Abstract
The objective of this study was to determine the shortest period of time necessary to detect histologic evidence of estrous cycle disruption in Sprague-Dawley rats treated for up to 28 days with the aromatase inhibitor exemestane at 1,000 mg/kg. Rats were evaluated on day 5, 8, 15, or 29. Vaginal mucification, uterine and cervical epithelial atrophy, uterine luminal epithelial vacuolation, decreased uterine granulocytes, and hypertrophy/hyperplasia of mammary ducts and alveoli were noted by day 5 and persisted throughout the study. From day 8 to day 29, absence of recent basophilic corpora lutea, increased atresia of antral follicles, interstitial cell hyperplasia, and increased luteinized follicles were present in the ovaries of treated rats. Vaginal smears detected persistent diestrus, confirming estrous cycle disruption between days 5 and 8. Ovary and uterine weights were largely unaffected. Serum hormone levels were not useful due to the study design employed. Other effects of exemestane included decreased adrenal weights and decreased cell size in both the adrenal zona fasciculata and the pituitary pars distalis. While early histologic changes were evident on day 5, only after 8 days of treatment were findings considered sufficient to clearly identify exemestane-induced estrous cycle disruption using microscopy alone.
Introduction
The female reproductive tract undergoes recurring physiological and morphological changes that are driven by the cyclical fluctuations in hormone levels that occur across the ovarian cycle. In the laboratory rat, these changes have been well characterized cytologically and histologically, and their assessment in preclinical toxicology studies serves as a means of monitoring the health and functionality of the hypothalamic-pituitary-ovarian (HPO) axis (reviewed in Goldman, Murr, and Cooper 2007; Davis, Travlos, and McShane 2001; Westwood 2008). Before a new drug candidate may enter clinical trials, repeat-dose preclinical toxicology studies are conducted to assess potential hazards and risk for human exposure. In general, these safety studies are conducted in two mammalian species (one rodent, one nonrodent) for periods that equal or exceed the duration of the planned human clinical trials. However, consistent with internationally recognized guidelines for the conduct of clinical trials for medicinal products, candidate compounds are routinely evaluated in human clinical trials prior to formal preclinical safety testing for effects on female reproductive function (ICH 2009). Histopathologic examination of the female reproductive tract in repeated-dose preclinical toxicity studies of at least 2 weeks' duration is considered sufficiently sensitive to evaluate the risks for female reproductive tract toxicity in early human clinical trials (Sanbuissho et al. 2009; ICH 2009).
From the standpoint of the pharmaceutical developer, the early identification of potential adverse outcomes of a candidate compound is desirable. This enables the developer to make informed decisions on compound selection and risk before significant time, money, and resources have been invested. In many pharmaceutical companies, this has led to the development of short-term exploratory preclinical studies designed to detect adverse effects in those organs with the greatest potential to delay or prohibit further development. In our experience, we have found that repeated-dose studies of as few as 4 days can help in identifying adverse effects in various organs. In cases of toxicity related to the female reproductive tract or the HPO axis, though, the minimum time required to detect toxicity is unclear. In most cases, 2-week toxicity studies in the rat, covering 2½ to 3 ovarian cycles, have been shown to be sufficient to identify ovarian toxicity (Sanbuissho et al. 2009). However, we were interested in establishing whether studies of less than 2 weeks might be used to detect compounds with the potential to induce reproductive dysfunction. Furthermore, we were interested in determining whether the specific pattern of histological changes in the female reproductive tract would provide useful future guidance on the mechanism of disruption within the HPO axis.
The present work, therefore, was conducted as part of a larger project to examine the temporal effects of treatment (up to 28 days) with compounds known to affect the female reproductive axis. The goal of these studies, when completed, will be to determine whether there are histologic changes that are unique or characteristic to specific mechanisms of hormonal disruption and whether they may be identified in a short-term toxicology study. In the current study, exemestane, an inhibitor of the cytochrome-P450 aromatase (P450arom) required for estradiol synthesis, was used to decrease estrogen in intact female rats. While the effects of decreased estrogen or its inhibition have been variably detailed in rodents, much of the work has been conducted in ovariectomized animals or genetically modified mice. Those approaches have enabled researchers to isolate the influence of estradiol on specific structures and functions of the reproductive tract. However, preclinical safety studies of pharmaceutical or chemical compounds are conducted using intact females in which potential compound effects are superimposed upon an intact and functioning reproductive tract. A limited number of studies have described histologic findings in the reproductive tract of intact female rats after 2 to 4 weeks of treatment with aromatase inhibitors or estrogen receptor antagonists (Shirai et al. 2009; Tsujioka et al. 2009). There are few reports detailing the histopathology of the reproductive tract in intact female rats treated with similar compounds for less than 2 weeks and focusing on the earliest changes. Therefore, a study was conducted, based on a design used in our preclinical toxicity studies, to identify the histologic effects of aromatase inhibition after 4, 7, 14, or 28 days of treatment with exemestane (1,000 mg/kg/day). Selected organ weights, vaginal cytology, and serum hormone levels were evaluated to assess their relative value for detecting HPO perturbation. This information will contribute to a future analysis to determine whether the pattern of histologic changes in the reproductive tract in early preclinical toxicity studies might be useful for distinguishing different mechanisms of hormonal dysregulation.
Materials and Methods
Test Article
Exemestane was provided by Pfizer. The vehicle for preparing the exemestane dosing formulation (100 mg/mL) and to dose the control animals was 0.5% methylcellulose (Dow Chemical, Midland, MI) containing 0.1% Tween 80 (USB Corporation, Cleveland, OH).
Animals
Six- to 7-week-old female Sprague-Dawley (Crl:CD®[SD]) rats, weighing 125 to 175 g, were obtained from Charles River Laboratories (Kingston, NY). Rats were housed individually in polycarbonate shoebox cages in a single room maintained at a room temperature of 70°F ± 5°F, relative humidity of 50% ± 10%, with a 12-hour light/dark cycle (lights on 6:00 a.m. to 6:00 p.m.), and a minimum of 12 air changes per hour. The animals were allowed free access to reverse osmosis-purified tap water and a pelleted diet (Certified Rodent Diet 5002 [PMI® Nutrition International, LLC, St. Paul, MN]) throughout the acclimation and study periods. Rats were acclimated for approximately 1 week prior to the start of the study. Following the acclimation period, vaginal smears were collected and examined during a 2-week observation period prior to initiation of dosing. All procedures involving animals were conducted under an animal use protocol approved by Pfizer’s Institutional Animal Care and Use Committee and in compliance with the Guide for Care and Use of Laboratory Animals and all applicable federal regulations.
Experimental Design
After the acclimation and observation periods, female rats were randomized by body weight into 2 groups (control or exemestane-treated) of 20 animals each. All animals included in the study exhibited at least two 4- to 5-day estrous cycles (based on vaginal cytology) without increased numbers of estrus days (> 2 consecutive) or diestrus days (> 3 consecutive) during the 2-week observation period. Control animals were administered the vehicle in a volume of 10 mL/kg by oral gavage. Animals assigned to the exemestane-treated group received a single daily dose of 1,000 mg/kg exemestane using the same route and dose volume as controls. This dose level for exemestane was selected because it had previously been shown to induce changes in the reproductive tract and was otherwise well tolerated after 4 weeks of treatment (FDA CDER 1999). During the treatment period, clinical signs were recorded twice daily, body weights were collected twice weekly, and food consumption was measured weekly. Vaginal smears were collected and examined daily (in the morning) during the treatment period and on the day of necropsy. On the day of necropsy (days 5, 8, 15, or 29), 5 rats/group were anesthetized using CO2 and blood samples were obtained from the caudal vena cava for hormone analysis; the rats were then killed under anesthesia by exsanguination. All necropsies were conducted between 9 and 11 a.m. The following organs were weighed: ovaries (paired), uterus (with cervix), and adrenal gland (paired). Relative organ weights were calculated on the basis of the final body weight measured prior to necropsy. The ovaries, uterus, cervix, vagina, mammary gland, adrenal gland, and pituitary gland were collected and fixed in 10% neutral buffered formalin. After fixation, all tissues were trimmed, dehydrated, embedded in paraffin, sectioned, mounted on glass slides, and stained with hematoxylin and eosin for microscopic evaluation.
Microscopic Examination of the Reproductive Tract
Microscopic examination of tissues was conducted as for a standard toxicology study, including a single transverse section of each ovary (with the goal of examining the greatest area at the hilus), a single section of each oviduct along the longitudinal axis, a single transverse section of each uterine horn, a longitudinal section of the cervix from the vaginal fornix into the distal uterus, and a single longitudinal section of the vagina. Additional sections were examined only for cases in which the original slide did not contain a complete section or in which there was excessive artifactual distortion of the tissue. For tubular organs in which mucosa could not be obtained, the tissue was designated as not examined.
Hormone Analysis
Serum was separated from blood samples collected at necropsy and frozen in aliquots at –80°C until analyzed for hormone concentrations. Serum was analyzed using radioimmunoassays for estradiol (Coat-A-Count; Siemens Diagnostic Healthcare, Deerfield, IL), progesterone (Coat-A-Count; Siemens Diagnostic Healthcare), prolactin (Prolactin [Rat] RIA; ALPCO Diagnostics, Salem, NH), and luteinizing hormone (LH) (Luteinizing Hormone [Rat] RIA; ALPCO Diagnostics) on a Perkin Elmer Gamma Counter. Follicle stimulating hormone (FSH) was measured on a Luminex Analyzer using a non-isotopic immunoassay (Milliplex Rat Pituitary Panel; Millipore, Billerica, MA). Assay parameters are included in Table 1 . Estradiol levels are not included in this report. Following completion of the hormone analysis, very high levels of estradiol were observed in sera from exemestane-treated animals at all time points (data not shown). Investigation of this finding using the test article spiked into pooled control rat sera indicated that exemestane, a steroidal compound, demonstrated significant cross-reactivity in the estradiol assay. Therefore, reliable values for estradiol could not be determined due to exemestane’s interference with the assay.
Hormone assay parameters.
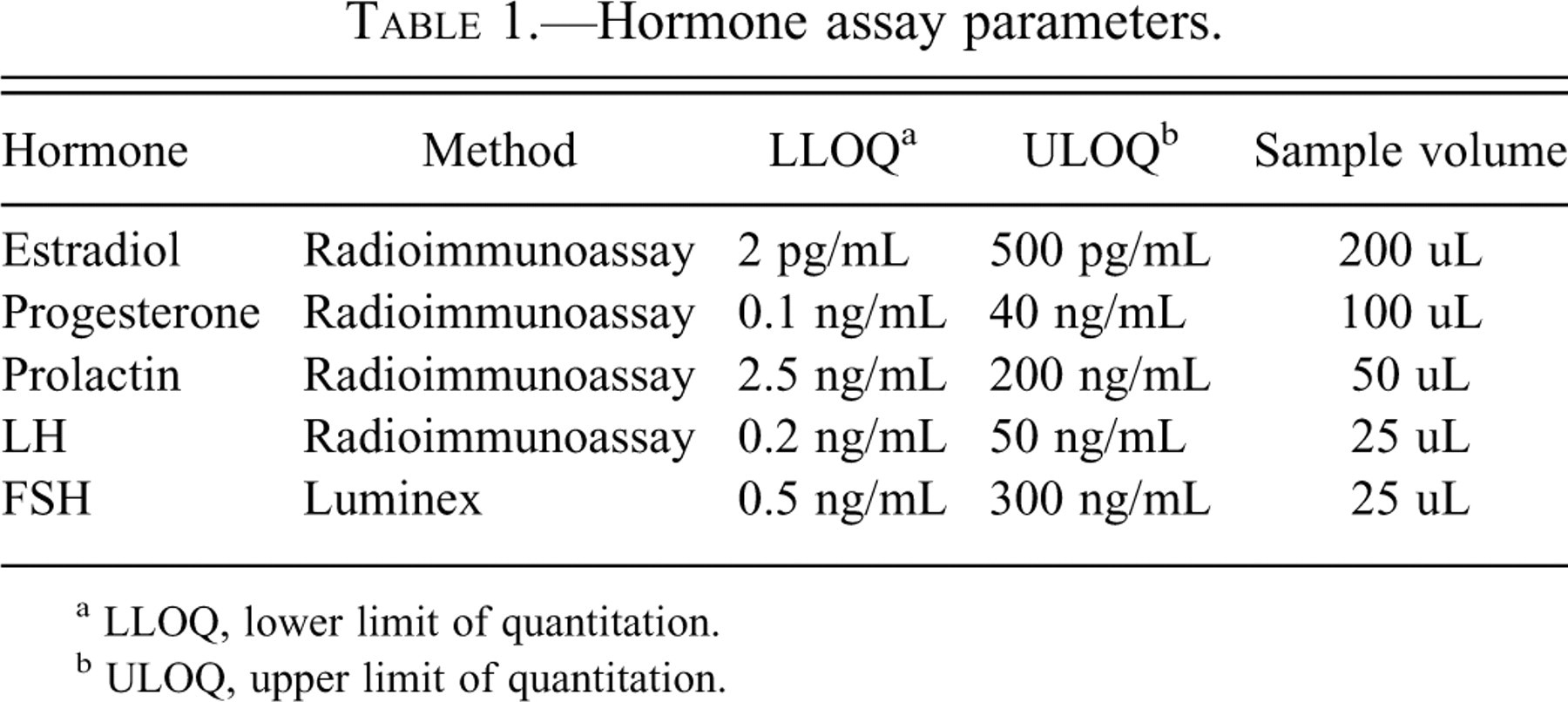
a LLOQ, lower limit of quantitation.
b ULOQ, upper limit of quantitation.
Statistical Analysis
Differences in terminal body weight, food consumption, serum hormone concentrations, and absolute and relative organ weights at each time point were examined using a two-sample t-test with an adjustment for unequal variance. Differences were considered to be statistically significant at p ≤ .05.
Results
Clinical Observations
No clinical signs or mortality related to treatment were observed in this study. Food consumption (Table 2 ) was decreased (74–81% of control mean) for the first 2 weeks of the study in exemestane-treated rats (although the decrease was statistically significant only at the end of the 1st week). Thereafter, food consumption was similar to control animals for the remainder of the study. Body weight in exemestane-treated rats was significantly decreased (83% of control mean) at the end of the 2nd week of treatment, likely reflecting decreased food consumption during the first 2 weeks of the study (Table 2). However, by the end of the study, body weight for the treated animals was similar to control.
Summary of weekly food consumption and terminal body weights.
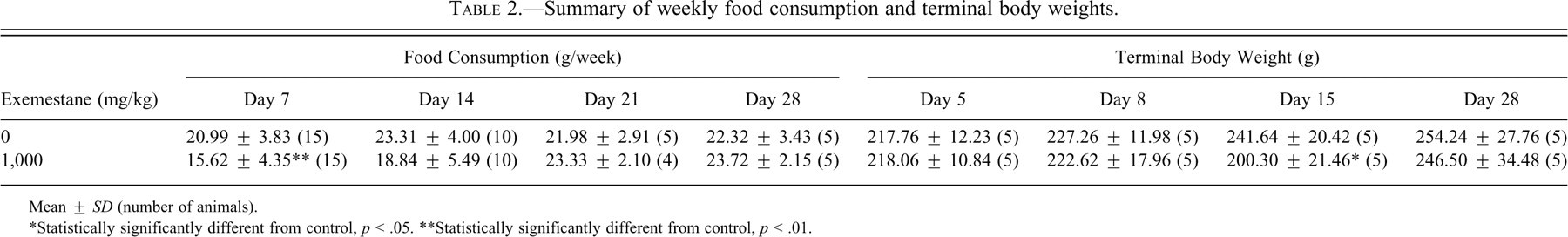
Mean ± SD (number of animals).
*Statistically significantly different from control, p < .05.
**Statistically significantly different from control, p < .01.
Estrous Cycles
Animals were not harmonized with regards to the stage of estrus and, therefore, were in various stages of the estrous cycle on the 1st day of dosing (Table 3 ). By the 4th and 5th day of dosing, it was clear that exemestane had disrupted estrous cyclicity, as all but 1 treated rat exhibited 2 consecutive days of vaginal diestrus while a similar pattern was observed in only 11 of 20 control animals (data not shown). All exemestane-treated animals were considered to be in persistent diestrus (≥3 consecutive days of diestrus) by day 6 or the subsequent day of metestrus (data not shown). Thereafter, all exemestane-treated rats remained in vaginal diestrus for the remainder of the study, with the exception of 1 animal that exhibited metestrus on day 22 of dosing and then reentered diestrus.
Number of animals at each stage of the estrous cycle, Days 1–5 a .

P, proestrus; E, estrus; M, metestrus; D, Diestrus.
a Stage of estrous cycle evaluated by vaginal cytology.
Hormone Levels
Statistically significant decreases were observed for serum LH on days 5 and 29 and for progesterone on day 29 (Table 4 ). A statistically significant increase in serum FSH was observed on day 5 (Table 4). Exemestane interfered with the determination of estradiol levels (see above); therefore, results for this hormone were unavailable.
Summary of serum hormone concentrations (ng/mL).
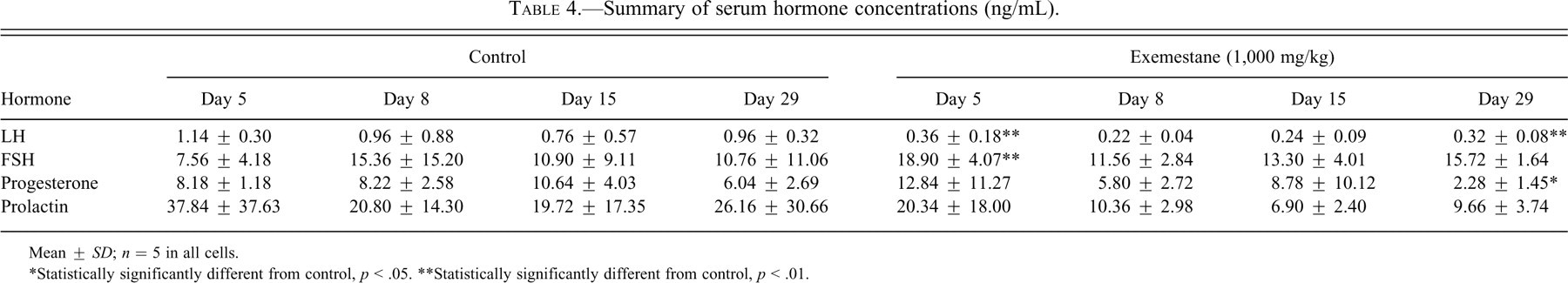
Mean ± SD; n = 5 in all cells.
*Statistically significantly different from control, p < .05.
**Statistically significantly different from control, p < .01.
Organ Weights
Absolute and relative adrenal weights of exemestane-treated rats were decreased throughout the 4 weeks of the study (Table 5 ). By day 5, both parameters were approximately 73% of control mean values, and they remained decreased up to day 28, when they were 65% of control mean values. Uterine weights were significantly decreased in exemestane-treated rats on day 5, relative to controls.
Summary of absolute (g) and relative (g/g body weight) organ weights.
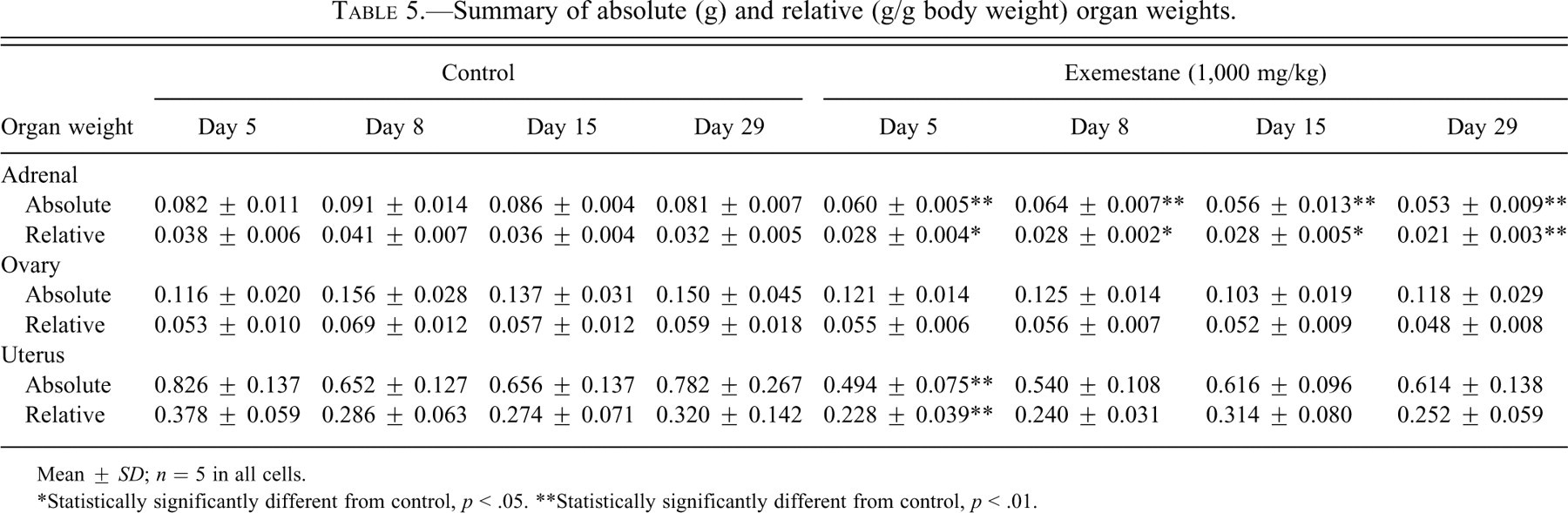
Mean ± SD; n = 5 in all cells.
*Statistically significantly different from control, p < .05.
**Statistically significantly different from control, p < .01.
Histopathology
In general, treatment of female rats with exemestane resulted in anticipated pharmacologic effects in tissues of the endocrine and reproductive systems (Table 6 ). The effects of treatment were consistent and progressive from day 5 to day 15, but by day 29 the effects appeared to diminish in some of the treated animals. The divergence in these effects was attributed to decreased systemic exposure as exemestane induces an increase in its own metabolism with repeated dosing in rodents (FDA CDER 1999; Vignati et al. 2004).
Incidence and grading of effects of exemestane treatment over time.
Incidence is given as total observations/number of animals examined. Grading is indicated parenthetically as P = present (listed with diagnosis) or as subjective severity scores (listed with incidence), where 1 = minimal, 2 = mild, 3 = moderate, and 4 = marked.
Pituitary Gland
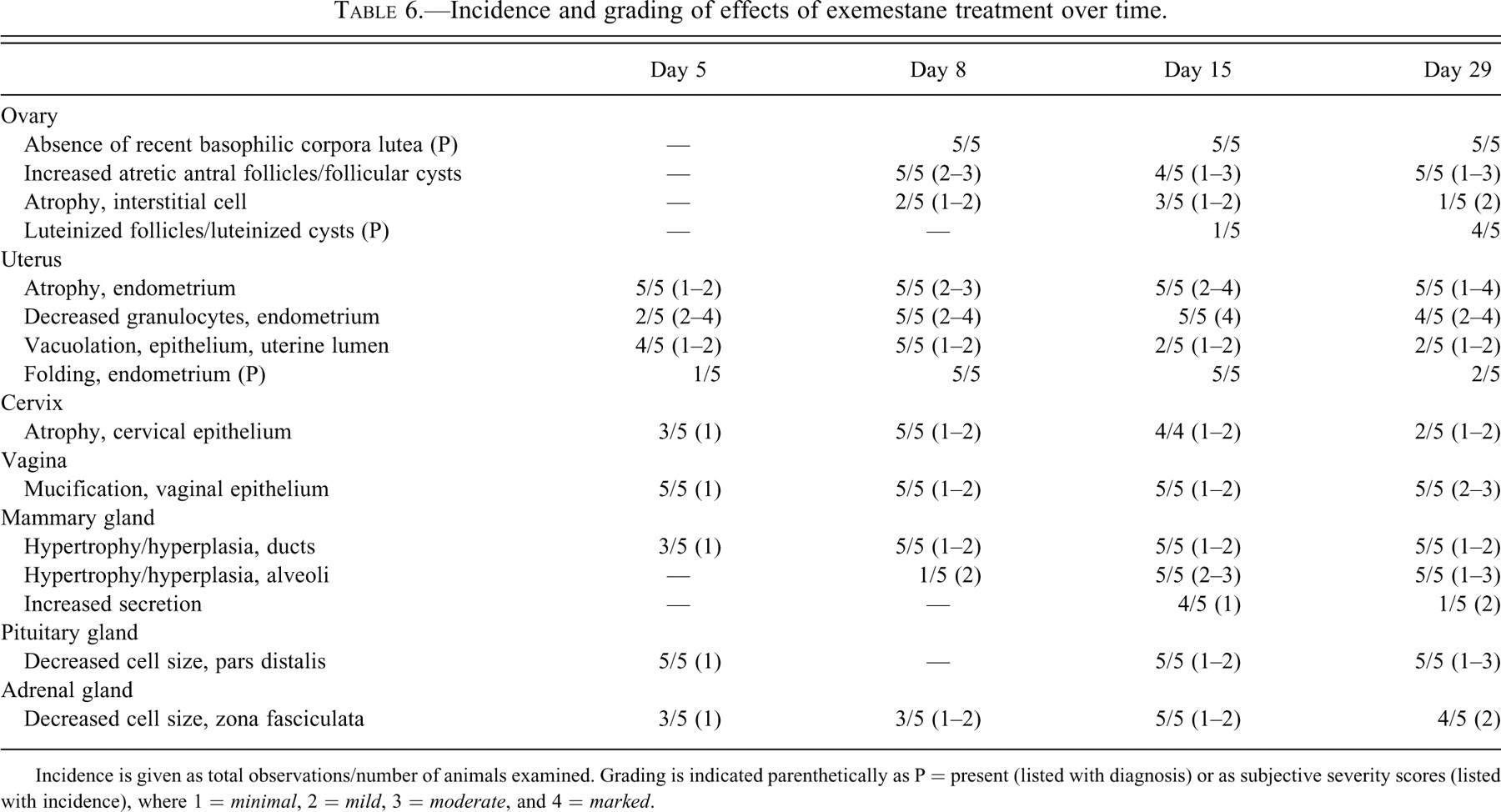
On day 5, a slight decrease in cell size (relative to control, Fig. 1a ) was noted in the pars distalis of exemestane-treated rats (Fig. 1b). Decreased cell size was accompanied by decreased cytoplasmic rarefaction and increased density of nuclei within the distalis. On day 8, there was no apparent difference in the cytoplasmic features or size of cells in the pars distalis. However, on days 15 (Fig. 1e) and 29 (Fig. 1f), cells of the pars distalis in exemestane-treated rats were again decreased in size with severity ranging from slight to mild on day 15 and mild to moderate on day 29, relative to controls (Fig. 1b and c, respectively).
Histology of the anterior pituitary gland of rats treated with vehicle only on days 5 (a), 15 (b), and 29 (c). In pituitary glands from rats treated with exemestane at 1,000 mg/kg, there was a generalized decrease in cell size (relative to control) on days 5 (d), 15 (e), and 29 (f), the severity of which tended to increase with the duration of treatment and resulting in increased density of nuclei within the gland. (H&E. Original objective lens magnification: 40×.)
Adrenal Gland
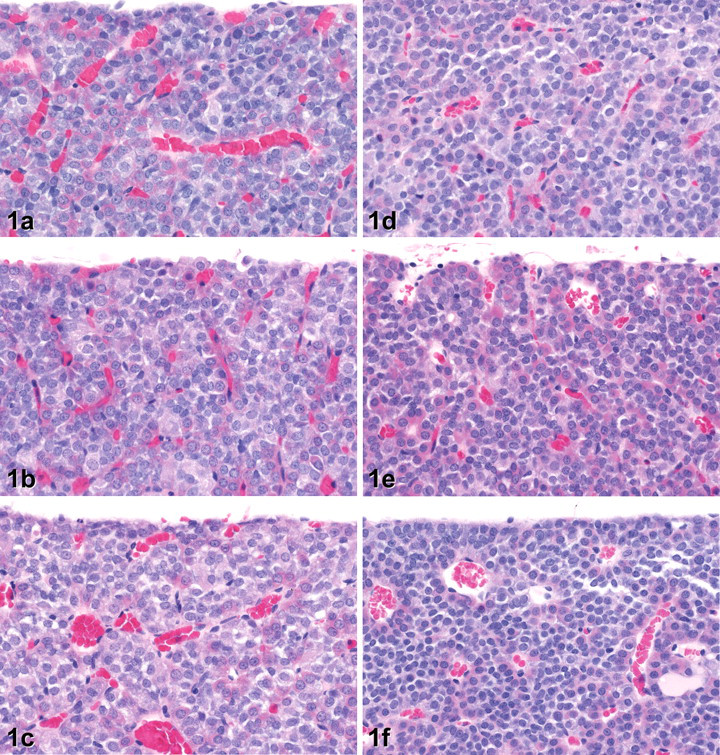
Treatment with exemestane was associated with a decrease in the size of the cells in the adrenal zona fasciculata in comparison with untreated vehicle controls (Fig. 2 ). A slight decrease in cell size was noted in a few animals on days 5 and 8, accompanied by a corresponding increase in the density of nuclei within this region. On days 15 and 29, decreased cell size resulted in an obvious decrease in the thickness of the fasciculata with attenuation of cords and increased width and prominence of sinusoids in most of the treated animals. In general, these changes in the adrenal cortex corresponded with decreases observed in adrenal weights.
Histology of the adrenal gland on day 15 in rats treated with vehicle only (a) or exemestane at 1,000 mg/kg (b). In the exemestane-treated rat, there is decreased thickness of the zona fasciculata in the adrenal cortex. Cells of the zona fasciculata are generally smaller than in controls with decreased cytoplasmic vacuolation and sinusoids are correspondingly enlarged and prominent. (H&E. Original objective lens magnification: 10×.)
Mammary Gland
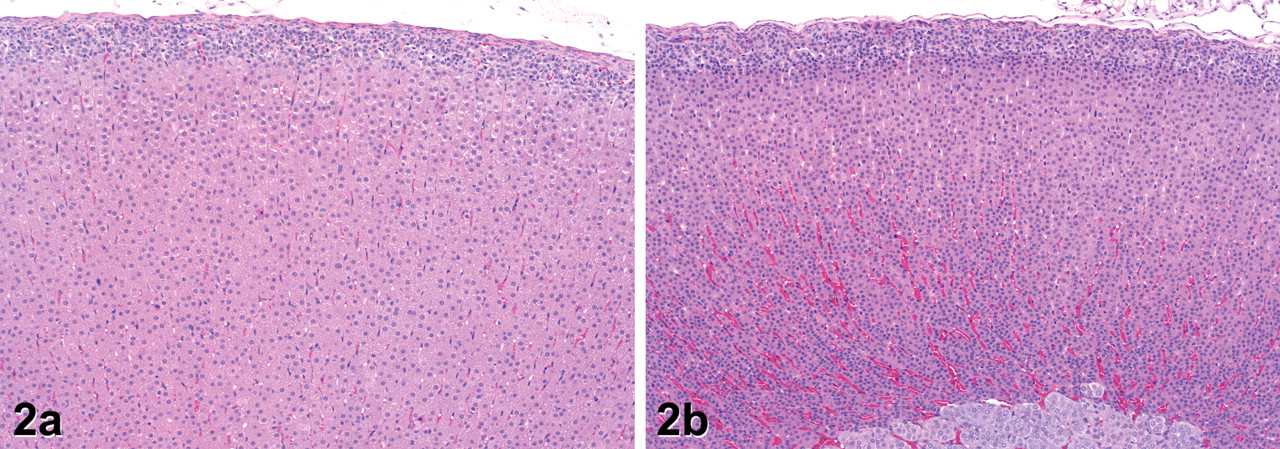
In the mammary gland, treatment with exemestane resulted in hypertrophy/hyperplasia of the mammary gland epithelium with varying degrees of glandular secretion. In a few treated animals on day 5, the epithelium lining occasional small ductules exhibited subtle apical cytoplasmic blebbing (Fig. 3 ) reminiscent of decapitation secretion and lactation (Masso-Welch et al. 2000); similar changes were not observed in control animals in this study. The first clear effects of treatment were noted on day 8, with obvious hypertrophy of epithelial cells in ductules and alveoli (Fig. 4b ). In a few ductules, the epithelium had started to acquire a more columnar appearance, and vacuolation of ductular and alveolar epithelium was evident. The apical blebbing first noted on day 5 was more common in both ductules and alveoli, and alveolar lumina often contained small quantities of residue, though comparable quantities of secretory material were occasionally noted in a few alveoli in control rats, particularly at metestrus. On day 15, there was a slight increase in the quantity of glandular tissue in treated rats. Most of the exemestane-treated rats had generalized ductular epithelial hypertrophy as well as cytoplasmic vacuolation of alveolar epithelium, and many alveoli contained amphophilic luminal secretion. Ductular epithelium varied from cuboidal to columnar with occasional focal pseudostratification. In some ductules, piling of the slightly hyperplastic epithelium was noted with occasional mitotic figures present, but without obvious cellular atypia. On day 29, there was a divergence in the appearance of the mammary tissue in treated rats. In some cases, the hypertrophy, hyperplasia, and secretion noted on day 15 persisted or became even more prominent (Fig. 4c, d), occasionally taking on the appearance of a lactating gland. In other animals, however, epithelial hypertrophy, vacuolation, and apical blebbing were decreased relative to day 15 (though not to controls) and were accompanied by apoptotic epithelial cells indicative of regression.
Histology of a mammary ductule from a rat treated with exemestane at 1,000 mg/kg for 4 days. Ductule epithelium has prominent apical bleb formation and decapitation reminiscent of lipid secretion during lactation. (H&E. Original objective lens magnification: 40×.)
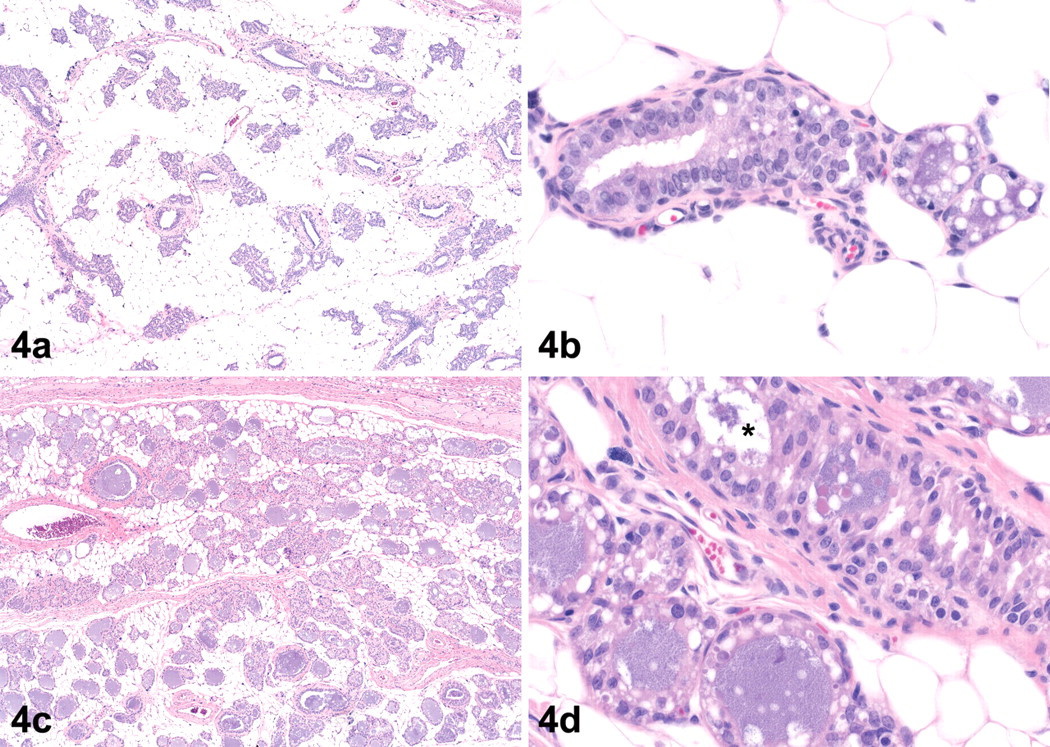
Histopathology of the mammary gland in rats treated with exemestane at 1,000 mg/kg on days 8 (a, b) and 29 (c, d). (a) Low-magnification view of mammary gland in a rat treated for 7 days is similar in appearance to that of vehicle control (not shown), while at higher magnification (b) there is hypertrophy of ductal and alveolar epithelial cells, increased cytoplasmic vacuolation of alveolar epithelial cells, and luminal secretory residue. (c) After 28 days of treatment, there is an increase in glandular tissue with prominent luminal secretion, and at higher magnification (d) ductule (*) epithelium is hypertrophied, often with a cylindrical or columnar appearance, and hyperplastic with multiple layers of cells lining basal lamina in some places. The alveolar epithelium is cuboidal and vacuolated, and residue fills the alveolar lumen. (H&E. Original objective lens magnification: a and c, 4×; b and d, 40×.)
Female Reproductive Tract
Disruption of the estrous cycle was microscopically evident in tissues of the female reproductive tract as early as day 5, and many of these findings persisted throughout the treatment period. Microscopic findings in the reproductive tract were largely attributable to the hormonal dysregulation induced by exemestane. The only organ in which treatment-related changes were not clearly observed during the course of the study was the oviduct. The following summary is presented by day of necropsy to provide a temporal perspective of the changes induced by exemestane among the various tissues of the reproductive tract.
Day 5
Ovarian histology was not clearly affected at day 5, and luteal morphology in exemestane-treated rats was generally consistent with diestrus or proestrus while control animals were staged at proestrus or estrus. In the uterus, the earliest microscopic observation was a minimal endometrial atrophy characterized mainly by decreased size of the glandular epithelium (relative to those of controls at diestrus from later time points; Fig. 5a ), which was cuboidal with decreased cytoplasmic basophilia, small, uniform nuclei, and an absence of mitotic activity (Fig. 5b). In contrast, luminal epithelial cells were strikingly uniform, columnar to tall columnar and, in most animals, contained clear, subnuclear or perinuclear cytoplasmic vacuoles (Fig. 5b). Other endometrial changes included decreased endometrial granulocytes and, in 1 animal, exaggerated folding of the endometrial surface similar to that observed during early pseudopregnancy. In the cervix, atrophy of the mucosa was characterized by a decrease in thickness of the epithelium, which often consisted of 1 to 2 layers of predominantly cuboidal cells and an absence of squamous differentiation (Fig. 5d) relative to control cervix at diestrus (Fig. 5c). In the vagina, the epithelium lining the lumen was slightly mucified and characterized by a thin (1–2 layers) stratum germinativum overlain by a single surface layer of low to tall cuboidal cells containing amphophilic cytoplasm (Fig. 5f). At low magnification, the vaginal histology mimicked that observed in our laboratory on the day of metestrus (early diestrus per Westwood 2008), but lacked the typical characteristics of squamous differentiation at this stage of the estrous cycle. Vaginal mucification was generally accompanied by slight epithelial degeneration, a mixed inflammatory cell infiltrate, and a slight mucocellular luminal exudate.
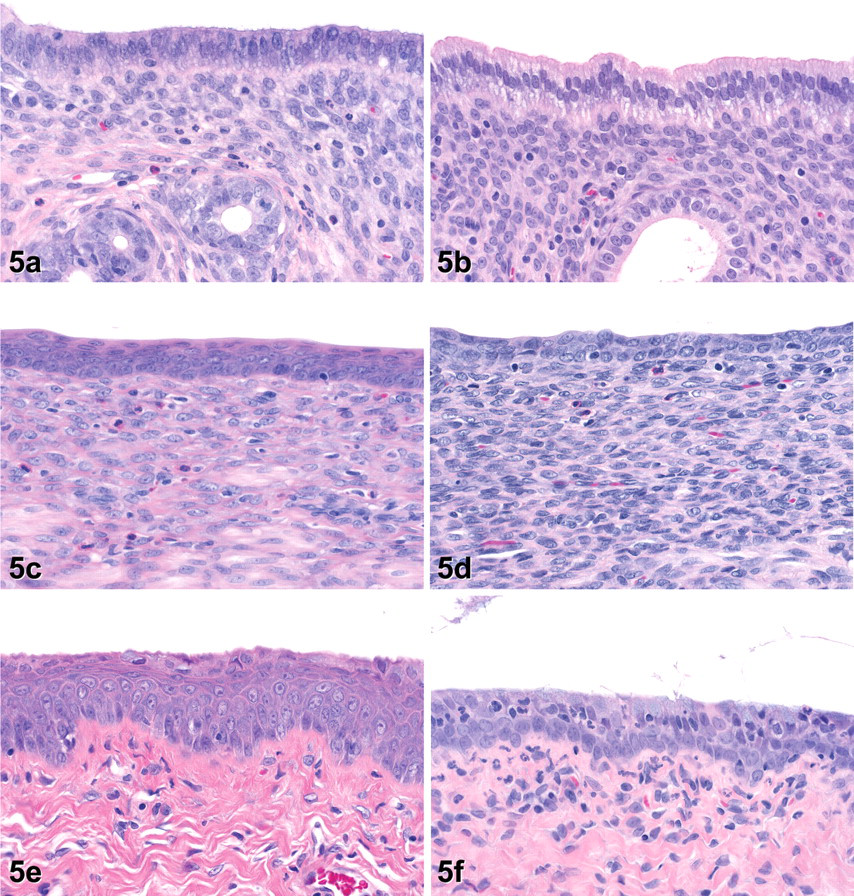
Histopathology of the female reproductive tract on day 5 in rats treated with vehicle only (a, c, e) or with 1,000 mg/kg exemestane (b, d, f). (a) Control uterus at diestrus. (b) In exemestane-treated rats, the uterine luminal epithelium is tall columnar with prominent basal cytoplasmic clearing and vacuolation. Epithelium lining endometrial glands is uniform and eosinophilic relative to control at diestrus. (c) Control cervix at diestrus. Epithelium is 3 to 4 layers thick, and superficial layers are flattened and eosinophilic. (d) In the exemestane-treated rat, the cervical luminal epithelium is 2 layers thick, and there is an absence of superficial squamous differentiation. (e) Control vagina at diestrus. (f) The vaginal epithelium in the exemestane-treated rat is atrophied and infiltrated by inflammatory cells; the stratum germinativum is 1 to 2 layers thick with an overlying layer of larger cuboidal cells, some of them containing scant mucinous material. (H&E. Original objective lens magnification: 40×.)
Day 8
By day 8, ovaries of all exemestane-treated rats lacked the recent (basophilic) corpora lutea (CLs) seen in control animals (Fig. 6a ), indicating that ovulation had not occurred since day 5. Ovaries of exemestane-treated rats often contained increased atretic antral follicles, many of which had retained a relatively large lumen lined by a thin layer of granulosa cells, or occasional larger cysts lined by a thin rim of granulosa or luteinized cells (Fig. 6b). Slight atrophy of ovarian interstitial cells was noted in some animals (Fig. 6d). In treated rats, uterine atrophy had progressed to mild (with decreased uterine size and increased uniformity of luminal surface epithelium) or moderate (with decreased size of stromal cells in addition to epithelial changes). Vacuolation of the uterine luminal epithelium, decreases in endometrial granulocytes, and slight endometrial folding persisted in all/most treated animals (Fig. 6e). Microscopic findings in the cervix and vagina were generally similar to those noted on day 5, although the severity of vaginal mucification increased based upon increased prominence of mucinous material within some cells lining the lumen (Fig. 6f).
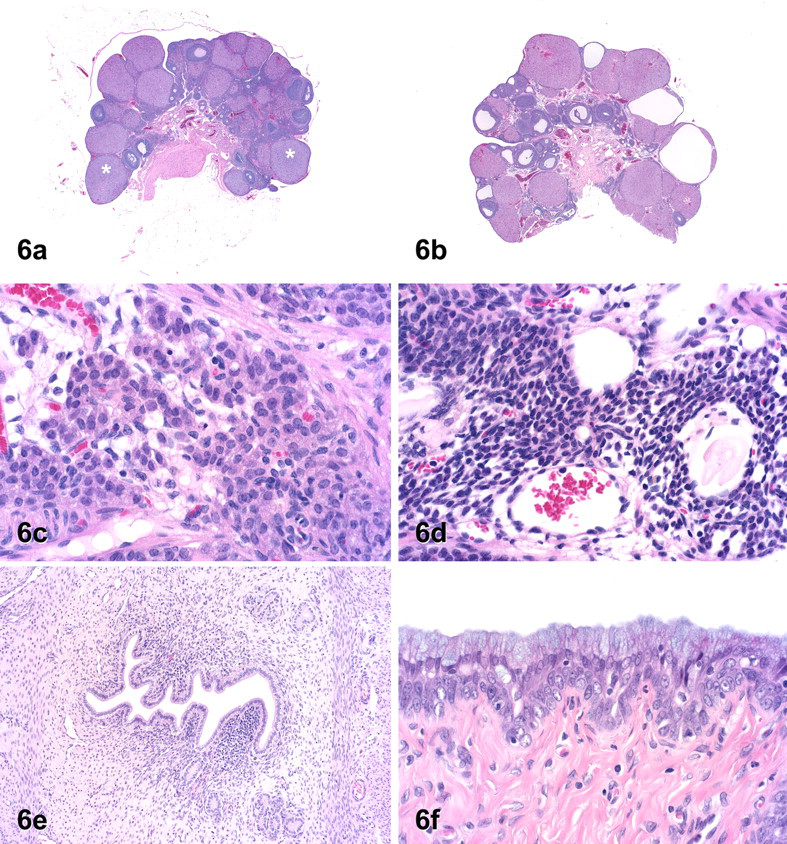
Histopathology of the female reproductive tract on day 8 in rats treated with vehicle only (a, c) or with exemestane at 1,000 mg/kg (b, d, e, f). (a) Control ovary at diestrus with recent corpora lutea (indicated by *). (b) Ovary from an exemestane-treated rat contains 2 cysts with central fluid-filled cavities lined by a thin rim of luteinized tissue, and there is an absence of recent (basophilic) corpora lutea. (c) Interstitial cells at diestrus in control ovary. (d) Interstitial cells in exemestane-treated rat are small with scant cytoplasm and dark, condensed nuclei. (e) Uterus of an exemestane-treated rat with mild uterine atrophy and exaggerated folding of the endometrium lining the lumen. (f) The vaginal epithelium from an exemestane-treated rat is composed of a thin stratum germinativum, 2 cells thick, and an overlying layer of cuboidal to columnar mucified cells. (H&E. Original objective lens magnification: a and b, 0.125×; c, d, and f, 40×; e, 20×.)
Day 15
Microscopic findings in the reproductive tracts of treated rats were generally similar to those noted on day 8, though with the following differences. Corpora lutea in the ovaries were consistently eosinophilic and generally appeared smaller than at previous time points. In the uterus, endometrial atrophy was marked in some animals with an obvious decrease in endometrial area and increased prominence of the endometrial folding. However, vacuolation of the endometrial epithelium became less common. In the cervix, occasional cells of the surface layer appeared to contain mucinous material.
Day 29
In the ovary, mild to moderate atresia of large antral follicles and/or follicular cysts (Fig. 7a ) were again observed in all treated rats and typical basophilic CLs of the current cycle were not present, indicating that ovulation had not occurred within the last 4 to 5 days. In 3/5 rats, partial or complete luteinization of follicles, some composed of luteinized cells surrounding a retained oocyte, was present (Fig. 7b). Additionally, ovaries of 3 treated rats contained a few luteinized structures similar to CLs that were suggestive of ovulatory activity between days 15 and 25 as well as older, small CLs. The luteinized structures were slightly more basophilic and composed of larger cells than were older CLs and contained scattered apoptotic cells; in 1 animal, they contained large central cavities resembling CLs of metestrus (Westwood 2008) (Fig. 7c–f). However, the cystic luteinized structures (Fig. 7f) were composed of a smaller population of less vacuolated cells than are typical CLs of metestrus (Fig. 7e), and the cyst lumen was lined by a thin fibrous membrane (Fig. 7f). Atrophy of ovarian interstitial cells was noted in 1 animal. In the uterus, minimal to marked endometrial atrophy (decreased endometrial thickness in addition to other findings) was still noted in all animals, usually in association with decreased granulocytes. In 2 rats, luminal epithelial changes and folding of the superficial endometrium persisted as at previous time points. The remaining 3 animals, however, had dilatation of the endometrial lumen suggestive of rising levels of estrogen during normal proestrus; in 2 of these animals, apoptotic cells (not typically present during proestrus) were scattered within the luminal and/or glandular epithelium (Fig. 7g). In the cervix, epithelial atrophy persisted in those animals with uterine atrophy, while the cervical epithelium of animals with uterine dilatation tended to be of normal thickness. In the vagina, epithelial mucification with associated cellular degeneration and/or inflammatory cell infiltration persisted and the degree of mucification was slightly greater in those animals with uterine dilatation (Fig. 7h).
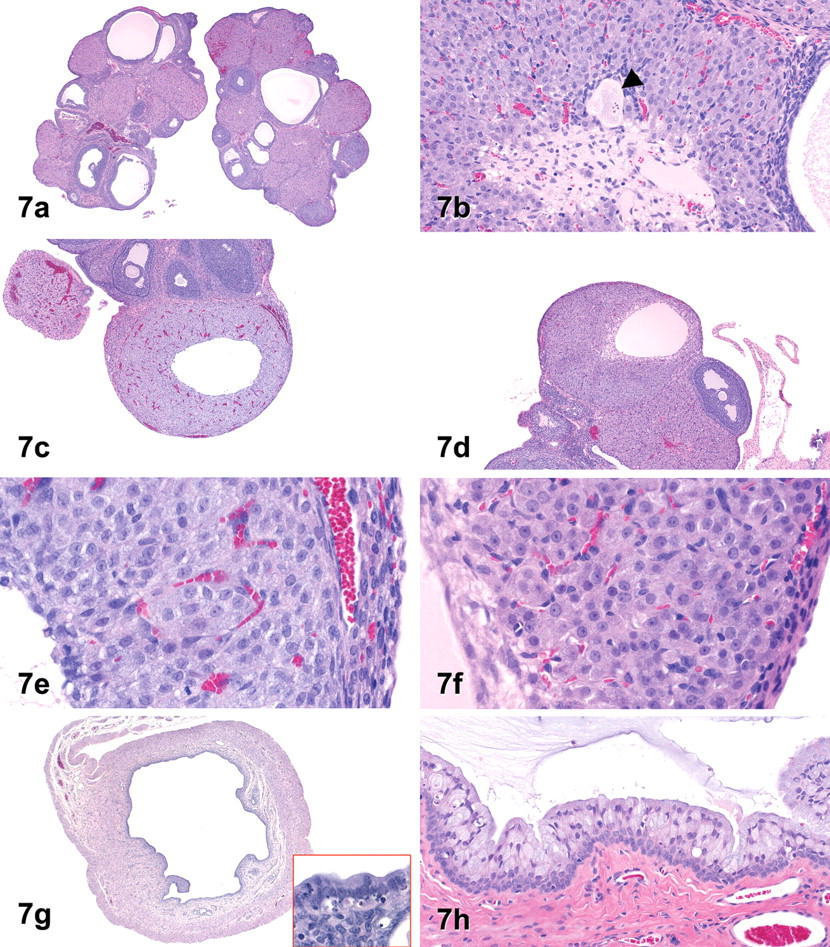
Histopathology of the female reproductive tract on day 29 in rats treated with vehicle only (c, e) or with exemestane at 1,000 mg/kg (a, b, d, f, g, h). (a) Ovaries from an exemestane-treated rat contain multiple fluid-filled follicular cysts, each lined by a thin layer of granulosa cells. (b) A degenerated retained oocyte (arrowhead) is surrounded by luteinized cells within an otherwise normal appearing luteal structure. (c, e) Current corpus luteum of metestrus containing a central cavity, from a control ovary. (d, f) Luteinized cyst from an exemestane-treated rat mimicking a corpus luteum of metestrus. At higher magnification, the luteinized cells are less basophilic and less vacuolated (relative to control in 7e) and are separated from the cyst cavity by a modest fibrovascular membrane. (g) Uterine luminal dilatation, resembling proestrus, in an exemestane-treated rat. Inset (40x): Apoptosis in the endometrial epithelium lining the uterine lumen and endometrial glands. (h) Exacerbation of vaginal epithelial mucification. (H&E. Original objective lens magnification: a and g, 2×; b and h, 20×; c and d, 4×; e and f, 40×.)
Discussion
This study was one of a series of experiments conducted to determine, first, the earliest time point at which hormonal dysregulation of the HPO axis in female rats would be detectable using an exploratory preclinical toxicology study design, and second, whether changes related to the administration of known hormonal modulators could be used to help guide the mechanistic interpretation of histologically observed reproductive dysfunction in short-term toxicology studies. This report details the time course for detection of histologic findings related to exemestane administration to decrease estradiol in intact female rats. Exemestane is a synthetic steroid that inhibits the conversion of androgens (principally testosterone and androstenedione) to estradiol by irreversibly binding to aromatase (di Salle et al. 1987). The dose of exemestane was chosen based on previous data to try to ensure that effects of decreased estradiol would be observed during the course of the study while avoiding obvious systemic toxicity.
In the present study, many of the findings noted were similar to observations in other reports detailing the treatment of intact female rats with other aromatase inhibitors for 2 to 4 weeks (Yano, Ikegami, and Nakao 1996; Shirai et al. 2009). On day 5, we found relatively subtle histologic effects of treatment, consistent with changes expected due to decreased estrogen, in the target organs (cervix, uterus, vagina) on which this hormone exerts its actions. These early changes were generally of minimal severity and included endometrial (glandular) atrophy and decreased granulocytes; mucification of the vaginal epithelium without normal, cyclical epithelial proliferation; and atrophy of the cervical epithelium. Additional changes observed in the uterus, which may have reflected the loss of estradiol and/or effects of an altered estrogen/progesterone ratio, included increased folding of the endometrium and hypertrophy and vacuolation of the luminal epithelium (Ljungkvist 1971; Westwood 2008). The epithelial vacuolation was somewhat surprising as the rat does not usually develop this endometrial change seen during the secretory (or luteal) phase in other species. This is because luteal progesterone secretion in the non-gravid rat is limited to a brief period in metestrus and is insufficient to support decidual changes in the uterus (Smith, Freeman, and Neill 1975; Freeman 2006). By day 8, the histologic appearance of the reproductive tract provided a much more robust signal indicative of exemestane-induced changes in the HPO axis. Effects of treatment were now readily apparent in the ovary and included an obvious increase in atretic large antral follicles and/or follicular cysts, an absence of current basophilic CLs, and atrophy of interstitial cells. These changes reflected expected effects of decreased estradiol, including a failure of hypothalamic stimulation for the gonadotropin surge and impaired ovulation. In the tubular organs of the tract, changes noted on day 5 tended to persist for the remainder of the study and increase in severity and/or incidence. However, some animals on day 29 exhibited uterine luminal dilatation, reestablishment of squamous differentiation of the cervical mucosa, and slight exacerbation of vaginal mucification (similar to a gravid state) (Barker and Walker 1966; Westwood 2008). The changes in these animals suggested a partial recovery of hormonal function likely related to auto-induction of exemestane metabolism and decreased exposure as previously reported in rodents (FDA CDER 1999; Vignati et al. 2004).
This study demonstrated that histologic changes required for detection of exemestane-induced HPO dysregulation were clearly and uniformly evident in all tissues of the reproductive tract (except the oviduct) by day 8. The most obvious microscopic findings indicative of estrous cycle disruption were vaginal mucification and the presence of increased large atretic follicles, follicular cysts, an absence of current basophilic CLs, and, less frequently, interstitial cell atrophy in the ovary. Changes in the uterus and cervix, while clearly evident, were less valuable for screening as, even on day 8, these tissues generally required more extensive comparison with control tissues.
In contrast, the microscopic changes in the reproductive tract on day 5 tended to be more subtle, and many were less consistently observed. Given the small group size examined at each time point, an independent evaluation of each of the reproductive organs could have led to a conclusion that there was no effect of treatment, particularly if there were no prior evidence or expectation for an endocrine effect of the tested compound. However, a coordinated assessment of all tissues of the reproductive tract in a standard toxicology study would be expected to highlight the observed asynchrony between the ovary (features consistent with diestrus/proestrus) and the vagina (features consistent with metestrus/anestrus) and highlight the need for additional attention (Li and Davis 2007; Westwood 2008). In general, evaluation of ovary and uterus weights using this study design provided no additional information that might have guided detection of disruption of the HPO axis.
Previous work has shown that evaluation of histopathology in toxicology studies of at least 2 weeks' duration will identify a majority of female reproductive toxicants with the possible exception of some cytotoxic compounds (Sanbuissho et al. 2009). However, we frequently conduct studies of less than 2 weeks' duration using relatively small numbers of animals to help prioritize compound selection or to quickly identify the greatest safety hazards of new compounds. A small group size of 5 to 6 animals has previously been shown to be sufficient for detection of endocrine effects after 28 days of treatment, and this group size (based on sex alone) was incorporated into the updated OECD guidelines (TG 407) for the testing of chemicals including the detection of endocrine activity (Andrews et al. 2001; OECD 2007). In our case, we were interested in determining whether treating 5 or 6 female rats for a period of 4 to 7 days was sufficient to detect effects on the HPO axis. Our results demonstrated that with this small group size it was possible to clearly detect dysregulation of the female reproductive tract after 8 days of treatment with a high dose of an aromatase inhibitor. Furthermore, with prior knowledge or expectations for hormonal activity in an investigational compound and a careful examination of the reproductive tract, it may be possible to detect effects on the female HPO axis after as little as 4 days of treatment.
The pituitary, adrenal, and mammary glands were also examined in this study to determine whether the microscopic examination of these tissues might contribute to detection of altered function of the HPO axis. We observed that findings consistent with HPO axis dysregulation were detectable in these tissues, but they were considered likely to be of limited value in studies of less than 2 weeks' duration with a small sample size.
In the pituitary gland, decreased cell size in the pars distalis was a fairly subtle finding that increased in severity later in the study. It is not clear whether the absence of this effect on day 8 may have been related to the small group size and subtlety of the response or a histologic reflection of endocrine change related to the duration of treatment. Though not previously described in association with exemestane treatment, the change in the pars distalis was consistent with reports of decreased pituitary weight in earlier studies of exemestane (at similar or lower doses) as well as some other aromatase inhibitors (Zaccheo et al. 1991; Matsuda et al. 1997; FDA CDER 1997; Shirai et al. 2009). Pituitary weights were not determined in the present study. In the case of exemestane, lower pituitary weights have been attributed to activity of the compound or its major metabolite, 17-hydroexemestane, at the pituitary androgen receptor, resulting in suppression of LH secretion (Zaccheo et al. 1991). A reduction in estrogen-mediated stimulation of prolactin production may also have contributed to this finding (Childs 2006), although such a decrease was not clearly demonstrated in this study.
Decreases in adrenal weight and associated microscopic changes were attributed mainly to exemestane treatment. Decreased adrenal weight and/or thickness of the zona fasciculata have been recognized in ovariectomized adult female rats (Winter and Emery 1936; Blumenfeld 1939), indicating the influence of ovarian hormones on normal adrenocortical homeostasis. Decreases in adrenal weights have also been previously observed in female rats treated with aromatase inhibitors, although histologic correlates have not been described (Zaccheo et al. 1991; Dukes et al. 1996; FDA CDER 1997, 1999). Aromatase inhibitor–mediated decreases in adrenal weight have been ascribed to effects of estrogen depletion on corticosteroid binding globulin (Dukes et al. 1996). Interestingly, decreased adrenal size and weight were recently described in aromatase knockout (ArKO) mice in association with telomere shortening and reduced cell proliferation, findings that were reversed with estrogen supplementation (Bayne et al. 2008).
In the mammary gland, exemestane treatment resulted in hypertrophy/hyperplasia of the small terminal ductules and alveoli in association with increased glandular secretion, and these effects were attributed to endocrine dysregulation at the high dose administered. These findings were very different from the expected effects of decreased estrogen, which include atrophy of both ductular and alveolar elements (Lucas et al. 2007). Effects of exemestane in the mammary gland have not previously been described in rats treated for up to 26 weeks at doses similar to those used in the present study (FDA CDER 1999). However, mammary gland duct hyperplasia with increased secretion in intact rats has been described in association with administration of another aromatase inhibitor, letrozole (Cohen et al. 2002), and similar findings have been noted after treatment with testosterone (Laquer and Fluhmann 1942). Histologically, it was not possible to clearly determine whether tubuloalveolar (expansion of the normal female glandular structure) or lobuloalveolar (masculinization) hyperplasia was occurring in exemestane-treated rats, and this observation has been noted by others (Creasy et al. 2008). For this reason, the less specific term “hypertrophy/hyperplasia” was chosen for use in this study. The ability to distinguish these two forms of endocrine-mediated hyperplasia may be useful in understanding whether the stimulation is estrogenic/prolactigenic or androgenic (Lucas et al. 2007; Creasy et al. 2008). In the present study, given that estrogen levels were probably decreased and prolactin levels showed no clear increases, the changes in the mammary gland may have been at least partially related to androgenic stimulation by exemestane and its major metabolite and/or increased endogenous androgen levels as previously noted in animals treated with an aromatase inhibitor (Baravalle et al. 2006). Exogenous androgens, including the non-aromatizable androgen dihydrotestosterone, induce lobuloalveolar hyperplasia in ovariectomized female rats (Sourla et al. 1998). Lobuloalveolar hyperplasia has also been induced in intact female rats with testosterone or as a result of hyperandrogenemia associated with the administration of an estrogen receptor antagonist (Laquer and Fluhmann 1942; Rudmann et al. 2005). Furthermore, administration of flutamide, an androgen receptor antagonist, has been shown to inhibit the hyperplastic and secretory effects of androgens on the mammary gland, indicating that the effects are mediated through the androgen receptor (Sourla et al. 1998; Rudmann et al. 2005). It has been suggested that the stimulatory activity of androgens in the female rat, in the absence of estrogen receptor activity or increased prolactin, may be facilitated in the mammary gland, although this hypothesis has yet to be tested (Rudmann et al. 2005). In the present study, it was considered likely that the effects of exemestane would be reversible as there was some evidence of their regression in a few animals on day 29, presumably as a result of decreased systemic exposure as noted earlier.
In comparison with histology, estrous cycle disruption was evident by day 6 using vaginal cytology when most of the exemestane-treated rats were classified as being in persistent diestrus (≥3 consecutive days of diestrus). This confirmed the utility and sensitivity of vaginal cytology as a tool to identify hormonal effects on the female reproductive system (Goldman, Murr, and Cooper 2007). Persistent diestrus is indicative of deficits in the estrogen surge necessary to stimulate ovulation and has been previously observed in rats treated with exemestane (FDA CDER 1999). It is clear that daily assessment of reproductive cyclicity using vaginal cytology enables slightly earlier detection of xenobiotic disruption of the HPO axis than does histology. However, from a practical standpoint, it is unlikely that vaginal cytology would be consistently used in preclinical safety screening studies. This is because of the relatively large number of compounds examined in exploratory studies; the additional resources required; the small number of compounds that might be detected as modulators of the HPO axis; and the fact that histology would be likely to detect the same endpoint with, at most, only a few additional days of treatment.
Hormone measurements obtained in this study were considered unreliable for detection of HPO dysregulation. Reasons for this included the small sample size and the single time point at which hormones were measured, but two additional factors were also involved. First, the stress and manipulations involved in blood collection procedures similar to those used in this study have been shown to impose significant variability in measurements of circulating hormones, particularly prolactin, but also LH and progesterone among others (Hawkins et al. 1975; Döhler et al. 1977; Böehm et al. 1982). Second, we intentionally restricted ourselves to our standard screening study design, which gives no consideration to the stage of the estrous cycle at the beginning or end of the study. Because of normal hormonal fluctuation across the estrous cycle and variability in the number of normal animals at any given stage of the cycle (Table 3), we could readily predict that it would be difficult to detect a consistent hormonal signal related to treatment difficult, and this proved to be the case. Given the high variability we saw in most of these parameters (Table 4), it is clear that measurement of serum hormones using this study design offered no advantage over histology for detection of HPO dysregulation.
However, while recognizing the limited utility of the hormone measurements in this study design in general, the uniformly low values for LH in treated rats were consistent with expectations of effects of exemestane (Zaccheo et al. 1991). These lower values were supported by histologic findings including the absence of recent ovulation and atrophy of ovarian interstitial cells (Erickson et al. 1985; Freeman 2006). Additionally, the luteinized structures observed in some animals on day 29, while suggestive of ovulation during the preceding 2 weeks, were considered more likely to be luteinized unruptured follicles (LUF) rather than post-ovulatory CLs. This conclusion was supported by the presence of oocytes within other luteinized structures in some animals. Low LH levels have been linked to the formation of LUF in rats (Mattheij and Swarts 1995), and this may explain why this phenomenon has not been reported with other aromatase inhibitors, which typically increase serum LH (Houjou, Wada, and Yasutomi 1993; Kilic-Okman, Kucuk, and Altaner 2003; Kafali et al. 2004).
The use of exemestane in this study was intended to aid in characterizing the temporal effects of a low estrogen state on the histology of the female reproductive tract. While documentation of a decrease in estrogen levels was not possible due to exemestane’s interference with the assay, the observed changes in vaginal cytology and the histomorphology of the reproductive tract in treated rats were consistent with aromatase inhibition resulting in decreased estradiol levels (Yano, Ikegami, and Nakao 1996; Baravelle et al. 2006; Shirai et al. 2009). Furthermore, in rats, treatment with lower doses of exemestane has been shown to decrease ovarian aromatase activity and plasma estradiol levels (Zaccheo et al. 1991). However, in the course of this study, we recognized several factors complicating the interpretation of our results. First, as noted earlier, exemestane and its principal metabolite have weak androgenic activity (Zaccheo et al. 1991; Ariazi et al. 2007). Furthermore, inhibition of aromatase has been shown to increase circulating levels of endogenous testosterone (Baravalle et al. 2006). In the rat, the administration of exogenous androgens has been associated with decreased circulating LH (consistent with decreased pituitary size and ovarian interstitial cell atrophy), vaginal mucification, uterine folding, and mammary gland hyperplasia and secretion independently of conversion to estrogens (Laquer and Fluhmann 1942; Zaccheo et al. 1991; Sourla et al. 1998; Allon et al. 2005; Rudmann et al. 2005). Therefore, we believe it likely that at least some of the effects observed at the high dose of exemestane administered resulted from a combination of decreased estrogen and some degree of androgenic stimulation.
While the effects on the HPO axis observed in this study were attributed primarily to exemestane-mediated endocrine modulation, decreased food intake and weight gain during the first 2 weeks of this study also imposed an unexpected confounding factor. In preclinical toxicology studies of 4 to 52 weeks in duration conducted prior to registration, exemestane treatment at doses ≤1,000 mg/kg typically resulted in slight increases in body weight and/or body weight gain while food consumption was unchanged or slightly greater than control (data not presented). Similar effects on body weight and food consumption have also been reported following the administration of aromatase inhibitors in female rats (Zaccheo et al. 1991; Mannerås et al. 2007; Shirai et al. 2009). A cause for the effects on food consumption, other than treatment itself, was not apparent in this study. Restricted food intake in Sprague Dawley rats resulting in greater than 30% lower body weight (relative to control) has been associated with alterations in the estrous cycle and associated histopathological findings in reproductive tissues (Seki et al. 1997; Terry et al. 2005). However, lesser decrements in body weight resulting from feed restriction have not clearly impacted estrous cyclicity or histology of the reproductive tract, and even when minor changes have been observed, they have been sporadic (Chapin et al. 1993; Seki et al. 1997; Tropp and Markus 2001; Terry et al. 2005). While the characteristics of the changes observed in this study, their high incidence, and persistence throughout the 28 days of study were consistent with effects of exemestane-mediated hormonal dysregulation, the possibility that lower feed intake and resulting body weight loss may have contributed to these findings could not be entirely excluded.
In conclusion, treatment of female rats with exemestane resulted in vaginal mucification; uterine and cervical atrophy; and, in the ovary, increased atresia of antral follicles, follicular cysts, an absence of current (basophilic) corpora lutea, luteinization of non-ovulatory follicles, and atrophy of interstitial cells. While vaginal cytology detected treatment-related changes between days 5 and 8 of treatment, microscopic evaluation of the female reproductive tract alone provided sufficient information to clearly identify effects of oral treatment with exemestane, an aromatase inhibitor, in a small group of animals after 7 days of treatment. Therefore, while vaginal cytology may provide an earlier signal, histopathology alone may be useful for the successful detection of female reproductive toxicity after 7 days of treatment, particularly when there are expectations or concerns for effects on the HPO axis.
Footnotes
Acknowledgments
The authors are indebted to the Developmental and Reproductive Toxicology, Clinical Pathology, and Histology groups at Pfizer, Groton for technical support. We also wish to thank Drs. Ingrid Pruimboom-Brees and Dan Morton for their thoughtful discussions and comments during the conduct of this work and in the development of the article.
