Abstract
Administration of lersivirine, a nonnucleotide reverse transcriptase inhibitor, daily by oral gavage to Sprague-Dawley rats for up to 2 yr was associated with decreased survival, decreased body weights, and an increase in neoplasms and related proliferative lesions in the liver, thyroid, kidney, and urinary bladder. Thyroid follicular adenoma and carcinoma, the associated thyroid follicular hypertrophy/hyperplasia, hepatocellular adenoma/adenocarcinoma, altered cell foci, and hepatocellular hypertrophy were consistent with lersivirine-related induction of hepatic microsomal enzymes. Renal tubular adenoma and renal tubular hyperplasia were attributed to the lersivirine-related exacerbation of chronic progressive nephropathy (CPN), while urinary bladder hyperplasia and transitional cell carcinoma in the renal pelvis and urinary bladder were attributed to urinary calculi. Renal tubular neoplasms associated with increased incidence and severity of CPN, neoplasms of transitional epithelium attributed to crystalluria, and thyroid follicular and hepatocellular neoplasms related to hepatic enzyme induction have low relevance for human risk assessment.
Introduction
As a key component of antiretroviral therapy, nonnucleoside reverse transcriptase inhibitors (NNRTIs) have been used to treat HIV-infected patients for several decades. The mechanism of action of NNRTI is to inhibit the reverse transcriptase enzyme that is critical for converting the viral RNA into complementary DNA (cDNA), which in turn is essential for hijacking the host normal cellular processes to facilitate viral replication, assembly, and transmission. Current therapeutic regimens for treatment-naive patients include combination therapy with an NNRTI and additional antiretroviral agents. NNRTIs have been inherently more efficacious and safer than protease inhibitors; however, the existing first-generation NNRTI drugs present issues due to their adverse event profiles and low genetic barrier to viral mutations that bestow resistance (Jayaweera and Dilanchian 2012). Accordingly, several next-generation NNRTIs have been developed to overcome disadvantages such as viral resistance mutations associated with first-generation NNRTIs. NNRTIs currently approved in the United States include efavirenz (Sustiva®; Bristol-Myers Squibb Company, Princeton, NJ), nevirapine (Viramune®; Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT), delavirdine (Rescriptor®; Pfizer, New York, NY), etravirine (Intelence®; Janssen Therapeutics, Titusville, NJ), and rilpivirine (Edurant®; Janssen Therapeutics, Titusville, NJ).
Lersivirine was developed as a next-generation NNRTI for treating HIV-infected patients. Lersivirine was designed to bind to the reverse transcriptase enzyme in a novel way and was shown to be a highly potent and selective NNRTI, with excellent efficacy against NNRTI-resistant viruses (Corbau et al. 2010). In this report, we describe neoplastic and nonneoplastic findings that were observed in a 2-yr carcinogenicity study in rats administered lersivirine. Lersivirine, like other NNRTIs, is an autoinducer of hepatic microsomal enzymes in preclinical animal species but not in humans (Walker et al. 2009). Short-term and chronic studies have shown evidence of liver and thyroid effects consistent with enzyme induction (Houle et al. 2013).
Materials and Methods
Animals and Husbandry
Male and female Sprague-Dawley (Crl:CD[SD]) rats 6 to 7 weeks old were obtained from Charles River Laboratories (Portage, Michigan). Seventy animals/sex were randomly assigned to the control and high-dose groups to enable a longer dosing period if high mortality was observed in either group (Table 1). Sixty animals per sex were assigned to the low- and intermediate-dose groups. Additional animals (6/sex/group) were assigned to separate groups for toxicokinetic analysis. Animals were identified with implantable microchips and cage cards. Male and female rats were housed individually in polycarbonate cages with paper bedding and offered Certified Rodent Diet #2016C (Harlan Laboratories, Inc., Madison, WI) ad libitum (except when fasted prior to the scheduled necropsy) and water ad libitum. Environmental controls were set to maintain the following animal room conditions: temperature range of 18°C to 26°C, relative humidity range of 30 to 70%, 10 or greater air changes/hr, and a 12-hr light/12-hr dark cycle. All procedures were approved by the institutional animal care and use committee and performed in compliance with the US Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and federal regulations.
Design for 2-yr carcinogenicity study of lersivirine in Sprague-Dawley rats.
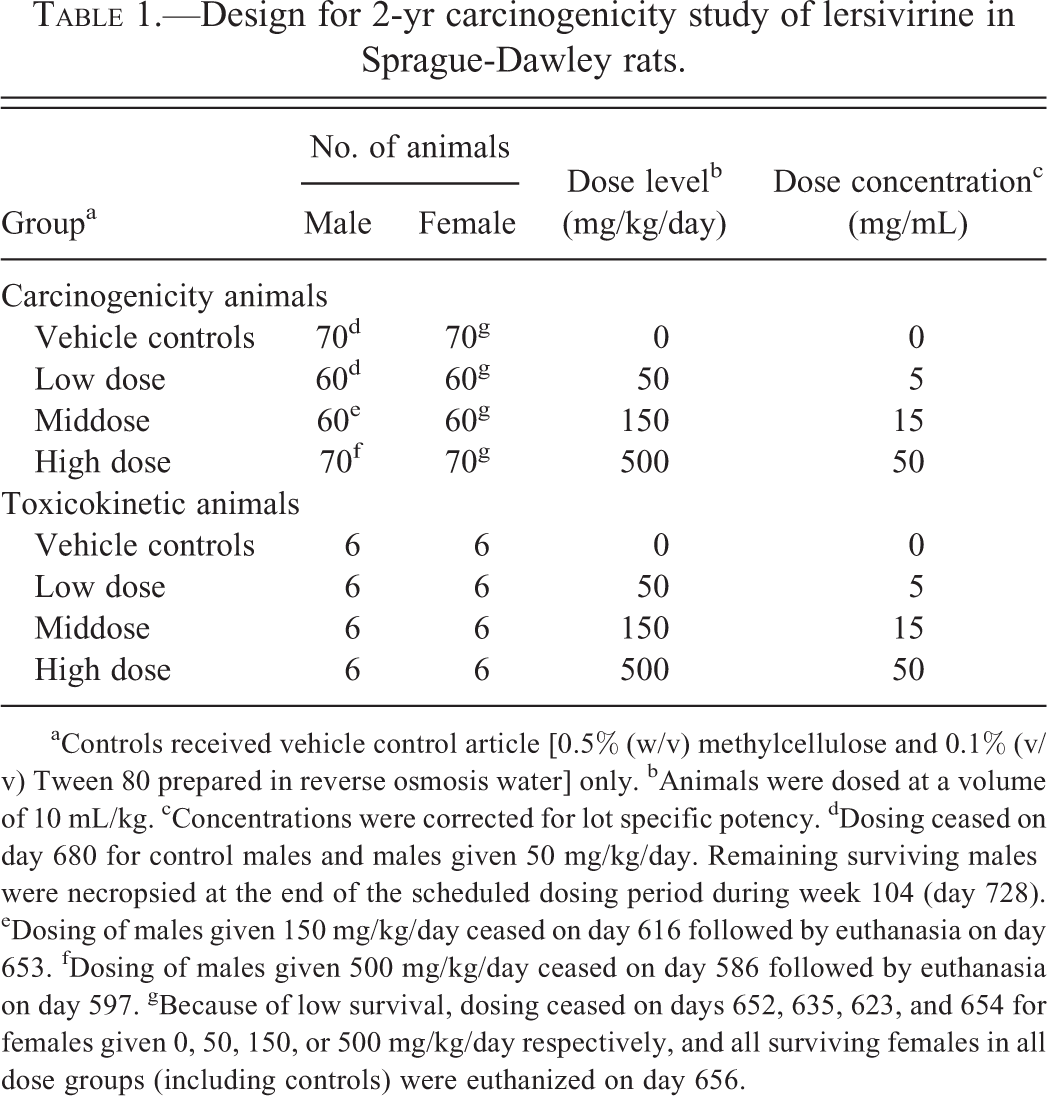
aControls received vehicle control article [0.5% (w/v) methylcellulose and 0.1% (v/v) Tween 80 prepared in reverse osmosis water] only. bAnimals were dosed at a volume of 10 mL/kg. cConcentrations were corrected for lot specific potency. dDosing ceased on day 680 for control males and males given 50 mg/kg/day. Remaining surviving males were necropsied at the end of the scheduled dosing period during week 104 (day 728). eDosing of males given 150 mg/kg/day ceased on day 616 followed by euthanasia on day 653. fDosing of males given 500 mg/kg/day ceased on day 586 followed by euthanasia on day 597. gBecause of low survival, dosing ceased on days 652, 635, 623, and 654 for females given 0, 50, 150, or 500 mg/kg/day respectively, and all surviving females in all dose groups (including controls) were euthanized on day 656.
Dose Selection and Administration
Dosages of 100, 300, and 1,000 mg/kg/day administered by oral gavage were suggested to the US Food and Drug Administration (FDA) Carcinogenicity Assessment Committee (CAC). This was based on observed mortality in animals given 1,500 mg/kg/day and the exposure plateau achieved in males given 500 mg/kg/day by gavage in a 13-week range finding study. The dose of 1,000 mg/kg/day was near the maximum tolerated dose (MTD) in the 13-week study; this was based on the unexplained death of 1 rat at 1,000 mg/kg/day and findings such as decreased body weight (males only), hepatocellular degeneration/necrosis and inflammation, increased kidney weights, regeneration of renal tubules (males), renal tubular basophilia (females), and granular material in the bladder with hypertrophy/hyperplasia of transitional epithelium at
Pre-term Euthanasia Criteria
High mortality was observed in males at 150 and 500 mg/kg/day and in female control rats. Prespecified criteria were developed in consultation with the CAC to halt dosing when survival for each group and sex (except controls) reached
Clinical Examination
Assessments during the treatment period included clinical observations, body weight, body weight gain, food consumption, ophthalmic examinations, and urinalysis. During the dosing period, animals were observed twice daily and detailed observations were recorded once weekly. Beginning at week 26, animals were examined for visible or palpable masses once weekly through the duration of the study. Weekly through week 26 and monthly thereafter, body weight and food consumption were assessed. Before the administration of the first dose and during week 53, eyes were dilated with a mydriatic agent and ophthalmic examinations were performed.
Toxicokinetics
Blood was collected from toxicokinetic animals before dose administration and at 1, 3, 6, and 24 hr after dose administration during week 26. Plasma samples were analyzed for lersivirine concentrations and maximal plasma concentrations and areas under the drug concentration curve were estimated for each dose level and sex. The toxicokinetic animals were euthanized without necropsy after the last TK samples were collected.
Postmortem Examination
At necropsy, a comprehensive set of tissues from each animal was preserved in 10% neutral-buffered formalin, with the exception of the eyes, Harderian glands, optic nerves, and testes. These were collected in modified Davidson’s fixative and stored in 10% neutral-buffered formalin. Tissues identified in the protocol from all animals were dehydrated in graded ethanol, embedded in paraffin, sectioned, stained with hematoxylin and eosin, and examined by light microscopy. All kidneys and urinary bladders were also examined using polarized light to identify crystals if present.
A high incidence of enlarged and pale kidneys was observed at necropsy in animals that died or were euthanized up to week 27 of the study. After week 27, urine was collected by cystocentesis at necropsy from all animals.
Statistical Analysis
All analyses were performed separately for each sex. Body weight, body weight change, food consumption, and numerical urinalysis data were analyzed by one-way analysis of variance (ANOVA), and if the ANOVA was significant (p
Survival data were analyzed using the National Cancer Institute (NCI) life table package (Thomas, Breslow, and Gart 1977). The methods consisted of the Cox Tarone binary regression and the Gehan Breslow nonparametric test on life tables. One-sided probabilities for trend and group comparisons were calculated and evaluated at a significance level of p
The occult (i.e., internal organ or unobservable) tumors (incidental alone or incidental and fatal combined) were analyzed by the International Agency for Research on Cancer asymptotic fixed interval-based prevalence test (Peto et al. 1980). The cutoff points for the interval-based test were weeks 0-52, 53-78, 79-92, and 93 through before terminal euthanasia, and terminal euthanasia. Nominal dose levels (i.e., the starting arithmetic dose levels) were used in the analyses. The test was implemented using PROC MULTTEST in the SAS system (SAS 2002–2008). In the case of sparse tables (<12 total in all groups combined), the exact form of the test was used. Palpable (superficial as in mammary or skin) tumors were analyzed by the Cox-Tarone binary regression method using the death time or the first palpation time as a surrogate for the tumor onset time. One-sided positive trends and control versus high-dose group comparisons in common (background incidence rate >1%) and rare (background incidence rate <1%) tumors (FDA Draft Guidance for Industry 2001) as observed in the concurrent control, defined by the principal investigator for anatomic pathology, or based on historical control data from the conducting laboratory and/or Charles River Laboratories (Giknis and Clifford 2004) were evaluated at the significance levels of .01 and .05, respectively (Haseman 1983). Other pairwise, one-sided group comparisons were evaluated at a significance level of p
Results
Mortality
Lersivirine produced a statistically significant increase in mortality in males given
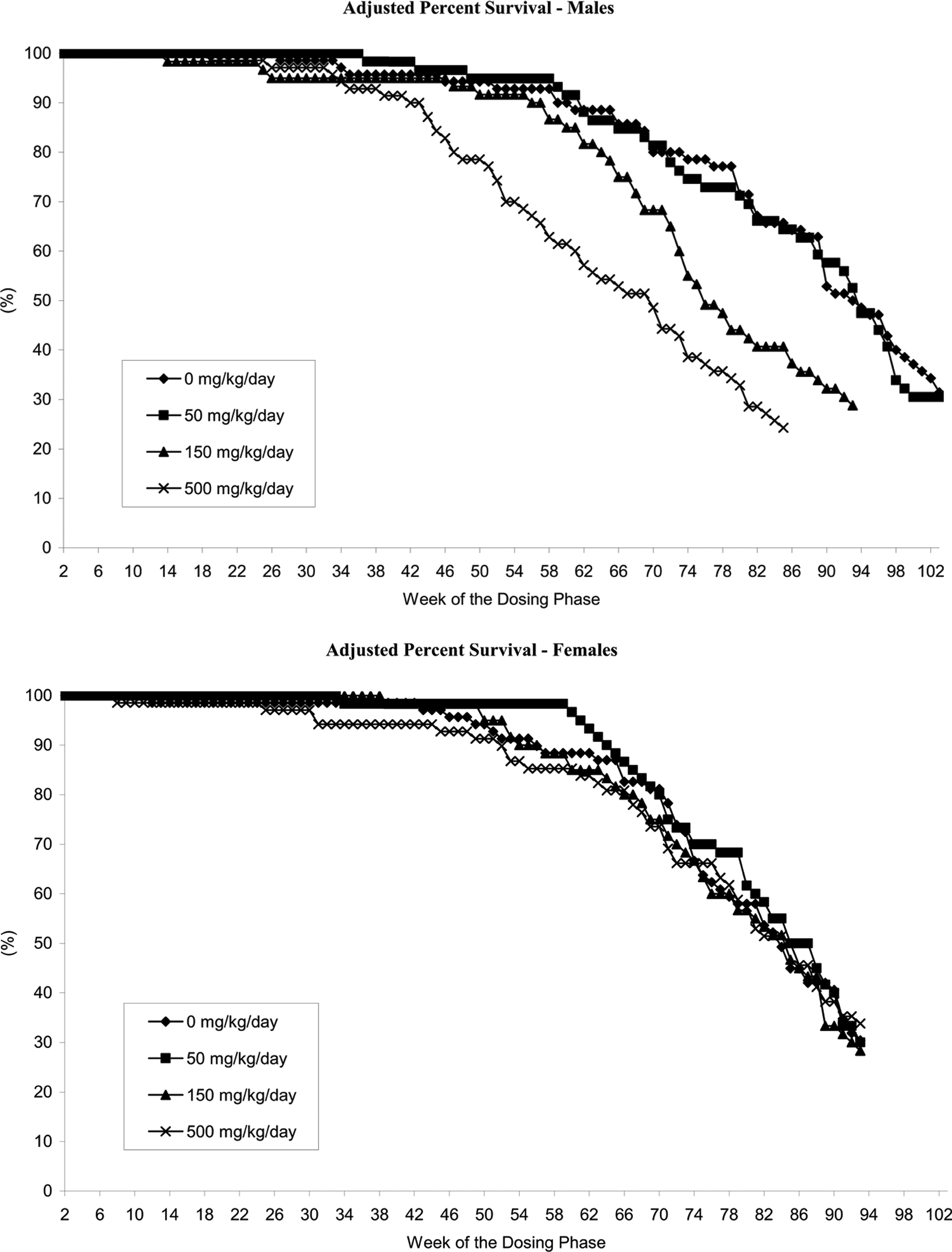
Adjusted percentage Kaplan-Meier survival of males and females in the 2-yr carcinogenicity study with lersivirine in Sprague-Dawley (SD) rats.
Body Weight and Food Consumption
Mean body weights in males and females given 500 mg/kg/day were lower than control values (28–34%) beginning at approximately weeks 34 and 30, respectively, and remained so until all animals in this group were necropsied (data not shown). At necropsy, mean body weights in surviving males given 150 mg/kg/day and 500 mg/kg/day were 9% and 28%, respectively, lower than controls. Similarly, mean body weights in females given 500 mg/kg/day were 34% lower than controls (data not shown). These reductions in body weight were noted despite increases in food consumption in both males and females (data not shown).
Urinalysis
Urinalysis conducted on males euthanized after day 181 and females euthanized after day 185 demonstrated higher incidences and/or severities of urine protein for males given
Toxicokinetics
Exposure data for drug in plasma are summarized in Table 2. Exposures measured as free lersivirine in this study were relatively low, but higher exposures were not possible as a result of the enzyme induction by lersivirine (Walker et al. 2009). The estimated margin above human exposure at a dose of 150 mg (AUC free 3,700 ng-hr/mL) based on area under the free drug concentration curve (AUC) was 0.3× for males and 1.5× for females given 500 mg/kg/day. The free fractions of lersivirine were 0.266 (rat) and 0.424 (human).
Mean toxicokinetic parameters in rats after oral administration of lersivirine for 26 weeks.

Note: Overall = combines male and female. Fraction of unbound (free) drug in rat plasma = 0.266.
Macroscopic and Microscopic Findings
Liver
Lersivirine-related neoplastic findings in the liver included increases in the incidences of hepatocellular adenoma and/or carcinoma in females given
Incidence of test article-related liver findings.
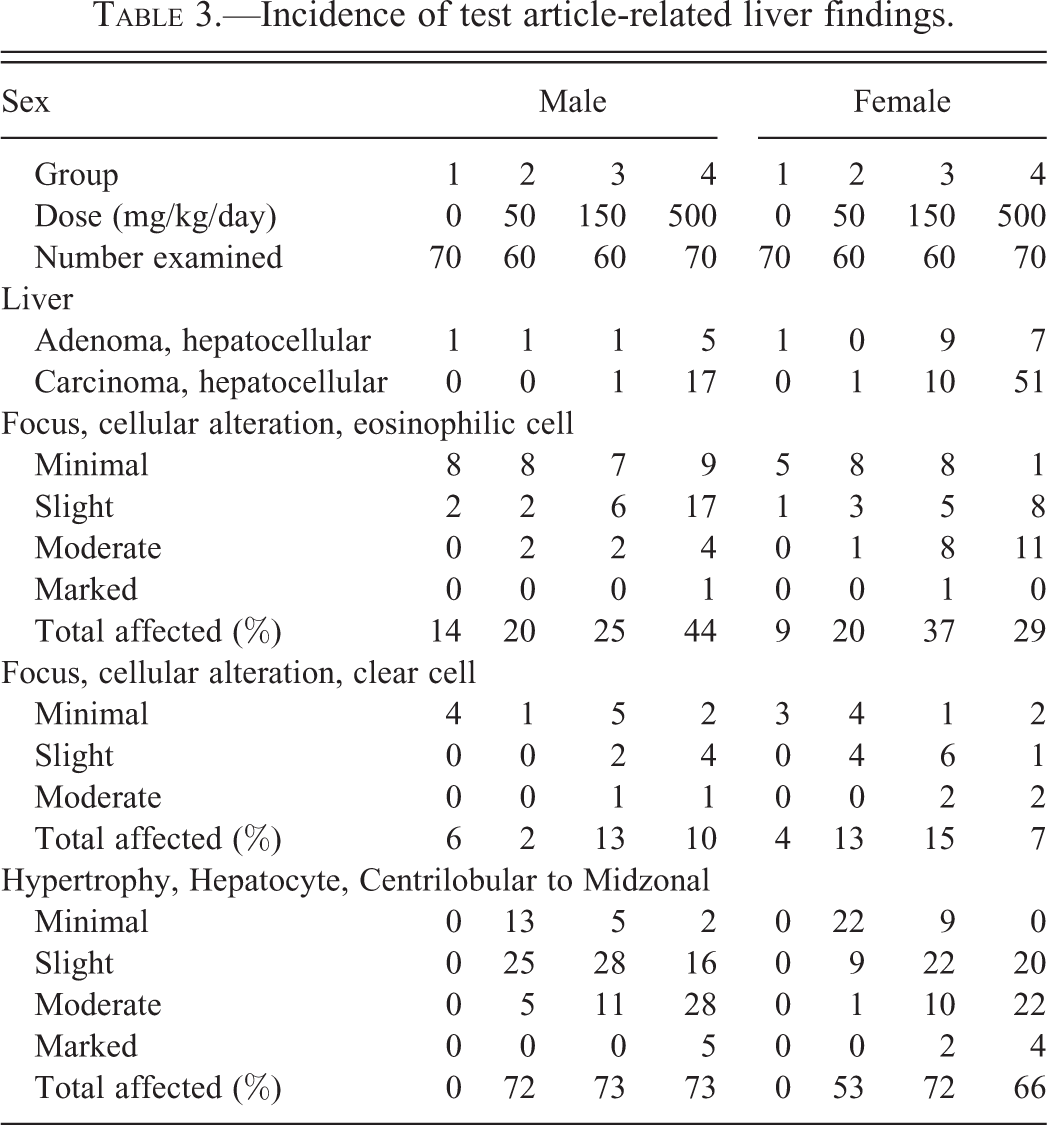
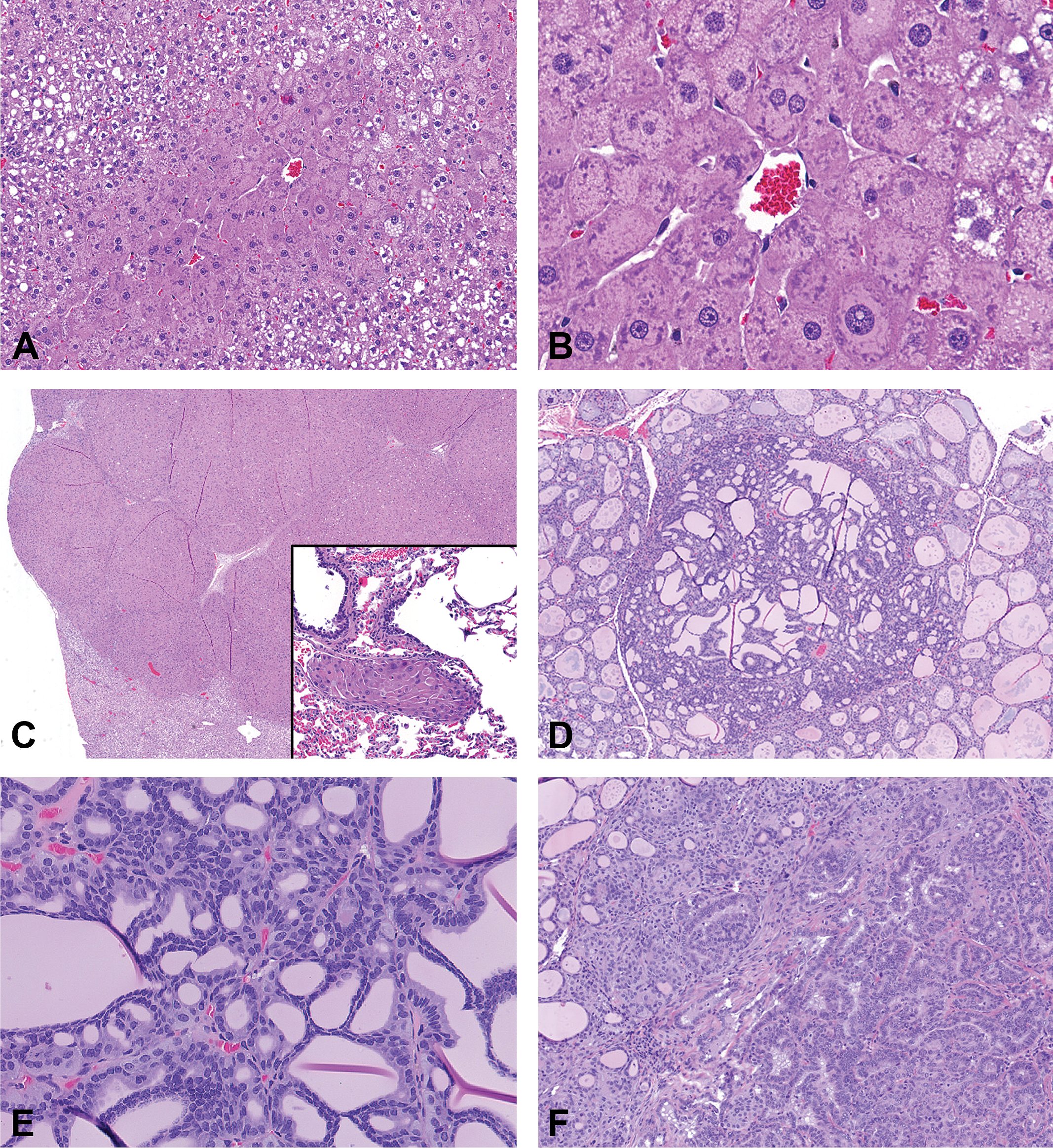
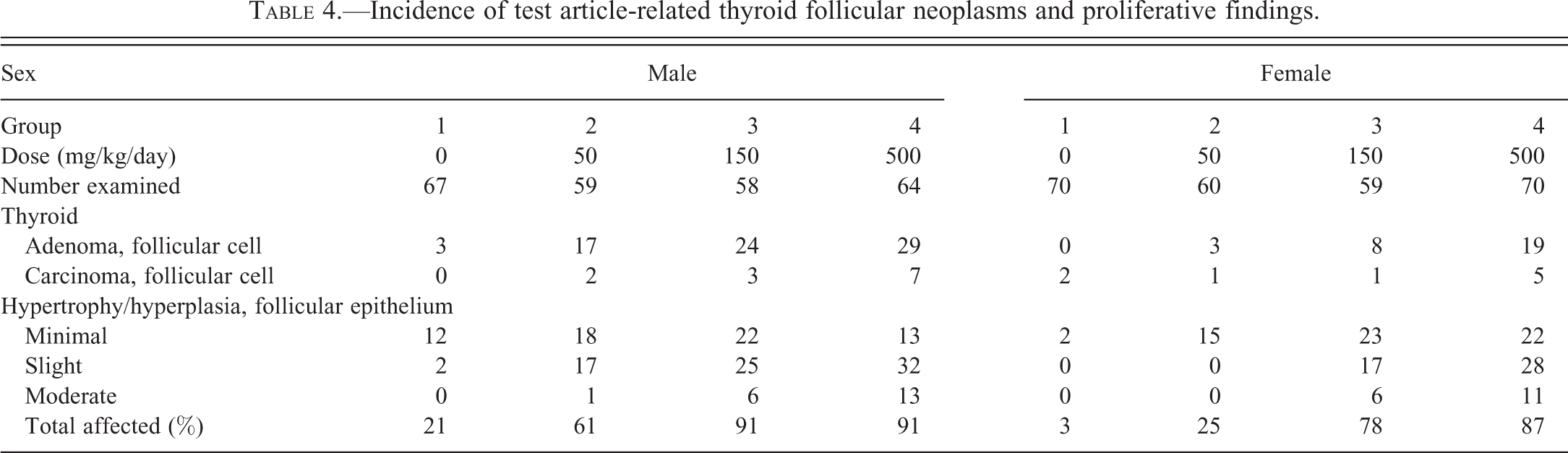
Histopathology of lersivirine-related thyroid and liver tumors in the 2-yr carcinogenicity study in Sprague-Dawley (SD) rats. (A) Low magnification view of centrilobular hypertrophy characterized by large hepatocytes around a central vein with darker eosinophilic cytoplasm. (B) High magnification view of the centrilobular hypertrophy with expanded hepatocellular cytoplasm. (C) Hepatocellular carcinoma that effaces the underlying parenchyma; inset shows a tumor embolus within the lung of the same animal. (D) Low magnification view of a thyroid follicular adenoma in a high dose male. (E) Higher magnification of the panel D. (F) Thyroid follicular adenocarcinoma in a high dose male.
Thyroid
Lersivirine-related increases in the incidences of discolored and/or large thyroid gland occurred in males given
Lersivirine administration increased the incidences of thyroid follicular cell adenoma/carcinoma in a dose-dependent manner in both sexes (Figure 2F). Statistically significantly increased incidences of follicular cell adenoma occurred in males given
Incidence of test article-related thyroid follicular neoplasms and proliferative findings.
Kidney and Urinary Bladder
Transitional cell neoplasms and hyperplasia: Lersivirine-related papilloma and/or transitional cell carcinoma occurred in the urinary bladder of males given 500 mg/kg/day (Table 5). These neoplasms were often associated with macroscopic changes in the urinary bladder including presence of calculi, masses, and distended bladder (Figure 3). The increased incidence of urinary bladder distension was likely associated with urinary obstruction due to calculi and/or neoplasia. Transitional cell carcinoma was also noted in the renal pelvis of 1 male given 500 mg/kg/day.
Incidence of test article-related transitional cell neoplasms and related urinary bladder and kidney findings.
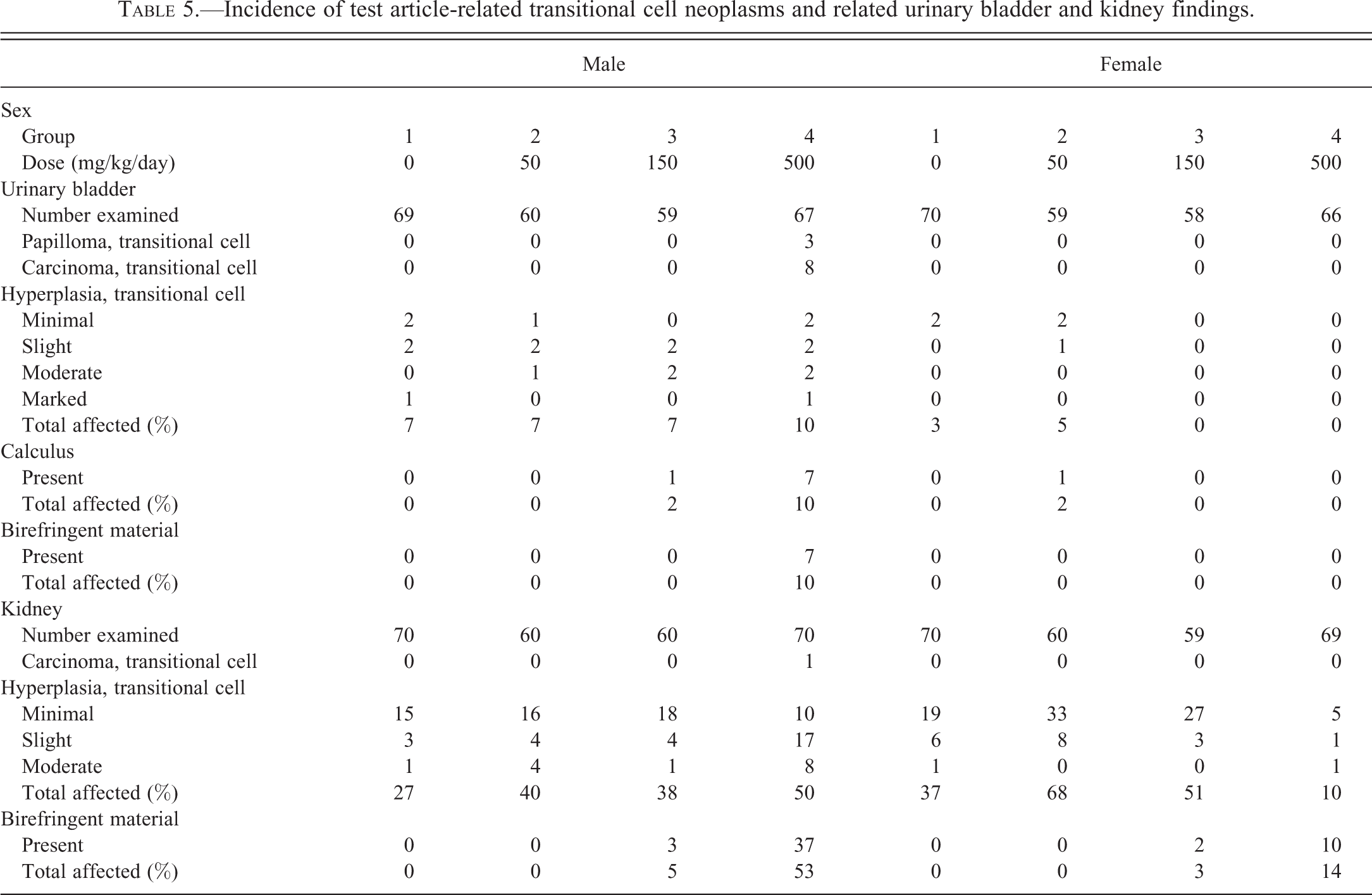
Histopathology of lersivirine-related urinary bladder neoplastic and related hyperplastic findings in high dose males. (A) Invasive transitional cell carcinoma that completely effaces the existing architecture. (B) Higher magnification of panel A showing the pleomorphic cells arranged in cords and nests with evidence of a few mitotic figures. (C) Low magnification view of an invasive transitional cell carcinoma that invades the wall of the urinary bladder. (D) High magnification of an invasive transitional cell carcinoma (see inset) with neoplastic cells arranged in thin cords. (E) Transitional cell hyperplasia within the urinary bladder of a high dose male rat. (F) Evidence of foreign material within the lumen of the urinary bladder admixed with debris. Inset shows the birefringent nature of the foreign material.
Lersivirine-related findings associated with urinary bladder neoplasms included increased incidence and severity of transitional cell hyperplasia in males given 500 mg/kg/day and presence of calculi within the urinary bladders of males given
Renal tubule cell adenoma and hyperplasia: Lersivirine-related renal tubule cell adenoma and increased incidences of renal tubule cell hyperplasia occurred in males and females given 500 mg/kg/day (Figure 4 and Table 6). Renal tubular adenoma and hyperplasia have been reported with increased severity of CPN (Hard, Betz, and Seely 2012), as was observed in this study in both sexes (CPN changes described subsequently). In fact, most renal tubular tumors were observed only in rats with CPN grades of 2 or higher (data not shown).
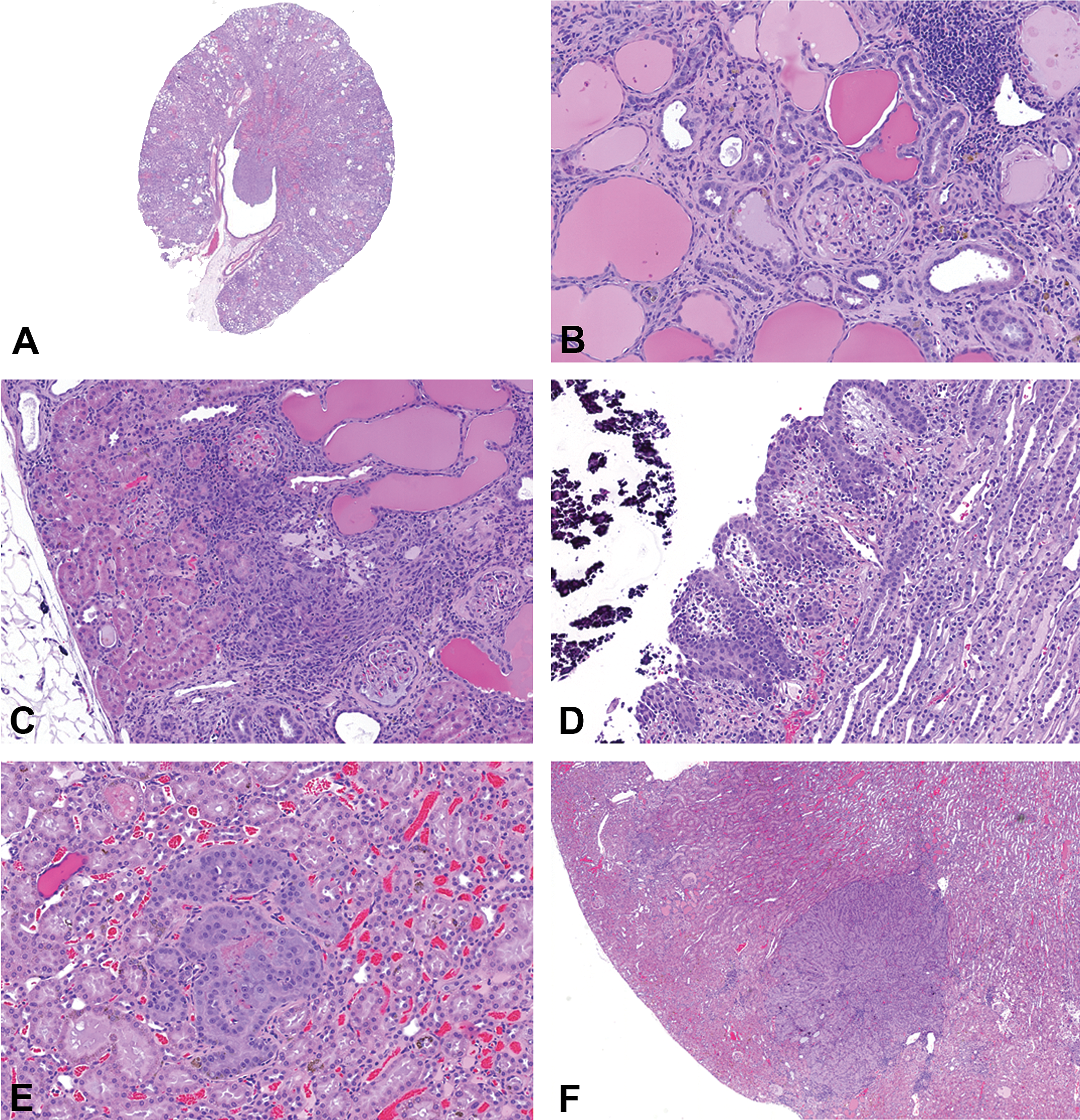
Histopathology of lersivirine-related findings in the kidney. (A) Note enlarged kidneys with dilated protein filled tubules chronic progressive nephropathy (CPN). (B) Higher magnification shows intratubular protein casts, stromal inflammation and fibrosis. (C) Areas of fibrosis, protein casts, and focal areas of prominent normal looking cortical renal tubules. (D) Focally extensive hyperplasia of renal pelvic urothelium with evidence of calculi within the pelvis. (E) Focal cortical renal tubular hyperplasia without any compression of the adjacent parenchyma. (F) Focal and discrete cortical renal tubular adenoma that compresses the adjacent parenchyma.
Incidence and severity of lersivirine-related kidney tubular cell neoplasms and chronic progressive nephropathy (CPN) in the kidney.
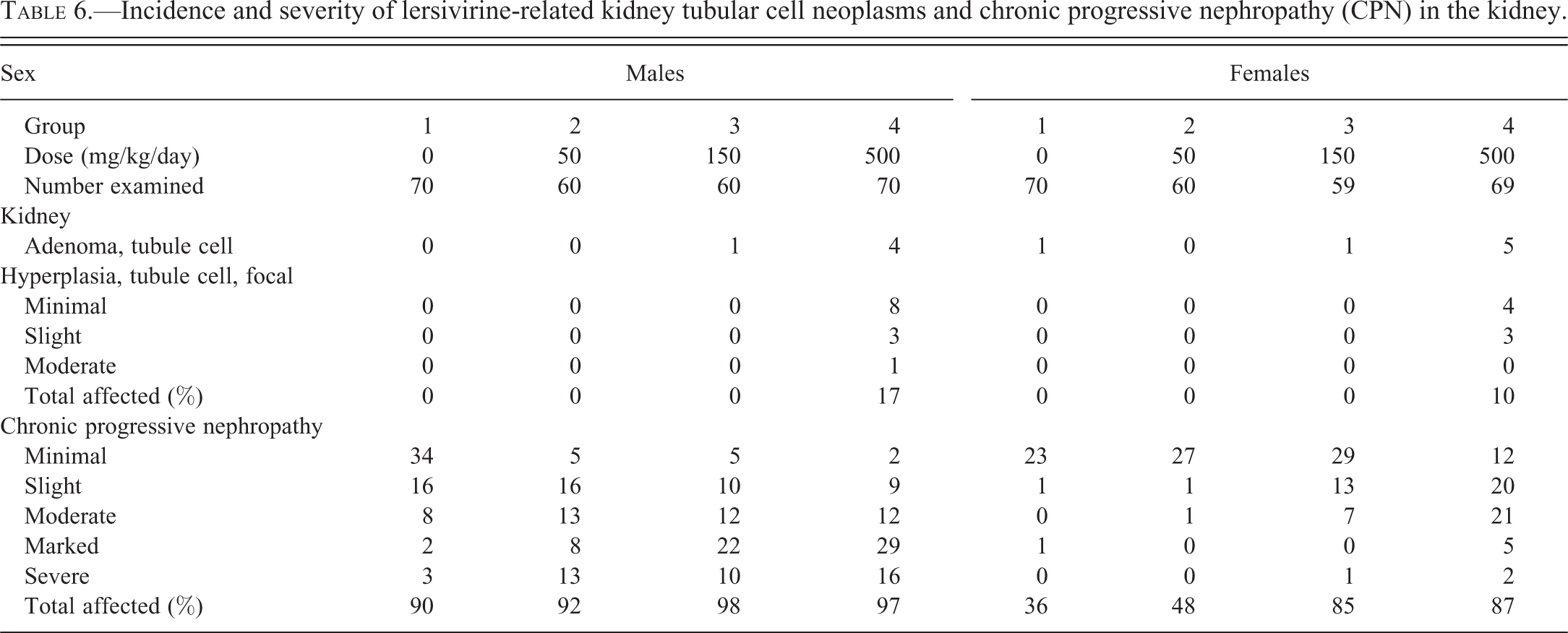
CPN: Lersivirine administration increased the incidences and severity of CPN in males given
Additional nonneoplastic findings attributed to lersivirine administration included polyarteritis (vascular/perivascular inflammation) in multiple organs in individual males and females given
Discussion
Lersivirine is a next-generation NNRTI for treating HIV-infected patients. For several of the previously approved first generation of NNRTI drugs, rodent carcinogenicity studies appear to have been supplied as postmarketing commitments. Since the issuance of the EMA guidance on antiretroviral therapies (EMA 2007), the general expectation is that carcinogenicity studies should be conducted prior to the granting of marketing authorization in the European Union. This is because the life expectancy of HIV-infected patients has increased significantly since antiretroviral therapies were first introduced. In this report, we describe neoplastic and nonneoplastic findings that were observed in a 2-yr carcinogenicity study in rats administered lersivirine.
Enzyme induction is commonly seen with NNRTI (Berruet et al. 2005; FDA 2012a, 2012b; FDA 2013a, 2013b; Hughes et al. 2009; Scholler-Gyure et al. 2009; Walker et al. 2009; Zabka et al. 2011). In humans and rats administered NNRTI, the CYP3A isoforms are the most commonly induced microsomal enzymes. Lersivirine, like other NNRTI drugs, is an autoinducer of hepatic microsomal enzymes in preclinical animal species (Walker et al. 2009). Autoinduction decreases lersivirine exposure over time in rats and mice and to a lesser extent in dogs. There was a modest increase in CYP3A activity over time in humans that did not affect human clearance of lersivirine (Walker et al. 2009). Short-term and chronic nonclinical studies with lersivirine have demonstrated liver and thyroid effects consistent with hepatic enzyme induction. Evidence of hepatic enzyme induction in rats included increased liver weights; hepatocellular hypertrophy; increased hepatocellular smooth endoplasmic reticulum; increased hepatic CYP 3A1, 2B1, and 2B2; increased hepatic uridine diphosphate glucuronosyl transferase (UDP-GT); and thyroid hypertrophy/hyperplasia (Houle et al. 2013; Walker et al. 2009).
The known class effects of NNRTI (being potent inducers of cytochrome p450s) and presence of proliferative changes in the rat dose-range finding study consistent with enzyme induction predicted a high likelihood of tumor development in the thyroid gland and liver (weight of evidence approach) in the rat carcinogenicity study. Indeed, administration of lersivirine to rats for up to 2 yr was associated with increases in neoplasms and related proliferative lesions in the expected target organs—liver and thyroid gland. Thus, the neoplastic and proliferative findings in the liver and thyroid gland in the 2-yr rat carcinogenicity study were attributed to the long-term effects of hepatocellular microsomal enzyme induction, associated hepatocellular hypertrophy (an adaptive response), and alteration of normal endocrine feedback mechanisms (Morton 2013; Zabka et al. 2011). No microscopic findings or neoplasms in the pituitary gland related to administration of lersivirine have been observed in any studies conducted in rats, mice, and dogs. The cause of the greater incidence of hepatocellular carcinomas in females compared to males is unknown.
Hepatocellular neoplasms in rodents that are attributed to hepatic microsomal enzyme induction generally have low relevance for human risk assessment if the mechanism of action has been appropriately characterized (Graham and Lake 2008; Greaves 2012; Holsapple et al. 2006; Walker et al. 2009). The absence of significant hepatic microsomal autoinduction in humans treated with lersivirine further diminishes the importance of this finding in preclinical animal models for human risk assessment.
Thyroid follicular cell hypertrophy and thyroid neoplasms were observed in rats treated with lersivirine. Induction of UDP-GT results in increased clearance of thyroxine (T4), chronic low-grade hypothyroidism, compensatory increase in thyroid-stimulating hormone, and subsequent activation, hypertrophy, and hyperplasia of thyroid follicular epithelium in rats that lead to thyroid follicular adenoma and carcinoma (Graham and Lake 2008; Hood, Hashmi, and Klaassen 1999; McClain 1989, 1992). Thyroid follicular cell hypertrophy secondary to UDP-GT induction and increased thyroid-stimulating hormone has been demonstrated in rats, mice, and dogs given lersivirine (Houle et al. 2013). Humans are much less sensitive than rats to perturbation of thyroid hormone metabolism and subsequent increases in thyroid-stimulating hormones. Thyroid follicular neoplasms in rodents that are related to increased clearance of thyroid hormone by increased UDP-GT are therefore not relevant to human risk assessment (Graham and Lake 2008; McClain 1989, 1992; Wu and Farrelly 2006).
A review of NNTRI approved in the United States and the European Union shows that a variety of neoplastic findings were observed in rats, mice, or both species (Table 7). Studies conducted in both rats and mice were 2-yr carcinogenicity bioassays while alternative 6-mo transgenic mouse bioassays do not appear to have been conducted for any of the approved NNTRIs. All compounds were noted to be enzyme inducers in rodents and, accordingly, exposure margins in comparison with human exposures tended to be relatively low. In all cases, hepatocellular tumors were noted, most notably in mice but also occasionally in rats. A 2-yr mouse carcinogenicity for lersivirine was not conducted. Instead, a 6-mo carcinogenicity study in rasH2 mice was planned, but it was canceled prior to initiation but after the development of lersivirine was terminated. Similar to rats, a 6-mo mouse general toxicity study and a 6-mo mouse investigative study demonstrated thyroid effects (follicular cell hypertrophy, increased thyroid weights, and increased thyroid-stimulating hormone and triiodothyronine levels) and hepatic effects (increased liver weights, increased total cytochrome p450 content, induction of UDP-GT, hepatocellular hypertrophy, eosinophilic foci, and hepatocellular adenomas). Product labels usually describe these findings in rats and mice as either of unknown relevance to humans, or as being specific to rodents, and therefore of limited relevance to human risk assessment.
Summary of carcinogenicity findings for other NNTRIs.

The 13-week rat dose range finding study for lersivirine had identified urinary bladder hyperplasia along with urinary bladder calculi in some of the animals in the high-dosage (nontolerated) groups. These nontolerated exposures were an order of magnitude higher than the exposures in the high-dosage group tested in the carcinogenicity study. However, despite this microscopic risk factor for positive cancer outcomes in rats based on the dose-ranging study, it was unclear whether long-term dosing with lersivirine would result in renal or bladder tumors. This was because bladder proliferation was noted only in the nontolerated high-dose animals and in association with calculi.
At the end of the 2-year study, transitional cell carcinoma in the renal pelvis and urinary bladder, urinary bladder hyperplasia, and renal tubular adenoma and renal tubular hyperplasia were associated with lersivirine administration. The proliferative finding of transitional cell hyperplasia and neopasia were attributed to irritation caused by calculi. Calculi have been reported as a source of mucosal irritation leading to hyperplasia of the transitional cell epithelium and subsequent neoplasia in rodents (Boorman et al. 1990; Dominick et al. 2006; National Toxicology Program 1983; Suzuki et al. 2010). Birefringent material noted in rare tubules of the kidneys of a few treated animals and in the urinary bladders of a few males was likely associated with drug material. However, attempts to biochemically characterize the crystals and prove that the crystals were drug substance were not successful.
Extensive CPN contributed to the deteriorating clinical condition of many animals, particularly males given
Footnotes
Author Contribution
Prashant R. Nambiar and Daniel Morton contributed equally to this manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
