Abstract
The development of novel safety or efficacy biomarkers has increasingly been used to improve safety monitoring and minimize attrition during drug development; however, for new biomarkers, the failure rate can equal or exceed that of new chemical entities. Drug-induced kidney injury is recognized to occur throughout the drug development process, with histopathology considered to be the gold standard for preclinical toxicologic screening. Renal biomarkers used clinically are primarily biomarkers of renal function and are considered insensitive for the detection of drug-induced kidney injury during first-in-man studies, particularly for compounds known to induce renal injury in preclinical species. Recent efforts by public–private partnerships have led to unprecedented success in the identification, development, and qualification of several new translatable biomarkers of kidney injury in the rat. To optimize the chance of success in current and future biomarker efforts in preclinical species and man, selection and development of biomarkers should emphasize biological considerations including marker variability and biology in both health and disease. The research to support the qualification of novel renal safety markers for routine use in the clinical setting is currently underway, and results from this work are greatly anticipated.
Introduction
Acute kidney injury (AKI), acute renal failure, and acute tubular necrosis are used synonymously in the clinical literature and are a major cause of morbidity in the clinic, approximating up to 50% of deaths in the critical care setting (Lieberthal and Nigam 2000). AKI is defined based on changes in renal function, specifically by changes in glomerular filtration rate (GFR) as reflected by changes in plasma or serum creatinine (sCR) and urine output, and are as described in the consensus Risk, Injury, Failure, Loss of kidney function, and End-stage kidney disease (RIFLE) and Acute Kidney Injury Network (AKIN) criteria (Bellomo et al. 2004; Kellum et al. 2005). It is widely recognized that these functional parameters are highly insensitive due to the large functional reserve capacity of the kidney, such that a sudden decrease in GFR is followed by a delayed rise in sCr that often requires several days to report compromised renal function. Kidney injury may also occur in the absence of effects on renal function, as can be seen with septic AKI, the predominant cause of AKI in the critical care setting (Murugan and Kellum 2011). In addition, changes in GFR and urine output can be manifestations of disease as well as adaptive responses to changes in extracellular fluid volume or renal blood flow, termed “acute renal success” (Kellum 2008). The recognition that kidney injury may precede, follow, or occur in the absence of functional renal impairment has led to the widespread evaluation of a number of urinary proteins as markers of structural kidney damage, with the intent to localize kidney injury to specific nephron segments or pathologic processes, and also to help differentiate intrinsic renal from prerenal injury (Murugan and Kellum 2011; Kellum 2008; Tsigou et al. 2013). The failure to identify kidney injury early has major clinical and economic consequences due to the lack of successful interventions; therefore, the identification and development of biomarkers of AKI are of great interest.
Disconnect between Human and Animal AKI
The development of any new biomarker requires the use of both robust reference standards and relevant animal models; thus, the development of novel markers of AKI poses several unique challenges. In addition to the low sensitivity and delayed onset of change in markers of renal function such as sCr, the most commonly used clinical or preclinical reference standard, there are profound structural differences between human and animal AKI. In contrast with animal models of AKI where severe, submassive tubular injury is relatively common, changes in kidney histopathology in human AKI are extremely rare (Heyman, Rosenberger, and Rosen 2011). When noted, tubular necrosis with AKI in humans is usually focal and minimal, primarily affecting the outer medulla, specifically the medullary thick ascending limb of the loop of Henle and collecting duct initially, with subsequent involvement of the proximal segment (Heyman, Rosenberger, and Rosen 2010). Similarly, the widespread proximal tubular necrosis seen in the rat warm ischemia-reperfusion (IR) model, a commonly used model for human AKI, is rarely seen in human ischemic nephropathies such as transplant nephropathy. Rather, extensive proximal tubular necrosis in humans is restricted to severe hemodynamic and ischemic events, as can be seen during prolonged resuscitation or with postpartum hemorrhage (Heyman, Rosenberger, and Rosen 2010). Acute tubular necrosis in humans is thus generally defined as a clinical syndrome in which there is sufficient renal perfusion for tubular preservation, but insufficient renal perfusion for the preservation of glomerular filtration (Rosen and Heyman 2001; Kellum 2008).
In preclinical studies, proximal tubular necrosis is one of the most common manifestations of drug-induced AKI. This is due to the unique structural and functional features of the proximal tubule (PT), where reabsorption and concentration of the glomerular filtrate occur through localized expression of receptors and transporters that mediate protein, glucose, and electrolyte absorption. The PT also expresses numerous transporters including organic anionic transporters (OAT) or organic cationic transporters (OCT), multidrug and toxic compound extrusion transporters (MATEs), and adenosine triphosphate (ATP)-binding cassette (ABC) or other agent-specific transporters that predispose the PT to injury caused by accumulation of toxicants or their metabolites.
Although animal models are used extensively to investigate both the pathophysiology and the treatment of acute renal failure, acute renal failure is actually difficult to model in preclinical species. For example, hypovolemia and shock are the most common causes of ischemic AKI; however, severe and prolonged ischemia (partial aortic ligation) is required to cause renal failure in the rat (Zager 1988). Unlike animal models of IR, AKI in humans following hemodynamic insult is often preceded by lower-magnitude states of hypovolemia. Sublethal and reversible kidney injury as might be caused by comorbidities such as hypovolemia is recognized as a contributing factor in the development of renal tubular dysfunction, and this is readily amenable to investigation using a combination of
Animal models of drug-induced kidney injury following administration of nephrotoxic agents often fail to recapitulate the clinical scenario because they cause disproportionately severe structural injury (Figure 1). Further, there are extensive structural differences among species that can affect vulnerability to kidney injury. For example, in the rat, cisplatin causes extensive necrosis of the pars recta of the PT in the outer stripe of the outer medulla (OSOM; Gautier et al. 2010); yet humans and primates lack a defined OSOM. Also, in humans, cisplatin causes injury to the PT, distal tubule, and collecting duct (Tanaka et al. 1986). Preclinical models of nephrotoxic injury generally lack the comorbid factors commonly seen with AKI in the clinic (Okusa 2010). Exceptions include rat models of contrast media or endotoxin-mediated nephropathy, where administration of either alone does not cause acute renal injury but, when combined with other renal insults such as volume depletion, uninephrectomy, indomethacin, furosemide, or salt depletion, will cause kidney injury (Lieberthal and Nigam 2000; Rouse et al. 2013).
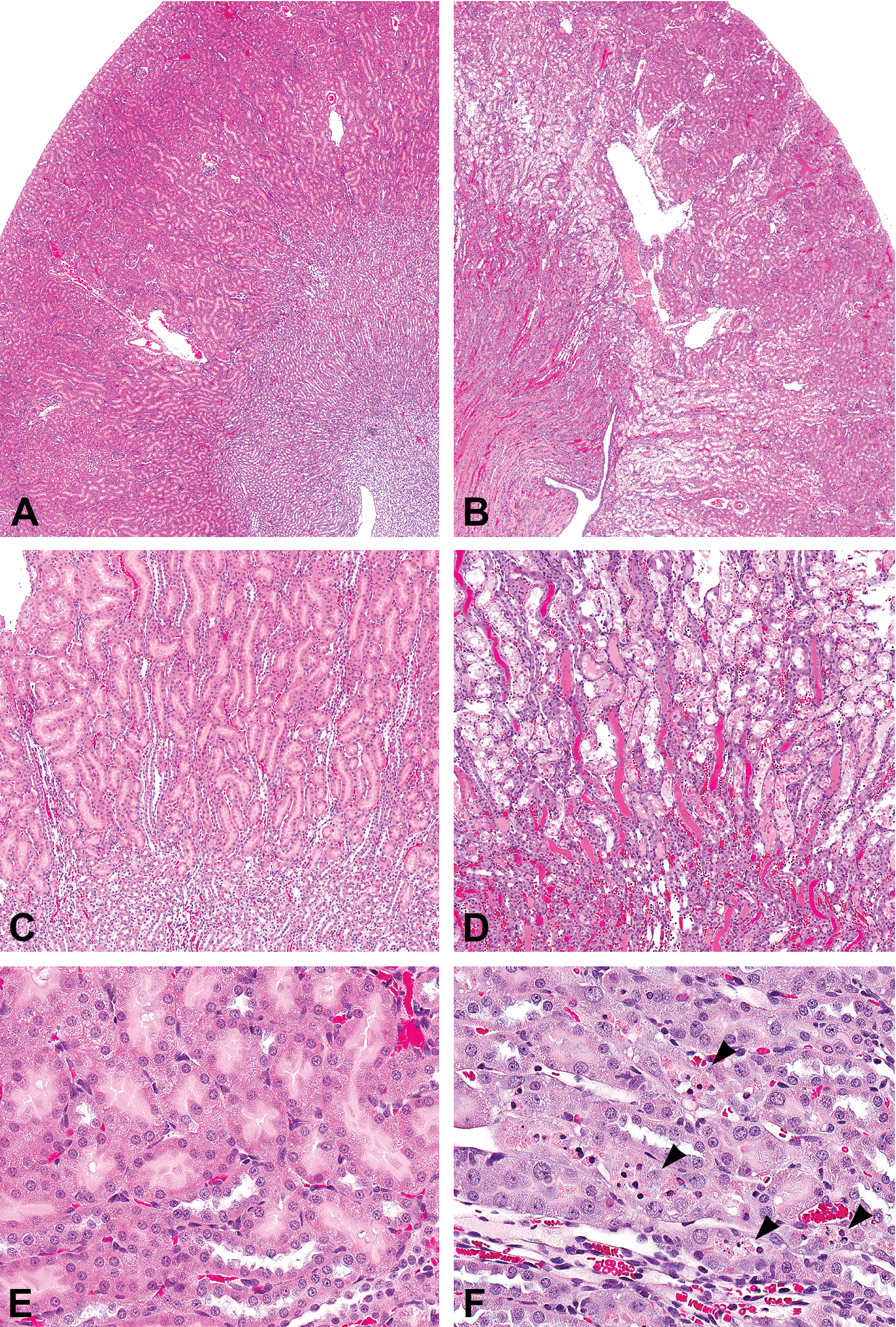
Cisplatin-induced nephrotoxicity in Crl:WI(Han) rats. (A, C, and E) Control kidney. (B and D) Moderate to marked tubular necrosis of the S3 segment of the proximal tubule within the outer stripe of the outer medulla on day 5 after a single 3 mg/kg intraperitoneal (IP) dose of cisplatin. (F) Minimal to mild single-cell necrosis on day 5 after a single 1 mg/kg dose of cisplatin IP (arrowheads). Hematoxylin and eosin, original magnification = 40×.
Identification and Development of Candidate Markers of Renal Injury
Successful development and progression of any new biomarker are aided by understanding the biological or pathophysiological function of that marker within the target organ of interest. With AKI, biomarkers can appear in the urine due to a variety of mechanisms, including impaired reabsorption of filtered proteins (as seen with the low-molecular-weight proteins cystatin C, β2-microglobulin, or α1-microglobulin that freely cross the glomerular filtration barrier and are normally reabsorbed in the PT), induction of intrarenal synthesis (as seen with Kim-1 or clusterin), or production by infiltrating inflammatory cells (e.g., neutrophil gelatinase–associated lipocalin [NGAL]). In addition, multiple mechanisms for increases in AKI markers can coexist. For example, NGAL, a low-molecular-weight, acute-phase protein originally identified in neutrophil-specific granules, is found in kidney and other tissues including liver, lung, and gastrointestinal tract, where it is produced in response to tissue injury, inflammation, or sepsis (Tsigou et al. 2013). NGAL plays a role in adaptive immunity as well as protection against oxidative stress through its activity as a siderophore or iron chelator. Normally, concentrations of plasma or serum NGAL are maintained at low levels by renal clearance through megalin-cubulin-mediated endocytosis (Charlton, Portilla, and Okusa 2014). Upregulation of NGAL mRNA in extrarenal tissues in response to sepsis, local tissue injury, or AKI causes increases in circulating NGAL concentrations (Devarajan 2010). With AKI, profound and rapid upregulation of renal NGAL mRNA occurs predominantly in the distal nephron, where it likely occurs as an adaptive response, as reflected by the protective effects of infusion of recombinant NGAL in rodent models of AKI (Charlton, Portilla, and Okusa 2014). Because NGAL has a low molecular weight of approximately 25 kD, increases in urinary NGAL excretion can reflect impaired proximal tubular reabsorption, as with other low-molecular-weight proteins; however, increased renal NGAL synthesis is generally considered to be the primary source of increased urinary NGAL excretion in AKI (Devarajan 2010). NGAL is rapidly and markedly upregulated in response to AKI and is often the earliest clinical signal, often preceding increases in sCR by days (Devarajan 2010). Thus, plasma and urinary NGAL have demonstrated a high sensitivity for kidney injury in both clinical and nonclinical studies that is related to diverse pathophysiological mechanisms.
Renal transcriptomics have played a key role in the identification of candidate markers of AKI, particularly for proteins such as Kim-1, osteopontin, clusterin, and NGAL that are upregulated within the kidney in response to AKI (Amin et al. 2004; Thukral et al. 2005). Urinary proteomics is another promising platform for identifying candidate biomarkers. Routine urinalysis techniques generally measure only a subset of the total urinary protein because the soluble (supernatant) fraction contains approximately 49% of the total urinary protein output, while another 48% is present in the sediment and 3% is found in exosomes (Pisitkun, Johnstone, and Knepper 2006). The importance of evaluating these urinary fractions relates to the differential localization of renal biomarkers as exemplified by glomerular podocyte markers—for example, the protein podocalyxin has greater abundance in the supernatant than in the sediment in both health and disease, while podocalyxin-specific granular structures and podocyte-specific mRNAs are detected in the sediment fractions (Sekulic and Sekulic 2013). Evaluation of the proteome of the exosomal fraction has great potential for use in targeted proteomics discovery because the proteins contained in exosomes directly reflect their cell of origin (Pisitkun, Shen, and Knepper 2004). For example, the presence of segment-specific proteins such as the Na-K-Cl cotransporter type2, the Na-Cl cotransporter (NCC), or aquaporin-2 can be used to localize human exosomes to the thick ascending limb of the loop of Henle, the distal convoluted tubule, or the collecting duct, respectively (Biner et al. 2002). However, it should also be noted that there are species differences in the localization of these segment-specific markers (Plotkin et al. 1996). Importantly, evaluation of the urinary exosomal proteome enriches the less-abundant cohort of urinary proteins for improved detection by mass spectrometry or immunochemistry (Pisitkun, Shen, and Knepper 2004).
Although profiling the urinary proteome using mass spectrometry has identified more than 1,000 different proteins, the selection and successful development of potential renal biomarkers remain challenging. As with any candidate new biomarker, the marker should have relevance to the intended target or disease process. Successful biomarker development also requires knowledge of post-translational modifications and understanding of the marker biology (e.g., effects of intrarenal processing, phosphorylation state, and time course).
Meta-analyses of preclinical and clinical data play a pivotal role in the evaluation of the diagnostic utility of a candidate biomarker. In contrast with studies using single nephrotoxicants, the curation of study data across multiple studies and combination into large data sets enable statistically robust analyses that most accurately describe the diagnostic performance of a novel biomarker. Receiver operating characteristic (ROC) analysis is one of the most commonly used methods for the evaluation of biomarker performance. ROC analysis is a truly unbiased evaluation because it evaluates every possible sensitivity and specificity pair within a data set without specification of a diagnostic threshold. Other statistical methodologies include integrated discrimination index (IDI) and net reclassification index (NRI), methods that are increasingly being used to evaluate the incremental added value of biomarkers with other predictors of risk.
Evaluation of biomarker performance requires use of a robust reference standard, which for preclinical studies is generally histopathology. The standardization of histopathological diagnoses using agreed upon diagnostic lexicons is widely employed in preclinical meta-analyses (Harpur et al. 2011; Sistare et al. 2010). In addition, curation of histopathology data is commonly performed to consolidate diagnostic redundancies and optimize statistical evaluation through the “binning” of diagnoses that are analogous or represent a continuum of histopathological change (Harpur et al. 2011; Vlasakova et al. 2014). Another extremely important aspect of biomarker performance evaluation is the use of relevant disease populations. It should be noted that the use of classical or “tool” nephrotoxicants that produce severe nephrotoxicity such as cisplatin (Figure 1) often yields marked changes in kidney injury biomarkers that far exceed changes seen in the preclinical or clinical nephrotoxic “space” (Table 1). The disparity in the magnitude of biomarker changes between qualification studies of “tool” toxicants and regulatory studies of development compounds can be considerable and has profound impact on the resultant diagnostic performance metrics in preclinical meta-analyses. This is because as the magnitude of biomarker change increases, so does the sensitivity, leading to potential overestimation of the diagnostic value of a new biomarker outside of the reference population.
Urinary biomarker changes in rats given cisplatin.
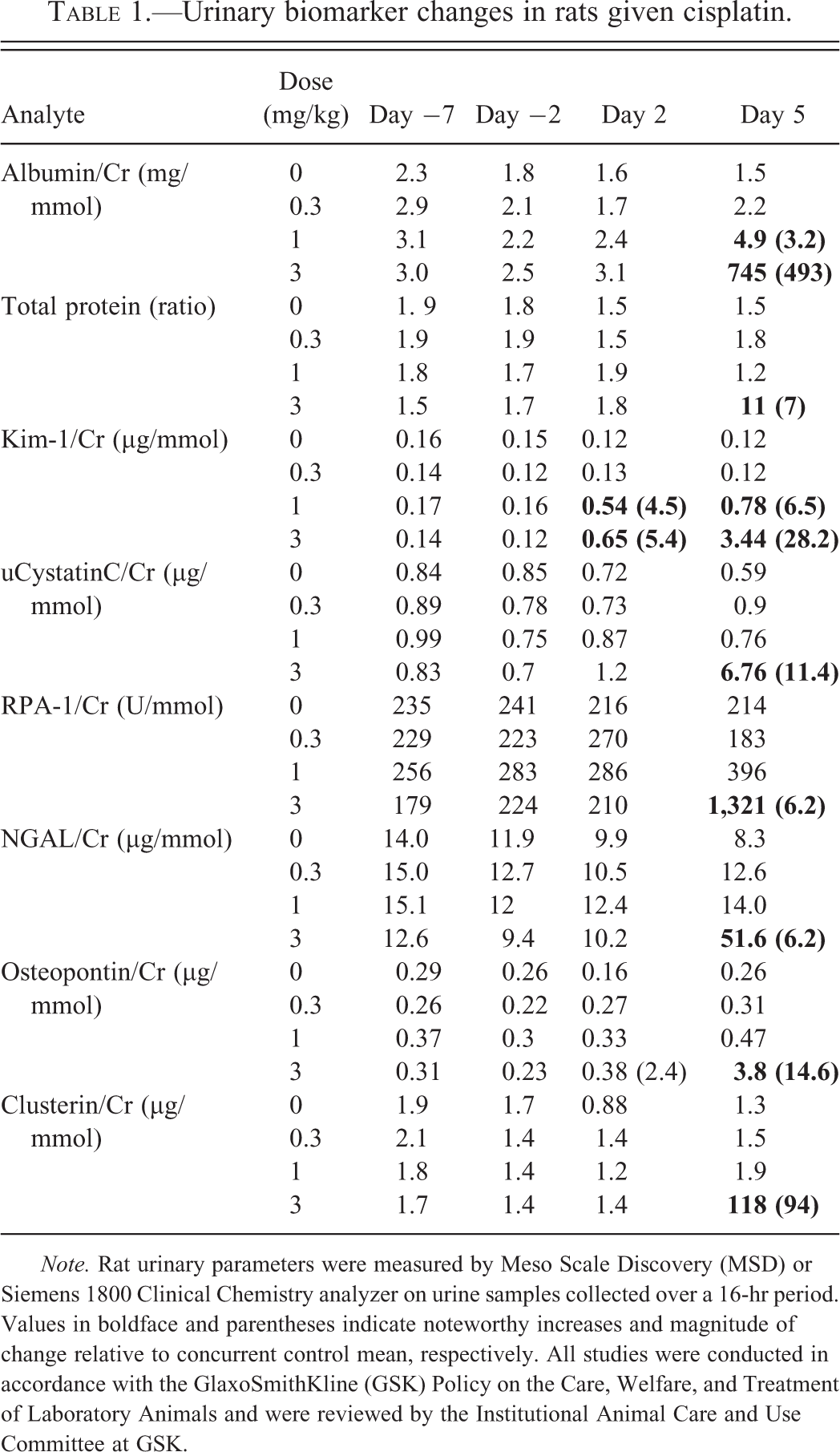
It is also important to understand the normal biological variability of every new biomarker. Unlike blood biomarkers, which circulate within a relatively constant volume at relatively stable concentrations, urine is a highly unstable matrix of variable or undefined volume that is vulnerable to other preanalytic variables such as food consumption, collection procedures, age and sex differences, diurnal variation, pH, osmolality, and so on. To diminish variability in urinary parameters, timed urine collections are commonly performed, as well as normalization of urinary parameters to urinary creatinine excretion, which is considered to be relatively constant under steady-state conditions. In early AKI, however, normalization to urinary creatinine may not be appropriate, because urinary creatinine excretion is highly unstable in early renal injury. For example, following a sudden decrease in GFR, urinary creatinine excretion will drop acutely and then increase in parallel with sCr because urinary creatinine excretion is the product of plasma or serum Cr clearance. Therefore, in early AKI, the evaluation of urinary analytes as concentrations rather than normalized to urinary creatinine may be more appropriate (Tonomura et al. 2011). Irrespective of normalization method, normal biological variability of urinary biomarkers in the rat and human may be up to 2-fold or 100% (Pinches, Betts, Bickerton, Beattie et al. 2012; Pinches, Betts, Bickerton, Burdett 2012; Brott et al. 2014). Diagnostic meta-analyses are often used to predict diagnostic thresholds, for example, as the magnitude of marker change at the highest specificity in a ROC analysis; however, the resultant predictions are often of low magnitude and within the range of biological variability (Vlaskova et al. 2014). While statistical meta-analyses are excellent tools for characterization of the diagnostic usefulness of a marker, they do not inform clinical or preclinical decision making. Rather, interpretation of novel biomarker data requires experience through use, combined with understanding of the unique aspects of each marker related to its dynamic range, time course, and so on.
The Current Preclinical Kidney Biomarker Landscape
Over the past decade, several markers of kidney injury have been evaluated and qualified in the rat by public–private partnerships (Betton et al. 2012; Dieterle et al. 2010; Gautier et al. 2010; Harpur et al. 2011; Ozer et al. 2010; Vaidya et al. 2010). A summary of the preclinical markers of kidney injury recently qualified or evaluated by these consortia in the rat is provided in Table 2. With the exception of renal papillary antigen-1 (RPA-1), a rat-specific collecting duct marker, all of these markers are currently undergoing evaluation in the dog and nonhuman primate. Several of these and other markers of renal injury have also been evaluated in healthy human volunteers for the determination of confidence intervals, subject variability, and analytic and preanalytic variables (Brott et al. 2014), and are currently undergoing evaluation in patients.
Rat kidney injury biomarkers evaluated by the PSTC and HESI.
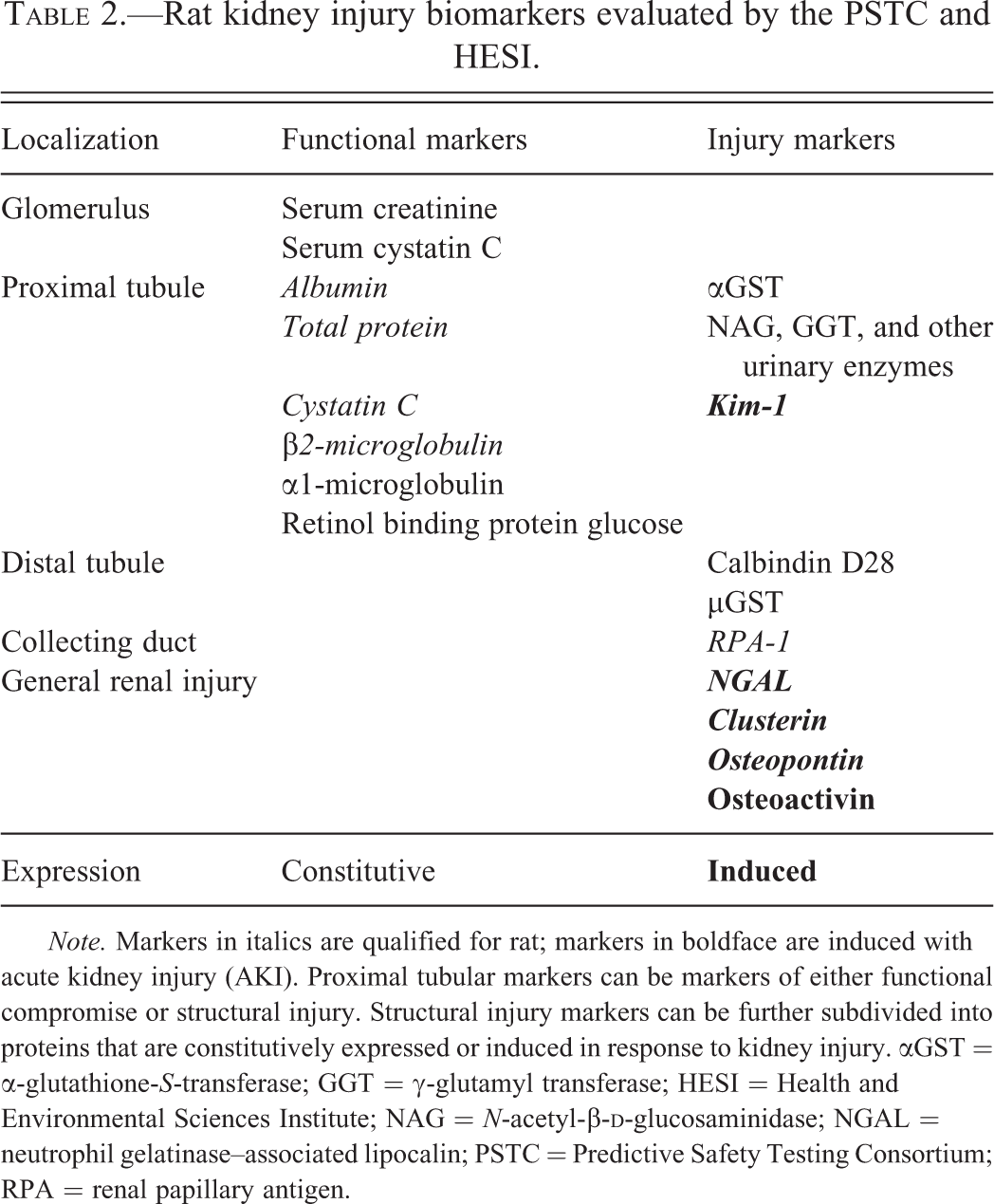
With the exception of the markers of glomerular filtration, serum (or plasma) creatinine, urea, or cystatin C, the kidney injury markers that have been evaluated preclinically are urinary proteins. These proteins can be subcategorized based on localization to nephron segment, role as functional or injury marker, and constitutive or induced expression. There is redundancy among these markers, and selection of one marker over another for use in a species is determined by a variety of factors such as assay availability and stability. For example, β2-microglobulin was qualified for the rat by the Predictive Safety Testing Consortium (PSTC; Dieterle et al. 2010); however, this protein is highly unstable in acid urine and therefore of limited value in dogs and humans, where α1-microglobulin is the preferred marker. The pattern of change in the excretion of urinary analytes can provide information on the functional status and severity of injury. For example, increases in urine glucose excretion in the absence of hyperglycemia could reflect an abnormality of proximal tubular glucose reabsorption, as the PT normally reabsorbs >99% of the urinary glucose. Similarly, the glomerular filtrate normally contains proteins of varying molecular weight, including albumin, which are predominantly absorbed within the PT; thus, proteinuria can reflect glomerular and/or tubular injury. The presence of a low-molecular-weight proteinuria (as reflected by increases in urinary β2-microglobulin, α1-microglobulin, cystatin C, retinol binding protein, and NGAL) could reflect an early protein overload nephropathy, as these proteins readily pass the glomerular filtration barrier and are normally reclaimed by the PT. In contrast, a high-molecular-weight proteinuria (i.e., increased urinary albumin) might reflect progression of both glomerular and tubular injury with increased passage of large-molecular-weight proteins through the glomerulus and loss into the urine due to saturation of the proximal tubular absorption capacity. Markers released in response to PT injury include the constitutively expressed proteins α-glutathione-
Inducible renal injury markers such as Kim-1, NGAL, clusterin, and osteopontin are cellular stress-related markers with different biological activities in AKI. For example, unlike NGAL, which is rapidly and transiently upregulated in response to renal injury, Kim-1 induction within the PT persists until there is recovery from renal injury. As with NGAL, induction of Kim-1 in AKI is an adaptive response related to tissue remodeling, where apical expression of Kim-1 on proximal tubular epithelium mediates the removal of apoptotic and necrotic cellular debris through phosphatidylserine-mediated phagocytosis (Ichimura et al. 1998). Kim-1 expression also co-localizes with dedifferentiation and proliferation markers (Ichimura et al. 1998), consistent with this model of kidney repair dedifferentiation, migration, and proliferation (Bonventre 2003). Chronic upregulation of Kim-1 can be associated with tubulointerstitial inflammation and fibrosis, however (Humphreys et al. 2013); thus, Kim-1 expression in renal injury can have both adaptive and deleterious effects.
Considerations for Implementation and Development of Preclinical Markers
Broader implementation of these new biomarkers, including assessment of interim changes, reversibility, and age and sex differences, is needed to gain understanding and experience with their use in preclinical studies. For example, while most of the qualification studies to date were performed in male rats, striking differences in the dynamic range and response to injury for several urinary biomarkers are reported for female rats (Gautier et al. 2014; Pinches, Betts, Bickerton, Burdett 2012 ). Strategic design of preclinical studies can aid in incorporating these new markers while simultaneously minimizing animal usage. For instance, if nephrotoxic injury was noted in a dose range study that did not include urinalysis, bridging the doses used in the dose range study to those in a subsequent study, and timing of urine collections in the second study to match the duration of the dose range study will enable use of histopathology data from the dose range study to inform the changes in the interim urinalysis data for the second study. Thus, animal usage is minimized, and potential time-related biomarker changes are measured for comparison with the final urinalysis and histopathology data at study termination.
While the existing meta-analyses based on studies using tool toxicants may overestimate the diagnostic utility of the novel kidney injury markers due to spectrum bias resulting from disproportionately severe structural injury, the broader implementation of these markers in preclinical drug development will both inform interpretation of novel biomarker data and provide the opportunity for additional meta-analyses using data from studies of development compounds to obtain realistic estimates of their diagnostic utility. In these additional meta-analyses, the performance of the novel biomarkers should also be compared with that of traditional quantitative analysis parameters such as urinary albumin, protein, glucose, or NAG. Ultimately, multi-marker panels are likely to be the end result of these biomarker qualification efforts, and species differences in their content are to be expected.
Clinical Evaluation Strategy for Renal Biomarkers
The qualification of novel renal safety biomarkers for preclinical use has set the framework for the development and qualification of clinical renal safety biomarkers. During the drug development process, it is important to be able to translate information learned in preclinical species to humans, and, in particular, to be able to monitor for specific safety findings as the compound moves through advanced testing in human healthy subjects and patients. The process of qualification is complex and requires multiple steps in order for clinical biomarkers to achieve acceptance by health authorities. Due to the complexity of biomarker development, public–private consortia have formed to join forces in generating the data to support clinical biomarker qualification. The Critical Path Institute PSTC and the Innovative Medicines Initiative Safer and Faster Evidence-based Translation Consortium (IMI SAFE-T) are two such organizations that are supporting the effort to develop and qualify novel renal safety biomarkers.
One of the most critical aspects of biomarker qualification is to determine the appropriate and specific context of use for the biomarker/biomarkers being developed. This context of use will then establish the prospective clinical trial to be designed that will support the use of that biomarker. If there are additional ways in which that biomarker is to be used, then additional studies should be designed to support its use in that manner. If a biomarker is qualified in this way, it would be a progressive qualification and similar in some ways to the development of a drug moving from phase 1 (in healthy subjects) through phase 3 (specific patient populations).
Once the context of use has been established, the biomarker expression profile must be well characterized in a healthy subject population to establish the normal range and variability. Typically, the establishment of true reference intervals requires 120 to 400 subjects. To date, the consortia have been able to collect and evaluate data from several individual studies of healthy volunteers and to pool data from these studies in an attempt to establish normal ranges for novel renal biomarkers.
Once a normal range has been established, it is important to understand the dynamic range of the biomarker in the context of a known nephrotoxicant such as aminoglycosides or cisplatin. Several prospective trials are currently underway in support of developing biomarker profiles in subjects exposed to agents known to induce renal injury. Intensive statistical analyses will be required to understand if the novel biomarker responses provide additional information related to the identification of drug-induced kidney injury as compared to the traditional clinical biomarker sCr.
In addition to these aspects of clinical biomarker development, preanalytic factors need to be considered, including the appropriate timing of sample collection, handling of samples, storage conditions, and the need to add stabilizer. These are important considerations as the biomarkers are transitioned into routine use, because these factors will clearly be critical for integration into clinical study protocols and planning for project teams. There is a tremendous amount of work ahead in this area of biomarker development, but the rewards are great, as these tools allow further development of novel medicines and increase the safety of subjects and patients participating in drug development trials.
Footnotes
Author Contribution
Authors contributed to data acquisition, analysis, interpretation (DE); drafting the manuscript (DE); and/or critically revising the manuscript (SA). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
