Abstract
Orally administered small molecule agonists of soluble guanylate cyclase (sGC) induced increased numbers of osteoclasts, multifocal bone resorption, increased porosity, and new bone formation in the appendicular and axial skeleton of Sprague-Dawley rats. Similar histopathological bone changes were observed in both young (7- to 9-week-old) and aged (42- to 46-week-old) rats when dosed by oral gavage with 3 different heme-dependent sGC agonist (sGCa) compounds or 1 structurally distinct heme-independent sGCa compound. In a 7-day time course study in 7- to 9-week-old rats, bone changes were observed as early as 2 to 3 days following once daily compound administration. Bone changes were mostly reversed following a 14-day recovery period, with complete reversal after 35 days. The mechanism responsible for the bone changes was investigated in the thyroparathyroidectomized rat model that creates a low state of bone modeling and remodeling due to deprivation of thyroid hormone, calcitonin (CT), and parathyroid hormone (PTH). The sGCa compounds tested increased both bone resorption and formation, thereby increasing bone remodeling independent of calciotropic hormones PTH and CT. Based on these studies, we conclude that the bone changes in rats were likely caused by increased sGC activity.
Introduction
Soluble guanylate cyclase (sGC) is believed to be the primary intracellular receptor for nitric oxide (NO), a key signaling molecule regulating many biological and physiological processes in mammalian cells (Murad 2006). Binding of NO to sGC stimulates the formation of the second messenger, guanosine 3',5'-cyclic monophosphate (cGMP), which subsequently interacts with specific cGMP-dependent protein kinases (PKG), cGMP-gated ion channels, and cGMP-regulated phosphodiesterases (PDEs; Figure 1) to modulate various cellular functions (Koesling et al. 2005), including vasorelaxation and reduction of blood pressure (Stasch, Pacher, and Evganov 2011). Decreased NO availability and/or decreased responsiveness to endogenous NO are implicated in the development of numerous pathological conditions, including systemic hypertension, cardiopulmonary and renal diseases, and male erectile dysfunction (Hobbs 2002; Priviero and Webb 2010; Stasch, Pacher, and Evganov 2011). During the past several years, new classes of small molecule sGC stimulators or activators independent of NO have been identified (Evgenov et al. 2006; Ghofrani and Grimminger 2009; Priviero and Webb 2010; Stasch, Pacher, and Evganov 2011). These compounds have distinct profiles for their ability to induce cGMP production both in vitro and in vivo and may offer considerable benefit over current therapies for a broad range of conditions.
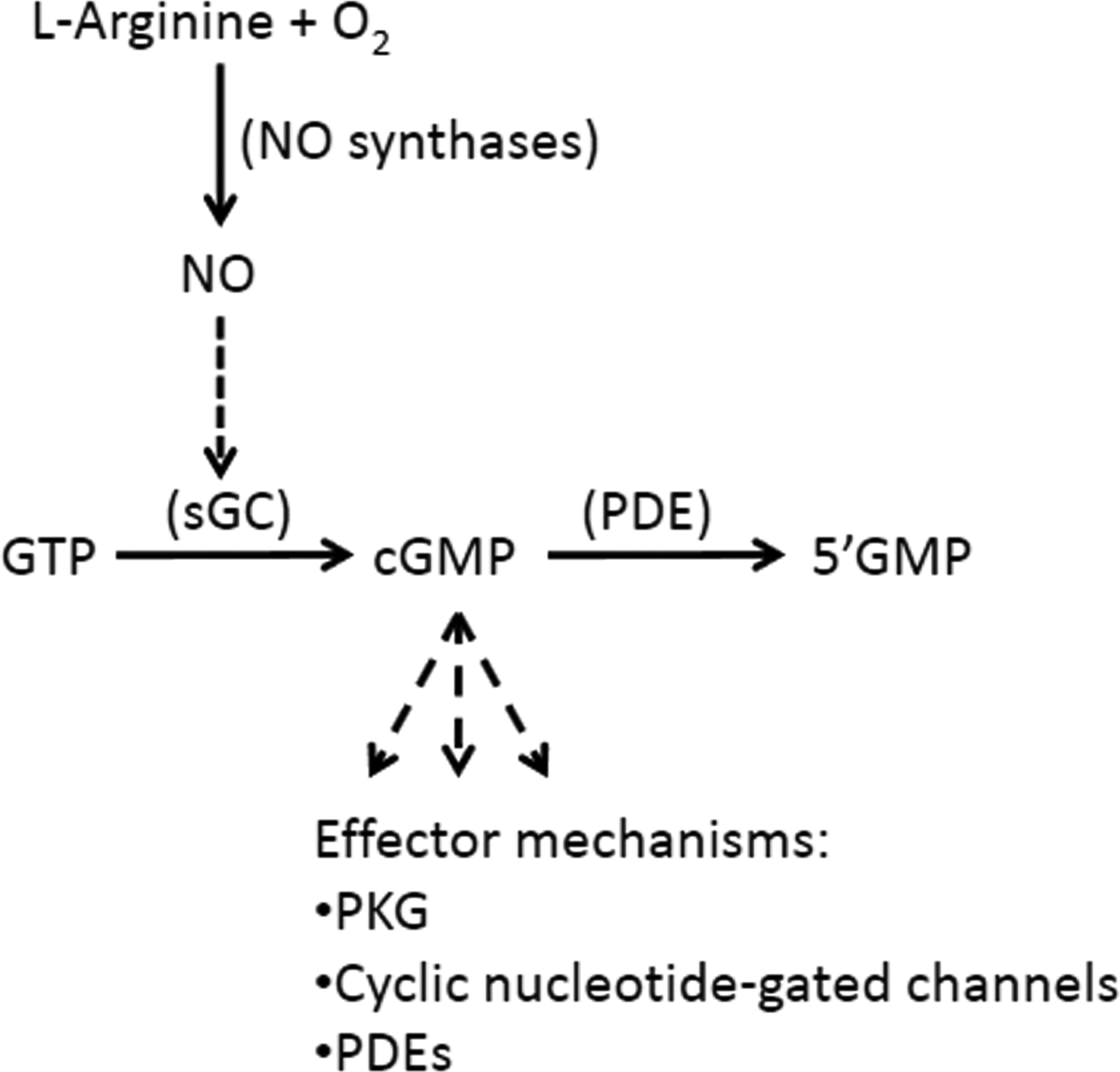
Nitric oxide–soluble guanylate cyclase–cGMP pathway.
Three antihypertensive small molecule heme-dependent (HD) sGC agonist (sGCa) compounds and a structurally unrelated antihypertensive heme-independent (HI) sGCa compound were evaluated in rats in 7-day exploratory toxicity studies. Treatment consistently resulted in varying degrees of osteoclasia, remodeling, and new bone formation in the femur and tibia. Therefore, investigative studies were designed to better characterize the effect of HD- and HI-sGCa compounds on bone in groups of rats of various ages, the time course and reversibility of bone changes, the distribution of bone lesions in appendicular and axial skeleton, and the role of PTH, CT, and thyroid hormone deprivation on the development of the bone changes. These studies were helpful to better understand the mechanism of the bone changes by sGCa compounds and risk assessment of the bone lesions.
Materials and Methods
Compounds and dosing formulations: Initially, three 7-day exploratory toxicity studies were conducted, each with a different antihypertensive heme-dependent soluble guanylate cyclase agonist (HD-sGCa; Table 1). These compounds (HD-sGCa-A, HD-sGCa-B, and HD-sGCa-C; Figure 2) were administered by oral gavage, at a dose volume of 10 ml/kg, as suspensions of the free base in a vehicle of 0.5% Avicel RC-591 (w/v) and 0.1% Tween 80 (v/v) in deionized or distilled water. An exploratory toxicity study with an antihypertensive HI compound, HI-sGCa-D (Table 1; Figure 2; same duration, dose volume, and vehicle), was conducted to test the hypothesis that the bone effect was a sGCa effect and was not a chemotype-associated off-target effect. In addition, HD-sGCa-A was administered in a time course study; HD-sGCa-B was administered in a reversibility study; HD-sGCa-C was administered in an aged rat study; and HD-sGCa-B, HD-sGCa-C, and HI-sGCa-D were each administered to thyroparathyroidectomized (TPTx) rats.
Exploratory toxicity studies with sGC agonist compounds in male rats.
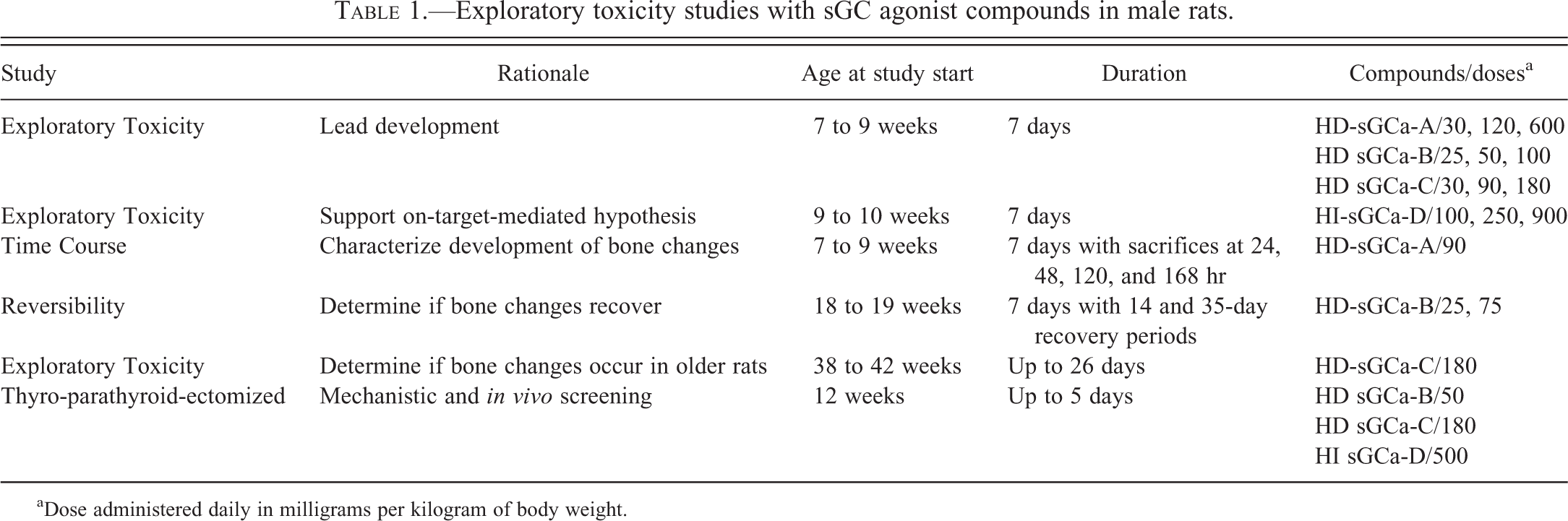
aDose administered daily in milligrams per kilogram of body weight.
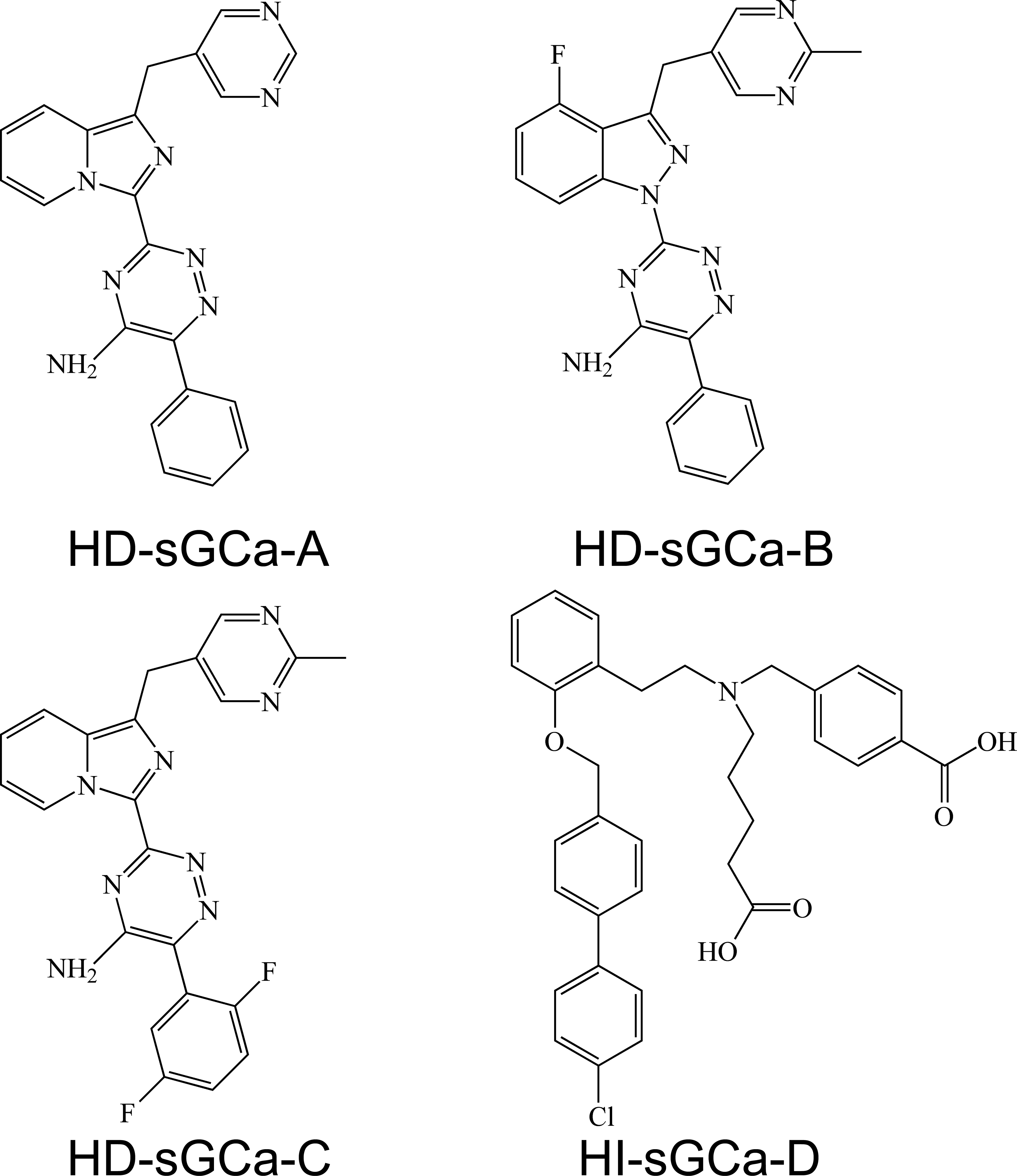
Chemical structures of sGCa compounds administered in toxicity and investigative studies. HD molecules are from same chemical series. HI molecule is structurally unrelated to the HD molecules and was administered to support the hypothesis that bone changes were related to sGC stimulation or activation.
Animals: All studies were approved by the Institutional Animal Care and Use Committee (IACUC) and were conducted in approved facilities in accordance with the current guidelines for animal welfare (Animals Scientific Procedures Act 1986 and ILAR Guide for the Care and Use of Laboratory Animals; National Research Council 1996). The animal facilities were accredited by Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC), International. All animals were male Sprague-Dawley (SD) rats [Crl: CD(SD) IGS BR rats, Charles River Laboratories, Wilmington, MA]. All compounds were administered by oral gavage.
In the exploratory toxicity and time course studies, HD-sGCa compounds were administered to 7- to 9-week-old rats weighing 175 to 250 g (Table 1). In the HI-sGCa-D compound toxicity study, rats were 9 to 10 weeks old, weighing 275 to 350 g. In the reversibility study, rats were 18 to 19 weeks old, weighing 475 to 525 g at study initiation. The aged rat exploratory toxicity study was conducted in 38- to 42-week-old males, weighing 600 to 800 g at study initiation. The TPTx model study was conducted in 12-week-old TPTx rats weighing approximately 350 g.
Rats were housed in individual cages in environmentally controlled rooms set to maintain a temperature of 22°C ± 3°C, humidity of 30 to 70%, and an approximate 12-hr light and 12-hr dark cycle. Water from the municipal source and a commercial pelleted rodent diet (LabDiet 5002, Purina, Richmond, IN) were available ad libitum. In lieu of plain water, TPTx rats were maintained on 1% calcium gluconate water during the surgical recovery period at the vendor and on 1% calcium lactate water during the experiment.
Younger rats were acclimated in the animal facilities for 1 week prior to the studies. Rats obtained for the aged rat study were 14 weeks old when purchased and were housed in the animal facility until used in the study.
Study Designs
Exploratory toxicity studies: In the initial 4 studies conducted with HD or the HI compounds, various daily doses of HD-sGCa-A, -B or -C or HI-sGCa-D were administered to groups of 5 rats (Table 1), and vehicle was administered to 5 vehicle control rats by oral gavage, once daily for 7 days. All rats were sacrificed 24 hr after the last dose. Blood was collected at intervals for 24 hr post-dose on day 1 and the last day of dosing for measurement of plasma drug concentration. At necropsy, right hind legs were collected, surrounding skin and muscle were trimmed away, and tissues were immersed in 10% neutral buffered formalin. In addition to bone, representative sections of mesentery with mesenteric arteries, brain, heart, liver, spleen, lung, kidney, thymus, mesenteric lymph node, stomach, small and large intestine, pancreas, skeletal muscle, testis, and epididymis were collected and processed for microscopic examination. Bone specimens were decalcified and sectioned for processing: single cross sections were cut through the proximal femoral and distal tibial diaphyses and the remaining distal femur and proximal tibia were sliced mid-sagittally, with the patella and ligaments of the stifle joint intact. All tissues were then processed through graded alcohol and xylene, infiltrated and embedded in paraffin, sectioned, stained with hematoxylin and eosin (H&E), and evaluated by light microscopy.
Time course study: In the time course study, HD-sGCa-A was administered to 20 rats and vehicle was administered to 5 rats by oral gavage, once daily at doses of 0 (vehicle only) and 90 mg/kg/day for up to 7 days (Table 1). The dose selected was based on the maximum tolerated dose identified in an exploratory toxicity study with this compound. This exposure approximated a 10-fold multiple over predicted human efficacious area under the concentration curve (AUC) defined by efficacious blood pressure lowering (10–20 mm Hg in mean arterial pressure) in the spontaneously hypertensive rat (SHR) model (Hughes et al. 2010; Webb et al. 1998). Five HD-sGCa-A-dosed rats each were sacrificed 24 hr after dosing for 1, 2, 5, and 7 days. The vehicle control rats were sacrificed 24 hr after 7 days of dosing. At necropsy, right hind leg and lumbar spine were collected. Legs were processed as described for the toxicity studies. After the lumbar spine was collected, surrounding skin and muscle were trimmed away, and tissues were immersed in 10% neutral buffered formalin. Spines were left intact, with the spinal cord in situ, decalcified and cut sagittally prior to processing.
Reversibility study: In the reversibility study (Table 1), HD-sGCa-B was administered to 2 groups of 24 rats, and vehicle was administered to 15 rats by oral gavage, once daily, at doses of 0 (vehicle only), 25, and 75 mg/kg/day for 7 days. At 24 hr after the last dose, 5 vehicle-dosed rats and 8 rats from each HD-sCGa-B dose group were sacrificed. The remaining rats entered into a recovery period lasting 2 weeks (5 vehicle-dosed rats and 8 rats from each HD-sCGa-B dose group) and 5 weeks (remaining 5 vehicle-dosed rats and 8 rats from each HD-sCGa-B dose group). The plasma drug concentrations for the doses selected approximated 10-fold and 20-fold multiples over predicted human efficacious AUC defined by efficacious blood pressure lowering in the SHR model. At necropsy, right hind leg, lumbar spine, and heads (with attached mandibles) were collected. The skin and muscle of the head were trimmed away; the caudal half of the skull was removed; and tissues, with brain in situ, were immersed in 10% neutral buffered formalin. Legs and spines were processed as described previously. Calvaria with attached mandibles were decalcified and sliced coronally prior to processing.
Aged rat study: Rats, aged 38 to 42 weeks, were administered either HD-sGCa-C 180 mg/kg/day (N = 13) or vehicle (N = 10) via oral gavage once daily for up to 26 days (Table 1). The dose selected was derived from an exposure that resulted in bone findings in 7- to 9-week-old rats within a 7-day dosing period. A 1-week pilot study in 38- to 40-week-old rats showed that targeted exposure could be achieved. Blood was collected at intervals for 24 hr post-dose on days 7, 14, 21 and 25 for measurement of plasma drug concentration. If a rat was sacrificed prior to day 26, the final plasma drug concentration was not measured. At necropsy, hind legs, lumbar spines, heads, and mandibles were collected and processed as described previously.
TPTx rat study: The TPTx rat model used in this study was described previously by Andresen et al. (2006). Study animals were assigned to the following 5 groups: vehicle control (7 control rats dosed with vehicle alone), TPTx vehicle (8 TPTx rats dosed with vehicle alone), TPTx HI-sGCa-D (8TPTx rats dosed with HI-sGCa-D at 500 mg/kg/day), TPTx HD-sGCa-C (8 TPTx rats dosed with HD-sGCa-C at 180 mg/kg/day), and TPTx HD-sGCa-B (8 TPTx rats dosed with HD-sGCa-B at 50 mg/kg/day; Table 1). Doses were administered by oral gavage once daily for 5 days, except for rats dosed with HD-sGCa-B or HI-sGCa-D, which were dosed for 4 and 2 days, respectively. These 2 groups of rats were sacrificed early due to signs of illness such as depressed activity, reduced body temperature, abnormal posture, and reduced response to stimulation. Blood (0.5 ml) for biomarker analyses was collected by the retro-orbital route under light CO2 anesthesia 3 hr following the final dose on day 5 for the vehicle and HD-sGCa-C dosed rats, on day 2 for the HI-sGCa-D dosed rats, and on day 4 for the HD-sGCa-B dosed rats. Immediately after the retro-orbital blood collection for biomarker analyses, terminal blood samples for plasma drug concentrations were collected from the caudal vena cava at necropsy. Tibias and distal femurs of all vehicle-dosed rats and rats administered HD-sGCa-C were collected at necropsy (day 5) and prepared for light microscopy as described previously. Serum analyses included assessment of markers in all dose groups, including rats dosed with HD-sGCa-D and HD-sGCa-B: C-terminal telopeptide of type I collagen (CTX) for bone resorption and osteocalcin (OC) for bone formation. Commercially available enzyme-linked immunosorbent assay (ELISA) kits were utilized for determination of CTX and OC (Nordic Bioscience, Denmark). Data from biochemical analyses were presented as mean ± SD. Statistical analysis of the data was performed by unpaired Student’s t test. Differences were considered to be statistically significant at a p value of <.05.
Bone severity grading criteria: Severity grading of multifocal bone resorption and porosity and of new bone formation was semiquantitative and based on recommended criteria for toxicity studies (Shackelford et al. 2002). The following criteria were used for bone resorption and porosity: 0 = normal bone; 1 (minimal) = 1 small area of cortical bone resorption, barely noticeable on low magnification 4× objective, containing 0 to 3 osteoclasts; 2 (mild) = 1 or 2 areas of bone resorption, more noticeable on low magnification, with more prominent scalloped edges (Howship’s lacunae), and containing 3 to 5 osteoclasts; 3 (moderate) = 1 or more porous areas of cortical bone resorption, obvious on low magnification, observed in both midsagittal and cross sections of bone, with 5 or more prominent osteoclasts; 4 (marked) = severity similar to 3, but more widespread involvement of bone including areas of trabecular bone resorption in the epiphysis and metaphysis, in addition to porous foci of bone resorption in the cortex of the femoral or tibial diaphysis. Criteria used for scoring new subperiosteal or endosteal woven bone were 0 = normal; 1 (minimal) = 1 thin area of new woven bone, barely noticeable on low magnification; 2 (mild) = 1 area of new woven bone, more obvious on low magnification; 3 (moderate) = 1 or 2 areas of subperiosteal and/or endosteal new bone formation, prominent on low magnification; 4 (marked) = noticeable displacement of periosteum or bone marrow elements by prominent new woven bone.
Results
Effects of sGCa Compound Administration on the Skeleton
After 7 days of dosing, the incidence and severity of bone morphological changes were dose-related and similar for the 3 HD and the HI sGCa compounds. Bone changes included increased numbers of osteoclasts and osteoclastic bone resorption and porosity in the tibia and femur, with variable formation of new woven bone lined by plump osteoblasts (Figure 3). The changes were multifocal and tended to be more marked in the cortical bone of the metaphysis and diaphysis (Figure 3B and C). With increasing severity, osteoclastic bone resorption also occurred in trabecular bone of the epiphyses, especially along the anterior aspect adjacent to the growth plate, and in the anterior aspect of the patella. In the most severe lesions, increased numbers of osteoclasts and osteoclastic bone resorption occurred in the spongiosa adjacent to the growth plates. No sGCa-related growth plate changes were observed. Porosity occurred multifocally in the cortex, and was most marked along the caudal aspect of the femur and anterior aspect of the tibia, particularly where muscles or tendons attached. Bone encompassing porous areas of resorption was often scalloped and lined with osteoclasts within Howship’s lacunae. In areas of marked osteoclastic resorption or increased cortical porosity in which foci of bone resorption were contiguous with the medullary cavity, bone marrow was sometimes replaced by stromal tissue (Figure 3D). Very occasional foci of necrosis were presented in the marrow cavity. Areas of bone remodeling and new bone formation in the metaphysis and diaphysis were most prominent in subperiosteal and endosteal cortical bone (Figure 3C and E). These areas were characterized by formation of woven cancellous bone and dilation of vascular spaces, occasionally with woven bone extending into the medullary cavity (Figure 3F). In some areas, new woven bone appeared to increase cortical thickness. Woven trabecular bone was often oriented in a different plane than the adjacent lamellar cortical bone.
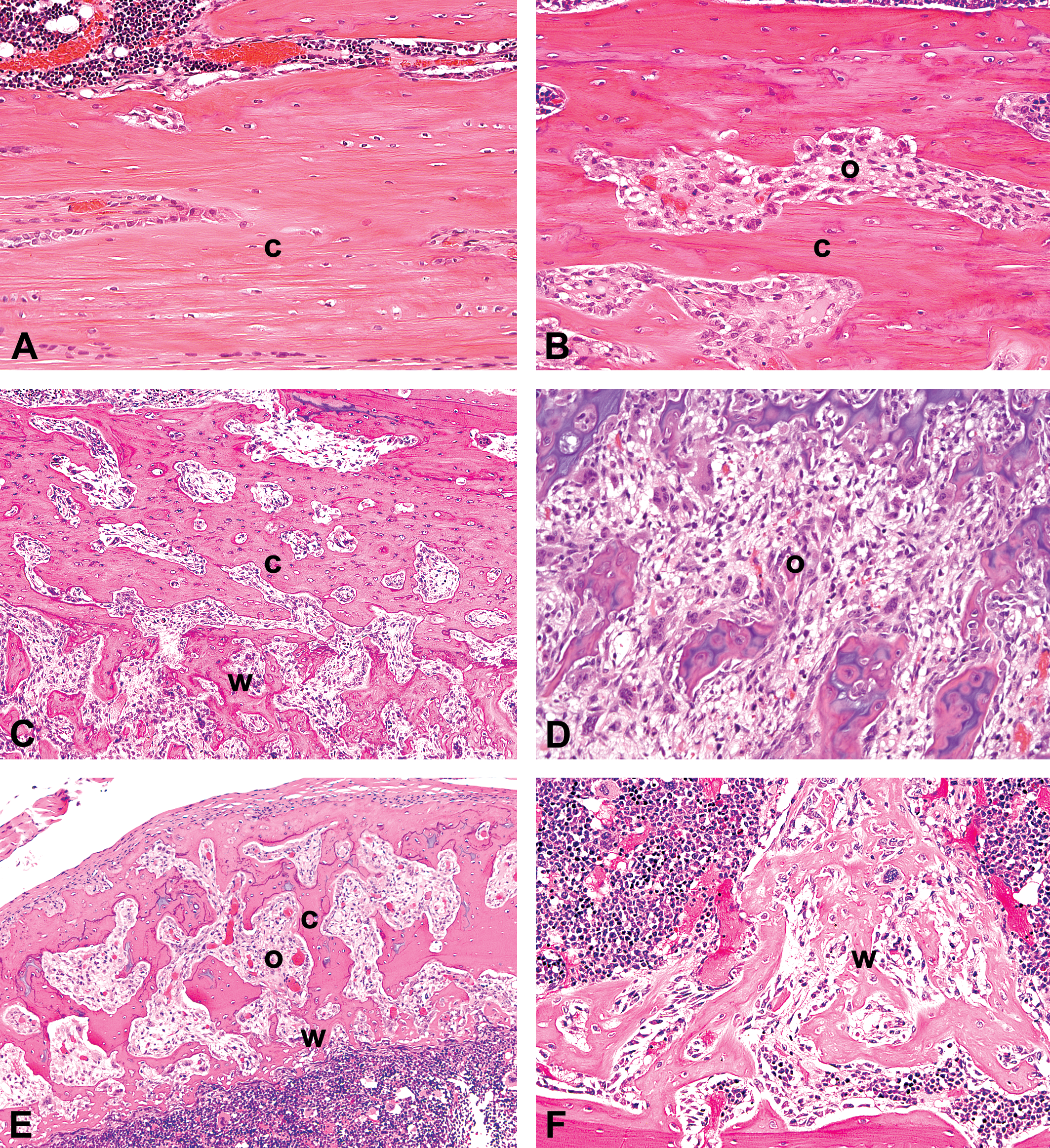
Photomicrographs of H&E stained longitudinal (A–D) and cross sections (E and F) of tibia and femur from a control rat and rats administered sGCa molecules daily in 7-day exploratory toxicity studies. (A) Control rat tibial diaphysis (×200). (B) Tibial diaphysis after dosing with HD-sGCa-B showing scalloped cortical bone lined by increased numbers of osteoclasts (×200). (C) Femoral diaphysis after dosing with HD-sGCa-B showing increased osteoclastic cortical bone resorption and porosity, and subperiosteal woven bone formation (×100). (D) Femoral metaphysis after dosing with HD-sGCa-B showing osteoclasia, loss of bone trabeculae in the primary and secondary spongiosa of the metaphysis, and marrow replaced by stromal tissue (×200). (E) Tibia after dosing with HI-sGCa-D showing increased cortical bone porosity and endosteal woven bone formation with increased osteoclasts (×100). (F) Tibia after dosing with HD-sGCa-A showing endosteal new woven bone trabeculae extending into the medullary cavity (×100). c = cortical bone; o = osteoclastic bone resorption; w = woven bone.
In follow-on studies in which spine and head were collected, changes were similar to those in the long bones after 7 days of dosing (Figure 4). Increased osteoclasts, osteoclastic bone resorption, and increased porosity were the predominant changes, occurring in the ventral cortex of the vertebral body (Figure 4B) and in the trabecular bone adjacent to the intervertebral discs. In more severe lesions, bone resorption also occurred in the dorsal vertebral arch and spinous processes. New bone formation tended to occur in areas of increased osteoclastic resorption and in the subperiosteum. Increased osteoclasts, osteoclastic bone resorption, and porosity were the predominant changes in the bones of the jaw, occurring in the ramus of the mandible, the mandibular fossa, palatine bone, and tendinous areas of muscle attachment along the skull. Morphological changes associated with remodeling and new bone formation were similar to those in the long bones and occurred multifocally in the subperiosteum of the mandibular ramus (Figure 4D) and in the dorsal aspects of the frontal bones.
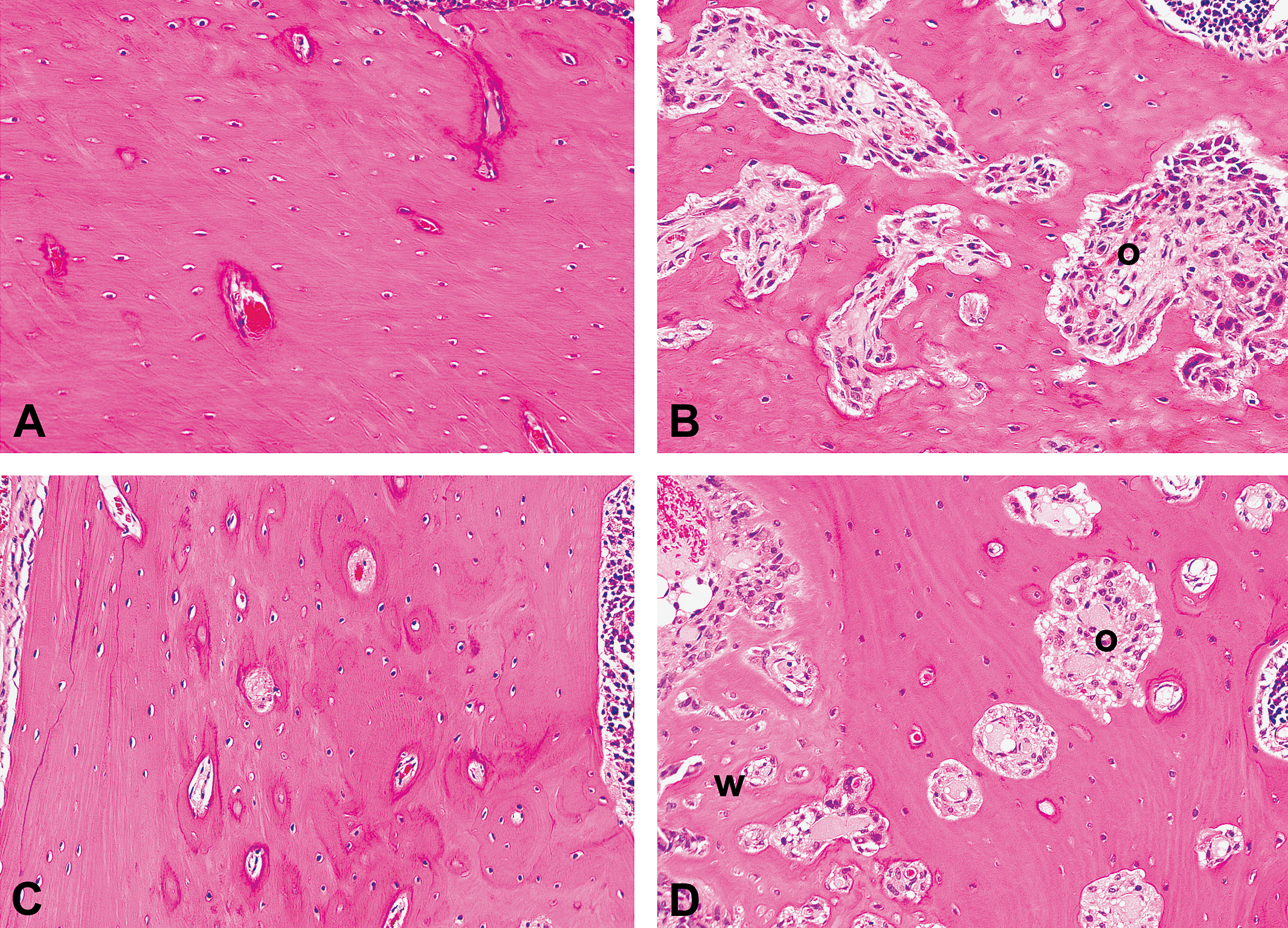
Photomicrographs of H&E-stained sections of spine and mandible of control rats and rats dosed with HD-sGCa-B after the 7-day dosing phase of a reversibility study (×200). (A) Control rat vertebral body. (B) Vertebral body after dosing with HD-sGCa-B showing increased osteoclastic bone resorption (o). (C) Control rat mandible. (D) Mandible after dosing with HD-sGCa-B showing increased osteoclastic bone resorption (o) and subperiosteal woven bone (w).
HD-sGCa-A time course study
In rats administered HD-sGCa-A at 90 mg/kg/day for up to 7 days, increased osteoclastic bone resorption, porosity, and new bone formation were observed in the tibia, femur, patella, and spine. Following 1 day of dosing, there was minimally increased tibial and femoral osteoclastic resorption of the cortical diaphyseal bone in 1 of the 5 rats (Table 2). After 2 days of dosing, minimal to mild tibial and femoral osteoclastic bone resorption was observed in all 5 rats and was observed in the spine of 4 rats. Minimal subperiosteal new bone formation was observed in the tibia of 1 rat. Thereafter, variable osteoclastic bone resorption, cortical porosity, and new bone formation were observed in all rats sacrificed after 5 and 7 days of dosing. Cortical porosity occurred after the onset of osteoclastic bone resorption. The degree of new bone formation became more prominent than osteoclasia after 7 days of dosing. Minimal to mild osteoclastic bone resorption and new bone formation also occurred adjacent to hyaline articular cartilage and tendinous fibrocartilage in the patella of 3 rats.
HD-sGCa-A time course study bone findings following various days of dosing.
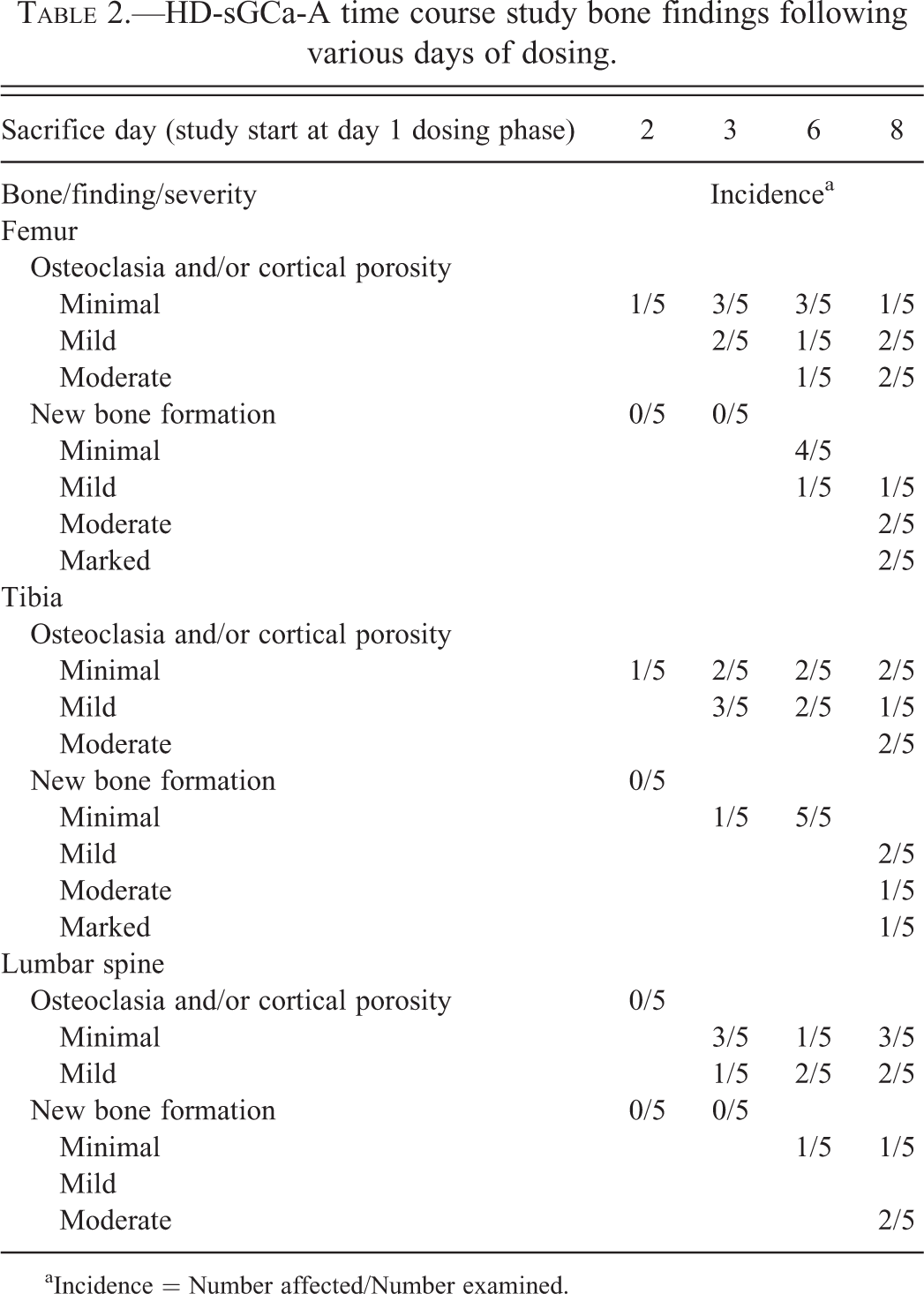
aIncidence = Number affected/Number examined.
HD-sGCa-B reversibility study
Dose-related skeletal effects were observed in tibias and femurs of all rats from both dose groups sacrificed after 7 days of dosing, similar to those observed in the exploratory toxicity studies. The compound half-life was approximately 4 hr and, after 7 days of recovery, the plasma concentration of HD-sGCa-B was below lower limits of quantification. After the 2-week recovery period, there was mildly increased porosity of the femur in 1 rat from the 75 mg/kg/day dose group. In this animal and 2 other rats in this dose group, there were either minimally to mildly increased porosity (N = 2) or new bone formation (N = 1) of bones of the jaw. No HD-sGCa-B-related bony changes were noted in the spine. No HD-sGCa-B-related bony changes were noted in rats sacrificed after the 5-week recovery period.
HD-sGCa-C aged rat study
Six of the 13 rats were sacrificed between days 9 and 21 of the study due to weight loss, dehydration, and listlessness. At sacrifice, bone changes were observed in the femur, tibia, spine, skull, and jaw in 11 of the 13 HD-sGCa-C-dosed rats, including the 6 rats sacrificed early (Figure 5; Table 3). In vehicle-dosed rats, growth plates were considerably thinner than those of 12-week-old rats; there was a reduced proliferative zone, marked reduction or loss of hypertrophic zone, acellular areas, and a thin horizontal band of bone at the metaphyseal edge of the growth plate (Figure 5A), consistent with normal growth plate morphology of rats in this age group. The effect of compound on multifocal osteoclastic bone resorption was more marked than the effect on new bone formation (Figure 5B–D). The distribution of changes in the various bones was similar to that observed in the younger rats. The severity of bone lesions appeared to correlate with AUC exposure in rats that survived to scheduled necropsy or with early sacrifices due to declining health status of the animal. Plasma concentrations of drug were only assessed at weekly intervals. If an animal did not survive until scheduled sacrifice, then a final weekly plasma concentration of drug was not assessed for that animal.
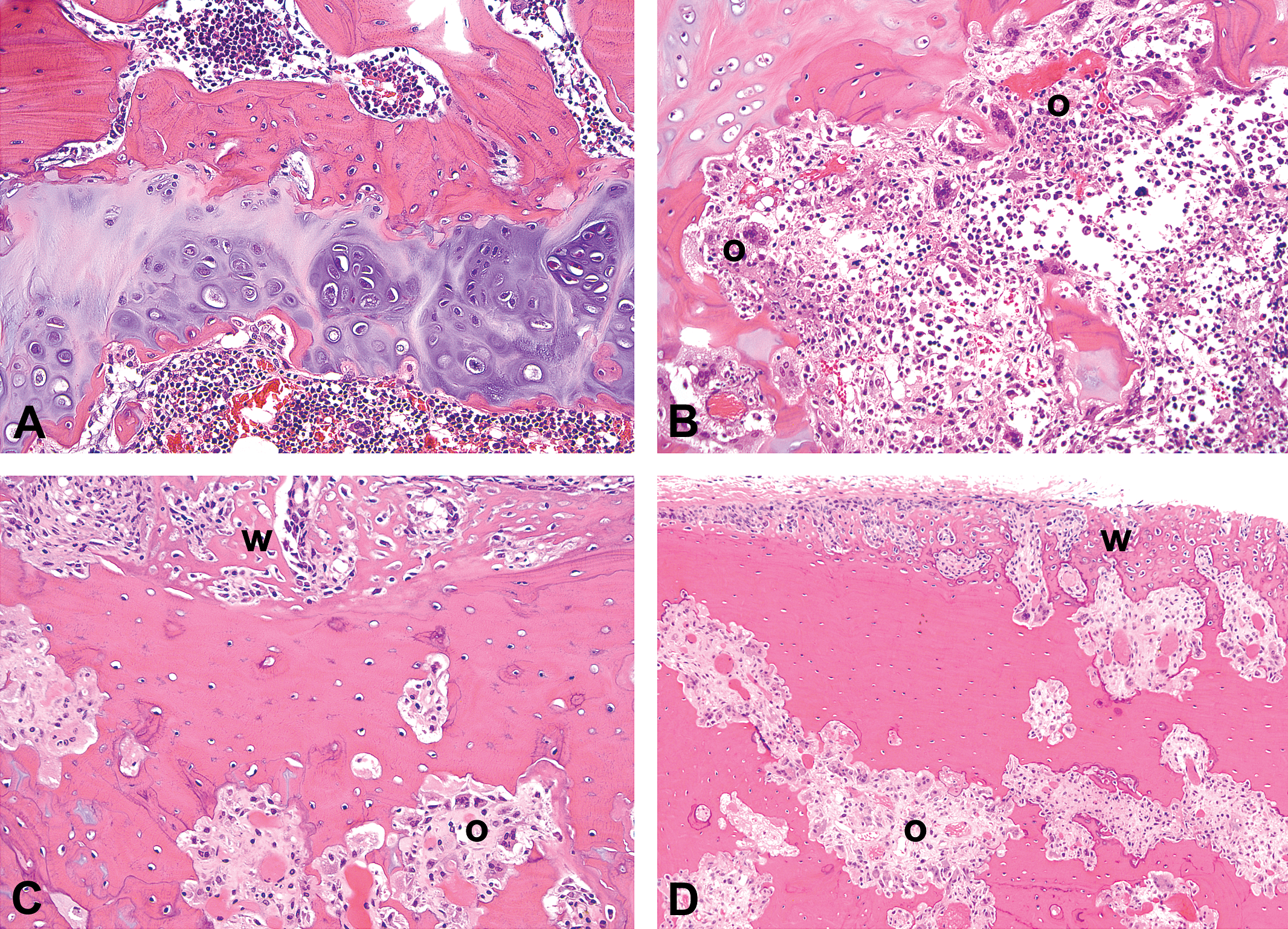
Photomicrographs of H&E-stained sections of tibia, femur, and skull of a control aged rat and aged rats dosed with HD-sGCa-C for 26 days. (A) Control rat growth plate and epiphysis showing growth plate changes consistent with a normal 44-week-old rat (×200). (B) Tibia after dosing with HD-sGCa-C showing marked epiphyseal osteoclastic bone resorption (o) (×200). (C) Cross section of femur after dosing with HD-sGCa-C showing cortical osteoclastic bone resorption (o) and endosteal woven bone (w) (×200). (D) Skull after dosing with HD-sGCa-C showing marked osteoclastic bone resorption (o) and subperiosteal woven bone (w) (×100).
HD-sGCa-C aged rat study bone findings at early sacrifice and end of study.
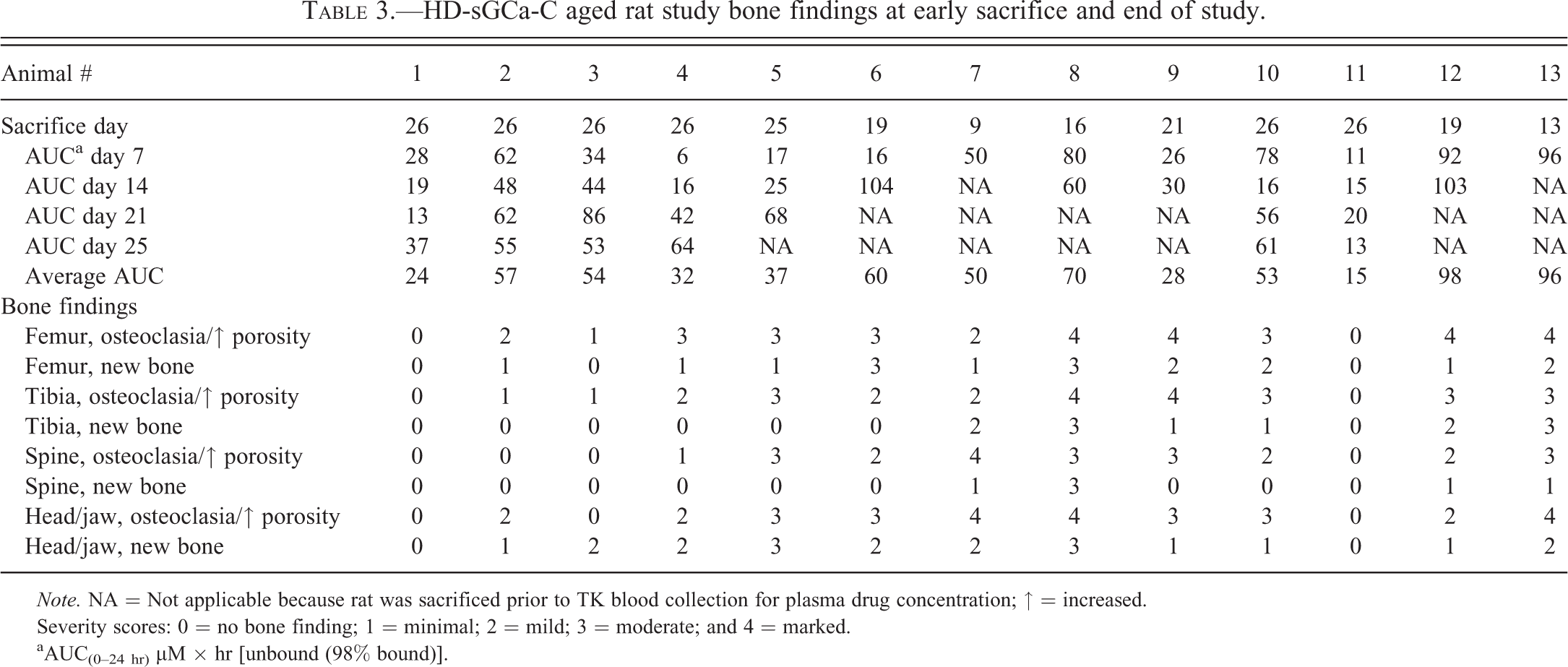
Note. NA = Not applicable because rat was sacrificed prior to TK blood collection for plasma drug concentration; ↑ = increased.
Severity scores: 0 = no bone finding; 1 = minimal; 2 = mild; 3 = moderate; and 4 = marked.
aAUC(0–24 hr) µM × hr [unbound (98% bound)].
TPTx rat model study
Tibias and femurs were examined only from groups of rats that survived to day 5 of study: control rats dosed with vehicle alone (Figure 6A), TPTx rats dosed with vehicle alone (Figure 6B), and TPTx rats dosed with HD-sGCa-C (Figure 6C). TPTx rats dosed with vehicle alone exhibited thinner growth plates, less bone in the primary and secondary spongiosa, and inactive bone surfaces, compared to vehicle-dosed control rats with intact thyroid and parathyroid glands. The primary bone changes observed in tibia and femur of HD-sGCa-C-dosed TPTx rats were increased numbers and size of osteoclasts and bone resorption adjacent to the metaphyseal edge of the growth plate, and affecting trabeculae in the primary and secondary spongiosa near the junction of the growth plate (Figure 6C). The cellularity of the hypertrophic zone of the growth plate was reduced compared to the vehicle-dosed TPTx rats, and there was a thin horizontal band of bone at the metaphyseal edge of the growth plate. Mean baseline and terminal serum CTX and OC were lower in TPTx vehicle rats than in vehicle control rats. After dosing with sGCa compounds, mean terminal serum CTX and OC concentrations were significantly higher in all sGCa-dosed groups compared to the predose baseline and to the mean terminal serum concentrations in the vehicle-dosed TPTx rats (Figure 7).
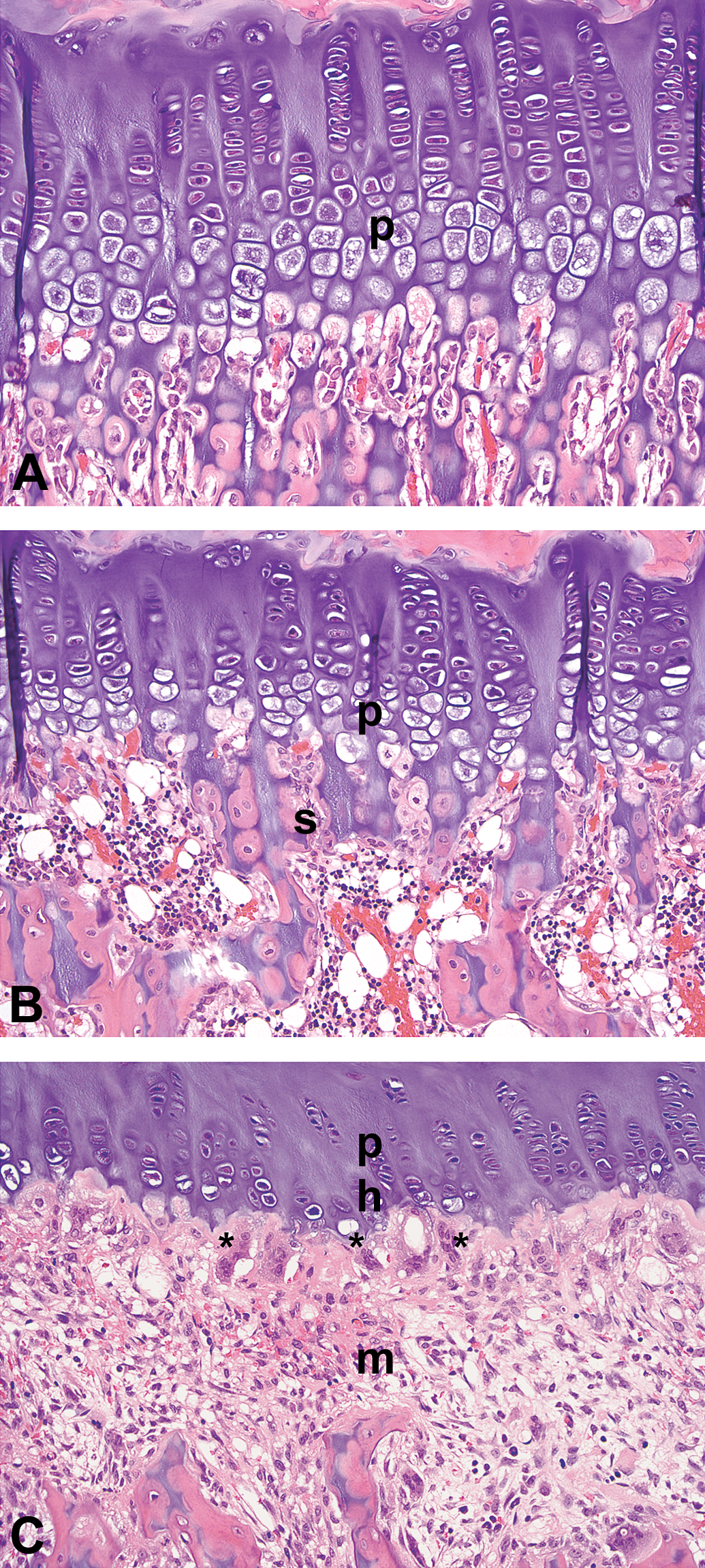
Photomicrographs of H&E-stained sections of tibial growth plate (p) of a control rat with intact thyroid and parathyroid, a TPTx rat dosed only with vehicle and a TPTx rat dosed daily with HD-sGCa-C for 5 days (×200). (A) Intact control tibial growth plate. (B) Vehicle-dosed TPTx tibia showing thinning of the growth plate and decreased bone in the spongiosa (s). (C) TPTx tibia after dosing with HD-sGCa-C showing increased number and size of osteoclasts (*) adjacent to the metaphyseal edge of the growth plate, reduced cellularity of the growth plate hypertrophic zone (h), and absence of bone trabeculae in the metaphysis (m).
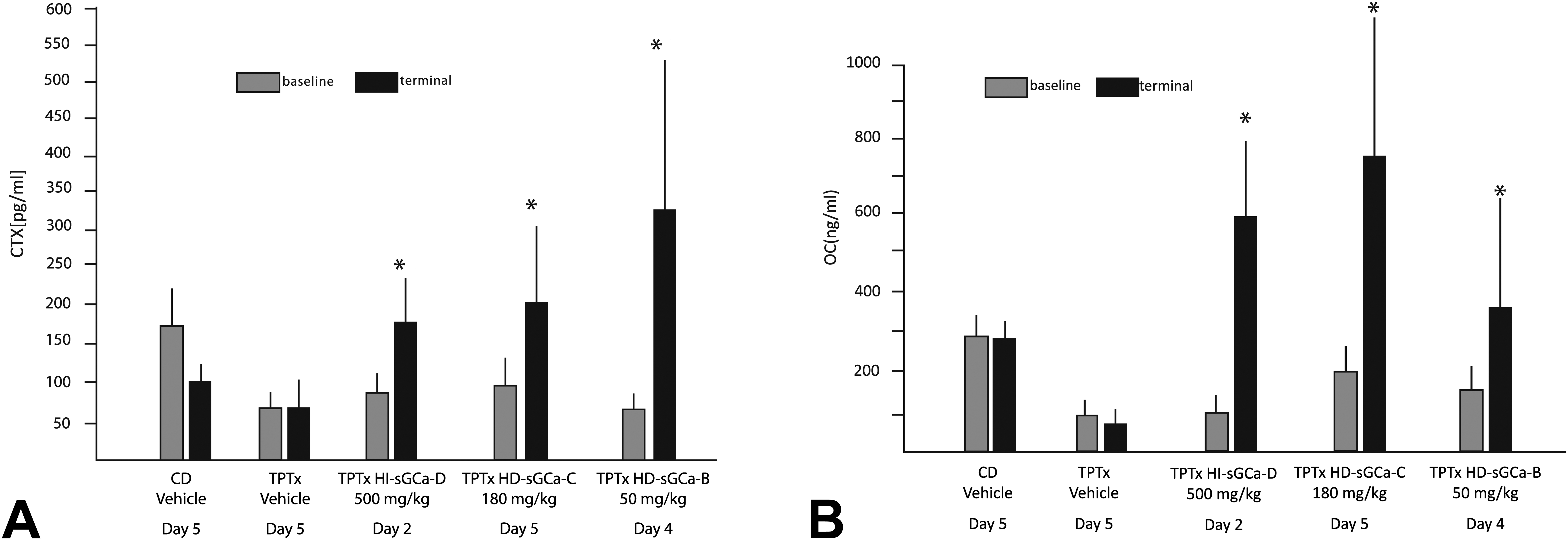
Graphs of mean serum concentrations of C-terminal telopeptide of type I collagen (CTX) (A) and osteocalcin (OC) (B) showing higher terminal concentrations in all sGCa-dosed groups compared to the mean baseline concentration and mean terminal concentration of the vehicle-dosed TPTx rats. *p < .05 for terminal biomarker concentration versus baseline and TPTx vehicle–dosed rats in the unpaired t test.
Other significant lesions
In addition to the bone changes in sGCa-dosed rats, the only other significant pathologic finding observed was varying degrees of hemorrhage, degeneration, and necrosis of mesenteric arteries, especially at bifurcations (Figure 8). The arteriopathy was characterized by intimal endothelial swelling; extravasated erythrocytes, smooth muscle degeneration and nuclear pyknosis, fibrinoid necrosis and scattered infiltrates of neutrophils and mononuclear leukocytes in the tunica media; and minimal to mild mostly mononuclear infiltrates in the tunica adventitia.
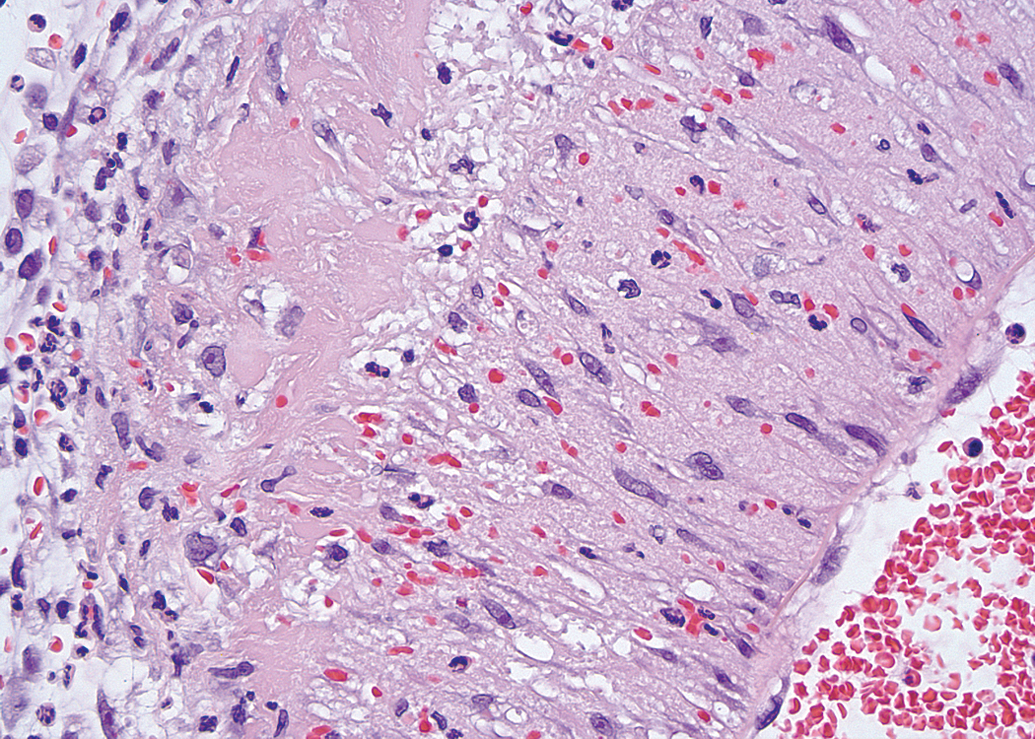
Wall of mesenteric artery of a rat dosed with HD-sGCa-C showing intimal endothelial swelling, hemorrhage, vacuolation, and fibrinoid necrosis of the tunica media, and mixed leukocytic infiltrates in the tunica adventitia (×400).
Discussion
Several in vivo studies were conducted to better understand the pathophysiology and risk assessment of sGCa-associated bone changes in SD rats. After observing increased tibial and femoral osteoclastic bone resorption, bone porosity, and new bone formation in sGCa studies associated with 3 different compounds from a HD series, several investigative in vivo studies were conducted.
In the initial three 7-day exploratory toxicity studies, the only bones examined were the femur and tibia. In the follow-on investigative studies, the lumbar spine and coronal sections of the head (including bones of the skull, frontal sinus, maxilla, and mandible) were assessed to determine whether the bone findings were regionally specific. Bone changes were observed in the appendicular and axial skeleton, head and jaw, indicating that they were not limited to bones with growth plates. Other than the TPTx model study, no sGCa-related growth plate changes were observed.
A primary objective of these studies was to determine whether the bone changes were directly related to the intended pharmacologic stimulation or activation of sGC. Several strategies have been proposed to address whether a toxicologic effect is caused by the intended mechanism of action (Rudmann 2013). Factors considered in exploring the mechanism responsible for the bone changes caused by sGCa compounds included relevant published literature information; increasing the potency of the molecule to provide the desired pharmacologic effect of reducing blood pressure while reducing the undesirable bone effect; synthesizing and testing structurally similar but pharmacologically inactive compounds or pharmacologically active, structurally unrelated compounds; using an in vivo model to manipulate factors that regulate bone metabolism; and in vitro testing in osteoblast and osteoclast cultures or organotypic assays. Increasing the potency and reducing the dose of lead molecules had no impact on reducing the development of bone changes. However, off-target and chemical-based effects could not be ruled out, as compounds from the same imidazopyridine chemical series (Roberts et al. 2011) were administered in the initial toxicity studies. Therefore, an active, structurally unrelated HI compound, HI-sGCa-D, was synthesized (Figure 2). The hypothesis was that if blood pressure lowering (expected pharmacology), mesenteric arteriopathy and bone changes occurred pursuant to administration of an HI-sGCa, the bone effects would likely be mediated through sGC. The 7-day toxicity study with an HI-sGCa resulted in arterial and bone changes similar to those observed with administration of HD sGCa compounds, supporting the hypothesis that sGC stimulation or activation was responsible for the bone changes. This suggests that all sGCa compounds will induce some degree of the bone changes.
The initial objective for conducting the time-course study was to develop an in vivo strategy for screening compounds against the bone effects, provided that these effects were not related to sGC activity. Increased osteoclastic bone resorption was observed in all 5 rats sacrificed on day 3, after only 2 days of once daily compound administration. Increased cortical bone porosity was not observed until day 6, indicating that osteoclasia likely preceded an increase in porosity. Although there was evidence of bone remodeling resulting in increased incidence and degree of subperiosteal and endosteal bone formation with longer duration of compound administration, the changes were multifocal and were not always observed in the same area of bone. The findings following administration of HI-sGCa-D combined with information from the published scientific literature support the hypothesis that the observed bone changes are a direct effect of sGCa activity. Therefore, further work to develop assays to screen away from the bone effects was not justified. The primary focus turned to determining reversibility of the bone changes and whether bone changes would be relevant in an older (adult) patient population.
Bone changes had mostly recovered within 2 weeks of the end of dosing phase. No HD-sCGa-B-related bone changes were observed after 5 weeks of recovery. Reversibility was anticipated since the processes involved in bone remodeling at the end of dosing were intact, as characterized by the presence of increased osteoclasts, scalloped surfaces of bone indicative of bone resorption, and formation of new woven bone lined by plump osteoblasts (Clarke 2008).
Longitudinal bone growth rate diminishes in rats after skeletal maturity at approximately 13 weeks of age, and virtually ceases at approximately 26 weeks. Growth plates in 42- to 46-week-old rats (age at study termination) are considerably thinner than those of 9- to 12-week-old rats and characterized by a reduced proliferative zone, loss of hypertrophic zone, a thin horizontal band of bone at the metaphyseal edge of the growth plate, and acellular areas (Roach et al. 2003). The similarity of the bone changes in the aged HD-sGCa-C-dosed rats to those of younger sGCa-dosed rats suggested that the bone changes would be relevant to an older population targeted for antihypertensive therapy. There was considerable variation in plasma drug concentrations between individual animals, with up to a 16-fold difference between lowest and highest concentrations measured at day 7, and less variation in rats surviving to study day 26. It is uncertain how this variability might have impacted the bone morphological changes, but there appeared to be greater severity of bone changes in animals with higher plasma drug concentrations, particularly during the first half of the study.
Multiple factors including PTH and CT control bone remodeling, regulation of bone mass, and differentiation and function of osteoclasts and osteoblasts (Andresen et al. 2006; Harada and Rodan 2003; Boyle, Simonet, and Lacey 2003). The goal of the TPTx study was to assess the effect of sGCa compounds on bone remodeling in the absence of parathyroid and thyroid hormonal effects to help understand the pathogenesis of the bone changes. In the TPTx rat model, the deprivation of thyroid and parathyroid hormones and calcitonin creates a low state of bone modeling and remodeling. This model is ideal for in vivo screening of bone effects of novel therapies for increased bone formation and/or bone resorption because of the sensitivity, fast readout (as early as 24–48 hr), and small amount of compound needed to effect bone changes in this model (Trechsel, Stutzer, and Fleish 1987; Millest et al. 1995; Andresen et al. 2006). The model also allows for correlation of pharmacokinetic, pharmacodynamic, and safety endpoints.
The primary bone change observed in the HD-sGCa-C-dosed animals in the TPTx study was that of osteoclasia. However, the bone biomarker responses observed in all sGCa-dosed animals were indicative of bone resorption (CTX) and bone formation (OC; Singer and Eyre 2008). CTX and OC will increase in states of high bone turnover associated with activity of osteoclasts and osteoblasts. Therefore, data from the TPTx model study supported evidence that sGCa compounds have the potential to directly affect bone remodeling.
Agonists of cGC have been explored for the treatment of high blood pressure. Following the initial identification of the compound YC-1 as a pharmacological stimulator of sGC (Ko et al. 1994), others reported more potent and selective agonists of sGC of the same mechanistic class as YC-1 (Schindler et al. 2006; Stasch, Alonso-Alija, et al. 2002; Stasch et al. 2001) and distinct mechanistic classes (Stasch, Schmidt, et al. 2002). YC-1 and other compounds of the same class, such as BAY 41-2272 and BAY 41-8543, stimulate sGC in the absence of NO and have been referred to as NO-independent for this reason (Stasch et al. 2001). These compounds, referred to as HD sGC stimulators, require the presence of a reduced heme prosthetic group in the enzyme (Evgenov et al. 2006; Priviero and Webb 2010). All of these compounds cause vasorelaxation and lower blood pressure. Riociguat (Adempas®, Bayer Healthcare Pharmaceuticals, Whippany, NJ), a member of this class, was recently approved by the U.S. Food and Drug Administration (FDA) for treatment of pulmonary hypertension (Ghofrani et al. 2013; Schermuly et al. 2011). In contrast, sGC agonists such as BAY 58-2667 (Cinaciguat, Bayer Healthcare Pharmaceuticals) and HMR1766 do not require the presence of prosthetic heme and are therefore described as HI sGC agonists (Evgenov et al. 2006; Erdmann et al. 2013; Priviero and Webb 2010). Another distinguishing characteristic of these 2 mechanistic classes is that the activity of HD-sGCa compounds is completely abolished by oxidation of the sGC heme moiety while that of HI-sGCa compounds is potentiated under these conditions.
The NO-cGMP signaling pathway has an important role in bone metabolism and remodeling (Van’t Hof and Ralston 2001; Blair, Zaidi, and Schlesinger 2002; Broderick et al. 2007). In vitro studies have demonstrated effects of this pathway on both osteoblasts (Hikiji et al. 1997) and osteoclasts (Yaroslavskiy et al. 2005). Both cell types express 1 or more forms of nitric oxide synthase (NOS), including endothelial, neuronal, and inducible isoforms (Van’t Hof and Ralston 2001), and both produce NO (Blair, Zaidi, and Schlesinger 2002). The in vitro NO effect on osteoclasts and osteoblasts may be variable and has been described as biphasic (Van’t Hof and Ralston 2001). Low concentrations of NO stimulated cGMP-dependent primary osteoblast proliferation and high concentrations inhibited proliferation and induced apoptosis (Mancini et al. 2000). Low NO concentrations inhibited osteoclast formation and activity during the osteoclast differentiation stage in murine marrow cultures, but 100-fold higher concentrations were required to inhibit activity of mature osteoclasts (Holliday et al. 1997). Conversely, low NO concentrations may potentiate interleukin 1 (IL-1) and prostaglandin induced bone resorption (Ralston and Grabowski 1996). Various concentrations of NO regulated motility, attachment and detachment of osteoclasts to bone matrix via cGMP-dependent protein kinase I and vasodilator-stimulated phosphoprotein (Yaroslavskiy et al. 2005). Most of these cellular effects have been studied in culture systems or organotypic assays, where specific humoral factors and cytokines were added to the system, and this raises the question of whether there is a pharmacologic effect of sGC activation on bone in vivo.
NO is the primary activator of sGC, resulting in increased cGMP (Derbyshire and Marletta 2012). Therefore, it is certainly possible that NO-like effects on bone could be mediated through in vivo sGC stimulation or activation, regardless of whether the agonist is NO or an administered pharmaceutical sGCa. Bone mass, maintained by homeostatic balance between osteoclastic bone resorption and osteoblastic bone formation, can be modulated by pharmacological mediators of the NO-cGMP pathway (Pan et al. 2005; Van’t Hof and Ralston 2001). In an ovariectomized rat model, once daily administration of 0.2 mg nitroglycerin, an NO donor, prevented ovariectomy-induced spinal bone mineral density (BMD) loss, but bone loss was not prevented when the same dose of nitroglycerin was administered twice or three times daily (Wimalawansa et al. 2000). Administration of aminoguanidine, a selective inhibitor of NO synthase, decreased spinal BMD in sham-operated rats and potentiated spinal BMD loss in ovariectomized rats (Kasten et al. 1994). The FDA pharmacology toxicology review of riociguat in various rat toxicity studies reported that riociguat was associated with increased bone resorption and a concurrent increase in new bone formation (U.S. Food and Drug Administration 2013). The reviewing pathologist concluded that there was acceleration of bone turnover, but with a net increase in bone. This provides further support that sGCa compounds directly affect bone. Experiments, using non-rodent species, may have been useful for risk assessment of the bone changes observed in rats, provided that the animals could tolerate the blood pressure lowering effect at pharmacologic doses. Histomorphological effects on bone were not reported for the dog toxicity studies in the FDA pharmacology toxicology review of riociguat (U.S. Food and Drug Administration 2013). The reviewer suggested that this was due to the slower rate of bone turnover in dogs than rats.
Mesenteric arteriopathy occurred in rats dosed with a concentration of sGCa that lowered blood pressure (Hughes et al. 2010). While blood pressure and heart rate were not assessed in the studies described in this report, a cardiovascular functional study was conducted in normal telemetered SD rats using HD-sGCa-A. HD-sGCa-A produced 35 and 50 mm Hg decreases in mean arterial blood pressure, respectively, associated with a maximum 75 beat/min increase in heart rate after single oral doses of 30 or 120 mg/kg (data not shown). sGCa stimulators relax the mesenteric artery through both cGMP-dependent and independent mechanisms (Teixeira, Priviero, and Webb 2006), resulting in vasodilatation. Vasoactive compounds that induce vasodilatation in rats, whether via NO or another pathway, are known to result in mesenteric arterial vasculopathy (Brott, Richardson, and Louden 2012; Slim et al. 2003). The vasculopathy is associated with exaggerated pharmacology, and the mechanism is thought to involve marked vasodilatation of susceptible regional vasculature, resulting in increased blood flow and biomechanical stress to endothelial cells and the blood vessel wall (Kerns et al. 2005). In rats, the mesenteric arterial bed is a predilection site for vasoactive molecule-associated vasculopathy.
In summary, in vivo studies demonstrated that administration of sGCa compounds induced bone resorption, remodeling, and new bone formation in the rat independent of age and thyroid and parathyroid hormones. The bone changes occurred rapidly, but were reversible. These effects were directly related to the intended pharmacologic activity of these sGCa compounds.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
