Abstract
Pineal gland tumors are very rare brain lesions in rats as well as in other species including humans. A total of 8 (out of 1,360 examined) Wistar rats from 3 different combined chronic toxicity/carcinogenicity or mere carcinogenicity studies revealed pineal gland tumors. The tumors were regarded to be spontaneous and unrelated to treatment. The morphology and immunohistochemical evaluation led to the diagnosis malignant pinealoma. The main characteristics that were variably developed within the tumors were the following: cellular atypia, high mitotic index, giant cells, necrosis, Homer Wright rosettes, Flexner-Wintersteiner rosettes and pseudorosettes, positive immunohistochemical reaction for synaptophysin, and neuron-specific enolase. The pineal gland is not a protocol organ for histopathological examination in carcinogenicity studies. Nevertheless, the pineal gland can occasionally be encountered on the routine brain section or if it is the origin of a tumor protruding into the brain, the finding will be recorded. Therefore, although known to be a rare tumor in rats, pineal neoplasms should be included in the list of possible differential diagnoses for brain tumors, especially when the tumor is located in the region of the pineal body.
Introduction
Pineal gland tumors occur extremely rare in humans (0.2–0.7% of all intracranial neoplasms) as well as in other mammalian species including the rat (0.01–0.8%; Al Zubaidy and Malinowski 1984; Krinke et al. 1985; Surawicz et al. 1999). There is no report on chemically induced pineal gland tumors in rats (Maekawa and Mitsumori 1990). In the literature, rodent neoplasms of the pineal gland are often just reported as pineal gland tumors or pinealoma. In human pathology, the differentiation is made between pineocytoma, pineal parenchymal tumor (PPT) of intermediate differentiation, and pineoblastoma (Louis et al. 2007). Since 2007, a new entity is adapted in the human World Health Organization (WHO) classification: papillary tumor of the pineal region.
The pineal gland (pineal body and corpus pineale) is located in the brain midline close to the skull between the cerebral hemispheres and cerebellum. In the rat, the pineal gland occurs as spherical structure, approximately 1 mm in diameter, adjacent to the occipital lobes of the cortex dorsal to the caudal colliculus and posterior to the wall of the third ventricle. The pineal gland contains cells related to retinal photoreceptors which are sensitive to light and secrete melatonin which acts as neuroendocrine regulator (Reiter 1991).
In 3 combined chronic toxicity/carcinogenicity or carcinogenicity studies performed at BASF SE (Ludwigshafen, Germany), 8 tumors in total in the region of the pineal gland were observed. The studies were performed between the early 1990s and 2012. Highest incidence of pineal gland tumors were observed in the 2012 performed study with occurrence of 5 tumors. The findings in H&E diagnostics as well of immunohistochemistry are described.
Material and Methods
All studies were conducted at the BASF Experimental Toxicology and Ecology Facility (Ludwigshafen, Germany) in compliance with Good Laboratory Practice Principles of the German “Chemikaliengesetz” (Chemicals Act) and the Organization for Economic Cooperation and Development (OECD), and designed to meet or exceed the known requirements of test guidelines OECD # 453, Commission Regulation (EC) No 440/2008, OPPTS 870.4300, and JMAFF No. 12-Nousan-8147. The studies were performed in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)-accredited laboratory.
Most tumor-bearing animals (5) were from a combined chronic toxicity/carcinogenicity study that was performed at BASF Experimental Toxicology and Ecology (study I) with Wistar rats (Crl:WI(Han)) and was finalized in 2012. The animals were about 35 days old at receipt and were obtained from Charles River Laboratories (Sulzfeld, Germany). The animals were held individually in polysulfonate cages with a floor area about 2,065 cm2 (TECNIPLAST, Hohenpeißenberg, Germany) covered with small amounts of bedding. The cages were closed with wire covers (Ehret, Emmendingen, Germany). Dust-free wooden bedding was used and wooden gnawing blocks (Typ NGM E-022) were added (Abedd® Lab. and Vet. Service GmbH, Vienna, Austria) for environmental enrichment. The animal room was completely disinfected prior to the study using a disinfector (“AUTEX,” fully automatic, formalin-ammonia-based terminal disinfector). The floor and the walls were cleaned once a week with water containing an appropriate disinfectant. The animal room was ventilated with fresh air, with 15 air changes per hour; the temperature was maintained at 20° to 24°C with relative humidity 30% to 70% and a 12 hr light–dark cycle. The animals were allowed to acclimatize for 1 week before the initiation of experiments with food and water available ad libitum. Animals that had to be sacrificed received Narcoren® anesthesia and were then killed by decapitation.
All affected animals were males and were sacrificed moribund (between 318 and 434 days after start of exposure) or died throughout the study (359 days after start of exposure), except for 1 animal of the chronic group that was sacrificed as scheduled after 12 months. The tumor-bearing animals were distributed as follows between the test groups (control/low-dose group/mid-dose group/high-dose group): 0/1/2/1. The affected animal of the chronic (1-year treatment) group was from the low-dose group.
Retrospectively, carcinogenicity studies performed at BASF Experimental Toxicology and Ecology were reviewed for the occurrence of brain tumors with regard to the new International Harmonization of Nomenclature and Diagnostic criteria (INHAND) initiative. In 2 older studies, glioma or glioblastoma was diagnosed in the brain but after reevaluation found to be pineal gland tumors. In 1 study finalized in 1994, 2 females, 1 control and 1 high-dose animal, revealed lesions in the pineal gland region (study II). In a third study (study III), finalized in 2001, a female control animal showed a pineal gland lesion. These two studies (studies II and III) were performed with Wistar rats (Chbb:THOM (SPF)) from a different breeder.
All tumors were fixed in 10% neutral-buffered formalin, routinely processed (trimming, paraplast-embedding, and cutting), stained with hematoxylin & eosin (H&E), and examined by light microscopy. The following immunohistochemical stains were performed: synaptophysin (SYN, Zytomed 1:100), neuron-specific enolase (NSE, DBS Pleasanton, California, ready to use), neurofilament (NF, BioGenex, 1:100), glial fibrillary acidic protein (GFAP, BioGenex, 1:200), S-100 protein (S100, Zytomed, 1:100), and pan-cytokeratin (PanCK, Sigma, 1:500). Detection system was ZytoChem Plus Polymer Kits (HRP) and DAB served as chromogen.
Results
In study I (finalized in 2012), 5 of the 480 animals (incidence of 1%) revealed pineal gland tumors. The tumor-bearing animals were distributed as follows between the test groups (control/low dose group/mid dose group/high-dose group): 0/1/2/1. The affected animal of the chronic (1-year treatment) group was from the low dose group.
In study II (finalized 1994), a glioma and a glioblastoma in females of the control and high-dose group were observed out of 400 animals examined (incidence 0.5%).
In study III (finalized 2001), 1 male of the low-dose group revealed a malignant pinealoma and in one female of the control group a glioma was diagnosed out of a total of 480 animals (incidence 0.42%). All the gliomas and glioblastomas were reevaluated and diagnosed as pinealomas.
Macroscopy
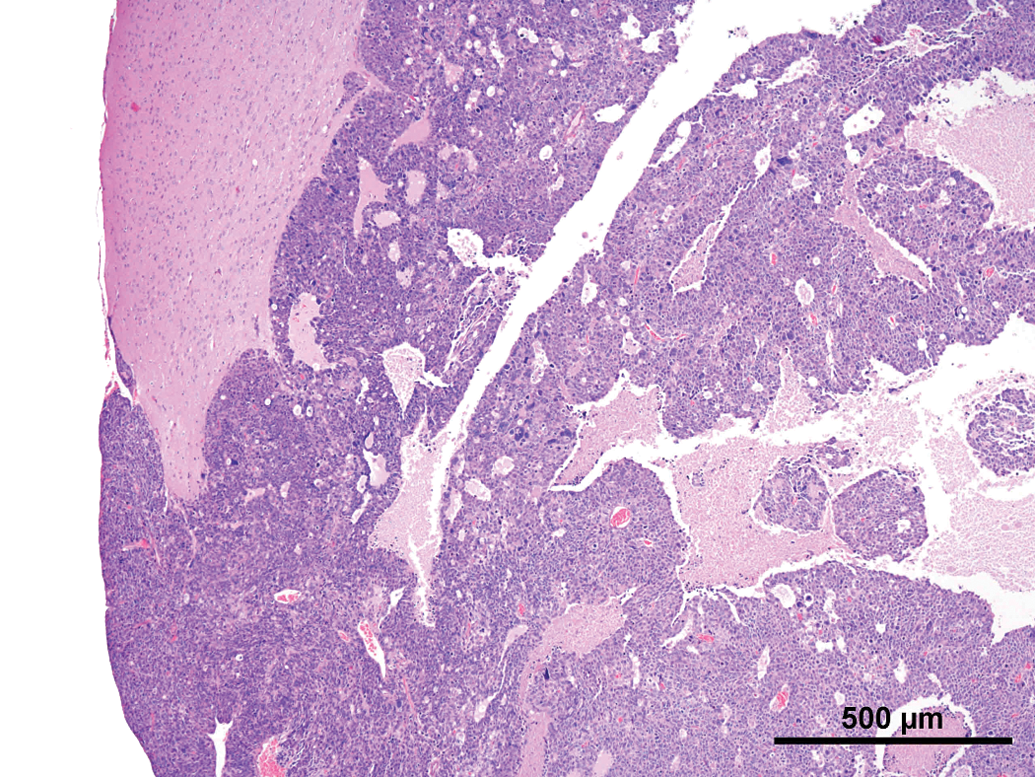
The lesions were described as masses in the region of the pineal gland (located between the cerebral hemispheres and the cerebellum). The tumors were between 8 mm and 10 mm in diameter with beige, light brownish or reddish color (Figure 1).
Rat, study I, brain, red-brown mass in the area between the cerebral hemispheres and the cerebellum and infiltrating both.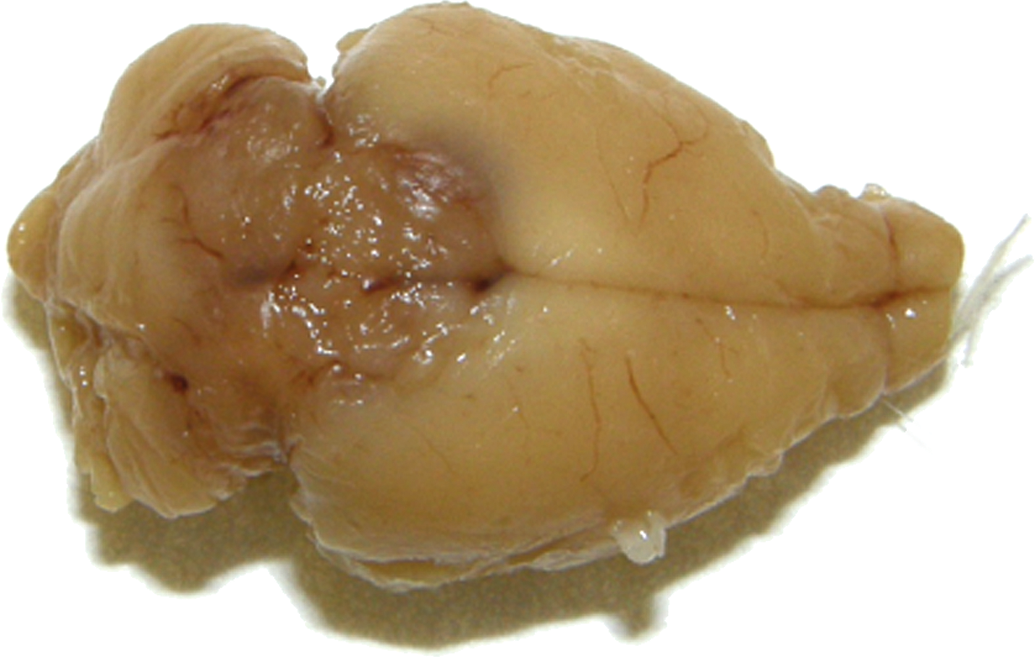
Microscopy
H&E
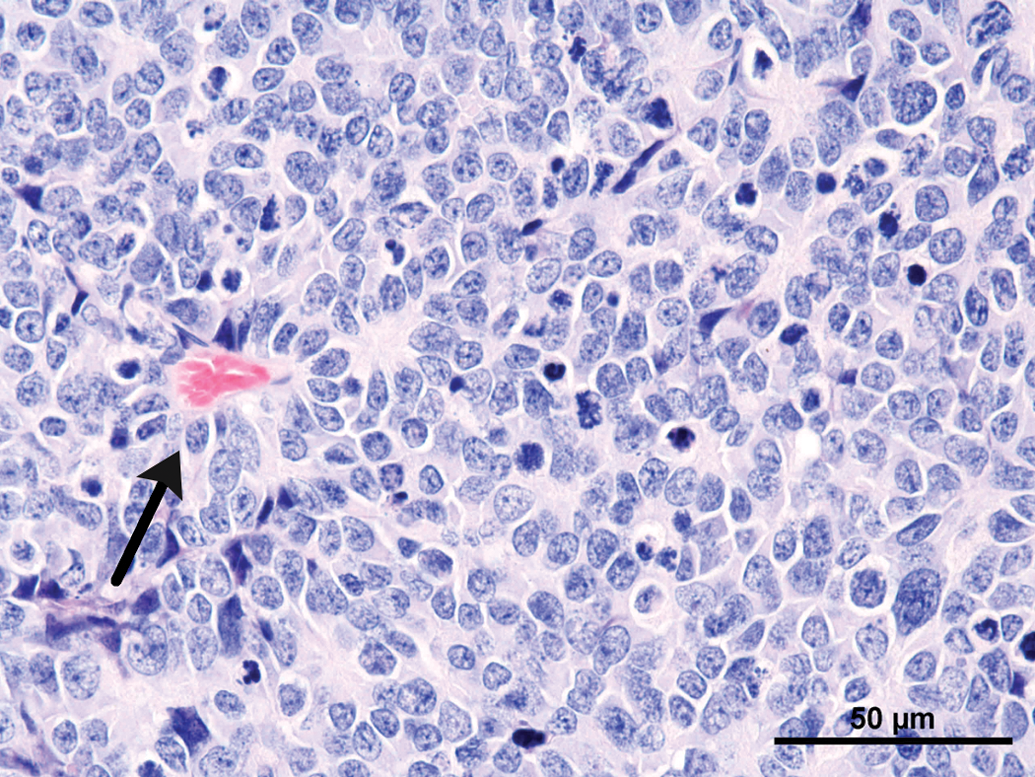
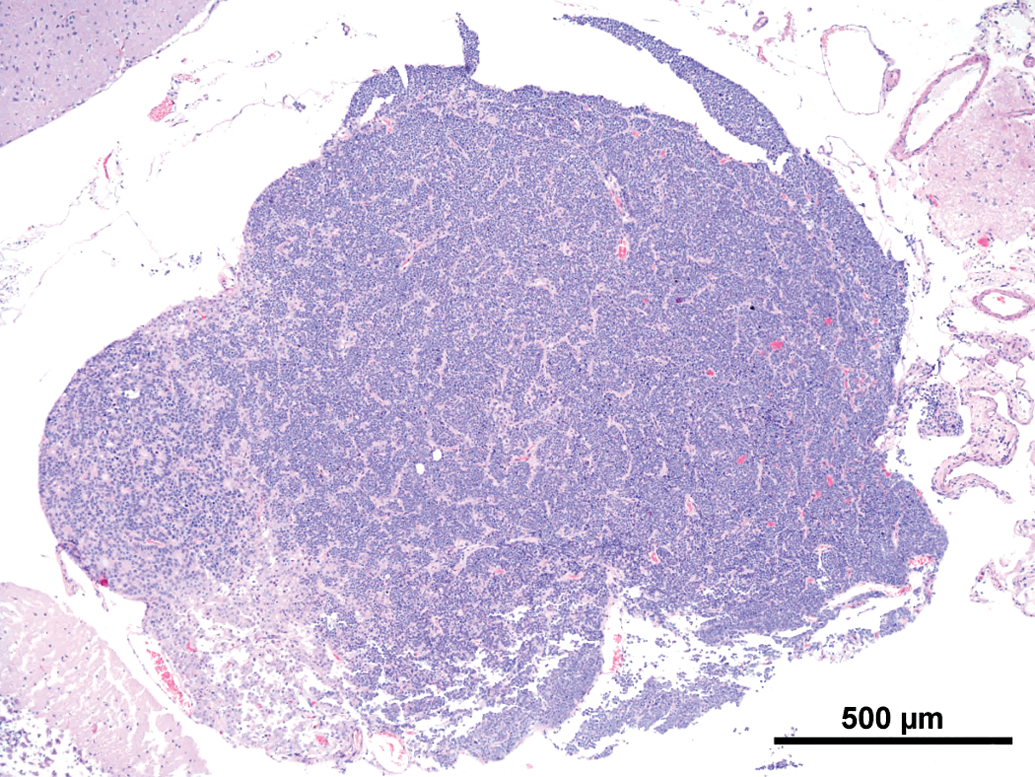
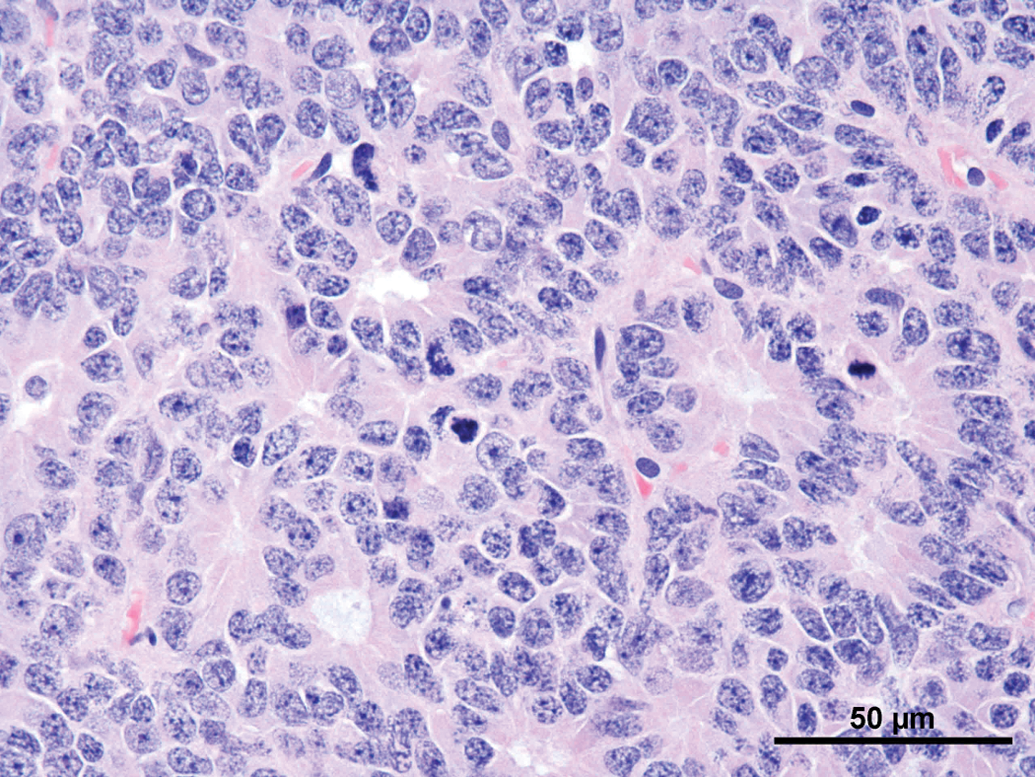
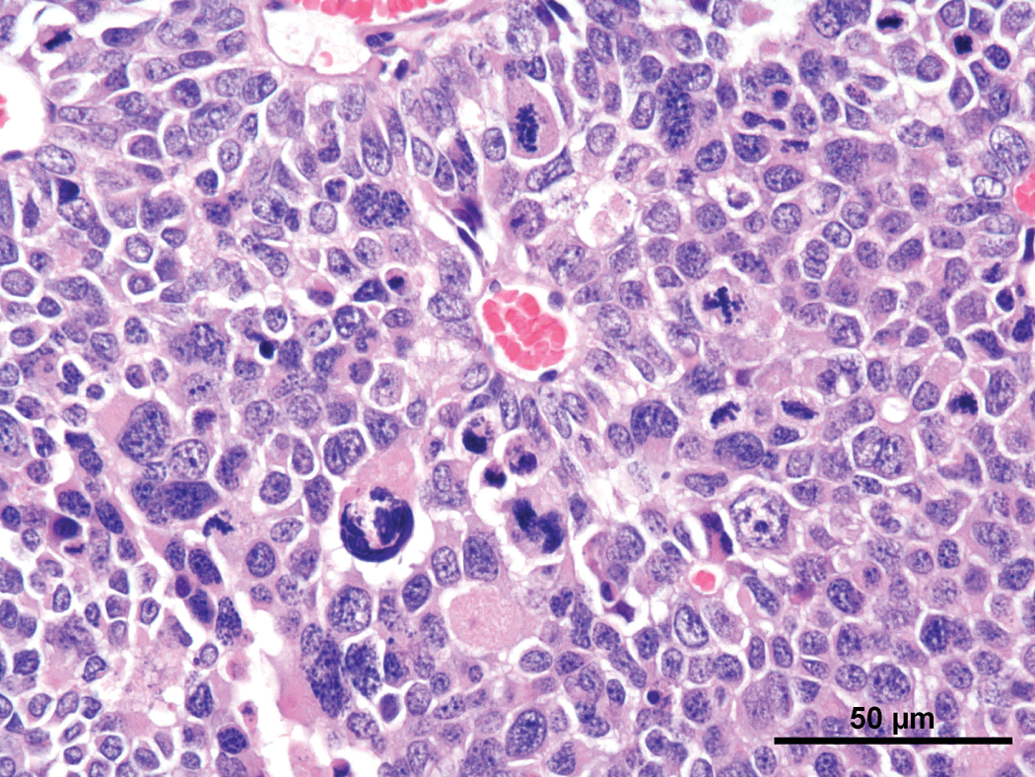
Common features observed in tumors of the current as well the older studies were (not every feature was present in every tumor) as follows: Compression and/or invasion of the adjacent brain tissue and the ventricles (Figures 2 and 6). Distant metastasis was not observed. In 1 case, infiltration into the meninges occurred. Only 1 tumor stayed within the original borders of the pineal gland (Figure 4). The growth pattern consisted mainly of solid sheets, occasionally showing palisading with some tumors forming papillary growth patterns. Focally, the formation of Homer Wright rosettes, Flexner-Wintersteiner rosettes, and perivascular pseudorosettes could be observed (Figures 3 and 5). The tumors were moderately to highly vascularized and often showed areas of necrosis and/or hemorrhage (Figures 2 and 6). The tumor cells were moderately pleomorphic and consisted mainly of large, polyhedral cells with abundant light eosinophilic cytoplasm separated by small amounts of delicate fibrovascular tissue. The nuclei were moderately to highly pleomorphic, round to oval, vesicular with fine to coarsely clumped chromatin and 1 to 3 prominent nucleoli (Figure 5). Frequently giant or multinucleated cells were seen scattered throughout the tumor (Figure 7). The mitotic activity was in general high and ranged from 6 to 25 mitotic figures per high power field.
Rat, study I, pineal gland, malignant pinealoma, mass protruding in between and invading the brain with focal areas of necrosis and hemorrhage. H&E, 4×. Higher magnification of Figure 2, large, monomorphic tumor cells with large vesicular nuclei with finely stippled chromatin and one to three nucleoli. Focal necrotic area, numerous mitotic figures and formation of a pseudo rosette (arrow). H&E, 40×. Rat, study I pineal gland, malignant pinealoma, this was the most differentiated of all tumors. No protrusion or invasion into the brain. H&E, 4×. Higher magnification of Figure 4, tumor cells forming Homer Wright and Flexner-Wintersteiner rosettes, numerous mitotic figures. H&E, 40×. Rat, study II, pineal gland, lobulated mass invading the brain with large necrotic areas. H&E, 4×. Higher magnification of Figure 6, pleomorphic tumor cells forming pseudo rosettes, very large vesicular nuclei, and high mitotic ratio. H&E, 40×.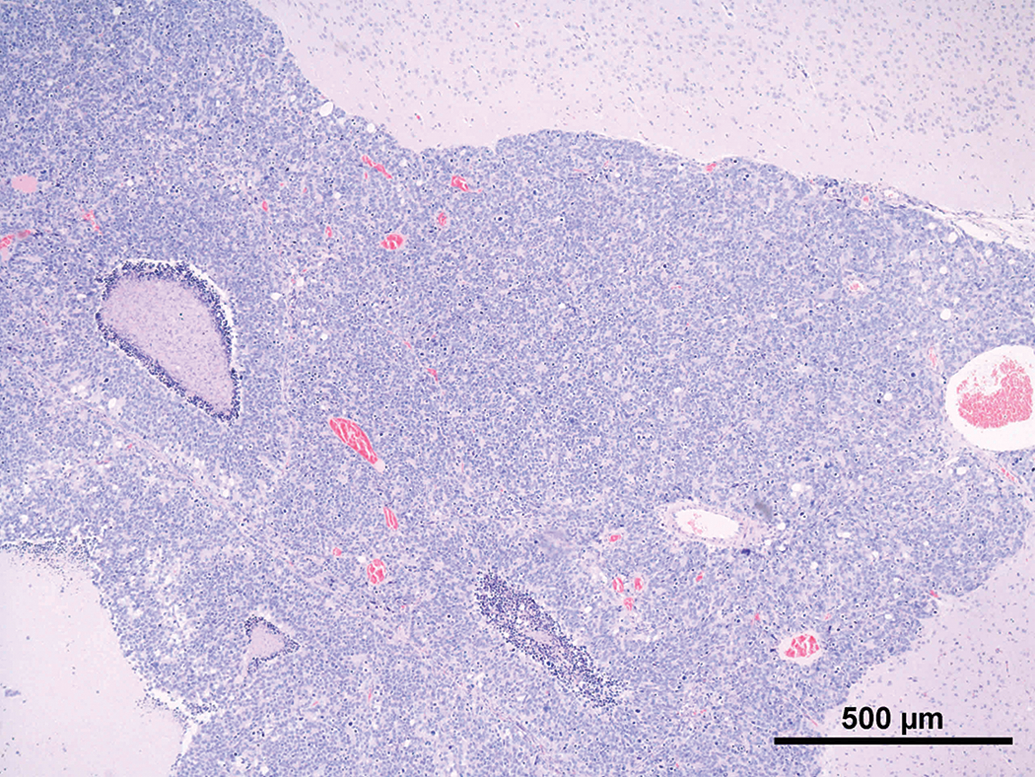
Immunohistochemistry
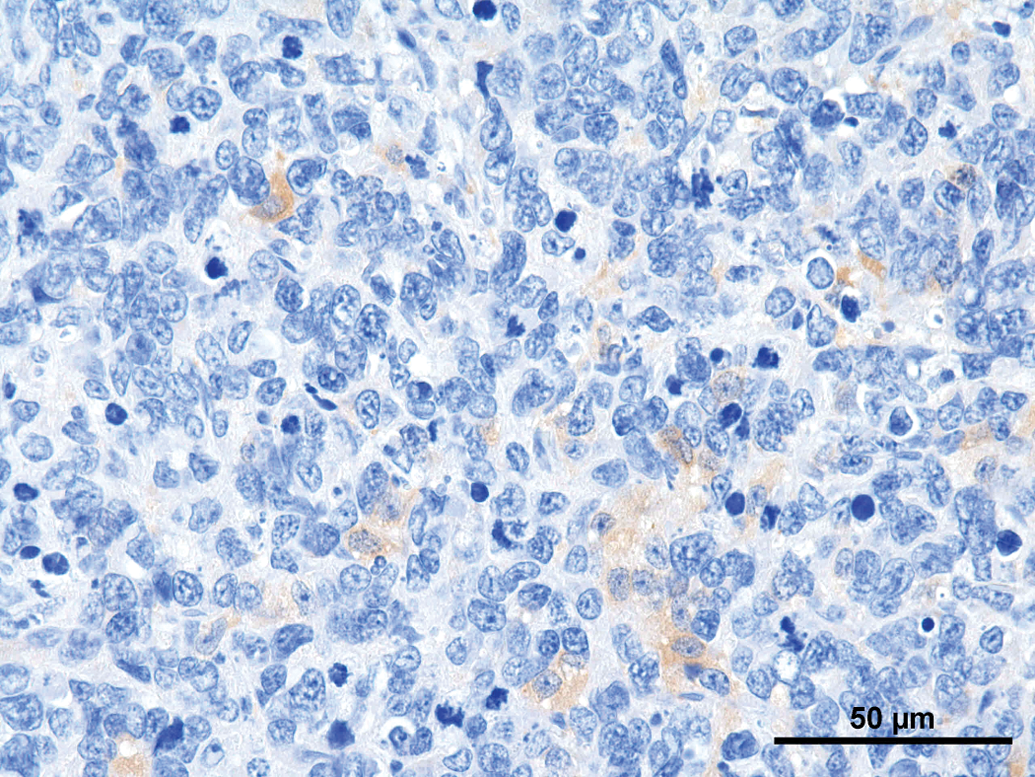
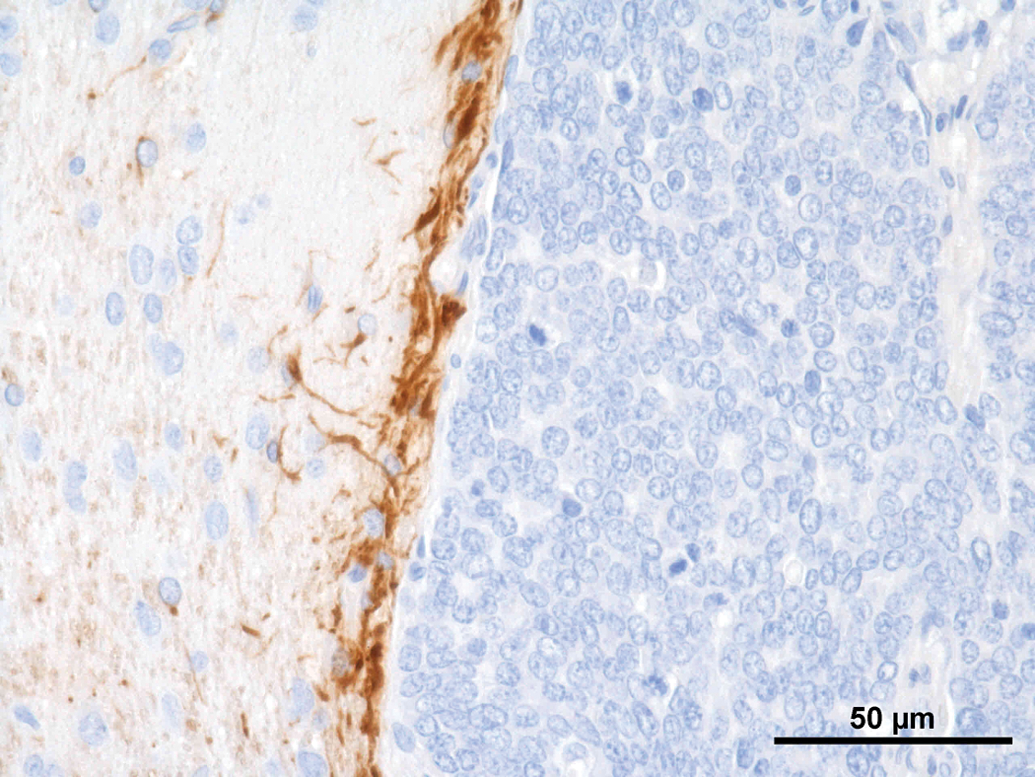
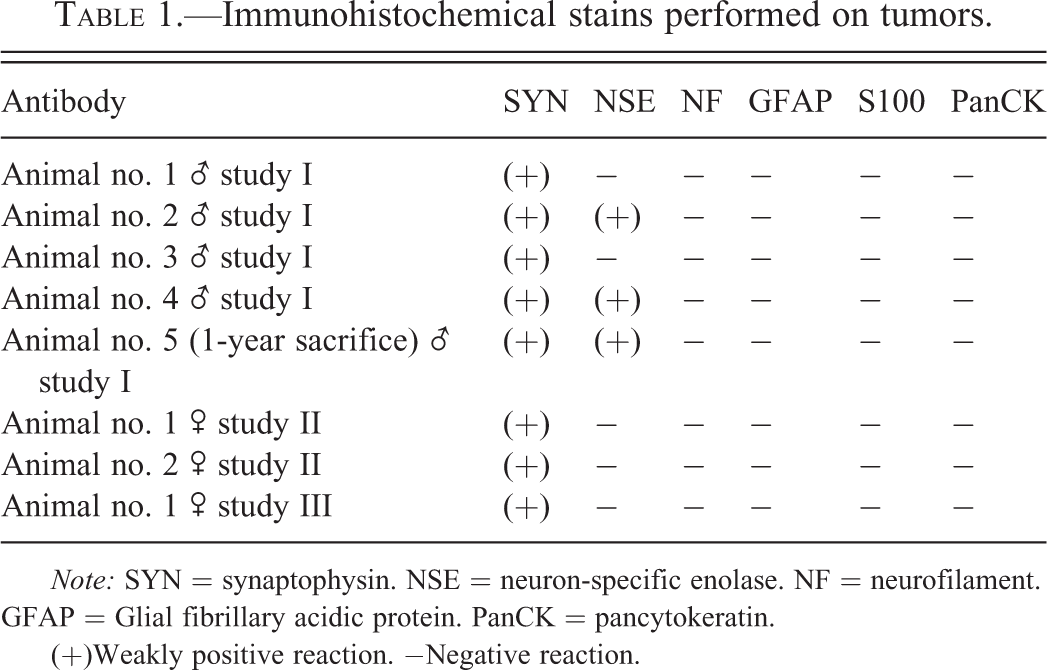
A set of immunohistochemical stains was performed on the different tumors (Table 1). Synaptophysin was weakly positive in all tumors (Figure 8); NSE revealed a weak positive reaction in 3 of the 8 tumors (Figure 9). All other immunohistochemical stains performed were negative. GFAP revealed a strong positive reaction right at the border to the unaltered brain (Figure 10).
Rat, study I, pineal gland, single tumor cells staining positive for synaptophysin. Immunostain against SYN, 40×. Rat, study I, pineal gland, few cells staining positive for neuron specific enolase (NSE). Immunostain against NSE, 40×. Rat, study I, pineal gland, tumor cells stain negative for glial fibrillary acidic protein, but there is a strong positive staining demarcating rim at the border to the brain. Immunostain against Glial fibrillary acidic protein (GFAP), 40×. Immunohistochemical stains performed on tumors.
Note: SYN = synaptophysin. NSE = neuron-specific enolase. NF = neurofilament. GFAP = Glial fibrillary acidic protein. PanCK = pancytokeratin. (+)Weakly positive reaction. −Negative reaction.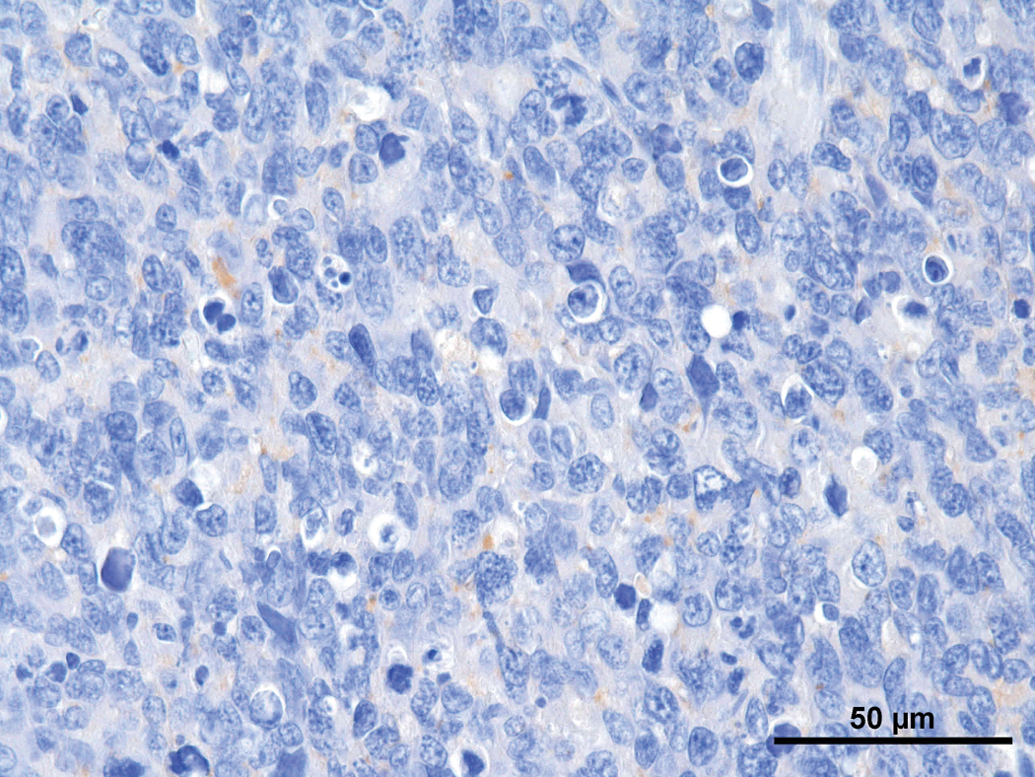
Discussion
Although known to be a rare tumor in rats as well as in humans, pineal gland tumors may appear in notable incidences in carcinogenicity studies. As the pineal gland is not a protocol organ in carcinogenicity studies and therefore only irregularly examined, one should be aware of it as potential origin of neoplasia in the brain. In a combined subchronic/chronic rat study finalized in 2012 at BASF Experimental Toxicology and Ecology in Wistar rats, pineal gland tumors were encountered at an incidence of 1%. The lesions were distributed among almost all test groups and the tumors occurred only in male animals. According to the literature, in laboratory animals, pineal gland tumors are diagnosed as pinealoma, malignant or pinealoma, benign, (Botts et al. 1994) or are referred to as pineocytomas (Al Zubaidy and Malinowsky 1984), whereas the less differentiated pineoblastomas are thought to be nonexistent in rats (Weber et al. 2011). In contrast to the rat, there are much more differentiated diagnostic terms in human pineal gland tumors. In the 2007 revised WHO classification, PPTs were subdivided into well-differentiated pineocytomas, poorly differentiated pineoblastomas, and PPTs with intermediate differentiation (Louis et al. 2007). Pineocytomas were referred to as slow-growing, well-differentiated tumors composed of mature cells resembling pineocytes with large fibrillary pineocytomatous rosettes formed by the tumor cells. Tumor cells show an intensive positive staining for NSE and synaptophysin, positivity for NF is present in most tumors (Jouvet et al. 2000). The pleomorphic variant of pineocytomas contains ganglioid cells with hyperchromatic and bizarre nuclei, but usually no mitotic figures or necrosis are seen (Jouvet et al. 2000). They usually show an intense immunolabelling for NSE and synaptophysin. Pineoblastomas are the most primitive tumors and reveal a highly malignant behavior with high numbers of mitotic figures and often necrosis. Homer Wright and Flexner-Wintersteiner rosettes are often observed but no pineocytomatous rosettes (Sato and Kubato 2009). NSE and anti-synaptophysin immunolabelling was found regularly in the pineoblastomas, but at a lower intensity compared with other PPT. Very few tumor cells were labeled for NF (Jouvet et al. 2000). All tumors have in common a positive GFAP labeling only of resident cells but not the tumor cells themselves. Taking all this information together, the malignant pinealomas from studies performed at BASF Experimental Toxicology and Ecology could be reconsidered to be pineoblastomas according to the WHO classification for humans.
As differential diagnosis, medulloblastoma should be taken into account. Medulloblastomas in rodents are localized within the cerebellum and are highly cellular masses comprised of embryonic neuroepithelial tumor that exhibit mainly neuronal differentiation. These tumors are composed of generally uniform cells resembling the granular cell layer of the cerebellar cortex with round to elongated, hypochromatic nucleus, and prominent nucleoli. The medulloblastomas often show formation of whorls and pseudorosettes. The pseudorosettes may have centrally located fibrillary material. An invasive growth pattern, often replacing cerebellar folia, occur frequently (Gould et al. 1990; Kaufmann 2012; Krinke et al. 2000; Solleveld and Boorman 1990). In our cases, especially the 5 tumors of the recent study looked very alike. The location of tumor growth was always the pineal gland area, the cerebellum was not the main affected area, and in 1 tumor still remnants of pineal gland tissue were observed. This led us to the diagnosis pinealoma, malignant, in combination with the immunohistochemical staining pattern.
Most of the tumors observed at BASF Experimental Toxicology and Ecology from all three studies showed an aggressive behavior with infiltration of the brain and meninges although no distant metastasis was observed. Secondarily, most of the tumors showed focal areas of necrosis and/or hemorrhage. Some tumor cells revealed a high grade of pleomorphism with very large cells containing a giant nucleus with highly stippled chromatin. The mitotic rate was generally high (6–25 mitotic figures per high power field). The tumors revealed Flexner-Wintersteiner rosettes, Homer Wright rosettes, and perivascular pseudorosettes, but no pineocytomatous rosettes, which frequently are observed in benign pineal gland tumors. Furthermore, only single tumor cells were positive for synaptophysin immunolabeling and only 3 tumors revealed positive labeled cells for NSE. All other immunostains performed (pancytokeratin, GFAP, NF, S100) were negative. Considering all these findings together, the tumors were classified as malignant pinealomas according to current diagnostic criteria for laboratory animals (Botts et al. 1994). These rodent tumors should be considered to be pineoblastoma based on the similarity to the tumor described in man (Sato et al. 2009).
In 2005, the Strategic and Regulatory Policy Committee of the American Society of Toxicologic Pathology has started the initiative “Revision of Standardized Nomenclature for Lesions in the Rat and Mouse” (http://www.goreni.org/inhand_v1_nov_21_2005.pdf). The INHAND initiative is currently working on the revised article for lesions in the pineal gland. Until now, pineoblastomas are subsumed under the diagnosis “pinealoma, malignant.” A more precise diagnosis can be made on neoplastic lesions in rodent pineal gland. Pineoblastoma could be used as a subtype of malignant pinealoma.
Due to the fact that there was quite a high incidence of pineal gland tumors (1%) in a recent combined chronic/carcinogenicity study (study I), a genetic background was speculated. Drift of spontaneously occurring tumors in rats is a well-known phenomenon. It is noted in all strains of laboratory rats and affects approximately 60% to 70% of the guideline organs investigated in carcinogenicity studies (Tennekes et al. 2004a , 2004b). According to recent work, epigenetic changes may also occur due to environmental stress factors such as social environment, and dissolved organic matter concentrations, nutrition or water deficiency, grazing, light, or temperature alterations. Moreover, genetic drift can also play an important role in the spontaneous occurrence of (rare) tumors (Kleinjans and van Ravenzwaay, 2014; van Ravenzwaay, 2014). The last 5 factors should not play a role in animal studies performed under standardized conditions, but the epigenetic influence on tumor development should be kept in mind when evaluating data from animal studies.
No information could be received from the breeder about the relationship of the single animals. In addition, we tried to perform DNA extraction for detecting genetic relatedness of the affected animals. So far, it was not possible to isolate DNA that would be of adequate quality for relatedness testing, as only formalin fixed tissue is at our disposal. Until this problem is solved, the familial background for these lesions must stay speculative. A similar occurrence of nontreatment related pineal gland tumors in Wistar Han rats (spontaneous occurrence at 9/960 animals) have been observed by Miller and colleagues in their study in Wistar Han rats exposed for up to 30 months to exhaust from a modern heavy-duty diesel engine, compliant with U.S. emissions 2007 standards; the occurrences of these tumors was not related to the exposures (R. A. Miller, EPL, Inc., personal communication, October 8, 2014).
In our view, the fact that in the recent study performed at BASF Toxicology and Ecology such a high incidence of pineal gland tumors that was not regarded to be treatment related (no dose–response relationship, the most differentiated tumor was observed in the high-dose group, pinealomas occurred in the older studies also in the control group), the tumors very much alike to each other with regard to their histopathologic appearance, and revealed similar immunohistochemical staining patterns, a familial background for these lesions must be considered to be possible. Even more as other pathologists (R. A. Miller, EPL, Inc., personal communication, October 8, 2014) observed similar incidence of pineal gland tumors within the same rat strain, which was not treatment related, points toward a genetic background for susceptibility for pineal glands in this strain.
In conclusion, pineal gland tumors may occur in quite reasonable numbers within 1 study (up to 1%, observed at BASF), although known from the literature to be a very rare tumor. The tumors detected in our study revealed a high concordance with the described WHO criteria for pineoblastomas in humans. The pinealblastomas will be covered in the recent INHAND manuscript under “pinealoma, malignant.” Still it could be considered whether the subtype “pineoblastoma” could be included for further characterization of this variant of pineal gland tumors. Known to be a rare tumor in rats, pineal neoplasms should be included in the list of possible differential diagnoses for brain tumors, especially when the tumor is located in the region of the pineal body.
Footnotes
Author Contribution
Authors contributed to conception or design (ST, SG); data acquisition, analysis, or interpretation (ST, RB, JE, BV); drafting the manuscript (ST); and critically revising the manuscript (RB, SG, JE, BV). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
