Abstract
This article presents an overview of microscopy and its ability to assist in understanding what happens in cells and tissues. From the 1960s to 1980s, electron microscopy was the best way to understand cell processes, but the advent in the mid-1980s of light microscopy and the ability to do fluorescence imaging displaced electron microscopy in this area. However, the 21st century has seen several improvements in electron microscopy that, along with the need for more detailed ultrastructural information, make it again very attractive in the study of cells, tissues, and organs, and electron microscopy has resumed its place as the preeminent method in understanding cell processes.
Keywords
For those unfamiliar with electron microscopy, some explanation of terminology is in order. An instrument called a
Let us start by examining what is new in the world of TEM. Improvements in computer control have resulted in new software that makes automation of the microscope available to every lab. On the purely mechanical side, this automation allows the microscope to do electron optical system alignments, align the electron source, focus, and in general reduce time to results. This automation also allows for better integration of different types of signals to help make the analytical process much simpler. We can integrate images and signals from many different sources such as x-ray detectors, secondary electron detectors, signals from bright field and dark field detectors, as well as imaging from electron energy loss spectroscopy, all of which can then contribute to a coherent understanding of what is happening inside a tissue. As for applications of the technique, this automation allows rapid and automated examination of things such as very large two-dimensional (2D) maps and the collection of 2D data that will eventually be turned into 3D volumes.
The traditional role for TEM has been the examination of fixed embedded and thin section samples. This fairly simple technique has allowed scientists to monitor the progression of disease and observe the process of viral infections for more than 60 years. Observing morphological changes alone cannot give a full set of answers to today’s complex questions, but the development of new gold labeling of antibodies has helped us better understand specific cellular processes. Also, the ability to incorporate fluorescent proteins into cells and bacteria has helped us better understand many biological processes, and this knowledge has allowed us to take real steps in doing correlated electron microscopy. Correlative microscopy can incorporate any number of techniques, from light microscopy to in vivo imaging, which can help give a much fuller understanding of ongoing biological processes. Using labeled fluorescent light and electron microscopy has allowed us both to see the location of certain molecules and to fully understand how those molecules are transported and stored or moved between cells (Figure 1).
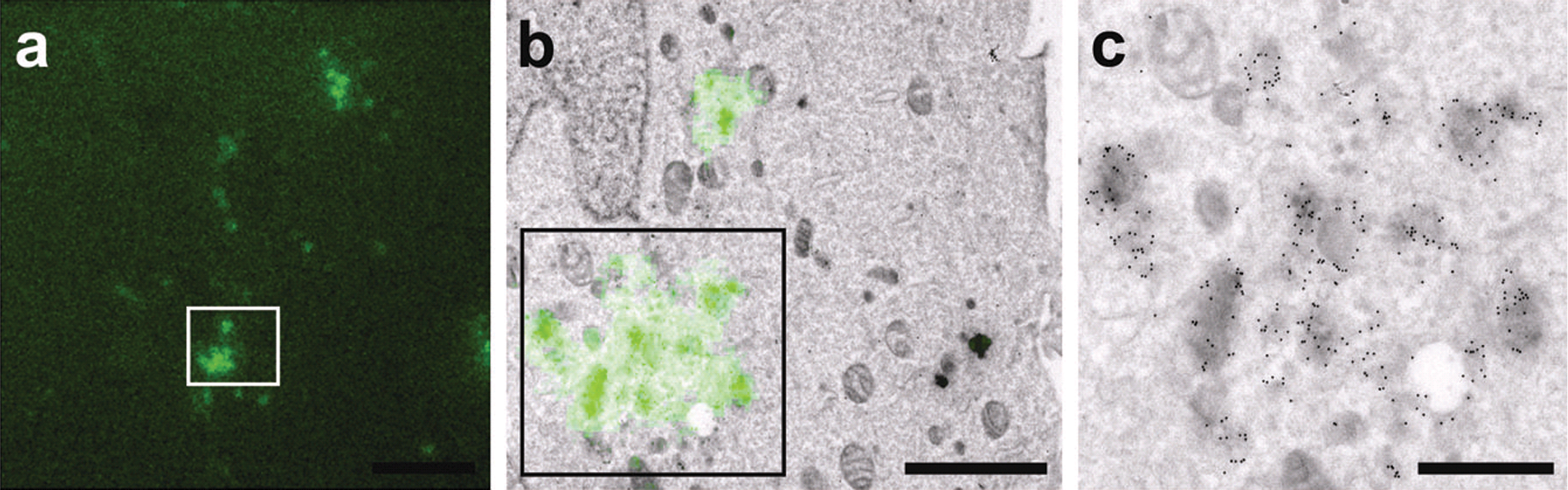
(a) Fluorescence image of cells labeled with a mouse monoclonal anti-LAMP2. (b) Ten-nanometer protein A-gold followed by Alexa488 conjugated goat anti-mouse. (c) Detail of boxed area in (b). (Karreman et al. 2011).
Traditional imaging in the TEM produces only 2D images. But by using a technique called
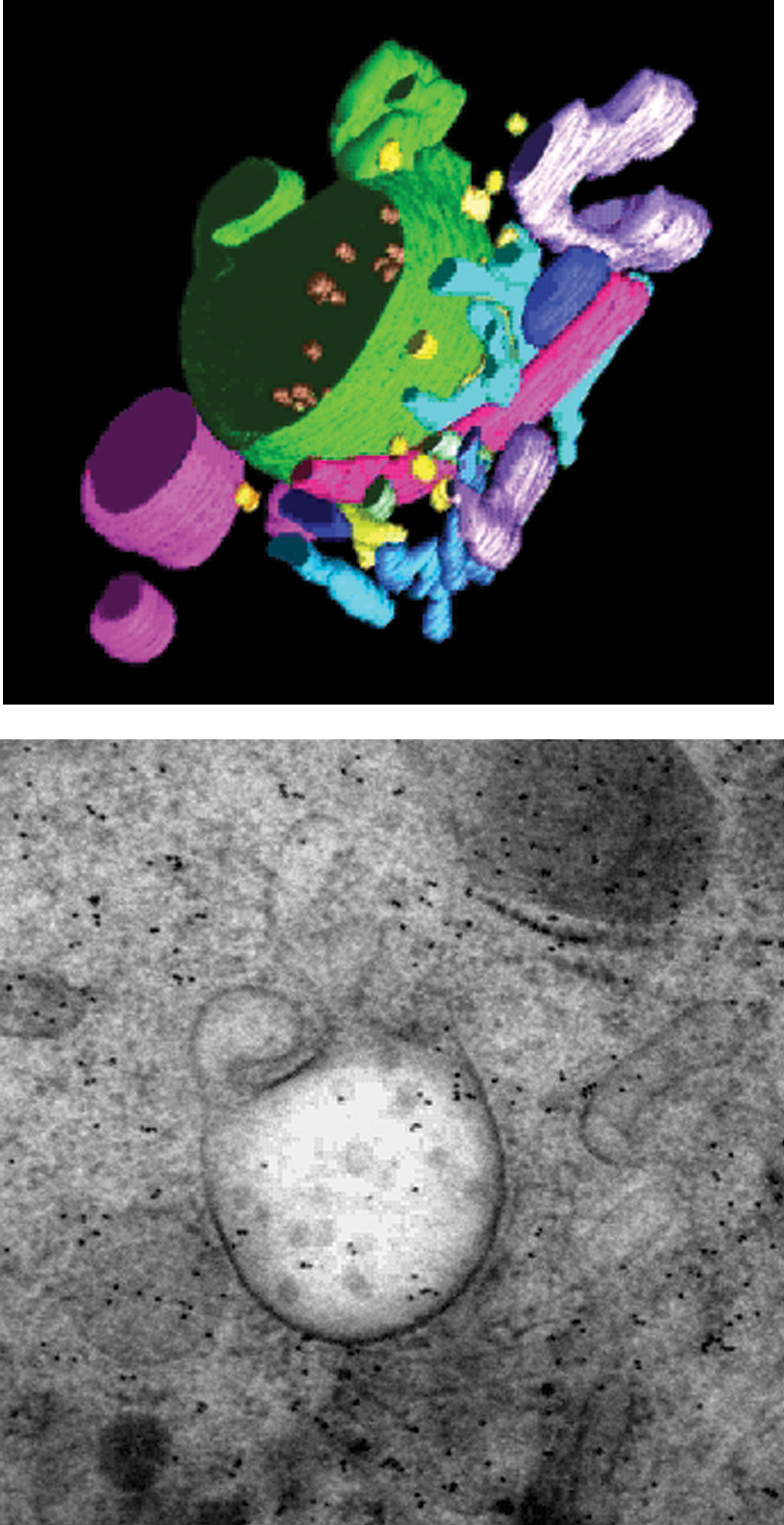
Tomography and segmentation. See supplementary material at http://tpx.sagepub.com/supplemental for Movie 1 depicting segmentation of the multivesicular compartment in a 250-nm-thick section of high-pressure frozen, freeze-substituted, and Epon-embedded mouse dendritic cells. (enhanced online). Multivesicular compartment in a 250-nm-thick section of high-pressure frozen, freeze-substituted, and Epon-embedded mouse dendritic cells. Courtesy of J. L. Murk, B. M. Humbel, U. Ziese, H. J. Geuze, A. J. Koster, and M. J. Kleijmeer.
In the TEM, there is a technique called
See supplementary material at http://tpx.sagepub.com/supplemental for Movie 2 depicting STEM tomography volume of yeast cells (1.0 µm) thick section (enhanced online). Yeast cells (Schizosaccharomyces pombe cells, wild strain). Cytokinesis, the thick (1.0 µm) sectioned image of high-pressure frozen, freeze-substituted cells showed the invaginated cell membrane at the medial region of septum formation. These images of dividing cells clearly showed nuclei, vacuoles, and secretory vesicles in the cytoplasm (Aoyama et al. 2008).
As mentioned earlier, with computer control of the microscope, we have available to us a wide variety of signals in the TEM.

Images from an 80-nm section of a
There have been significant improvements in the application of STEM in instruments that are commonly called ESEM. An ESEM is an environmental scanning microscope, and that refers to the environment that the sample experiences, which is closer to a normal biological environment than the vacuum of deep space, as is found in normal electron microscopes. This technique is called
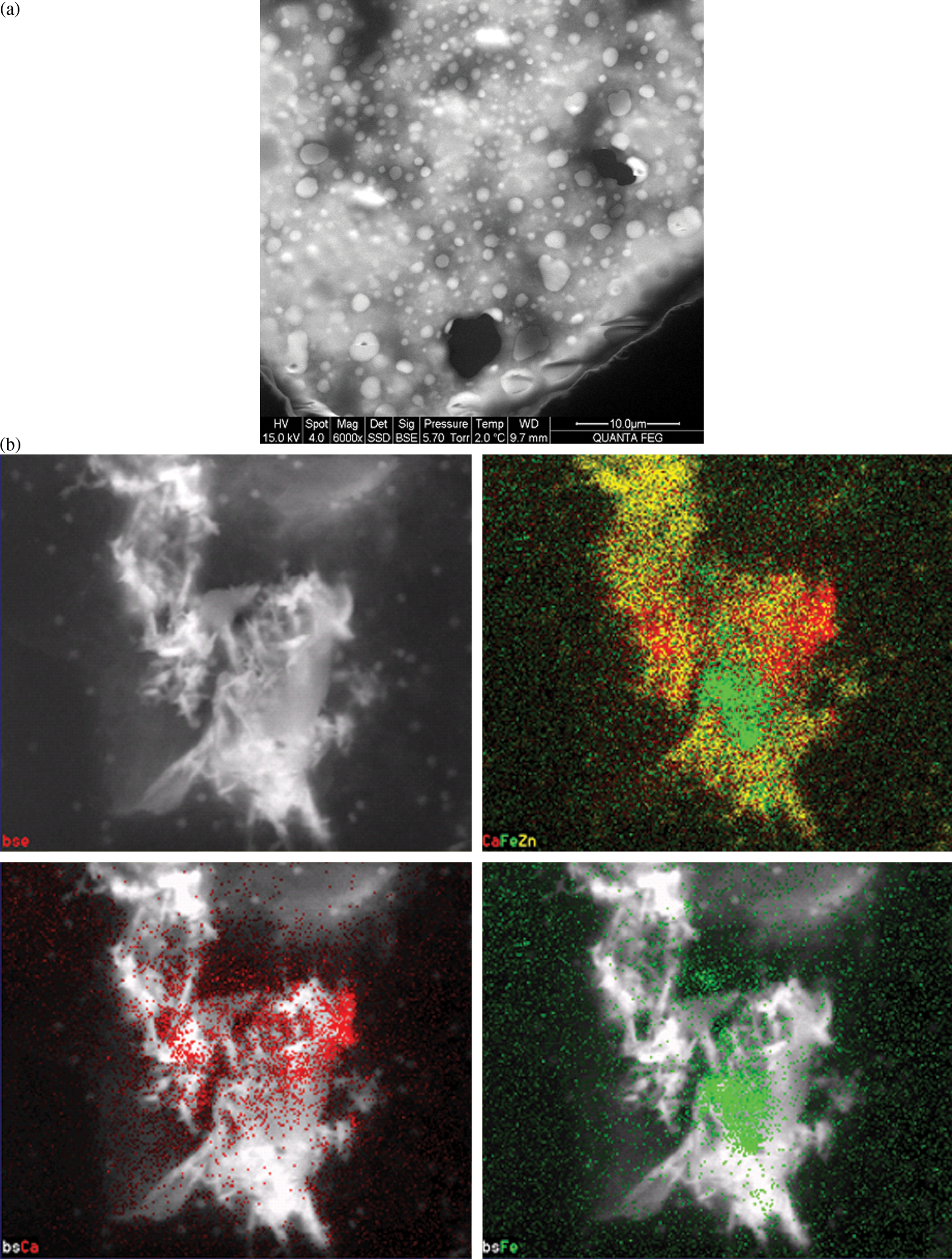
Imaging in wet scanning transmission electron microscopy. (a) Nanophases in structured liquids. (b) EDX elemental map of dispersion (wet) of metal nanoparticles. Unpublished Results from Marc Castagna, FEI.
Another extremely useful advance in SEM instrumentation is the development of extreme high-resolution imaging at very low accelerating voltages. In the past, high resolution was normally coupled directly to high KV, which came with its own set of issues and limitations. High KV brought phenomena such as penetration of the surface and charging artifacts, both limiting the ability to obtain high-resolution images of small or thin and fine surface structures (Figure 6).

Extreme high-resolution imaging, 2 kV. Intact bacteriophage on substrate, also visible is expelled genetic material from adjacent collapsed bacteriophage. Unpublished results from Ingo Gestmann, FEI.
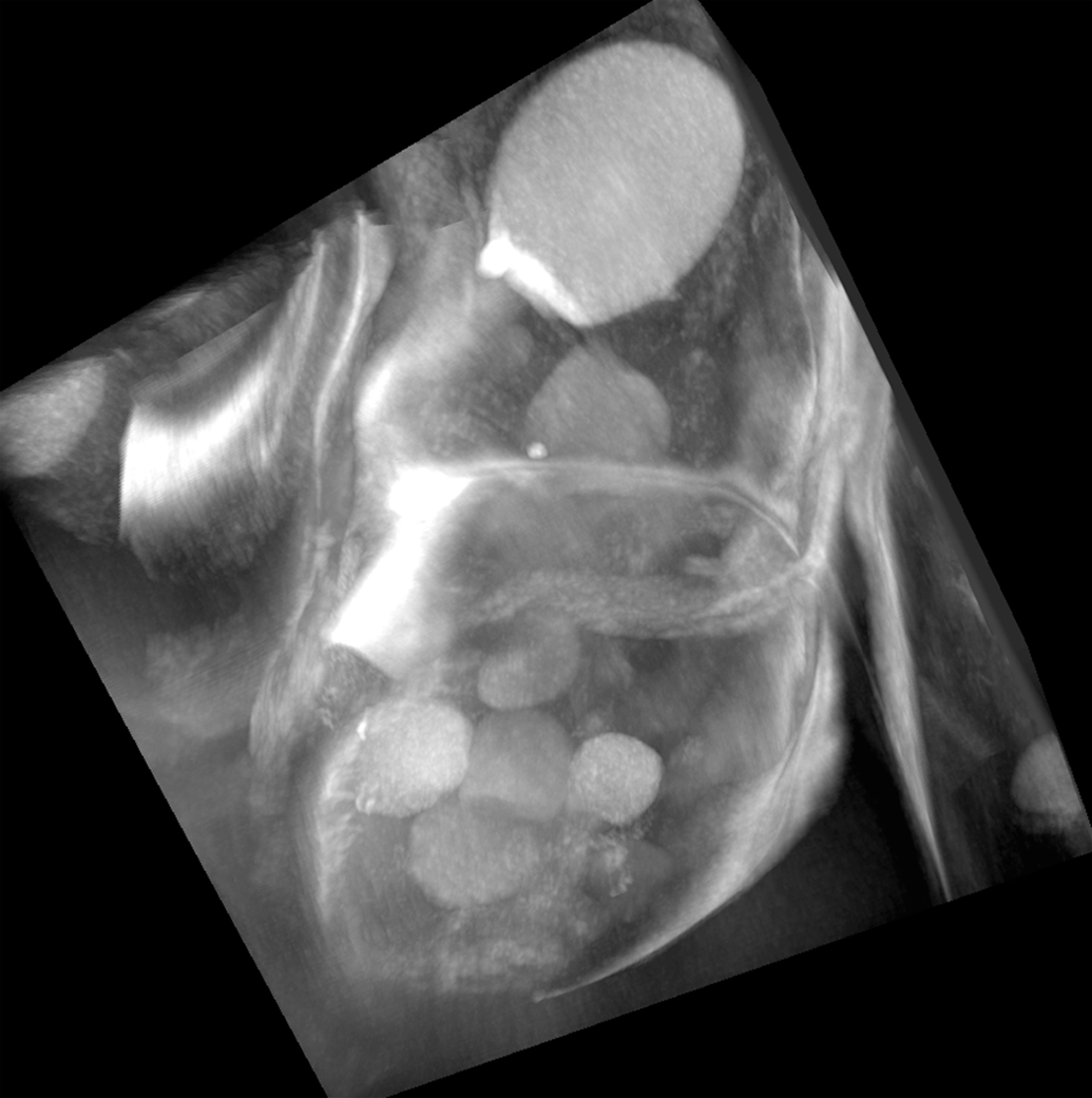
Taking only a slight detour, we can move now into the area of the
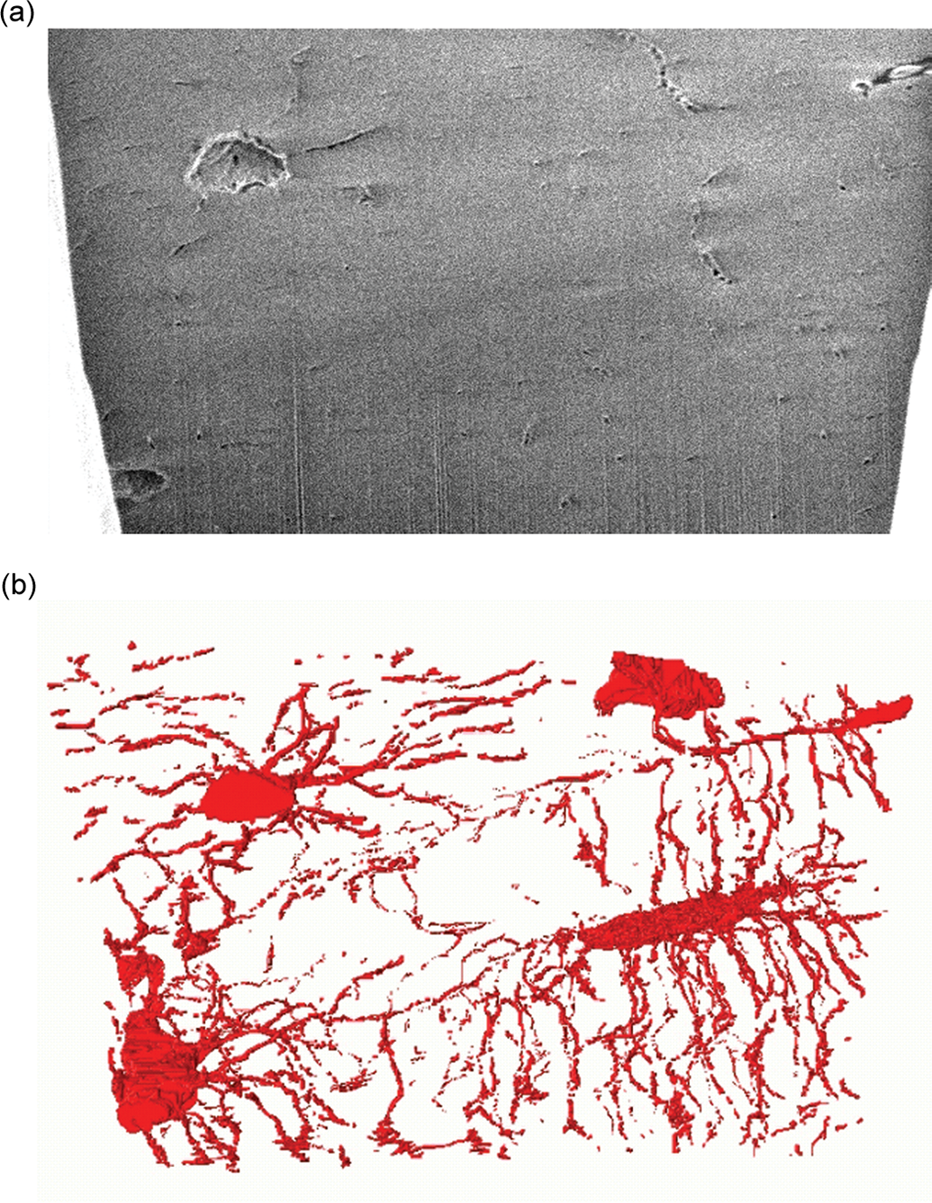
Osteoporosis studies and the potential for 3D imaging. (a) Part of a set of secondary electron images from an area sliced by the DualBeam (b) 3D reconstruction via the image data set in (a), showing osteocyte cells networked to each other via long processes called canaliculi, which are used in the exchange of nutrients and waste. Osteocytes are the most abundant cell found in bone. Stokes et al.,
Another advantage of the

See supplementary material at http://tpx.sagepub.com/supplemental for Movie 3 depicting slice and views through intestinal epithelium tissue morphology (enhanced online). See supplementary material at http://tpx.sagepub.com/supplemental for Movie 4 depicting three-dimensional reconstruction of slice and view through intestinal epithelium tissue morphology (enhanced online). Courtesy: Paul Matsudaira, National University of Singapore Intestinal epithelium tissue morphology.
One last area that I would like to examine is the ability to look at liquids, gels, or emulsions in the
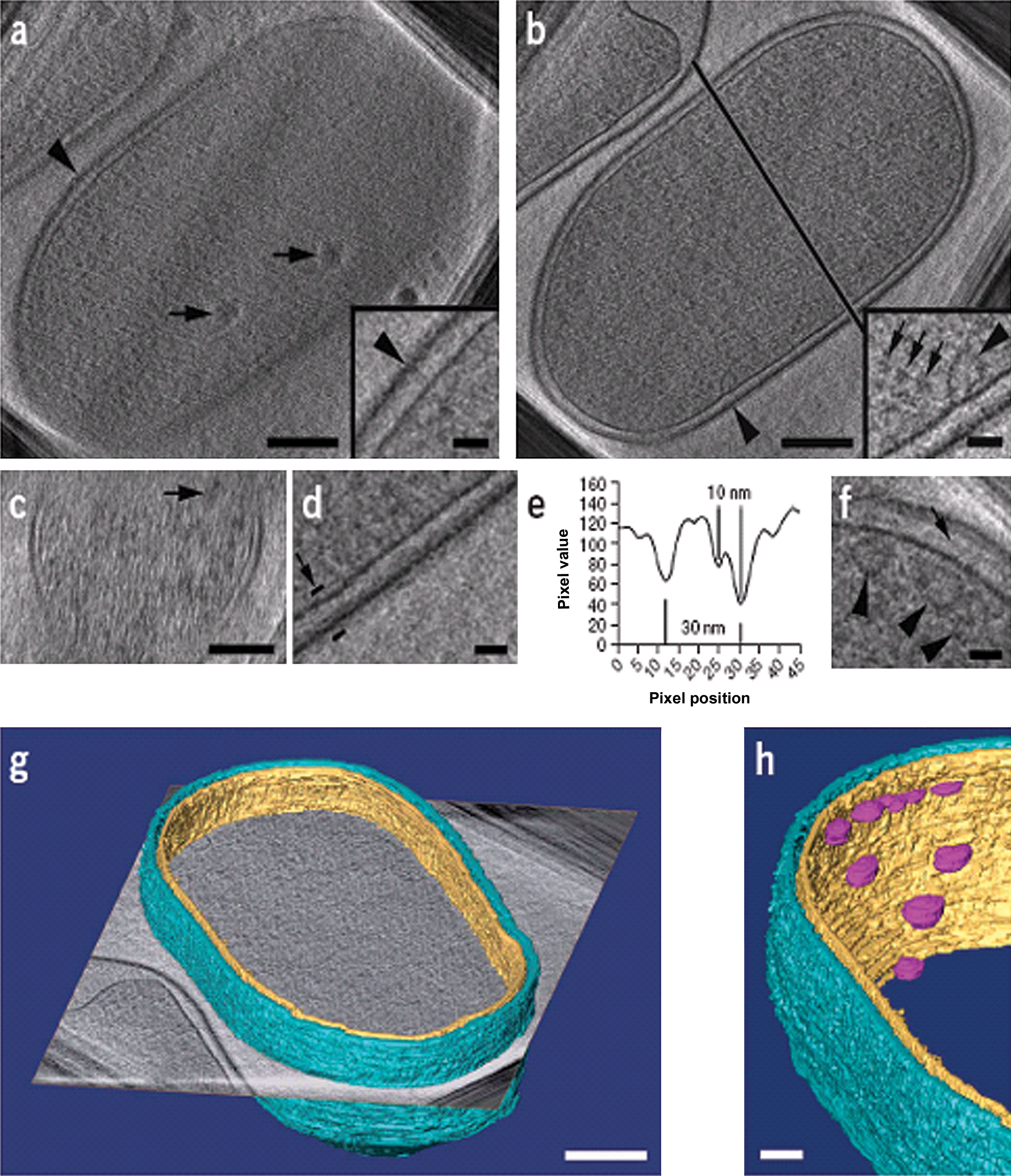
Focused Ion Beam sample for Cryo-electron Tomography of Escherichia coli cells. Marko et al., Nature Methods, 2007.
In summary, the numbers of advances that have been engineered and developed in the past few years have made it possible to see things never before observed and in much more natural states than in the past. These new tools are within reach of just about any laboratory and because of the high degree of automation, they have very short learning curves and allow rapid time to results. Whatever size or type of particle you are looking to track, there is an instrument that will allow you to do that. Whatever substrate or tissue you need to examine, there is an instrument available that will allow you to do that. Whatever type of system changes you are trying to understand, there is an instrument available that will help. Today we cannot think of electron microscopy as business as usual because business as usual is changing very rapidly. In many cases where time to results is critical, new techniques are being developed and explored that allow us to answer questions that we were not able to ask in the past. It is now possible to monitor changes in cells and systems at high resolution in large volumes. We can both ask and answer questions that have major impacts on health and wellness and get answers that repeatedly verify the value of these new technologies we use to explore them.
Footnotes
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. DualBeam is a trademark of FEI Company.
The author received no financial support for the research, authorship, and/or publication of this article.
