Abstract
The Syrian hamster, Mesocricetus auratus, is a suitable rodent species for standard regulatory toxicity studies. However, little is published about the female Syrian hamster reproductive system. It has unique anatomic features that differ from the other rodent species. In the hamster, the upper cervix is composed of 2 canals and the vagina shows 2 lateral pouches where keratin debris accumulates. These pouches must be distinguished from the vagina in order to stage the estrous cycle properly. The microscopic changes occurring during all the estrous cycle stages show some differences with the other rodents, the lower cervix and upper vagina presenting the more dramatic changes. The aim of this work was to produce a practical guide to staging the cycle and to highlight some of the differences between the rat and hamster reproductive system.
Introduction
According to the most recent publication available (Strandberg 1987), the Syrian golden hamster, Mesocricetus auratus, is cited as the fifth most commonly used laboratory research animal after mice, rats, rabbits, and guinea pigs. This desert rodent originates from northwest Syria, and nowadays most laboratory and pet hamsters are derived from 3 or 4 littermates captured in 1930 by Professor Aharoni of the Hebrew University of Jerusalem, Israel (Clark 1987). Although other strains of hamster are used in research, the Syrian hamster represents approximately 90% of those used (Newcomer et al. 1987).
The Syrian hamster is a suitable rodent species for standard regulatory toxicity studies, ranging from short-term studies through to 2-year carcinogenicity studies. In the majority of cases, the hamster is selected when good systemic drug exposure cannot be achieved in the mouse or rat, and they are also used for testing agents such as peroxisome proliferator–activated receptor α compounds that induce hepatic metabolic changes in rats and mice (Choudhury et al. 2000). Hamsters are also good models for studying induced carcinogenesis and immunobiology (Streilin 1987). It was originally reported that hamsters had a relatively low incidence of naturally occurring tumors when compared to other rodents (Trentin 1987), but recent publications indicate that this is probably not the case, and, in particular, vaginal squamous hyperplasia and papillomas are frequently observed in carcinogenicity studies (McInnes, Ernst, and Germann 2013, 2015). Hamster are highly susceptible to viral oncogenesis (including viruses from other species). It has been shown that several human adenoviruses can produce sarcomas at the site of injection in hamsters, and this susceptibility has been used to study the role of tumor antigens and defense mechanisms (Trentin 1987). In addition, due to the similar morphology and clinical condition of pancreatic tumors in hamster and man, the hamster is a good model to study pancreatic carcinogenesis (Van Hoosier and Ladiges 1984; Standop et al. 2001). An anatomical feature peculiar to hamsters is a pair of well-developed cheek pouches used to carry food or nesting material. These pouches have been referred to as “immunologically privileged” sites because tumors can be induced or transplanted easily into them without subsequent rejection by the immune system, and the reason for this lack of rejection is, however, poorly understood. The fact that hamsters lack suppressor T cells, have atypical cytotoxic T cells, and, unlike other mammals, have functionally monomorphic class I major histocompatibility complex genes, and proteins might play a role (Newcomer et al. 1987). The epithelium lining the pouches is similar to that lining the human mouth, and therefore the hamster has been used as a model for, and to study the features of, oral tumors in man (Newcomer et al. 1987).
Little is published about the female Syrian hamster reproductive system. Hamsters have a short and consistent estrous cycle (4 days), a predictable time of ovulation, and a short gestation period (16 days). Their reproductive cycle is photoperiod sensitive, and hamsters cease reproductive activity during short daylight seasons, a process mediated through melatonin production/secretion by the pineal gland (Newcomer et al. 1987).
It is well known that during preclinical drug safety evaluation, many drugs can interfere with female rat reproductive function (Yuan and Foley 2002). These changes usually manifest through alterations in the normal morphology of the reproductive tract or through disturbances of the estrous cycle (e.g., changes in stage duration, loss of synchronicity between the various reproductive organs, overrepresentation of a particular stage of the cycle in a group, etc.). In many instances, the changes can be subtle and their detection requires a good understanding by pathologists of the normal morphology and dynamics of the estrous cycle of the species under investigation. The excellent review of the female rat cycle by Westwood (2008) provides a very useful first approach to staging the cycle in other species, for example, mouse and also the hamster, but in the hamster, the anatomy and macroscopic and microscopic appearance of some regions of the female reproductive tract differ significantly from other rodents, including rats. The aim of this work was to better understand the anatomy and histology of the female hamster reproductive system to produce a visual guide to staging the cycle and finally to highlight some of the differences between the rat and hamster reproductive system.
Materials and Methods
The reproductive tracts of 47 sexually mature female Syrian hamsters (HsdHanTM:AURA, Harlan UK), approximately 10–11 weeks of age at the end of the study, were examined. These hamsters formed part of a standard 14-day good laboratory practices oral regulatory toxicology study. The study was ethically reviewed and carried out in accordance with Animals (Scientific Procedures) Act 1986 and the GSK Policy on the Care, Welfare and Treatment of Animals.
The animals were grouped housed (groups of 3) in cages conforming to the code of practice for the housing and care of animals used in scientific procedures (Home Office, London 1989). The photoperiod was a 12-hr light/dark cycle.
For the purposes of this work, ovaries, uterine horns, cervix, and vagina were examined. Tissues taken at necropsy were fixed using standard fixation procedures in 10% neutral buffered formalin. They were then paraffin embedded and sectioned at 4 μm to produce medial sections of both ovaries, transverse sections of the mid-portion of the uterine horns, and a complete longitudinal section of the distal uterus with adjacent conjoined cervix and vagina. A routine H&E stain was used to produce the final slides.
Sexual Maturity and Anatomy of the Female Hamster Reproductive System
In the female hamster, reproductive maturity is usually reached between 8 and 10 weeks of age when the average body weight is 90–100 g (Diamond and Yanagimachi 1970; Fitzgerald and Zucker 1976). Vaginal opening occurs at around 10 days of age. This is different from other laboratory rodents where vaginal opening and sexual maturity occur at the same time.
The anatomy of the female hamster upper reproductive tract is similar to that of the rat. Ovaries are 3–4 mm long, oval shaped, and each is completely enclosed within a bursa. The hamster has 2 uterine horns leading to the cervix, which in turn leads to the vagina and the vulva. The hamster cervix is different to the rat cervix in that it is composed of 2 parts, an upper dual cervix leading to the 2 uterine horns and a lower cervix composed of a single canal (the latter being comparable to the rat cervix) that leads to the upper vagina (Figure 1A).
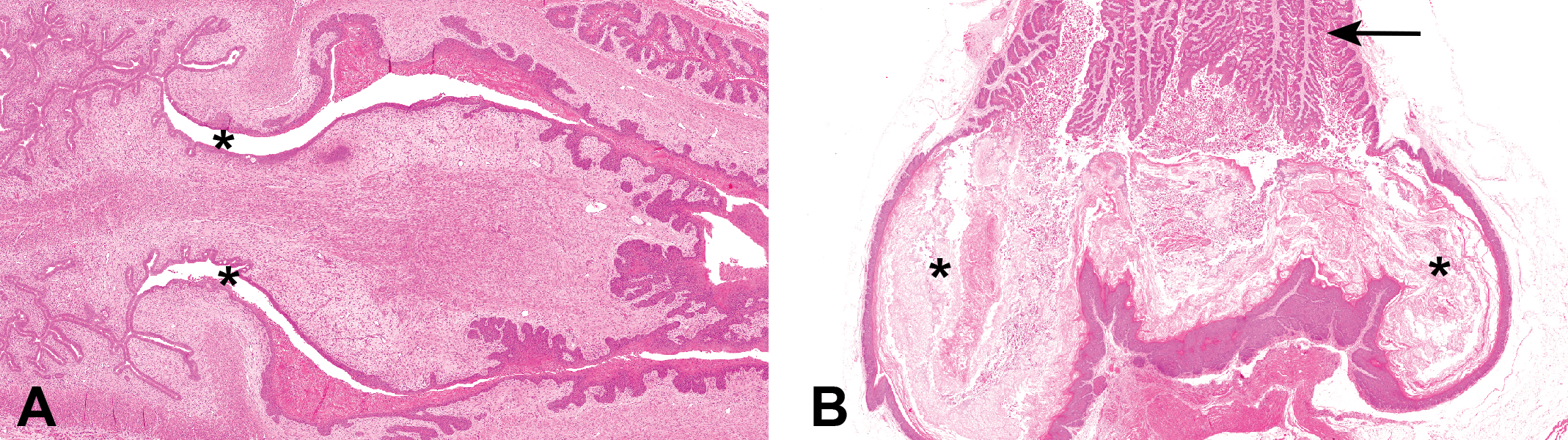
Microanatomical appearance of the female hamster reproductive tract. (A) Hamster upper cervix. Note the dual canals (*), each leading to the dual uterine horns (on the left of the photomicrograph). (B) Hamster lower vagina showing the lateral wall pouches (*). Note the accumulation of keratin debris within the pouches, which could potentially result in incorrect determination of estrous stage. Note also the histological appearance of the upper vagina (arrow), which is completely different to that of the lower vagina.
The microanatomy of the hamster vagina is also more complex than that of the rat, since it also is composed of 2 distinct regions. The upper vagina adjacent to the cervix is lined by an epithelium whose appearance, as in the rat, is responsive to the hormonal effects of the estrous cycle. In the lower vagina of the hamster, however, a distinct pair of pouches protrudes from the lateral walls. These pouches extend caudally and also laterally and ventrally before terminating close to the vaginal orifice (Figure 1B). The vaginal pouches are a unique feature of the hamster reproductive tract and contain keratin and other cell debris throughout the cycle. For this reason, in contrast to the rat, the histological appearance of the lower vagina, and/or the presence of keratin during random in-life vaginal smear evaluation, cannot be relied on to determine the estrous stage of the hamster. Daily in-life daily smearing of the vagina, however, prevents accumulation of cells from earlier stages of the cycle, and the smears obtained more nearly mirror the estrus-stage condition of the tract (Bivin, Olsen, and Murray 1987).
In all other aspects, the structural histology of the ovary, uterus, and vagina of the hamster is similar to that described for the rat and mouse (Champlin, Dorr, and Gates 1973; Westwood 2008). Histological changes associated with aging are also similar, for example, an increase in the ratio of atretic to normal follicles in the ovary and increased fibrous connective tissue in the ovary, uterus, and vagina (Rolle and Charipper 1949).
Estrous Cycle Characteristics of the Hamster Reproductive Tract
As in the rat, female hamsters have a 4-day estrous cycle. The stages of the cycle are similar in hamsters and rats, but the duration of the stages differs between the 2 species; proestrus and metestrus are significantly shorter in the hamster than in the rat, whereas diestrus is significantly longer—almost 20 hr longer in the hamster than in the rat. The duration of estrus is similar in both species (Table 1).
Estrous Cycle Duration in Rats and Hamsters.
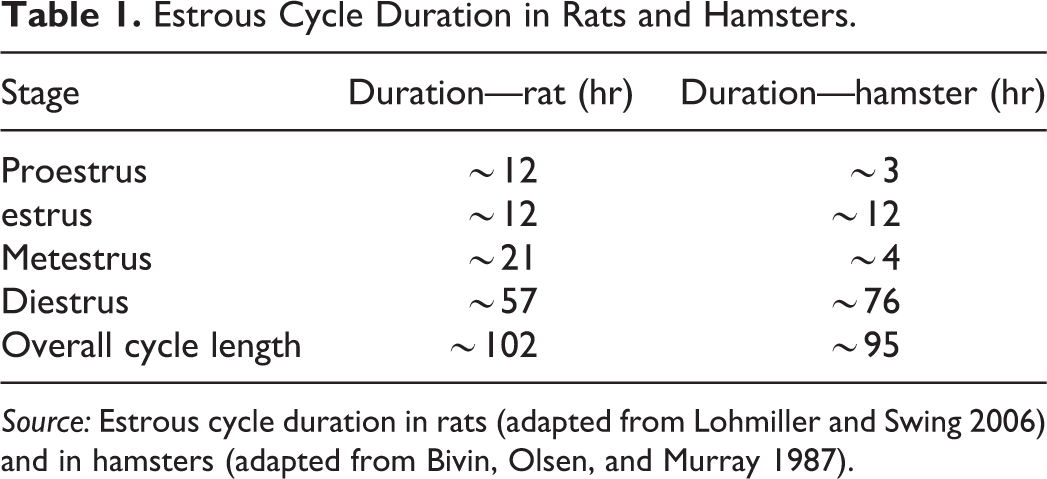
Source: Estrous cycle duration in rats (adapted from Lohmiller and Swing 2006) and in hamsters (adapted from Bivin, Olsen, and Murray 1987).
In the hamster, once ovulation has occurred the corpus luteum organizes very rapidly. Unlike the rat and mouse, where retention of several sets of corpora lutea from previous cycles is visible in the ovary (polycyclic species), in the hamster, there is rapid development and regression of corpora lutea during a single cycle which means there is never more than one generation of corpora lutea visible histologically in the ovary (Greenwald 1985). In addition, due to the short duration of proestrus, its detection may be dependent on the time of day of necropsy. Another feature unique to the hamster is the presence of a copious postovulatory discharge the day after ovulation, which can be used as an in-life tool to help determine the stage of the cycle (Lisk 1985).
Histological Variations in the Reproductive Tract throughout the Cycle
As in the rat and mouse, the histological changes seen in the reproductive tract of the hamster during proestrus, estrus/metestrus, and diestrus have distinct morphologic features allowing stage-specific identification of the cycle to be determined. The nature of these stage-specific changes in some regions of the hamster reproductive tract, however, is notably different to those seen at comparable stages in the rat or mouse, these differences being most prominent in the ovaries and particularly in the cervix and upper vagina. Also, due to the extremely short duration of metestrus in the hamster, determining estrus from metestrus changes in the tract is challenging, so these stages are usually merged and termed estrus/metestrus.
As mentioned previously, the hamster, unlike the rat, is not a polycyclic species and ovarian corpora lutea regress quickly during a single cycle. Hamster proestrus ovaries, therefore, only contain one generation of corpora lutea, that is, those from the most recent cycle, and these corpora lutea will already generally show advanced luteolysis characterized by the presence of apoptotic and degenerate luteal cells (Figures 2A and 3B). Developing follicles, including tertiary follicles, are also usually present in proestrus ovaries (Figure 2A). It is the authors’ experience that unlike the rat, detection of ruptured nonluteinized postovulatory follicles (hallmark of estrus in the rat) is rare in the hamster ovary since luteinization in this species is such a rapid process. For this reason, it is probably more practical in the hamster to use the merged term estrus/metestrus to describe the next cycle stage in the ovary, which is depicted histologically either by the absence of distinguishable corpora lutea or more commonly by the presence of small, early corpora lutea, containing a mixture of fusiform and round luteal cells sometimes surrounding a central cavity (Figure 2C). Mature corpora lutea are only found in diestrus ovaries (Figure 2E) where large corpora lutea containing mature, round intact luteal cells are present (secretory phase). Occasionally, in late diestrus, very early degeneration of the luteal cells may be present as the cycle approaches proestrus once more.
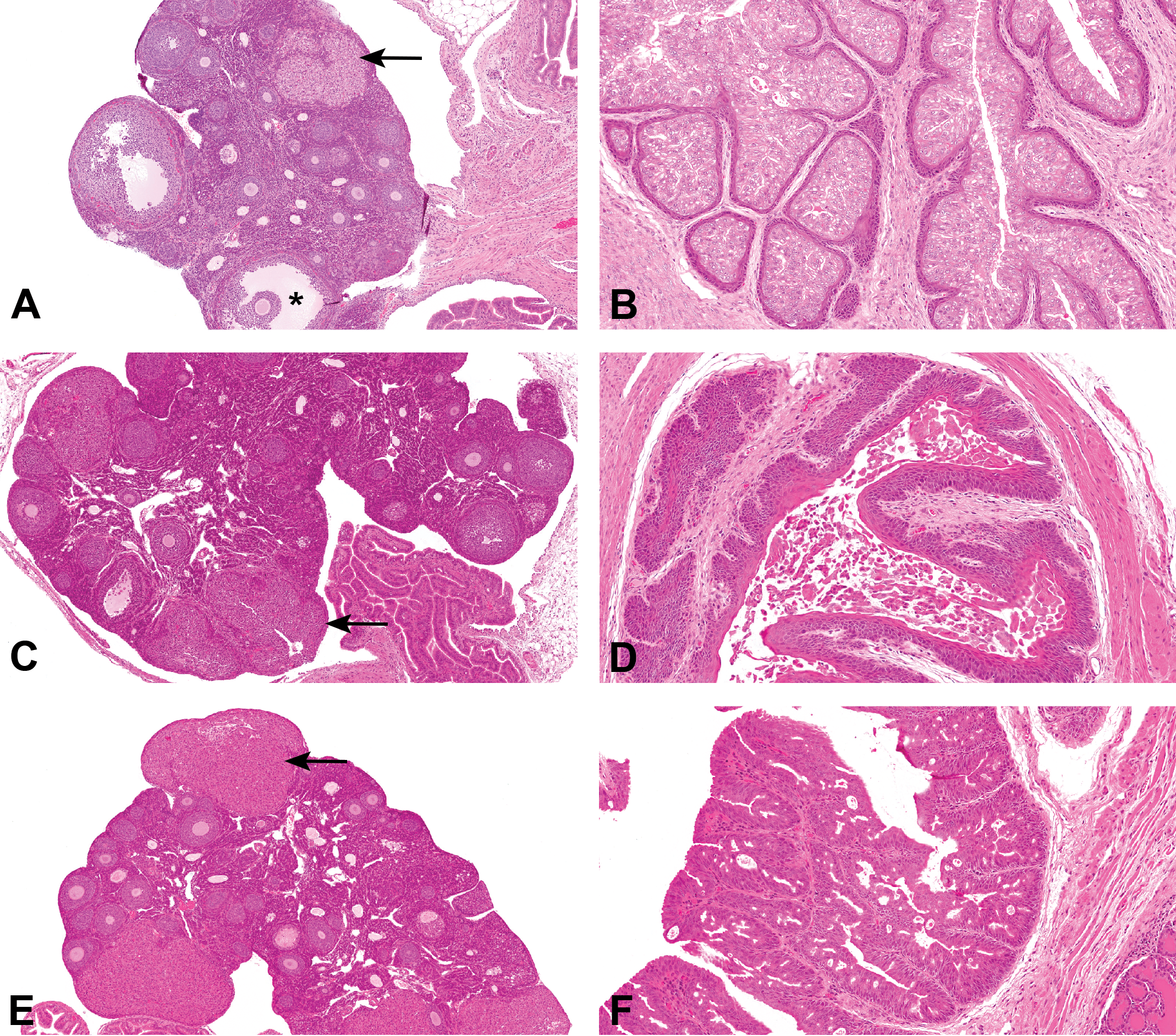
Visual guide to staging the female hamster estrous cycle. Ovarian and cervical/vaginal changes. (A) Hamster ovary: proestrus. Corpus luteum (arrow) showing apoptosis/degeneration of luteal cells. Note absence of corpora lutea from previous cycles. Developing follicles, including antral follicles (*), also present. (B) Hamster lower cervix/upper vagina: proestrus. Note foliaceous appearance of luminal epithelium and underlying thin, squamous, nonkeratinized squamous epithelium. (C) Hamster ovary: estrus–metestrus, luteinizing follicles, and early corpora lutea are present (arrow). Detection of recently ruptured postovulation follicles is rare in the hamster. (D) Hamster lower cervix/upper vagina: estrus–metestrus. Note thickening of basal squamous epithelial layer with prominent “rete peg” appearance and surface keratinization. Mucous cell layer and keratin are exfoliated into lumen. (E) Hamster ovary: diestrus. Well-developed mature corpora lutea are present (arrow). (F) Hamster lower cervix/upper vagina: diestrus. Epithelium is stratified and thicker with prominent pseudo-vacuoles containing pale eosinophilic fluid.
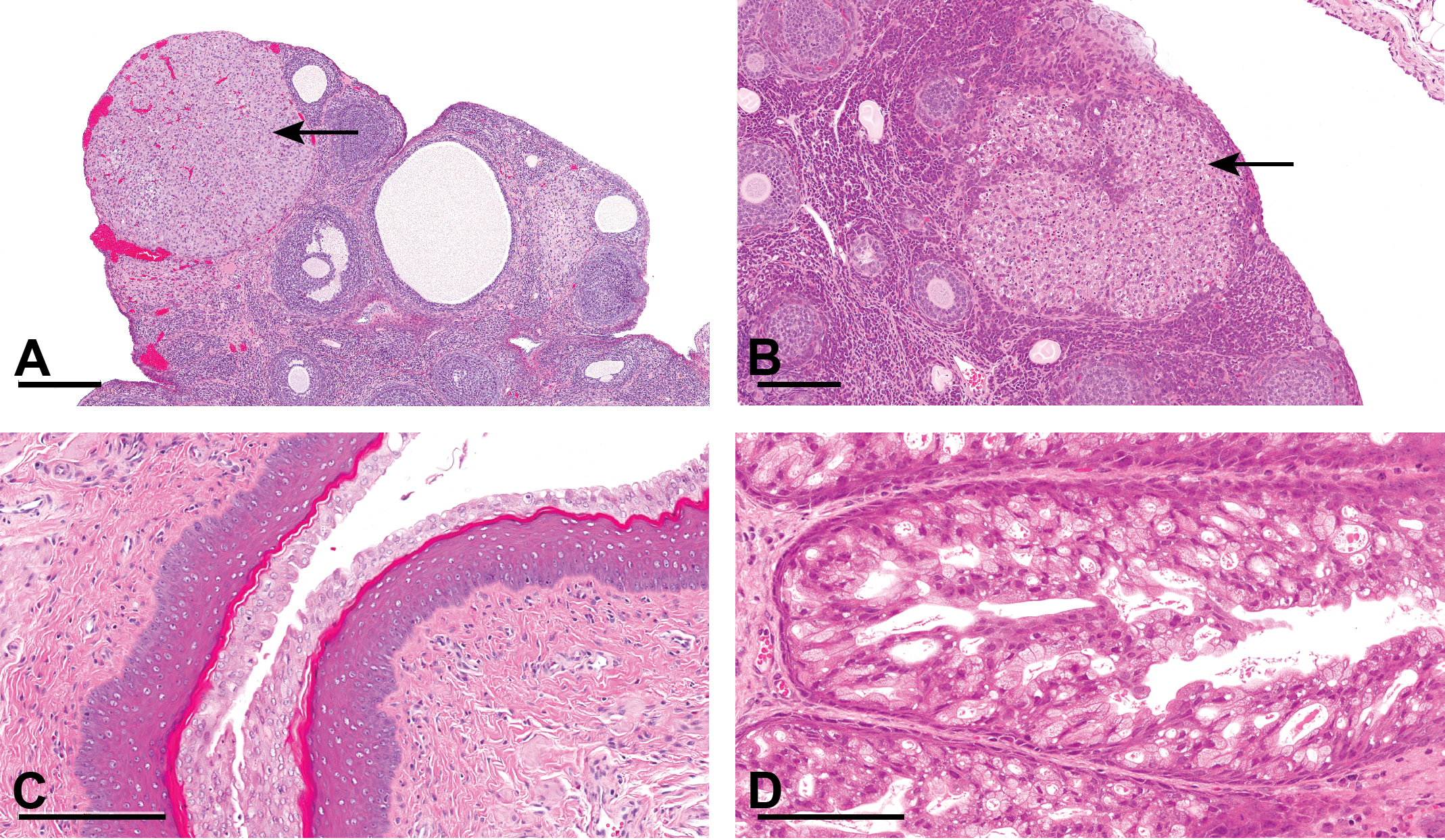
Comparison between rat and hamster ovary and vagina/upper vagina during proestrus. (A) Rat ovary during proestrus. Note the most recent corpus luteum in this ovary, which is showing apoptosis and degeneration of luteal cells (arrow), the hallmark of proestrus in the rat. Also notice the presence of a corpus luteum from a previous cycle (*). (B) Hamster ovary during proestrus. Note here also the most recent corpus luteum in this ovary, which is showing apoptosis and degeneration of luteal cells (arrow), the hallmark of proestrus in the hamster. Unlike the rat, corpora lutea from previous cycles will not be present in the hamster ovary as they undergo such rapid and complete luteolysis. (C) Rat vagina during proestrus. Note the typical and prominent mucified layer overlying a thick layer of squamous epithelium showing early, but prominent, keratinization. (D) Hamster upper vagina during proestrus. Note the foliaceous appearance of the epithelium, which is composed of elongated and vacuolated mucous-type cells overlying a thin layer of nonkeratinized squamous epithelium.
The morphological changes in the uterus of the hamster at the various stages of the estrous cycle are broadly comparable to those seen in the rat, although in comparison with the rat, the changes are less distinctive and less prominent. In proestrus, the luminal epithelium of the hamster uterus is tall and columnar and tends to be pseudostratified. In the luminal and glandular epithelium, prominent mitotic figures are visible while apoptosis is absent (Figure 4A). Additional findings associated with this stage are luminal dilation and subtle edema of the uterine stroma. No real distinguishing features differentiate between estrus and metestrus in the hamster uterus. The luminal epithelium generally remains tall and pseudostratified but in contrast to proestrus, prominent apoptosis of the luminal and glandular epithelium is present while mitotic figures are generally absent (Figure 4B). Although minimal leukocytes may occasionally be visible in the stroma and/or epithelium of the uterus during this stage, in contrast to the rat, they do not appear to be either a prominent or a consistent feature of estrus/metestrus in the hamster. In early diestrus, the uterine luminal epithelium is low and columnar in appearance, and the predominant change in both the luminal and the glandular epithelium is apoptosis (Figure 4C), although a few mitotic figures may also be present. In late diestrus, the luminal epithelium takes on a taller columnar appearance, and the predominant change in the luminal and glandular epithelium is usually the presence of mitotic figures. In the authors’ opinion, the uterus is the least useful region of the reproductive tract for staging the estrous cycle in hamsters.
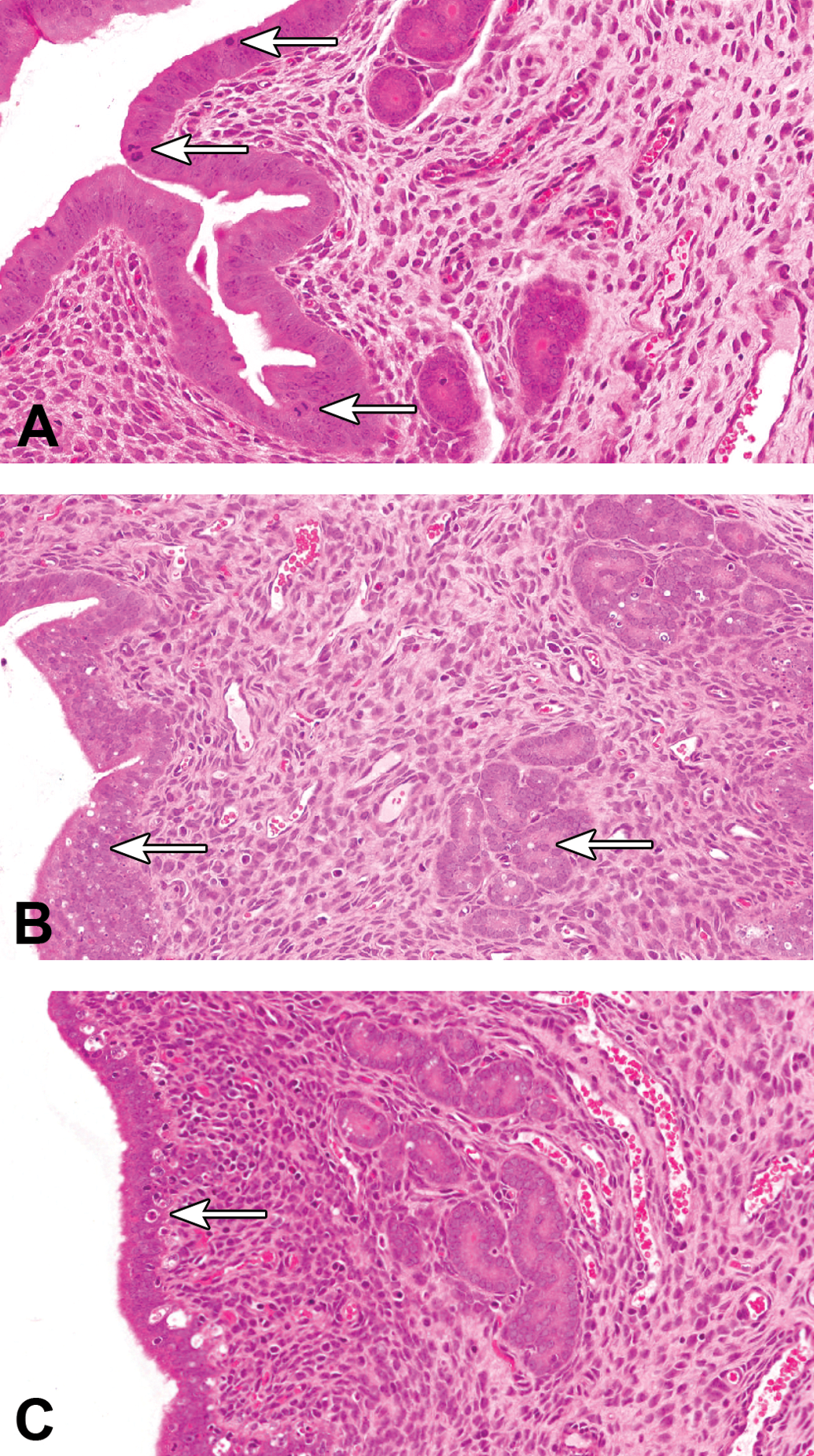
Visual guide to staging the female hamster estrous cycle. Uterine changes. (A) Hamster uterus: proestrus. Note the tall columnar, pseudostratified luminal epithelium and presence of mitotic figures (arrows). Epithelial apoptosis/vacuolation is absent in luminal and glandular epithelium. (B) Hamster uterus: estrus–metestrus. The luminal epithelium is still tall, but now prominent apoptosis/vacuolation of the luminal and glandular epithelium is present (arrows). Mitotic figures are absent. (C) Hamster uterus: early diestrus. Luminal epithelium is low columnar. Note some apoptosis/vacuolation is still present in the luminal epithelium (arrows).
Morphologically, the most distinct and unique estrous stage changes in the hamster occur in the lower cervix/upper vagina, and for this reason, this region of the reproductive tract is the most useful for assessing the cycle stage. During proestrus, the epithelium of the cervix /upper vagina has a foliaceous appearance characterized by the presence of prominent, elongated, and vacuolated mucous-type cells sitting on a more compact, thin layer of squamous epithelium (Figures 2B and 3D). During estrus/metestrus, the underlying epithelium becomes thicker and keratinized, with a “rete peg” type appearance, and exfoliation of the mucous cell and keratin layers takes place, with maximum exfoliation occurring during metestrus (Figure 2D). This luminal exfoliation accounts for the clinical postovulatory discharge mentioned previously. Luminal inflammatory cells may also be present during this stage. During diestrus, the appearance of the vaginal epithelium changes again as surface cells elongate toward the lumen, epithelial mitotic activity is visible, and the epithelium becomes stratified and thicker with prominent pseudo-vacuoles. In early diestrus, clusters of inflammatory cells may be present within the epithelium/vacuoles. In late diestrus, the vacuoles tend to contain pale eosinophilic fluid (Figures 2F and 5D).
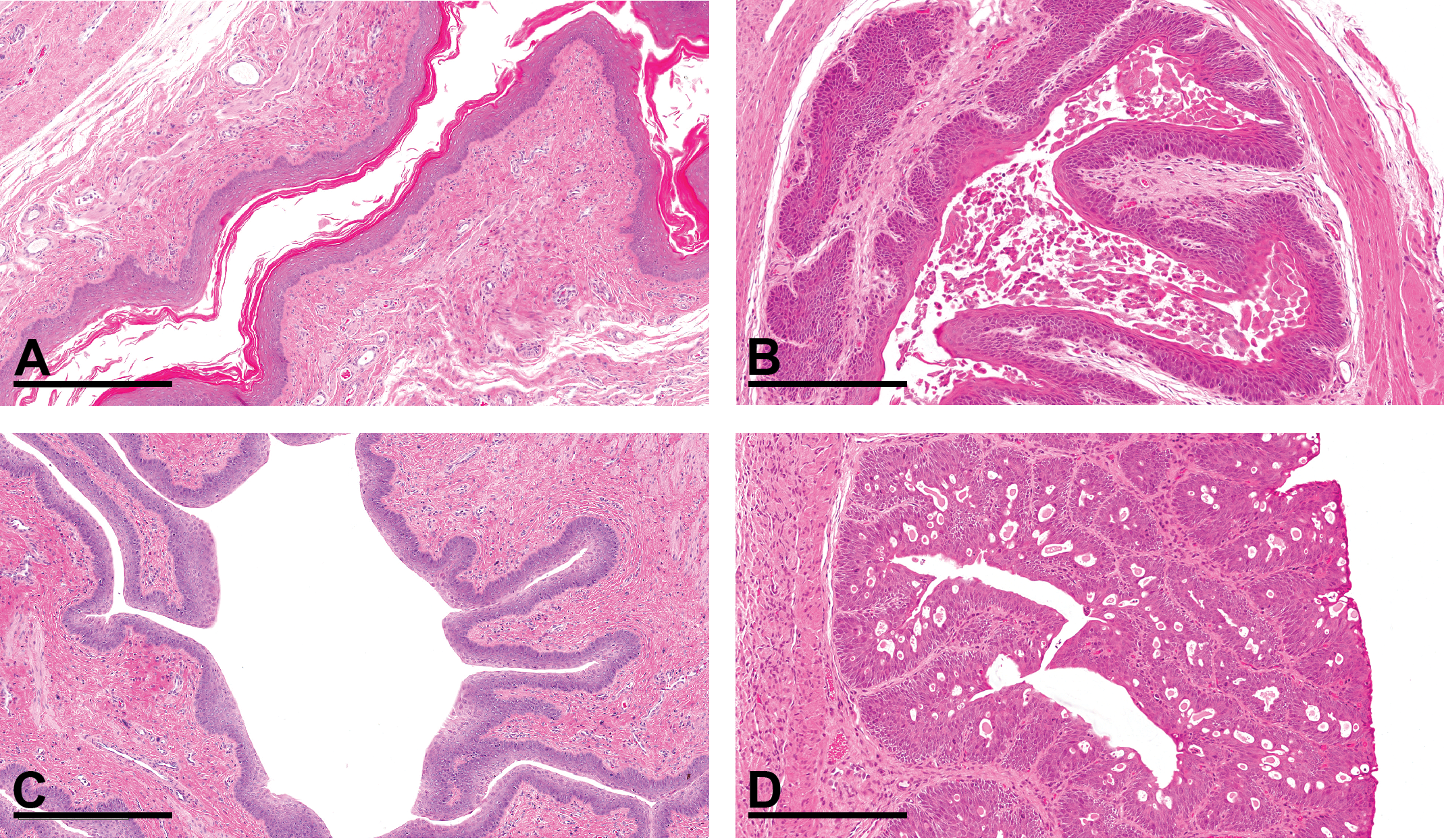
Comparison between rat and hamster vagina/upper vagina during estrus/metestrus and diestrus (bar: 300 μm). (A) Rat vagina during estrus. Note the thickened squamous epithelium and shedding of the keratin layer into the lumen. (B) Hamster upper vagina during estrus/metestrus. Note thickening of basal squamous epithelial layer with prominent “rete peg” appearance and surface keratinization. Mucous cell layer and keratin are exfoliated into lumen. (C) Rat vagina during late diestrus. Note the stratified epithelium and very early mucification of the apical layer (latter indicative that this is late diestrus). (D) Hamster upper vagina during late diestrus. Note the florid, pseudostratified appearance of the epithelium and the presence of pseudo-vacuoles containing pink eosinophilic fluid.
Table 2 summarizes the main stage-specific changes seen in the hamster reproductive tract during the estrous cycle. As with any species used in regulatory toxicity studies, examination of the whole reproductive tract is essential in order to accurately determine the stage of the cycle. It is the authors’ experience that in the rat, and to a lesser extent in the mouse, it is useful in the first instance to use the specific appearance of the corpora lutea in the ovaries to determine the cycle stage, following this, to then assess the histological appearance of the uterus, and finally, the vagina to help confirm the stage-specific findings seen in the ovary. Other pathologists prefer to assess the vaginal morphology first and then that of the ovary and uterus. Regardless of the initial approach, the goal is to assess synchronicity of changes in the ovary, uterus, and vagina. The presence/appearance of ovarian corpora lutea is also useful in the hamster when assessing estrous stage, but the preferred region for this assessment in the hamster is the lower cervix/upper vagina, due to the distinct and easily recognizable histological changes associated with each stage.
Summary of the Histological Features of the Hamster Female Reproductive Tract during the Estrous Cycle.
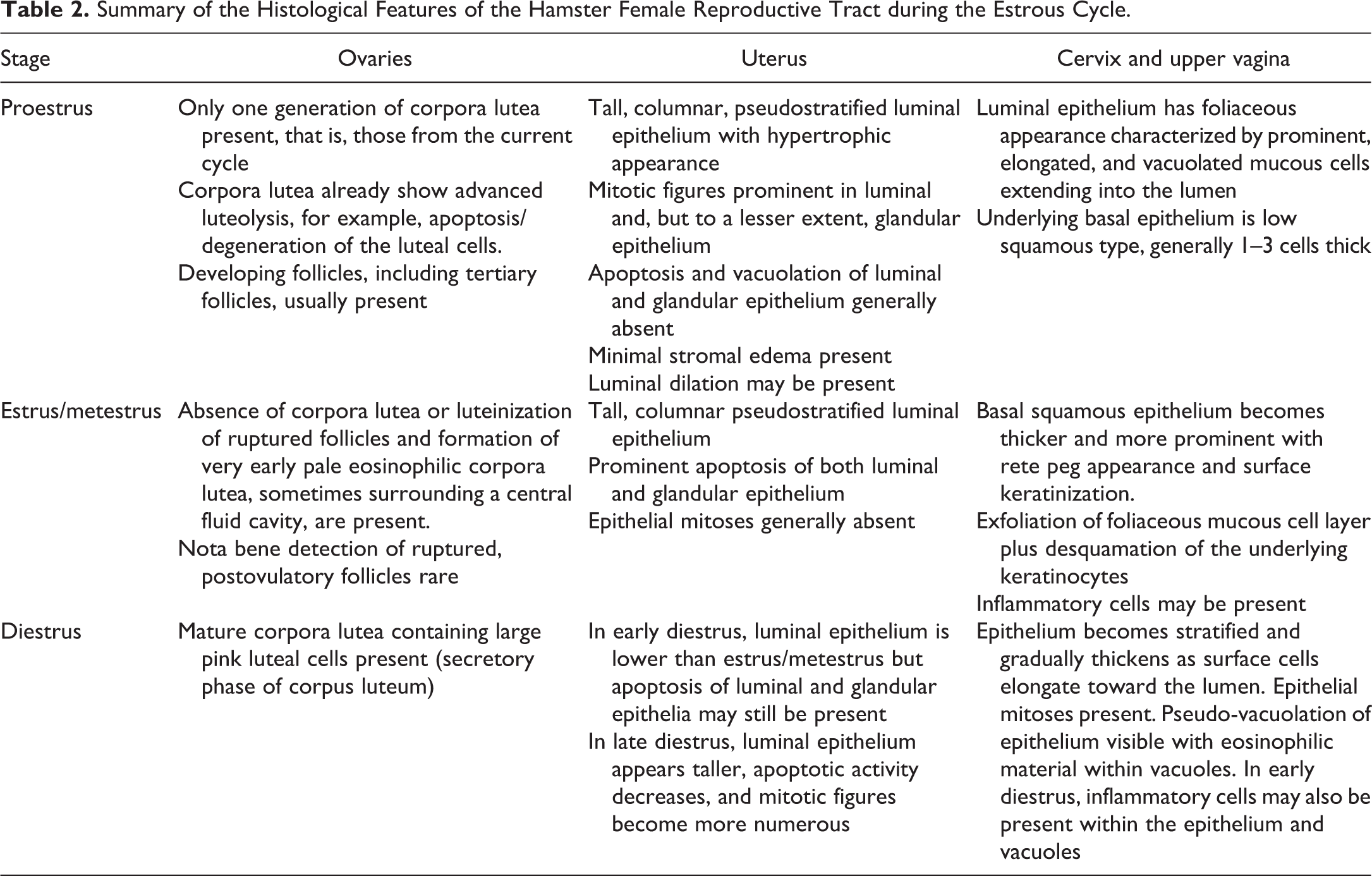
Figure 3 illustrates the major differences in the ovary and vagina/upper vagina of the rat and hamster during proestrus. Figure 5 illustrates the major differences in the vagina/upper vagina of the rat and hamster during estrus/metestrus and diestrus.
Conclusions
The purpose of this article was to guide the toxicological pathologist toward a pragmatic approach to staging the estrous cycle in the hamster, through descriptions and illustrations of the changes present in the reproductive tract at various stages of the cycle, and also to illustrate some of the differences between the histological appearance of the stages in the hamster compared to the rat.
The cervix and vagina of the hamster are anatomically distinctly different from the rat, and, for this reason, tissue selection, processing, and sectioning of these regions in the hamster, particularly the vagina, require close monitoring. To avoid confusion, we suggest that the whole length of the hamster vagina is trimmed and sectioned as a single longitudinal sample attached to the cervix as this will allow good visualization of the upper vagina (with its distinct hormonally responsive epithelium) as well as the lower vagina and the vaginal pouches (which are not hormonally responsive). If a transverse section of vagina is required, then this should be taken from the upper vagina to avoid accidental sampling of the pouches and then incorrectly determining estrous stage because of the constantly keratinized appearance of the pouch epithelium.
In the hamster, the histological appearance of the cervix/upper vagina and, to some extent, the ovaries is key in assessing the stage of the estrous cycle as they are the tissues which show the most prominent and distinguishable cyclical changes. The uterus is the least useful region of the tract for assessing estrous stage but should be examined as diligently as possible as additional confirmation of the stage and to confirm synchronicity with the rest of the reproductive tract. Furthermore, since the histological appearance of all regions of the tract in the hamster during estrus and metestrus appears to be very similar (possibly influenced by the short duration of metestrus in the hamster), we suggest that it may be practical in the hamster to merge these 2 stages into one and use the term estrus/metestrus.
In this study, the most commonly observed stage of the cycle was diestrus (60%), followed by estrus/metestrus (24%), and finally proestrus (17%). Given the respective lengths of each cycle stage, these percentages would be the expected frequency for these observations.
Footnotes
Author Contributions
All authors (FC and AW) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
