Abstract
Diethylnitrosamine (DEN), a hepatocarcinogen, is found in a variety of smoked and fried foods and was reported to be hepatotoxic in mice. Butylated hydroxytoluene (BHT) is a potent antioxidant used in cosmetic formulations and as a food additive and preservative. As a result, BHT was studied as a potential inhibitor in the early stages of diethylnitrosamine (DEN)-induced HCC. Male Wistar albino rats (n = 24) were equally subdivided. Group 1 was the negative control; Group 2 and 3 administered BHT and DEN, respectively; Group 4 received BHT followed by DEN. Blood samples and rat livers were taken for biochemical and histological investigation. Hepatotoxicity was assessed by increased liver enzymes and HCC indicators, along with reduced antioxidant and pro-apoptotic factors. AFP, AFPL3, GPC3, GSH, SOD, MDA, CASP3 and BAX expression increased significantly after DEN treatment. DEN also reduced GPx, CAT, and CYP2E1 activity, and BCl-2 expression. Moreover, in the hepatic parenchyma, the DEN caused histological alterations. Pretreatment with BHT enhanced antioxidant status while preventing histopathological and most biochemical alterations. BHT pretreatment suppresses DEN-initiated HCC by decreasing oxidative stress, triggering intrinsic mitotic apoptosis, and preventing histopathological changes in liver tissue.
Keywords
Introduction
Liver cancer is the sixth most commonly occurring cancer worldwide and the fourth leading cause of cancer mortality. 1 Hepatocellular carcinoma (HCC) occurs in the context of chronic liver disease. 2 The most prevalent cause of HCC is cirrhosis. 3 One of the main reasons of liver cancer is hepatitis B virus, accounting for 33% of all cases. Other causes include alcohol (30%), hepatitis C virus (HCV) (21%), and other factors (16%). 4 The liver being a vital organ is responsible for a wide range of metabolic-related functions, in addition to detoxifying various metabolites. 5 Reactive oxygen species (ROS) are byproducts to either normal cellular metabolism or from being exposed to a variety of xenobiotics and drugs. ROS play an essential role in liver carcinogenesis. 6 Chemoprevention may be a viable option for lowering the incidence, morbidity and mortality associated with hepatocarcinogenesis. 7
Diethylnitrosamine (DEN) is found in a variety of smoked and fried foods and was reported to be hepatotoxic in mice. Moreover, DEN-induced HCC in animals is histologically and biochemically identical to human HCC. 8 DEN is converted into alkylating agents that form mutagenic DNA adducts in the liver. Oncogenic mutations introduced in actively proliferating hepatocytes cause dysplastic lesions that may proceed to cancer. 9 DEN was reported to increase the malondialdehyde (MDA) levels because of increasing the ROS. 10 Inflammation is likely important for DEN-induced hepatocarcinogenesis. DEN is also a hepatotoxic substance that causes necrotic cell death. This injury initiates inflammation that leads to increased expression of mitogens, such as interleukin-6, that drive compensatory hepatocyte proliferation.10,11
Butylated hydroxytoluene (BHT) is a highly lipophilic antioxidant that depletes peroxyl radicals and thus arrests oxidative processes. The antioxidant is used in cosmetic formulations and as a food additive and preservative. 12 It was reported that BHT is safe when used in cosmetic formulations. The protective effect of BHT against many types of cancers were previously detected. BHT decreased the incidence of mammary cancer, lymphoma, liver neoplasms and pulmonary adenoma.12,13 However, the protective effect of BHT on the incidence of liver tumors was still controversial.14,15
Therefore, the goal of this study was to assess the role of BHT for protecting against DEN-induced HCC and discuss underlying metabolic pathways.
Materials and methods
Chemicals
BHT and DEN were obtained from Sigma-Aldrich (St Louis, MO, USA). All assay kits were purchased from Biodiagnostics (Cairo, Egypt). Primers were synthesized by Bioline Inc. (Taunton, MA, USA). All other chemicals were of analytical grades.
Animals
Wistar albino rats obtained from the animal house of Ahram Canadian University, Cairo, Egypt, weighing 150–170 g at the beginning of the experiments were used throughout the study. The animals were acclimatized to standard laboratory conditions (temperature 24 + 1°C, relative humidity 55 + 5% and a 12 h photoperiod) and were housed in Tarson Cages (three rats per cage) for 1 week before the commencement of the experiment. During the entire period of study, the rats were supplied with a semi-purified basal diet (El Nasr Co, Cairo, Egypt) and water ad libitum. All experimental techniques followed the guidelines of the Ethical Committee of Faculty of Pharmacy, Ahram Canadian University (Approval no. CLT221). All recommendations for the care and use of laboratory were strictly followed throughout the study.
Experimental design
The initiation experiment was designed based on a previous report of Chakraborty et al.
16
The experiment extended for 8 days and was performed on rats at 9 weeks of age. Different sets of rats were randomly divided into four experimental groups (n = 6) for carrying out molecular studies (short-term studies) as shown in Figure 1. Groups I and II rats were the normal and BHT controls, respectively. Group III rats were the DEN control, while Group IV received BHT followed by DEN. Animals in Groups II and IV were provided BHT (0.8 mg/kg/day) in drinking water for 7 days ad libitum. Groups III and IV rats were the DEN-treated groups that received a single, necrogenic, and intraperitoneal (i.p.) injection of DEN (200 mg/kg body weight in 0.9% saline) on day 8 (after 1 week). A retro-orbital venous plexus hemorrhage was used to obtain blood samples under light ether anesthesia. After overnight fasting, all the rats were sacrificed by decapitation between 09:00 and 11:00 h under proper light ether anesthesia. The basic experimental regimen for molecular studies.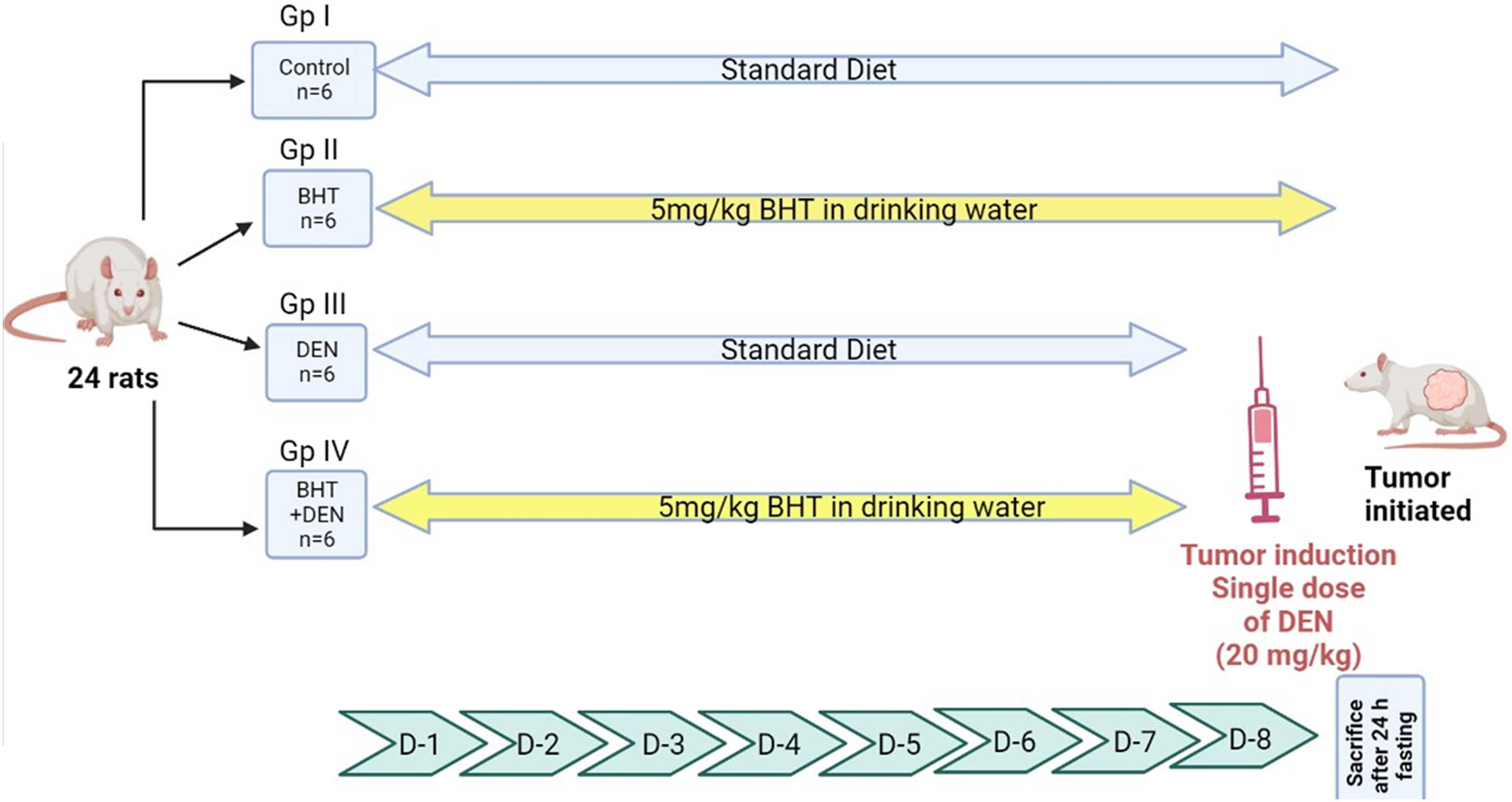
Methods
Liver function tests
Whole blood was centrifuged at 4000 r/min for 10 min to get serum which was then stored at −80°C for subsequent examination. Colorimetric assay kits were used for estimating serum levels of ALT, AST 17 and total proteins. 18
Hepatic antioxidant status
Homogenization of liver tissue was done in ice-cold phosphate buffer (20% w/v homogenate) and centrifuged at 3000 r/min for 15 min. Oxidative stress markers were assessed in supernatants, including reduced glutathione (GSH), superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), cytochrome p450 family 2 subfamily E member 1 (CYP2E1) and MDA.
GSH levels were assayed using 5,5′-dithiobis (2-nitrobenzoic acid) and the produced color was quantified with a spectrophotometer (Beckman DU-64 UV/VIS) using absorption at 405 nm. 18 SOD activity was also measured spectrophotometrically using inhibition of superoxide-driven NADH oxidation. 19 Antioxidant enzymatic activity of CAT was assessed by rate of H2O2 conversion to water and O2 at 510 nm. 20 GPx activity was estimated as previously described. 21 CYP2E1 was measured with an ELISA kit (MyBioSource, Cat # MBS453581, San Diego, CA, USA). MDA was evaluated colorimetrically at 535 nm as previously described. 22
Alpha fetoprotein (AFP) and AFP L3
Their serum levels were assessed quantitatively using ELISA kits (Elabscience, Cat # E-EL-R0153, USA) and (LifeSpan BioSciences, Cat # LS-F40468, USA), respectively, following the manufacturer’s instructions.
Estimation of Glypican 3 (GPC3)
For protein extraction, liver tissue homogenization was done in Tris buffer pH 7.4, NaF, EDTA, β-glycerol phosphate, Na3VO4, sodium deoxycholate, PMSF, and a protease inhibitor cocktail (Sigma, P8340), then the mixture was centrifuged. A Bradford quantitative protein assay kit (Bio basic inc., Cat # BDE641, Ontario, Canada) was used to assess total protein in supernatants. Proteins were separated on a 12% SDS-PAGE before being transferred to PVDF membranes (Sigma-Aldrich, Cat # GE10600021, MO, USA). Membranes were blocked with TBS-T for 2 hours. Subsequently, membranes were incubated with primary rabbit monoclonal antibodies (Cell Signaling, Cat # 2870, UK) and mouse anti-actin (1:1000; Cell Signaling, Cat # 3700, UK)—at 4°C overnight. Finally, membranes were mixed with secondary labeled antimouse IgG antibodies (Cell Signaling, Cat # 7076, UK). An enhanced chemiluminescence kit (Bio-Rad, Cat #170-5060, CA, USA) was used to visualize protein bands then it was captured by a CCD camera-based imager. Image analysis software, the ChemiDoc MP imager utilized to compare the target proteins’ band intensity relative to the control housekeeping protein, beta actin.
Gene expression of BCl-2, BAX, and CASP3
Primer sequences.

Statistical analysis
Kolmogorov-Smirnov and Shapiro–Wilk tests were used to assess normality (p > 0.05), and we were able to use one-way ANOVA (parametric test). Tukey HSD tests were used post hoc due to homogeneity of variances. Means and standard deviations of continuous variables are reported. IBM SPSS Statistics software version 26 was used for analyses. p < 0.05 was used as the statistical significance threshold. GraphPad Prism Software version 9.1.1 was used to make the graphs.
Results
Estimation of liver functions
DEN-treated rats exhibited a significant elevation (p < 0.001) in the activities of ALT (Figure 2(a)) and AST (Figure 2(b)) and a significant decrease in total protein levels (p < 0.001) (Figure 2(c)) compared to control animals. In contrast, marker enzymes in DEN + BHT-pretreated rats (Group IV) were significantly (p < 0.001) reduced, and levels of total protein significantly (p < 0.001) increased compared to DEN-treated rats. BHT alone did not significantly change serum ALT or AST activities, or total protein levels compared to negative controls. Effects of DEN, BHT and their combination on serum ALT (Figure 1(a)), AST (Figure 1(b)), and total proteins (Figure 1(c)). **Statistically significant, p < 0.001. *** Statistically significant, p < 0.0001.
Measurement of HCC biomarkers
Impacts of BHT alone on levels of AFP, AFPL3, and GPC3 as carcinogenesis biomarkers were not significant. Conversely, DEN administration caused a significant elevation in AFP (4.2-fold), AFPL3 (3.5-fold), and GPC3 (3.4-fold) compared to controls (p < 0.0001). However, BHT pretreatment significantly (p < 0.0001) countered the impact of DEN alone on AFP, AFP-L3, and GPC3 levels by 68%, 64%, and 56.3%, respectively (Figure 3). Effects of DEN, BHT and their combination on HCC biomarkers, AFP (Figure 2(a)), AFP-L3 (Figure 2(b)), and GPC3 (Figure 2(c)). ***Statistically significant, p < 0.0001.
Lipid peroxidation and antioxidant levels
No significant differences in GSH (Figure 4(a)), CAT (Figure 4(b)), GPx (Figure 4(c)), SOD (Figure 4(d)), CYP2E1 (Figure 4(e)), and MDA levels (Figure 4(f)) were observed between negative controls and animals treated with BHT alone. However, DEN administration increased MDA (4.2-fold) and CYP2E1 (2.6-fold) when compared to the control group (p < 0.0001). Further, significant decreases were seen in hepatic GSH levels and activities of CAT, GPx, and SOD activity by 39%, 77%, 72% and 40%, respectively. Pretreatment of rats with BHT significantly reversed MDA and CYP2E1 levels, and rescued hepatic GSH level and activities of CAT, GPx, SOD. Interestingly, antioxidant levels were restored to near-normal (Figure 4) (p < 0.001). Effects of DEN, BHT and their combination on GSH levels (Figure 3(a)) and the activity of the antioxidant enzymes, CAT (Figure 3(b)), GPx (Figure 3(c)), SOD (Figure 3(d)), CYP2E1 (Figure 3(e)), and status of lipid peroxidation measured as MDA (Figure 3(f)). *Statistically significant, p < 0.01. ***Statistically significant, p < 0.0001.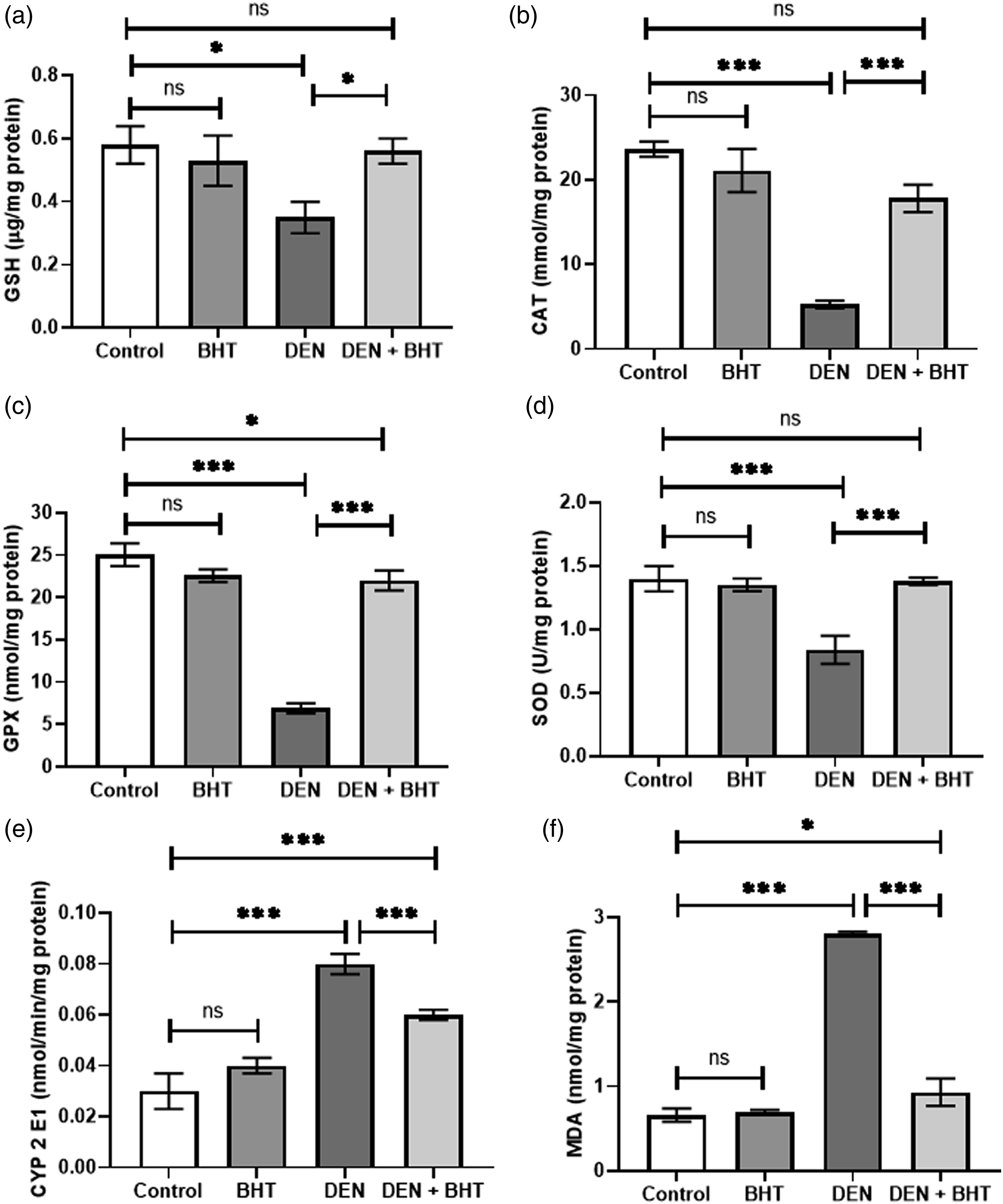
Apoptotic biomarkers
As shown in Figure 4, BHT treatment did not significantly change gene expression of BCL-2, pro-apoptotic BAX, BAX: BCL-2 ratio, or CASP3. DEN treatment upregulated BCL-2 by 24% but downregulated BAX and CASP3 by 50.4% and 50%, respectively. Pretreatment with BHT significantly reduced BCL-2 and elevated BAX and CASP3 gene expression compared to DEN-treated animals (Figure 5). Effects of DEN, BHT and their combination on the gene expression pro-apoptotic BAX (Figure 4(a)), anti-apoptotic Bcl-2 (Figure 4(b)), BAX: Bcl-2 ratio (Figure 4(c)) and CASP3 (Figure 4(d)). *Statistically significant, p < 0.01. **Statistically significant, p < 0.001. ***Statistically significant, p < 0.0001.
Liver histopathology
The H&E staining results are presented in Figure 6. No abnormal findings were observed in liver tissue from control or BHT-treated rats, indicating a lack of BHT toxicity (Figures 6(a) and (b), respectively). However, livers from DEN-treated rats showed a lack of typical hepatocyte architecture and lobules of neoplasm with areas of large fatty tumor cells characterized by irregular nuclei and numerous mitotic figures (Figure 6(c)). Hepatocytes pretreated treatment with BHT had fewer neoplastically altered cells and a near-normal architecture., implying that BHT might help prevent DEN-induced liver cancers. (Figure 6(d)). Representative photomicrographs of liver histopathology: (Figure 5(a)) The liver of control showed no abnormal findings. (Figure 5(b)) BHT-treated group showed no changes in liver tissues. (Figures 5(c)1–5) In the DEN-induced group, the liver showed hepatocytes with a lack of architecture and lobules of neoplasm with an area of large fatty tumor cells characterized by having various irregular nuclei and numerous mitotic cells. (Figures 5(d)1 and 2) Rats given BHT had fewer neoplastically altered cells, and hepatocytes exhibited nearly normal architecture.
Discussion
HCC is the most common liver malignancy worldwide and a principal cause of mortality. 24 BHT is a synthetically prepared phenolic antioxidant, which is mainly used in foods containing solid and liquid fat, packing materials, and cosmetic products. 25 Fermentation and other oxidation events can cause fat and fat-containing foods to deteriorate. BHT acts as radical scavenger against lipid peroxidation chain reactions and thereby protects foods from deterioration. 26 As per the FDA, BHT acceptable daily intake for man is 0.5 mg BHT/kg body weight.27–29 BHT, at a dose higher than 150 mg/kg body weight, forms a metabolic intermediate called quinone methide which is a reactive and electrophilic metabolite that can covalently bind to various cellular nucleophiles leading to acute hepatotoxicity and pneumotoxicity.30,31
This study explored the modulation and therapeutic potential of BHT on the initiation of DEN-induced HCC with a focus on mode of action. A rat model of HCC initiation was created using DEN activation by cytochrome P450 enzymes. This reaction leads to formation of electrophilic species responsible for cytotoxicity, mutagenicity and carcinogenesis. 32 DEN is a carcinogen that produces reactive oxygen species (ROS) that are normally scavenged by endogenous antioxidants. 33 This action helps prevent oxidative DNA destruction. 34
In our study, injection of rats with DEN-induced early HCC manifestations, including significant deterioration of liver architecture leading to substantial increases in serum AST and ALT activity. These enzymes are also elevated in HCC. 35 In addition, a previous study found hypertrophy and increase in mitosis in liver cells after using DEN once for a week. 36 In the current study, pretreatment with BHT (0.8 mg/kg/day) for a week significantly reduced serum enzyme activity and largely restored hepatic architecture. Thus, BHT exhibits protection against loss of structure and function after DEN administration. Low doses of BHT have been proven to suppress carcinogenesis by guarding against harmful chemicals formed from the degradation of nutrients and the oxidation of fatty acids in diets.32–34
The biomarker AFP is used to detect HCC. 37 AFP-L3, a malignant tumor-specific isoform of AFP, is utilized to determine the risk of developing HCC. AFP-L3 can identify HCC before AFP and aid in the diagnosis of AFP-negative HCC. 38 GPC-3 is vital for cell proliferation and differentiation and is variably expressed during invasive growth of liver tumors. Its expression may reflect liver cancer onset and progression. 39 Our results demonstrate significantly increased serum AFP, AFP-L3 and GPC3, decreased total protein levels, and malignant transformation of hepatocytes that indicate hepatic dysfunction upon DEN administration. HCC and chronic liver disorders are distinguished by AFP levels, which are increased in the early phases of HCC induction. 40 An increase in AFP levels in DEN-intoxicated rats was previously reported. 41 Also, early-stage HCC was characterized by high levels of GPC-3 in both serum and tissues. 42 When compared to DEN-intoxicated rats, pretreatment with BHT induced a considerable reduction in AFP, AFP-L3, and GPC3 levels. This action might reflect BHT anticancer properties. Suppression of HCC development is supported by a decrease in AFP and liver function enzyme levels compared to HCC-bearing rats. 26
DEN effect could be attributed to the fact that it generates ROS that damages DNA and leads to carcinogenesis.34,43,44 Endogenous antioxidants deactivate ROS via the elimination of free radicals before DNA damage occurs. 44 GPx and SOD enzymes are responsible for free radical elimination. SOD can eliminate highly toxic superoxide anions; GPx avoids the creation of harmful peroxyl radicals. 45 Hydrogen peroxide is also broken down immediately by CAT. 46 GSH scavenges dangerous electrophilic chemicals via conjugation to less harmful products. 47 The activity of SOD, CAT, and GPx enzymes in the present study, as well as the amount of GSH, were significantly reduced by DEN treatment; MDA levels were, however, significantly increased, indicating that the liver’s antioxidant defense system had been entirely compromised. The dose of DEN exceeded the capacity of liver cells to counter oxidative stress and led to disruption of liver histology. This stress led to membrane breakdown, inflammatory cell infiltration, and initiation of HCC. 48
Liver tissue DEN-induced HCC models showed increased ROS generation and decreased antioxidant enzymes.49,50 Rats pretreated with BHT showed significant restoration of antioxidant activity via significantly increasing SOD, CAT, and GPx activity and GSH concentrations. Likewise, BHT supplementation rescued hepatic damage markers to normal levels, lowered MDA levels, and significantly improved antioxidant status. Further, BHT treatment reduced the negative histopathological impacts on hepatocytes and partially restored normal histological architecture. These effects may reflect significant activity of BHT for scavenging free radicals and mitigating their harmful impacts.28,51,52
Cytochrome P450s are important enzymes in cancer initiation and treatment. 53 CYP2E1 is a cytochrome P450 isozyme that often converts exogenous chemicals to electrophilic metabolites that produce oxidative stress, 54 and contributes to the metabolic activation of certain procarcinogens. 55 Our results showed overexpression of the CYP2E1 gene in the liver of DEN-treated rats. CYP2E1 is known to metabolize DEN that results in the formation of toxic metabolite, which can cause DNA damage and, subsequently, carcinogenesis. In comparison to the DEN-treated group, BHT-treated mice displayed significantly lower CYP2E1 level. Conversely, the protective effect of BHT against hepatotoxicity, that was induced by acetaminophen, does not involve changes in CYP2E1 enzyme levels. 56
Bcl-2 is an anti-apoptotic protein, and BAX, a pro-apoptotic protein. These proteins are well-known for regulating cell apoptosis. 55 Moreover, activation of a caspase cascade is crucial in producing cell death. 57 Dysfunction in apoptosis makes cancer treatment more difficult and aids tumor progression. 55 In our study, a significant upregulation in Bcl-2 and downregulation in BAX and CASP3 were detected in livers of DEN-treated animals. BHT initiated apoptosis via reversing this DEN’s effect on Bcl-2, BAX and CASP3. This modulation of gene expression that might be the crucial mechanism for preventing HCC. These results are consistent with previously reported DEN-induced cell proliferation. 58
The current study showed that the synthetic phenolic antioxidant BHT, added to human and animal food, can lower the incidence of cancer caused by chemical compounds as evidenced by counteracting the adverse effects of DEN and protecting the liver from injury and carcinogenesis. BHT lowers HCC biomarkers, oxidative stress, and lipid peroxidation and increases antioxidative enzyme activity and activates apoptotic signaling.
Footnotes
Author contributions
Conception and design: SF and OB; Development of methodology: SI and SF; Acquisition of data (provided animals, provided facilities, etc.): SI; Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): ST and SF; Writing, review, and/or revision of the manuscript: ST, SF, and SI, Study supervision: OB. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
