Abstract
Amoxicillin used in early childhood may be associated with enamel hypomineralization. Our aim was to assess disturbances of amelogenesis in mice lower incisors induced by chronic administration of amoxicillin/clavulanic acid (AMC). Twenty-eight C57BL/6 male mice, of similar age, randomly divided into a control and 3 treatment groups (n = 7) received subcutaneous injection, once per day, for 60 days: 50, 100, and 150 mg/kg BW of AMC. Scanning electron microscopy/energy dispersive X-ray spectroscopy analysis in AMC treatment groups showed higher content in F and a decrease in P and Ca. Morphology changes ranged from scratched patterns, and small isolated pits-like enamel loss, to generalized demineralized enamel surface, giving a rough, foamy, scaly, or even cracked eggshell appearance to the affected areas. Histological analysis showed disturbances of maturation ameloblasts, which were less organized, with increased amounts of clear vacuoles in the cytoplasm and slightly more elongated and less condensed nucleus. Additionally, they were often detached from the enamel matrix. Transitional ameloblasts formed underlying the cysts of varied sizes. In conclusion, AMC dose-dependently affect ameloblast functions especially in the maturation phase, causing hypomineralized enamel formation with quantitative and/or qualitative defects.
Keywords
Introduction
Dental enamel is the most mineralized tissue of vertebrates and contains 96% anorganic matter, 3% water, and only 1% organic matter. The anorganic content of the enamel is mostly composed of calcium phosphates (90%) consisting of hydroxyapatite (Ca5(PO4)3(OH)) and small parts of fluorapatite (Ca5(PO4)3F; Gardner et al. 1992; Hong et al. 2005; Line and Novaes 2005). Elements such as CO3 −2, Na, Mg, K, Cl, N, and Fe are present in smaller amounts, which can partially replace calcium or hydroxyl ions from hydroxyapatite during the enamel formation (Kumar 2014). The organic matter consists of some residual proteins, mostly amelogenins (90%), and enamelin, ameloblastin, and tuftelin (10%; Laisi et al. 2009). Ameloblasts are the cells responsible for enamel formation (amelogenesis). Amelogenesis consists of secretory, transition, and maturation stages (Hu et al. 2007; Simmer et al. 2010). During the secretory stage, ameloblasts secrete enamel proteins into the enamel matrix. This extracellular matrix undergoes enzymatic modification by enamelysin (matrix metalloproteinase-20 [MMP-20]) and kallikrein 4 (KLK4) in the transition and maturation stages, which facilitates the continued deposition of minerals required for the formation of mature enamel (Hu et al. 2007; Smith et al. 2011; Mohazab et al. 2013). Amelogenesis is genetically controlled, but it is sensitive to several environmental and genetic factors. If these factors act during critical periods of amelogenesis, disturbances in tooth development such as numerical size and shape variations of the teeth, defects in the mineralized dental tissues (enamel, dentin, and cementum), may occur (Suckling, Thurley, and Nelson 1988; Hu et al. 2007; Kuscu et al. 2013). Developmental defects of enamel are classified as hypoplasia (quantitative defects) or hypomineralization (qualitative defects). Hypoplasia occurs if the matrix formation is affected during the secretory stage. Hypomineralization occurs if the maturation stage is lacking, incomplete, or if there exists a disturbance during initial calcification (Kuscu et al. 2013; Sahlberg et al. 2013). One of the clinical forms of enamel hypomineralization is the so-called molar incisor hypomineralization (MIH), which affects most severely the first permanent molars and is usually accompanied by less severe defects in the incisors (R. A. Farah, Monk, et al. 2010; Baroni and Marchionni 2011). Teeth enamel affected by MIH has normal thickness; presents demarcated, asymmetrical opacities of white to yellow-brown coloration; and is soft and porous, which usually induce posteruptive breakdown in enamel (R. Farah, Drummond, et al. 2010). It was established that MIH have multifactorial etiology, which consist of additional or even synergistical interaction between genetic predisposition, environmental factors, systemic condition/insults, and medical status (childhood infectious illness) occurring during mineralization of the first permanent molars and permanent incisors (i.e., prenatally and in the first 3 years of life; Alaluusua 2010). Several retrospective and experimental studies suggested that amoxicillin can be involved in the etiology of MIH, but results were inconclusive, and the need for further controlled studies on test animals was highlighted (Hong et al. 2011; Ciarrocchi et al. 2012; Kumazawa et al. 2012). Amoxicillin/clavulanic acid (AMC) is widely prescribed as a first choice antibiotic agent in pediatrics and pedodontics. Amoxicillin is a semisynthetic wide-spectrum penicillin, with bactericide activity against many gram-positive and gram-negative microorganisms. Clavulanic acid protects amoxicillin from being degraded by β-lactamase enzymes, therefore extending the antibacterial spectrum of amoxicillin to include bacteria normally resistant to amoxicillin, other penicillins, and cephalosporins (Gottberg et al. 2014).
The aims of our study were to assess variations in enamel mineral content and to evaluate changes in outer enamel morphology and histological changes in the ameloblastic layer of mice lower incisors’ enamel induced by chronic administration of AMC in different doses.
Materials and Methods
Drugs and Chemicals
AMC (Amoxiplus®; Antibiotice S.A., Romania) powder for injection or perfusion and sterile water for injections (10 ml vials, Antibiotice S.A., Romania) were purchased from a reputable pharmaceutical company. Each 1.2 g vial of amoxiplus contains 1,000 mg amoxicillin (as sodium salt) and 200 mg clavulanic acid (as potassium salt).
Experimental Animals
The experimental design was fully approved by the Research Ethics Committee of the “Grigore T. Popa” University of Medicine and Pharmacy Iasi, Romania (Reg. No. 15488/30.VII.2013). The 28 C57BL/6 inbred strain adult male mice (8 mice were 8 weeks old and 20 were 10 weeks old) used were purchased from the Baneasa Station, unit of the “Cantacuzino” National Institute in Research and Development in Microbiology and Immunology (Bucharest, Romania). All mice were housed within the Center for the Study and Therapy of Pain, “Grigore T. Popa” University of Medicine and Pharmacy Iasi, Romania. Mice were kept at 24° ± 1°C in boxed caging, under a 12-hr light–dark cycle and allowed ad libitum access to food and distilled water. They were fed with a standardized laboratory rodent pellet diet (18.8% proteins, 2.3% fats, and 6.1% fibers; “Cantacuzino,” Romania). After 2 weeks of acclimatization, mice were randomly divided into 1 control group and 3 treatment groups of 7 mice each (n = 7). Age distribution per group was 5 mice of 10 weeks old and 2 mice of 8 weeks old.
Experimental Study Design
Group 1: Control group received only solvent (0.1 ml sterile water once per day);
Group 2: Treated with 50 mg/kg body weight (BW) of AMC once per day;
Group 3: Treated with 100 mg/kg BW of AMC once per day; and
Group 4: Treated with 150 mg/kg BW of AMC once per day.
AMC was dissolved in sterile water and injected subcutaneously into the loose skin of the posterior neck in a single dose per day for 60 days. In order to provide a proper concentration of amoxicillin or to adjust dosage, every 10 days the mean value of BW was assessed for each group. Maximum injectable volume was set at 0.1 ml.
Mandible and Teeth Sampling
This part of the study took place at the Center for Simulation and Training in Surgery, “Grigore T. Popa” University of Medicine and Pharmacy Iasi, Romania. After 60 days of AMC treatment, the mice were deeply anesthetized with isoflurane using an anesthetic machine (Komesaroff, Medical Developments Australia) and sacrificed by decapitation (using sharp scissors). In order to assess the gross appearance of the incisors, immediately after decapitation, photographs were taken with a D-SLR camera (Canon EOS 700D, F64 STUDIO S.R.L., Romania) along with a macro zoom lens (Canon EF 100 mm f/2.8 mm Macro USM, F64 STUDIO S.R.L., Romania). Afterward, complete bilateral mandibles were harvested and surrounding tissues of the bone partially removed. Lower incisor crowns were sectioned tangential to the gingival margin in a transversal plan, using a diamond cutting disc adapted to dental micromotor (Marathon Multi 600, Megadental GmbH), as shown in Figure 1.
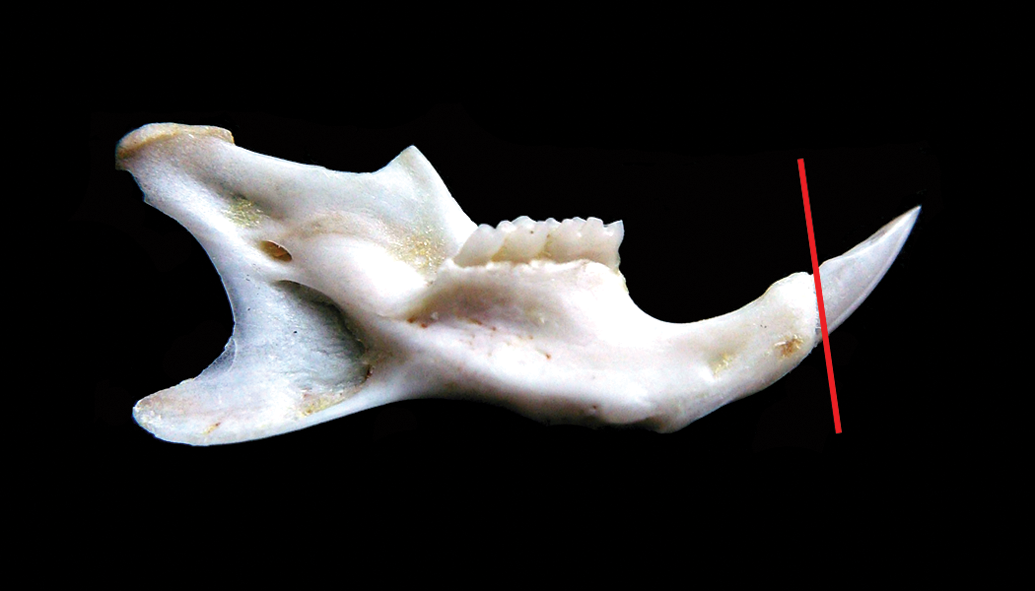
Mandible and teeth sampling. After mandible harvesting and soft tissue removal, crowns of lower incisors were sectioned tangential to the gingival margin in a transversal plane (red line) and used for scanning electron microscopy/energy dispersive X-ray spectroscopy analysis. The remaining mandibular bone (including the roots of the incisors) was used in order to perform histological analysis.
In order to perform SEM (scanning electron microscopy) and EDX (energy dispersive X-ray spectroscopy) analysis, each incisor crown was washed with distilled water, fixed for 24 h in glutaraldehyde 2% and dehydrated in ascending ethanol concentrations (70%, 85%, and 99% for 1 day for each concentration). In order to perform histological analysis, remaining mandibles (including the roots of the incisors) were washed with 0.01% PBS (phosphate-buffered saline) solution and then fixed in 10% neutral-buffered formalin for 48 hr at room temperature.
SEM and EDX Analyses
For both SEM and EDX analyses, all data were collected by assessing the middle third of the incisors enamel. Thus, each lower incisor crown was mounted on an aluminum stub for outer enamel structural defects examination by SEM (FEI Quanta 200, Eindhoven, the Netherlands) operating at 20 kV in low-vacuum mode for secondary electron imaging. The Quanta 200 SEM was equipped with an EDX system for qualitative and quantitative analyses and elemental mapping. To acquire information about elemental content of the enamel, all the obtained spectra across it were analyzed using EDAX Inc. Genesis Spectrum SEM Quanta ZAF Software (version 6.10). Through EDX analysis, only the relative amounts of C, N, O, F, Na, P, Cl, Ca, and Fe were assessed as wt% (percentage by mass); afterward C/O, Ca/P, and F/Fe ratios were calculated.
Histological Analysis
Fixed in 10% buffered formalin, each mandible underwent decalcification process with 10% trichloroacetic acid (TCA) for 4 days at room temperature. After ensuring complete decalcification, in order to remove the excess of the TCA, mandibles were washed in 96% ethanol. Afterward, one hemimandible per mice was embedded in paraffin with a tissue processor Leica TP1020 (Leica Microsystems GmbH, Germany). Parasagittal sections of 5 μm thickness were obtained with a Microtome SLEE CUT 6062 (SLEE Medical GmbH, Germany), deparaffinized, and stained by the Masson’s trichrome technique. The qualitative histology was performed on stained sections using a light microscope Leica DM 750 (Leica Microsystems GmbH) with an attached digital camera Leica ICC50 HD (Leica Microsystems GmbH). The photomicrographs were captured with Leica Application Suit (LAS) Software version 4.2, and the morphology of ameloblasts during the development of mice incisors in each group was examined.
Statistical Analysis
All statistical analyses were performed using SPSS Software version 21.0 (IBM Corp., Chicago, IL). Data were represented as mean ± standard deviation (SD). On the basis of the obtained results from Levene’s test (applied to evaluate variance of homogeneity), we decided to apply Mann–Whitney U test to evaluate data obtained from EDX analysis. Values of p < .05 were considered statistically significant.
Results
Gross Appearance and Morphology of the Incisors
Photographic examining of the lower incisors revealed changes in color and translucency of the enamel between control and AMC treatment groups. The enamel of incisors of mice in the control group was of dark yellow or orange-yellow, smooth, and translucent. In contrast, the enamel of lower incisors of the AMC-treated mice appeared significantly different, with partial loss of translucency and yellow pigmentation, with chalky/whitish enamel in some of the areas. There were no evident visible changes in the morphology of the incisors between groups, just slight roughness of the enamel in the AMC-treated groups (Figure 2). Moreover, there were no differences between groups regarding appearance and morphology of the upper incisors (not shown).
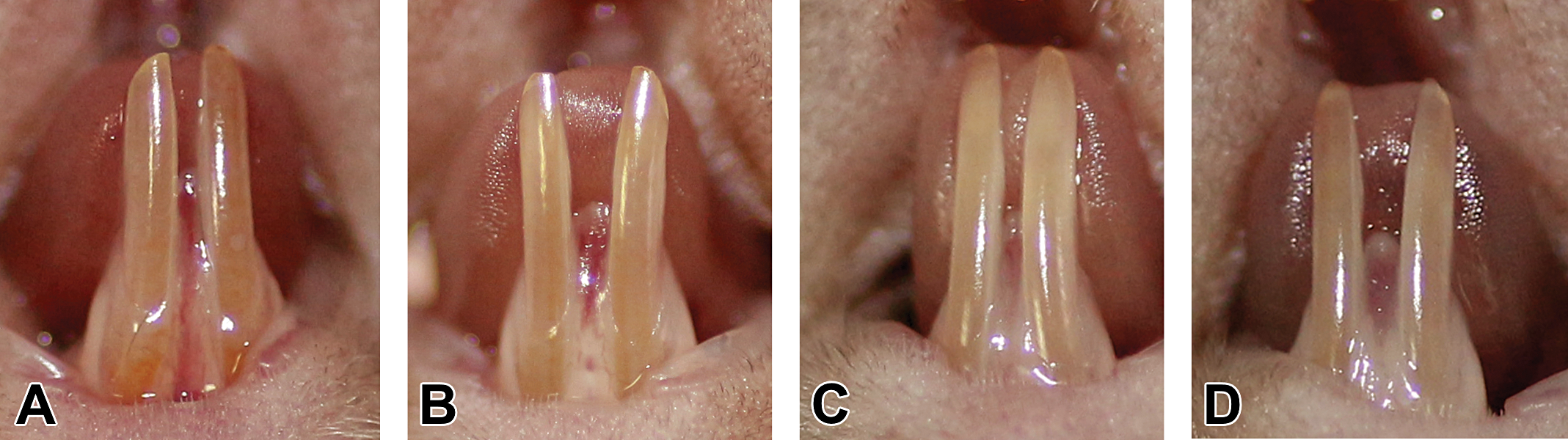
Gross appearance and morphology of the incisors. (A) Macroscopic aspect of lower incisors in the control group was of a dark yellow/orange-yellow color, smooth, and translucent. (B–D) The enamel of lower incisors of amoxicillin/clavulanic acid–treated mice appeared with partial loss of translucency and yellow pigmentation, with chalky/whitish enamel in some of the areas.
SEM Observation and EDX Elemental Analyses
SEM observations of the incisors enamel in the control group showed homogeneous, regular, and smooth surfaces with several scarcely visible point-like pits (Figure 3A, B, and C). In the group treated with 50 mg/kg BW of AMC, we found the following several defects: irregular scratched enamel pattern in all specimens (Figure 3D, E, and F); occasional, isolated enamel loss with almost or perfectly regular margins, with variable size, smooth base, and which extend from outer to inner enamel (Figure 3D); conglomerate of superficial pits in outer enamel with regular margins with variable size and depth, giving a moth-eaten appearance to the affected areas (Figure 3E); and grouped fissures in the outer enamel, with variable size, depth, and irregular margins, which can be accompanied by scratches (Figure 3F). We found the same defects in the group treated with 100 mg/kg BW of AMC, but these were bigger in size, more commonly found, and often combined with each other (Figure 4A and C), and in several specimens we found demineralized areas in outer enamel with exposed enamel prisms (Figure 4B). The most severe microstructural modifications in outer enamel were found in the group treated with 150 mg/kg BW of AMC. In addition to the changes already described we found, all the examined surfaces were demineralized giving a rough, almost foamy (Figure 4D) or scaly (Figure 4F) microstructural appearance to the affected areas. Also, in some specimens we found changes in outer enamel with cracked eggshell appearance (Figure 4E).
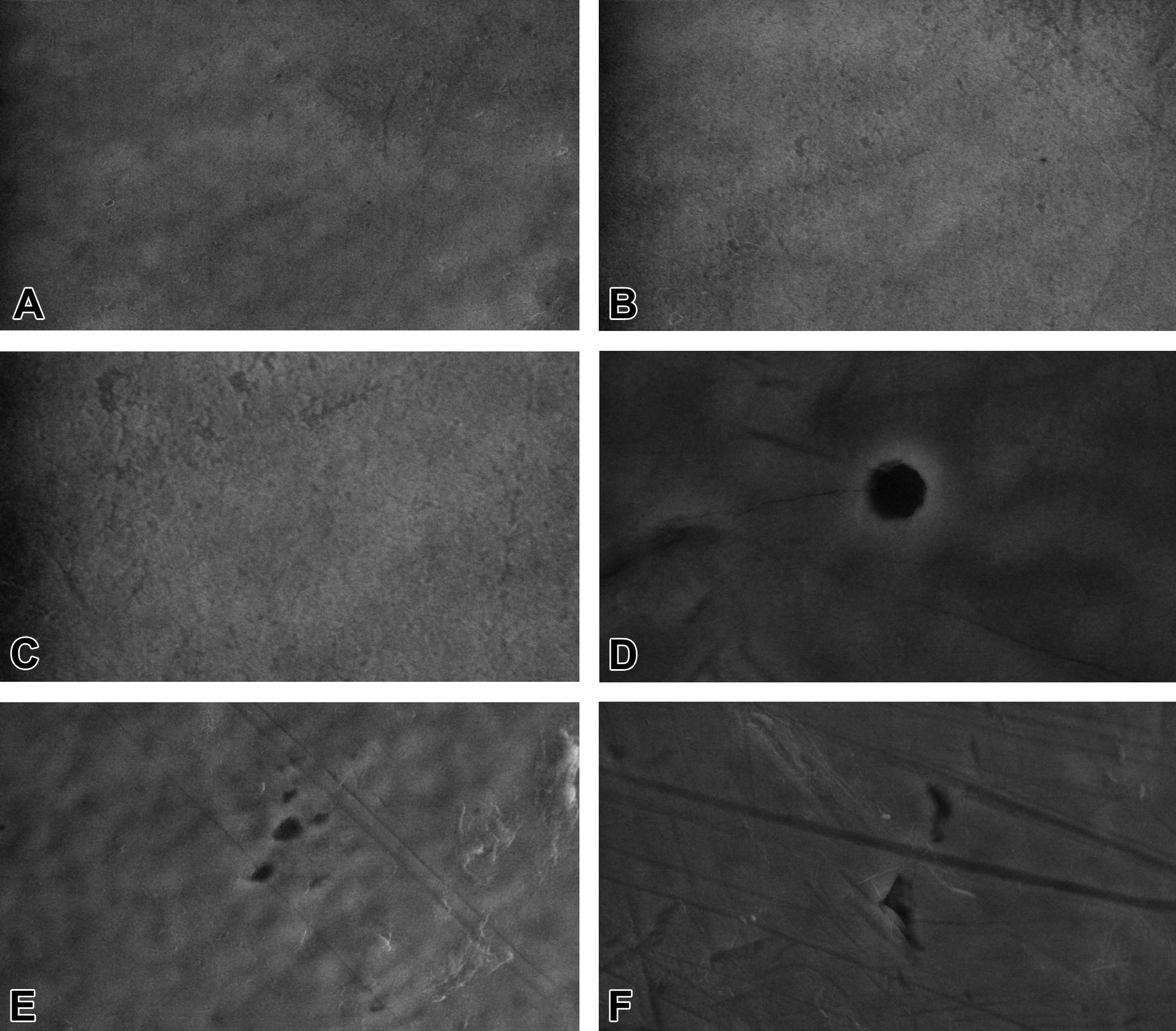
Ultrastructure of lower incisors’ outer enamel by scanning electron microscopy. (A–C) Control group: homogeneous, regular, and smooth surfaces with several scarcely visible point-like pits. (D–F) 50 mg/kg body weight of amoxicillin/clavulanic acid treatment group: irregular scratched pattern in all specimens; isolated enamel loss with perfectly regular margins, smooth base, which extends from outer to inner enamel (D); conglomerate of superficial pits in outer enamel with regular margins with variable size and depth, giving a moth-eaten appearance to the affected areas (E); and grouped fissures in the outer enamel with variable size, depth, and irregular margins (F) (magnification = A: ×1,000; C and D: ×10,000; and B, E, and F: ×5,000).
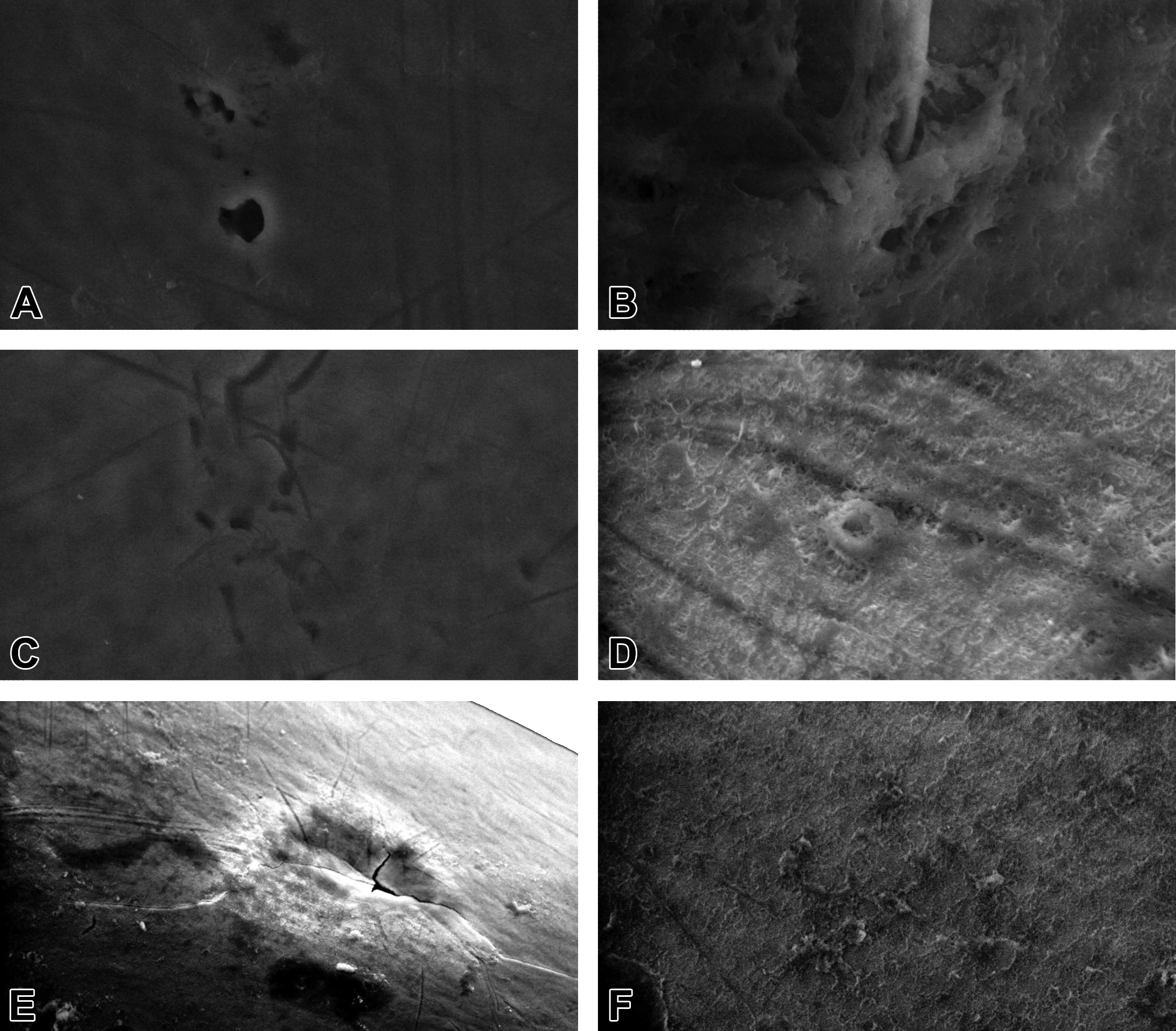
Ultrastructure of lower incisors’ outer enamel by scanning electron microscopy. (A–C) 100 mg/kg body weight (BW) of amoxicillin/clavulanic acid (AMC) treatment group: conglomerate of superficial pits in the outer enamel with almost or perfectly regular margins of variable size and depth (A); demineralized enamel area with exposed prisms (B); several superficial pits with regular margins associated with scratches (C); and (D–F) 150 mg/kg BW of AMC treatment group: all examined surfaces were demineralized resulting in a rough, foamy (D), scaly (F), or even cracked eggshell (E) appearance in the affected areas (magnification = E: ×1,000; A, C, and F: ×5,000; and B and D: ×10,000).
Even though EDX analysis on the enamel in AMC treatment groups showed an increase in mean values of C, N, F, Na, and F/Fe ratio and a decrease in mean values of P, Cl, Ca, Fe, and Ca/P ratio as compared to the control group, Mann–Whitney U test revealed significant differences only for F, P, and Ca (Table 1). In group 2 (50 mg/kg BW of AMC), the values of F were significantly increased as compared to the control group. Furthermore, meaningful differences were found between groups 2 (50 mg/kg BW of AMC) and 3 (100 mg/kg BW of AMC), with increased levels of Ca and P in group 3. The results of EDX analysis were similar between mice of 10 and 8 weeks old.
Comparison between Elemental Content of Control Group Incisors Enamel versus AMC Treatment Groups Incisors Enamel.
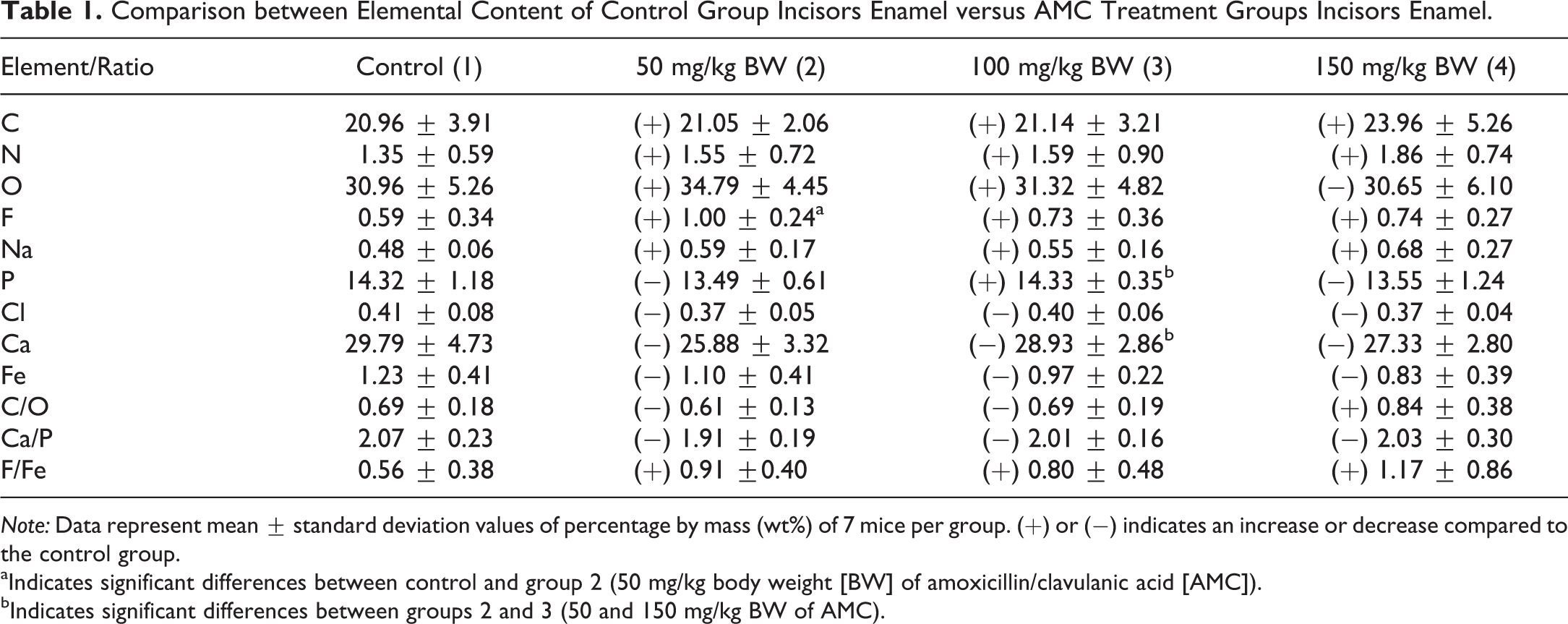
Note: Data represent mean ± standard deviation values of percentage by mass (wt%) of 7 mice per group. (+) or (−) indicates an increase or decrease compared to the control group.
aIndicates significant differences between control and group 2 (50 mg/kg body weight [BW] of amoxicillin/clavulanic acid [AMC]).
bIndicates significant differences between groups 2 and 3 (50 and 150 mg/kg BW of AMC).
Histological Findings
The maturation ameloblasts (MAs) layer, in the AMC treatment groups, was less organized and had increased amounts of clear vacuoles in the cytoplasm. Additionally, the nuclei were slightly elongated and less condensed, especially in the 100 and 150 mg/kg BW of AMC groups as compared to the control one. Also, MAs were often detached from enamel matrix (Figure 5). Moreover, the specimens from these 2 groups showed changes in transitional ameloblasts (TAs), which were disorganized and had formed underlying cyst-like lesions of varied sizes (Figure 6).
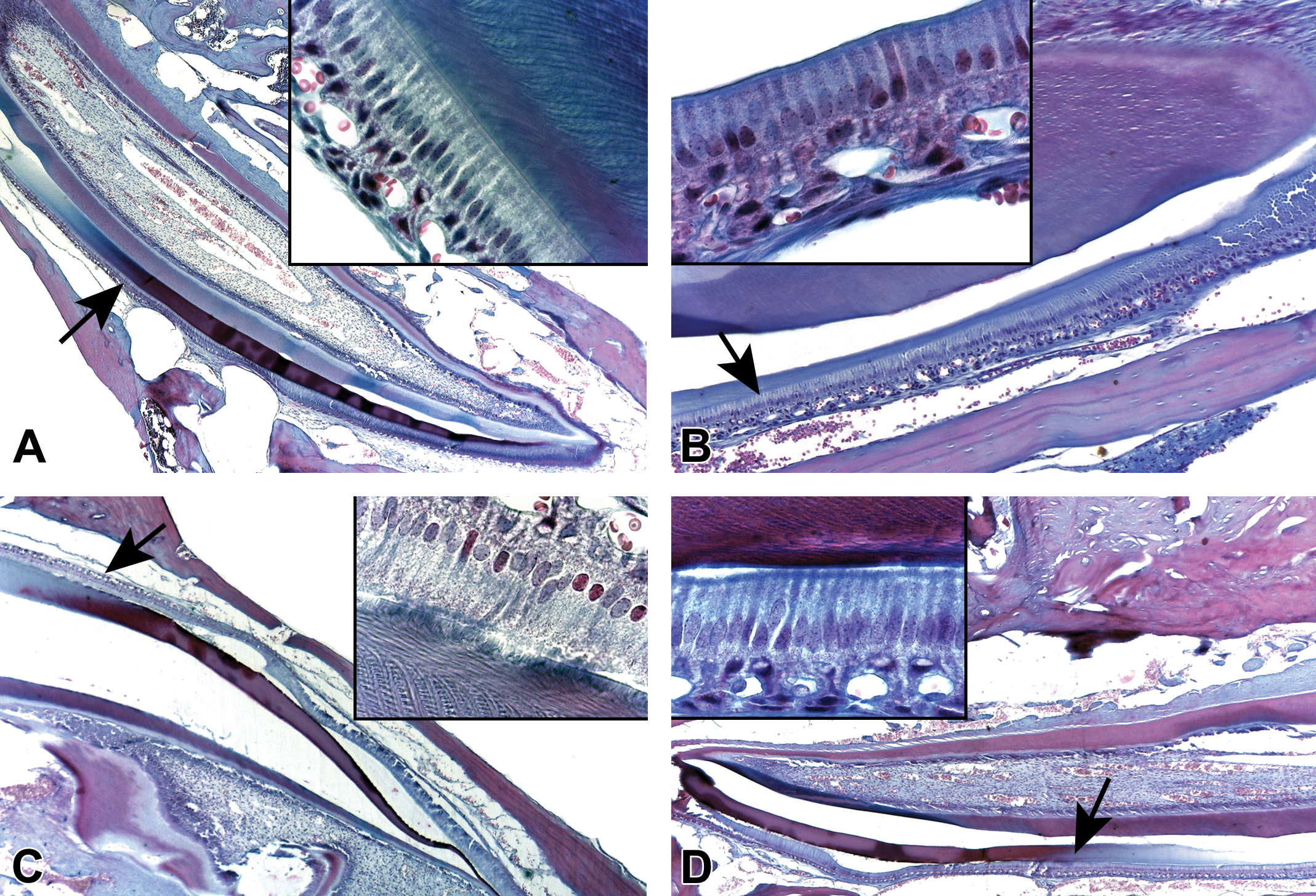
Photomicrographs of Masson’s trichrome-stained sections of mouse incisors (insets: higher magnification of maturation ameloblasts from the areas pointed by arrows). (A) Control group. (B–D) Amoxicillin/clavulanic acid treatment groups. In the treated groups (B–D), maturation ameloblasts’ layer was less organized and had increased amounts of clear vacuoles in the cytoplasm, and the nuclei were slightly more elongated and less condensed (original objective used for main images is ×4 and original objective used for insets is ×100).
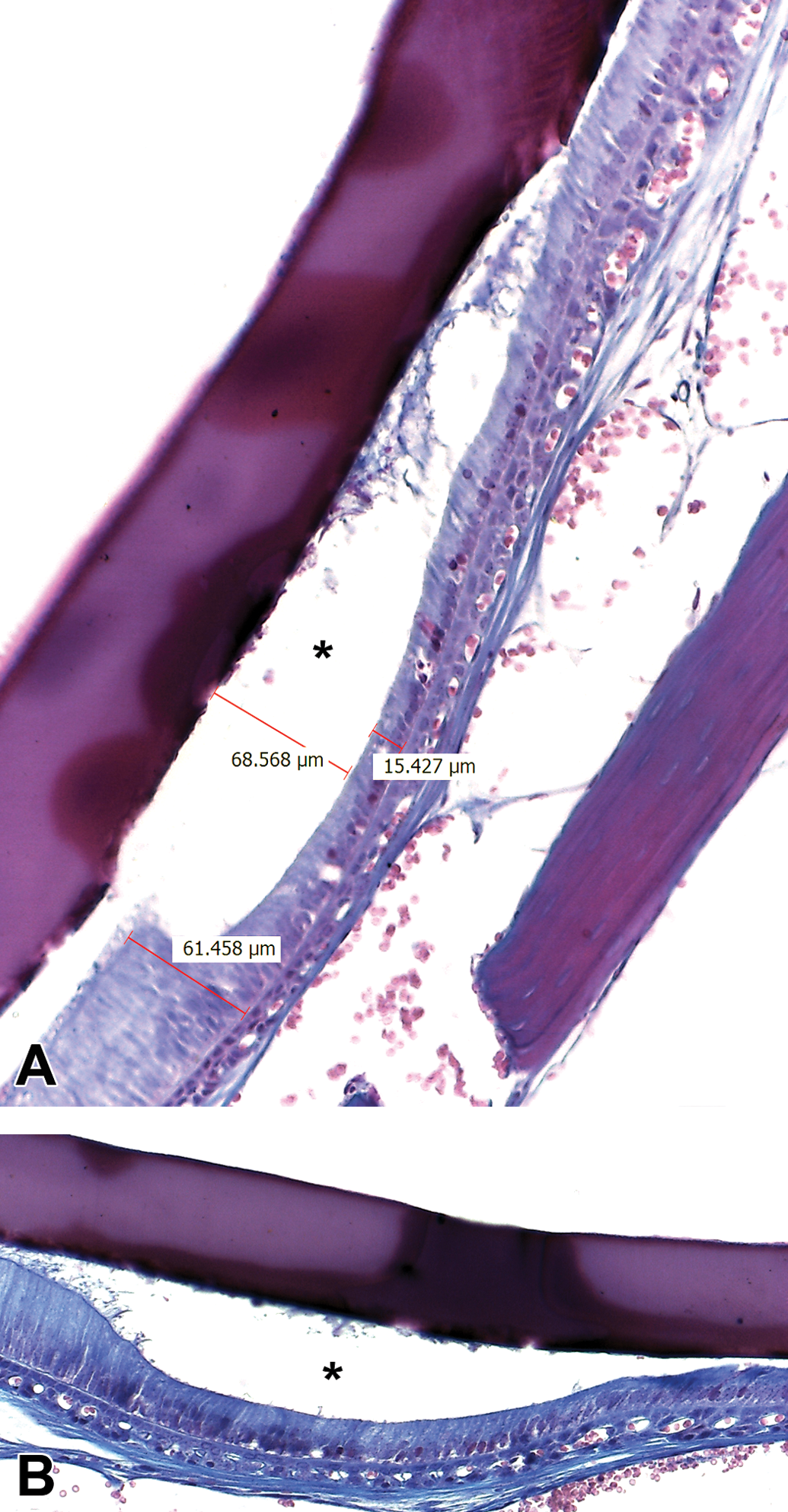
Photomicrographs of Masson’s trichrome-stained sections of mice incisors. (A) 100 and (B) 150 mg/kg BW of AMC treatment groups. Cyst-like lesions (asterisk) with varied dimensions have been formed under transitional ameloblasts (original objective used for both images is ×20).
No obvious morphological changes were detected in secretory ameloblasts (SAs) as compared to the control group. SAs appeared elongated (columnar), polarized, with cell nuclei distant from the basement membrane. Beside this, SAs had a “picket-fence’’ appearance and were clearly aligned Tomes’ processes, which extended and penetrated into the developing organic enamel matrix layer.
Discussion
Recent literature review suggests that the prevalence of MIH varies from 2.4% to 40.2% in different countries (Jälevik 2010). This wide range could be explained by methodological variability by different sociodemographic and ethnical characteristics of the samples. In terms of amoxicillin involvement in the etiology of MIH, some studies found that the use of amoxicillin increases the risk of MIH during the first year of life (Hong et al. 2005; Laisi et al. 2009) or during the first 4 years of life (Whatling and Fearne 2008). Other studies conclude that the MIH prevalence did not differ between children who had received amoxicillin during their first 3 years and those who had not (Alaluusua 2010), even more, was lower (Koch et al. 1987). These types of studies are based on the clinical data about antibiotic administration during prenatal, perinatal, postnatal, or during infancy, which is obtained by questionnaires, interviews, medical records, or medical notebooks. Therefore, the data could be insufficient to draw evident conclusions about the existence of the link between amoxicillin and MIH.
To avoid these inconveniences of retrospective studies, mice were chosen for the study since their teeth have the same development stages as humans do, and incisors are continuously growing (active amelogenesis; Bronckers, Lyaruu, and DenBesten 2009), which facilitates the investigation of amoxicillin effects on tooth enamel development at any time during the animal’s life. Furthermore, for our study we chose to test AMC instead of amoxicillin alone, since this drug combination is one of the most commonly prescribed antibiotics (Fallon 2005). Also, the C57BL/6 inbred strain mice proved sensitivity in developing defects in enamel (like dental fluorosis; Everett 2011). Moreover, C57BL/6 mice has been reported to reach an adult age on an average of 6.8 weeks (Kempermann, Kuhn, and Gage 1997), which means that mice used by us either of 8 or of 10 weeks old were adult, and age distribution should not influence the results of the study. In mice, the normal rate of eruption (which equals the rate of wear, so that the incisors remain a constant size in adult mice) is approximately 2 mm per week for the upper incisors and 2.8 mm per week for the lower incisors. This results in a turnover of the entire tooth in 35 to 50 days (Satoh et al. 2001; The Jackson Laboratory [JAX] 2003). Based on these findings, we decided to administer AMC for 60 days.
In humans, the usual dosage of amoxicillin or AMC is 50 mg/kg BW per day and the maximum adult dose of amoxicillin can be between 2 and 3 g/day (Kuscu et al. 2013). According to these standards, we chose to use 50 mg/kg BW per day of AMC and also decided to use an experimental dosage of 100 and 150 mg/kg BW per day of AMC. The bioavailability of amoxicillin was reported more than 80% in humans but only 44% in rats when administered orally (Joint FAO/WHO Expert Committee on Food Additives [JECFA] 2012). Low bioavailability of the drug in small rodents (mice and rats) was related to presystemic degradation of amoxicillin in the intestine due to the lack of hepatic first-pass metabolism for amoxicillin (Chesa-Jiménez et al. 1994). To avoid these inconveniences, we decided to use subcutaneous injections as route of administration. Beside this, there were reported hepatotoxicity and nephrotoxicity of amoxicillin in usual dosage in humans (Gresser 2001; Salvo et al. 2007). However, according to the JECFA (2012) report, 26-week study of amoxicillin toxicity on small rodents did not reveal any changes that may suggest hepatotoxicity or nephrotoxicity. Furthermore, the no observed adverse effect level for this study was 2,000 mg/kg BW of amoxicillin per day, which is far less than the doses used in our study. Thus, we considered that 50 to 150 mg/kg BW of AMC is safe from the hepatic and renal point of view, according to the abovementioned report.
Based on our knowledge, there are no experimental studies regarding the role of exposure to AMC on the enamel defects. There are only a few case reports which describe extrinsic tooth discolorations related to the administration of AMC in humans (Garcia-López et al. 2001). In our study, photographic examining of the lower incisors revealed intrinsic discoloration (loss of yellowish pigmentation) and translucency changes in AMC treatment groups. Yellowish pigmentation of rodents’ incisors is due to a higher presence of iron in the outer enamel layer and it is a normal phenomenon. Also, it was suggested that iron incorporation in rodents’ enamel serves as a strengthening agent to resist cracking and abrasion (Pugach and Gibson 2014). According to our EDX analysis, AMC administration dose-dependently decreased iron levels in the incisors’ outer enamel (Table 1), which has led to partial loss of yellowish pigmentation (Figure 2). Moreover, increased levels of fluoride in AMC treatment groups could be involved in translucency loss, as was previously reported by Everett (2011).
In our study, SEM was used to observe the ultrastructure of mice lower incisors’ outer enamel, and we found significant differences in all AMC treatment groups. In groups treated with 50 and 100 mg/kg BW of AMC, most of the defects were pits or fissures with variable incidence, size, depth, and usually with regular, smooth margins (Figures 3 and 4). These quantitative defects reveal enamel hypoplasia (dos Santos and Maia 2012), which is inconsistent with characteristics of teeth affected by MIH. Thus, we can suppose that AMC affects ameloblasts during the maturation phase. In moderate/severe forms of the MIH, affected areas can present quantitative defects due to posteruptive breakdown (under masticatory forces). In this case, the margins of the disintegrated areas are irregular (Kuscu et al. 2013), which is opposite to our SEM findings. Only in the group treated with 150 mg/kg BW of AMC, we found signs of hypomineralization not as individual lesions but associated with hypoplastic ones, described previously. Rough and foamy appearance in the affected areas (Figure 4D and F) is a sign of porosity of the enamel surface, which indicates inadequate maturation of enamel (Sahlberg et al. 2013). Cracked eggshell appearance (Figure 4E) indicates hypomineralization within inner enamel associated with breakdowns in outer enamel. The presence of hypoplastic lesions relates with results from several clinical studies, which suggest that amoxicillin may also cause fluorosis-like lesions (Hong et al. 2005; Laisi et al. 2009, Gottberg et al. 2014).
Through EDX analysis (Table 1), we found that all AMC treatment groups tended to show the lower content of Ca and P in enamel, as a sign of hypomineralization. Also, all groups had almost similar Ca/P ratios, which indicates that calcium phosphate was a greater part of enamel inorganic content (Jälevik et al. 2001, Mahoney et al. 2004).
No obvious histological changes were detected in the ameloblasts of the lower incisors in the group treated with 50 mg/kg BW of AMC as compared to the control one. Higher doses of AMC have affected predominantly MAs, and severities of the modifications were dosage dependent (Figure 5). Formation of the cyst-like lesions under the TAs in the 100 and 150 mg/kg BW of AMC groups (Figure 6) can be associated with local enamel hypoplasia seen as pits at the enamel surface of erupted teeth (Lyaruu, Bervoets, and Bronckers 2006).
Nevertheless, our results are opposite, with several studies suggesting that amoxicillin treatment during the first 3 years of life causes dysfunction in SAs (Hog et al. 2005) or that amoxicillin increased enamel thickness in exposed explants with enhancement of ameloblastic function (Laisi et al. 2009). Probably these differences occur due to different study protocols. Furthermore, based on our knowledge there are no comparative studies between amoxicillin and AMC or clavulanic acid alone effects on teeth development. Regarding the abovementioned drugs, we consider that further studies are required in order to fully elucidate the involved mechanisms and their effects on enamel mineralization.
In conclusion, our results denote that chronic administration of AMC through subcutaneous injection in C57BL/6 mice leads to disturbances in the formation of lower incisors’ enamel, mainly as a dysfunction in the maturation and TAs, resulting in hypomineralized enamel, with quantitative and/or qualitative dose-dependent defects.
Footnotes
Acknowledgment
The authors wish to thank the members of the Center for Simulation and Training in Surgery at “Grigore T. Popa” University of Medicine and Pharmacy Iasi for the excellent assistance.
Author Contributions
Authors contributed to conception or design (EM); data acquisition, analysis, or interpretation (EM, LM, AC, NG, AP, and SP); drafting the manuscript (EM, LM, and SP); and critically revising the manuscript (EM, LM, AC, NG, and AP). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received financial support through the “Program of Excellence in multidisciplinary doctoral and postdoctoral research in chronic diseases,” contract no. POSDRU/159/1.5/S/133377, project cofinanced by the European Social Fund Operational Programme “Human Resources Development” for 2007–2013.
