Abstract
Medical imaging is a rapidly advancing field enabling the repeated, noninvasive assessment of physiological structure and function. These beneficial characteristics can supplement studies in swine by mirroring the clinical functions of detection, diagnosis, and monitoring in humans. In addition, swine may serve as a human surrogate, facilitating the development and comparison of new imaging protocols for translation to humans. This study presents methods for pulmonary imaging developed for monitoring pulmonary disease initiation and progression in a pig exposure model with computed tomography and magnetic resonance imaging. In particular, a focus was placed on systematic processes, including positioning, image acquisition, and structured reporting to monitor longitudinal change. The image-based monitoring procedure was applied to 6 Yucatan miniature pigs. A subset of animals (
Keywords
Introduction
Medical imaging is a rapidly advancing field enabling the repeated, noninvasive assessment of physiologic structure and function. As a clinical imaging modality, computed tomography (CT) has a fast acquisition time, moderate cost, and superior structural resolution while magnetic resonance imaging (MRI) involves no ionizing radiation and has excellent soft tissue contrast. These modalities provide the opportunity to noninvasively characterize and compare progressive changes in pigs that can result from an intervention, exposure, or genetic manipulation. Longitudinal monitoring within the same animal can provide valuable information about the etiology of disease while also keeping the number of animals needed for a study to a minimum.
Animal models have been relied upon in medical research to advance biomedical and translational research for humans. Specifically, pig models can serve as a valuable human surrogate in imaging studies due to their similar anatomy, physiology, life span, and size to humans (Swindle et al. 2012; Svendsen 2006). This allows for testing and validation of novel imaging methods on clinical imaging systems with direct comparison between multiple modalities, a feat that is challenging to accomplish in humans. For example, the pig lung anatomy is comparable to human structure, with similar lung volumes, airway dimensions, and cardiopulmonary rates, facilitating investigations of lung function (Kotoulas et al. 2014), novel bronchoscopy techniques (Henne et al. 2015; Tan et al. 2012), and novel medical imaging methods (O’Connell et al. 2015). Recently, genetically modified pig models have been developed for cancer and cystic fibrosis in which medical imaging has been used to provide valuable in-vivo insight (J. C. Sieren et al. 2014; Rogers, Stoltz, et al. 2008; Rogers, Abraham, et al. 2008). Focus on longitudinal pulmonary assessment is of interest for these genetically modified models as well as for toxicological exposure models in pigs.
In this article, we outline the methods used for multimodality, longitudinal image acquisition in a pig model of silicosis. Silicosis is a pneumoconiosis caused by crystalline silica particles in the lung. In humans, inhaled crystalline silica causes pulmonary inflammation, progressive fibrosis, airway wall thickening, and nodules. These conditions cause structural changes in the lung that can be observed through medical imaging (Matsumoto et al. 1998; Mosiewicz et al. 2004). We apply and evaluate the ability of these imaging techniques to track disease development in a pig model of silicosis, presenting a systematic approach for comparing the sensitivity of CT to MR for pulmonary assessment.
Material and Method
Model Development
Six female Yucatan miniature swine were imaged for a total of 6 screening time points beginning at 1 year of age, as displayed in Table 1. All animals were heterozygotes with genetic modification of a common tumor suppressor gene (
Subjects and Screening Time Points.

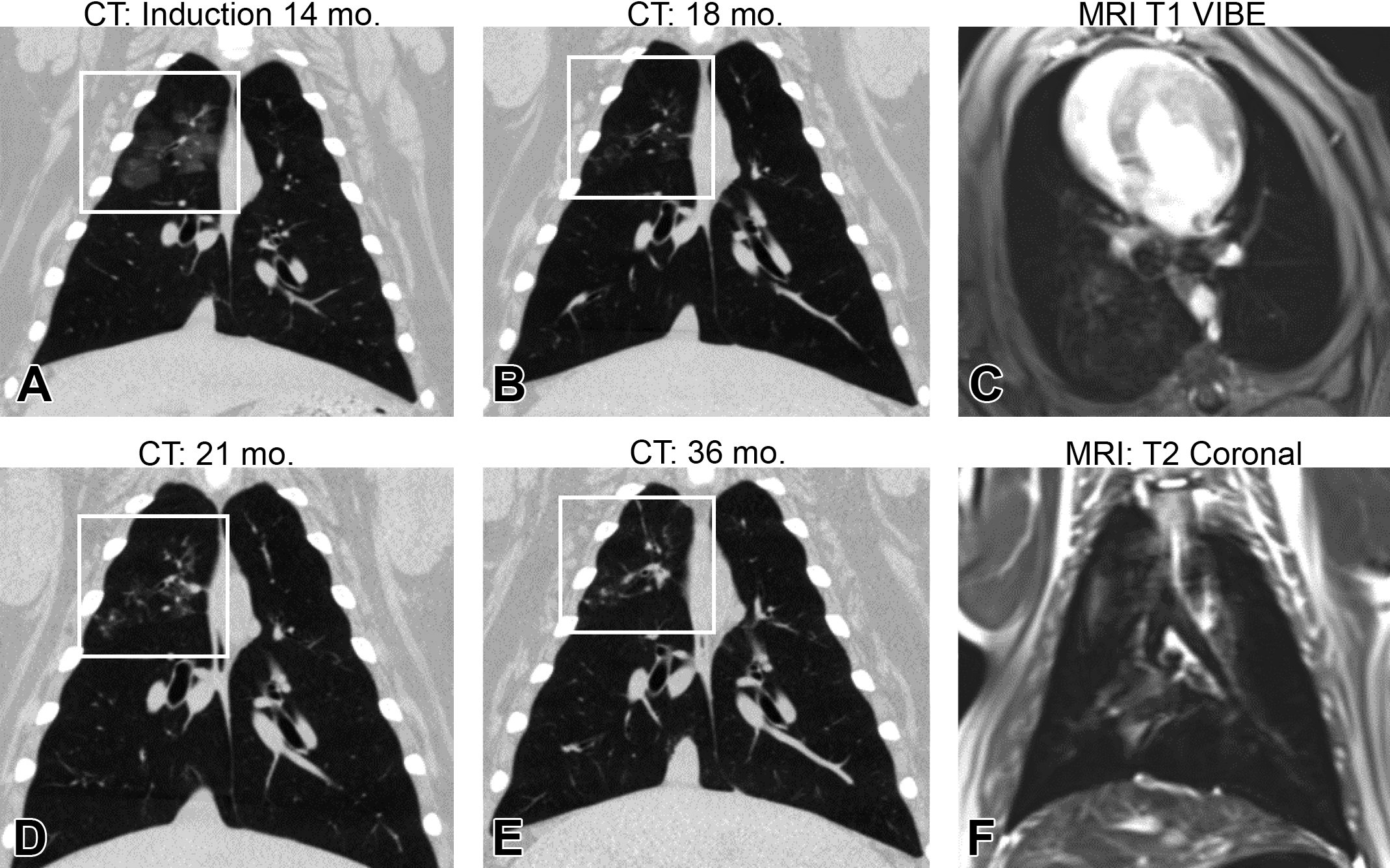
Longitudinal development of disease and lung images. The progression of disease, highlighted by the white box, in subject 4 from (A) deposition of silica at 14 months and disease progression at (B) 16 months, (C) 21 months, and (D) 36 months of age. Example magnetic resonance imaging images from subject 3 at 18 months (E) postcontrast T1-weighted scan VIBE, and (F) turbo spin echo T2-weighted scan.
Animal Preparation
All procedures were performed under anesthesia induced with an intramuscular injection mixture of telazol (2.2 mg/kg), ketamine (1.1 mg/kg), and xylazine (1.1 mg/kg) and maintained with 3–5% isoflurane. All swine were mechanically ventilated through tracheal intubation with a balloon cuffed, 7–8 mm diameter endotracheal tube. Ventilation was performed with a 5 cm H2O positive end expiratory pressure (PEEP) and an approximate tidal volume of 10 ml/kg using an MRI compatible, large animal anesthesia machine (DRE Premier XP, Louisville, KY). Enforced breath holds were used to prevent respiratory motion through short imaging acquisition protocols achieved by suspending mechanical ventilation with a 20 cm H2O PEEP bidirectional flow guard (ACCU-PEEP; Vital Signs, Nashville, TN). The ventilation protocol used a respiratory rate of 10–16 breaths/min to maintain an end-tidal carbon dioxide pressure between 35 and 45 mmHg and a blood oxygen saturation pressure of 100%. Peripheral intravenous access was obtained via ear vein cannula (20 Gauge) for the administration of contrast and was maintained with heparinized saline (3–5 ml, 500 units/L) flushes.
Animals were placed supine in a positioning unit designed to maintain reproducible positioning during imaging (Hammond et al. 2015). Physiologic monitoring was maintained through the duration of each animal imaging study, in CT with a Phillips physiologic monitor (IntelliVue MP90; Philips Healthcare, Best, the Netherlands), in transit with a portable physiologic monitor (Propaq Encore; Welch Allyn Inc., Skaneateles Falls, NY), and in MRI with an MRI-compatible physiologic monitor (Invivo MAGNITUDE:3150M; Medeco, Boise, ID).
Imaging Protocols
The strengths and weaknesses of CT and MRI for longitudinal lung disease assessment were evaluated in this model. CT was chosen for fast acquisition, high resolution, and quantitative imaging capabilities. Although MRI is not typically used clinically for pulmonary imaging, it was selected in this study as an alternative modality that does not require ionizing radiation, which is advantageous for longitudinal screening.
Chest CT scans were obtained with a 128-multidetector dual-source scanner (Somatom Definition Flash; Siemens Healthcare, Erlangen, Germany) to assess lung density and anatomical detail (210 mAs and 120 kV, a rotation time of 0.5 second, pitch of 1, and a CTDIvol of 14.2 mGy). No resultant effects were observed from this exposure, which is slightly higher than a standard clinical high-resolution chest protocol and less than a CT angiography exam. Scans were acquired at an inspiratory breath hold of 20 cm H2O following 2 min of pulmonary recruitment at an equivalent PEEP. Mouth pressure was monitored with in-house LabVIEW software (National Instruments, Austin, TX) to confirm consistent inspiratory breath holds during imaging. Reconstructions were performed with a B35f Siemens kernel at slice thicknesses of 3 mm and 0.75 mm with 0.75 mm isotropic in-plane resolutions.
Chest MRI scans were acquired with a 3-Tesla MRI system (TIM Trio 3T; Siemens Healthcare) with standard surface coils. Turbo spin echo T2-weighted scans (TR of 5280 ms, TE of 156 ms, flip angle of 120°, and echo train length of 109) acquired in the axial and coronal planes were used to assess areas of increased fluid content, such as tumor development or inflammation. Slice thicknesses of 5 mm were acquired with 1.4 mm × 1.8 mm and 1.5 mm × 2.0 mm in-plane resolution for the axial and coronal scans, respectively. Respiratory gating was accomplished during this MRI sequence by using navigator echoes to eliminate respiratory-induced motion artifacts. An additional postcontrast three-dimensional (3D) single breath hold T1-weighted VIBE (Volume Interpolated Breath-hold Examination) thoracic MRI scan (repetition time [TR] of 4.3 ms, echo time [TE] of 1.92 ms, and flip angle of 12°) was acquired postintravenous administration of 0.2 ml/kg gadolinium (Magnevist; Berlex Inc., Wayne, NJ) for increased anatomical detail in the lungs. In-plane resolutions of 1 mm × 1.8 mm were acquired at slice thicknesses of 3 mm. Breath holds at an inspiratory pressure of 20 cm H2O were used to reduce motion artifact for all VIBE scans.
Data Analysis
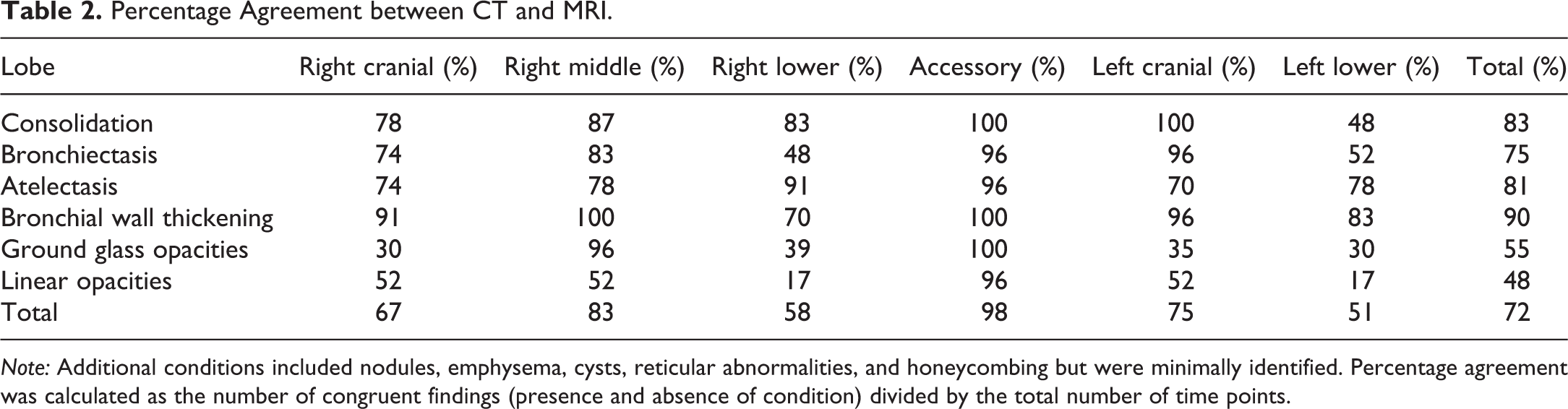
The completion of each screening time point using the above imaging protocol resulted in 1 CT scan and 3 MRI scans per subject. Comprehensive structured reports were developed for each modality, modeled after the Radiological Society of North America radiology reporting initiative (Kahn et al. 2009), to provide consistent and complete visual interpretation of the lungs and surrounding anatomies. Each structured report included the assessment of the diagnostic quality of the scan and targeted anatomic structures of interest. The anatomic structures of interest included the large airways, pleura, heart and pericardium, mediastinum and hila, chest wall and lower neck, vessels, and bones. Within the lungs, a total of 11 conditions were prompted for within each lobe of the lungs. These included consolidation, bronchiectasis, atelectasis, bronchial wall thickening, ground glass opacities, linear opacities, nodules, emphysema, cysts, reticular abnormalities, and honeycombing. A subset of screening time points were analyzed using the structured reports to assess the agreement between the 2 imaging modalities, with CT considered the gold standard for lung imaging. To minimize reader bias, scans were randomized by subject and time point within each modality resulting in pseudo time points read in 1 week intervals by a board-certified chest radiologist (author J. D. N.). Structured reports were defined for each modality, subject, and screening time point and were matched with the corresponding randomized set. The T2-weighted thoracic images and the T1-weighted thoracic images were read in combination for the MRI structured reporting results. For each detected condition within a lobe, the number of incidences where both CT and MRI resulted in a congruent finding (presence and absence of the condition) divided by the total number of imaging time points was used to calculate the percentage agreement.
Quantitative analysis of the CT data focused on the right cranial lobe of the lung to assess disease progression in the airways and surrounding lung parenchyma related to the silica exposure. Parenchymal analysis was performed with in-house software (Pulmonary Analysis Software Suite [PASS]; Guo et al. 2008). A 3D region of interest was placed in the right cranial lobe for each CT data set, and the mean CT density in that region was calculated (Figure 2A). To account for the variation in lung volume across and within subjects, density measures were normalized to the mean density of the entire lung. The apical bronchus and corresponding airway tree were automatically segmented from the chest CT scan using the Apollo quantitative lung imaging software (VIDA Diagnostics, Coralville, IA, Version 2.0.3.16; Figure 2B). The wall area fraction (wall area as a percentage of the total airway area) was measured and used to assess change in the airways between the exposure and control cohorts.
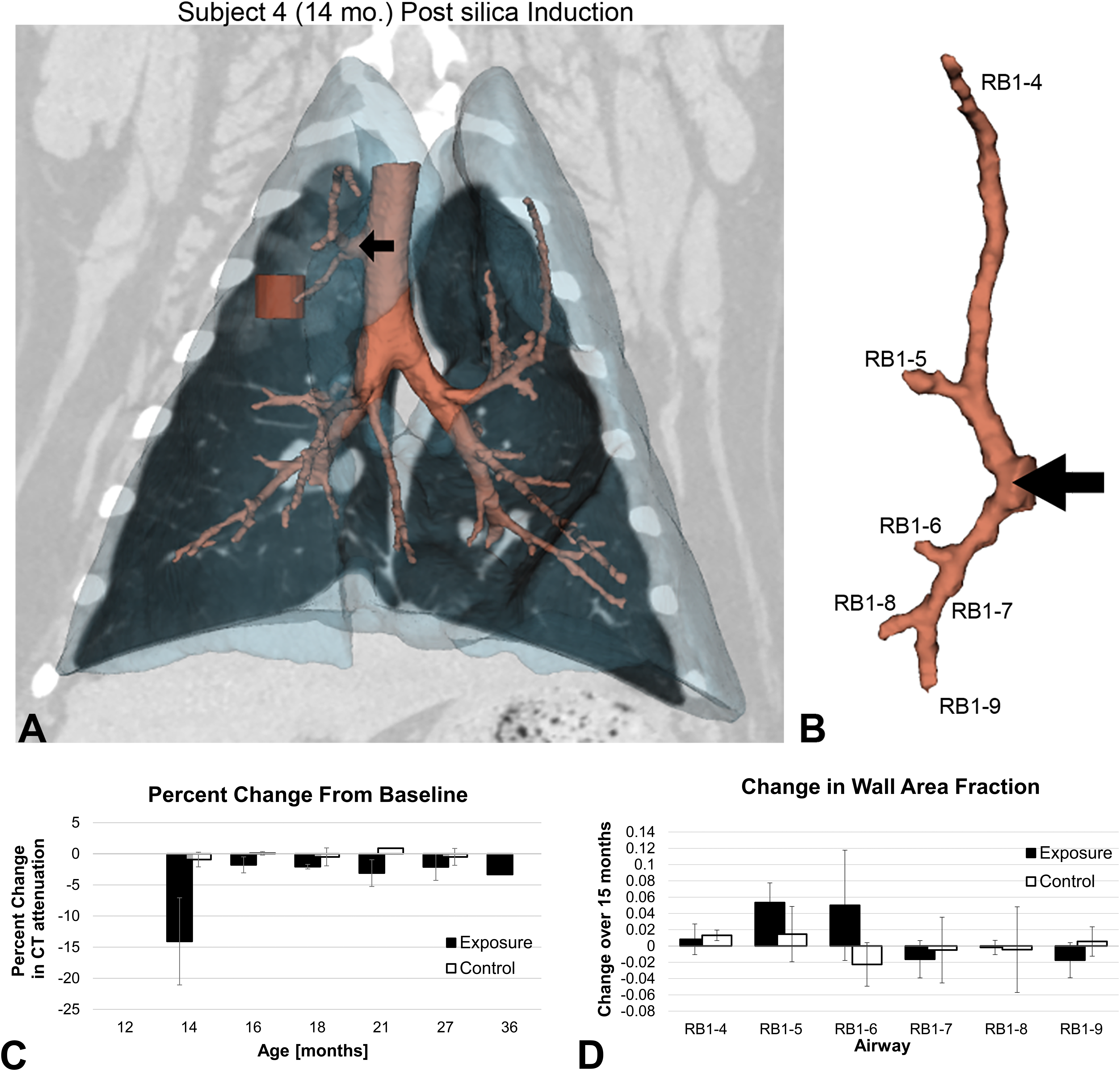
Silica deposition and corresponding quantitative measures. (A) Volumetric rendering of the lungs (translucent blue) with the corresponding airway tree (peach) from subject 4. The red cylinder within the upper lung indicates the location of the volumetric region of interest (ROI) used for computed tomography (CT) attenuation analysis. The corresponding CT coronal section shows the deposition of silica as noted by the increased attenuation in the surrounding area. (B) The apical bronchus airway tree and the labeling system used to identify each branch. The black arrow shows the site of silica deposition with the bronchoscope corresponding to the black arrow in (A). (C) Percentage change from baseline (at 12 months of age) of lung attenuation (measured in Hounsfield Units [HU]) in the ROI for exposure and control animals. Due to the nature of CT in the lungs (negative HU values), an increase in attenuation results in a negative percentage change. (D) Change in wall area fraction in the peripheral airways of the apical bronchus tree. Change is determined as the difference between wall area fraction measures at 15 months postexposure (27 months of age) versus those acquired at baseline (12 months of age).
Results
Screening time points required an average of 2 h (
Structured report results were tabulated and the percentage agreement between modalities was tabulated (Table 2). There was 72% overall agreement in the type and location of pulmonary findings between the 2 modalities. MRI proved useful in the detection of consolidation, bronchiectasis, atelectasis, and bronchial wall thickening with a level of agreement of 75% or above. At only 55% agreement, MRI systematically overestimated ground glass opacities in the lungs, particularly in the cranial and caudal lung lobes (Figure 3). In addition, MRI underestimated linear opacities (48%), mainly due to poor congruency in the lower lobes (17%) when compared to CT. With respect to lobar location in the lungs, the cranial lobes showed higher agreement (67 and 75%) than the lower lobes (58 and 51%), with the right middle (83%) and accessory (98%) lobe showing the greatest agreement; however, minimal findings occurred in the right middle and accessory lobes with both modalities.
Percentage Agreement between CT and MRI.
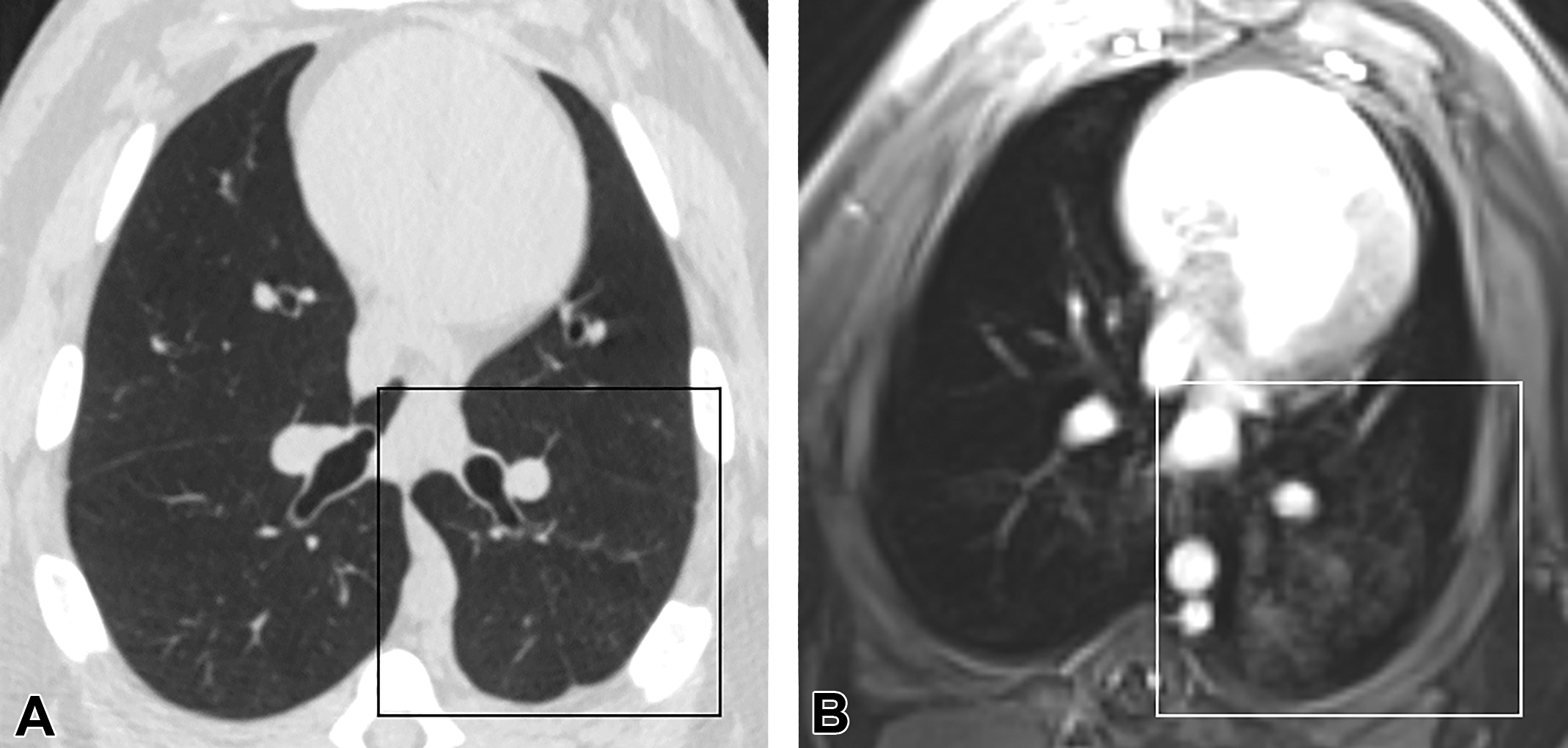
Ground glass artifact in magnetic resonance imaging (MRI) thoracic scan. The high detection of ground glass in the (B) MRI data sets in the upper lung compared to the low detection of ground class in the corresponding (A) computed tomography images indicated the presence of an artifact in the T1 postcontrast images as indicated by the boxes. Image data from subject 6 at 18 months.
The effects of silica instillation on the right cranial lobe in the exposure cohort generated new findings during serial imaging of this cohort, including ground glass opacities and the identification of multiple small nodules (2–3 mm) at 21 months in subject 4 (Figure 1). Via the structured reporting mechanism, the cranial lobes in both exposure and control animals had a high number of reported findings, with multiple minor transient changes reported. Quantitative CT-based density and airway measures were obtained to objectively compare the severity and progression of structural changes in the exposure versus the control cohorts (Figure 2). CT attenuation is expressed in Hounsfield Units (HUs) and is representative of tissue density, with water represented as 0 HU and air as −1,000 HU. Normal mean lung CT attenuation values are reported to be −861 (
Pathological confirmation of silica deposits with corresponding fibrosis in the upper right lung (Figure 1) was obtained for subject 4 at 36 months of age (Figure 4). Necropsy occurred immediately following the final imaging time point with CT. Prosection of the lung was guided by the previously acquired MRI and CT images through 3 plane viewing and volume rendering to gauge correspondence between and within structures. The remaining 2
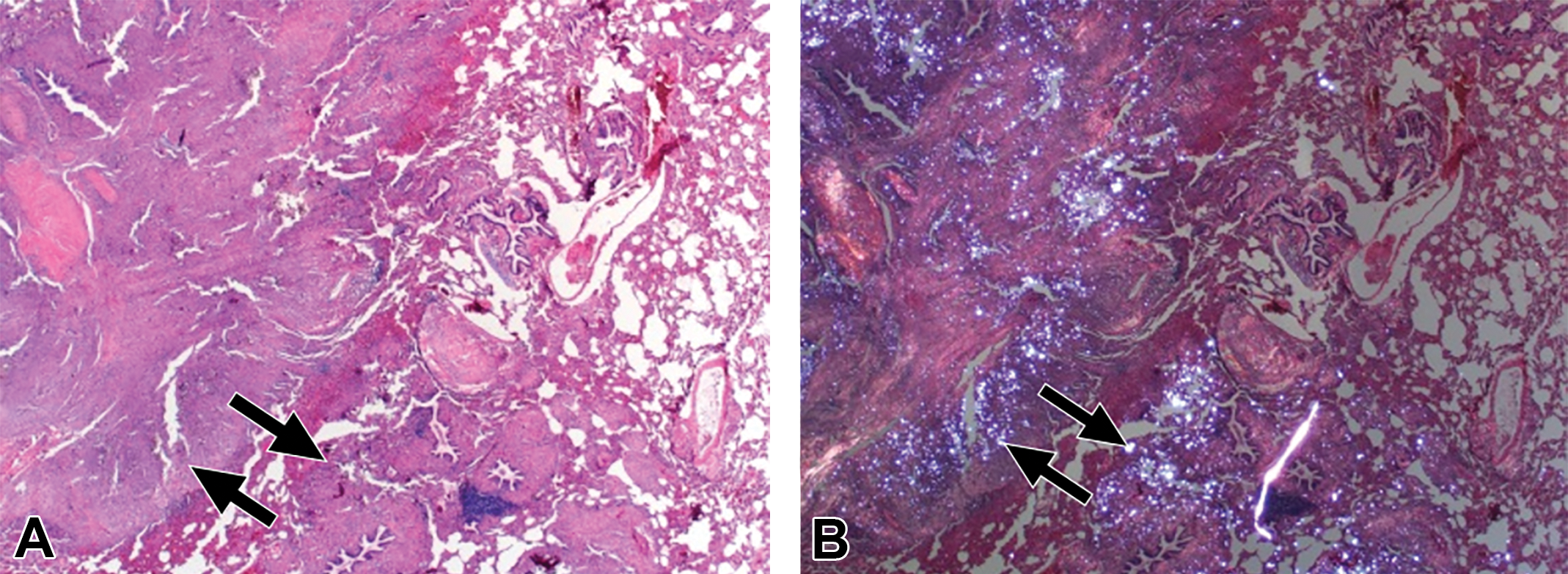
Pathological confirmation of silica deposits and disease development. (A) Histopathological appearance of the lung (from subject 4, see also Figure 1D) showed locally extensive remodeling accentuated by fibrosis (arrows) and inflammation (hematoxylin and eosin [H&E] stain, 20×). (B) Same section as (A) but examined with polarized light. Note that the white (refractile) silica granules (arrows) are admixed within pulmonary remodeling (H&E stain, 20×).
Discussion
This study presents a method for noninvasively characterizing lung disease development in a pig model exposed to crystalline silica using CT and MRI. A pulmonary structured reporting method was developed to record pertinent information useful in the systematic, longitudinal phenotyping of pig models. With respect to imaging modalities, CT was selected for its fast acquisition of high-resolution volumetric data, broad applicability, and wide availability in the clinical imaging field. MRI provided a strong complement with the lack of ionizing radiation making it ideal for longitudinal acquisition. This study shows that pig models can be used for controlled longitudinal studies directly comparing CT and MRI protocols that is not easily accomplished with humans and with direct translatability not found with small animals.
The developed thoracic structured reporting method focused on conditions noted in the lungs and provided a comparison between MRI and CT for lung disease screening using CT as the gold standard. Predictably, CT was superior over MRI in the detection of fine structural details, such as linear opacities. Comparatively, MRI was shown to be reliable in the detection of bronchiectasis, atelectasis, and bronchial wall thickening. However, the results indicated a large disagreement in the incidence of ground glass detected in MRI compared to CT. It was inferred that these ground glass abnormalities observed in the T1 postcontrast VIBE scans were artifacts which may be due to cardiac motion and/or the presence of contrast. In addition, it is possible these were real transient findings related to the fact that the MRI scans were obtained after the CT acquisitions. It follows that the animal had been on the ventilator longer so atelectasis and increased secretions may have been present, specifically in the lower lobes despite lung recruitment maneuvers. We did not acquire ground truth pathology confirmation for the ground glass opacities detected with MRI. While the desired diagnostic quality was still attained in a majority of the scans with the management of respiratory motion, improved quality may occur with the use of cardiac gating and increased frequency of pulmonary recruitment. Due to these factors, routine imaging with CT is preferred for the early structural assessment of silicosis progression in pigs. Later stage disease may be adequately captured using MRI.
An additional advantage of CT data acquisition in this study was the ability to quantitatively assess the lung for density and airway structural changes over time using CT analysis software developed for human pulmonary assessment. The structural reporting mechanism aided in the comparison of findings between modalities and also assisted in distinguishing transient from persistent structural changes within a modality. However, the structured report did not capture the severity of a finding in a particular lobe and an approximately equal number of findings were reported in the right cranial lobes of exposure and control animals. Objective characterization of differences between the cohorts across time was achieved using quantitative CT image analysis of lung density and airway wall structure. Having established these methodologies, the findings support further exploration of these quantitative measures in a larger cohort to validate these biomarkers for the assessment of silicosis in pigs.
Incorporating medical imaging surveillance has proven to be a useful tool for pathological analysis of pig models. Imaging modalities can screen large volumes of tissue more effectively and efficiently than can be done at a typical necropsy. We have previously used whole-body CT and MRI monitoring to identify tumor formation in a Li-Fraumeni
Due to their comparable size, the use of large animals has been previously reported for the development and translation of CT protocols to aid in the quantitative CT characterization of human emphysema (Won et al. 2003; Alford et al. 2010; Dakin et al. 2008). Furthermore, pig models have similar metabolic activity and life span, providing a more robust surrogate for targeted therapies developed in large animal models for subsequent translation to humans (Ellinwood and Clay 2009; Bode et al. 2010). The imaging protocols for this study were chosen to parallel clinical protocols with an emphasis on lung structure assessment. Data acquisition was facilitated by the availability of dedicated research CT and MRI scanners and technologists at our institution. Recent upgrade of our dedicated research CT scanner (Somatom Force; Siemens Healthcare) will allow future studies to incorporate advanced radiation dose reduction methods, including more efficient X-ray detectors and more advanced iterative reconstruction algorithms, while maintaining comparable quality image data (J. P. Sieren et al. 2014; Mobberley et al. 2013). In this study, the high-resolution, quantitative CT data were superior compared to the acquired structural MRI protocols in capturing early evidence of silicosis in a porcine model. It is important to note that our MRI protocols were optimized within the limitations of available MRI scanner (3T vs. 1.5T), coils, and software. Future application of ultrashort TE sequences on our 3T MRI system could further enhance lung structure assessment of MRI (Johnson et al. 2013; Bell et al. 2015). We can now use the developed structured reporting method and comparative analysis to assess performance of these methods. In addition, MRI can be advantageous for lung function assessment when incorporating hyperpolarized and/or inert fluorinated gas MRI, which will be future areas of development in this model (Fain et al. 2007; Couch et al. 2014).
To capture and track longitudinal changes in pulmonary structure due to disease development using medical imaging, we selected to expose a subset of
As sufficient data from this longitudinal project are collected, we will then be able to evaluate our imaging data and correlate this to macroscopic and microscopic pathological changes. The application of imaging to correlate with pathological changes has been described (Tempel-Brami et al. 2015) and has been employed to study small animal models such as mice (Ramot et al. 2014) and large animal models such as pigs (J. C. Sieren et al. 2014; Hariri et al. 2013). The longitudinal imaging and analysis methods presented in this article will be vital in the continued development and exploration of this pig model, including further histopathological confirmation of identified disease.
In conclusion, in this study, we have developed optimized CT and MRI imaging and reporting methods for longitudinal lung studies of the pig silica exposure model. We developed structured reporting approaches for standardizing radiologist interpretation and quantitatively cross comparing CT and MRI modality performance for the detection of pulmonary structural changes indicative of disease. The integration of these methods provides a procedure and valuable multimodality cross comparison for refinement of protocols developed for characterization of other pig models and consistently monitors potential areas of interest for guided biopsy and/or necropsy.
Footnotes
Acknowledgments
We thank Christopher Rogers, Frank Rohret, Judy Rohret, Jason Struzynski, Tony Smith, David Stoltz, Mahmoud Abou, Mark Hoegger, Chelsea Sloan, Melissa Shirk, Elizabeth Allard, Marla Kleingartner, Autumn Craig, Peter Thorne, and Andrea Dodd for technical assistance.
Authors’ Contribution
Authors contributed to conception or design (EH, JN, SD, JS, DT, EAH, and JCS); data acquisition, analysis, or interpretation (EH, JN, SD, NS, JM, DM, and JCS); drafting the manuscript (EH); and critically revising the manuscript (JN, SD, NS, JM, JS, DT, EAH, DM, and JCS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by the NIH (UL1 TR000442, P30 ES005605, and R01HL112986) along with in kind support from Exemplar Genetics.
