Abstract
Microscopic evaluation of the types of cells present in vaginal smears has long been used to document the stages of the estrous cycle in laboratory rats and mice and as an index of the functional status of the hypothalamic–pituitary–ovarian axis. The estrous cycle is generally divided into the four stages of proestrus, estrus, metestrus, and diestrus. On cytological evaluation, these stages are defined by the absence, presence, or proportion of 4 basic cell types as well as by the cell density and arrangement of the cells on the slide. Multiple references regarding the cytology of the rat and mouse estrous cycle are available. Many contemporary references and studies, however, have relatively abbreviated definitions of the stages, are in reference to direct wet mount preparations, or lack comprehensive illustrations. This has led to ambiguity and, in some cases, a loss of appreciation for the encountered nuances of dividing a steadily moving cycle into 4 stages. The aim of this review is to provide a detailed description, discussion, and illustration of vaginal cytology of the rat and mouse estrous cycle as it appears on smears stained with metachromatic stains.
Introduction
Microscopic evaluation of the types of cells present in vaginal smears has long been used to document the stages of the estrous cycle in laboratory rats and mice and as an index of the functional status of the hypothalamic–pituitary–ovarian axis. As such, assessment of the estrous cycle has been used both as a principal measure in determinations of reproductive cyclicity and as an ancillary test in reproductive toxicologic studies (Goldman, Murr, and Cooper 2007).
The estrous cycle in rats and mice averages 4–5 days and is a repetitive but dynamic process whereby different cell types appear and recede in waves throughout the cycle, reflecting changes in the levels of estradiol and progesterone secreted by the ovarian follicles. The estrous cycle is generally divided into four stages—proestrus, estrus, metestrus, and diestrus. However, depending on the investigator and the objectives of a study, it has been divided into as few as 3 stages—proestrus, estrus, and diestrus (Goldman, Murr, and Cooper 2007); alternatively, the four stages have been subcategorized into as many as 13 stages (Thung, Boot, and Mühlbock 1956). On cytological evaluation, these stages are defined by the absence, presence, or proportion of the cells present as well as by the cell density and arrangement of the cells on the slide.
Vaginal cytology can be evaluated immediately after collection as an unstained, wet mount preparation (direct cytology) or as a fixed and stained slide preparation. Although some of the discussion can be applied to the evaluation of direct wet mount preparations, this review will focus on the estrous cycle as evaluated on slide preparations that are dry-fixed and stained with metachromatic stains.
Multiple references regarding the cytology of the rat and mouse estrous cycle are available (Allen 1922; Byers et al. 2012; Caligioni 2009; Cooper and Goldman 1999; Goldman, Murr, and Cooper 2007; Hubscher, Brooks, and Johnson 2005; Long and Evans 1922; Mandl 1951; Organization for Economic Cooperation and Development [OECD] n.d.; Young, Boling, and Blandau 1941). Many of the contemporary references and studies, however, have relatively abbreviated definitions of the stages, are in reference to direct wet mount preparations, or lack comprehensive illustrations. This has led to ambiguity and, in some cases, a loss of appreciation for the encountered nuances of dividing a steadily moving cycle into 4 stages. The aim of this review is to provide a detailed description, discussion, and illustration of the vaginal cytology of the rat and mouse estrous cycle and give the reader an appreciation for the nuances and challenges in definitively characterizing this dynamic process. For information on ovarian hormone secretion and the subsequent impact on vaginal physiology as well as how the evaluation of vaginal cytology applies to toxicological research, the reader is directed elsewhere (Butcher, Collins, and Fugo 1974; Cooper and Goldman 1999; Goldman, Murr, and Cooper 2007; Kovacic and Parlow 1972; Li and Davis 2007; Nelson et al. 1981; Neill 2006). For a detailed review of the histopathology of the estrous cycle, see Westwood’s (2008) “The female rat reproductive cycle: a practical histological guide to staging.”
Historical Perspective
The term estrus
1
was first used (as oestrus
2
) in Heape’s (1900) publication, “The ‘sexual season’ of mammals and the relation of the ‘pro-oestrum’ to menstruation,” in which he defines estrus as the “special period of sexual desire in the female.” Estrus is a Latin adaptation of the Greek word oistros, which means gadfly, frenzy, or madness. It is from this base word that Heape named and defined the other stages of the mammalian estrous cycle: proestrus, an animal coming into heat; metestrus, a short period in the absence of conception, when “the activity of the [reproductive] organs gradually subside during a definite period”; diestrus, a period of short rest (during the breeding season); and anestrus, a period of rest (in the nonbreeding season). At that time, the stages were defined and based on changes in behavior (e.g., the acceptance of copulation) as well as gross morphological and histological changes observed in the reproductive tract of cycling females. Included in these were the macroscopic changes in the vulva (e.g., swelling), vaginal excretions (e.g., hemorrhage, mucous), and uterus (e.g., congestion) together with the microscopic changes in the uterine tissue and ovaries. However, the macroscopic observations were not always reliable in small rodents, and tissue pathology was and still is an invasive procedure and not feasible for in-life estrous cycle staging. It was the need for a more reliable, noninvasive method for defining the stages and determining the time of ovulation in a laboratory setting that lead to Stockard and Papanicolaou’s (1917) landmark article, “The existence of a typical estrous cycle in the guinea pig with a study of its histological and physiological changes.” It was with this article that, for the first time in any species, the changes that occur in the vaginal canal during the estrous cycle were thoroughly characterized both macroscopically and microscopically (on histology and cytology). Additionally, these vaginal changes were correlated to the changes that occur in other reproductive organs during the estrous cycle. This publication influenced three other pioneers of reproductive physiology—Joseph Long, Herbert Evans, and Edgar Allen—in their pursuit to study and then describe the rat (Long and Evans 1922) and mouse (Allen 1922) estrous cycles (Figure 1). Long and Evans (1922) eloquently expressed the importance of Stockard and Papanicolaou’s (1917) findings with the following:
Historical representation of select stages of the rat estrous cycle as presented in Long and Evans (1922). These eloquent pencil drawings illustrate proestrus (A), metestrus (B), and diestrus (C). By the fortunate discovery that in the guinea pig these [vaginal] mucosal transformations are accompanied by the dehiscence of epithelial cells so that at times the lumen of the vagina has a characteristic cell content, it has been possible for Stockard and Papanicolaou to show us that we may discover with ease in the living animal the exact occurrence and progress of these cycles. (p. 6)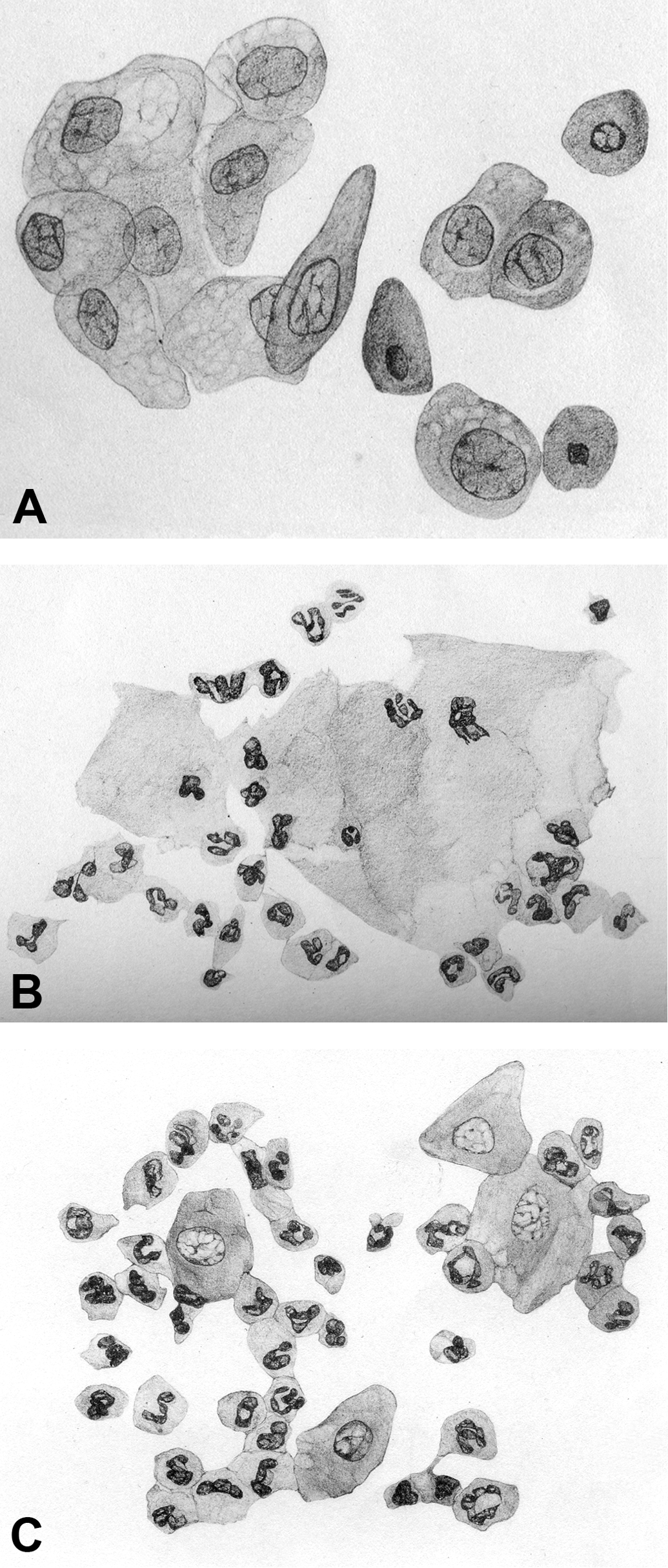
And so, it is from these early twentieth-century publications that the present-day definitions of proestrus, estrus, metestrus, and diestrus arose.
It would be a great disservice not to take this opportunity to state that the abovementioned scientists were brilliant researchers and made many other significant contributions to the fields of endocrine and reproductive physiology—laying the foundation for many others working in these fields. The following sentences highlight but a few of their contributions. The Pap smear or test, developed by Papanicolaou and used as a screening test for cervical cancer, is now recognized as the most significant advancement in the control of cancer in the twentieth century (Vilos 1998). In addition to developing the Long–Evans stain of rat, Evans and colleagues went on to characterize the hormones of the anterior pituitary (e.g., luteinizing hormone and follicle-stimulating hormone) and define interactions between this gland, the ovary, and uterus (Lindsey and Baker 2006). Allen and Edward Doisy isolated the ovarian estrogenic hormone (Wells 1945). Additionally, Allen and associates also investigated the effects of estrogenic and androgenic hormones on various reproductive and endocrine organs as well as the relationship of estrogen concentrations to certain types of neoplasms (e.g., leukemia, mammary cancer; Wells 1945).
Methodology and Technical Considerations
To elucidate the length and, subsequently, the stages of the estrous cycle, vaginal cytology samples are collected over at least 14 consecutive days (Cooper and Goldman 1999). Vaginal cytology samples can be collected at any time of the day, but the practice of most laboratories is to collect them early in the morning after the lights are turned on. Regardless of when samples are collected, it should be done at approximately the same time of the day over the course of the collection period to reduce variability. Collection of the samples can be done either by vaginal lavage or swabbing. Lavage typically yields a higher cellularity sample and is the preferred method of the authors. Regardless of the methodology employed, accurate interpretation of vaginal cytology samples is dependent on the quality of the sample preparation (Figure 2).
Several examples of poor quality smears. In the first example (A), the smear is understained, resulting in a lack of cellular detail. Additionally, some cells are ruptured resulting in a background of scattered cellular debris. Due to the poor quality, this smear was ultimately deemed uninterruptable. Plate B has a background of eosinophilic streaming and debris (arrows), which most likely represents ruptured or disintegrated neutrophils. Depending on full evaluation of the smear and the experience of the evaluator, this smear could be misclassified as proestrus (instead of diestrus) or could be deemed an insufficient sample. Plate C has a large amount of eosinophilic material in the background, making visualization of the neutrophils that are present challenging. The background material either was due to poorly maintained lavage fluid or is stain precipitate due to the poorly maintained staining solutions. Modified Wright–Giemsa stain (A, B) and Toluidine blue O stain (C). Original objective magnification of 10× (A) or 20× (B, C).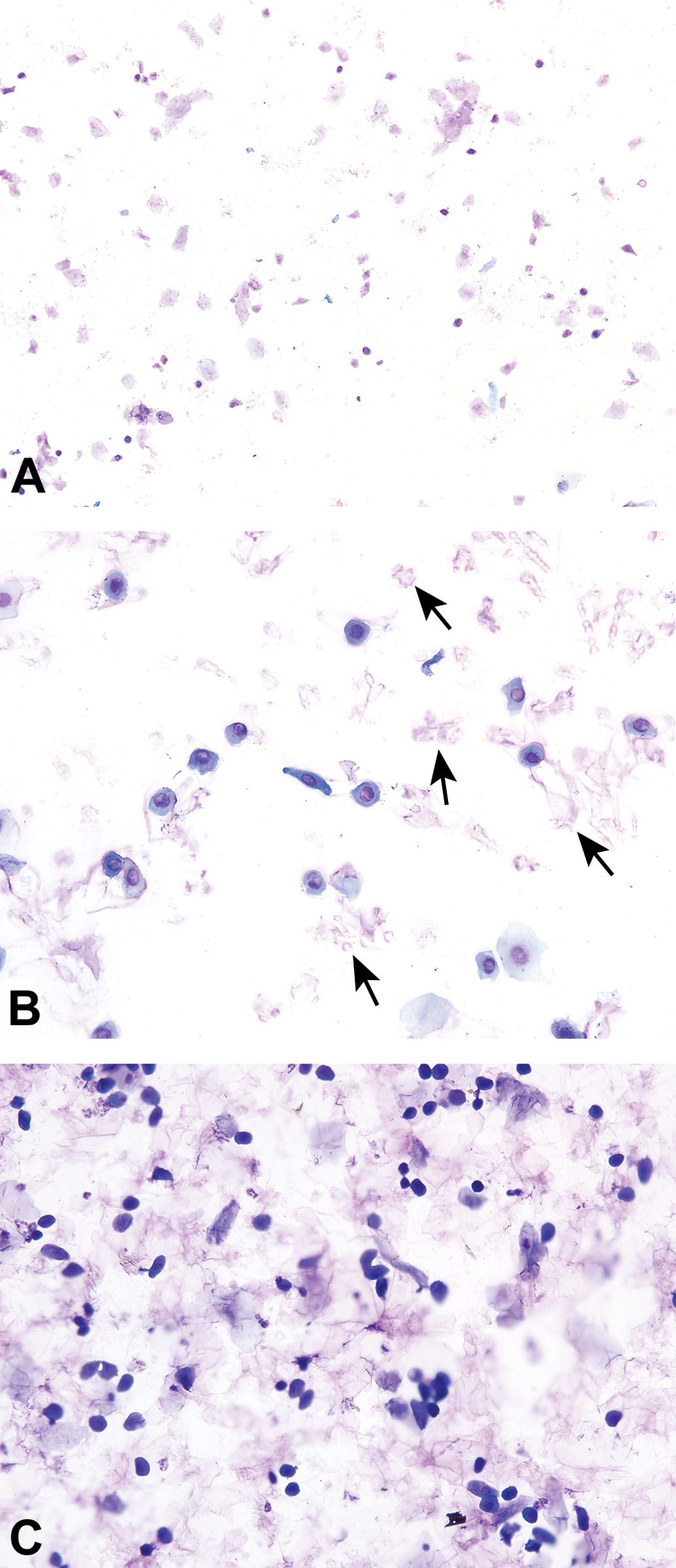
Materials
To perform vaginal lavage, one needs a pipette or eyedropper, saline, and glass slides. The pipette tips or eyedroppers need to be smooth and tapered with a recommended 1.5 mm internal tip bore for pipette tips (OECD n.d.). It is a best practice to use a new tip or dropper for each animal. If a single tip or dropper is to be used on more than 1 animal, it should be thoroughly rinsed with fresh distilled or sterile water between animals. Thorough rinsing is very important to prevent between-animal sample contamination that may lead to inaccuracies in staging or, rarely, infection of the reproductive tract.
Phosphate-buffered saline or normal saline are the preferred fluid to use for vaginal lavage. Some publications also list distilled water or tap water as options, and many research studies have used these in lieu of saline. Saline is an isotonic solution while water is hypotonic. The use of water can cause distorted epithelial cell morphologies and will invariably rupture neutrophils, which can lead to confusion or inaccuracies in the evaluation of vaginal cytologies. Water has seemingly been successfully used when cells are evaluated on direct preparations that are evaluated shortly after collection. Nevertheless, to avoid any issues with cell morphology and for the purposes of preparing stained slides, saline should be used.
For archiving purposes and ease of sample evaluation, it is advantageous to place 6–8 consecutive samples from 1 animal onto a basic 1 × 3 inch glass slide. Grids that form “boxes” or “squares” representing each day may be drawn on the slides with a black, solvent-resistant marker. Alternatively, glass slides with pre-drawn and numbered grids representing 8 days are also available at an additional cost (Figure 3). Some investigators use wax pencils to draw the grids on glass slides. An advantage of using a wax pencil is that the wax forms a barrier that will prevent 1 day’s sample from “bleeding” into another day’s sample. However, if an automated coverslipping machine is used to process the slides, the wax markings can lead to an irregular slide surface that may result in an uneven distribution of permount or prevent direct contact between the slide and coverslip, resulting in air bubbles and inadequate preservation. With care and practice, slides can be easily and successfully made without the use of a wax pencil. If wax pencils are to be used, removal of the wax by soaking the slides in xylene (or equivalent) prior to coverslipping with an automated machine is recommended.
Example of a commercially available 1′′ × 3′′ glass slide with a predrawn numbered grid for vaginal cytology sample collection. This slide contains samples that were collected daily for 8 consecutive days from an individual animal and, after dry fixation, were stained with a metachromatic stain.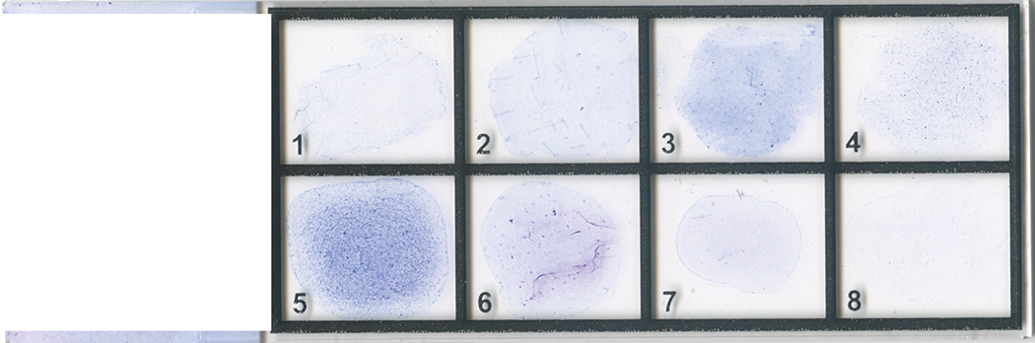
Collection and Processing
To collect cells from the vaginal canal, approximately 0.1 ml (mice) or 0.2 ml (rats) of saline is drawn into the pipette or dropper. The tip of the dropper is gently inserted into the vaginal orifice at a depth of approximately 1–2 mm in mice and 5–10 mm in rats and then the saline flushed into the vagina and back out 2 or 3 times. If after the first flush the fluid is cloudy, subsequent flushings may not be necessary (OECD n.d.). When inserting and flushing into the vaginal orifice, care must be taken not to insert the tip too deep so as to avoid cervical stimulation. Excessive stimulation can induce pseudopregnancy, which appears as a persistent diestrus for up to 14 days (Goldman, Murr, and Cooper 2007).
After the lavage, a small drop of the sample is then placed evenly on the slide in a thin layer (smear) and allowed to air-dry. Crystals may form on the dry unstained slides; these will be removed as part of the staining process. Several different metachromatic stains may be used to stain the dry fixed slides. Romanowsky-type stains (e.g., Modified Wright’s, Wright’s Giemsa) and Toluidine blue O are the commonly used cytological stains offering good coloration for the identification of the different cell types and are recommended for staining vaginal smears. It is important to avoid heavy staining and to rinse the slides thoroughly. Dry fixed slides do not require a spray fixative (e.g., Spray-CyteTM) prior to staining. A spray fixative is used when a stain requires wet fixation (i.e., application of the spray fixative onto the sample before the cells air-dry), such as is needed with the Papanicolaou stain.
Papanicolaou stain has also been used to stain vaginal cytology samples, especially in humans as a screening procedure for cervical cancer (i.e., Pap smear or test). This stain is multichromatic involving the use of 5 different dyes and requires a wet fixation of the sample before staining. Use of this stain offers better chromatin detail and stains the cytoplasm with shades of orange, pink, or blue, depending on the degree of cornification. This level of detail, however, is often not needed to identify the stages of the estrous cycle in rodents.
Slide Evaluation
In general, cells can be identified using a 10× objective; however, in some cases the use of higher objectives may be needed (e.g., to better visualize neutrophils). Care should be taken to examine the whole smear as cell types and numbers may vary in different areas on the slide. Evaluating daily smears with knowledge of the preceding or succeeding days’ stages (vs. “blindly” reading each day) is important for the accurate interpretation of the stages and cycling status because it puts each day into context. This is of particular importance when a smear is a transition between two consecutive stages (transitional stage) when it may be difficult to make an accurate determination without knowledge of the preceding or succeeding day’s smears. For example, in a rat, a smear transitioning from proestrus to estrus will have combinations of round nucleated cells and keratinized anucleated cells. Knowing the previous day’s findings will help one to identify the day in question as proestrus rather than late estrus, where similar cell types may be observed.
For the best viewing of stained cytology preparations, it is important that the microscope is properly configured for Köhler illumination. In laboratories where wet mount preparations or unstained specimens are evaluated, the microscope condenser and field diaphragm are usually adjusted for greater contrast with a resulting loss in image illumination and resolution; with these types of adjustments the microscope does not have proper Köhler illumination. When proper Köhler illumination is applied, a stained cytology image will have an even illumination with optimal resolution and contrast and minimal artifact or glare. Adjusting a microscope for Köhler illumination is relatively simple. The reader is directed elsewhere for details on Köhler illumination (Rottenfusser 2013).
One of the single most important factors in the evaluation of vaginal cytologies is proper training and experience
The evaluator needs to be aware of the inherent variations that may be encountered, be able to recognize artifactual changes (e.g., balled-up or ruptured neutrophils, understained cells), and understand that the smears represent a moment in time of a dynamic process and that the total impression of the smear is more important than the exact cell counts. Practice slide sets, whereby the trainee follows groups of individual animals for several cycles, is the best way to develop proficiency in reading vaginal smears. To reduce variability and ensure consistency, it is also important to limit the number of people evaluating the slides for a given study or within a given laboratory and to make certain there is agreement on the criteria for the classification of each stage. This is particularly important in the inevitable event of transitional stages, when the evaluation is prone to increased subjectivity despite established objective criteria. If more than one observer is involved in reading smears, discrepancies can be reduced by initially having them simultaneously evaluate the same slides to ensure consistency of stage interpretation (Cooper and Goldman 1999).
The Cells and Stages of the Estrous Cycle
As previously stated, estrous cycle length averages 4–5 days in both rats and mice, but occasional 6-day cycles may be observed in some individuals (Nelson et al. 1982; Mandl 1951; Morrissey et al. 1988; Long and Evans 1922; Goldman, Murr, and Cooper 2007; Parkes 1928; Blandau, Boling, and Young 1941). Compared to rats, mice have less regularity in cycle length or cycle contiguity; that is, the likelihood of a 4-day cycle being followed by a 5-day cycle instead of another 4-day cycle is higher in mice (Nelson et al. 1982). Many factors influence cycle length including light, age, temperature, noise, nutrition, stress, and social relationships (Li and Davis 2007; Nelson et al. 1981; Campbell, Ryan, and Schwartz 1976; Campbell and Schwartz 1980; Nelson et al. 1982; Whitten 1956; Goldman, Murr, and Cooper 2007). For example, mice housed singly cycle more regularly and about 1 day shorter than mice housed in multiples (Nelson et al. 1982). Female rodents cycle more regularly when males are present in the room (i.e., the Whitten effect), although mice are more sensitive to this phenomenon than rats (Whitten 1956; OECD n.d.).
The rodent estrous cycle is very sensitive to changes in the light–dark cycle. A 12:12 light–dark cycle is standard in most laboratories. In laboratories using a 14:10 light–dark cycle, a different ratio in the number of 4- and 5-day cycles may occur in comparison to a 12:12 cycle (OECD n.d.).
The length of the four stages varies between 6 and 72 hr depending on the stage and individual rodent, therefore, some short stages may be “missed” especially if samples are collected very early or late in the day. For example, proestrus, with an average length of 14 hr in rats, could be missed with an early morning sample collection because in many females proestrus does not start until midmorning and would end before the next collection. Sample collection can be shifted to late morning or early afternoon in an effort to minimize the incidences of missed stages.
Cell Types of the Estrous Cycle
For this discussion, cell descriptions are based on cytology preparations that were dry fixed and stained with a metachromatic stain.
Neutrophils
Neutrophils are also known as leukocytes or polymorphonuclear cells. These cells are round, very small, and possess multilobulated nuclei (Figure 4). During collection and preparation, these cells may condense or “ball-up,” making visualization of their nuclei difficult, but with experience, they can still be recognized as neutrophils (Figure 5A). If at low power cells are suspected to be condensed neutrophils, the magnification can be increased to a 20× or 40× objective. Usually on higher magnification, condensed neutrophils can be identified as such and neutrophils with a more normal appearance can sometimes be found on the periphery of the smear. Neutrophils are relatively delicate cells and can sometimes rupture during collection or processing (Figure 5B and C). It is important to recognize the appearance of ruptured neutrophils to avoid misinterpretation of a particular stage.
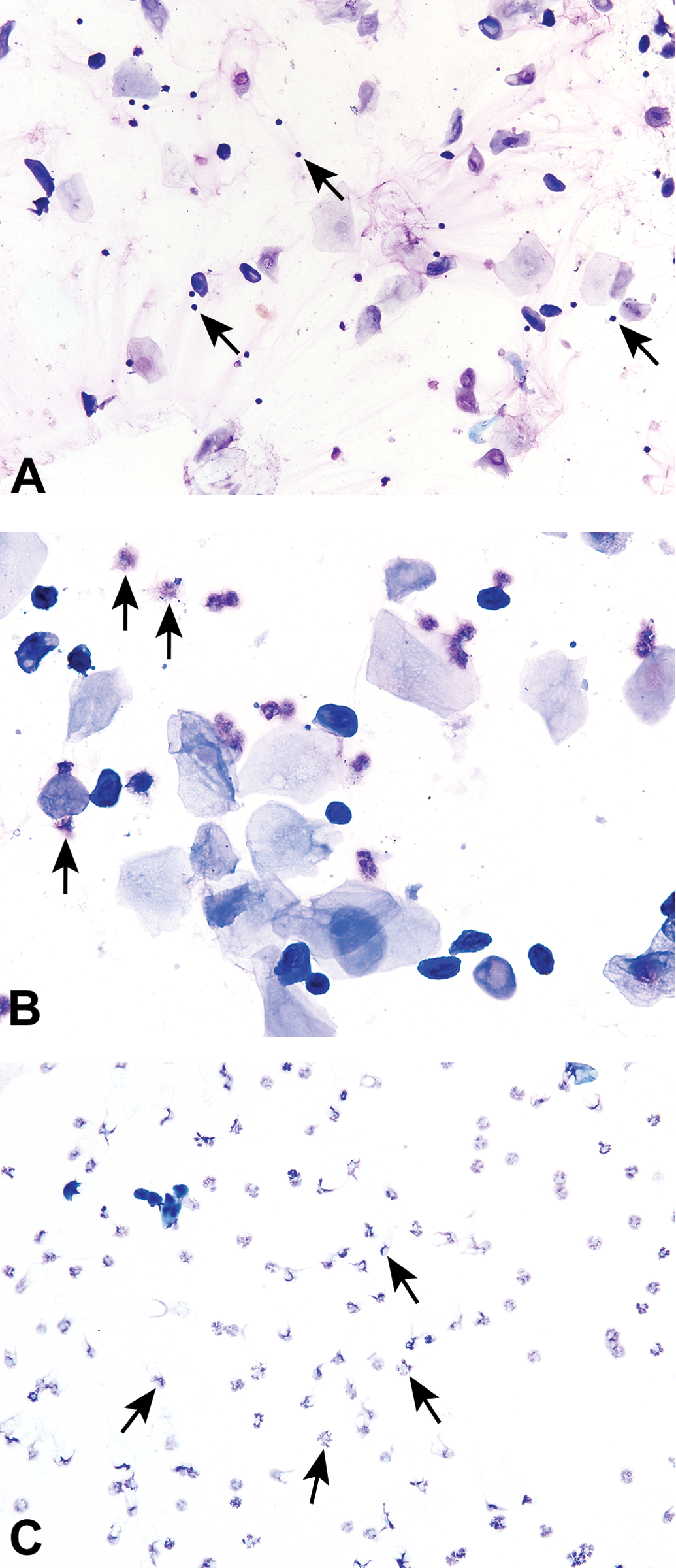
Many neutrophils (arrows), characterized by their very small size and multilobulated nuclei, and two large nucleated epithelial cells (arrowhead) are present on this diestrous vaginal smear from a Sprague-Dawley rat. Toluidine blue O stain. In Elmore et al. Proceedings of the 2014 National Toxicology Program satellite symposium. Toxicol Pathol, 43:10-40, 2015. Several condensed or “balled-up” neutrophils (A, arrows) are interspersed among the epithelial cells of a diestrous smear from a Sprague-Dawley rat. The pale pink background is due to the presence of mucous. Plates B and C demonstrate ruptured neutrophils (arrows) from vaginal cytology smears of a Sprague-Dawley rat (B) and B6C3F1/N mouse (C). Modified Wright–Giemsa stain (A) and Toluidine blue O stain (B, C). Original objective magnification of 20× (A, B) or 40× (C).
Small Nucleated Epithelial Cells
These cells are small, round to oval, and nonkeratinized (Figure 6). They have a high nuclear to cytoplasmic (N:C) ratio relative to large epithelial cells, a round nucleus, and blue cytoplasm. They may stain very dark or basophilic, precluding visualization of the nucleus. Small cytoplasmic vacuoles may be seen in occasional small epithelial cells of proestrus.
Small nucleated epithelial cells from a proestrous vaginal smear of a Sprague-Dawley rat. Occasional nucleated epithelial cells containing small cytoplasmic vacuoles (arrow) and a few anucleated epithelial cells are also present. Toluidine blue O stain. Original objective magnification of 40×.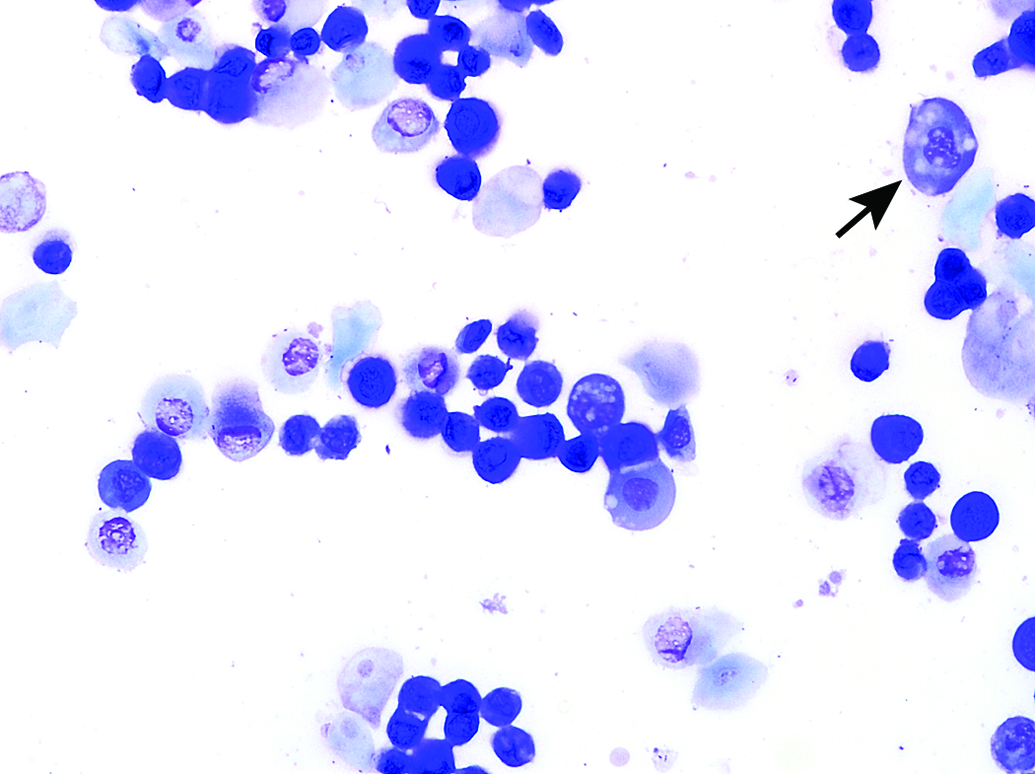
Large Nucleated Epithelial Cells
These cells are larger with moderate to abundant amounts of blue to sky blue cytoplasm and a lower N:C ratio in comparison to small epithelial cells (Figures 4 and 7). They are round to polygonal and may have irregular, jagged, or angular borders. Large epithelial cells can have some degree of keratinization and possess nuclei that may be intact, degenerate, or pyknotic. Occasionally, large nucleated epithelial cells will contain finely stippled cytoplasmic granules (Figure 8).
Several large nucleated epithelial cells (arrows) are intermixed with anucleated epithelial cells (arrowheads) in a vaginal smear of late estrus from a Sprague-Dawley rat. Numerous bacteria are observed adhered to many of the epithelial cells. Modified Wright–Giemsa stain. Original objective magnification of 40×. In Elmore et al. Proceedings of the 2014 National Toxicology Program satellite symposium. Toxicol Pathol, 43:10-40, 2015. A large nucleated epithelial cell containing many small cytoplasmic granules (arrow) from a Sprague-Dawley rat. Modified Wright–Giemsa stain. Original oil objective magnification of 100×.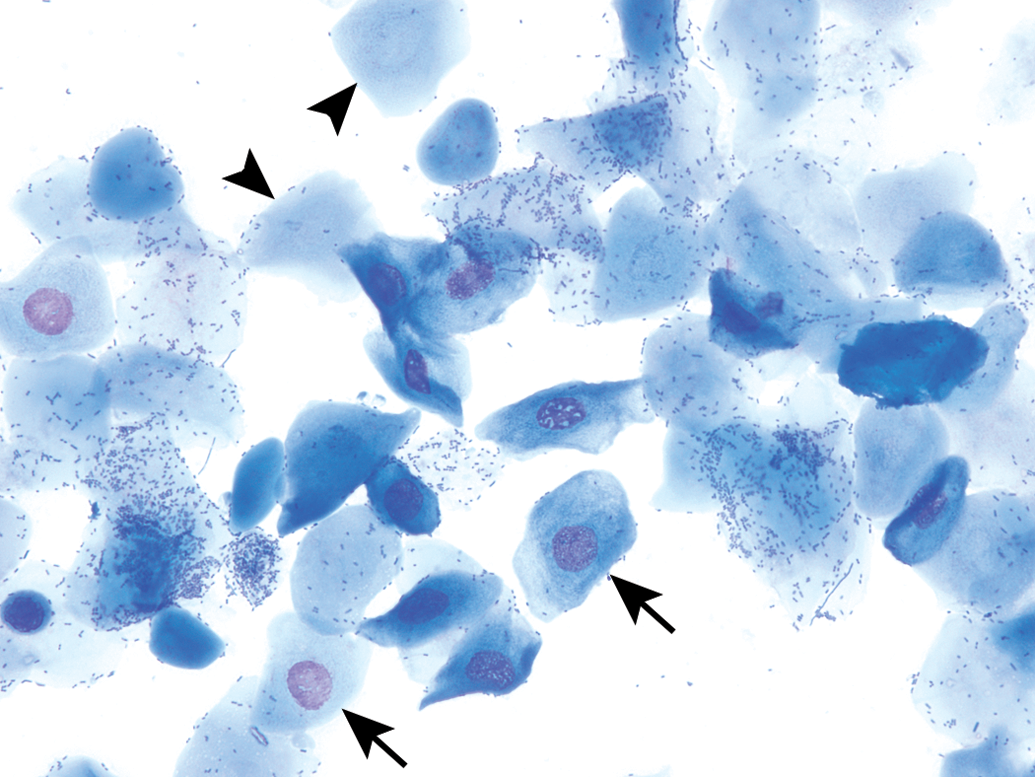
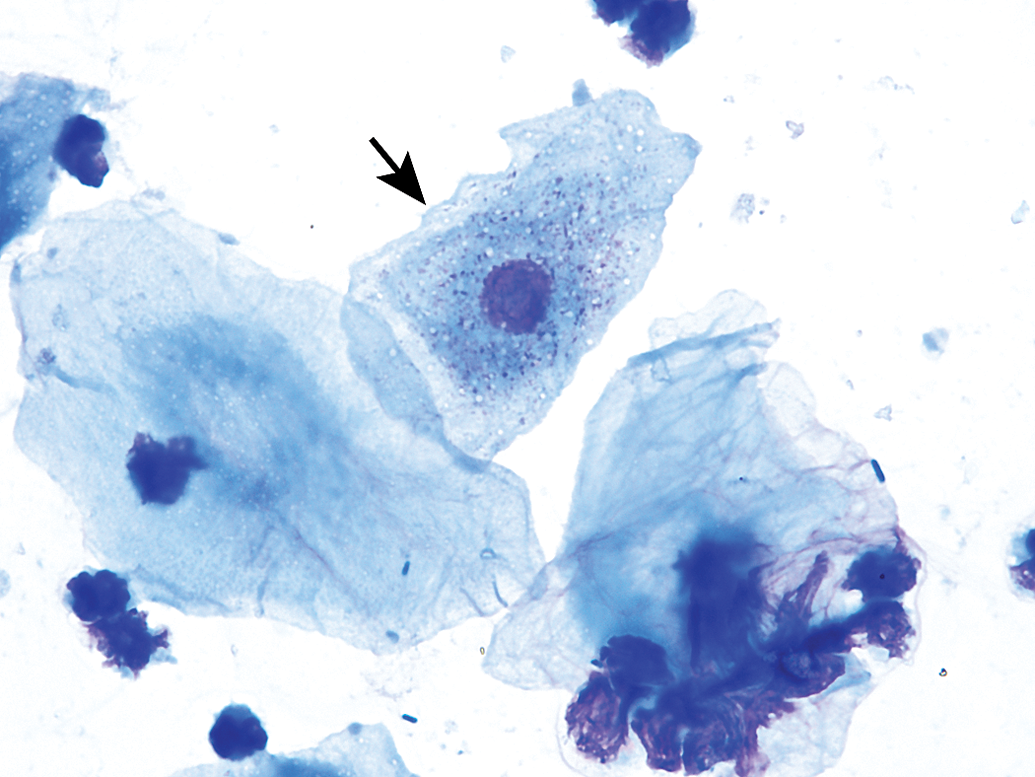
Anucleated Keratinized Epithelial Cells
Anucleated epithelial cells are also known as squames or “cornflakes.” They are aged cells and are characterized by an abundant blue to sky blue cytoplasm with jagged or angular edges (Figures 7 and 9). As the name infers, they lack nuclei, although pale round areas representing where a nucleus had existed (nuclear “ghost”) may be visualized (Figure 9). These epithelial cells can fold onto themselves or break apart, creating jagged elongated structures referred to as “keratin bars” (Figure 10).
Anucleated epithelial cells from a vaginal smear of estrus in a B6C3F1/N mouse. Some cells possess ghost nuclei (arrows). Modified Wright–Giemsa stain. Original objective magnification of 40×. In Elmore et al. Proceedings of the 2014 National Toxicology Program satellite symposium. Toxicol Pathol, 43:10-40, 2015. A keratin bar (arrow) is observed in a vaginal smear of estrus from a Sprague-Dawley rat. Note that many bacteria are either adhered to the cells or free in the background. Modified Wright–Giemsa stain. Original objective magnification of 40×.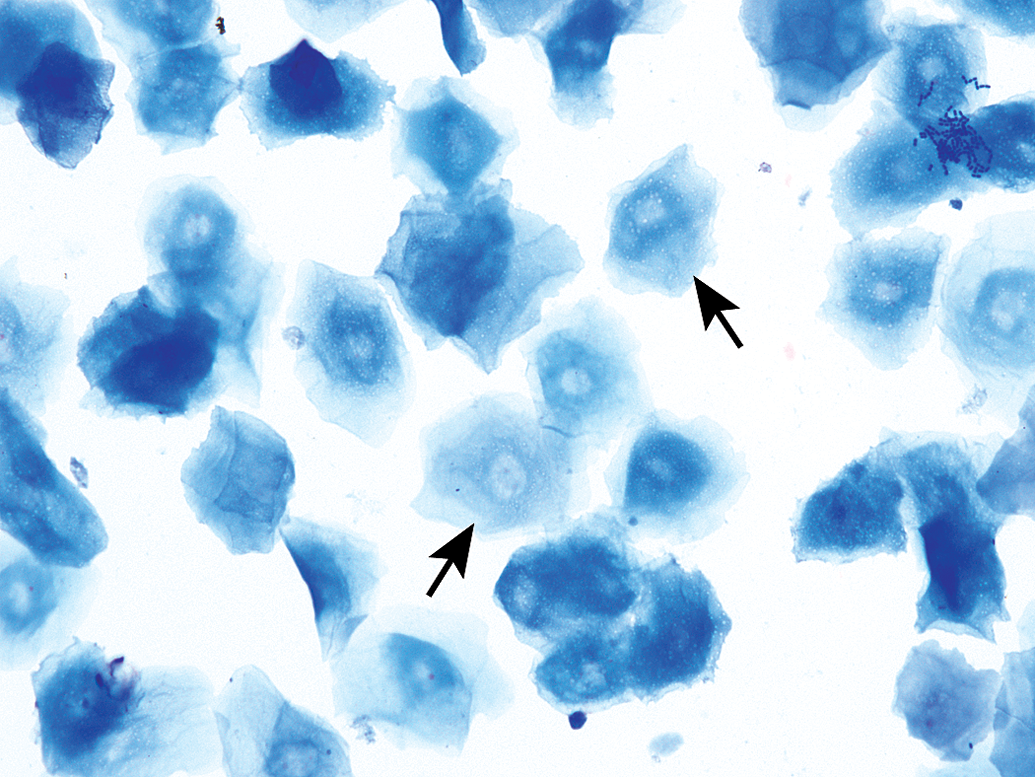
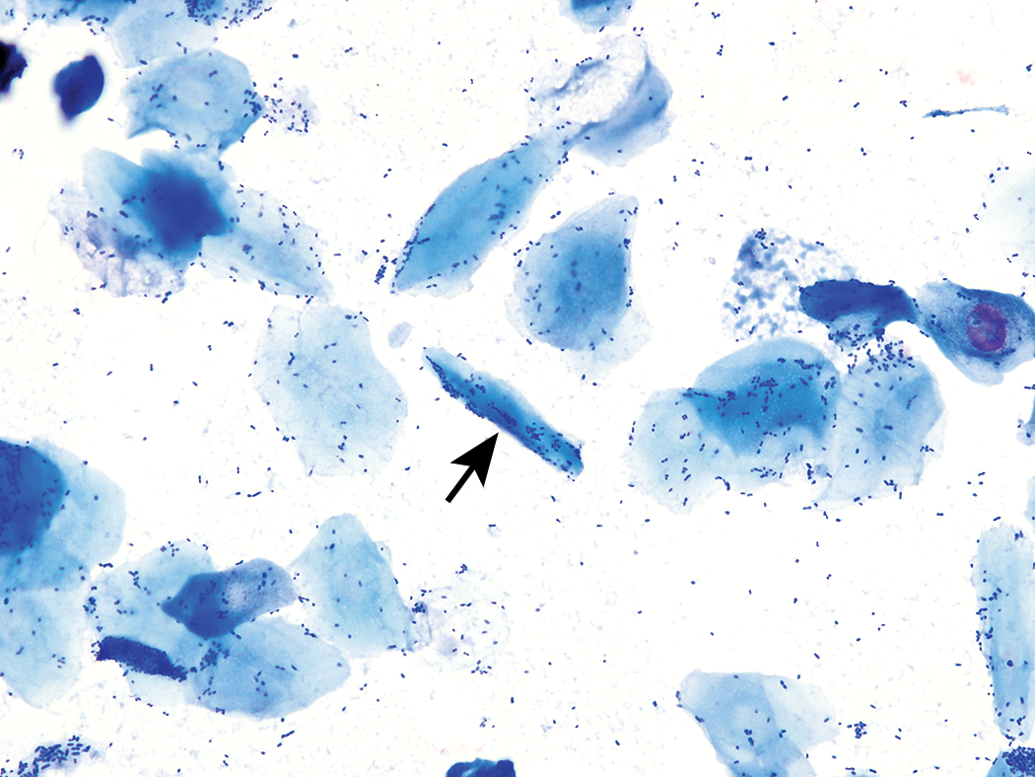
It should be noted that the distal one-fourth to one-third of the vaginal lumen is covered by squamous epithelial cells and is permanently keratinized. Therefore, it is possible that during sample collection, cells from this area of the vagina may be collected and is one reason why low numbers of anucleated keratinized cells can be seen in proestrus and diestrus. Additionally, glass slides may be contaminated with keratin if not handled properly (i.e., touching the surface of the slide with bare hands).
Stages of the Estrous Cycle
The stages of the estrous cycle are identified by the absence, presence, or proportion of the described four basic cell types as well as by the cell density and arrangement of the cells on the slide. Conventionally, the cycle is divided into the four stages of proestrus, estrus, metestrus, and diestrus; however, some researchers use a simple proestrus–estrus–diestrus classification, while others subdivide the four stages, record transitional stages, or roughly quantify the individual cell populations (Goldman, Murr, and Cooper 2007). The following will define and characterize the range in appearance of the four classic stages, including species differences and a brief discussion of transitional stages, from which criteria for stage subdivisions or transitional stages can be based.
One general approach to staging vaginal smears is to first assess for the presence of neutrophils—are there a good number of neutrophils observed or just an occasional one? If neutrophils are a dominant feature, or consistently observed, the stage is either metestrus or diestrus; if neutrophils are rare to absent, the stage is either proestrus or estrus. Figure 11 and Table 1 are graphical and tabular representations of the estrous cycle adapted from various sources and serve as good adjuncts to the subsequent narrative.
Estrous cycle wheels. Visual representation of the cell types and relative proportion of each cell type present during the 4 stages of the estrous cycle in the mouse and rat. The size of each quadrant does not directly correlate to the length of each stage. Adapted from Byers et al. (2012). The mouse wheel was modified from its original form to create the rat wheel. Adaptations and modifications by David Sabio. Basic classification of the stages of estrous cycle based on cell types and relative numbers of these cell types in vaginal smears.
Note: 0 = none; + = few; ++ = moderate; +++ = high.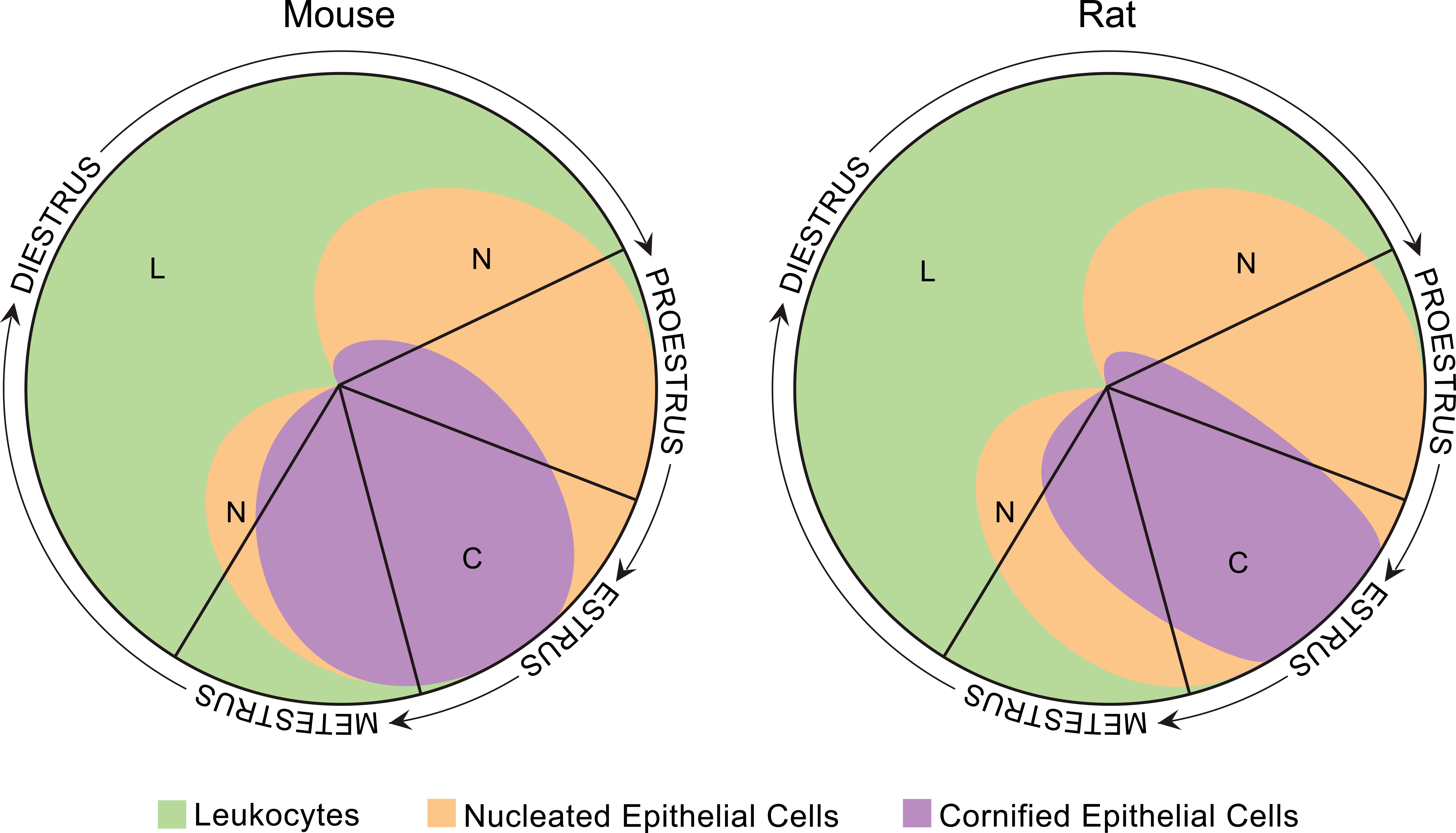

Proestrus
Proestrus is a short stage, lasting an average of 14 hr in rats and less than 24 hr in mice (Astwood 1939; Long and Evans 1922; Mandl 1951; Allen 1922; Grasso, Rozhavskaya, and Riechert 1998). The predominant feature of this stage is the presence of small, round, nucleated epithelial cells of relatively uniform appearance and size (Figures 6 and 12); these cells may stain deeply basophilic. Often they will be seen in cohesive clusters (grape clusters), sheets, or strands. However, cohesive clusters, sheets, and strands are not always observed, especially in low cellularity samples, and should not be thought of as a prerequisite in the determination of proestrus; cells may only be seen individually throughout the smear. Typically, no neutrophils will be seen. That said, rare to occasional neutrophils can be found in early proestrus as the rodent would have recently transitioned from diestrus into proestrus. Relatively low numbers of large epithelial cells and keratinized anucleated cells may also be observed. As the cycle approaches estrus, keratinized cells will become more abundant. The presence of low numbers of neutrophils, or large and anucleated epithelial cells, does not preclude the diagnosis of proestrus when the predominating feature of the smear are the small, round epithelial cell population.
Vaginal smears of proestrus from Sprague-Dawley rats characterized by high numbers of small nucleated epithelial cells found individually and in cohesive clusters (arrows). Proestrus appears the same in mice. Plate D shows the appearance of a so-called epithelial “strand” that is typical of proestrus. Modified Wright–Giemsa stain (A, D) and Toluidine blue O stain (B, C). Original objective magnification of 10× (A, B), 20× (C), or 40× (D). Plate C is in Elmore et al. Proceedings of the 2014 National Toxicology Program satellite symposium. Toxicol Pathol, 43:10-40, 2015.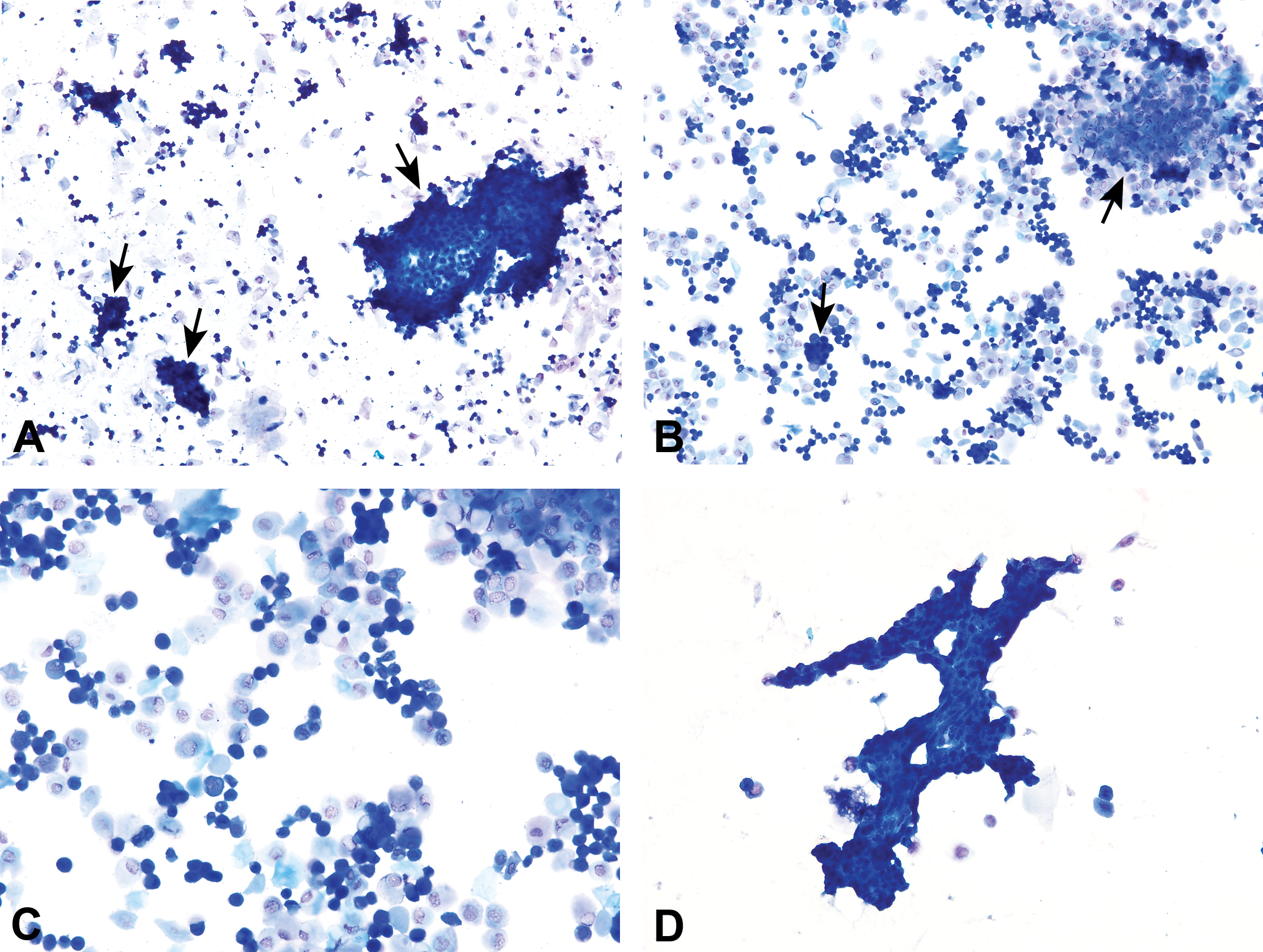
Estrus
Estrus duration ranges between 24 and 48 hr in rats and between 12 and 48 hr in mice (Astwood 1939; Long and Evans 1922; Mandl 1951; Allen 1922; Grasso, Rozhavskaya, and Riechert 1998). It is characterized by the presence of predominately anucleated keratinized epithelial cells (Figures 13 and 14). Numerous bacteria may be observed adhered to the cells or free in the background. Occasional nucleated epithelial cells can be observed throughout the stage of estrus, and neutrophils are absent or occasionally observed in late estrus. The first and second half of estrus, while consisting mostly of anucleated keratinized cells, have distinct differences in their appearances and also differ between rats and mice. These differences were distinct enough that Young, Boling, and Blandau (1941) and Allen (1922) had different terminology or subdivisions for the first and second “phases” of estrus in rats and mice, respectively. Long and Evans (1922) described these different phases in rats but did not distinguish them as subdivisions of estrus.
Representative photomicrographs of the first (A, C) and second (B, D) “phases” of estrus in mice. The first phase typically has smaller anuclear epithelial cells that are arranged in loose clusters reminiscent of proestrus. The anuclear epithelial cells of the second phase are generally larger, more evenly dispersed, and higher in numbers. Plates C and D are 2 consecutive days of estrus from the same B6C3F1/N mouse. Modified Wright–Giemsa stain (A, B) and Toluidine blue O stain (C, D). Original objective magnification of 10×. Vaginal smears of estrus (A, B) and late estrus (C–F) in several Sprague-Dawley rats. Plates A and B represent a typical estrus stage with high numbers of anucleated epithelial cells. Vaginal smears of the late estrus phase (C–F) are characterized by the presence of round-, oval-, and spindle-shaped nucleated epithelial cells interspersed among anucleated epithelial cells. Modified Wright–Giemsa stain (A–C, E) and Toluidine blue O stain (D, F). Original objective magnification of 10× (A, C, D), 20× (F), or 40× (B, E). Plate A is in Elmore et al. Proceedings of the 2014 National Toxicology Program satellite symposium. Toxicol Pathol, 43:10-40, 2015.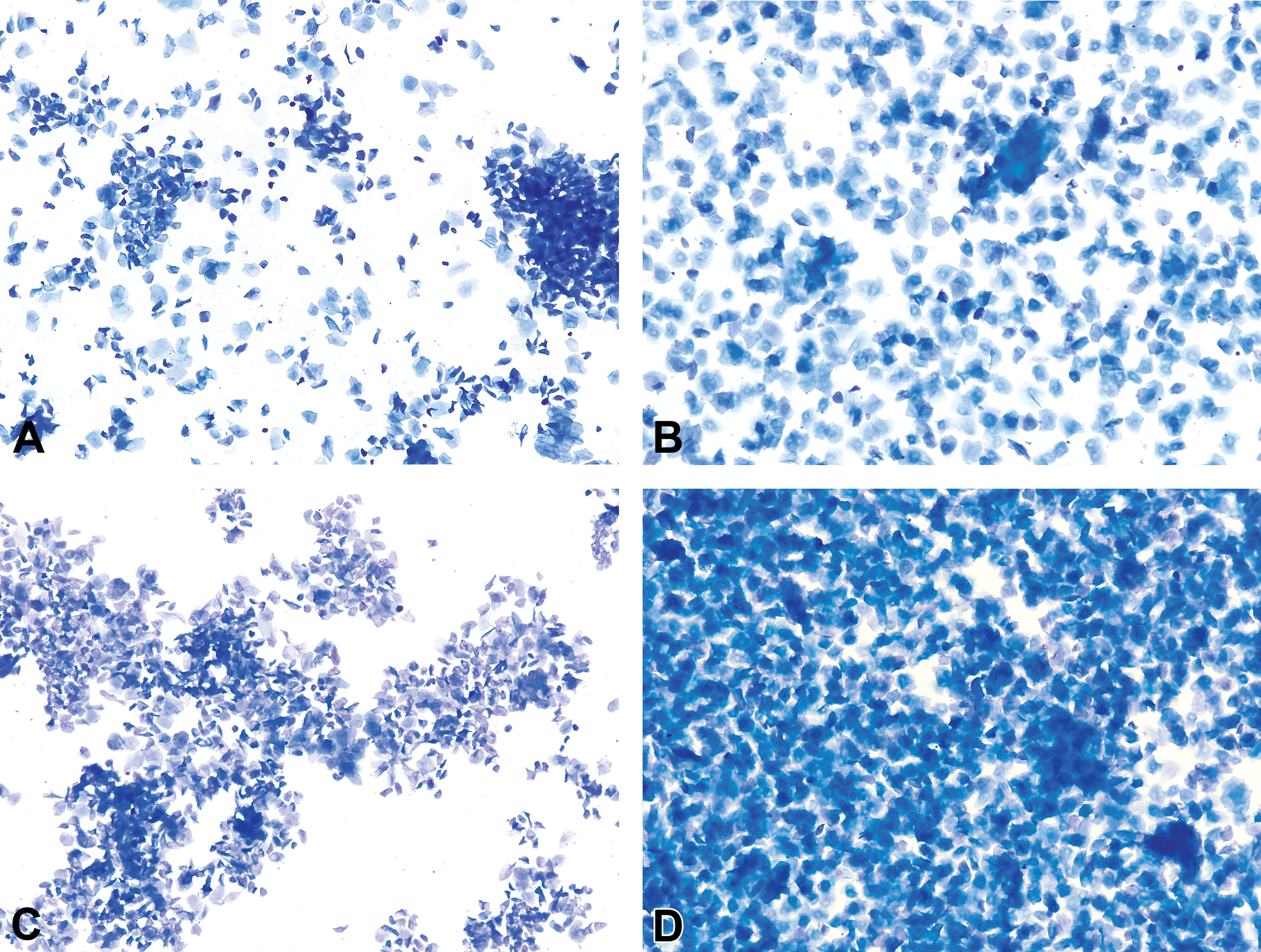
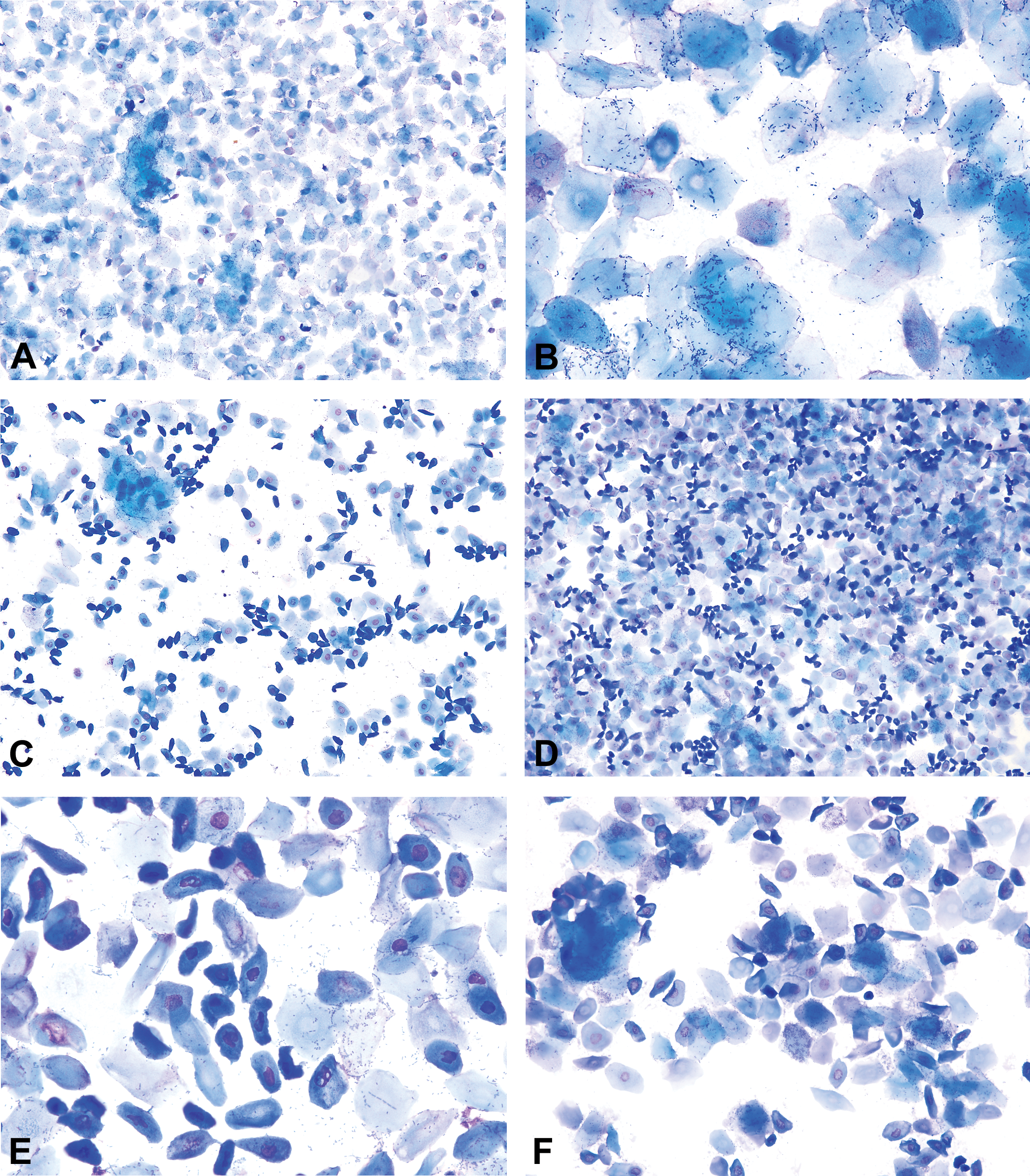
In a mouse when 2 consecutive days of estrus are observed, these different phases are generally appreciated; however, the second phase in rats occurs late in estrus and will not always be seen (i.e., it will be missed) even if 2 days of estrus are recorded. In mice, at the start of estrus, the anucleated cells are smaller and are typically arranged in loose clusters or sheets reminiscent of proestrus (Figure 13A and C). As estrus progresses, the cell numbers generally increase, and the cells appear larger and more evenly dispersed and can be seen in stacks or layers (Figures 9 and 13B, D). Allen (1922) notes this distinction in mice as follows: By standardizing the technique of smear preparation,… [the different phases of estrus] are easily differentiated, although the basis for this division is the number and clumping rather than any change in the [anucleated, keratinized] character of the cells themselves. (p. 306) It is interesting that in a certain proportion of animals the epithelial desquamation of the mucosa may proceed somewhat further than to the cornified cell layer before leucocytes come in, and hence we may have… Stage Three [estrus] immediately succeeded by a few hours of a stage in which rather large, spindle-like, nucleated epithelial cells are shed. In such cases, however, vestiges of the cornified cell are always present and when leucocytes appear three cell types area actually found. (p. 21)
In conclusion, estrus is the stage predominated by keratinized anucleated epithelial cells. Neutrophils may be occasionally seen in late estrus, as the rodent is transitioning to metestrus; but once they are observed with regularity throughout all fields, the stage should be interpreted as metestrus. Relatively high numbers of nucleated epithelial cells are interspersed with the keratinized cells in late estrus of rats.
Metestrus
It is well documented that metestrus is a short stage of 6–8 hr in rats (Astwood 1939; Long and Evans 1922; Mandl 1951). Based on the limited reports in the literature and personal experience, the duration of metestrus in mice seems to be longer than rats and can last up to 24 hr (Allen 1922; Grasso, Rozhavskaya, and Riechert 1998) and, although infrequent, metestrus (as described herein) has been identified for 2 consecutive days in control mice on reproductive studies (personal experience; Figure 15A and D).
Metestrous smears from several B6C3F1/N mice. Metestrus begins with the emergence of neutrophils interspersed or clumped (arrow) among the anucleated epithelial cells (A–D). Occasional nucleated epithelial cells may also be present. As metestrus progresses, neutrophil numbers increase resulting in smears of very high cellularity (E, F). Neutrophil and epithelial cell numbers decrease as the mouse transitions to diestrus (G, H). Plates A and D illustrate 2 consecutive days from a control B6C3F1/N mouse, representing the uncommon occurrence of a metestrus stage being recorded for 2 days in a row in a mouse. Modified Wright–Giemsa stain (A–D, G, H) and Toluidine blue O stain (E, F). Original objective magnification of 10× (A, C–E, G), 20× (F, H), or 40× (B). Plate F is in Elmore et al. Proceedings of the 2014 National Toxicology Program satellite symposium. Toxicol Pathol, 43:10-40, 2015.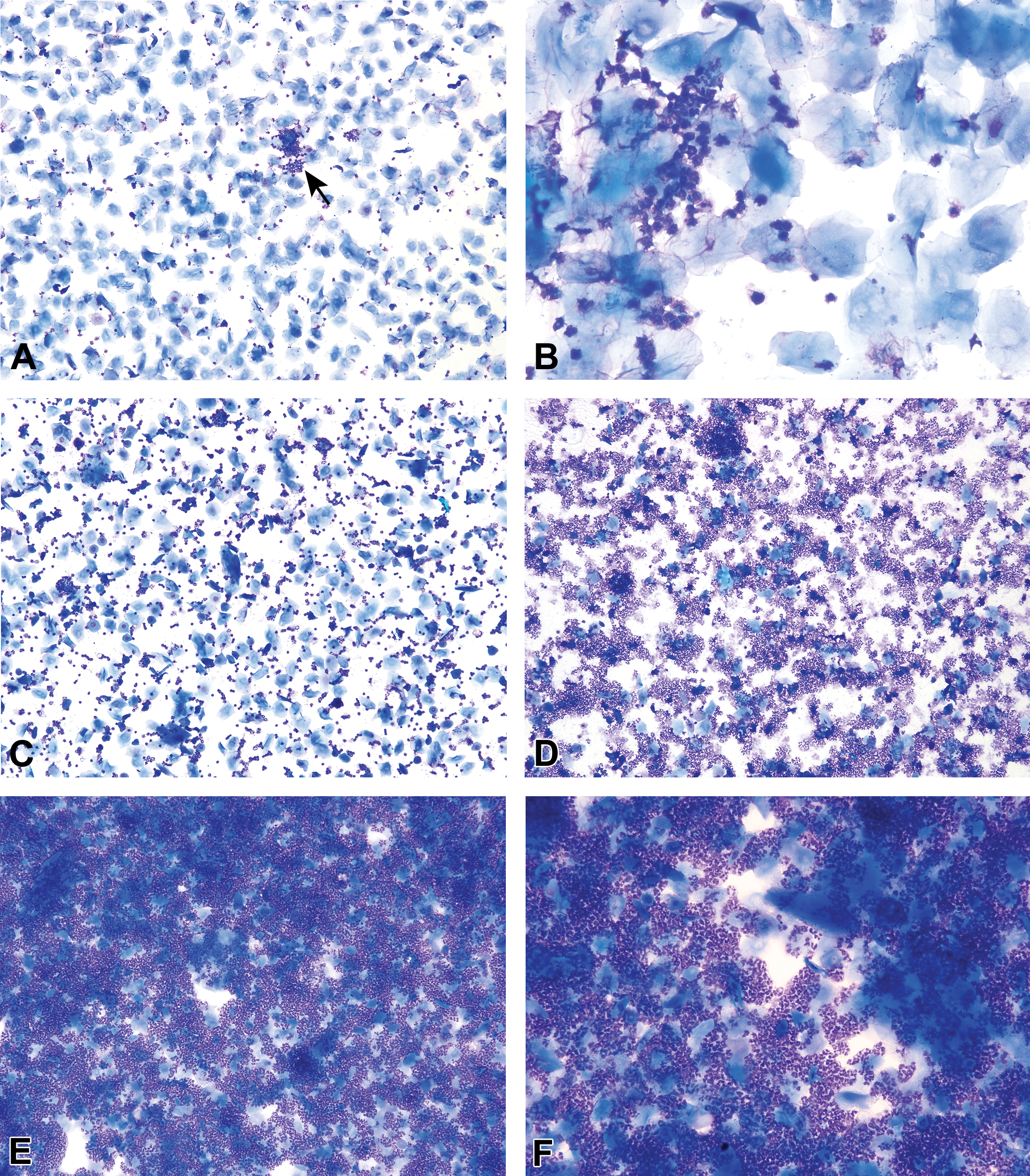
Metestrus is characterized by a combination of anucleated keratinized epithelial cells and neutrophils. In mice, nucleated cells may appear occasionally throughout metestrus (Figure 15). In rats, the small and large nucleated cells of late estrus are present in low to moderate numbers throughout the stage (Figure 16).
Metestrous vaginal smears from several Sprague-Dawley rats. Metestrus begins with the emergence of neutrophils interspersed or clumped among the nucleated and anucleated epithelial cells (A, B). Note that the appearance of metestrus differs in rats and mice, as rats have much higher numbers of nucleated epithelial cells. As metestrus progresses, neutrophil numbers increase resulting in smears of high cellularity (C–F). Toluidine blue O stain. Original objective magnification of 10× (A, C, E) or 40× (B, D, F).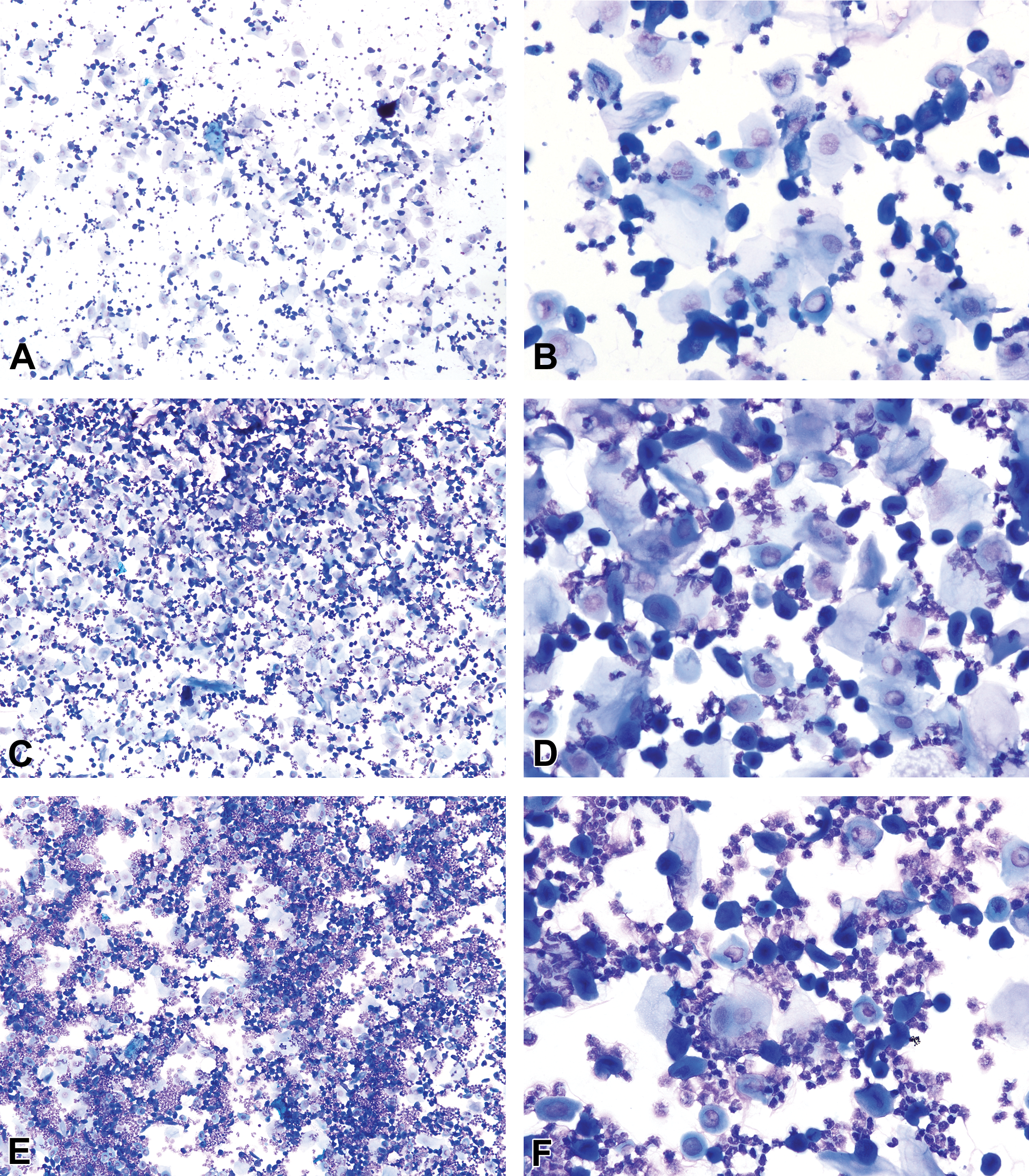
In early metestrus, neutrophils are interspersed with the epithelial cells and are sometimes tightly packed together or clumped around the cells; the epithelial cells usually predominate but may be in equal proportion to the neutrophils. As metestrus progresses, neutrophils become very high in number—outnumbering epithelial cells by as much as 10-fold (Hubscher, Brooks, and Johnson 2005)—and the smear will be highly cellular and dense. Neutrophil and epithelial cell numbers decrease by late metestrus, with a decrease in smear cellularity, before the transition to diestrus. Allen (1922) gives the following simple description of metestrus in mice: … the smear shows polymorphonuclear leukocytes among the red, horny elements [anucleated epithelial cells]. A few polymorphs and many cornified cells is an early [metestrus] stage, and a heavy leucocytic infiltration and decrease in number of the cornified cells is a late one. (p. 306)
Diestrus
Diestrus is the longest stage of the estrus cycle with an average duration of 48–72 hr in both mice and rats (Astwood 1939; Long and Evans 1922; Mandl 1951; Allen 1922; Grasso, Rozhavskaya, and Riechert 1998). This stage is characterized by a substantial decrease in the number of anucleated keratinized epithelial cells (but not necessarily an absence of) as the animal transitions out of metestrus. Overall cellularity is moderate to low with a combination of neutrophils, small and large nucleated epithelial cells, and low numbers of anucleated keratinized cells (Figures 4 and 17). Neutrophil numbers can vary but are usually higher in number relative to the epithelial cells with smears sometimes being exclusively neutrophilic. Occasionally, in early diestrus, neutrophils may still appear in clumps. It is not unusual for diestrus smears to have a very low cellularity, especially on day 2 or 3 of diestrus, with only a sparse scattering of neutrophils and epithelial cells (Figure 17E). In late diestrus, the epithelial cells may become more round or be organized in small clumps, indicating proestrus the next day; however, neutrophils will still be consistently observed (Figure 17F).
Diestrous vaginal smears from B6C3F1/N mice (A–E) and a Sprague-Dawley rat (F) characterized by a moderate to low cellularity with predominately neutrophils that are mixed with lower numbers of epithelial cells. A small clump of round epithelial cells (arrow) is present on a diestrous smear taken the day before the emergence of proestrus. Modified Wright–Giemsa stain (C–E) and Toluidine blue O stain (A, B, F). Original objective magnification of 10× (A, C, E, F) or 40× (B, D). Plate E is in Elmore et al. Proceedings of the 2014 National Toxicology Program satellite symposium. Toxicol Pathol, 43:10-40, 2015.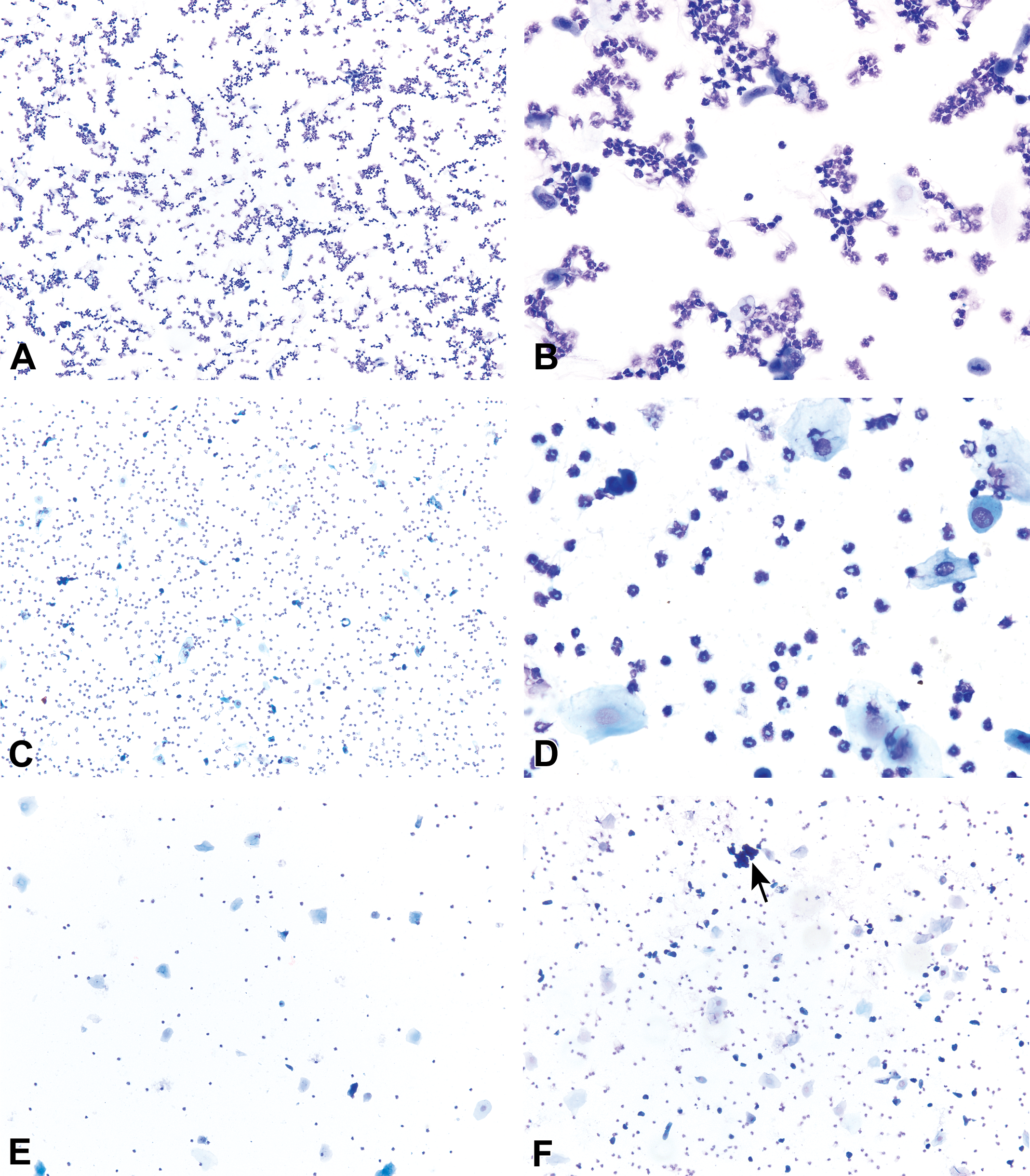
During diestrus, macroscopic vaginal excretions are low. When extrections are present, however, they can be viscous and stringy (i.e., mucous) and this is especially true during times of persistent diestrus. This excretion is sometimes aspirated during vaginal cytology sample collection. It will stain and appear as a thick pink to blue-violet material with entrapped neutrophils and epithelial cells. The cells may be distorted or elongated in the strings of mucous (Figure 18).
Diestrous vaginal smears from several Sprague-Dawley rats. Basophilic to eosinophilic staining mucous has entrapped and distorted the epithelial cells and neutrophils. A classic mucous “swirl” is seen in plate B (arrow). Modified Wright–Giemsa stain (B) and Toluidine blue O stain (A, C).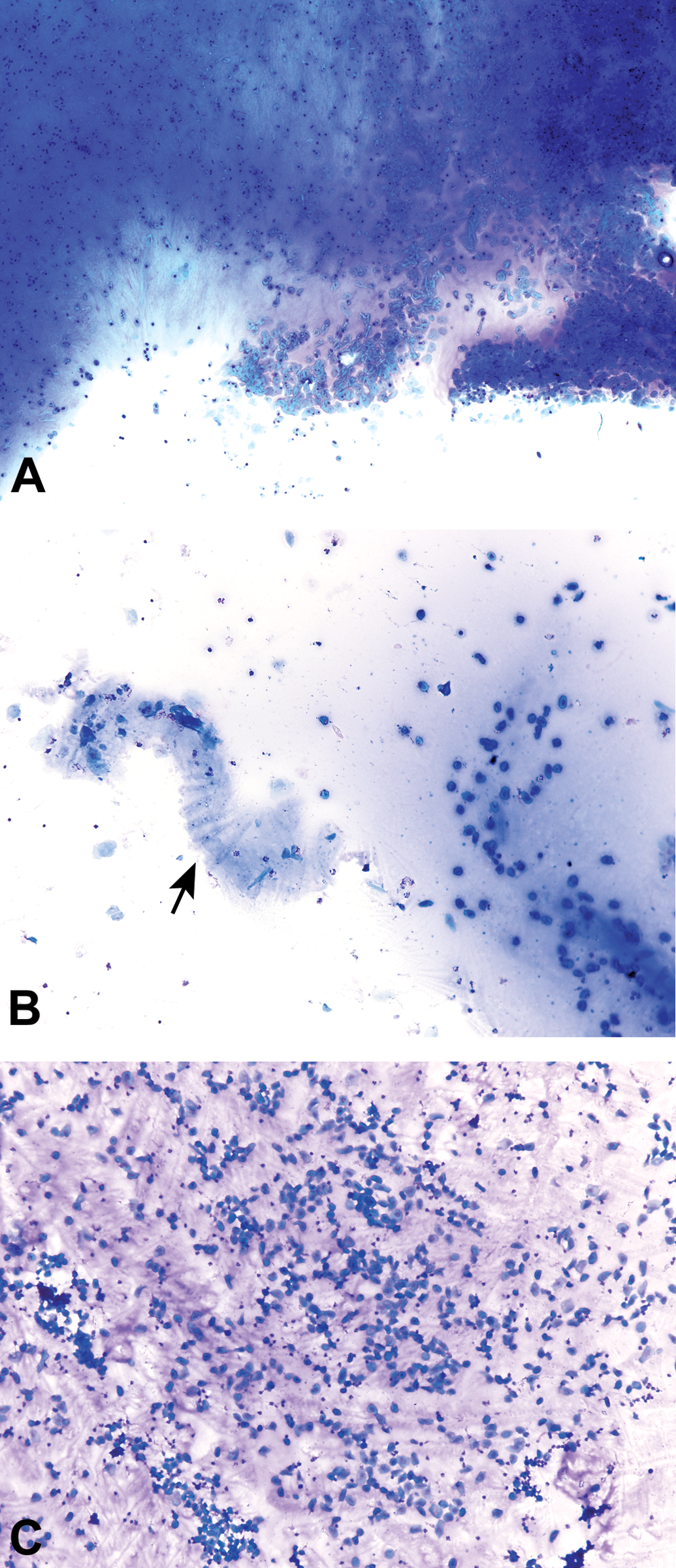
Transitional stages
For any given study, transitional stages will be encountered (Figure 19). There are several strategies or paradigms that can be used for the recording of a transitional stage. The strategy used would depend on the reason or purpose for documenting the stages of the estrous cycle; for example, a study designed to document potential changes in the frequency or duration of a particular estrous stage may require a different transitional stage paradigm than a study evaluating the estrous cycle in a group of young rodents to determine sexual maturity or their first ovulation. Following are 2 of the more common approaches for dealing with or recording transitional stages.
Lower (original objective magnification of 10×) and higher (20×) magnifications of a vaginal cytology smear taken from a Sprague-Dawley rat representing a proestrus-to-estrus transition. Individual and clumps of small epithelial cells are seen among anucleated epithelial cells. Based on complete review of this smear, the recorded stage (based on the 4-stage paradigm) was proestrus or “P.” Using the other classification paradigm discussed in the article, it could also be classified as “P(e).” Please refer to Transitional Stages section for further details. Modified Wright–Giemsa stain.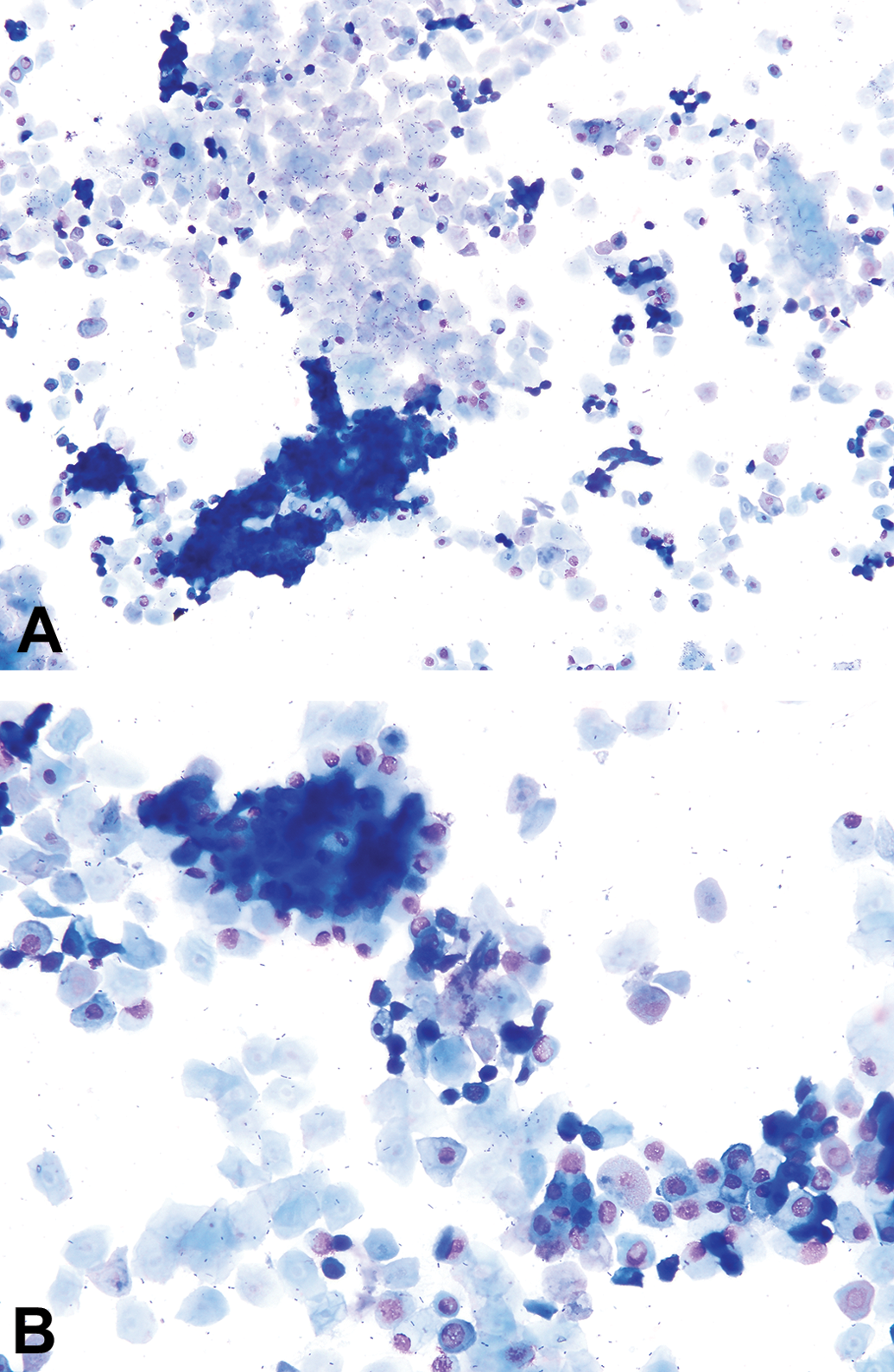
In studies where only the 4 classic stages are to be used to identify the stages of the estrous cycle, then it is recommended that with a transitional smear the predominating feature(s) of the slide be used to make the final call. For instance, when confronted with a proestrus–estrus transition smear, if the impression is that more than 50% of the cells are small and nucleated, then the smear would be classified as proestrus. Another strategy for the classification of transition stages is to use uppercase and lowercase letters. For example, the previously described smear would be designated as P(e) indicating that while the day was called proestrus (denoted by “P”), anucleated keratinized cells were also present in significant numbers, indicating that a transitioning toward estrus was occurring (denoted by “e”). Similarly, if clumps of small nucleated cells are observed in late diestrus, the smear can be recorded as D(p), indicating a transition from diestrus into proestrus. Regardless of what classification paradigm is used, to stay consistent and thus minimize variability, it is important that a concerted effort be made to ensure agreement on the criteria for stage definitions.
Cellularity
The cell density or cellularity of a cytology preparation may aid in defining a stage. Cellularity of a vaginal cytology, however, should not be used in isolation because it can vary for numerous reasons including those independent of the actual stage of the estrous cycle. The overall cellularity and ratio of cells during the estrous cycle can differ between individual rodents and this is particularly true for diestrus. Long and Evans (1922) reference this with the statement that “both types of cell [neutrophils, nucleated epithelial cells] may occur [during diestrus] in considerable numbers in some animals or may be both of them very sparse in others.” (p. 18). For a given animal, cellularity and cellular composition is usually consistent from cycle to cycle. For example, one animal may consistently have almost exclusively neutrophilic diestrus smears, while another will have a good mixture of neutrophils and epithelial cells. Thus, comparison of one animal to another should be avoided, and comparison of day-to-day changes for each animal individually is the best approach for identifying and predicting in advance a given animal’s cyclic pattern (Hubscher, Brooks, and Johnson 2005).
Further, there are preanalytical sources of variation that can affect cell density on a vaginal cytology preparation, including, collection technique, compliance of the rat or mouse during collection and preparing more than one smear for each collection day. More specifically, preparation of more than one smear for each day can alter the cellularity enough as to cause a metestrus smear to be classified as diestrus, for example. Although this is not a common issue, one should be aware that this phenomenon could occur when making more than 1 smear for each day.
Conclusion
Evaluation of sequential vaginal cytology smears is a time-honored tool for the assessment of the estrous cycle in laboratory rats and mice. Collection and preparation of vaginal cytology samples is an essentially noninvasive procedure that, with proper training, is easily mastered. For reliable and consistent data generated from vaginal cytology studies, evaluations of the cytology smears must be based on well-established standardized criteria. Smear review and, ultimately, the data obtained, however, can only be as “good” as the technical expertise and experience of the evaluator performing the microscopic examination. Thus, it is recommended that individual laboratories define and codify their respective processes via laboratory procedures, and the proficiency of the personnel reviewing the cytology materials must be established and is achieved through appropriate training, the use of vaginal cytology study sets of known diagnoses, and experience. As highlighted in this review, the cytological appearance of the rat and mouse estrous cycle differs in several ways. Thus, regardless of general similarities, when it comes to the estrous cycle, a “mouse is not a rat” and proper training is required for each species to garner full understanding of the differences.
Footnotes
Acknowledgments
The authors would like to thank Drs. Mark Cesta, NTP, NIEHS, and Barry McIntyre, NTP, for their helpful suggestions concerning the article, and Beth Mahler, EPL, for her help with all the photomicrographs.
Author Contribution
Authors contributed to conception or design (MC, LK, GT); drafting the article (MC); and critically revising the article (MC, LK, GT). All authors gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported (in part) by the National Institutes of Health, National Institute of Environmental Health Sciences.
1.
Note that the word estrus is a noun, while the word estrous with an “o” is an adjective, as in “the estrous cycle”; the two words are not interchangeable.
2.
As is widely known, the British English, Greek-derived spelling of estrus with an “o” as in oestrus and the oestrous cycle and the use of an “o” in the spelling of the stages (pro-oestrus, metoestrus, dioestrus) is still used today in British-based publications. Additionally, older literature, regardless of country of origin, generally used British English. This is important to note when undertaking a comprehensive literature search on the subject of the estrous cycle and its stages.
