Abstract
Regenerative biology/tissue engineering offers potential solutions for the repair and augmentation of diseased tissues and organs. Tissue engineering technology platforms currently under development for organ regeneration may function in part by recapitulating key mechanistic and signaling pathways associated with embryonic organogenesis. Temporal observations of observed morphological outcomes from the regeneration of tubular organs provide insights into the mechanisms of action associated with the activation of regenerative pathways in preclinical animal models and humans. These include induction of a neo-blastema, regeneration of laminarily organized mural elements (i.e.,
Introduction
Organ regeneration technologies aim to restore the original structure and functionality of a diseased organ. Typically, healing responses within mammals are characterized by fibrosis and scar tissue formation, not regeneration. Nevertheless, developing mammalian fetuses demonstrate wound healing without fibrosis and scar tissue formation during the first trimester (Adzick and Lorenz 1994). Additionally, compensatory hyperplasia of mammalian liver or kidney secondary to partial hepatectomy or nephrectomy, remodeling of bone, and epidermis subsequent to injury and regeneration of limb digit tips in humans and mice following amputation are all examples of regenerative outcomes in adult mammals suggestive of the existence of an innate regenerative potential within adult mammals (reviewed by Roy and Gatien 2008).
However, the clearest examples of regenerative outcomes secondary to injury have been established in model organisms such as Stages in Regeneration of Urodele Limb Secondary to Amputation The open wound is covered by wound epithelium to form a permissive epithelial structure called the apical ectodermal cap (AEC). Upregulation of matrix metalloproteinase (MMP) expression mediates structural reorganization of the ECM. Dedifferentiation of cell populations occurs proximal to the plane of amputation. Proliferation and migration of dedifferentiated cells are observed under the AEC. Formation of a
An understanding of these stages has already been applied to accelerate regenerative outcomes in mammals. For example, application of MMP1 to digit remnants of adult mice with amputation at the mid-second phalanx significantly improved regeneration of soft tissue and observed rates of wound closure. More multipotent progenitor cells, capillary vasculature, and neuromuscular related tissues were also noted (Mu et al. 2013). Furthermore, recent data on regenerative outcomes in mammals from tissue engineering of bladder, esophagus, and intestine provide additional evidence of the existence of a regenerative pathway in adult mammals mimicking aspects of that observed in urodeles, including formation of a neo-blastema (reviewed by Basu and Ludlow 2010, 2011). This regenerative pathway is characterized by a dependence on adequate vascularization and innervation at the site of regeneration. Importantly, these observations provide insight into a potential mechanism of action for tissue-engineered products characterized broadly as instructive signaling between mesenchymal cells of the regenerative construct and host epithelial cell populations. Such interactions recapitulate established signaling networks between epithelial and mesenchymal populations during organogenesis and/or regeneration in systems such as salamander or chick limb regeneration and mammalian kidney development (Figure 1).
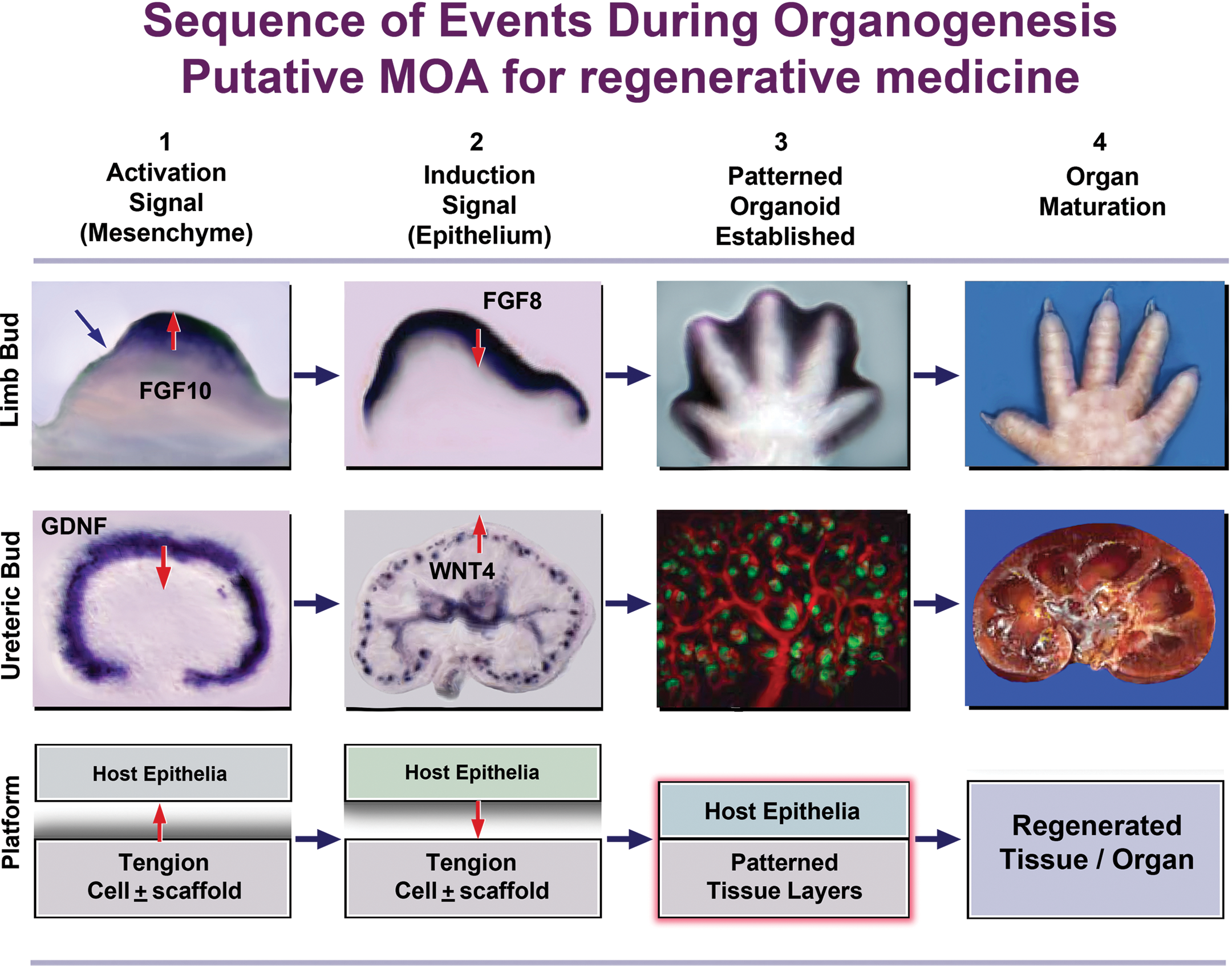
Technology platforms for the tissue engineering of tubular organs recapitulate aspects of instructive mesenchymal–epithelial signaling observed during organogenesis.
Classical experiments dissecting key signaling mechanisms underlying chick limb development and regeneration applied beads soaked in candidate signaling growth factors to elucidate the nature of the limb inducing element, together with grafting of candidate inductive and receptive tissues (reviewed by Martin 1998). FGF8 produced in the potential forelimb niche mesenchyme induces expression of FGF10 in flanking mesenchymal cell populations, which in turn then induces FGF8 expression in the overlying surface ectoderm. FGF8 secreted by the prospective limb ectoderm in conjunction with FGF10 in the mesoderm acts to maintain outgrowth of cells from the putative limb mesenchyme region. FGF8 also functions to induce expression of Sonic Hedgehog and thus establish a zone of proliferative activity within the developing limb bud (Figure 1). In the mammalian kidney, bidirectional signaling between the ureteric bud and the meta-nephric mesenchyme is ultimately responsible for mediating the seminal events of nephrogenesis. In the mouse, the ureteric bud, an outgrowth of the nephric duct at E10.5, signals to the surrounding meta-nephric mesenchyme, inducing condensation of meta-nephric mesenchymal cells around the tips of the invading ureteric bud. These mesenchymal condensates then undergo a mesenchymal–epithelial transition (MET) to form primitive epithelial vesicles known as renal vesicles. Continued branching of the ureteric bud leads to development of components of the collecting duct system and renal pelvis. Meanwhile, the renal vesicles undergo a systematic series of morphological changes, eventually fusing with the ureteric bud epithelia to form a continuous epithelial tubule, the S-shaped body. Infiltration of the S-shaped body by endothelial cells leads to formation of the glomerular vasculature. Continued branching morphogenesis from the ureteric bud epithelia in response to signaling from the neighboring meta-nephric mesenchyme in turn leads to induction of new aggregates of meta-nephric mesenchyme at ureteric bud tips and continued nephrogenic events. This iterative process of ureteric bud branching morphogenesis and induction of additional mesenchymal condensates continues along the radial axis of the developing kidney with the youngest nephrons induced toward the periphery. Induction of the ureteric bud is triggered through upregulation of Glial cell Derived Neurotrophic Factor (GDNF), whereas MET during conversion of metanephric mesechymal condensates to renal vesicles is controlled by members of the WNT family, including WNT4. Ultimately, it is the complex interplay between multiple overlapping signaling pathways, the ECM and ECM modifying factors across the meta-nephric mesenchyme and pro-nephric duct that mediates branching morphogenesis of the ureteric bud and subsequent assembly of the nephron (reviewed by Basu and Ludlow 2012). This insight may be harnessed to facilitate development of novel neo-organ products that recapitulate these signaling elements (Figure 1). Here, we review recent progress in regeneration of tubular organs with a particular focus on the GI tract.
A Technology Platform for Organ Regeneration
Tubular organ regeneration involves a specific, temporal sequence of cellular infiltration, vasculogenesis, neurogenesis, and the defined differentiation of mucosal, stromal, and parenchymal laminar tissue architectures (reviewed by Basu and Ludlow 2010). Strategies for organ and tissue regeneration must therefore achieve the dual objectives of triggering a true regenerative response while ameliorating any tendency toward fibrotic repair. The methodology first pioneered for regeneration of the bladder may serve as a foundational platform for the regeneration of any tubular organ including penis, vagina, lung, small intestine, stomach, and vessels.
Illustrated by Tengion’s Neo-Bladder Augment™ (NBA), this approach initially applied committed cell populations of target organ sourced (bladder)-derived urothelial cells (UC) and smooth muscle cells (SMC) complexed with a biodegradable, synthetic scaffold to trigger

Scanning electron microscope (SEM) images of an organ regeneration platform: smooth muscle cells complexed on a synthetic, biodegradable polymer matrix. Modification of the properties of the polymer permit creation of context-appropriate construct.
Maturation of a tubular regenerative construct into a laminarly organized neo-organ. Polymer matrix of construct degrades following implantation to be replaced by infiltrating smooth muscle cells, mesenchymal, endothelial, neuronal, and other cell populations.
Examination of the temporal sequence of neo-bladder regeneration in canine clinical studies illustrates the dichotomy in outcomes between implantation of acellular and cellularized scaffolds (Jayo, Jain, Wagner, et al. 2008; Jayo, Jain, Ludlow, et al. 2008). This distinction is a principal theme of most studies describing the regeneration of tubular organs. Cell-seeded scaffolds mediated a regenerative response within 1-month postimplantation, characterized by induction of heavily vascularized, smooth muscle-like parenchyma. In contrast, acellular scaffolds triggered a principally fibrotic, reparative outcome characterized by randomly organized collagen fibers with minimal vascularization. Baseline urodynamics were reconstituted within 4 months of implantation with cell-seeded scaffolds, whereas the urodynamic profile of animals implanted with acellular scaffolds remained abnormal throughout the trial period (Jayo, Jain, Wagner, et al. 2008). In a related dog study, restoration of tri-laminar bladder wall architecture occurred within 3 months of postimplantation and normal compliance characteristics of a urinary bladder wall developed by 12 months (Jayo, Jain, Ludlow, et al. 2008). Regenerated bladders were functionally and structurally stable for up to 2 years postimplantation (Jayo, Jain, Ludlow, et al. 2008). Importantly, although the construct volume was constant at implantation within variably sized dogs, the ratio of the regenerated bladder’s volume to host body weight adapted to the recipient animal’s size, demonstrating that the neo-organ responds to homeostatic mechanisms regulating organ volume (Jayo, Jain, Ludlow, et al. 2008).
Although these canine studies utilized both bladder-derived urothelial and smooth muscle cells, urothelial cells have been shown to be
NUC
Therapeutic urocystectomy removes a cancerous or malformed bladder and requires reconstructing a channel to divert urine outside the body. The most common treatment involves harvesting a segment of bowel to replace the removed organ; however, the use of gastrointestinal tissue in the urinary tract is associated with ∼70% of patients having long-term comorbidities linked to absorption of excreted metabolites and altered metabolic conditions including acid–base and electrolyte imbalances. Additionally, these patients can suffer from recurrent and chronic urinary tract infections. As an alternative to the use of intestinal tissue, Tengion is developing a urinary diversion that regenerates non-absorptive urinary tissue mucosa from a regenerative construct, Neo-Urinary Conduit (NUC). The NUC is produced by complexing an autologous population of adipose-sourced SMC and a biodegradable PLGA scaffold. Preclinical studies in a porcine model of cystectomy demonstrated regeneration of a urinary diversion within 3 months following implantation (Basu, Jayo, et al. 2012). In these studies, histological assessment of stromal and parenchymal events demonstrated formation of urinary tissue that was histologically characterized by the presence of all layers of a native urethra and appropriate immunohistochemical markers specific for urothelium, epithelium, and smooth muscle. The luminal surface was covered by urothelium extending from the ureteral–conduit junction (UCJ) to the mid segment of the NUC, followed by the presence of squamous epithelium extending into the distal end of the conduit to form a native-like mucocutaneous junction (similar to structure and function to native anterior urethra) and ending out in the stoma–skin interface (Rivera et al. 2013). Presence of smooth muscle layers or bundles was predominantly observed in the proximal and middle segments of the NUC, with less organization toward the distal end and adjacent to the stoma. Similar regenerative outcomes have been observed in humans and support application of a broad-based organ regeneration technology platform to trigger an innate regenerative induction of urinary-like tissue in adult humans (Rivera et al. 2013).
Regeneration of Muco-cutaneous Region at Skin–conduit Junction
Successful implantation of NUC requires the regeneration of a native-like muco-cutaneous region at the skin–conduit junction following implantation. A muco-cutaneous junction is a region where the
In the case of the urinary system, urine exits the body via the
GI Tract
Signaling interactions between endodermal epithelium and mesenchymal cells derived from splanchic mesoderm mediate axial differentiation of the primitive gut tube during embryogenesis into distinctive functional domains.
These individual components of the GI including the oral cavity, esophagus, stomach, small intestine, and colon represent locally specialized iterative variations of essentially the same laminarly organized tubular histologic architecture as the bladder. The molecular genetics underlying these differentiative signaling cascades is complex, but is broadly understood to involve members of the
Esophagus
A number of independent experiments suggest that regeneration of esophagus with the bladder-derived organ regeneration platform may be possible. For example, patch defects made in the abdominal esophagus of 27 female rats were repaired with cell-free scaffolds generated from gastric acellular matrix (Urita et al. 2007). Of the 24 survivors, none showed evidence of lamina muscularis mucosae regeneration, even at 18 months postimplantation. In contrast, in a canine model of esophageal resection and replacement, poly-glycolic acid (PGA) tubes seeded with a mixture of keratinocytes and fibroblasts triggered regeneration of smooth muscle laminar organization similar to native esophagus within 3 weeks postimplantation, whereas acellular PGA tubes formed esophageal strictures and led to near-complete obstruction within 2 to 3 weeks (Nakase et al. 2008).
In another canine study, cervical esophageal defects were patched with either SI sub-mucosa (SIS) alone, or with SIS (5 cm × 2.5 cm) seeded with 1 × 107 autologous oral mucosal epithelial cells (OMECs). After 4 weeks, dogs implanted with cell-seeded SIS showed almost complete reepithelialization and minimal evidence of inflammation, and, by 8 weeks post-surgery, regeneration of the underlying smooth muscle layer. Acellular SIS-grafted animals presented only partial reepithelialization and a more extensive inflammatory response by 4 weeks, and no muscular regeneration by 8 weeks (Wei et al. 2009). Attempts to introduce an acellular SIS tubular construct into the cervical esophagus of piglets were also unsuccessful, demonstrating scarification and a minimal regenerative response (Doede et al. 2009).
Preclinical rodent studies were initiated in our laboratory to evaluate the ability of constructs composed of SMC-seeded, PLGA-coated PGA patches to support regeneration of esophagus. In these studies, small (3–5 mm2) defects were introduced within the esophagus of rodents, such that the entire esophageal wall was removed within the defect. Injuries were subsequently repaired with cell-seeded, patch constructs and allowed to regenerate
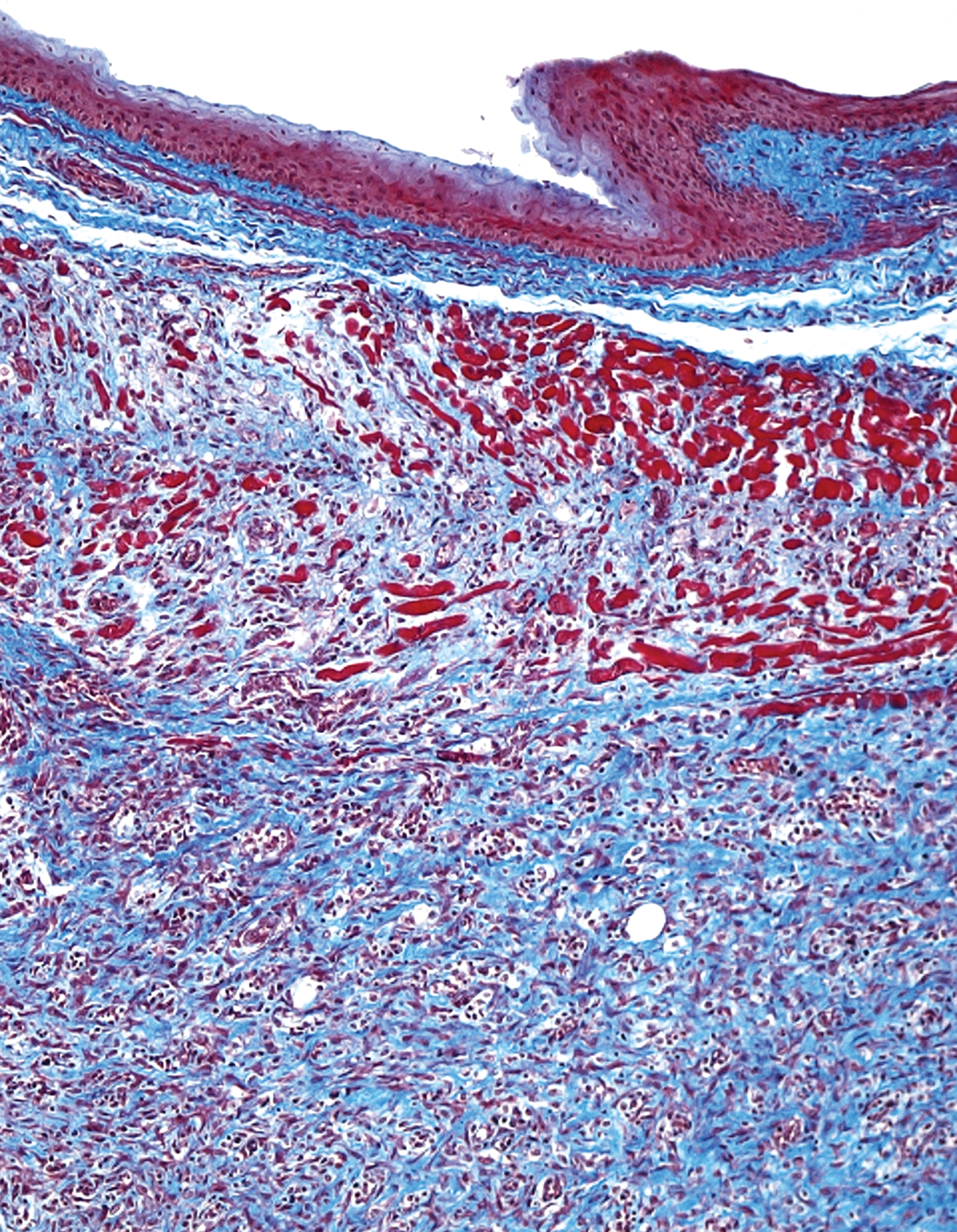
Early events in esophageal regeneration. Masson’s trichrome stain, transverse section of rodent esophagus postimplantation of regenerative construct at injury site (top). Bundles of developing smooth muscle are evident. 10×.
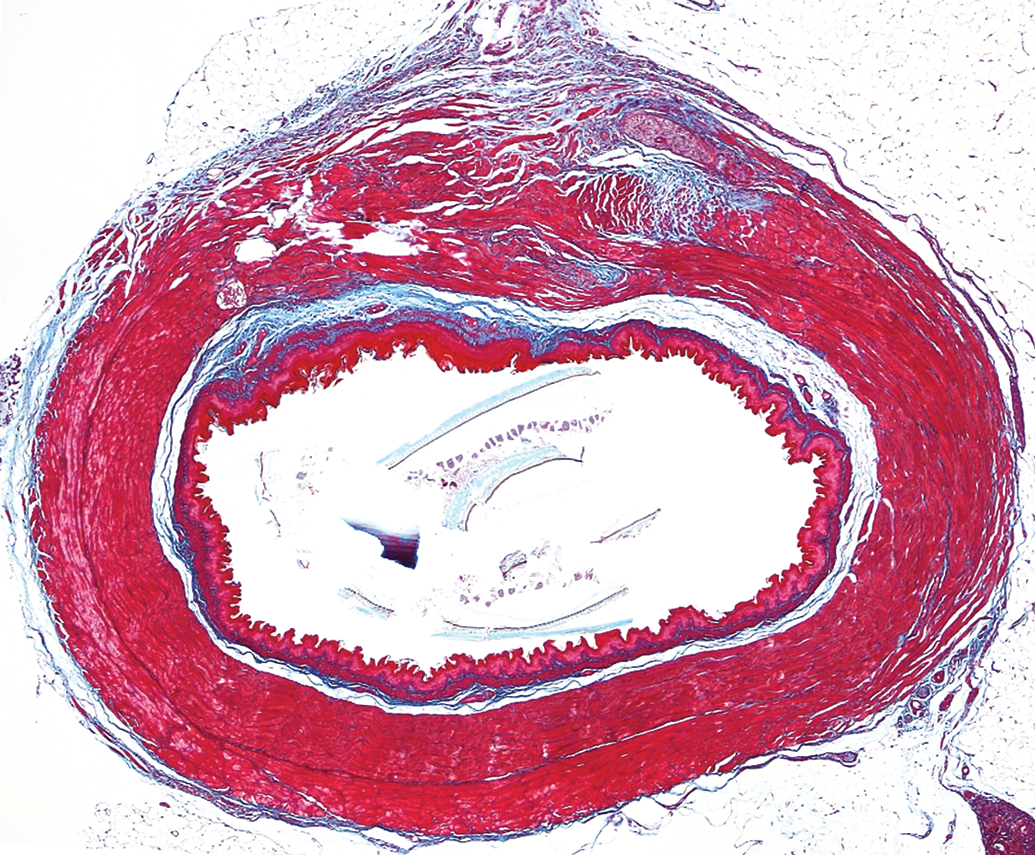
Regeneration of esophagus from patch-based implant in rodent esophageal injury model, 3 months postimplant. Note development of laminarly organized, muscular layers together with luminal epithelial layer. Area of regeneration, top of section. Native control, bottom of section Masson’s trichrome stain, 4×.
Small Intestine
Within the GI, the small intestine (SI) represents the most pressing unmet clinical need by far. For example, small bowel transplantation is an unsatisfactory current standard of care for pediatric small bowel syndrome, owing to technical difficulties inherent in surgery, the requirement for extended immunosuppression and high rates of graft rejection (Warner 2004). Intestinal epithelium is highly regenerative in adult mammals; therefore, it may reasonably be expected to be amenable to tissue engineering or regenerative medicine methodologies. Discrete multipotent intestinal stem cells positive for the Lgr5 marker, typically reside at the base of intestinal villi and are capable of self-organizing
Autologous organoid units, composed of incompletely dissociated clusters of epithelial and mesenchymal cells, were generated by partial digestion of intestinal epithelium (presumably containing resident intestinal stem cells) and used to seed poly-
An organ regeneration platform approach composed of seeded bio-polymeric scaffolds with SMCs may be applicable for SI regeneration. As with regeneration of the bladder and bladder derivatives, regenerative outcomes are catalyzed by instructive signaling interactions between mesenchymal cells on the regenerative construct and host-derived epithelial cell populations, potentially mimicking events associated with gastrulation. To this end, collagen sponge scaffolds with (SMC+) or without (SMC−) stomach-derived SMCs have been each grafted onto two 1 cm × 1 cm defects in surgically isolated, ileal loops of dogs (Nakase et al. 2006). SMC+ sponges were seeded at a density of 5 × 106 cells per cm3. The two loops were converted into bioreactors by constructing a double ileostomy on the anterior abdominal wall. At 8 weeks post-ileostomy, one of the loops was re-anastamosed to native intestine in 4 animals (2 dogs received the SMC+ loop, 2 received the SMC− loop) for an additional 4 weeks. Macroscopic analysis of tissue at the SMC+ implant sites demonstrated regeneration of native-like neo-mucosa. However, tissue at the SMC− implant sites remained ulcerated. Significantly enhanced vascularization, epithelialization, and circular muscle organization were also observed at the SMC+ implant sites relative to SMC− implant sites (Nakase et al. 2007). Increasing the number of SMCs seeded onto the scaffold resulted in a greater area of regenerated SI tissue, although no concomitant increase in the thickness of the smooth muscle layer was observed, indicating that there may not be a simple, linear relationship between the density of initial scaffold seeding and the final regenerative outcome (Nakase et al. 2007). Nevertheless, these data suggest that a platform technology of biodegradable scaffolds seeded with SMCs may be adequate to facilitate SI regeneration.
To this end, patch-based constructs composed of adipose derived, SMC-seeded, PGA felts were observed to support full regenerative outcomes upon implantation within rodent models of SI injury (Basu, Mihalko, et al. 2011). Regeneration of intestinal epithelia with microvilli as well as regenerated bundles of SMC were observed within 3 months postimplantation. Studies were also initiated to evaluate the regenerative potential of cell-seeded biomaterial constructs within the context of a tube. In addition to supporting the proliferation and migration of SI derived cells, such tubular constructs must have appropriate physical and mechanical properties that complement those of native tissue. In particular, within the GI tract, tubular constructs must be capable of supporting peristaltic movement of food or fecal matter. Commercially sourced, PGA-braided tubes were observed to have physical properties reminiscent of SI and were therefore applied in the context of tubular, cell-seeded constructs for evaluation of regenerative outcomes within rodent SI (Basu, Mihalko, et al. 2011).
As with the patch, complete regeneration of intestinal epithelia and partial regeneration of bundles of SMC were observed at 3 months postimplantation (Figure 6; Basu, Mihalko, et al. 2011). Implanted animals were capable of eating normally, gained weight, and passed fecal material in a manner comparable to healthy controls. A detailed analysis of regenerative outcomes in rodents implanted with SMC-seeded tubular constructs has been previously presented (Basu, Mihalko, et al. 2011). As with all other tubular organs, the presence of a cellular component on the regenerative construct is essential to a successful regenerative outcome. These data notwithstanding, the observed difficulties in regeneration of SI musculature from simple constructs derived from biomaterials used in bladder regeneration suggest that there are elements unique to SI currently missing from the current generation of construct prototypes. One such potential factor is the role of the enteric nervous system in facilitating regenerative outcomes within the GI tract.
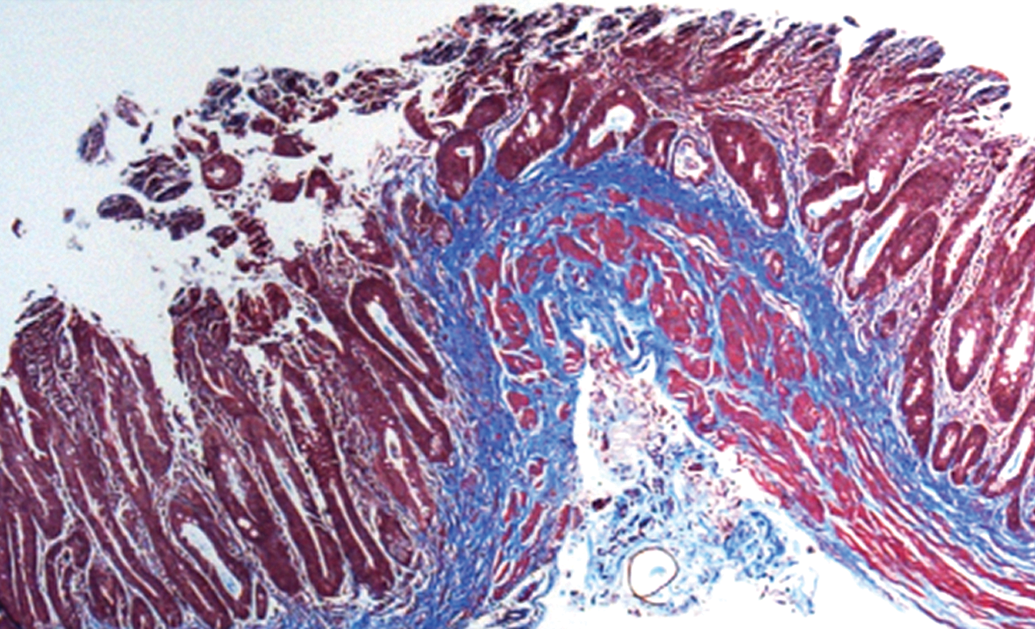
Regeneration of small intestine from tube-based implant in rodent small intestine injury model, 3 months postimplant. Note development of laminarly organized, muscular layers together with luminal epithelial layer. Masson’s trichrome stain, 10×.
Role of Nervous System in Regeneration
Classical experiments with limb regeneration in the salamander demonstrate that regenerative outcomes are dependent on the presence of nerves (Wessels 1977). If one forelimb of a salamander is enervated, and following recovery, both forefeet are amputated, regeneration of the missing limb is observed on the forefoot with intact nerves but not from the enervated forelimb (Wessels 1977). In a related experiment, a salamander is shielded from X-irradiation exposing only the forelimb region that is then irradiated to the point that limb regeneration is blocked. This failure to regenerate can be rescued by implantation of a piece of unirradiated nerve axon and surrounding Schwann sheath into the irradiated forelimb (reviewed by Wessels 1977). At a cellular level, the AEC forms, but is not maintained in the absence of nerve axons, leading to failure of blastema induction. Regenerating axons induce the synthesis and secretion of anterior gradient protein (AGP) by distal Schwann cells during dedifferentiation and by the gland cells of the AEC during blastema growth stages. AGP promotes the regeneration of a denervated limb to digit stages when electroporated into the limb during dedifferentiation. Once a critical mass of blastema cells has been attained, the blastema can undergo morphogenesis in the absence of the nerve, but the regenerated limb will be a miniature, because the nerve is no longer inducing the AEC to carry out its AGP-mediated proliferative function. AGP expression by both Schwann cells and the AEC is induced by axons, but the nature of the inductive agent remains unclear (Stocum 2011).
With these observations in mind, it is possible that incorporating neuronal cells within the regenerative construct or implanting the construct with an awareness of a neighboring nerve plexus may be important in promoting adequate regenerative outcomes. To this end, studies are currently underway to definitively establish a role for the nervous system in mediating regeneration of the GI tract.
Summary
An organ regeneration technology platform composed of adipose-sourced SMC complexed with a synthetic, biodegradable scaffold is broadly applicable to regeneration of multiple tubular organs. Constructs function by manipulating fundamental signaling pathways between mesenchymal and epithelial cell populations to trigger regenerative outcomes mimicking organogenesis during development and regeneration in organisms such as urodeles, including induction of a neo-blastema. Tubular organ regeneration is influenced by adequate vascularization and cellular constituents present at the local site. Mucosal regeneration is influenced by blood supply with organ-specific events controlling biological events. A role for nerve tissue in gastrointestinal regeneration remains to be established.
This ability to apply the same fundamental signaling elements that mediate the regenerative process through application of the same combination of cells and biomaterials toward regeneration of multiple tubular neo-organs will greatly facilitate development of a manufacturing platform with commonalities throughout bio-processing.
Footnotes
Acknowledgment
We thank Randal McKenzie (McKenzie Illustrations, rmac3@att.net) for the graphics.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors declare an equity and intellectual property interest related to the content of this work in Tengion, Inc.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
