Abstract
Diethylnitrosamine (DEN) is a chemical broadly used in animal models as a hepatocarcinogen, reported to also cause pulmonary neoplasms in mice. The original objective was to evaluate the impact of a Western diet with or without 10% broccoli on DEN-induced on liver cancer. We administered DEN (45 mg/kg) intraperitoneally to young adult male B6C3F1 mice by 6 weekly injections and evaluated liver cancer 6 months after the DEN treatments. Here, we report unexpected primary tumorigenesis in nasal epithelium, independent of dietary treatment. More than 50% of DEN-treated B6C3F1 mice developed nasal neoplasm-related lesions, not reported previously in the literature. Only one of these neoplasms was visible externally prior to postmortem examination. Intraperitoneal DEN treatment used as a model for liver cancer can have a carcinogenic effect on the nasal epithelium in B6C3F1 mice, which should be carefully monitored in future liver cancer studies.
Introduction
Diethylnitrosamine (DEN), which can be found in cigarette smoke, dried seafood, and processed meat, is classified as a probable human carcinogen (B2 level) according to the U.S. Environmental Protection Agency (USEPA 2003; Tricker and Preussmann 1991). The carcinogenicity of DEN is due to its bioactivation by cytochrome (Cyp) P450 enzymes, specifically Cyp 2E1, which produce ethyl diazonium ions, which then form DNA adducts (Verna et al. 1996). In animal models, DEN has been shown to cause tumorigenesis in different tissues, depending on species (Verna et al. 1996). Different routes of DEN administration can also alter the site of neoplasm formation (Clapp and Craig 1967).
It has been reported that B6C3F1 mice develop both hepatic and pulmonary neoplasms following intraperitoneal (ip) injection of DEN, with a greater number of hepatic neoplasms than C57BL/6J mice (Vesselinovitch et al. 1984; Goldsworthy and Fransson-Steen 2002). Nasal neoplasms have been reported in hamsters given DEN ip (Herrold and Dunham 1963). However, the development of nasal cavity neoplasm has not been reported for any route of DEN administration in the mouse model. As part of a larger study of the impact of DEN and a Western (high fat/ high sugar) diet on liver health (Chen et al. 2016), we observed difficulty in breathing in many DEN-treated animals, independent of dietary treatment. Here, we report the appearance of multiple nasal epithelial neoplastic lesions, visible externally in only one instance, induced by ip treatment with DEN.
Materials and Methods
Experimental design and diet formulae are described in a previous report (Chen et al. 2016) and summarized below. Seventy-two male, 4-week-old B6C3F1 (male C3H/HeJ × female C57Bl/6J) mice purchased from the Jackson Laboratory (Bar Harbor, ME). DEN (Sigma-Aldrich, St. Louise, MO) was diluted in normal saline for injection. Ip administration of DEN was chosen to avoid mixing DEN with dietary treatments and to provide better control of dose delivery to the liver; only 1 sex (male) was chosen to decrease variability in response. Five-week-old male mice were divided into 4 groups and provided control, control + 10% broccoli, Western, or Western + 10% broccoli diets. Animals were housed individually and given water and feed ad libitum under a 12-hr light/dark cycle at 22°C and 60% humidity. DEN (n = 12/group; 45 mg/kg ip) or saline (n = 6/group) was given to mice once a week for 6 weeks, at 6, 7, 8, 10, 11, and 12 weeks of age. Mice were killed at 36 weeks old, and tissue samples were collected. Animal care was in compliance with the Institutional Animal Care and Use Committee at the University of Illinois, according to the National Institutes of Health guidelines.
At the termination of the study, heads of all mice were fixed in 10% neutral-buffered formalin for 7 weeks and decalcified (DeltaFORM; Delta Medical, Aurora, IL) for 5 days before paraffin embedding. Upon sectioning of the nasal cavities, the presence of grossly visible neoplasms within the nasal cavities of 28 of 64 mice warranted additional histologic examination. Nasal cavity sections (3 µm) at 4 levels as previously described (Harkema et al. 2006) were stained with hematoxylin and eosin (H&E) for the examination of nonneoplastic proliferative changes (hyperplasia and metaplasia), adenomas, and carcinomas. Slides were prepared in the Veterinary Diagnostic Laboratory at the University of Illinois (Urbana, IL). All neoplasm-related lesions were identified by a board-certified veterinary pathologist, based on the report from the International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in rats and mice (INHAND) project (Renne et al. 2009).
The relationships between DEN/dietary treatments and nasal epithelial lesions were evaluated using the χ2 test. A p value of less than .05 was considered significant. All statistical analyses were performed using Statistical Analysis System 9.4 Software (SAS Institute Inc., Cary, NC).
Results
Mice treated with DEN had variable nasal nonneoplastic and neoplastic lesions in the nasal cavity. In only 1 animal, a nasal neoplasm was visible externally. In all DEN-treated groups, there was chronic multifocal inflammation of variable intensity and in variable locations, although not all mice showed proliferative or neoplastic lesions. Normal respiratory, transitional, and olfactory epithelia were affected, to variable degrees, exhibiting varied distribution of hyperplasia (respiratory, transitional, basal cell, glandular, and olfactory), metaplasia (respiratory metaplasia of olfactory epithelium), adenoma, adenocarcinoma, and neuroepithelial carcinoma (Figure 1). The incidence of total nasal proliferative lesions was low in the saline-treated animal (1/24; 4%) but high in the DEN-treated animal (27/40; 67%), showing ip DEN increased the risk of nasal neoplasm-related lesions (Table 1). However, neither the Western diet nor broccoli had any impact on the formation of nasal epithelial neoplasm-related lesions.
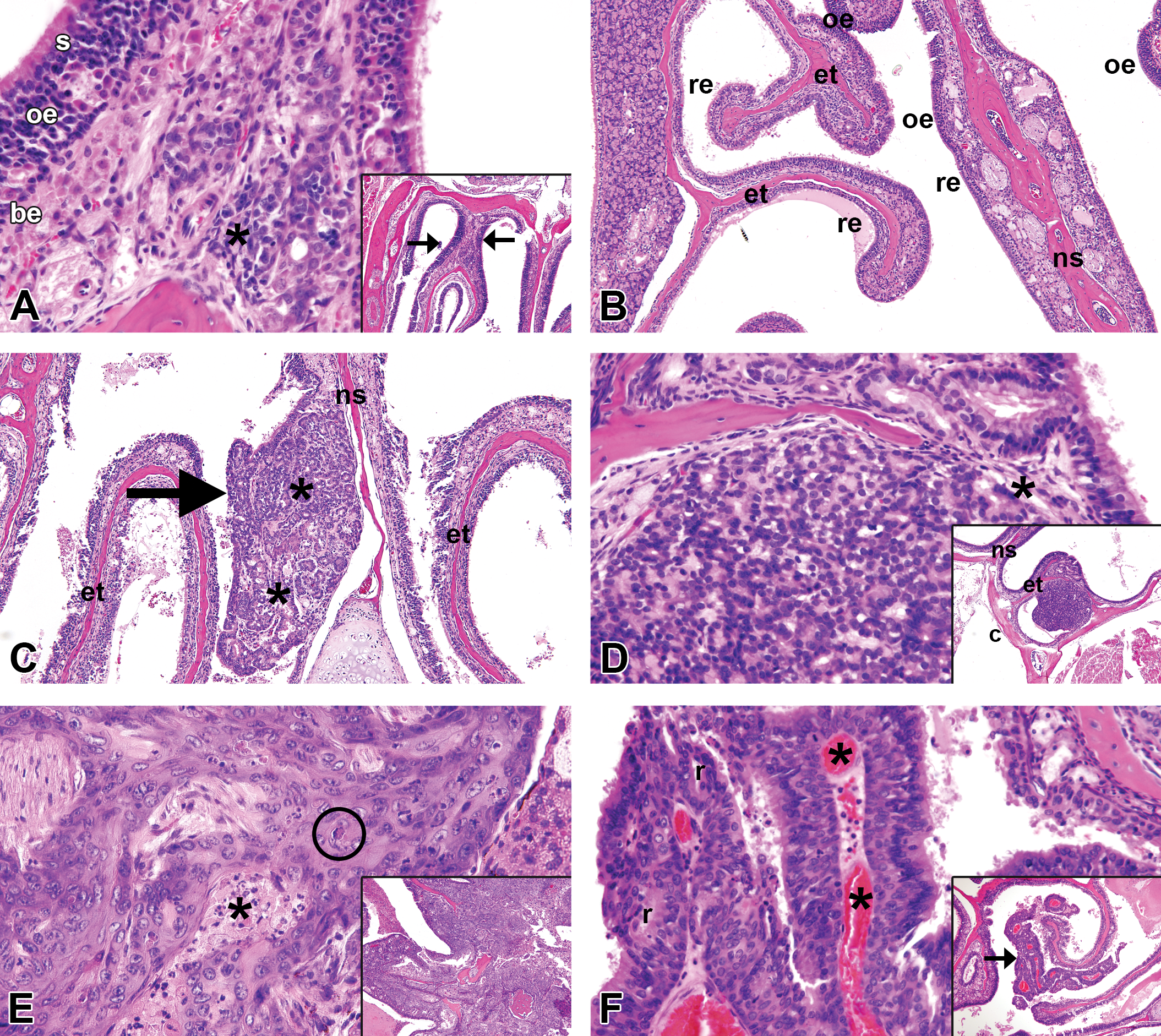
Nasal neoplasm-related lesions in DEN-treated mice. (A) Olfactory epithelial hyperplasia originating in the dorsal T2 region of the nasal cavity, original objective 40×. The olfactory epithelium is thickened due to an increase in olfactory sensory epithelial (oe) cells between basal epithelium (be) and larger surface sustentacular epithelial cells (s). Low-grade inflammation (asterisks) is also present. Inset, original objective 10×, shows the location of hyperplastic lesion (arrows), here bilateral. (B) Respiratory epithelial metaplasia of olfactory epithelium in the T3 (ethmoturbinate) region of the nasal cavity, 10×. Multilayered olfactory epithelium (oe) covering the ethmoturbinates (et) and nasal septum (ns) is replaced by columnar or pseudostratified columnar respiratory epithelium (re). (C) Adenoma of glandular origin arising in the nasal septum of the T4 region, original objective 10× (more typically arising in the anterior nasal cavity). The neoplasm is typically exophytic (arrow) with glandular morphology (asterisk). (D) Adenocarcinoma of glandular origin arising in the caudal T4 region (see inset, original objective 10×) near the floor of the calvarium (c), original objective 40×. The neoplasm is less well organized and less differentiated than an adenoma, with poor delineation of glandular structures and early invasion (asterisk) through the ethnoturbinate bone. (E) Squamous cell carcinoma, original objective 40×. This was the only nasal neoplasm visible externally, with point of origin unknown, due to profound invasion and effacement of nasal and adjacent cranial structures (inset, original objective 10×). The neoplasm is composed of irregular intersecting masses and cords of anaplastic squamous epithelial cells, with foci of necrosis (asterisk) and dyskeratosis (encircled). (F) Neuroepithelial carcinoma arising from an ethnotubinate (arrow; inset, original objective 10×) in the T3 region, original objective 40×. The neoplasm is highly vascularized (asterisks) with fine fibrous stroma. Nuclei are uniform in morphology and rosette-like structures (r) are present. Sections were stained with H&E.
Nasal Cavity Neoplasm–related Lesions.
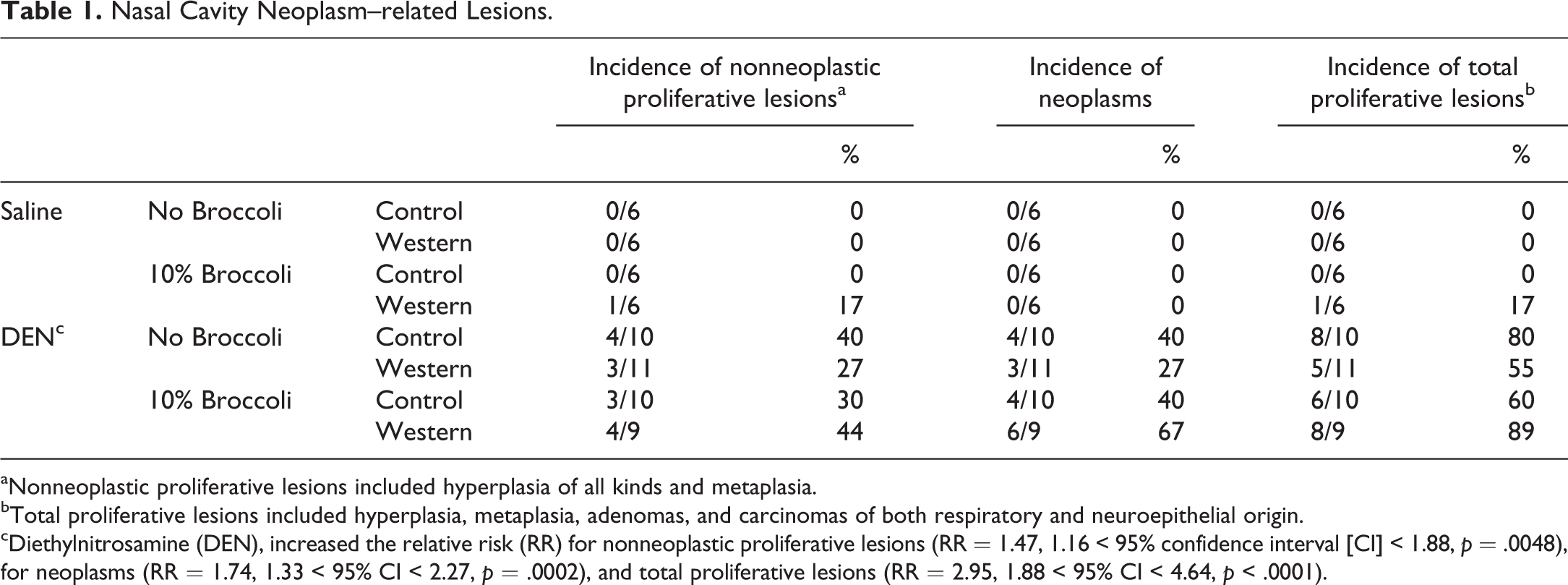
aNonneoplastic proliferative lesions included hyperplasia of all kinds and metaplasia.
bTotal proliferative lesions included hyperplasia, metaplasia, adenomas, and carcinomas of both respiratory and neuroepithelial origin.
cDiethylnitrosamine (DEN), increased the relative risk (RR) for nonneoplastic proliferative lesions (RR = 1.47, 1.16 < 95% confidence interval [CI] < 1.88, p = .0048), for neoplasms (RR = 1.74, 1.33 < 95% CI < 2.27, p = .0002), and total proliferative lesions (RR = 2.95, 1.88 < 95% CI < 4.64, p < .0001).
Six mice exhibited multiplicity of neoplasms, including 1 each in the DEN-treated control and control + 10% broccoli groups and 2 mice each in the Western and Western + 10% broccoli groups. The predominant neoplasm was adenocarcinoma (22 total), distributed among zones 1–4 of the nasal cavity. There were 2 adenomas (in mice with multiple neoplasms), 1 squamous cell carcinoma that was grossly visible at postmortem (Figure 1E), and 3 neuroepithelial carcinomas, all arising in zone 4, the most caudal part of the nasal cavity, lined almost entirely by olfactory epithelium.
Discussion
The hybrid mouse strain B6C3F1 is often used for the study of carcinogenesis. However, this strain also develops spontaneous neoplasms in multiple organs. The incidence of spontaneous hepatic adenoma/carcinoma and pulmonary adenoma/carcinoma in male B6C1F1 mice is high, reported to be 42% (range from 10% to 68%) and 20% (range from 4% to 23%), respectively (Haseman et al. 1998). Spontaneous nasal neoplasms are rare in B6C3F1 mice; the reported incidence is less than 1% (Brown et al. 1991; Haseman et al. 1998). Unexpectedly, we found a greater than 50% incidence of nasal lesions in the ip DEN-treated B6C3F1 mice, suggesting this may not be an unusual lesion in this model even though we are not aware of any report of DEN-induced nasal lesions in mice previously. Since most nasal lesions were not evident externally, such lesions may have been missed in earlier studies designed to evaluate hepatic neoplasms and thus sections of head would not have been taken.
DEN is known to be highly metabolized in the nasal cavity of C57BL/6J mice. Using [14C]-labeled DEN, a high level of nonvolatile DEN metabolites was observed in the nasal cavity, trachea, bronchi, salivary gland, liver, and tongue (Brittebo et al. 1981). This may provide the underlying reason for the high incidence of nasal neoplasms in this study. Microsomal Cyp 2E1 can activate DEN, producing carcinogenic metabolites (Kang et al. 2007). In the rodent nasal cavity, Cyp 2E1 is highly expressed in the olfactory epithelium, especially in the Bowman’s glands (Wang et al. 2002), which has been proposed as the site that N-nitrosamine-induced olfactory neoplasms are typically initiated (Brittebo 1997). Our results are consistent with this, showing a high quantity of neoplasms in the olfactory epithelium. To date, the Syrian hamster has been shown to respond to DEN with changes in the nasal cavity independent of route of DEN administration, including adenocarcinomas, carcinomas, and olfactory neuroepithelial neoplasms (Herrold 1964; Herrold and Dunham 1963). The high nasal neoplasm incidence in the DEN-treated Syrian hamster compared to rat or mouse has been suggested to reflect the relatively higher concentration of microsomal Cyp enzymes in nasal mucosa of the Syrian hamster (Longo et al. 1986; Sarkar 1992).
In conclusion, DEN induced not only liver and lung cancer but also nasal cancer in B6C3F1 mice, independent of dietary treatments. We are not aware of other reports of ip DEN-induced nasal neoplasia in B6C3F1 mice. Our findings suggest that possible metabolism of DEN in the nasal cavity, with subsequent induction of neoplasia, should be considered when choosing to use the B6C3F1-DEN liver cancer model.
Footnotes
Author Contributions
Authors contributed to conception or design (YC, MW, EJ); data acquisition, analysis, or interpretation (YC, MW, EJ); drafting the manuscript (YC); and critically revising the manuscript (YC, MW, EJ). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant number 5RO3CA162539-02 from the National Cancer Institute to EHJ.
