Abstract
Soft tissue filler products have become very popular in recent years, with ever-increasing medical and aesthetic indications. While generally considered safe, the number of reported complications with tissue fillers is growing. Nevertheless, there is no specific animal model that is considered as the gold standard for assessing safety or efficacy of tissue fillers, and there are very little data on interspecies differences in reaction to these products. Here, we report on interspecies differences in reaction to a subcutaneous injectable co-polyester, composed of castor oil and citric acid. Comparison of the histopathological local tissue changes following 1-month postimplantation, indicated that in rats the reaction consisted of cavities, surrounded by relatively thin fibrotic enveloping capsule. In contrast, an unexpected severe inflammatory granulomatous reaction was noticed in Sinclair minipigs. To our knowledge, this is the first report on significant interspecies differences in sensitivity to tissue fillers. It emphasizes the importance of using the appropriate animal model for performing preclinical biocompatibility assays for biodegradable polymers, tissue fillers, and implanted medical devices in general. It also makes the Sinclair minipig subject for scrutiny as an animal model in future biocompatibility studies.
Soft tissue filler products have become very popular in recent years, with increasing market size, which is estimated to currently include 160 products from more than 50 companies (Funt and Pavicic 2013). While at first fillers were used mainly in aesthetic medicine for correction of rhytides and folds (Rzany et al. 2010), they are now being used also for volume replacement and enhancement procedures (Goldberg 2006). With growing use, the number of complications reported with filler injections has increased proportionally, and adverse reactions have been described with all injectable fillers (Zielke et al. 2008). The time until reaction differs between the various fillers, with a mean of 4.9 months for biodegradable fillers and 18.3 months for nonbiodegradable fillers (Zielke et al. 2008). While most of the complications related to fillers are of mild severity and transient, severe reactions have been reported, including tissue necrosis, severe infections, and disfiguring nodules, which are mostly related to the formation of foreign body granuloma (Sachdev et al. 2010; Funt and Pavicic 2013). Many of the more severe reactions are permanent and recalcitrant to treatment (Alijotas-Reig and Garcia-Gimenez 2008; Lowe, Maxwell, and Patnaik 2005). It is on this basis that proper and reliable preclinical animal-based biocompatibility studies are warranted for each new filler compound (Schuh 2008). Although previously several animal models have been used, including rabbits (Pitaru et al. 2007), pigs (Farkas et al. 2008; Shumaker et al. 2006), rats (Hillel et al. 2012), and mice (Ramires et al. 2005), there is no specific animal model that is considered a gold standard for assessing safety or efficacy of tissue fillers (Hillel et al. 2012). Furthermore, to our knowledge, there is no reliable data source for interspecies differences in reaction to the use of tissue fillers. This information is of utmost importance when assessing human health risk from tissue fillers. Here, we describe such interspecies difference in reaction to an injectable co-polyester designed for tissue augmentation, observed as part of preclinical safety studies. Knowledge of such interspecies differences can help in the design of future preclinical safety studies for tissue fillers.
The test material in these studies was a mixture of branched viscous polyester composed of castor oil EP (obtained from Tamar Pharma, Rishon LeZion, Israel) and citric acid USP (obtained from BioLab, Jerusalem, Israel) with a ratio of 93.5:6.5 w/w, and a cross-linked semisolid polymer of the same compounds, with a ratio of 92.5:7.5 w/w. The ratio between the branched polymer and the crossed-linked one was 66.6 to 33.3, respectively. The preparation of the product was performed as described by Domb et al. (2011). As control, Macrolane, large particle biphasic hyaluronic acid, obtained from Q-Med Sweden, was used. This is a commonly used product for volume restoration of soft tissues and is known to be safe and with almost no side effects (Cerqua and Angelucci 2013).
Three female Sprague-Dawley rats (obtained from Harlan, Jerusalem, Israel) were injected subcutaneously under anesthesia with the test material and with Macrolane as control in the posteriolateral flank, according to the previously described protocol with a 16-G needle (Vaisman et al. 2013). Each rat was injected twice with each compound in each side of the body. Rat experiments were approved by the Ethics committee of Hadassah–Hebrew University Medical Center (Jerusalem, Israel). Four Sinclair minipigs (obtained from Ben Meir farm, Yokneam, Israel) were injected subcutaneously under anesthesia in the ventral area lateral to the mammary gland with a 14 G cannula, following a 1 mm incision in the skin. A total of 10 injections were performed, 5 on each side of the animal. One minipig received 5 injections of Macrolane as control. Minipig experiments were approved by the Institutional Animal Care and Use Committee–Assaf Harofeh Medical Center, Be’er Yaakov, Israel. After 1 month, animals were euthanized, and tissue samples of the implantation sites and regional lymph nodes were harvested, preserved in 4% formaldehyde solution, and processed for histological examination. Tissues were trimmed, embedded in paraffin, sectioned at a thickness of approximately 5 microns, and stained with hematoxylin & eosin (H&E). We report only the findings from the implantation sites, as no pathologic findings were observed in the regional lymph nodes. Subcutaneous changes noted at the implantation sites of rats consisted of multiple empty, large cavities, surrounded by a relatively thin enveloping fibrotic capsule, consisting of fibroblasts, collagen, and mild aggregates of mononuclear cells (Figure 1A and B). No evidence of granulomatous reaction was noted, indicating that the test compound is nonirritant (i.e., excellent tolerability). All injection sites showed a similar response.
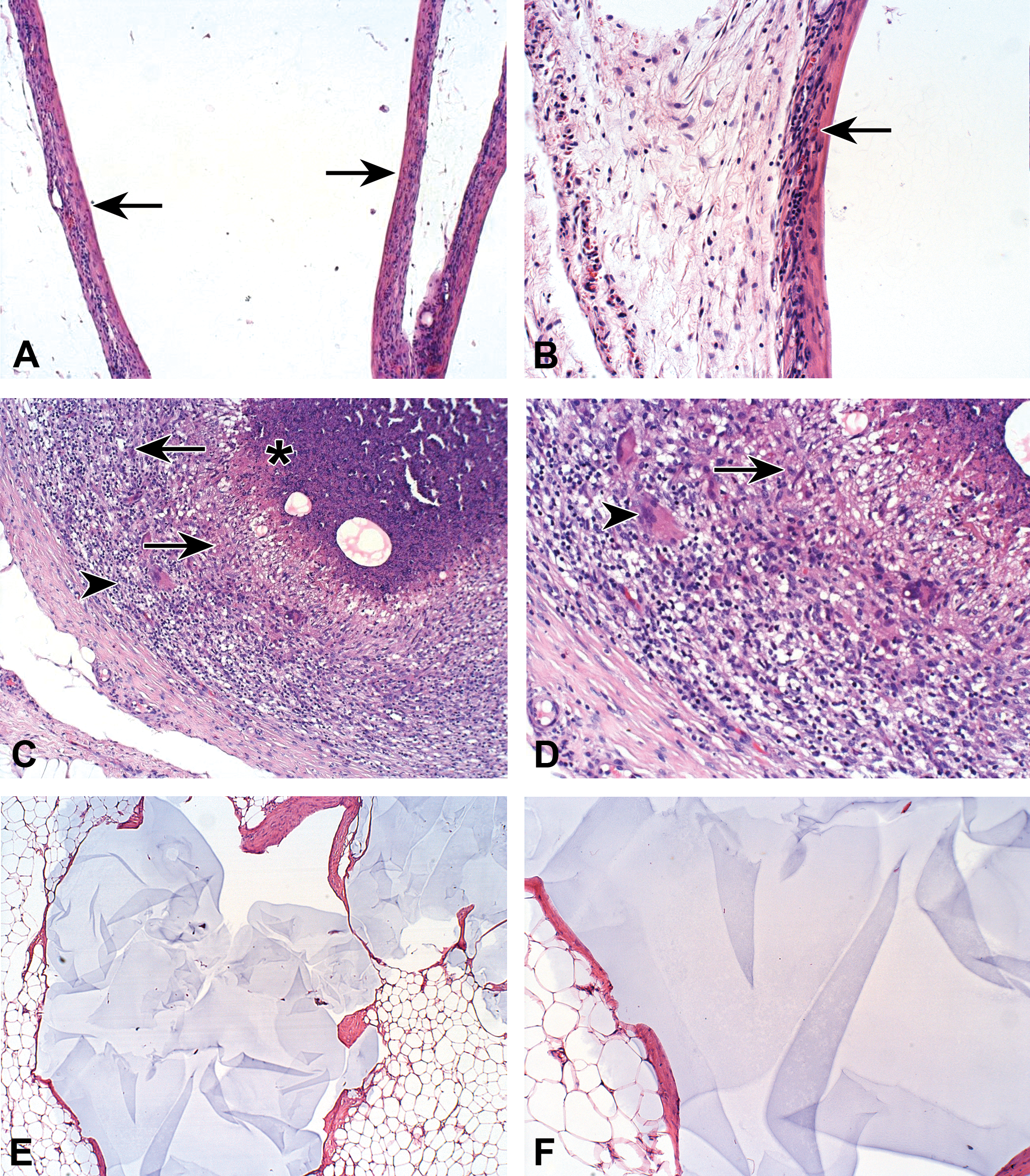
(A) Subcutaneous implantation site of co-polyester, composed of castor oil and citric acid, in the rat. Low magnification view of the capsular reaction (arrows) surrounding the implant. Note the capsule is composed of fibroblasts, collagen, and mild aggregates of mononuclear cells, suggesting excellent tolerability of the test compound (100×, H&E). (B) High magnification view of (A). Note the presence of mononuclear cells within the capsule (arrow; 200×, H&E). (C) Subcutaneous implantation site of co-polyester, composed of castor oil and citric acid, in the Sinclair minipig. Low magnification view of a granulomatous inflammation, each granuloma consists of a central area filled with necrotic debris (asterisk), surrounded by a relatively thick layer of macrophages (arrows). The outer thick layer is composed of a mixture of mononuclear cells, macrophages and sporadically distributed multinucleated giant cells (arrowhead). The findings are indicating severe reaction—irritation (i.e., nontolerance; 100×, H&E). The two circular empty spaces within the central necrotic area are reflecting cavities left by the washed-out dermal filler. (D) High magnification view of (C). Note the presence of macrophages (arrow) and sporadically distributed multinucleated giant cells (arrowhead; 200×, H&E). (E) Subcutaneous implantation site of Macrolane, composed of large particle biphasic hyaluronic acid, in the Sinclair minipig. The findings consist of cavity formation surrounded by capsule, containing amorphous material. The capsule surrounding the implanted material is relatively thin, composed of mature connective tissue, without any foreign body reaction. (40×, H&E). (F) High magnification view of (E). Note the presence of capsule lacking any inflammatory reaction (100×, H&E).
Subcutaneous changes noted at the implantation sites of the Sinclair minipig consisted of multiple foci of severe granulomatous inflammation, enveloped by a relatively thick fibrotic capsule. Each granuloma consisted of central area filled with necrotic debris, surrounded by a relatively thick layer of macrophages. Clear cavities reflecting washed-out dermal filler were also observed. The outer thick layer was composed of a mixture of mononuclear cells, macrophages and sporadically distributed multinucleated giant cells (Figure 1C and D). These findings indicate a severe irritation reaction (i.e., nontolerance), which was observed in all injection sites. In the samples that were implanted with Macrolane in the Sinclair minipig (Figure 1E and F), as well as in the rat, the findings were distinctly different, consisting of cavity formation surrounded by capsule in the subcutis, containing amorphous material. The capsule surrounding the implanted material was thin, composed of mature connective tissue, without any foreign body reaction.
Duranti et al. (1998) proposed a grading system for foreign body granuloma reaction based on the number and cell type present, and accordingly, the reaction observed in this Sinclair minipig study was judged to be of the highest degree (i.e., granuloma with encapsulated implants and a clear foreign body reaction). According to Lowe et al. (2005), what distinguishes granulomas is the collection of modified macrophages, epithelioid cells, and further fusion of these cells to form multinucleated giant cells, which may contain dozens of nuclei, randomly scattered throughout the cytoplasm. In fact, such giant cells were identified in our case.
These findings clearly demonstrate an interspecies difference in reaction to citric acid:castor oil polymer. While in rats no inflammation was observed after 1 month from implantation, a severe granulomatous inflammation was noticed in the Sinclair minipig. No such effects were shown in the sites injected with Macrolane hyaluronic acid gel as control. In order to confirm the specificity of the reaction to the injected compound, 2 sites in 2 Sinclair minipigs were injected with the same batch as the one injected to the rats, showing similar results. Of interest, 1 domestic pig was also injected subcutaneously with the test compound in the same manner as the Sinclair minipig, with no evidence of a granulomatous reaction (data not shown), in contrast to the effect observed in the Sinclair minipig. However, since only one domestic pig was studied, more experiments are needed in order to confirm this observation. Furthermore, minipig strains that are used more commonly in toxicity studies, such as the Goettingen and the Yucatan minipigs, should also be evaluated for potential dermal toxicity.
Swine models might offer a better suited animal model for the evaluation of cutaneous adverse reactions, due to their resemblance to human cutaneous anatomy and physiology (Jacobs 2006; Sambuco 1985). Indeed, minipigs have been used extensively in the past for dermal toxicity studies (Ganderup et al. 2012; Nuonoya et al. 2007; Swindle et al. 2012), and the most used types are the Goettingen and the Yucatan minipigs. The Sinclair minipig, which was developed at the University of Missouri Comparative Medicine Research Farm (i.e., the Sinclair Farm; Jones and Amoss 1982), is used comprehensively for melanoma research, due to its high incidence of cutaneous melanoma showing similar histopathology as in humans (Misfeldt and Grimm 1994; Okomo-Adhiambo et al. 2012). It is also used today for cardiovascular, musculoskeletal, urogenital, immunology, reproduction toxicology, gastroenterology, respiration, and nutrition research (Kohn 2012). However, its use as a model for dermal toxicity is less explored.
Foreign body granuloma formation following dermal filler injection is considered an overreaction, a complicating adverse effect (Lemperle, Rullan, and Gauthier-Hazan 2006; Winslow 2009). This reaction is not considered allergic, but rather caused by sudden stimulation of the memory of macrophages (Nyska et al. 2014). Clinically foreign body granulomas appear late, months to years after injection, and remain localized to the injection site (Winslow 2009). According to Lemperle et al. (2006), foreign body granulomas are occasionally seen with all dermal fillers at a rate of 0.01% to 1.0%, and if they occur, they appear after 6 to 24 months at all injected sites at the same time.
The reason for the exaggerated response of the Sinclair minipig to the injected polymer is not clear. In general, interspecies differences can result from variances in homology of genes, proteins, biochemistry, and physiology (Burgess-Herbert and Euling 2013). In the Sinclair minipig, a recent study has shown that surprisingly, the subcutaneous fat had more immune cells than the visceral fat (Booker 2010), in contrast to the reported normal distribution of immune cells, which are usually more abundant in the visceral fat. However, further analysis revealed that the macrophages in the subcutaneous fat layer of the Sinclair minipigs were mainly M2 macrophages, which have anti-inflammatory properties (Kang et al. 2008; Chawla et al. 2001). Nevertheless, it is possible that changes in numbers and type of additional infiltrating immune cells, such as neutrophils, or changes in the inflammatory cytokine secretion by adipocytes, which are yet to be revealed, stand at the basis of the observed exacerbated inflammation in the Sinclair minipig.
To our knowledge, this observation is the first to demonstrate significant interspecies differences in sensitivity to tissue fillers. It also underlines the importance of finding the appropriate animal model for preclinical biocompatibility assays for biodegradable compounds and tissue fillers. Further studies are needed to understand the biological mechanism that stands in the basis of the observed granulomatous inflammation in the Sinclair minipig, which makes this animal model subject for scrutiny in future biocompatibility studies.
Footnotes
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Abraham Nyska served as a consultant for Juvenis Ltd.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Juvenis Ltd., M.P Misgav, Israel.
