data added new information to the table, demonstrating that AR was not present in mammary epithelium nuclei until PND 15 in female controls and only very low expression through PND 70. DES-exposed animals were the only group to have nuclear ERα present at PND 15 and TCDD delayed the presence of PR until PND 46. AR expression was very low in females and often variable, but DES-exposed animals very rarely had any expression at any time point. The male ERα, PR, and AR-staining data elucidate the differences and similarities between males and females during key moments of mammary gland development that previously have not been studied. Differences in steroid hormone receptor populations give insight into when EDCs may have their maximal effect on the mammary gland. ERα expression, while it had very low overall expression levels in males, was present by PND 33, which is over a week later than it is seen in females and is present at least a week before the males undergo preputial separation. PR expression was not detectable in the male mammary glands at any time point evaluated. AR expression was much higher in male mammary glands compared to females and peaked at PND 46. DES treatment reduced the amount of AR expression at PND 46 compared to other treatments. The ranges of normal circulating serum hormone levels of nulliparous female rats starting at puberty and through adulthood are presented in Figure 16, but because of variation in collection and reporting in those studies, we were unable to condense the information into a more user-friendly presentation and present these data as ranges of hormone levels.
Although this study had limitations such as low power, which kept us from providing statistical differences, it is our hope that this pictorial atlas will serve as a guide for mammary gland development in fetal HSD rats and enhance the assessment of juvenile mammary gland evaluation in chemical screening and testing studies. The mammary gland is unique in that the epithelial bud originates during gestation, but the majority of development occurs after birth. This study highlights the potential immediate or later life effects on the developing mammary gland due to EDCs that might be missed in adult-only exposures rather than exposure during critical windows of mammary gland susceptibility.
We encourage scientists to produce and evaluate mammary gland sections or whole mounts from male and female offspring in their studies. There are now published protocols (Davis and Fenton 2013; Plante et al. 2011) that enhance any laboratory’s ability to make high-quality mammary gland whole mounts. Increased preparation/examination of whole mounts within every EDC study of rats or mice would help detect chemically induced changes that could easily be missed by routine processing and evaluation of a single section. If whole mounts are to be prepared, we suggest some of the ages for analysis that were chosen in this study, as they represent the ages most often used for mammary gland comparisons in a variety of research studies; PND 4 (culls from litter size equilibration), PND 21 (extra weanlings), PND 46 (about the age chemical carcinogen is delivered), and PND 70 (young adult, similar to PND 90). If histology sections are the only analysis of the mammary gland, longitudinal sections of the mammary gland, free of skin, are highly recommended over transverse cross sections that may include the skin, as they provide much more mammary tissue to evaluate in a single section and may include information from both 4th and 5th inguinal glands. In fact, whole mounts can now be evaluated, digitally documented, and sectioned for histological evaluation as described (Tucker et al. in press).
Mammary gland development in the HSD strain is similar to others rat strains but demonstrates key differences in rate of mammary gland growth (faster than others), dense branching by the time of puberty, and the fairly common absence of the 5th mammary gland in males. The female HSD rat expresses steroid hormone receptors in the mammary gland in a way that is similar to other strains and should provide useful information that will inform on mode of action for chemicals that shift steroid hormone receptor nuclear localization patterns. For example, an acceleration of lateral and longitudinal growth may indicate an estrogenic or antiandrogenic chemical effect, decreased branching may suggest that progesterone or growth factor signaling pathways are disrupted, or epigenetic changes may explain alterations that are persistent but are not apparently hormone related. In order to prevent breast cancer and precocious breast development in girls around the globe, we must understand some of the environmental factors adversely affecting the breast. Further evaluation of the male and female mammary gland in screening and test guideline studies will help produce the data needed to move this process forward.
Footnotes
Author Contribution
Authors contributed to conception or design (JF, SF); data acquisition, analysis, or interpretation (AB, AF, PB, LA, JF, SP, MT, BM, SH, CR, VC, SF); drafting the manuscript (AF, JF, SH, CR, VC, SF); and critically revising the manuscript (AB, AF, PB, LA, JF, SP, MT, BM, SH, CR, VC, SF). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Adam J. Filgo was funded by a NIEHS Individual Research Training Award. Aditi R. Borde, Lydia B. Alexander, and Pretish R. Borde were funded by the NIEHS Summer Intern Program.
