Abstract
Background:
Toxicant-associated fatty liver disease (TAFLD) is a recently identified form of nonalcoholic fatty liver disease (NAFLD) associated with exposure to industrial chemicals and environmental pollutants. Numerous studies have been conducted to test the association between industrial chemicals/environmental pollutants and fatty liver disease both in vivo and in vitro.
Objectives:
The objective of the article is to report a list of chemicals associated with TAFLD.
Methods:
Two federal databases of rodent toxicology studies—Toxicological Reference Database (ToxRefDB; Environmental Protection Agency) and Chemical Effects in Biological Systems (CEBS, National Toxicology Program)—were searched for liver end points. Combined, these 2 databases archive nearly 2,000 rodent studies. Toxicant-associated steatohepatitis (TASH) descriptors including fatty change, fatty necrosis, Oil red O-positive staining, steatosis, and lipid deposition were queried.
Results:
Using these search terms, 123 chemicals associated with fatty liver were identified. Pesticides and solvents were the most frequently identified chemicals, while polychlorinated biphenyls (PCBs)/dioxins were the most potent. About 44% of identified compounds were pesticides or their intermediates, and >10% of pesticide registration studies in ToxRefDB were associated with fatty liver. Fungicides and herbicides were more frequently associated with fatty liver than insecticides.
Conclusion:
More research on pesticides, solvents, metals, and PCBs/dioxins in NAFLD/TAFLD is warranted due to their association with liver damage.
Introduction
The liver is the first line of defense against potentially harmful xenobiotics, and it is therefore the target organ that is most commonly affected by commercially produced chemicals and environmental pollutants. Indeed, 33% of the 677 most common workplace chemicals reported in the National Institute of Occupational Safety and Health Pocket Guide are associated with hepatotoxicity (Tolman and Sirrine 1998). The pathologic liver lesions associated with chemical exposures are myriad and range from hepatitis, fibrosis, and cirrhosis to liver cancer (Cave, Falkner, and McClain 2011). However, following the description of toxicant-associated fatty liver disease (TAFLD) and its more severe form, toxicant-associated steatohepatitis (TASH), it now appears that fatty liver may be the most common pathologic hepatic response to chemical exposure (Brautbar and Williams 2002; Cave et al. 2012; Cave, Falkner, Costello, et al. 2011; Cave, Falkner, and McClain 2011; Wahlang et al. 2013). Identifying TAFLD/TASH in humans is challenging for several reasons. The entity is clinically underrecognized; routine clinical biomarkers are insensitive; and of the 88 million substances registered with the Chemical Abstracts Service (CAS) by 2014, there is no comprehensive list of chemicals correlated with TAFLD (CAS 2014). As such, TAFLD is a clinicopathologic diagnosis that relies on histologic examination.
We have recently reviewed pathologic grading and staging systems and known molecular mechanisms of fatty liver disease (Wahlang et al. 2013). The term “TASH” was initially coined in 2010 to describe steatohepatitis in human vinyl chloride (VC) workers (Cave, Falkner, et al. 2010). Liver biopsies from highly exposed workers resembled those from obese subjects (nonalcoholic steatohepatitis [NASH]) or alcoholics (alcoholic hepatitis), although these workers were neither obese nor consumed alcohol. Relatively more is known about histologic abnormalities in NASH, and this is primarily driven by pharmaceutical clinical trials including the National Institutes of Health (NIH)-sponsored NASH Clinical Research Network (Kleiner et al. 2005). The NASH CRN uses, in part, histological improvement to determine the efficacy of experimental medications. Important pathologic lesions including steatosis, inflammation, and fibrosis have been included in these studies. Steatosis has been defined as an accumulation of triglycerides in at least 5% of hepatocytes (Aly and Kleiner 2011; Canet et al. 2012). The transition from steatosis to steatohepatitis is characterized by centrilobular (zone 3)-centered injury and lobular inflammation (lymphocytes with neutrophils and activated Kupffer cells), hepatocyte ballooning, and Mallory-Denk bodies and fibrosis (Kleiner and Brunt 2012). Although these findings are typically present on H&E-stained slides, other stains have been used, such as Oil Red O that stains lipid droplets to quantify steatosis. Similar pathologic lesions have been observed in human TASH and in rodent models of steatohepatitis (Wahlang et al. 2013).
TASH development may have similar mechanisms to other forms of fatty liver disease. TASH may be a progressive “2 hit model” in which the “second hit” occurs on the background of steatosis and involves the elevation of inflammatory cytokines, mitochondrial dysfunction, insulin resistance, and oxidative stress, which causes steatohepatitis and fibrosis (Day and James 1998; Yilmaz 2012). With time and persistence of exposure to these conditions, which can arise secondary to chemical exposure, steatohepatitis may progress to fibrosis and cirrhosis (Cave, Falkner, et al. 2010; Cave, Falkner, Henry, et al. 2011; Wahlang et al. 2013).
Rodent models are widely utilized to study steatohepatitis. However, these models typically do not recapitulate all aspects of the human diseased form (McGonigle and Ruggeri 2014). It is therefore important to select the appropriate animal model that would best conform to the study end points (Larter and Yeh 2008). Although many chemical exposure studies in rodents in the literature have reported the development of steatosis descriptors, the significance of these findings was not appreciated. This is because steatosis was erroneously believed to be a benign finding, at least prior to the description of NASH in 1980 (Ludwig et al. 1980) and certainly TASH in 2010. The purpose of this study is to provide a list of chemicals that impact hepatic steatosis based on previously published rodent studies. A searchable archive of rodent studies provided in the websites of U.S. Environmental Protection Agency (EPA) and the National Institute of Environmental Health Sciences presented a unique opportunity to accomplish this objective. The identification of environmental chemicals associated with the development of TAFLD will enable subsequent mechanistic animal studies and clinical translation in exposed humans.
Materials and Methods
Searchable Databases
Two comprehensive chemical exposure and rodent pathology databases managed by the U.S. federal government were accessed for this study. The first was the EPA database known as the Toxicological Reference Database (ToxRefDB), which was designed by the National Center for Computational Toxicology and the EPA Office of Pesticide Programs and includes the past 30 years’ pesticide registration toxicity data and US$2 billion of animal studies results (EPA 2013). Using standardized vocabulary, ToxRefDB warehouses detailed study design, dosing, and observed treatment-related effects. The ToxRefDB also stores chemical toxicity data in detail through freely accessible and searchable databases (EPA 2013). ToxRefDB also connects with the Aggregated Computational Toxicology Resource (ACToR) in order to link it with public hazard, exposure, and risk resources (EPA 2013). Furthermore, ToxRefDB is connected to ToxCast, another EPA database and a high-throughput screening tool that links exposure to biological processes affected by chemicals (EPA 2013). ToxRefDB allows users to search congregate and group chemicals depending on the toxicological outcomes that are specific to the type of the study and target organ/effect categories (e.g., tumorigenicity; Martin et al. 2007). ToxRefDB classifies chemicals by their relative potency depending on specific end points/grouping of chemicals that also depends on the mechanism of action.
Currently, the ToxRefDB warehouses searchable pathologic information on 474 studies of pesticides and intermediates. In our study, the 474 rat/mouse studies were queried for histological nonalcoholic fatty liver disease (NAFLD) and TASH descriptors including “fatty change,” “Oil red O positive,” “steatosis,” and “lipid deposition.” The data were accessed in Fall 2013 at http://actor.epa.gov/toxrefdb/faces/Home.jsp. The following study types were queried: subchronic (SUB), chronic (CHR), and multigeneration reproductive (MGR). MGR are studies performed in rodents to identify parental and offspring systemic toxicity and the reproductive toxicity of pesticides, industrial chemicals, and pharmaceuticals (Martin et al. 2009). The effect type selected was “pathology (nonneoplastic).” The effect target was always the “liver” in the search, and the effect descriptions were “fatty change,” “lipid deposition,” “steatosis,” and “Oil red O” positivity in increased effect direction (see supplemental material, ToxRefDB search instructions). Compounds selection was based on the altered NAFLD and TASH descriptors at the lowest effect level (LEL). Compounds and their LELs were arranged and listed in tables.
The second rodent database utilized was the Chemical Effects in Biological Systems (CEBS) data repository developed by the National Toxicology Program (NTP 2004), which warehouses about 9,000 rodent toxicology studies (NTP). CEBS combines public toxicogenomics data including study design and time line, clinical chemistry and histopathology, and microarray and proteomics data (Waters et al. 2008). CEBS warehouses data from academic, industrial, and governmental laboratories, and it was mainly developed to allow public and free search through these data and studies (Sciences 2012; Waters et al. 2008). CEBS stores rats, mice, and human subjects studies, and it contains more than 4,000 microarray hybridizations, and 75 two-dimensional gel images with protein identification (Waters et al. 2008). Furthermore, CEBS comprises more than 1,500 animals’ clinical chemistry and histopathology data (Waters et al. 2008).
In Fall 2013, CEBS was accessed at: http://cebs.niehs.nih.gov. The queried assay domain was “histopathology,” and the diagnoses selected were “fatty change” and “toxic hepatopathy” as the latter 2 terms appear to have been used to describe fatty liver in several NTP reports on polychlorinated biphenyls (PCBs; National Toxicology 2010). “Liver and all its parts” was always the target organ selected, and all degrees of severity were included (see supplemental material, CEBS search instructions). The search initially returned 329 studies, but medications and natural products were subsequently manually excluded. Remaining compounds and their LELs were then arranged and listed in tables.
Results
ToxRefDB
At the LEL, 42 pesticides from 474 studies were associated with TAFLD pathologic descriptors including “fatty change,” “Oil red O positive,” “steatosis,” and “lipid deposition” (Table 1). The 42 compounds included 22 fungicides, 13 herbicides, 6 insecticides, and 1 miticide. These positive results came from both species (rat = 40 and mouse = 20) and from all queried study designs including SUB (n = 16), CHR (n = 34), and MGR (n = 10). Thus, >10% of pesticide studies were associated with the development of TAFLD. Not all of these pesticides may be clinically relevant mediators of steatohepatitis due to the high LEL values reported in some cases. However, 6 pesticides had LELs less than 10 mg/kg/day and that increases the likelihood that they could be clinically significant mediators of TAFLD depending on their crop application patterns. These pesticides were cyproconazole, dazomet, fluazinam, hexaconazole, pyrasulfotole metabolite (SXX 0665), and acequinocyl. Cyproconazole, dazomet, fluazinam, flusilazole, hexaconazole, paclobutrazol, triadimefon, vinclozolin, and fluthiacet-methyl pesticides were associated with the development of steatosis in more than 1 study. This reproducibility increases the likelihood that exposures to these chemicals do indeed result in steatosis. Two fungicides, dazomet and hexaconazole, were linked to steatosis in 3 studies and had LELs <10 mg/kg/day in at least 2 studies. Supplemental Table 1 lists the 395 chemicals and their studied doses that did not produce histologic descriptors of fatty liver disease in rodents in the ToxRefDB database.
Pesticides associated with fatty liver disease in ToxRefDB.
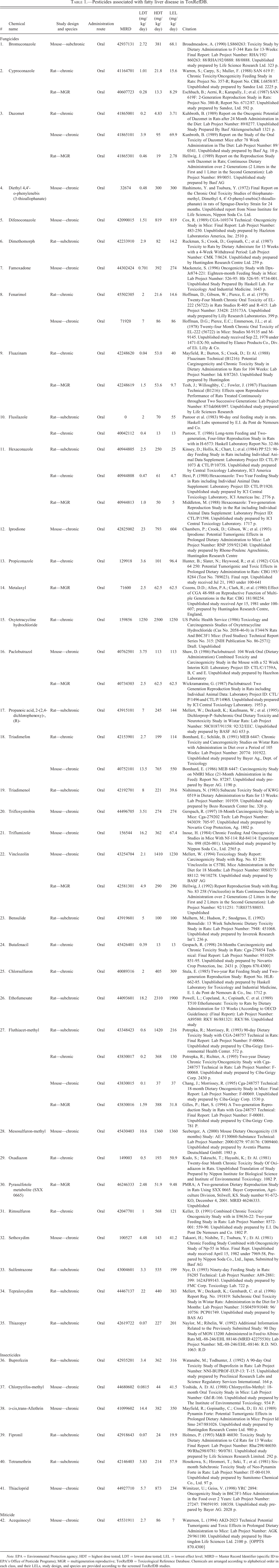
Note. EPA = Environmental Protection agency; HDT = highest dose tested; LDT = lowest dose tested; LEL = lowest effect level; MIRD = Master Record Identifier (specific ID to EPA's Office of Pesticide Programs); MGR = multigeneration reproductive; ToxRefDB = Toxicological Reference Database. Chemicals are arranged according to alphabetical order in each class, and their LELs, study design, and species are provided according to the screened ToxRefDB studies.
CEBS
Three hundred twenty-nine studies of 81 chemicals reported positive TAFLD descriptors (“toxic hepatopathy” and “fatty change”). These chemicals included 31 solvents, plasticizers, monomers, and chemical intermediates (Table 2); 14 miscellaneous chemicals (Table 3); 12 pesticides and pesticide intermediates (Table 4); 9 fragrances, cosmetics, and essential oils (Table 5); 9 paints, polishes, dyes, and food additives (Table 6); and 6 PCBs and dioxin-like molecules (Table 7). Several chemicals from each class produced steatosis with LELs ≤10 mg/kg (7 of 14 pesticides; 6 of 6 PCBs and dioxin-like compounds; 4 of 31 solvents, plasticizers, monomers, and chemical intermediates; 3 of 9 paints, polishes, and dyes; 3 of 14 miscellaneous chemicals; and 1 of 9 fragrances; cosmetics, and essential oils). In CEBS, steatosis was reported in 29 mouse studies and 57 rat studies including both acute (n = 9) and CHR (n = 72) exposure models.
Solvents, plasticizers, monomers, and chemical Intermediates associated with fatty liver disease in CEBS.
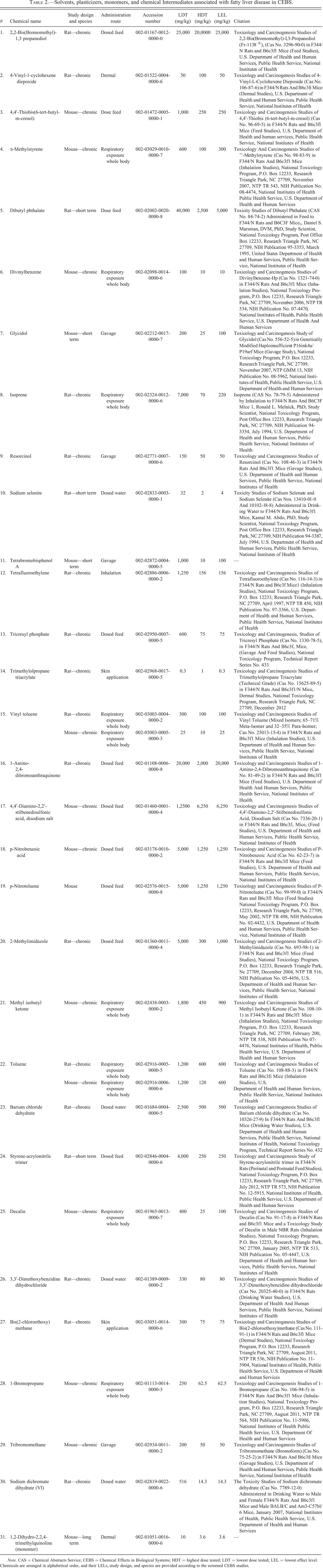
Note. CAS = Chemical Abstracts Service; CEBS = Chemical Effects in Biological Systems; HDT = highest dose tested; LDT = lowest dose tested; LEL = lowest effect level. Chemicals are arranged in alphabetical order, and their LELs, study design, and species are provided according to the screened CEBS studies.
Miscellaneous chemicals associated with fatty liver disease in CEBS.
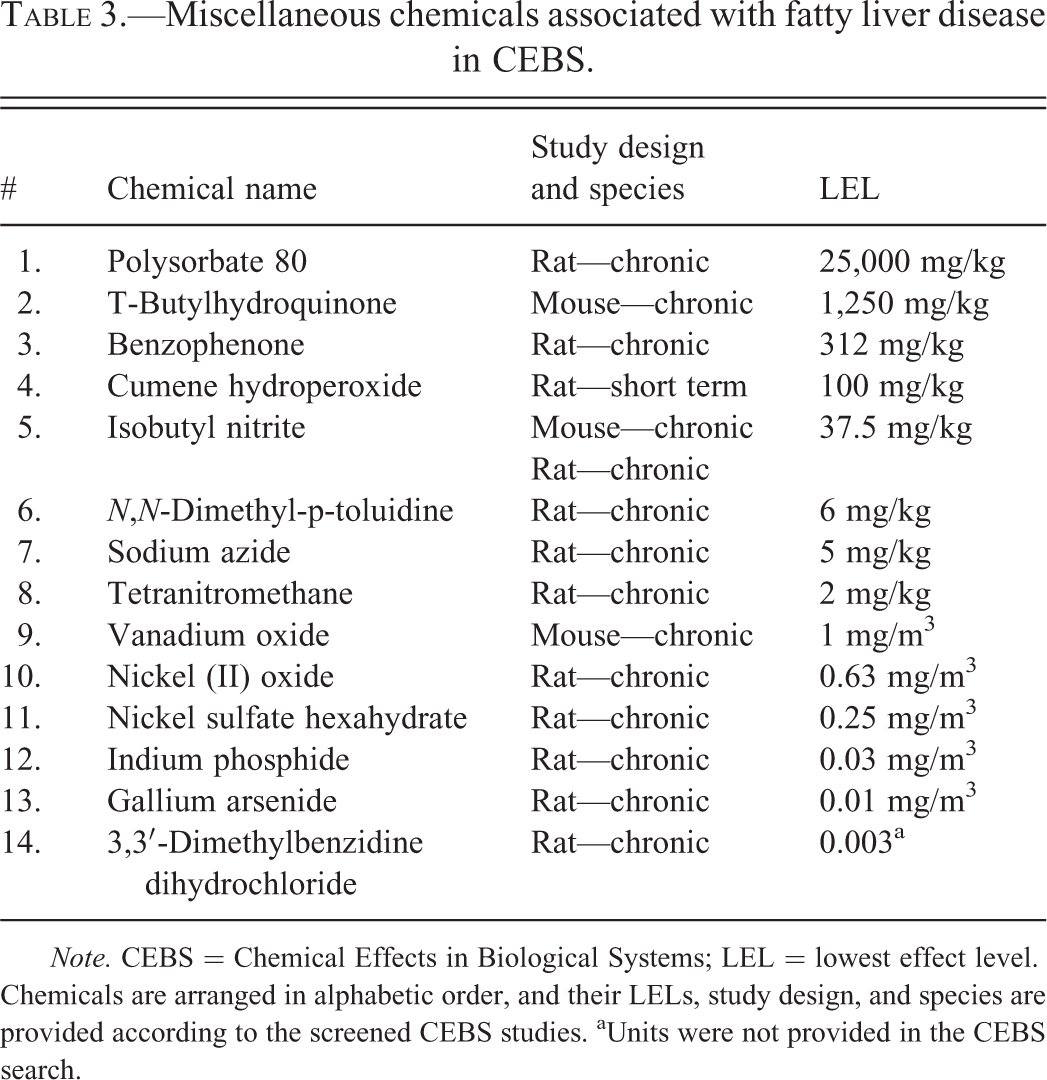
Note. CEBS = Chemical Effects in Biological Systems; LEL = lowest effect level. Chemicals are arranged in alphabetic order, and their LELs, study design, and species are provided according to the screened CEBS studies. aUnits were not provided in the CEBS search.
Pesticides associated with fatty liver disease in CEBS.
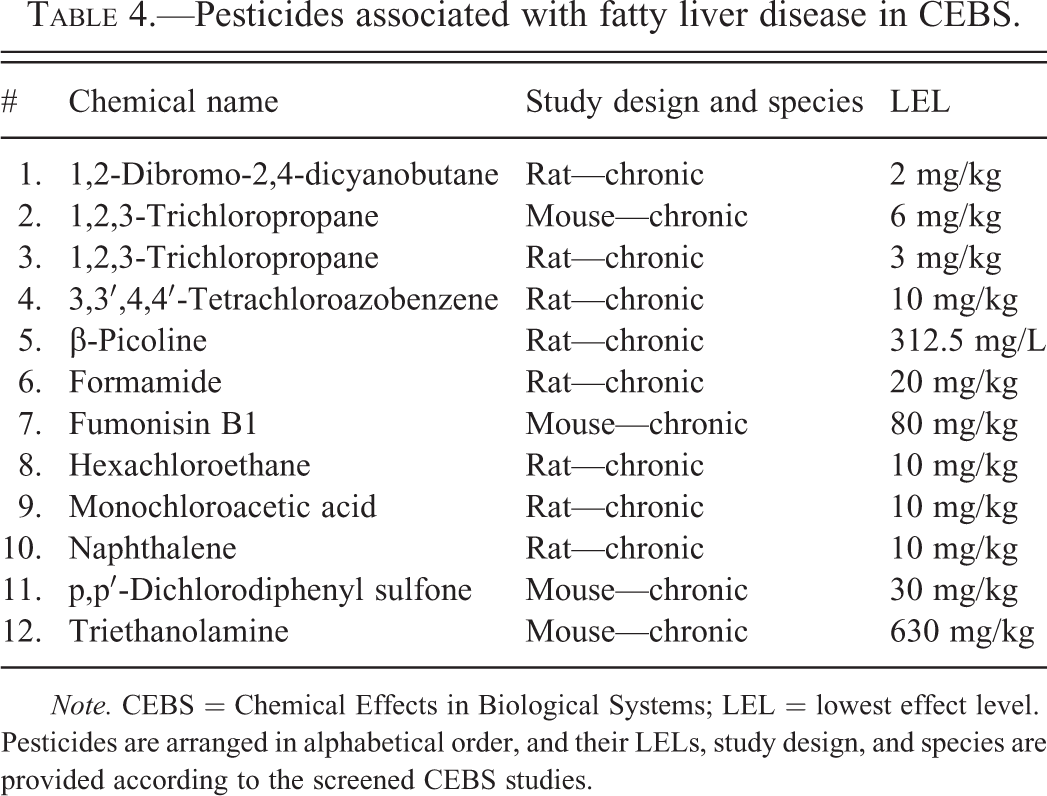
Note. CEBS = Chemical Effects in Biological Systems; LEL = lowest effect level. Pesticides are arranged in alphabetical order, and their LELs, study design, and species are provided according to the screened CEBS studies.
Fragrances, cosmetics, and essential oils associated with fatty liver disease in CEBS.
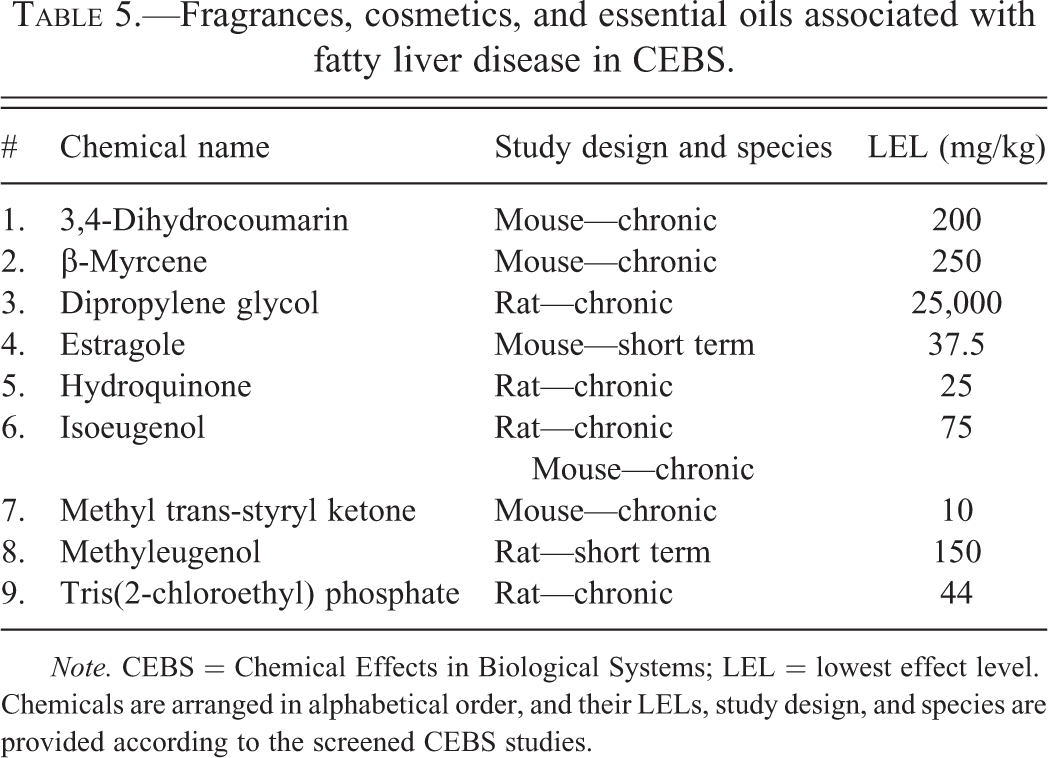
Note. CEBS = Chemical Effects in Biological Systems; LEL = lowest effect level. Chemicals are arranged in alphabetical order, and their LELs, study design, and species are provided according to the screened CEBS studies.
Paints, polishes, dyes, and food additives associated with fatty liver disease in CEBS.
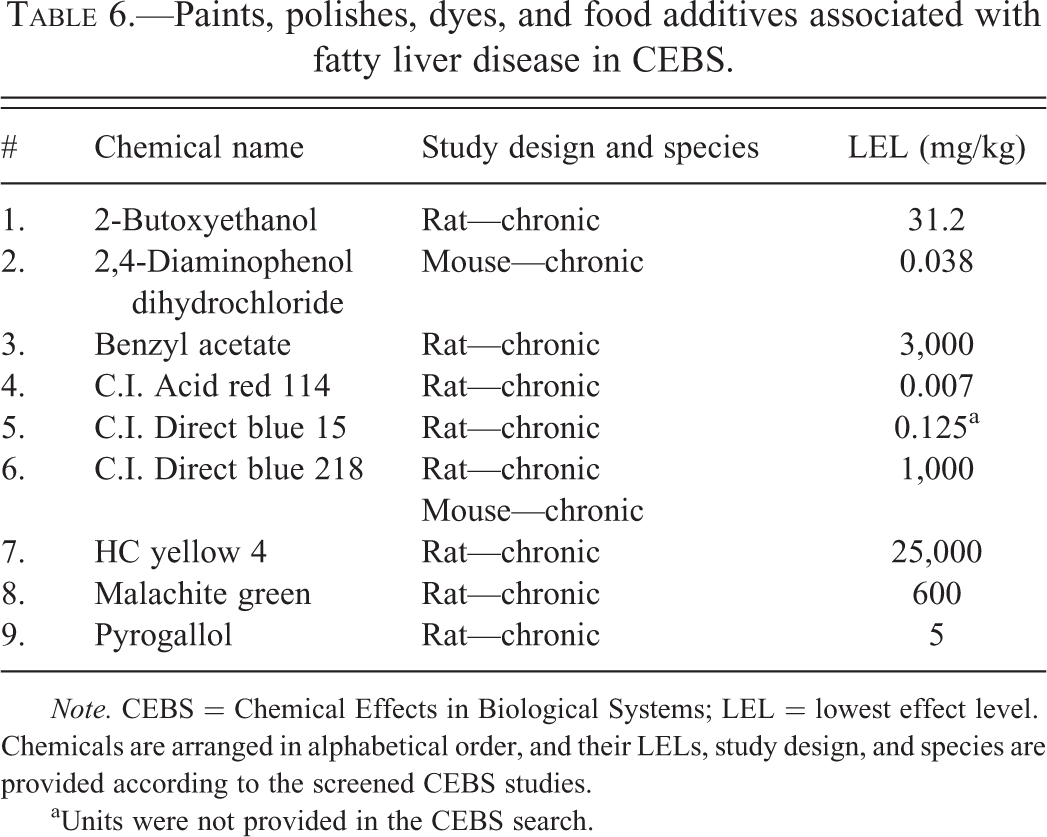
Note. CEBS = Chemical Effects in Biological Systems; LEL = lowest effect level. Chemicals are arranged in alphabetical order, and their LELs, study design, and species are provided according to the screened CEBS studies.
aUnits were not provided in the CEBS search.
PCBs and dioxin-like compounds associated with fatty liver disease in CEBS.

Note. CEBS = Chemical Effects in Biological Systems; LEL = lowest effect level. Chemicals are arranged in alphabetical order, and their LELs, study design, and species are provided according to the screened CEBS studies.
Discussion
Between CEBS and ToxRefDB, 371 studies linked 123 environmental chemicals to fatty liver disease in rodents. Pesticides composed almost 44% (54 of 123) of these chemicals and 14 of the 55 pesticides produced steatosis with LELs less than 10. According to the U.S. EPA, a pesticide is “any substance or mixture of substances intended for preventing, destroying, repelling, or mitigating any pest.” Although often misunderstood to refer only to insecticides, the term pesticide also applies to herbicides, fungicides, and various other substances used to control pests. Pesticides are a double-edged sword because they increase crop yields while simultaneously contaminating the food supply. The liver is responsible for the detoxification of these xenobiotic compounds primarily through cytochrome P450 enzymes, such as CYP3A and CYP2B families, that initiate the first step of the detoxification process (Nebert and Gonzalez 1987; Nelson et al. 1996). Thus, it is not surprising that the liver is a target organ for pesticide toxicity. Indeed, many pesticides have been previously associated with fatty liver disease and aminotransferases elevation (reviewed in Wahlang et al. 2013).
The present study increases the understanding of the role of pesticides in steatohepatitis. In particular, the potential roles of fungicides and herbicides in steatohepatitis appear to have been previously underestimated. Fungicides and herbicides are widely used for agricultural, residential, and industrial purposes (Reigart 2013). According to EPA, global annual fungicide application is nearly 500 million pounds (Reigart 2013). Some azole antifungals including triadimefon, propiconazole, and cyproconazole have been previously associated with hepatotoxicity and hepatomegaly in rats (Hester et al. 2006; Peffer et al. 2007). Interestingly, dazomet and hexaconazole were associated with fatty liver disease at relatively low LELs in multiple studies in ToxRefDB. Dazomet is a fungicide, herbicide, and nematicide that in CHR mice studies produced hepatomegaly combined with large droplet steatosis (EPA 2008; Chemical Review Section National Registration Authority 1997). Hexaconazole is a systemic triazole fungicide mainly used for the banana black and yellow sigatoka disease control (EPA 1999). Hexaconazole was associated with hepatic enzyme elevation, hepatocellular hypertrophy, and hepatic fatty infiltration/changes in rodent and dog studies (EPA 1999). Interestingly, in the present study, dazomet, hexaconazole, and 8 other pesticides in ToxRefDB were associated with steatosis descriptors in multigenerational reproductive studies. This may be the first evidence linking developmental pesticide exposures to fatty liver disease.
After pesticides, solvents were the class with the second highest number of chemicals (n = 31) associated with steatosis in rodents. Solvents have been associated with hepatotoxicity since the late 1800s (Brautbar and Williams 2002). Solvents are primarily used in industrial and military applications but also have residential uses and contaminate the environment (Brautbar and Williams 2002). Many solvents like VC are halogenated hydrocarbons. Previous studies link exposure to VC and other haloalkanes/haloalkenes to steatohepatitis (Cave et al. 2012). Furthermore, a Brazilian study on volatile petrochemical mixtures demonstrated that these chemicals caused NASH in exposed plant workers (Cotrim et al. 1999). It is therefore not surprising that our study yielded 31 chemicals from this class, which has historically been the class most associated with steatohepatitis.
Paints, polishes, and dyes have also been previously linked to liver disease. Abnormal serum transaminases have been reported in painters, but this may have been due to solvent coexposures (Dossing et al. 1983; Zimmerman 1999). Likewise, liver enzyme elevation was reported in 44% of shoe repairmen (Tomei et al. 1999). Moreover, Nigerian vat dye workers had increased serum transaminases levels (Soyinka, Adeniyi, and Ajose 2007). Three of the chemicals associated with TAFLD in CEBS were azo dyes. Azo dyes are used as well to color textile, fabric, leather, and papers. Exposure to azo food dyes including tartrazine and carmoisine resulted in aminotransferase elevation in a rodent study (Amin, Hameid, and Elsttar 2010). Four chemicals associated with TAFLD in this study are used for cosmetic purposes including hair coloring. Hair dyes have been proposed to influence the development of liver disease (Prince, Ducker, and James 2010). The potential role of food coloring additives and cosmetics in the development of NASH is intriguing due to their widespread use, and more data are needed.
Five PCBs/dioxins were associated with steatosis descriptors in this study; and all had very low LELs. PCBs are polychlorinated hydrocarbons that were commercially produced in the 1930s to 1970s (Silberhorn, Glauert, and Robertson 1990). PCBs are thermodynamically stable persistent organic pollutants, and thus PCB exposure remains relevant, although PCBs were banned in the 1970s. PCB exposures have been associated with suspected NALFD in epidemiological studies including the 2003 to 2004 adult National Health and Nutrition Examination Survey (NHANES; Cave, Appana, et al. 2010). PCBs were also found to cause hepatomegaly and slight increases in hepatic enzymes in workers in electrical capacitor factories in Taiwan (Yu et al. 1997). We recently demonstrated that a nondioxin-like PCB, PCB 153, worsened diet-induced obesity and steatosis and was associated with hepatic antioxidant depletion (Shi et al. 2012; Wahlang et al. 2011). Likewise, dioxins have been associated with hepatic steatosis, in part due to increased lipid transport into hepatocytes (Angrish, Dominici, and Zacharewski 2013; Angrish et al. 2012; Lee et al. 2010).
This study is not without several weaknesses, foremost of which are the pathologic descriptors used. Although TAFLD is a progressive disease characterized by steatosis, inflammation, and fibrosis, only steatosis descriptors were searched. “Steatohepatitis” is not a searchable term in either CEBS or ToxRefDB. Because inflammation/fibrosis queries could not be cross-matched with steatosis descriptors in CEBS/ToxRefDB, these terms were not included in the study as surrogates for steatohepatitis. This is because if hepatitis/fibrosis were present, it would be unclear whether the underlying disease was steatohepatitis or another form of liver injury. Compounding the problem, precise terms for fatty liver disease such as steatosis were not available in some studies. Steatosis was not quantified and neither photomicrographs nor original publications were available on ToxRefDB to allow for independent confirmation of the findings. Toxic hepatopathy might have been defined differently in different NTP studies. This study was solely dependent on pathologic descriptors and failed to address modes of action. Potential human relevance, especially for compounds with high LELs, is uncertain, and the effects of coexposures (e.g., alcohol or high fat diet) were not addressed. Furthermore, only the lowest doses associated with fatty liver disease histologic descriptors are available at ToxRefDB. Determining chemicals not associated with fatty liver disease in CEBS was difficult due to the complexity of the database and lack of histologic descriptors for fatty liver disease in most of the chemicals studies.
Conclusions
From 371 studies archived in federal databases, 123 unique environmental chemicals were possibly linked to some form of fatty liver disease in rodents. Pesticides composed almost 44% of these chemicals. Thus, >10% of pesticide registration toxicity studies reported the development of fatty liver disease. Some of these compounds were linked to liver disease at very low LELs, ≤10 mg/kg, suggesting that these compounds could contribute to the development of steatohepatitis at environmentally relevant doses. Pesticides and solvents were the most frequently identified chemicals, while PCBs/dioxins were the most potent (e.g., had the lowest LELs). Moreover, 395 chemicals were not associated with fatty liver disease at their studied doses in ToxRefDB. Given the high prevalence of both obesity and alcoholism, coexposure to environmental chemicals, especially pesticides, may contribute to the development and progression of fatty liver disease. However, the effects of diet and alcohol on xenobiotic metabolism impacting TASH require further investigation. Therefore, these findings suggest that more research on the effects of pesticides, solvents, metals, and PCBs/dioxins in steatohepatitis is required.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by NIHS grants R01ES021375 (Cave, Prough), 5T35ES011564 (Prough), and K23AA018399 (Cave).
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
