Abstract
Batracylin (NSC-320846) is a dual inhibitor of DNA topoisomerases I and II. Batracylin advanced as an anticancer agent to Phase I clinical trials where dose limiting hemorrhagic cystitis (bladder inflammation and bleeding) was observed. To further investigate batracylin’s mechanism of toxicity, studies were conducted in Fischer 344 rats. Once daily oral administration of 16 or 32 mg/kg batracylin to rats for 4 days caused overt toxicity. Abnormal clinical observations and adverse effects on clinical pathology, urinalysis, and histology indicated acute renal damage and urothelial damage and bone marrow dysfunction. Scanning electron microscopy revealed sloughing of the superficial and intermediate urothelial layers. DNA damage was evident in kidney and bone marrow as indicated by histone γ-H2AX immunofluorescence. After a single oral administration of 16 or 32 mg/kg, the majority of batracylin was converted to N-acetylbatracylin (NAB) with a half-life of 4 hr to 11 hr. Mesna (Mesnex™), a drug known to reduce the incidence of hemorrhagic cystitis induced by ifosfamide or cyclophosphamide, was administered to rats prior to batracylin, but did not alleviate batracylin-induced bladder and renal toxicity. These findings suggest that batracylin results in DNA damage-based mechanisms of toxicity and not an acrolein-based mechanism of toxicity as occurs after ifosfamide or cyclophosphamide administration.
Introduction
Batracylin (8-aminoisoindolo[1,2-b]quinazolin-12(10H)-one; NSC-320846) is a heterocyclic arylamine that was originally synthesized by Bayer AG, Leverkusen, Germany. The National Cancer Institute (NCI) acquired batracylin as an investigational anticancer agent based on its antitumor activity in the mouse refractory colon adenocarcinoma 38 model and its demonstrated activity against solid tumors and multidrug-resistant murine leukemia P-388 cell lines (Plowman et al. 1988; Mucci-LoRusso et al. 1989). Studies subsequently showed that batracylin induced ATP-independent topoisomerase II (Topo II) inhibition (Luo et al. 1993; Meegalla et al. 1994) and DNA strand breaks as detected using γ-H2AX as a marker of replication-mediated DNA damage (Luo et al. 1993; Meegalla et al. 1994; Rao et al. 2007).
Preclinical safety evaluations of batracylin revealed that rats are much more sensitive than mice or dogs to batracylin-induced toxicity. Oral administration of batracylin at one-tenth the dose that was lethal to 10% (LD10) of mice was lethal in rats whereas only slight toxicity was observed in dogs that received 2 times the mouse LD50 (Dixit, Stedham, Maldinger, Lopez, El-hawari 1989). Pharmacology studies indicated that rats extensively acetylate batracylin to produce N-acetylbatracylin (NAB; Figure 1); however, dogs do not produce the metabolite, and mice produce relatively small amounts of it (NCI). In addition, oral administration of batracylin (590 mg/m2) resulted in systemic levels of NAB that were approximately 9 times greater in the rat than in the mouse (Ames, Mathiesen, and Reid 1991). Since the observed interspecies variation in batracylin metabolism was coincidental with the interspecies variation in toxicity, and since N-acetyltransferase (NAT2) was shown to N-acetylate batracylin in human liver samples (Stevens, Burkey, and McQueen 2000), it was hypothesized that metabolism of batracylin to NAB via NAT2 was responsible for the observed toxicity.
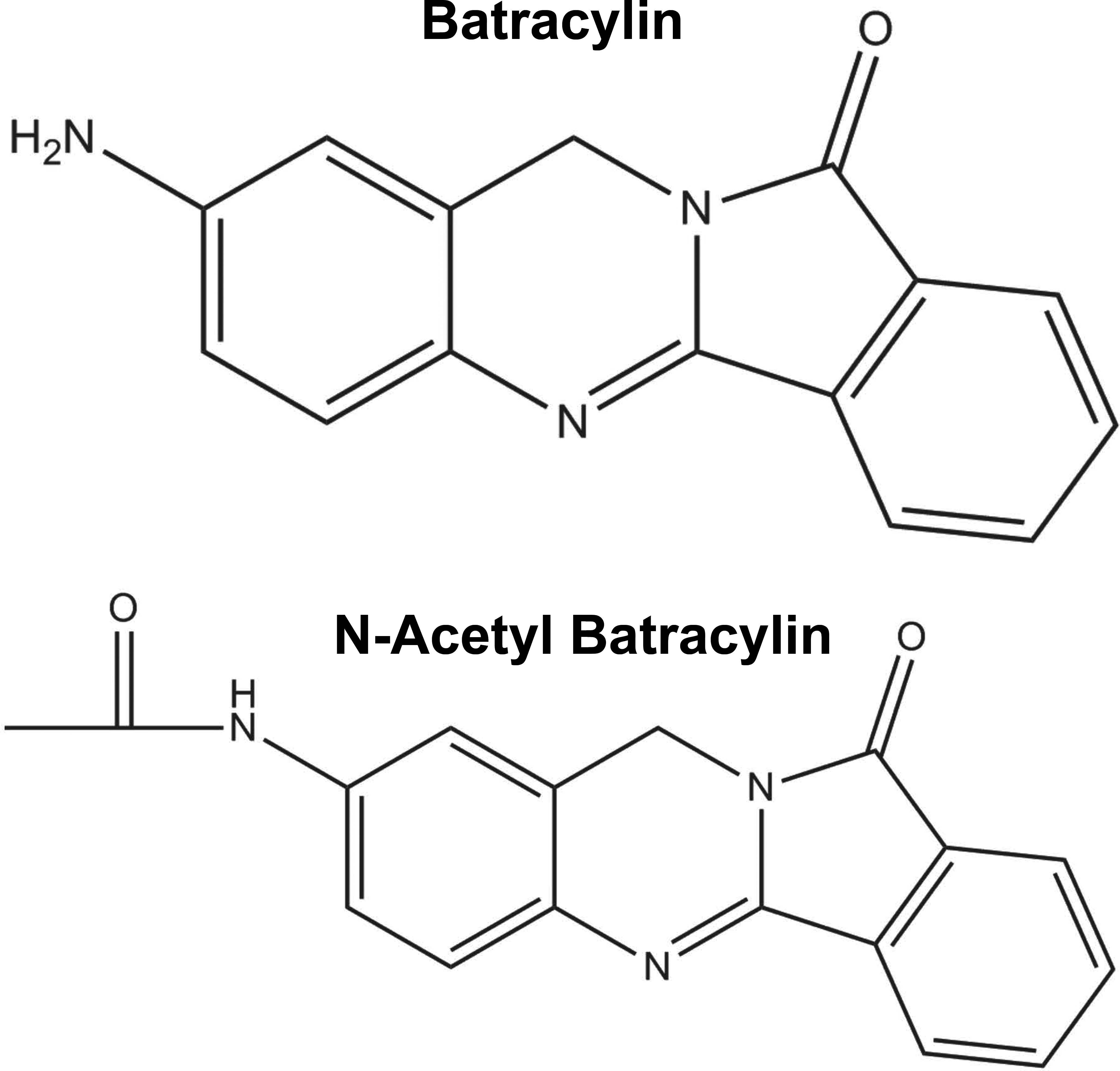
The chemical structures of batracylin and N-acetylbatracylin (NAB).
A phase I clinical trial to evaluate the safety and test the effectiveness of batracylin as a tumor-suppressing agent was sponsored by NCI and began in 2007 (Kummar et al. 2013). Trial eligibility criteria included the requirement that patients had a slow acetylator NAT2 genotype defined as NAT2*5, NAT2*6, NAT2*7, or NAT2*14. It was reasoned that batracylin might be safely administered in patients with a slow acetylator NAT2 genotype because, like dogs and mice and unlike rats, batracylin metabolism to NAB would be reduced in such patients. Despite the slow acetylator NAT2 genotype of the trial participants, dose limiting hemorrhagic cystitis was identified in the patients, and thus, bladder and kidney were postulated to be target organs of toxicity.
To further investigate the mechanism of batracylin-induced bladder and renal toxicity, a physiologically relevant animal model was needed. Rats were hypothesized to be an ideal model for such an investigation because earlier work showed rats to be particularly sensitive to batracylin. We also hypothesized that batracylin might cause hemorrhagic cystitis via a mechanism similar to that of other anticancer drugs such as ifosfamide and cyclophosphamide via reduction of protective cellular sulfhydryl groups within bladder and kidney (Brock, Pohl, and Stekar 1981).
Herein, we report the effect of oral batracylin administration on the rat bladder, bone marrow, and kidney using traditional toxicity assessments in combination with scanning electron microscopy (SEM) and document the extent of DNA damage in the kidney and bone marrow using γH2AX immunofluorescence (IMF). We also characterize the pharmacokinetics of batracylin and NAB in rat plasma, the amount of batracylin and NAB excreted in the urine, and the metabolites present in plasma and kidney homogenates. Once the rat model was characterized, we investigated whether mesna could alleviate batracylin-induced toxicity in rats. Our data suggest that batracylin-induced renal and urothelial damage in rats is related to DNA damage, a previously identified mechanism of action for batracylin. Our data also imply that the observed toxic effects of batracylin on bladder and kidney are not dependent on reactive intermediates that are detoxified by sulfhydryl conjugation as has been previously described for compounds like ifosfamide and cyclophosphamide.
Materials and Methods
Test System
Male and female Fischer 344 rats (8–12 weeks of age) from Harlan (Livermore, CA) were maintained on Purina Certified Rodent Chow 5002 (Richmond, IN) and reverse osmosis purified tap water ad libitum under controlled lighting (12-h light-dark cycle) in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)-accredited facility. Their use was approved by the facility Institutional Animal Care and Use Committee and procedures for animal care, and housing were in accordance with the National Research Council (NRC) Guide for the Care and Use of Laboratory Animals (1996) and the Animal Welfare Standards incorporated in 9 CFR Part 3, 1991. Animals were acclimated ≤5 days before initiation of treatment and housed 2 to 3 per micro-isolater cage except when housed singly in metabolism cages for sample collection.
Test Articles
Batracylin (NSC-320846; lot CK5-63-2; Starks Associates Inc, Buffalo, NY) and sodium 2-mercaptoethane sulfonate (mesna; lot 069L1613; Sigma Aldrich, St. Louis, MO) were supplied by Fisher BioServices (Rockville, MD). The vehicle for batracylin was 25%:75% Tween® 80 (Spectrum Chemical Mfg. Corp., Gardena, CA) and saline (0.9% sodium chloride for injection; USP; Baxter Healthcare, Marion, NC), and the vehicle for mesna was saline. Batracylin and mesna formulations were prepared under yellow light using aseptic technique by dissolving the test article in the vehicle to achieve the target concentrations, were used within 6 to 9 days or 24 hr of preparation, respectively, stored refrigerated, and protected from light. Batracylin suspensions were homogenized and sonicated before use. All formulations were mixed with a magnetic stirrer before use.
Study Designs
Animals were randomly assigned to treatment groups by a computerized body weight stratification procedure (Labcat In-Life v 8.0 SP2; Innovative Programming Associates, Inc, Lawrenceville, NJ). Dose levels for batracylin were based on previous toxicology studies conducted in rats in which renal toxicity was observed, but urinary bladder was not examined. Batracylin was administered by oral gavage (p.o.), once daily on days 1 to 4 to male and female rats (6/sex/group) at 0, 16, or 32 mg/kg (10 ml/kg) with sacrifice on day 4 for the toxicity assessment and was administered as a single 16 or 32 mg/kg p.o. dose to male rats for the pharmacokinetic assessment. To evaluate mesna as a mitigating co-treatment, 6 male rats/group were administered vehicle or 32-mg/kg batracylin p.o. once daily for 4 consecutive days and were also administered twice daily intraperitoneal (IP) injections of either vehicle or 80 mg/kg mesna (1 hr pre- and 4 hr post-batracylin administration) with sacrifice on day 5 (group 1 received both vehicles, group 2 received mesna and vehicle of batracylin, group 3 received batracylin and mesna, and group 4 received batracylin and vehicle of mesna).
Rats were examined for clinical signs of toxicity 2 hr to 4 hr post-batracylin dose. Body weights were recorded on day 1 and at sacrifice. Blood was collected from the retro-orbital sinus under 60% CO2/40% O2 anesthesia for clinical pathology (4 hr postdose on day 4 from 5 to 6/sex/group) and for pharmacokinetics (3 males/group at predose, 15-min, 30-min, and 60-min, 2-hr, 4-hr, 6-hr, 12-hr, and 24-hr post-batracylin dose) using K3EDTA anticoagulant. Urine was collected on day 4 without preservative from individual rats using metabolism cages (beginning ∼1 hr before dose administration and ending ∼4 hr postdose, 5 to 6/sex/group) for urinalysis and on wet ice 0- to 8- and 8-hr to 24-hr postdose for measurement of batracylin and NAB levels in urine (5 males/group). Kidneys were collected from 3 males/group at 4 hr, 12 hr, and 24 hr after a single batracylin dose for metabolite analysis. At 4-hr postdose on day 4 (5–6/sex/group), kidneys were collected for histopathology and γH2AX IMF analysis, urinary bladders were collected for histopathology and SEM analysis, and bone marrow was collected for γH2AX IMF analysis. For the mesna study, clinical pathology, urinalysis, and kidney and bladder histopathology and SEM analysis were evaluated from all surviving males on day 5.
Pharmacokinetic Analysis
Batracylin and NAB levels in plasma, urine, and kidney homogenates were quantified by liquid chromatography mass spectrometry (LCMS). Stock solutions of batracylin, NAB, and d3-datracylin (internal standard) were prepared in 1:1 (v/v) methanol: water and stored at –20°C. Working standard solutions were prepared daily by diluting the stock solutions with 1:1 (v/v) methanol: water and storing on ice. Assay standards containing batracylin and NAB were prepared by addition of 5.0 µl of working standard solutions. Rat kidney homogenates were prepared in cold phosphate buffered saline (PBS; 2 ml PBS/gm tissue). After addition of 5 μl 20 μg/ml d3-batracylin to 100 µl of sample, the proteins were precipitated with 300 μl methanol and allowed to stand at room temperature for 10 min. After centrifuge (14,000 rpm, 5 min, 25°C), the supernatants were filtered through a Captiva NDlipids plate (Agilent) and collected in a polypropylene 96-deep well plate. The supernatants were concentrated to dryness under a gentle stream of nitrogen, reconstituted in 100 μl 1:1 MeOH:H2O and transferred a glass autosampler vial for LC/MS/MS analysis. High-performance liquid chromatography (HPLC) separations were achieved on a reversed-phase column (Genesis C18, 2.1 × 100 mm, 4 µ particle size; Vydac) with a mobile phase consisting of 22:78 (v/v) acetonitrile:1 mM ammonium formate, pH 3.5. For quantitative determinations, a micromass triple quadrupole mass spectrometer (Waters Corporation, Milford, MA) in the positive electrospray mode was used. Detection was accomplished by multiple reaction monitoring mass spectrometry using the M+H m/z transition 250 > 130 for batracylin; the M+H m/z transition 292 > 130 for NAB; and the M+H m/z transition 253.1 > 130 for the internal standard (d3-batracylin). Metabolite detection was accomplished by scanning from a start mass of 50 m/z to an end mass of 500 m/z. Maximum plasma concnetration (C max), time to maximum plasma concentration (T max), half-life, and area under the concentration curve (AUC) values were estimated by standard non-compartmental analysis methods using WinNonlin (Professional Version 5.0; Pharsight Corp; Mountain View, CA).
Clinical Pathology and Urinalysis
Standard methods were used to measure hematology and clinical chemistry parameters (Advia 120 Analyzer, Bayer HealthCare, Tarrytown, NY and Cobas c501 Chemisty Analyzer, Roche Diagnostics, Indianapolis, IN). The following parameters were measured in addition to those listed in Table 1: monocytes; eosinophils; basophils; mean corpuscular hemoglobin, volume, and hemoglobin concentration; alkaline phosphatase; potassium; phosphorus; and calcium. Urine samples were examined for pH and levels of glucose, bilirubin, ketones, occult blood, protein, urobilinogen, nitrite, and leukocytes using Multistix 9 Reagent Strips and Clinitek Advantus Analyzer (Siemens Healthcare, Malvern, PA). Urine specific gravity was read using a digital refractometer (Atago USA, Bellevue, WA).
Summary of altered clinical pathology parameters after 4 daily doses of batracylin.
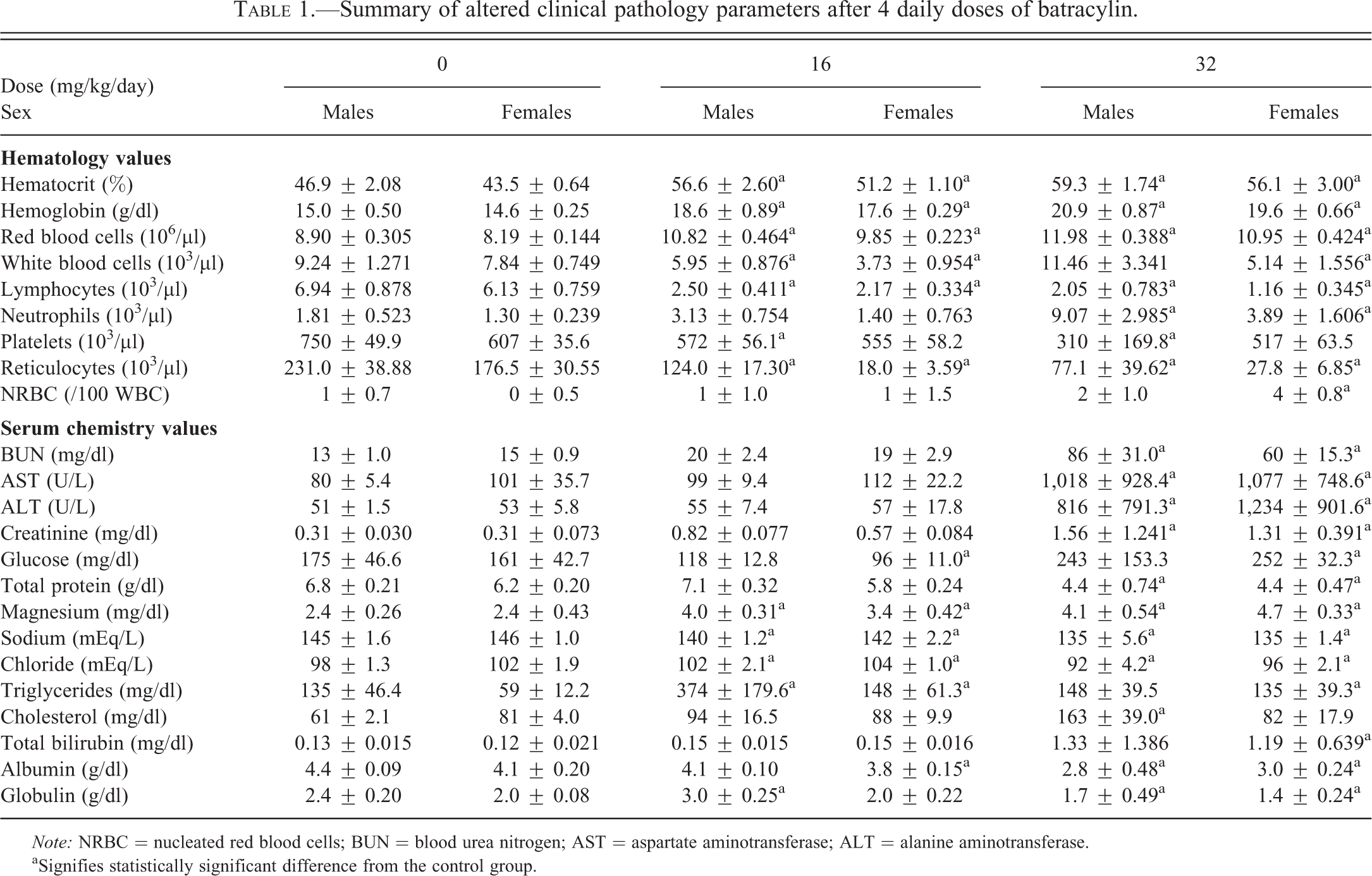
Note: NRBC = nucleated red blood cells; BUN = blood urea nitrogen; AST = aspartate aminotransferase; ALT = alanine aminotransferase.
aSignifies statistically significant difference from the control group.
Histopathology and SEM
The right kidney and half of the bladder were collected, fixed (formalin for kidney; 2% paraformaldehyde/2% glutaraldehyde/0.1 M phosphate buffer, pH 7.4 for bladder), embedded in paraffin, cut approximately 5-µm thick, and stained with hematoxylin and eosin for histopathologic examination. Bladders were inflated, harvested, fixed, and divided in half-longitudinally as per (Cohen et al. 2007). Half of the bladder from 2 rats/sex/group were processed for SEM as follows: dehydration through an ascending ethanol series; critical point dried out of ethanol (Autosamdri-814, Tousimis Research Corporation, Rockville, MD); mounted on SEM stubs using double sided, conductive carbon adhesive and “iron cross” cuts to flatten the tissue; gold coated by plasma sputtering at partial vacuum (0.17 mBar, 20 sec; Cressington 108 Sputter Coater, Cressington Scientific Instruments Ltd., Watford, England); and imaged at a working distance of 15 mm and an accelerating voltage of 5 kV at 20 μA using the SEM instrument (JEM 6700, JEOL Ltd, Tokyo, Japan). Representative SEM images were captured at 250×, 500×, 1,000×, and 2,500× magnification. A board-certified veterinary pathologist identified and scored lesions evident by light microscopy and SEM using a 4-step grading system (minimal, mild, moderate, and marked).
Biomarker Analysis
Kidney and bone marrow collection
Wedge biopsies centered about the hilum and angling through the medulla were cut from left kidneys to generate 2 top pieces of mostly cortex with cranial renal capsule and some underlying medulla, 2 wedge-shaped biopsies containing the hilum and medulla, and 2 bottom pieces of mostly cortex with caudal renal capsule and some medulla. Each piece was individually snap-frozen with liquid nitrogen and stored at ≤–60°C. Bone marrow was collected from both femurs via aspiration with PBS. Samples were gently triturated 5 times through a syringe needle. The total volume was adjusted to 8 ml with PBS then injected into a CellSave Preservative Tube (Veridex LLC, Raritan, NJ), mixed by inversion, and stored at room temperature.
γH2AX IMF staining of kidney sections
Determination of γH2AX in paraffin-embedded kidneys was performed with a biotin-conjugated preparation of anti-phospho-histone H2AX (Ser139), clone JBW301 (Millipore Corp., Billerica, MA). Biotin conjugates were developed with Streptavidin Alexa Fluor 488 (Life Technologies, Grand Island, NY). Staining procedures used the Bond-max Autostainer (Leica Microsystems, Buffalo Grove, IL), Bond-max reagents, dispense volumes of 150 μl, and working solutions of 10 μg/ml γH2AX biotinylated antibody prepared in Bond Primary Antibody Diluent and 10-μg/ml Steptavidin Alexa Fluor 488 prepared in 1× bond wash solution (BWS). Citrate buffer (pH 6) heated to 95°C was used for antigen retrieval. DAPI (4′,6-Diamidino-2-Phenylindole, Dihydrochloride; Life technologies, Grand Island, NY) was used as a counterstain for nuclei.
All 3 pieces of kidney were prepared on each slide in duplicate. Five fields were imaged from 1 slide from 1 rat/group as an aid in interpreting the acquired images. All images were captured on a Leica DM5500B (for phase contrast) or Nikon 90i fluorescent microscopes equipped with a Retiga 2000R CCD camera (QImaging). A semi-automated, custom macro capture script was designed and operated with Image-Pro Plus v6.2 software (Media Cybernetics, Rockville, MD). Images were acquired by pseudo-random selection of regions of interest after unacceptable areas were excluded using phase contrast in association with UV (DAPI) excitation. Fields were imaged in the blue channel (Chroma A4 filter, BP 360/40) to measure DAPI-signal nuclear staining and then in the green channel (Chroma L5 filter, BP 480/40) to measure Alexa Fluor 488–labeled conjugated γH2AX JBW301 antibody. At least three fields from each specimen section were selected in the DAPI channel (200× magnification) to minimize field selection bias and to ensure that selected fields had significant cellularity. Background autofluorescence was controlled by selecting a region with minimal cellularity and avoiding areas with high intrinsic fluorescence. Excitation exposure was adjusted by the computer script comparing, in real time, the calculated difference between the fluorescence channel intensity of the mean background intensity of the field being viewed to a predetermined intensity threshold. The resulting monochromatic capture images (DAPI and γH2AX) were merged into a single 8-bit RGB image for analysis. Illuminated pixels were counted in each channel (DAPI and γH2AX) and signal intensity was evaluated as a yes or no event relative to the dispersed background signal in the green channel (γH2AX). Quantitative analysis of γH2AX fluorescence was performed generally as outlined in Redon et al. (2011); Dzierzbicka, Januchta, and Skladanowski (2012).
γH2AX IMF staining and analysis of bone marrow
γH2AX analysis was carried out as outlined in Kummar et al. (2011). Cytospin spots of 1.5 × 106 fixed cells were prepared on Superfrost* Plus slides (Thermo Fisher Scientific Inc.) using a Shandon Cytospin 4 (Fisher Scientific). After air-drying (2 min), slides were stained at room temperature using the automated Bond-Max System (Leica Microsystems). Slides were rinsed once with 5% hydrogen peroxide and 3 times with BWS; incubated for 1 hr with 1.5-μg biotin-conjugated anti-γH2AX clone JBW301 in Bond Antibody Diluent; washed (3×, 5 min) with BWS; incubated for 30 min with 0.75 μg Alexa Fluor 555 goat anti-mouse immunoglobulin G (H+L; Molecular Probes, Life Technologies) in Bond Antibody Diluent; washed (2×, 5 min) with BWS; and washed (3×, 5 min) with deionized water. Cover slips were mounted using ProLong gold anti-fade reagent with DAPI (Life Technologies). Slides were examined using Zeiss LSM 510 meta (inverted) confocal laser scanning microscope with plan-apochromat 63×/1.40 oil DIC M27 objective and AIM software (Carl Zeiss Microscopy GmbH, Jena, Germany). For each sample, 3 to 6 images were captured to analyze a total of 200 to 300 nuclei. The percentage of nuclei positive for γH2AX was calculated using Image-Pro software (Media Cybernetics).
Statistical Evaluations
Mean and standard deviation were calculated for body weight, urinalysis (specific gravity and pH), and clinical pathology data and evaluated by one-way analysis of variance (ANOVA), followed by Dunnett’s test (if the ANOVA was significant) with p value ≤ .05.
Results
Batracylin Pharmacokinetics
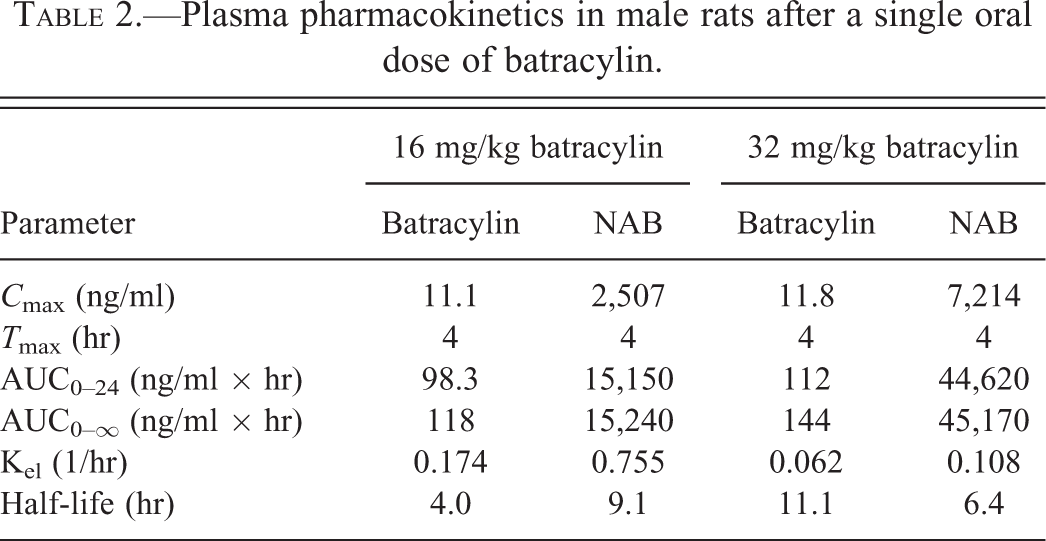
Plasma levels of batracylin and NAB in rats after oral administration of 16 or 32 mg/kg batracylin were determined. Nearly all the absorbed dose appeared in plasma as NAB. The plasma pharmacokinetic parameters are presented in Table 2 and plotted in Figure 2. Elimination half-lives were 4 and 11 hr for batracylin and 6 and 9 hr for NAB in the low- and high-dose groups, respectively. After high-dose administration, NAB C max and AUC0–24 hr values were 611 and 398 times greater, respectively, than the corresponding values for batracylin. The C max for batracylin did not increase substantially with dose, but the AUC0–24 hr increased 1.5-fold for a 2-fold increase in dose. The C max and AUC0–24 hr for NAB increased greater than proportionally with dose, with each parameter demonstrating increases of 2.9-fold. The T max was 4 hr for both compounds at both dose levels. Urinary excretion of both batracylin and NAB was low and comparable in both dose groups with an approximately 3% mean cumulative recovery of batracylin plus NAB over 24 hr. Consistent with the plasma pharmacokinetics, urinary excretion of NAB was substantially greater than that of batracylin.
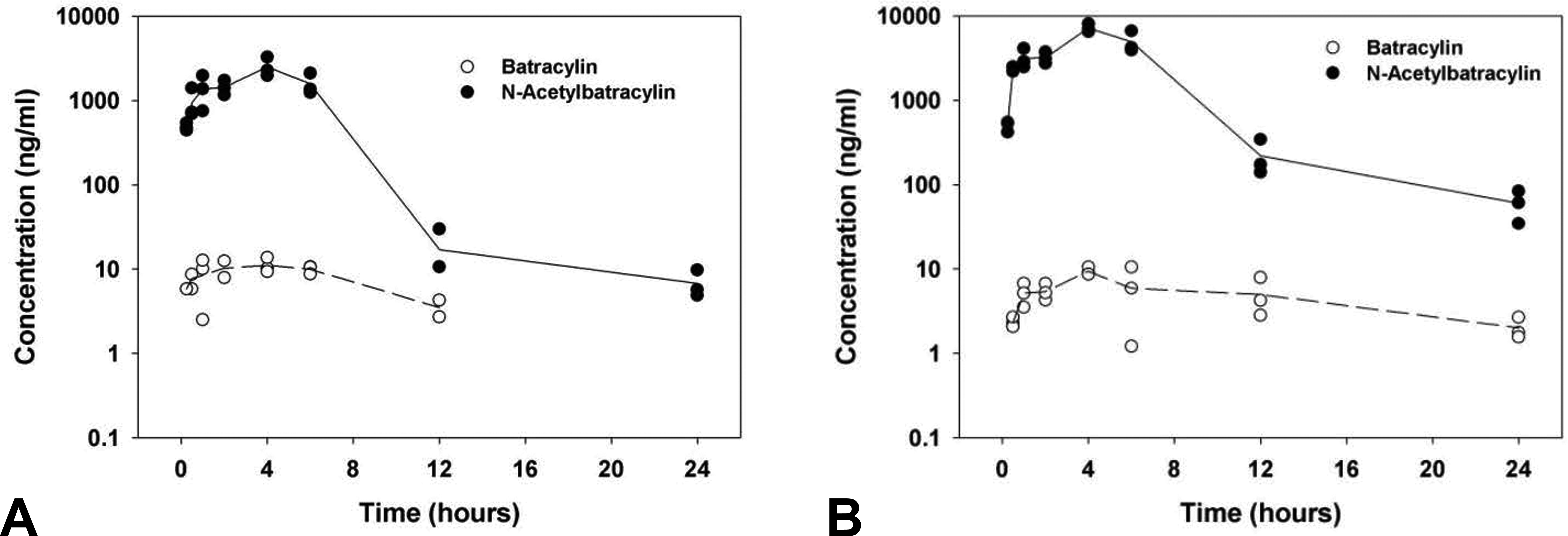
Plasma pharmacokinetics following oral administration of batracylin. Pharmacokinetic profiles are shown for plasma levels in rats following (A) 16 mg/kg or (B) 32 mg/kg batracylin.
Plasma pharmacokinetics in male rats after a single oral dose of batracylin.
Metabolites other than NAB were also detected in plasma and kidney homogenates. Specifically, hydroxyl-NAB (m/z 308), batracylin-N-glucuronide (m/z 426), dihydroxy-NAB (m/z 324), and other polar conjugates were detected in plasma and kidney while 3 additional putative glutathione conjugates were detected in kidney, batracylin-s-cysteine (m/z 369), batracylin-s-glutathione (m/z 555), and NAB-s-glutathione (m/z 597).
Clinical Signs and Body Weights
Rats administered batracylin at 16 and 32 mg/kg/day appeared normal on days 1 and 2 with the exception of a few instances of fecal stains, soft stool, and discolored red nostrils. On day 3, a third of the rats in the low-dose group and all of the rats in the high-dose group displayed evidence of toxicity (e.g., hunched posture, ruffled fur, hypoactivity, fecal and urine stains, soft stool, and diarrhea; n = 6/sex/group). By day 4, the severity and incidence of the clinical signs had increased, and findings were observed in all of the batracylin-treated rats. Most clinical signs on days 3 and 4 had severity scores of slight to moderate. Body weight loss was also observed in the batracylin-treated rats. Males and females weighed on average 10% and 15% less, respectively, on day 4 than they did on day 1, irrespective of dose level, while the mean body weight for the control rats increased approximately 1% over the 4-day interval.
Clinical Pathology Evaluations
Hematology
Administration of batracylin resulted in many hematologic changes as shown in Tables 1 and 3. Dose-dependent increases of 118% to 139% of control levels were observed in hematocrit, hemoglobin, and red blood cell counts suggesting slight dehydration. Large decreases of 46% to 90% were seen in absolute and percentage reticulocytes, and nucleated red blood cell counts increased in the high-dose group females, both of which suggest that batracylin has effects on the bone marrow. Platelet counts decreased in low- and high-dose group males (24% and 59%, respectively) and also in females, although the decreases reached statistical significance only in the males. Decreases were also seen in the total number of white blood cells (WBC), and there were dose-dependent changes in the composition of the WBC population in both sexes consistent with the observation of acute renal damage and associated blood loss. Specifically, neutrophils increased while lymphocytes, monocytes, basophils, and eosinophils decreased in response to batracylin administration. For example, neutrophils increased from 19% to 78% of the total WBC population in control group and high-dose group males, respectively, and lymphocytes decreased from 75% to 19% of the total WBC population in control group and high-dose group males, respectively.
Summary of altered urinalysis parameters after 4 daily doses of batracylin.a
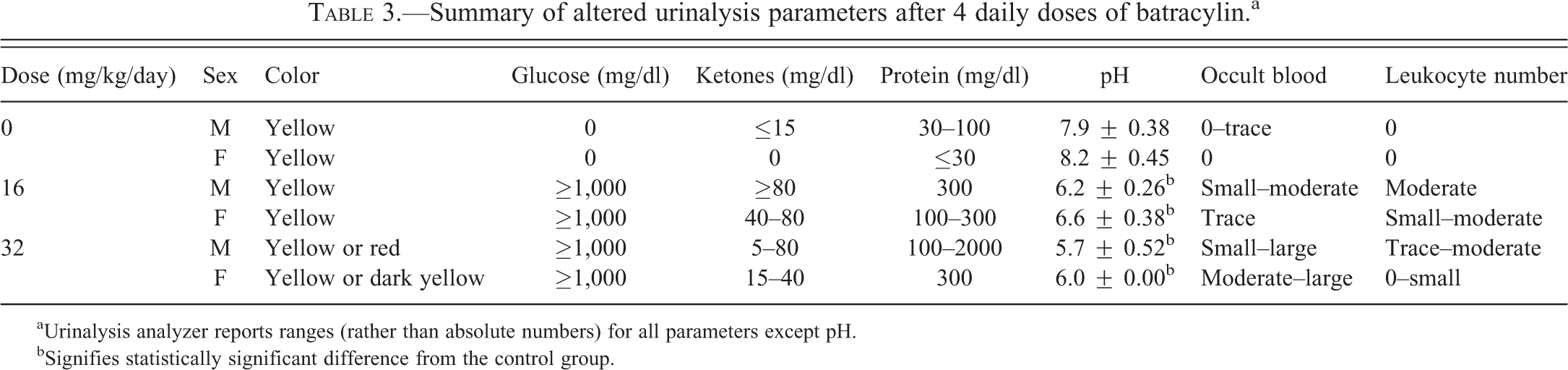
aUrinalysis analyzer reports ranges (rather than absolute numbers) for all parameters except pH.
bSignifies statistically significant difference from the control group.
Clinical chemistry
A number of clinical chemistry parameters were altered following batracylin administration as shown in Table 1 that are consistent with acute renal damage and associated liver dysfunction. The most notable increases in the high-dose group males and females compared with their control group counterparts were seen in blood urea nitrogen (BUN; 7- and 4-fold increases), aspartate aminotransferase (AST; 13- and 11-fold increases), alanine aminotransferase (ALT; 16- and 23-fold increases) and creatinine (5- and 4-fold increases). Imbalances in serum electrolytes such as magnesium, sodium, chloride, and calcium were also observed as were increases in triglycerides and total bilirubin, and decreases in total protein, albumin, and globulin. Glucose and cholesterol levels were altered in both dose groups but in opposite directions. For example, cholesterol levels were increased in the low-dose groups but decreased in the high-dose groups.
Urinalysis
A number of urinalysis parameters were altered in the batracylin-treated rats as shown in Table 3. Glucose is normally absent from urine; however, in all of the batracylin-treated rats, glucose levels were ≥ 1,000 mg/dl of urine. In addition, increases in ketone and protein levels and decreases in pH were observed in both dose groups. Occult blood was present in the urine of all batracylin-treated rats with the urine visibly red in 4 of the 6 high-dose male rats. Leukocytes were also present in the urine from both the low- and high-dose group males and in the low-dose group females.
Histopathology and SEM Evaluation
Histopathology evaluation revealed dose-dependent lesions in the kidneys and urinary bladder of batracylin-treated animals as detailed in Table 4. Treatment-related kidney lesions included protein casts (minimal to moderate), tubular dilation (minimal to mild), noninflammatory tubular degeneration, and congestion (mild to moderate). Treatment-related urinary bladder lesions included urothelial atrophy (minimal to moderate), vacuolation (minimal to mild), and desquamation (minimal to mild; Figure 3). Other histopathologic findings did not differ meaningfully between the control and batracylin-treated rats (data not shown). SEM images also showed treatment-related lesions of the urinary bladder in batracylin-treated rats including cell-cell junction disassembly; desquamation of superficial or intermediate urothelial cells; intermediate cells or deep connective tissue visible through defects in the superficial, or superficial and intermediate urothelial layers; intermediate layer cells lining the bladder surface in place of superficial urothelial cells; and smooth surfaces of superficial urothelial cells instead of the normal, heavily wrinkled surface (Figure 4).
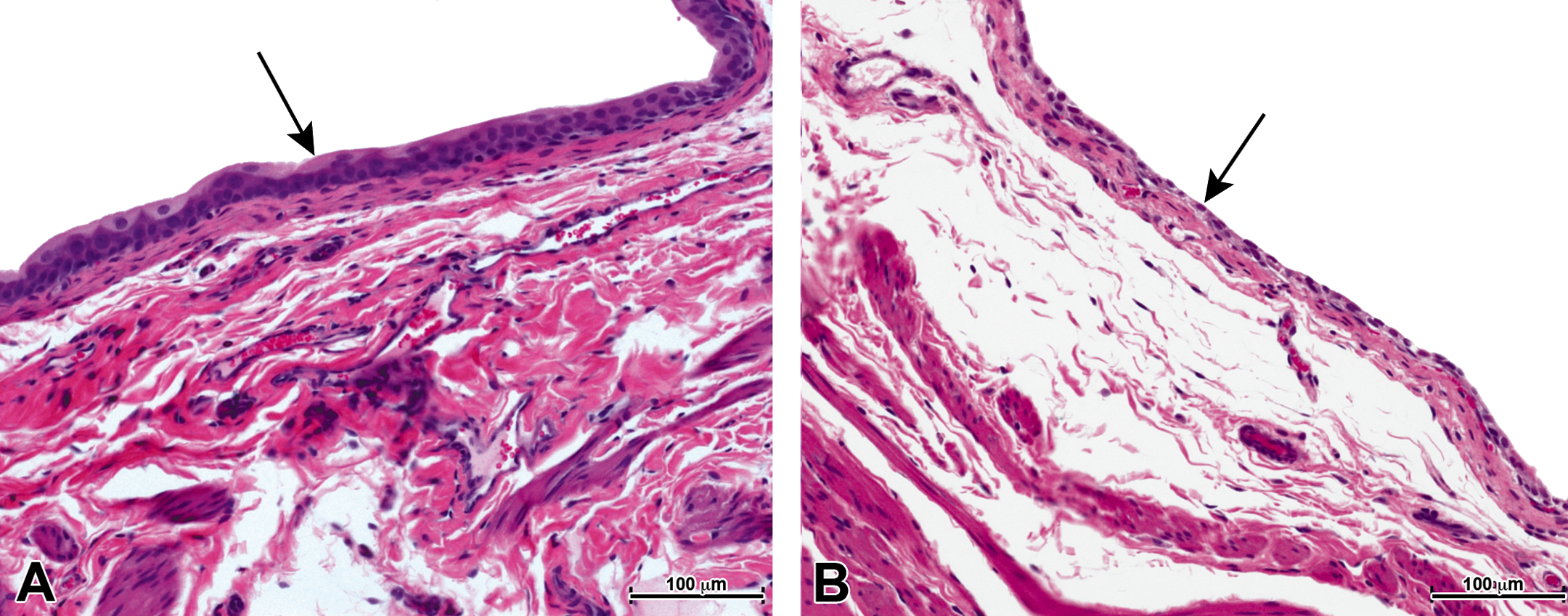
Light microscopic images of batracylin-related histopathology in urinary bladder. (A) Normal bladder from vehicle control rat showing normal height of uroepithelium (arrow). (B) Representative bladder from a rat administered batracylin orally (32 mg/kg, once daily for 4 days) showing urothelial atrophy and desquamation (arrow). Hematoxylin and eosin stain. Panels A and B 200× magnification; bar = 100 µm.
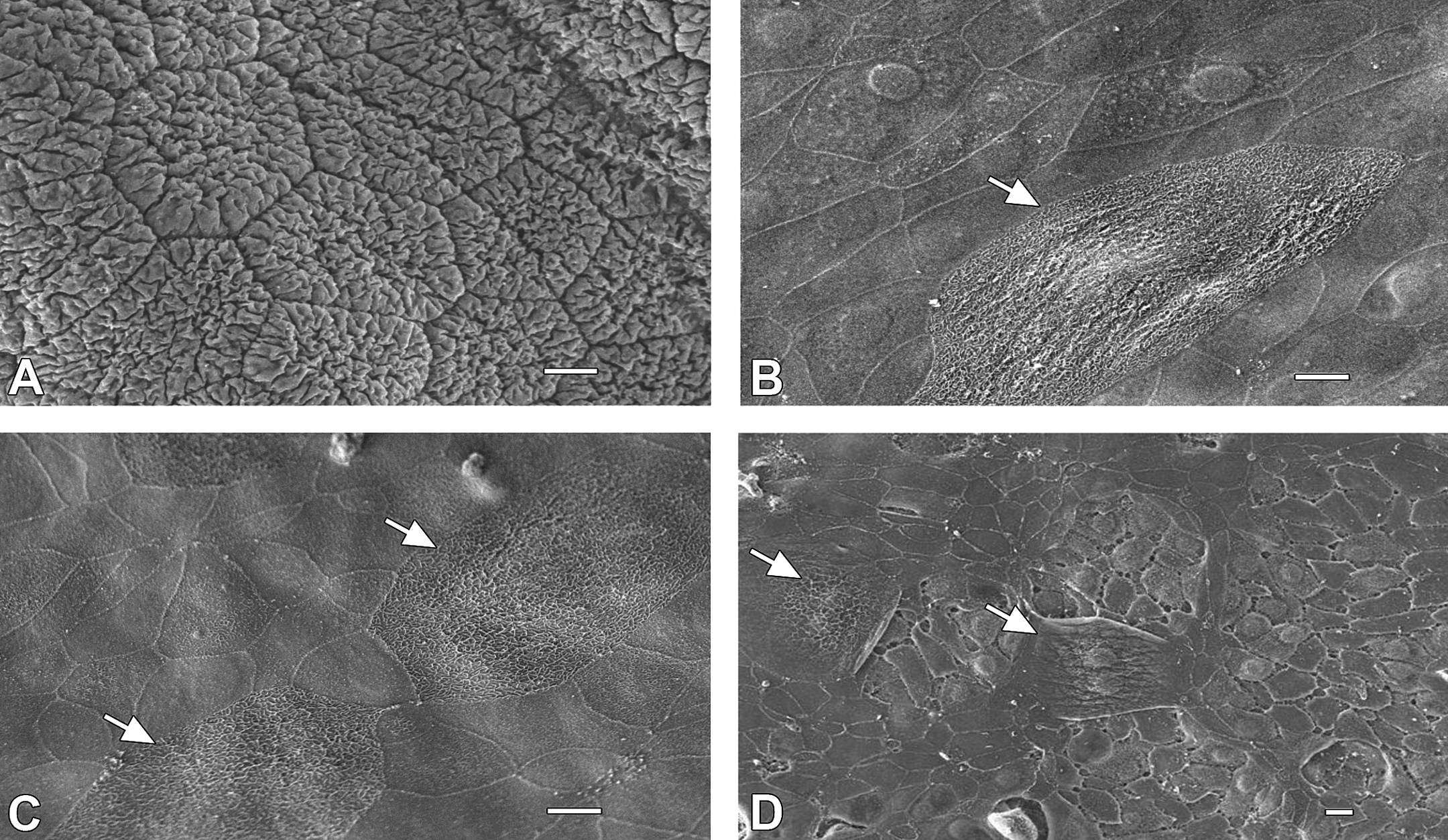
SEM images of batracylin-related urothelial lesions. (A) Normal bladder epithelium from control rats consists of tightly joined, large, heavily wrinkled, polygonal superficial cells. (B) Batracylin administered orally (32 mg/kg) results in desquamation of superficial or intermediate urothelial cells; intermediate cells or deep connective tissue visible through defects in the superficial, or superficial and intermediate urothelial layers; intermediate layer cells lining the bladder surface in place of superficial urothelial cells; and smooth surfaces of superficial urothelial cells instead of the normal, heavily wrinkled surface. (C) Urothelium was similarly damaged after administration of mesna plus 32 mg/kg batracylin. (D) In more severe cases, damage occurred to both the superficial and intermediate layer cells exposing deep connective tissue (32 mg/kg batracylin). Arrows in all panels pointing to cells that have surface corrugations and are enlarged. Panels A–C 1,000× magnification; bar = 10 μm and panel D 500× magnification; bar = 10 μm.
Severity and incidence of batracylin-related kidney and bladder lesions.
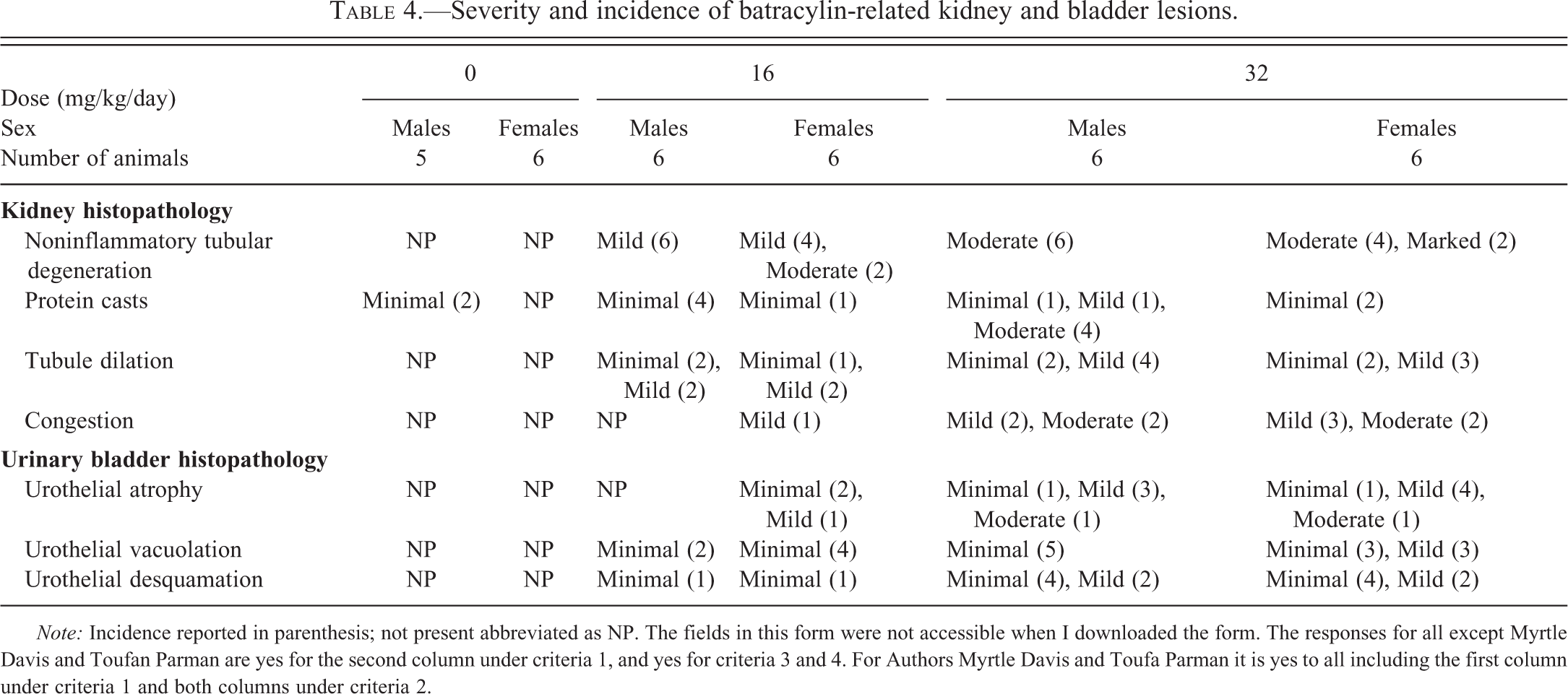
Note: Incidence reported in parenthesis; not present abbreviated as NP. The fields in this form were not accessible when I downloaded the form. The responses for all except Myrtle Davis and Toufan Parman are yes for the second column under criteria 1, and yes for criteria 3 and 4. For Authors Myrtle Davis and Toufa Parman it is yes to all including the first column under criteria 1 and both columns under criteria 2.
Effect of Batracylin on γH2AX IMF Staining of Kidney and Bone Marrow
IMF staining of sectioned kidneys and bone marrow revealed batracylin-induced phosphorylation of histone H2AX on serine 139. Phosphorylation at this site is termed γH2AX and occurs in response to DNA damage (Podhorecka, Skladanowski, and Bozko 2010). In kidneys from batracylin-treated rats, γH2AX expression was localized to the nuclei of tubules of the renal cortex with the staining usually appearing as ring structures along the inside nuclear membrane of the tubules (Figure 5). γH2AX localization was also related to chromatin margination and in some fields, apoptotic nuclei were evident (not shown). Glomerular nuclei and nuclei in the renal pelvis were both negative for γH2AX staining as were all sections from the vehicle control rats.
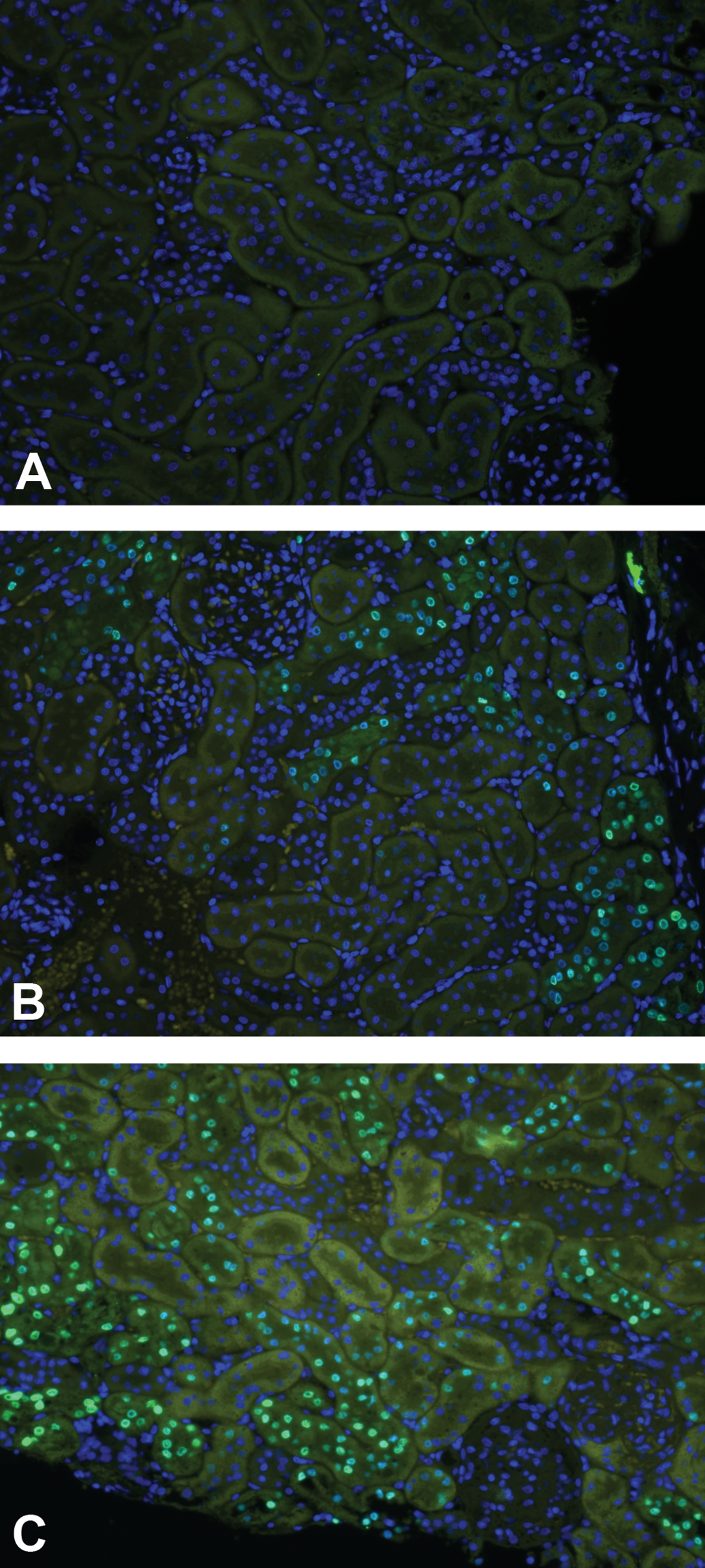
γH2AX staining in kidney sections from batracylin-treated rats. (A) Vehicle control kidneys do not express γH2AX. Less than 0.05% of the nuclei were γH2AX positive. Dose-dependent increases in the number of γH2AX positive renal tubule nuclei were observed in the renal cortex after oral administration of batracylin at (B) 16 mg/kg or (C) 32 mg/kg. Nuclei are stained blue with the DAPI counterstain. Specific γH2AX staining is considered to be nuclear staining (inner membrane appearing as green, nuclear rings). Positive nuclei are shown in the representative sections from rats treated with 16 or 32 mg/kg batracylin. Magnification 200×.
Upregulation of γH2AX in kidneys following batracylin administration was quantified by averaging the percentage of nuclei positive for γH2AX in 5 imaged fields from the renal cortex from 1 rat/sex/group (Figure 6). Less than 0.05% of the nuclei were γH2AX positive in vehicle controls; 13% and 22% of the nuclei were γH2AX positive in the male and female rat administered 16-mg/kg batracylin; and 29% and 15% of the nuclei were γH2AX positive in the male and female rat administered 32-mg/kg batracylin.
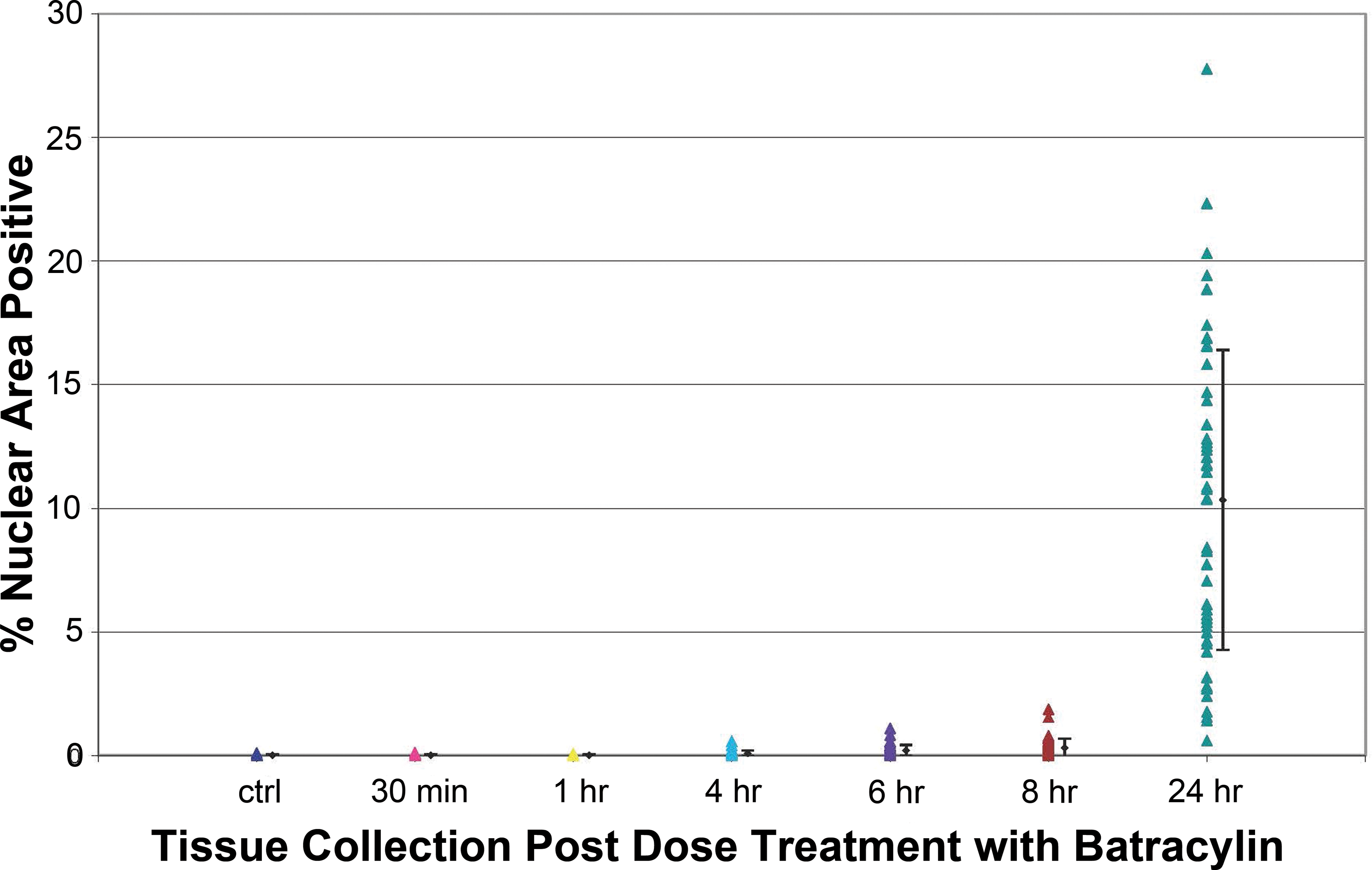
Quantitation of γH2AX in kidneys following batracylin administration. γH2AX was quantified by averaging the percentage of nuclei positive for γH2AX in 5 imaged fields from the renal cortex from 1 rat/sex/group for tissues collected at the times indicated on the x-axis. Individual tissues are represented by symbols and standard error bard are shown to the left of each group for clarity. Detailed analysis procedures are found in the Methods section.
Upregulation of γH2AX was also seen in cells collected from bone marrow, but occurred primarily after 32-mg/kg batracylin administration. Specifically, 1% to 9% of the bone marrow’s cellular nuclei were γH2AX positive in 5 of the 6 male and 5 of the 6 female rats from the high-dose group and 2 of the 6 female rats in the low-dose group. Less than 0.5% of the nuclei were γH2AX positive in all the vehicle controls, all 6 low-dose males, and in 4 of the 6 low-dose females.
Effect of Mesna on Batracylin-induced Toxicity
Twice daily administration of mesna 1 hr before and 4 hr after batracylin administration did not alleviate batracylin-induced renal or bladder toxicity. Body weight loss; hematology, clinical chemistry, and urinalysis changes; and kidney and bladder microscopic lesions were similar between the batracylin plus mesna group and batracylin alone group. Specifically, the male rats that received batracylin alone or batracylin plus mesna both lost approximately 18% of their day 1 body weight at sacrifice on day 5 while the control rats (mesna plus batracylin vehicle or vehicle for both compounds) gained 0.5% to 2% of their original body weight during the same time interval. In both the batracylin and batracylin plus mesna groups, hematology changes included large decreases in WBCs, platelets, reticulocytes, and many of the WBC differentials, and there were increases in the percentage of neutrophils. The serum chemistry changes for these two treatment groups included increases in BUN, AST, ALT, creatinine, glucose, magnesium, chloride, and cholesterol, and decreases in total protein, sodium, calcium, alkaline phosphatase and albumin. Urinalysis revealed red or orange colored urine of pH 6.0, large amounts of occult blood with trace leukocytes, and highly abnormal glucose, ketone, and protein levels in the urine of rats that received mesna and batracylin. Moreover, no mitigating action of mesna on batracylin-related kidney or urinary bladder histopathology were observed. Both groups had moderate noninflammatory tubular degeneration, mild to moderate protein casts, mild tubular epithelial regeneration, mild tubular dilation, mild to moderate urothelial atrophy, minimal to mild urothelial vacuolation, and intermediate, rather than superficial, cells lining the bladder.
Discussion
The main objective of this investigation was to characterize the effect of batracylin on rat urinary bladder and kidney histology using light microscopic and electron microscopic examination and to elucidate to some extent potential mechanisms of toxicity induced by batracylin on these organ systems. Batracylin administration caused acute renal damage as well as damage to the urothelium of the urinary bladder. Noninflammatory tubular degeneration, protein casts, tubular dilation, and congestion were observed in the kidney of batracylin-treated rats. The urinary bladder of these animals showed urothelial atrophy, urothelial vacuolation, and urothelial desquamation. SEM of the bladder revealed the presence of lesions that included cell-cell junction disassembly; desquamation of superficial or intermediate urothelial cells; intermediate cells or deep connective tissue visible through defects in the superficial, or superficial and intermediate urothelial layers; intermediate layer cells lining the bladder surface in place of superficial urothelial cells; and smooth surfaces of superficial urothelial cells instead of the normal, heavily wrinkled surface.
The clinical signs observed in these investigations (red discoloration of the nostrils, red discharge from the nostrils, hunched posture, ruffled fur, hypoactivity, fecal stain, soft stool, diarrhea, and urine stain) are consistent with the stress associated with the observed acute renal damage. The hematologic and the clinical chemistry measures are also consistent with acute renal damage and associated liver dysfunction following batracylin administration. The observed increase in total neutrophil and a decrease in total lymphocyte, eosinophil, and basophil counts are consistent with inflammation and/or stress; increases in RBC, HCT, and HGB and a decrease in MCV are associated with slight dehydration; decreases in WBC and total platelet counts are associated with loss of blood from the urinary tract; increases in serum BUN, CRE, and glucose and a decrease in serum total protein as well as an imbalance in serum electrolytes such as magnesium, sodium, chloride, and calcium are indicative of kidney damage. The significant increases in AST, ALT, total bilirubin, and cholesterol and significant decreases in ALB are all signs of liver dysfunction. The observation of substantial amounts of blood, protein, glucose, leukocytes, and ketones in the urine of treated animals as well as an increase in urine specific gravity and a decrease in pH in urine are consistent with acute renal damage and damage to the urinary bladder.
The nuclei of kidney and bone marrow cells from batracylin-treated rats stained positive for γH2AX and this indicates an increase in DNA double-strand breaks after treatment. This biomarker of DNA damage was seen in the tubules of the renal cortex, but not in the glomerular and renal pelvis nuclei. Moreover, γH2AX in the tubules of the renal cortex was localized to rings around the inside membrane of the nuclei, which suggests the observed events were coincident with initiation of apoptosis rather than late-stage apoptosis or necrosis (Qvarnstrom et al. 2009; Rogakou et al. 2000; Cook et al. 2009). The effect of batracylin on bone marrow was further supported by dose-dependent decreases in reticulocytes.
The toxic effects of batracylin (32 mg/kg administered for 4 consecutive days) on kidney and urinary bladder were not alleviated by twice per day administrations of mesna at 80 mg/kg. The chemotherapy agents ifosfamide and cyclophosphamide, like batracylin, induce hemorrhagic cystitis in humans. Acrolein, reactive oxygen species (ROS), and reactive nitrogen species are implicated in the bladder pathogenesis following administration with ifosfamide and cyclophosphamide. Mesna binds to acrolein when coadministered to patients taking ifosfamide and cyclophosphamide and prevents hemorrhagic cystitis (Elias et al. 1990). The fact that mesna did not protect when coadministered with batracylin is not surprising because there was no evidence from our pharmacokinetic analyses that batracylin leads to acrolien-like reactive intermediates, reactive free sulfhydryls, or other ROS. It must be noted, however, that local generation of reactive species in the target organs may not be revealed through the evaluation of metabolites in plasma. Analysis of tissues from batracylin-treated rats (and humans) for bound or unbound species might be required to rule out the possibility that reactive intermediates play a role in the observed toxicity.
We determined that bladder toxicity occurs in rats and that mesna is not protective, but without detailed histopathologic examination of human bladder derived from treated patients, we cannot determine if the organ pathology is the same in humans and rats after batracylin administration. Investigations to determine whether the observed renal and bladder toxicity are caused mainly by NAB or some other metabolite are next steps in order to build a translational bridge. After all, ratios of N-acetylation of arylamines, O-acetylation of arylhydroxylamines, and N, O-acetyltransfer of arylhydroxamic acids may be highly dependent on the conformation of the enzymes responsible and may contribute to species differences in the activation of aromatic amines. Published studies have suggested that CYP3A-mediated biotransformation of batracylin is involved in the formation of a cytotoxic product and this is likely to be species dependent (Stevens, Burkey, and McQueen 2000). The results of our study provide a solid basis for use of the rat model in toxicology studies to further investigate mechanisms of batracylin-induced toxicity.
Footnotes
Acknowledgment
The authors would like to thank Dr. Sandy Eldridge for donating her time and valuable assistance in formatting the figures for submission.
Authors’ Note
The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in part) by the Developmental Therapeutics Program in the Division of Cancer Treatment and Diagnosis of the National Cancer Institute under NCI Contracts N01-CM-42203, and N01-CM-2011-00028.
