Abstract
In order to harmonize diagnostic terminology, confirm diagnostic criteria, and describe aspects of tumor biology characteristic of different tumor types, a total of 165 cases of mesenchyme-related tumors and nephroblastomas of the rat kidney were reexamined from the National Toxicology Program (NTP) Archives. This survey demonstrated that renal mesenchymal tumor (RMT) was the most common spontaneous nonepithelial tumor in the rat kidney, also occurring more frequently in the NTP studies than nephroblastoma. Renal sarcoma was a distinct but very rare tumor entity, representing a malignant, monomorphous population of densely crowded, fibroblast-like cells, in which, unlike RMT, preexisting tubules did not persist. Nephroblastoma was characterized by early death of affected animals, suggesting an embryonal origin for this tumor type. Male and female rats were equally disposed to developing RMT, but most of the cases of nephroblastoma occurred in female rats and liposarcoma occurred mostly in male rats. This survey confirmed discrete histopathological and biological differences between the mesenchyme-related renal tumor types and between RMT and nephroblastoma. Statistical analysis also demonstrated a lack of any relationship of these renal tumor types to test article administration in the NTP data bank.
Keywords
Introduction
For many years, a distinct renal tumor entity has been recognized in rat kidney that has mesenchymal features, defining origin from mesenchymal and not epithelial tissue (Hard 1998). This neoplasm has been experimentally induced by certain genotoxic carcinogens, especially nitrosamines, the cycad toxin cycasin (Hirono, Laqueur, and Spatz 1968), methylazoxymethanol (Laqueur and Matsumoto 1966), and dimethylhydrazine (Sunter and Senior 1983). Using a single, high-incidence dose of dimethylnitrosamine (DMN), the morphology of a comprehensive range of these tumors was studied at the light and electron microscopic levels (Hard and Butler 1970a, 1971b), and based on the exclusive, mesenchymal nature of the tumors, was termed renal mesenchymal tumor (RMT). Furthermore, development of the tumors was traced directly to mesenchymal cells originating from interstitial cell lesions induced by DMN (Hard and Butler 1970b, 1971a). At the same time, the term stromal nephroma was coined by Riopelle and Jasmin for these DMN-induced renal tumors (Riopelle and Jasmin 1969), and many other synonyms have been used in the early and intervening years for this tumor type, including mixed malignant tumor, sarcomatoid tumor, hemangioendothelioma, interstitial tumor of kidney, and renal sarcoma (Hard 1998). However, the term RMT has persisted through the years and has become the nomenclature accepted by the Society of Toxicologic Pathology (STP) and other international toxicologic pathology organizations (Frazier et al. 2012).
As stated above, these tumors have been induced only by genotoxic carcinogens. However, they do occur sporadically across some 2-year carcinogenicity bioassays conducted in the National Cancer Institute (NCI)/NTP program for identifying chemical carcinogens. In this setting, mesenchymal tumors appear to have been given various names, including mesenchymal tumor benign, mesenchymal tumor malignant, stromal nephroma, mixed tumor (benign or malignant), myxoid tumor, sarcoma, and fibrosarcoma. There is therefore a need to determine whether these tumor examples all belong to the same entity as RMT, or if indeed, there are different subtypes of mesenchymal tumor in the National Toxicology Program (NTP) series. Any variation in nomenclature for the same tumor entity could alter statistical incidence and subsequent assessment of the chemical’s weight of evidence as a possible carcinogen. For example, in the NTP database of long-term studies, 1 chemical—methyl isobutyl ketone (MIBK)—was identified as causing RMT. In the 2-year carcinogenicity study of MIBK (NTP 2007), NTP concluded that there was equivocal evidence of carcinogenic activity in female rats based on the occurrence of 2 mesenchymal tumors observed in the high-dose group, and no record of this tumor type in control rat groups of any NTP study at that time. Using a consistent approach in the terminology of RMT would provide a statistical understanding of the scatter of such uncommon tumors through control and dose groups in order to place better perspective on incidence, and association or not, with chemical exposure.
All RMTs contain many remnants of preexisting, entrapped tubules and, sometimes, hyperplastic pelvic urothelium. The survival and hyperplastic nature of these elements are probably influenced by paracrine growth factors or other stimuli associated with the primitive mesenchymal tumor cells. This combination of malignant mesenchyme with nonneoplastic epithelium has been a frequent impediment for consistent and accurate identification of RMT in rats and its distinction from nephroblastoma, which is essentially an epithelial tumor (Hard and Butler 1970a; Hard 1978; Seely 2004). Consequently, aspects of RMT are sometimes confused with nephroblastoma (Hadjiolov 1968; Sadrudin, Lewin, and Cruse 1985; Mitsumori et al. 2002). For example, in the Toxicology Data Management System (TDMS) printout of the NTP tetralin 2-year bioassay, the primary kidney tumor in 1 animal was classified as nephroblastoma, but metastases to lung and lymph node in the same animal were diagnosed as stromal nephroma (synonymous with RMT). The NTP collection of renal tumors therefore provides an opportunity to emphasize the differences between nephroblastoma and RMT.
Accordingly, the aim of this study was to histologically reevaluate the large database of mesenchyme-related, nonepithelial renal tumors and nephroblastomas of rats in the NTP Archives, in order to harmonize the diagnostic terms used, to establish and/or highlight criteria that distinguish the various mesenchymal renal tumor types and nephroblastoma, and to determine any association of these tumor types with test article exposure.
Materials and Methods
Survey Plan
In the database of the NTP, test articles evaluated for toxicity or carcinogenicity are systematically assigned a number and are logged under either the Carcinogenicity Bioassay Data System, representing studies reported from 1971 to 1982, or the TDMS, containing the results of studies reported from 1982 to the present time. In this study of the occurrence of mesenchyme-related renal tumor types and nephroblastoma, the TDMS was surveyed from inception up to the beginning of 2015 for the diagnostic terms: mesenchymal tumor benign, mesenchymal tumor malignant, mixed tumor (benign or malignant), myxoid tumor, hamartoma, fibroma/fibrosarcoma, sarcoma, liposarcoma, hemangioma/hemangiosarcoma, stromal nephroma, and nephroblastoma. This electronic search retrieved 204 cases of rats recorded with 1 of these tumor types. Thirty-two of these specimens were held at the National Center for Toxicological Research (NCTR). Whole-slide imaging of these cases using the Aperio system by NCTR enabled evaluation of tumor identity at the premises of the NTP Archives in Research Triangle Park, North Carolina, along with the specimens held in the Archives. Of the 204 specimens identified in the search, only 6 were not available for microscopic reevaluation. The survey therefore reexamined the kidneys of a total of 198 tumor-bearing animals, representing studies on 116 chemicals, extracts, agents, or mixtures.
Because of the digitized histopathology lexicon used by NTP, in the TDMS, the term sarcoma retrieved cases of metastases to the kidney from nonrenal primary sites. Although examined, these were not considered relevant to the purpose of this survey of mesenchyme-related tumors of rat kidney origin and were excluded from consideration here. Also excluded from the survey was the term lipoma due to the relatively common occurrence of this proliferative lesion and a lack of any diagnostic uncertainty.
Statistical Methods
Cochran–Armitage trend tests and Fisher’s exact tests were used to assess the relationship between dose of chemical, agent, mixture, or extract administered and the occurrence of each specific tumor type, using Statistical Analysis System (SAS 2011) software. In addition to analyzing each chemical study separately, those having 4 (control, low dose, mid dose, and high dose) or 3 (control, low dose, and high dose) dose groups were combined and analyzed. In all tests, a p value of .05 was considered the limit for significance.
Results
Excluding cases of mesenchyme-related metastasis to the kidney from nonrenal sites, as well as a few hemangiomatous tumors that were considered by the authors to be long-standing, mature thrombi, or in 1 case, a stromal polyp arising from the renal pelvis lining, the evaluation found 165 specimens with a renal tumor type identified by 1 of the search terms of interest. The results of the survey are summarized in Table 1. Most of the studies used the F344 strain of rat, but some, such as the Toxic Equivalency Factor (TEF) studies, used Harlan Sprague-Dawley rats, and those with ethinyl estradiol and genistein, used the NCTR Sprague-Dawley (CD23/NCTR BR). One study used rats of the Osborne–Mendel strain. In general, there was very good concordance between the diagnoses made in this survey and the recorded NTP diagnoses when the use of synonyms of RMT was taken into account.
Summary of Incidence and Special Features of Mesenchyme-related Tumors and Nephroblastoma in the Database of the National Toxicology Program.
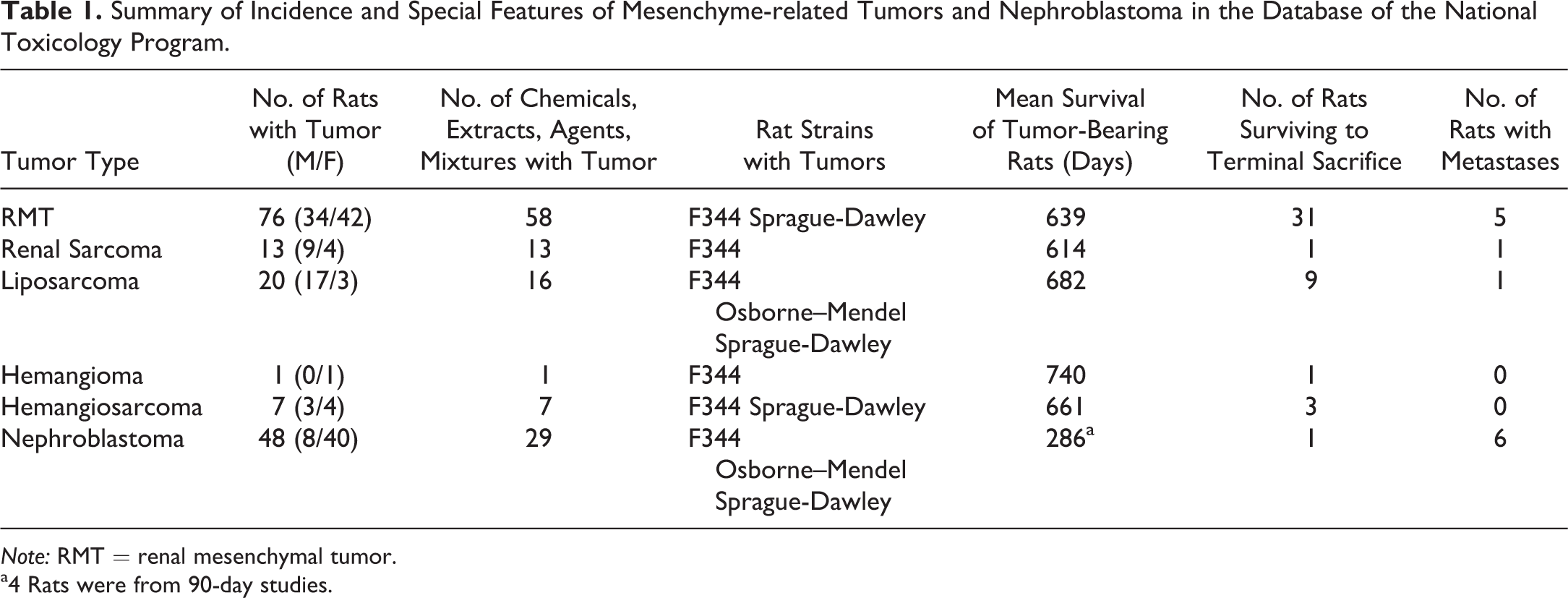
Note: RMT = renal mesenchymal tumor.
a4 Rats were from 90-day studies.
RMT
Of the 165 tumor specimens pertinent to this survey, 76 were RMTs, involving 58 chemicals, extracts, agents, or mixtures. These are listed, along with relevant data in Supplemental Table S1. All of the tumors diagnosed by NTP with the synonymous terms of mixed benign or malignant tumors, myxoid tumors, or stromal nephromas were RMTs. These tumors consisted of a heterogeneous mixture of primitive mesenchyme, fibroblasts, smooth muscle cells, and entrapped preexisting tubule profiles, which were frequently altered by dilation or simple hyperplasia (Figure 1A). Deposits of collagen were also prominent. Invasion into the renal pelvis was quite frequent, with preexisting urothelium forming islands (Figure 1B) or tracts ramifying through the invading tumor tissue. In an occasional tumor, there were islands of cartilage, osteoid, or rhabdoid elements. Mitotic figures were most conspicuous in tumor cells infiltrating in the interstitium between normal tubules at the invading edge of the tumors.
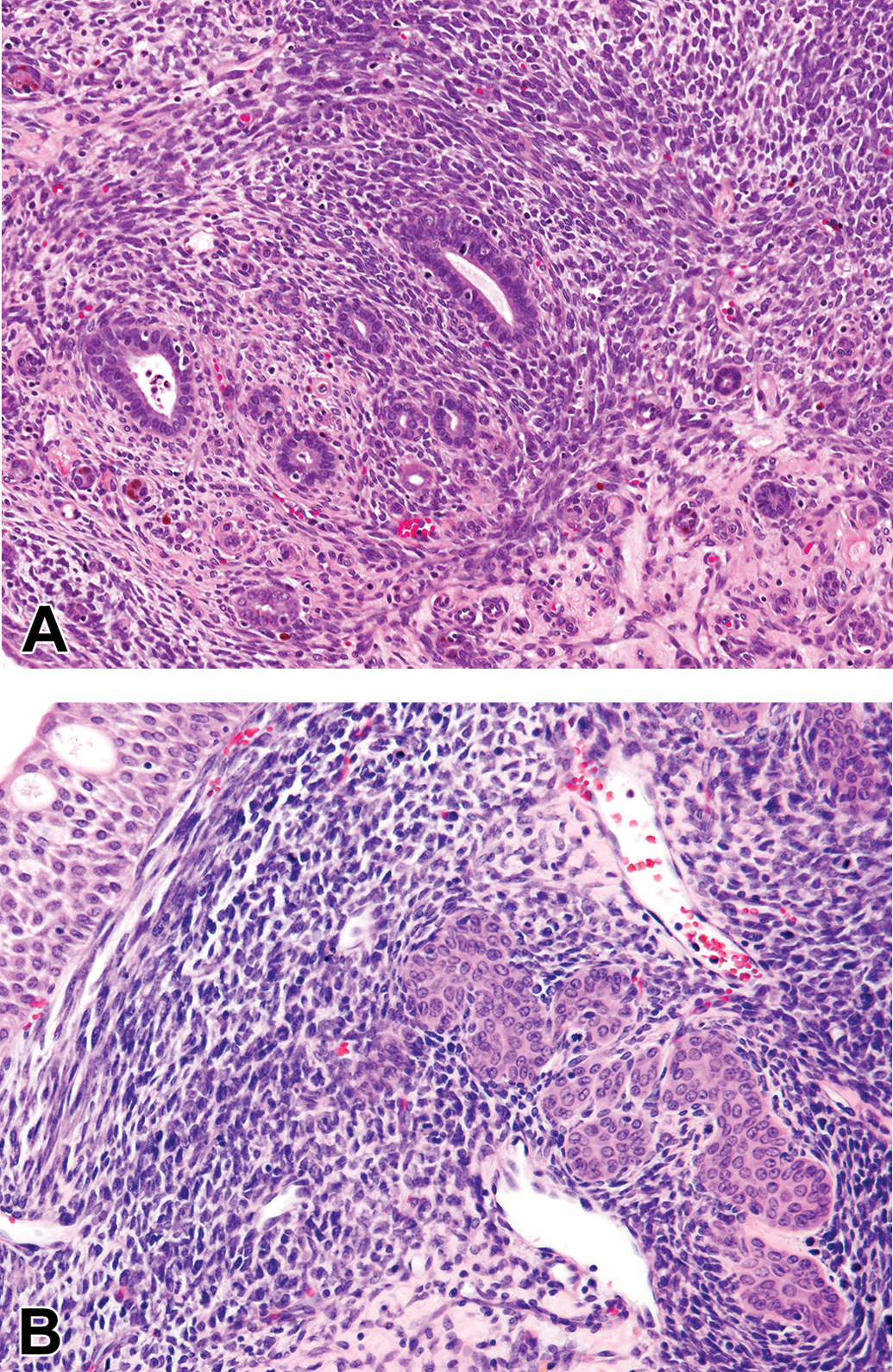
(A) Rat renal mesenchymal tumor (RMT) typically shows heterogeneous areas with primitive mesenchymal cells swirling around entrapped preexisting tubules, which often display simple hyperplasia of their lining. (B) Higher magnification of RMT showing mesenchymal tumor cells invading the renal pelvis. This common development leads to hyperplastic proliferation of the preexisting urothelial lining of the pelvis, which forms islands or ramifying cords of transitional cells within the RMT (hematoxylin and eosin).
RMTs were frequently very large and metastasis was observed in 5 cases. In 4 of these, metastasis involved the lung. Two of the tumors with lung metastasis had invasion of the mesentery as well, and 1 case also involved the visceral organs. The 5th animal had metastasis to the mesentery alone. The sexes were equally affected, with RMT being found in 33 animals of each sex in studies using both males and females. Although some early deaths occurred, many of the rats with RMT survived until the terminal sacrifice at 2 years, but in a number of cases, RMT was considered by NTP to have been the cause of death. The mean survival time for rats with this tumor type was 639 days.
More than 2 RMT occurred in carcinogenicity studies of ethylene thiourea (3 cases), nickel (II) oxide (3 cases), isobutene (3 cases), PCB 126 TEF evaluation (5 cases), and PCB 153 TEF evaluation (3 cases). Qualitatively, tumor occurrence was distributed evenly among control and treatment groups. For example, 3 rats in the isobutene study were diagnosed with RMT, 1 each in the control, low-dose, and high-dose groups. The Sprague-Dawley rats used in the TEF evaluation of PCB 126 appeared to have a higher incidence of RMT than any single study using F344 rats, with 5 cases of RMT in 423 female Sprague-Dawley rats. As observed with all RMTs in the survey, the distribution among the treatment groups of PCB 126 was random, with a single rat at each of exposure levels 3, 5, 6, 7, and 8 (of 8 exposure levels) having an RMT.
Renal Sarcoma
The 13 tumors diagnosed in this survey as renal sarcoma are listed in Supplemental Table S2. In all but one case, the tumor tissue was restricted to the kidney for those particular animals according to the NTP Report’s individual animal pathology pages, implicating the sarcoma as a primary tumor of the kidney. All 13 cases were seen in F344 rats with none in the Sprague-Dawley strain.
Renal sarcomas tended to be large tumors with areas of autolysis and hemorrhagic spaces (the latter probably due to postmortem change also) and were recorded as the primary cause of death by NTP in 9 cases. Histologically, these tumors consistently comprised a highly malignant, monomorphic, densely cellular sheet of spindle-shaped, fibrocyte-like cells, often arranged in fascicular or swirling pattern (Figure 2A and 2B). Collagen was absent or minimal and the tumor tissue lacked entrapped tubules or glomeruli, except sporadically at the tumor periphery. Tubules appeared to degenerate and disappear rapidly when entrapped, unlike the case with RMTs. When the tumor extended into the renal sinus, there was sometimes a trail of engulfed lipocytes present in the tumor tissue. Renal sarcomas appeared to grow mainly by expansion because parts of the tumor periphery were bordered by a band of fibrous tissue. However, local invasion into the renal parenchyma was also observed, and invasion into the viscera (pancreas and small intestine) occurred in 1 case. Mitotic figures were variable but frequent in some cases.
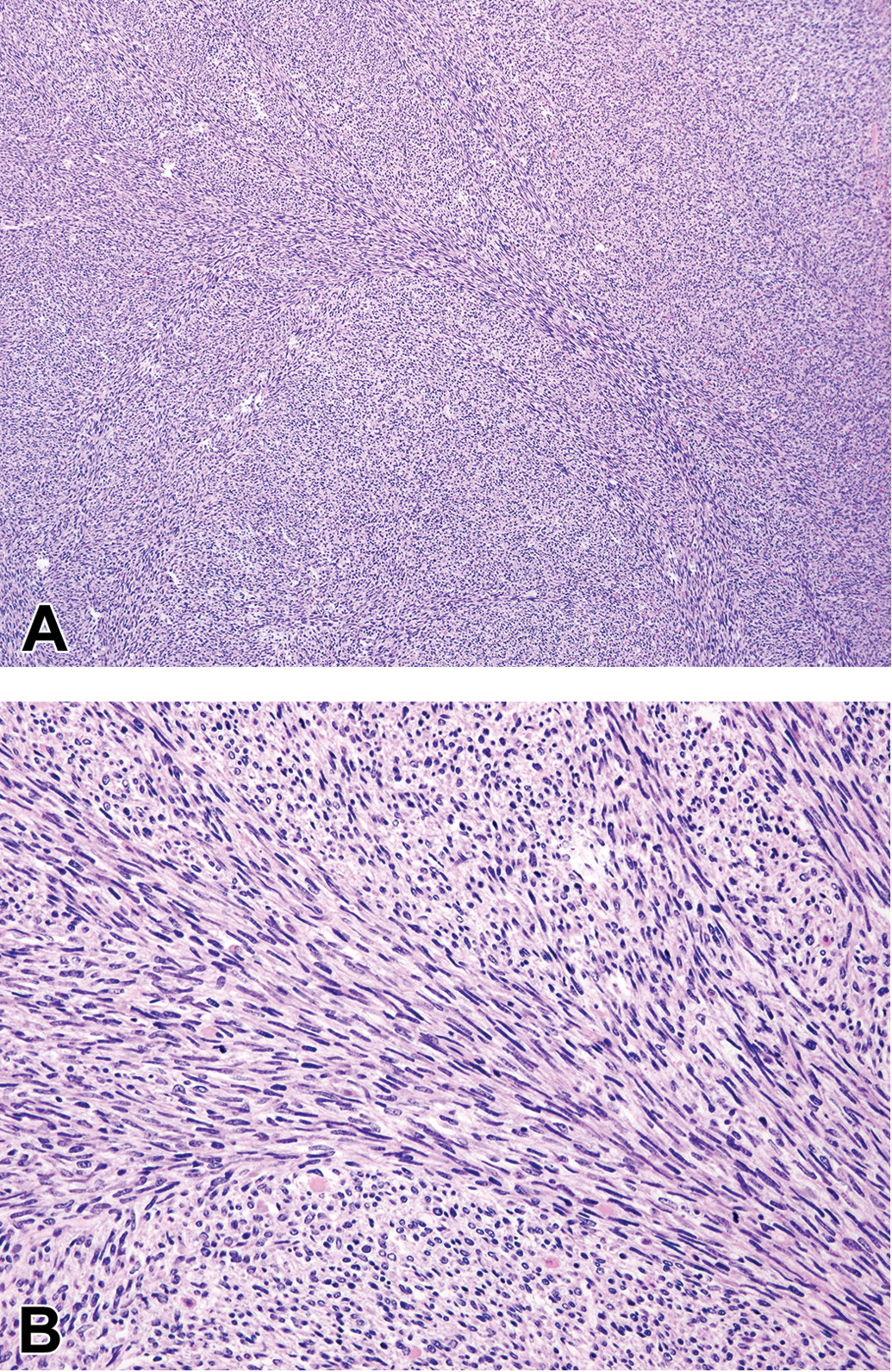
(A) Renal sarcoma is typified by homogeneous sheets of fibrosarcoma-like tissue without any entrapped parenchymal structures or prominent collagen deposition. (B). Higher magnification of renal sarcoma shows sarcomatous cells in a fascicular pattern, without prominent collagen formation (hematoxylin and eosin).
The mean survival for rats with renal sarcoma was 613.5 days. The gender ratio was approximately 2:1, with 9 of these renal tumors occurring in males and 4 in females.
Renal Liposarcoma
There were 20 renal liposarcomas in the series, which are listed in Supplemental Table S3. These were characterized by sheets of mature lipocytes infiltrating between preexisting renal tubules and glomeruli (Figure 3). Immature fat cells, lipoblasts, and islands of primitive mesenchymal cells were also present. Red blood cells were usually prominent, either in congested vessels or as extravasated clusters. Only 1 liposarcoma was considered to have metastasized to distant organs, namely, the lung and seminal vesicles. However, the diagnosis of this particular tumor was uncertain because it also contained smooth muscle, which has not been reported previously in rat liposarcomas.
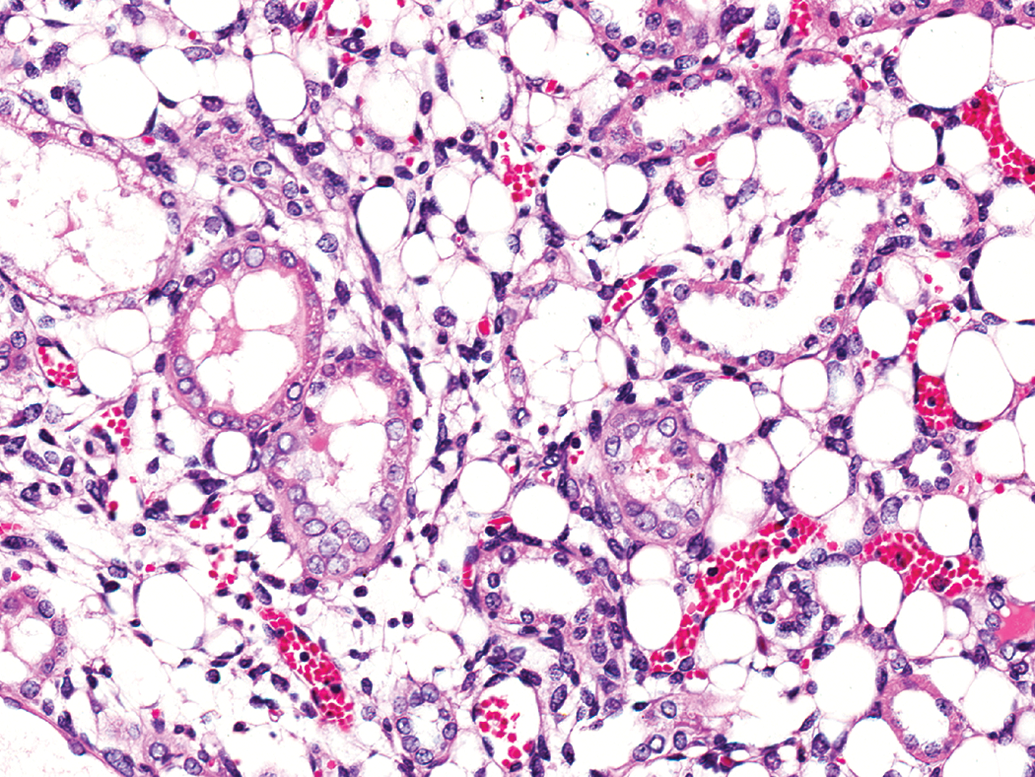
Liposarcoma is characterized, and differentiated from rat renal mesenchymal tumor, by sheets of mature fat cells interspersed with lipoblasts and primitive mesenchymal cells. Entrapped tubules usually show degenerative lining cells. Extravasated red cells and congested small blood vessels are often prominent (hematoxylin and eosin).
There was an apparent sex predisposition, with 13 males and 1 female having liposarcoma among the F344 rats and 4 males versus 1 female among the Sprague-Dawley rats. A third female rat with liposarcoma occurred in the single study using Osborne–Mendel rats, a strain considered to be predisposed to developing lipomas and liposarcomas (Goodman et al. 1980). The mean survival time for liposarcoma-bearing rats was 682 days, and this tumor was considered in the NTP records as being the cause of death in only a few animals.
Hemangiomatous Tumors
There was 1 hemangioma and 7 hemangiosarcomas, which are listed in Supplemental Table S4. The hemangioma was of a cavernous type. The hemangiosarcomas were characterized by proliferation solely of endothelial-like tissue enclosing vascular spaces with scattered red cells (Figure 4), and no other mesenchymal elements suggestive of RMT. The mean survival time for rats with hemangiosarcomas was 661 days. Males and females were equally disposed to bearing this tumor type, with 3 males and 4 females affected. In only 1 case was hemangiosarcoma recorded by NTP as the cause of death.
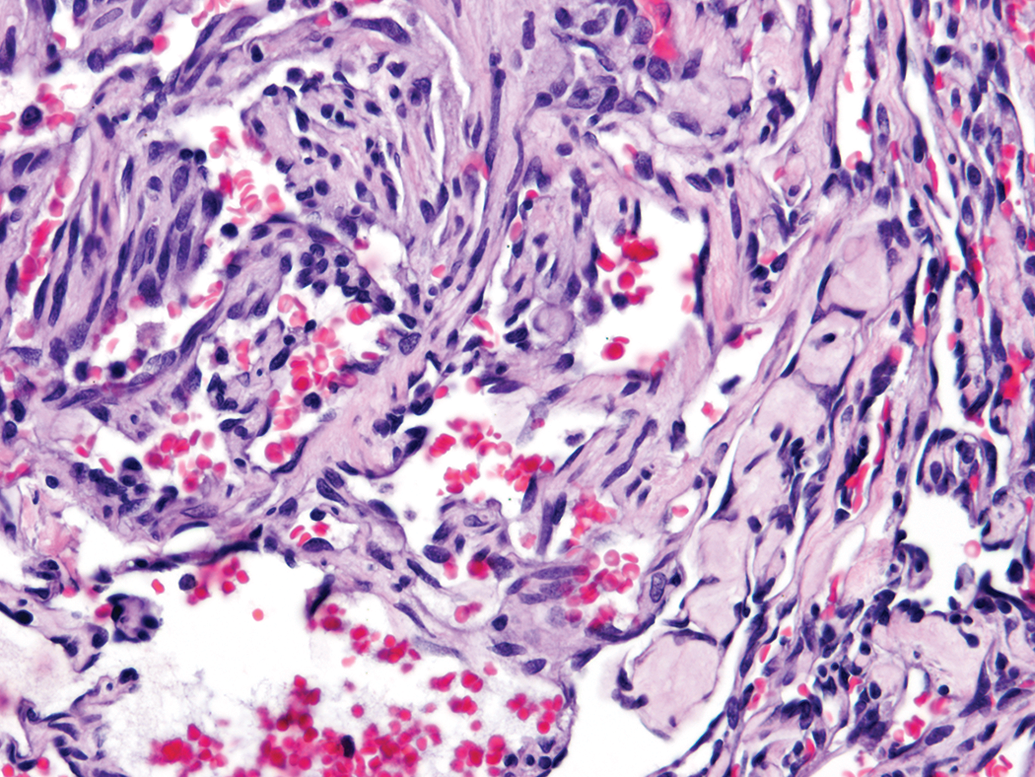
Renal hemangiosarcoma consists uniformly of proliferating endothelium-like cells enclosing vascular spaces. The lack of cellular heterogeneity differentiates it from rat renal mesenchymal tumor (hematoxylin and eosin).
Nephroblastoma
The 48 animals with nephroblastoma are listed in Supplemental Table S5. Nephroblastomas were characterized by the presence of dense basophilic aggregates or sheets of blastemal cells (Figure 5A and 5B) and usually some densely basophilic primitive tubules or rosette formation (Figure 5C). Blastemal cells were characterized by a prominent basophilic nucleus and scant cytoplasm, and they were densely crowded together. However, autolysis caused some disaggregation of these blastemal clusters, confounding accurate distinction from RMT. In some tumors, the blastemal cells assumed an alveolar pattern. Blastemal cell aggregates were commonly condensed around a bland, mature duct profile (Figure 5B), which in some cases was continuous with urothelial cell lining, suggesting possible origin of these ducts from preexisting renal pelvis lining. Mitotic figures were prominent in blastema and primitive tubules. One tumor exhibited primitive glomeruloid formation, consisting of invaginations of very small, dense epithelial cells, into small, usually rounded spaces, enclosed by a single layer of squamous cells and supported by an avascular, eosinophilic stalk (Figure 5D). This appearance is reminiscent of a stage in embryonic glomerulus development.
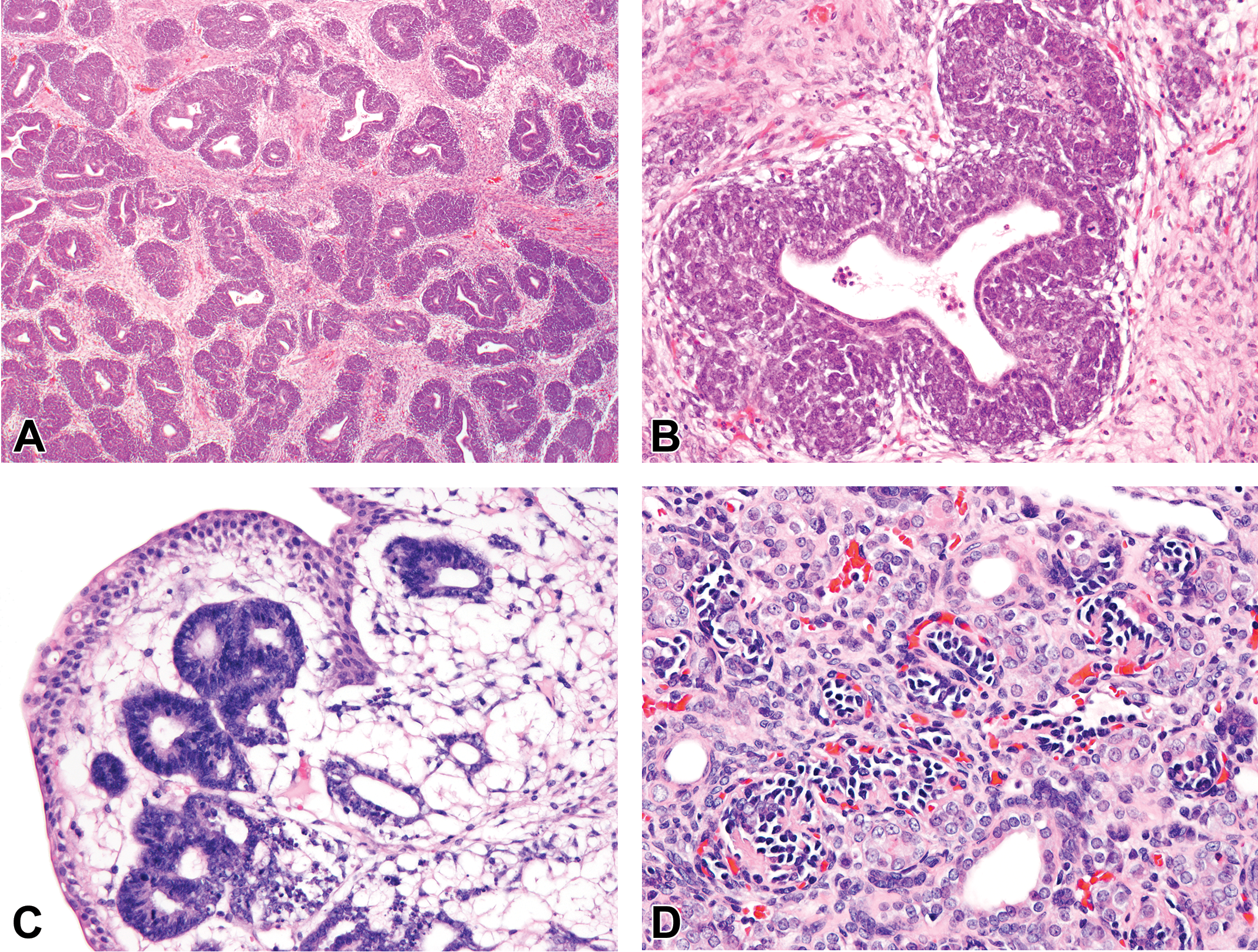
(A) Nephroblastoma in the rat usually consists of dense aggregates of intensely basophilic blastemal cells surrounding mature ducts. The clusters are separated by a mature, banal stroma. (B). Higher magnification of nephroblastoma highlights the arrangement of dense blastema with frequent mitotic figures, surrounding a mature duct. The stroma is mature and morphologically distinct from rat renal mesenchymal tumor (RMT) tissue. (C) Intensely basophilic ill-defined primitive tubule or rosette formation characterizes rat nephroblastoma and is morphologically distinct from preexisting tubules entrapped in RMT. The blastemal cells in this area are arranged in an alveolar pattern. (D) Rat nephroblastoma with formation of glomeruloid bodies consisting of invaginations of very small rounded cells into small spaces lined by flattened cells. The cellular clusters are supported by avascular, eosinophilic stalks (hematoxylin and eosin).
The interstitial stroma of nephroblastomas usually consisted of well-developed bands of mature fibrous tissue dissecting the nephroblastoma tissue (Figures 5A and 5B). The maturity of the fibrous stroma was morphologically distinct from RMT tumor tissue.
Nephroblastoma in this survey was a rapidly growing, lethal tumor, being frequently recorded as the primary cause of death in the NTP pathology records. Only 1 rat with nephroblastoma survived to the 2-year terminal sacrifice, the survival times of the remainder of the rats ranging from 92 to 717 days. The mean survival time of the rats with nephroblastoma, but excluding those from 90-day studies (4 cases) and interim sacrifices (2 cases), was 286 days, much lower than the means for the other kidney tumor types considered here. Metastases from nephroblastoma were observed in 8 cases. In 4 rats, the nephroblastoma metastasized to the lungs, while 3 tumors metastasized to the mesentery, some visceral organs or lymph nodes, and in 1 case, urinary bladder. In 1 rat with nephroblastoma, metastasis had been noted in the renal lymph node only in the NTP pathology pages. A single rat with nephroblastoma occurred in four 90-day toxicity studies, emphasizing the early development and probable embryonal origin of this neoplasm.
Of the studies that used both sexes, there were 18 females and 6 males with nephroblastoma. Thus, nephroblastoma in the NTP series of rats (predominantly F344) showed a sex predisposition with a female to male ratio of 3:1.
In the tetralin study, the primary kidney tumor was confirmed as nephroblastoma, but the lung and other organ metastases (diagnosed as stromal nephroma) were not RMT. They were typical of intracapillary spread of histiocytic sarcoma, in keeping with the NTP diagnosis of fibrous histiocytoma in the liver and skin for that animal.
Statistical Analysis
No statistical trends were found for any of the tumor types versus test article administration. For studies where there were 2 or more tumors in 1 group, Fisher’s exact test between control group and the group with multiple tumors was not significant. Where studies with 3 or 4 dose levels were combined, the Cochran–Armitage trend test and the individual Fisher’s exact tests were not significant for any of the tumor types.
Discussion
The NTP Archives constitutes one of the largest collections of laboratory animal histology specimens from toxicity and carcinogenesis safety evaluation studies in the world, enabling critical retrospective investigation of issues in toxicologic pathology. The large number of primary kidney tumor specimens (165) examined in this survey enabled comment on tumor identity, aspects of tumor biology, gender predisposition, and spontaneous versus treatment-related occurrence. The mesenchyme-related, nonepithelial tumor types of rat kidney found in this survey were RMT, renal sarcoma, renal liposarcoma, and renal hemangiomatous tumors. Although an epithelial tumor, nephroblastoma was included in the survey because of frequent confusion with RMT in the general scientific literature (Hadjiolov 1968; Sadrudin, Lewin, and Cruse 1985; Mitsumori et al. 2002).
RMT was the most common of the primary mesenchyme-related tumors occurring in the rat kidney and, in this survey, was more common than nephroblastoma. In the majority of studies, RMT occurred as a single tumor in a single-dose group. In the studies with more than 1 RMT, the tumors were not restricted to the higher doses of the test article but represented random distribution with no statistical association of RMT with test article exposure.
RMTs induced by genotoxic carcinogens can be very invasive and malignant and demonstrate a complex range of mesenchymal cell types, occasionally including hemangiomatous tissue, rhabdoid elements, islands of cartilage, or osteoid tissue. However, RMT usually consists mainly of spindle and stellate fibroblast-like cells, prominent and abnormal collagen deposition, and smooth muscle cells (Hard 1998). Depending on the predominant cell type, the histological range can include fibroma-like tumor of benign appearance, fibrosarcoma, hemangiosarcoma, or rhabdomyosarcoma (Hard and Butler 1970a, 1971b). Although it was anticipated that spontaneous RMT occurring in repeated exposure 2-year studies with epigenetic carcinogens would present with a more benign appearing morphology than the invasive RMT associated with genotoxic chemicals, such as single-dose DMN (Hard and Butler 1970a), this was not the case. In the NTP 2-year studies, RMTs showed the same heterogeneous range of neoplastic tissue types, large size, and metastatic capability as did DMN-induced RMTs (Hard and Butler 1970a, 1971b). Therefore, this tumor type should be regarded as potentially malignant.
Renal sarcoma is included as an entity in the STP papers on standardized nomenclature in toxicologic pathology (Hard et al. 1995) and in the most recent INHAND document on proliferative and nonproliferative lesions of the rat kidney (Frazier et al. 2012). Nevertheless, it has not been encountered very often, and the inclusion of this tumor in the STP papers has been based in part on experimental experience with renal sarcoma induced by systemic injection of polyoma virus into perinatal rats (Georgii, Prechtel, and Zobl 1969). Little is known about this tumor’s biology, true identity, or distinguishing features separating it from RMT. In the NTP Archives, 13 examples of this very rare tumor were diagnosed, and its restriction to the kidney (in all but 1 case), as recorded in the NTP individual animal pathology statements, strongly suggests that it is a primary renal tumor entity that can occur spontaneously. Distinctiveness from RMT is emphasized by its lack of cellular heterogeneity in particular. Thus, it was characterized by a monomorphic population of dense fibroblasts, often with fascicular pattern, and little or no apparent production of collagen. As a distinct feature from RMT, sporadic, engulfed preexisting renal structures were seen only at the periphery of the tumor adjacent to normal parenchyma. These tubules degenerated rapidly and were not present within deeper tumor tissue as epithelial remnants. Although it could invade into the adjacent kidney parenchyma, parts of the tumor were often associated with a fibrous capsule, which RMT lacked. The tumors also appeared to be rapidly growing, sometimes with a high mitotic rate, and capable of local invasion into the peritoneal cavity, attaching to some visceral organs.
The tumors diagnosed as liposarcoma were also uncommon renal lesions in rats used in the NTP studies. These showed definite sex predominance with 17 of 20 liposarcomas occurring in males. These were characterized by sheets of lipocytes containing lipoblasts, and other cells along the lipocyte differentiation pathway. Distinction from RMT rested on the presence of sheet-like arrangements of lipocytes as well as scattered lipoblasts. RMT can also contain a few mature lipocytes, but these only occurred in tumors that had breached the renal sinus fat pad, with the fat cells usually forming a tapering trail into the RMT tumor tissue.
Eight tumors occurring in the rat kidney were diagnosed as hemangiomatous tumors, 1 as hemangioma and 7 as hemangiosarcoma. Hemangioma and hemangiosarcoma have not been recognized as specific renal tumor types in rat kidney in the STP publications on nomenclature (Hard et al. 1995; Frazier et al. 2012), mainly because hemangiomatous areas are quite common as part of RMT. However, the 8 tumors examined in this survey appeared to have none of the cellular heterogeneity that characterizes RMT and were entirely hemangiomatous. In each of the affected animals, the lesions were restricted to the kidney, as reported in the NTP individual animal pathology records, and hence were considered to be primary tumors of the kidney.
Nephroblastomas were histologically quite distinct from RMT, being epithelial with a consistent morphology of dense clusters or sheets of blastemal cells. The primitive tubule formation usually present also was quite different from the preexisting remnants of tubules that survived as a feature of RMT, being ill-defined and intensely basophilic. In addition, the interstitial stromal tissue of nephroblastoma had a mature, banal character that was different from the mesenchymal tissue of RMT. Nephroblastomas were more rapidly growing than RMT, leading to early death of affected animals.
Of the studies using female Sprague-Dawley rats, the incidence of nephroblastoma was relatively high, suggesting a familial trait in these rats. The strain of rat used, termed the NCTR CD strain, was derived in 1972 from Sprague-Dawley rats obtained from Charles River Laboratories, and maintained since then as a closed colony in the NCTR breeding facility (NTP 2010). There are several records of closed colonies of rats developing relatively frequent occurrence of heritable nephroblastoma, for example, involving a subline of the Upjohn Sprague-Dawley (Mesfin and Breech 1996), the NB hooded rat (Noble, Hochachka, and King 1975), and an inbred colony of Wistar-derived WAB/Not rats (Middle, Robinson, and Embleton 1981).
Statistical analysis of the tumor data in this survey of NTP kidney mesenchymal neoplasms, and nephroblastomas, confirmed that their distribution among dose groups was random and that none of these tumor entities was associated with test article administration. Along with previous experience, RMT and nephroblastoma appear to be induced only by certain chemicals with genotoxic properties, while there is no credible information to date that sarcoma, liposarcoma, or hemangiosarcomatous tumor of the rat kidney have been induced by chemical agency.
Supplemental Material
Supplemental Material, RMT_Ms_Supplementary_Tables_for_online_only - A Survey of Mesenchyme-related Tumors of the Rat Kidney in the National Toxicology Program Archives, with Particular Reference to Renal Mesenchymal Tumor
Supplemental Material, RMT_Ms_Supplementary_Tables_for_online_only for A Survey of Mesenchyme-related Tumors of the Rat Kidney in the National Toxicology Program Archives, with Particular Reference to Renal Mesenchymal Tumor by Gordon C. Hard, John Curtis Seely and Laura J. Betz in Toxicologic Pathology
Footnotes
Acknowledgments
The authors wish to thank NTP for access to the NTP Archives, and EPL for providing logistical support, both of which enabled the conduct of this histopathological survey. In particular, we acknowledge EPL personnel, namely, Kylie Brockenfelt, who accessed the specimens and related paperwork, and Maureen Puccini and Emily Singletary for photographic assistance.
Author Contributions
Authors contributed to conception or design (GH, JS, LB); data acquisition, analysis, or interpretation (GH, JS, LB); drafting the manuscript (GH); and critically revising the manuscript (GH, JS, JB). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Gordon Hard and John Seely both contributed to the histopathological review and confirmation of lesion diagnoses, while Laura Betz conducted the statistical analysis of the data. All three contributed to the preparation of the manuscript and its revision.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Lyondell Chemical Company, Houston, TX, sponsored this project, with GCH receiving compensation for travel-related expenses and effort involved in conducting the study. Social and Security Systems (SSS), Inc, NC, received compensation from these project funds for LBJ’s time on statistical analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded in part by Lyondell Chemical Company, Houston, TX, USA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
