Abstract
In a 2-year rat carcinogenicity study, pegvisomant injected subcutaneously on a daily basis at doses of 0, 2, 8, or 20 mg/kg/day produced malignant fibrous histiocytomas (MFHs) at the injection sites of 3 male rats (5%) given 8 mg/kg/day and 5 males (8%) given 20 mg/kg/day. MFH was characterized by unencapsulated dermal and subcutaneous sheets of fusiform and spindle-shaped cells sometimes with areas of round and/or irregular, pleomorphic cells and variable numbers of large multinucleated giant cells. Some regions of MFH had a fibroblastic appearance with streaming cells forming storiform patterns, while other areas consisted primarily of round to plump irregular cells with more giant cells. Pegvisomant did not increase the incidence of MFH in female rats and did not produce any other neoplastic responses in rats. In the dermis and subcutis at the injection sites of many males and females, pegvisomant produced dose-related increased incidences and severity of histiocytic infiltrates consisting of vacuolated macrophages with variable mature or immature fibrous tissue. Neoplasms at injection sites did not result in marketing restrictions or a label warning for human cancer risk, highlighting that injection-site neoplasms in rats have low relevance for human risk assessment.
Introduction
Pegvisomant (Somavert®) is an analog of human growth hormone modified to be a growth hormone receptor antagonist. It has been approved in multiple geographies for the treatment of acromegaly, for example, in the European Union (EU) and United States in 2002 and 2003, respectively. Pegvisomant is a protein of recombinant deoxyribonucleic acid (DNA) origin containing 191 amino acid residues to which several polyethylene glycol (PEG) polymers are covalently bound (predominately 4–6 PEG molecules/protein molecule). The molecular weight of the protein is 21,998 Da. The molecular weight of each PEG molecule in pegvisomant is approximately 5,000 Da. The predominant molecular weights of pegvisomant are thus approximately 42,000, 47,000, and 52,000 Da. Pegvisomant is administered by subcutaneous (SC) injection.
The nonclinical data that supported the global marketing approvals included good laboratory practices (GLP) general toxicology studies of up to 6 months duration in rats and monkeys, genetic toxicology studies, reproductive toxicity studies, a local irritancy study, and human cancer cell line studies. As part of the approval in the United States, a commitment was made to conduct a 2-year rat carcinogenicity study. This article describes malignant fibrous histiocytomas (MFHs) at SC injection sites caused by pegvisomant.
Material and Methods
Animals and Husbandry
The study was conducted for Pfizer at MPI Research Inc. (Mattawan, MI). Five-week-old male and female Sprague-Dawley rats (Crl: CD®[SD] IGS BR, Strain Code 001) were obtained from Charles River Laboratories (Portage, MI). The rats were acclimated for at least 2 weeks prior to study start. Rats were randomly assigned to treatment groups based on body weight. Each rat was uniquely identified with an implanted microchip inserted at the base of the skull so as not to interfere with the nearest injection sites on the right and left lateral shoulders. Rats were individually housed in hanging stainless steel cages equipped with an automatic watering valve. The animal room environment was controlled (74°F ± 10°F, humidity 50% ± 20%, 12-hr light/dark cycle). Certified laboratory diet (LabDiet 5002, PMI Nutrition International, Inc., Richmond, IN) and drinking water were provided ad libitum. The protocol and any amendment/amendments or procedures involving the care and use of animals were reviewed by the Institutional Animal Care and Use Committee prior to the study. The animals care and experimental procedures for this study were conducted in compliance with the U.S. Animal Welfare Act and the ILAR Guide for the Care and Use of Laboratory Animals (National Research Council 1996).
Experimental Design, Test Substance, Observations, and Measurements
The study design, dose selection, and protocol were reviewed and approved by the Food and Drug Administration (FDA) prior to study initiation. The high dose of 20 mg/kg was selected based on significant toxicity at 30 mg/kg in a 26-week rat study including decreased body weight gain in males, nephropathy in females, and irritation, inflammation, fibrosis, and thickening of the skin at the injection sites. The SC route was used, as this is the clinical route of administration. Group size was 60 rats/sex/group and 2 vehicle control groups were used. Additional groups (6 rats/sex/group) were included for blood collection and used for toxicokinetic analyses at week 26. Rats were subcutaneously injected once daily for 2 years with 2-, 8-, or 20-mg/kg pegvisomant. Control group animals were injected subcutaneously with the vehicle.
Pegvisomant is a sterile, white lyophilized powder intended for SC injection. Commercial pegvisomant (Somavert®, 15-mg vials, Pfizer Inc, Kalamazoo, MI) was used for this study. (Each vial also contained 1.36 mg of glycine, 36.0 mg of mannitol, 1.04 mg of sodium phosphate dibasic anhydrous, and 0.36 mg of sodium phosphate monobasic monohydrate). Pegvisomant was diluted with Sterile Water for Injection (USP) to produce pegvisomant concentrations of 0.67, 2.67, and 6.67 mg/ml for animals receiving 2, 8, and 20 mg/kg, respectively. The vehicle control group received glycine, mannitol, sodium phosphate dibasic anhydrous, and sodium phosphate monobasic monohydrate diluted with Sterile Water for Injection (USP). The pH was adjusted, if necessary, to pH 7.4 ± 0.1 with 1N NaOH or 1N HCl. Injection volume was held constant at 3 ml/kg. Pegvisomant and vehicle dosing solutions were prepared daily and weekly, respectively, under sterile conditions.
Four injection sites were utilized on each rat in the study. They were the right shoulder, right flank, left shoulder, and left flank. Prior to dosing, the 4 dorsal dosing sites were clipped free of hair and the injection sites were outlined with an indelible marker. Sites were clipped weekly. Rats were dosed by SC injection every day for 2 years. Injections were rotated each day among the 4 injection sites. For example, on day 1 the right shoulder could be the injection site, on day 2 the right flank, on day 3 the left shoulder, and day 4 the left flank. On day 5, the right shoulder would be used again and so on for the duration of this 2-year study. Thus, each injection site received an SC injection every 4 days. Thus, if a rat was dosed for 2 years or 730 days, each of the 4 sites received a minimum of 182 injections.
Euthanasia of rats found moribund and at the scheduled terminal sacrifice was humanely conducted by carbon dioxide inhalation followed by exsanguinations via the abdominal vena cava.
All protocol tissues from all animals (with the exception of animals for toxicokinetic analysis) in all dose groups including all 4 injection sites from each animal were fixed in 10% neutral-buffered formalin except for eyes and testes, which were fixed in a modified Davidson’s fixative (Creasy and Jonnassen 1999; Latendresse et al. 2002), processed to paraffin blocks, sectioned at approximately 5 μM, stained with hematoxylin and eosin, and examined microscopically.
Statistical Analysis
Data from the 2 vehicle control groups were combined for statistical analyses (n = 120/sex). Male and female data were analyzed separately. Treatment group means were compared to the combined control group mean. Levene’s test was used to assess homogeneity of group variances. If Levene’s test was not significant, Dunnett’s test was used to compare each treatment group to the combined control group. If Levene’s test was significant, Welch’s t-test with a Bonferroni correction was used. The Peto method was used to evaluate tumor incidences.
Survival data were evaluated for a dose–response relationship and to determine if there was a difference between any treatment group and the combined control groups. The two-tailed Tarone’s test was used with all dose groups.
Results
Survival and Body Weight
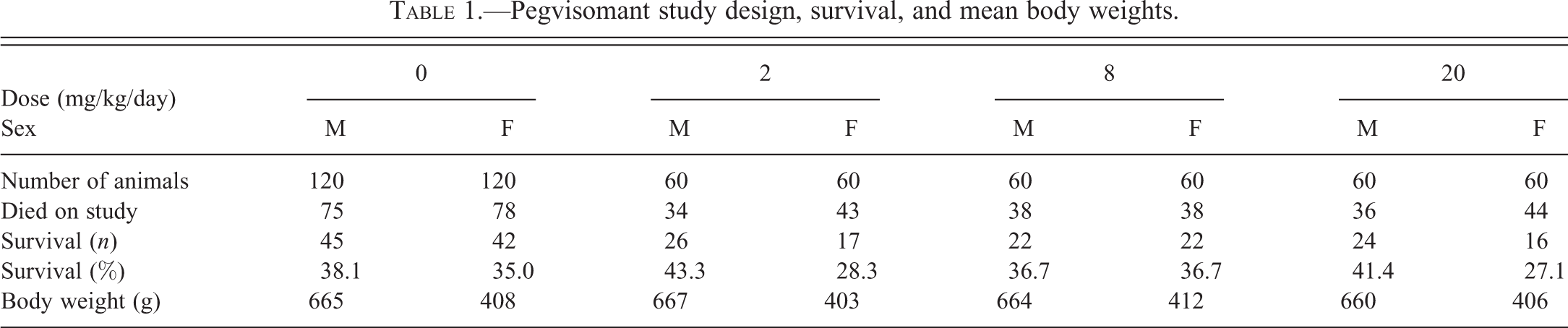
Pegvisomant treatment did not produce any effects on survival or body weight (Table 1).
Pegvisomant study design, survival, and mean body weights.
Exposure
Pegvisomant mean serum exposure expressed as area under the drug concentration curve (AUC) and maximal drug serum concentration (Cmax) increased with increasing dose (Table 2).
Mean rat serum exposure to pegvisomant and comparison to human exposure.
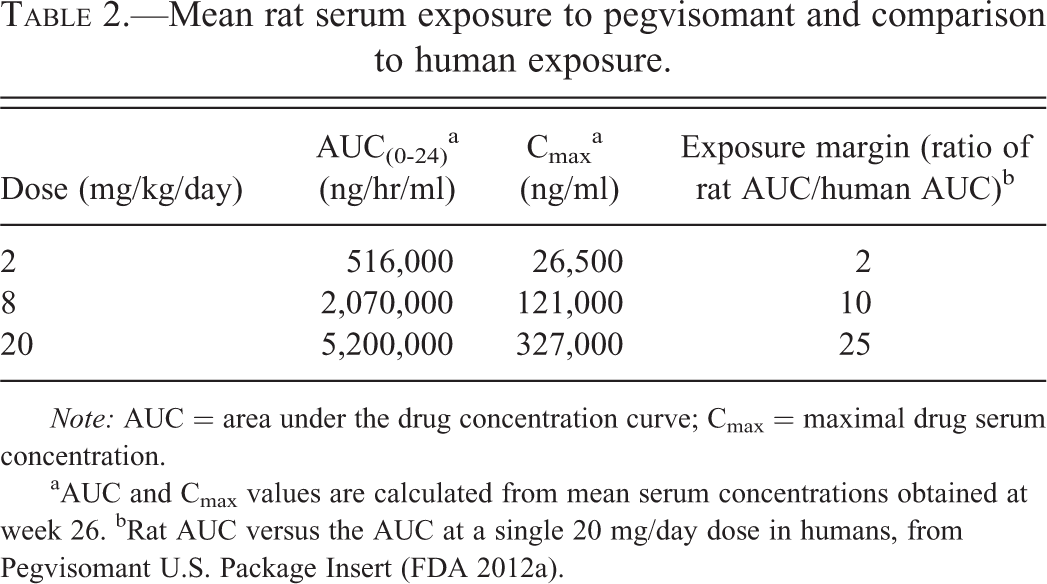
Note: AUC = area under the drug concentration curve; Cmax = maximal drug serum concentration.
aAUC and Cmax values are calculated from mean serum concentrations obtained at week 26. bRat AUC versus the AUC at a single 20 mg/day dose in humans, from Pegvisomant U.S. Package Insert (FDA 2012a).
Neoplastic Effects
In the 2-year rat carcinogenicity study, pegvisomant injected subcutaneously on a daily basis produced MFHs at the injection sites in male rats administered 8 and 20 mg/kg/day (Table 3). MFHs observed in this study were consistent with published descriptions of this neoplasm in rats (Greaves 1989, 1997, 2012; Greaves and Barsoum 1990; Schneider et al. 1999). These neoplasms consisted primarily of spindle-shaped to fusiform cells with less frequent areas including round to plump, irregular pleomorphic cells, and multinucleated giants cells with atypical, large nuclei, and variable nuclear condensation (Figure 1). In the fusiform cell areas, cell streaming in storiform arrangements was common. In some neoplasms, neoplastic fusiform cells were intermixed with collagen bundles, while in other regions, cells were arranged in solid sheets with minimal apparent stroma. Neoplastic cells formed solid masses in the deep dermis and/or subcutis that infiltrated through adjacent tissue components. Superficial ulceration and extensive deep necrosis were observed in some of the larger neoplasms. Metastases were not observed.
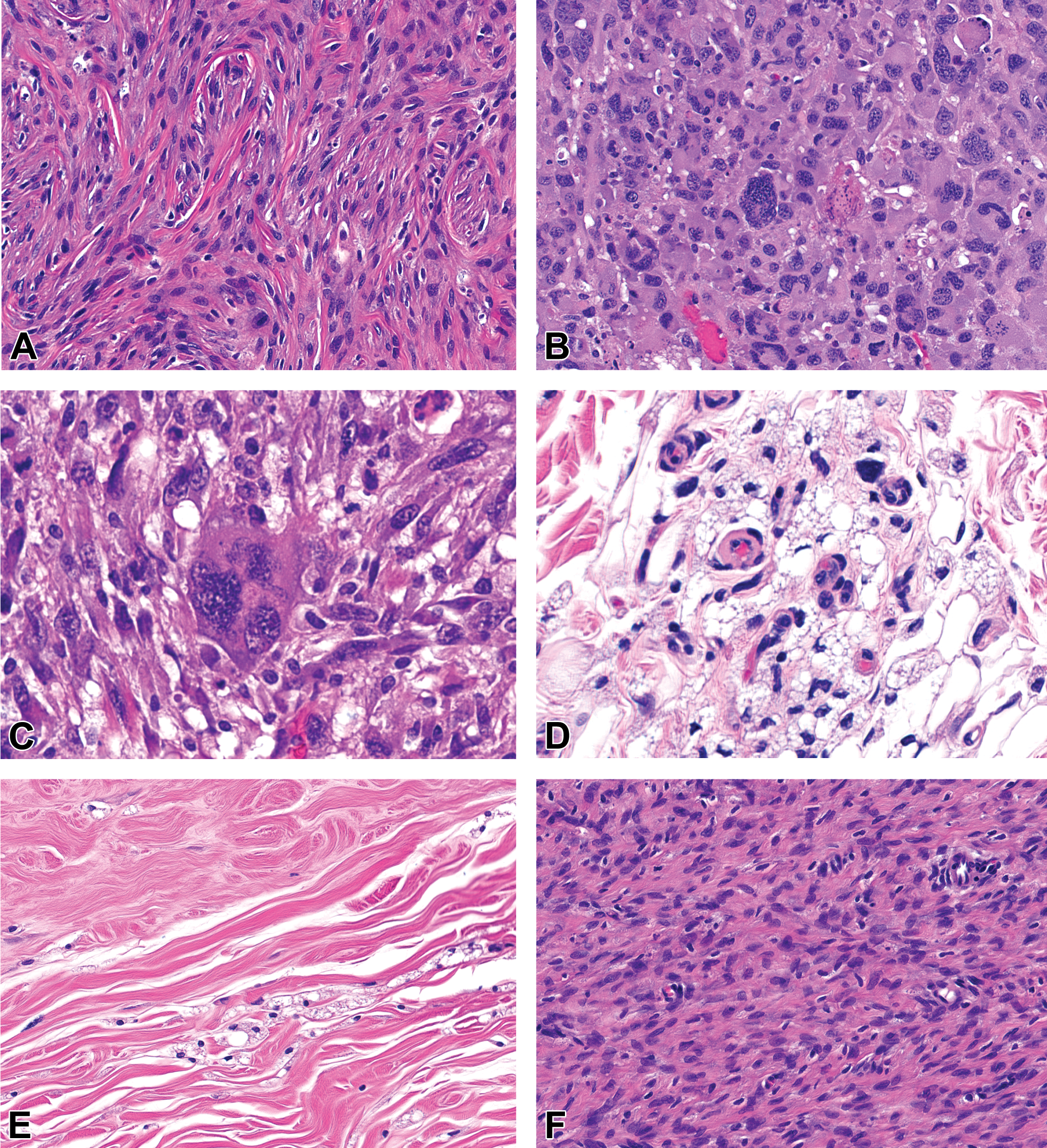
(A) MFH with fusiform cells streaming in a storiform pattern. H&E 20×. (B) MFH with sheets of variably sized, round, oval, and irregular neoplastic cells with pleomorphic, often bizarre hyperchromatic nuclei, atypical mitotic figures, and abundant cytoplasm. H&E 20×. (C) MFH with a very large neoplastic cell with multiple pleomorphic nuclei surrounded by fusiform cells with minimal stroma. H&E 40×. (D) Clusters of vacuolated macrophages in the deep dermis at an injection site. H&E 40×. (E) Vacuolated macrophages within an area of dense, mature fibrosis in the deep subcutis. H&E 20×. (F) Immature fibrous tissue in an area of extensive subcutaneous fibrosis at an injection site. H&E 40×. MFH = malignant fibrous histiocytoma; H&E = hematoxylin and eosin.
Incidence of injection-site malignant fibrous histiocytomas.

a p = .016 using a trend test performed excluding the group administered 20 mg/kg. b p = .000/0.004 trend test/multiplicity adjusted trend.
All 8 of the MFH tumors in male rats were observed at 1 of the 4 injection sites. Of the 8 affected males, at least 1 MFH occurred at each of the 4 sites (Table 4). Most injection sites showed no development of MFH as the incidence of MFH in the mid-dose group was only 3 of the 60 male rats (3 of the 240 injection sites) and in the high-dose group was 5 of the 60 male rats (5 of the 240 injection sites). The incidences of MFH (5.0% and 8.3% for mid- and high-dose males, respectively) increased in a dose-dependent manner, exceeding the historical control incidence (mean of 0.44%, ranging from 0% to 2.0% based on 675 control males at the testing facility).
Location and timing of malignant fibrous histiocytomas observed in male rats in the 2-year rat carcinogenicity study with pegvisomant.

Note: MFH = malignant fibrous histiocytoma; RS = right shoulder; RF = right flank; LS = left shoulder; LF = left flank.
aClinical signs at or near the injection site used as the first evidence of what was later diagnosed as MFH included swelling at the injection site or nearby region, for example, for Animal 1263, “Swelling, Axillary region/left” was first noted on day 680.
There was no apparent relationship between the dose and the time of MFH appearance or the time of necropsy (Table 4). The first appearance of a clinically observable sign (e.g., swelling) or palpable mass at or near the injection site ranged from 463 to 734 days in the 8 affected male rats. Whether or not these masses were truly MFH at the time of the first observation cannot be determined with certainty. The early deaths or scheduled sacrifices occurred between days 511 and 734 for the 8 affected male rats.
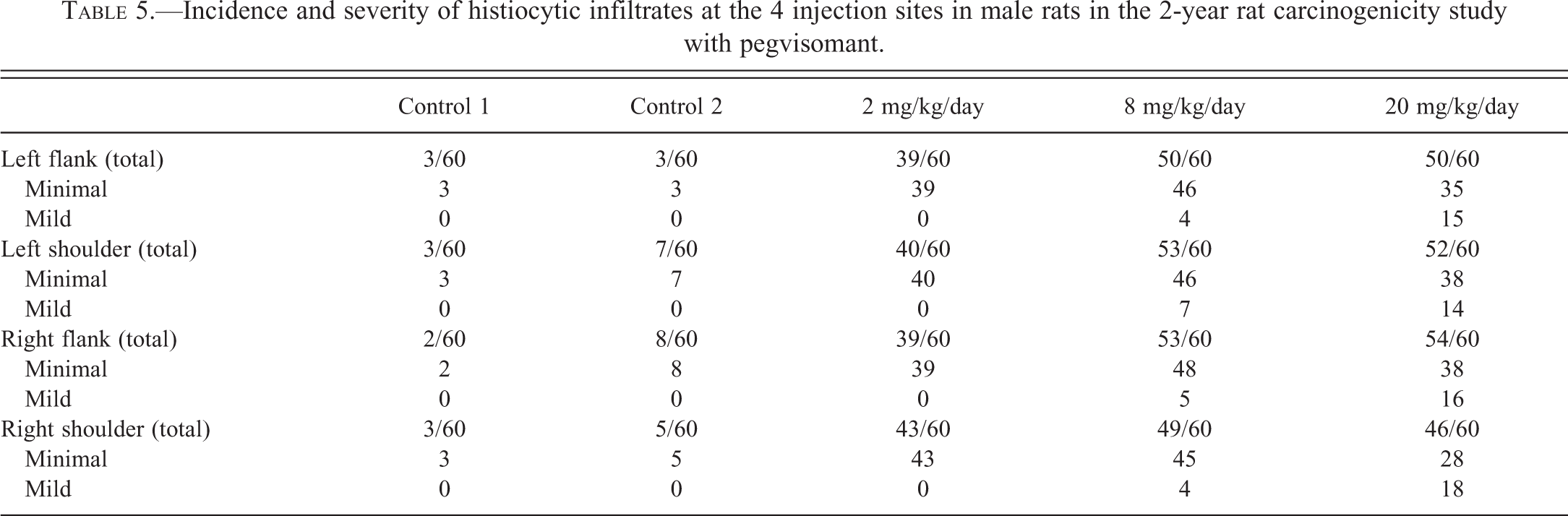
Most injection sites in rats treated with any of the pegvisomant dosages had histiocytic infiltrates at the injection sites (Table 5). Dose-dependent increases in the incidences and severities of histocytic infiltrates were observed. Dermal and SC vacuolated macrophages were described as a feature of the histiocytic infiltrates at injection sites (Figure 1). Although not documented independently, since they form part of the description, vacuolated macrophages were assumed to have been present when histiocytic infiltrates were recorded. Injection sites also had variable SC fibrosis. In some cases, the fibrous tissue was mature with widely scattered mature fibrocytes. In other injection sites, fibroblasts were immature, plump, and numerous. The relationship between histiocytic infiltrates and MFH was unclear.
Incidence and severity of histiocytic infiltrates at the 4 injection sites in male rats in the 2-year rat carcinogenicity study with pegvisomant.
Pegvisomant did not produce a statistically significant increase in MFH in female rats. Two cases of MFH were observed in the control females and 1 in the low-dose females. Pegvisomant treatment did not produce any other neoplastic responses in rats.
Discussion
MFH in humans is a rare neoplasm, yet historically it is one of the most common soft tissue sarcomas in humans (Clark et al. 2011; Fletcher 1992; Henderson and Hollmig 2012). MFH in humans arises from fibroblasts or primitive mesenchymals cells and is characterized by pleomorphic spindle cells, atypical (often very large) pleomorphic round to ovoid cells that may contain numerous and irregular mitotic figures, and multinucleated giant cells. Currently, MFH in humans usually is called undifferentiated pleomorphic soft tissue sarcoma. As diagnostic immunohistochemistry techniques and other diagnostic procedures have improved, many human neoplasms with morphology typical of MFH can be more accurately classified as other neoplasms and the diagnosis of MFH has declined (Fletcher 1992; Henderson and Hollmig 2012). Although risk factors for MFH development in humans have not been well characterized, MFH appears more commonly in patients with prior exposure to ionizing radiation (especially radiation therapy for cancer of the head and neck; Clark et al. 2011; Sturgis and Potter 2003). Isolated cases of MFH in humans associated with foreign bodies have also been reported (Kaplan and Lykosker 2012; Nishida et al. 2005; Theegarten, Sardisong, and Philippou 1995). Reports of MFH in humans associated with SC injection of drugs were not found, and these are assumed to be rare if they occur at all. It is interesting to note that similar to the results of this study in rats, most cases of human MFH (roughly two-thirds) are reported in males, usually during the 6th or 7th decade of life (Clark et al. 2011).
The increase in MFH in mid- and high-dose male rats does not represent an unexpected and serious risk to patients. Other nongenotoxic chemicals, inert biomaterials, and plant extracts can produce mesenchymal neoplasms (fibrous histiocytoma and sarcomas) in rats at the site of SC injection or implantation (Greaves 2012; Greaves and Baroum 1990). Concentrated solutions of glucose, other sugars, sodium chloride, food colorings, carboxymethylcellulose, high molecular weight dextrans, iron dextran, and surfactants that are not considered carcinogenic in humans can produce injection-site sarcomas in rodents when injected subcutaneously over long periods of time. Materials such as pegvisomant that produce relatively low numbers of neoplasms at injection sites without producing neoplasia in other tissues following prolonged repeated SC administration often are attributed to injection-site inflammation, tissue damage, prominent macrophage response (histiocytic infiltrates), and fibrosis (Greaves 2012). It is also worth noting that in the 26-week SC toxicology study in rats, vacuolated macrophages were observed at injection sites with high incidences and this finding was considered to be a nonspecific inflammatory reaction caused by repeated SC injections of high concentrations of pegvisomant.
In addition to pegvisomant, MFH has been reported with other drugs, chemicals, and inert materials administered via the SC route in rodent carcinogenicity studies or other rodent models (Tables 6 and 7). Octreotide and lantreotide, 2 drugs with a similar mechanism of action to pegvisomant (human growth hormone antagonism), have been reported to produce either MFH or fibrosarcomas in rodent carcinogenicity studies. However, growth hormone antagonists are not the only class of SC-administered drugs that produce MFH or fibrosarcomas. For example, a long-acting insulin product (insulin glargine) produced MFHs secondary to irritation and inflammation at injection sites in rats and mice that were considered rodent-specific (Stammberger et al. 2002). Liraglutide caused fibrosarcomas in the skin of male CD-1 mice attributed to the high local concentration of drug near the injection site, but did not produce similar neoplasms in female mice or in SD rats (European Medicines Agency [EMA] 2009; FDA 2012b). Mipomersen, an SC-administered lipid lowering oligonucleotide for familial hypercholesterolemia, produced MFH in male and female rats and fibrosarcomas in male mice (FDA 2013). Although these findings were mentioned in the package inserts for each of the compounds, injection-site neoplasia was not mentioned in the label warnings.
Marketed drugs that produce mesenchymal neoplasms at sites of administration.
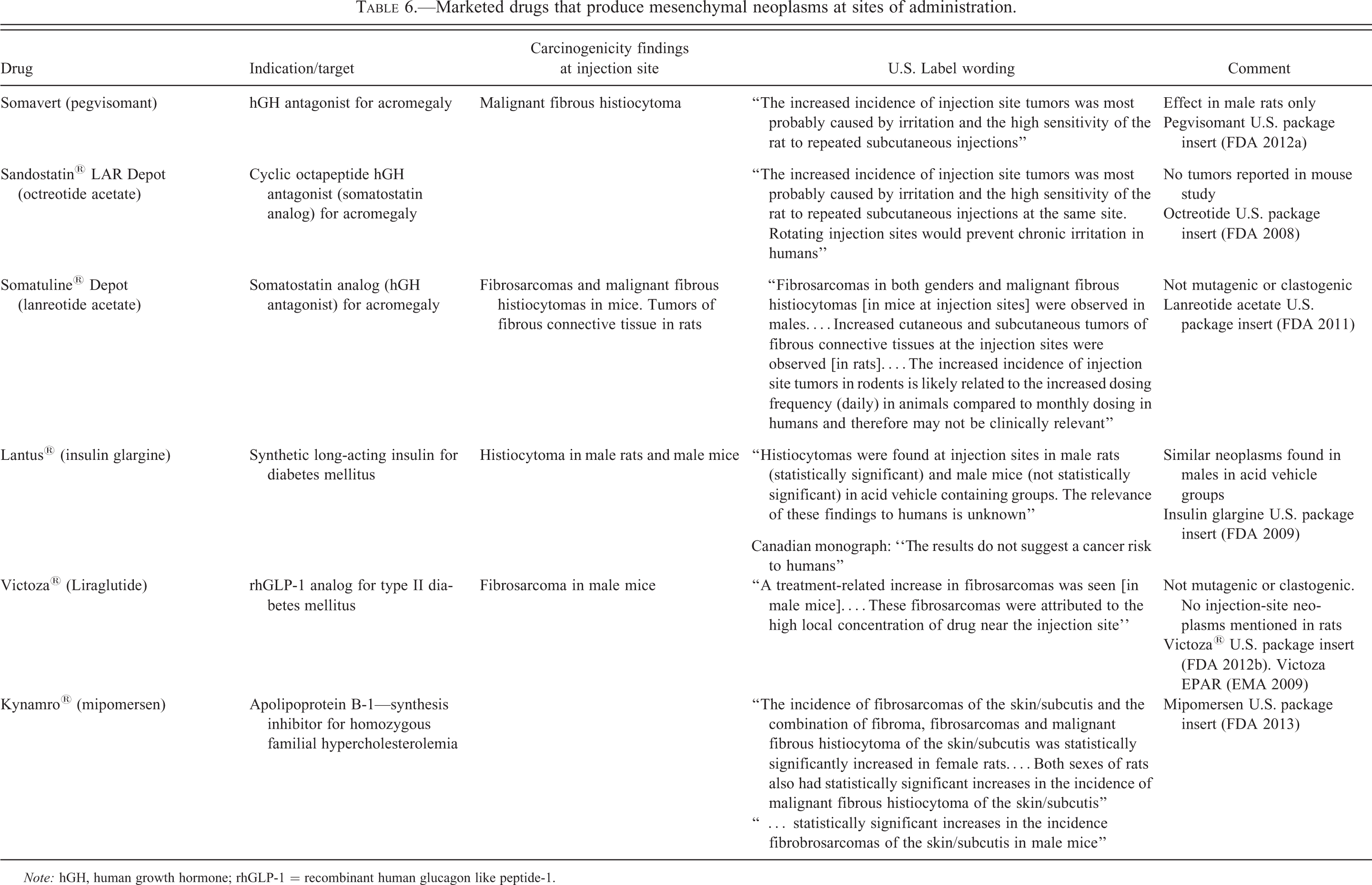
Note: hGH, human growth hormone; rhGLP-1 = recombinant human glucagon like peptide-1.
Chemicals and materials that produce mesenchymal neoplasms at sites of administration.

Note: MFH = malignant fibrous histiocytoma.
Pegvisomant produced fibrosis and histiocytic infiltrates (macrophage accumulation) at injection sites in rats that increased in incidence and severity with dose in both sexes. Similar degrees of histiocytic infiltrates and fibrosis were observed in males and females. MFHs were observed only in male rats at doses ≥8 mg/kg/day that also had significant fibrosis and histiocytic infiltrates at injection sites. Chronic irritation has been cited as the cause of injection-site neoplasms in rodents for a number of other drugs (Table 6). However, the hypothesis that MFH caused by pegvisomant results from chronic irritation does not explain why MFH occurred only in male rats when there was a similar degree of histiocytic infiltrates and fibrosis in both male and female rats.
The increased incidence of MFH in male rats observed with pegvisomant is not relevant for human risk assessment. There have been no reports of MFH or other injection-site neoplasms in patients (Schreiber et al. 2007; FDA 2012a; Pfizer, unpublished data). This conclusion is supported by over a decade of clinical experience with pegvisomant as well as experience with a number of other drugs administered subcutaneously (Table 6). Although injection-site reactions in patients have been described for pegvisomant, octreotide, lantreotide, insulin glargine, liraglutide, and mipromersen, injection-site neoplasms in patients are not reported in the U.S. labels and are not mentioned in the Warnings and Precautions section (FDA 2008, 2009, 2011, 2012a, 2012b, 2013). The package labeling suggests that injection-site neoplasms in rodents produced by nongenotoxic compounds raise little or no concern for human cancer risk.
Footnotes
Acknowledgments
The authors acknowledge Jonathan C. White, Theodore W Sloan Jr., Jean-Loic LeNet, Arthur Roth, Royal John Weaver, Dean Kendall, and Donald Beussing for their contributions to the pegvisomant carcinogenicity study.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
