Abstract
In this study, histopathologic changes following carbaryl exposure for 96 hr were investigated in the digestive tract of Levantine frog, Pelophylax bedriagae. Adult frogs were exposed to carbaryl once by oral gavage in concentrations of 0.05, 0.1, and 0.2 mg/g. Histopathological changes were more prominent in medium- (0.1 mg/g) and high-dose (0.2 mg/g) groups than in the low-dose (0.05 mg/g) group. Esophageal cells showed vacuolization, cellular swelling, nuclear pyknosis, karyolysis, and necrosis. Additionally, esophageal glandular atrophy and infiltration of inflammatory cells around esophageal glands were observed at medium and high doses. In the stomach, there were prominent histopathologic defects such as cellular swelling and necrosis in gastric glands, necrotic cells within the interstitial spaces, separation of epithelial cell layer, congested vessels, and hemorrhage at medium and high doses. In the intestine, detachment of epithelial layer, epithelial cell disorganization, inflammation, and necrosis were detected at medium and high doses. The results of this study showed that carbaryl caused adverse effects on the digestive tract of the Levantine frog, P. bedriagae.
Introduction
Carbaryl (1-naphthyl N-methylcarbamate) is a widely used carbamate insecticide that is used in the control of over 100 species of insects on citrus, fruit, forests, lawns, nuts, ornamentals, shade trees, and other crops, as well as on poultry, livestock, and pets (Environmental Protection Agency [EPA] 2004). It has the same general mode of action as other cholinesterase inhibitors: carbamates and organophosphates (Cox 2005). Carbamate insecticides are activated by carbamylating the esteratic active site of acetylcholinesterase (AChE; Watts and Wilkinson 1977). Thus, they are reversible inhibitors of the enzyme AChE (Gruber and Munn 1998; Mora et al. 2000). Amphibian declines have been linked to these insecticides (Davidson 2004). Carbaryl especially has adverse effects on endangered anuran populations (Garber, Jones, and Steeger 2007).
The adverse effects of carbaryl and carbamates on amphibians’ health have been conducted by many researchers (Bridges 1997, 2000; Bridges and Semlitsch 2000; Relyea and Mills 2001; Boone and Bridges 2003; Relyea 2003, 2005; Rohr et al. 2003; Boone et al. 2004; Metts, Hopkins, and Nestor 2005; Davidson et al. 2007; Bacchetta et al. 2008; Kang et al. 2010; Distel and Boone 2009, 2010; Bulen and Distel 2011). The striking point is that these studies are generally about the effect of carbaryl on tadpoles. Although extensive literature on the effects of pesticides on histopathological and biochemical aspects of fishes and other vertebrates is present, amphibians as a group have only recently been included in routine toxicological assessment of environmental chemicals (Renuka 2007).
Pelophylax bedriagae, which was formerly considered a subspecies of Pelophylax ridibundus primarily known as Rana ridibunda, is a widespread species along the Aegean coast and the southern part of the Anatolian highlands but ranges widely in the eastern Mediterranean in Turkey. P. bedriagae mainly feed on aquatic invertebrates and insects (Amin, Düşen, and Oğuz 2012). Çiçek and Mermer (2007) who investigated the food composition of the marsh frog populations inhabiting Turkey’s Lake District stated that the species mainly fed on invertebrates and especially on terrestrial preys belonging to arthropod groups (75.17%), and the most frequently consumed preys relating to numeric proportion were Diptera (19.85%), Coleoptera (12.72%), and Hymenoptera (10.02%). As clearly seen, adult frogs are likely to be affected by eating these kinds of insect species contaminated by insecticide. Carbaryl is extensively used in Turkey (Delen et al. 2005), which is home to a variety of animal species. There is a risk of this frog species like other animal species to be affected in regions where carbaryl is used. As is known, the digestion is an important life process. But there are few studies about the effects of pesticides on the digestive tract. To that end, this study was designed to investigate for the first time the histopathologic effects of carbaryl on the digestive tract of adult individuals of P. bedriagae.
Materials and Methods
Animal Groups and Experimental Design
The protocol was approved by the animal ethical committee of Ege University, Faculty of Medicine (2011-165). Adult individuals of P. bedriagae were caught around İzmir/Turkey. There were no significant differences in weight (24.9 g ± 0.04) between control and experimental frogs. They were maintained under laboratory conditions at a 12-hr dark–light cycle, 22 ± 3°C temperature, and 45 ± 5% relative humidity in the Herpetology Research Laboratory at Ege University Campus, Bornova, İzmir. After 15 days of acclimation, the frogs were randomly assigned to either the carbaryl-treated groups or the control group, each consisting of 8 frogs (4 males and 4 females). Each group was maintained in plastic cages in which lower surface was covered with water and ad libitum crickets for food. As noted formerly, adult frogs mostly fed on insects (Çiçek and Mermer 2007). Therefore, they may be exposed to carbaryl by consuming insects that are covered in carbaryl. Fair, Kennedy, and McEwen (1995) found that grasshoppers had mean residues of 17 µg/g 2 days following rangeland application of 0.5-kg active ingredient/ha. Using this insect residue data and carbaryl application rates, which can vary from 1.12- to 22.42-kg active ingredient/ha (EPA 2004), DuRant, Hopkins, and Talent (2007) determined that a 10-g lizard consuming 1-g prey could ingest dose concentrations ranging between 3.9- and 78.5-µg/g 2 days following carbaryl application. Taking these into account, the chosen concentrations of carbaryl in this study (low dose: 0.05 mg/g, medium dose: 0.1 mg/g, and high dose: 0.2 mg/g) are the ones which frogs could encounter in the environment. Carbaryl (purity 98%) was supplied by AgroBest Grup (İzmir, Turkey). Carbaryl was dissolved in acetone. Acetone was given to the control group during the experiments. It did not cause any important histologic effect on digestive tract. Carbaryl was administered to frogs once via oral gavage using an Eppendorf micropipette (0.5–20 µl) to examine histologic changes. After 96 hr following exposure to carbaryl, frogs were euthanized by decapitation with a guillotine under ether anesthesia, and their digestive tracts were quickly removed.
Histological Analyses
For light microscopic examination, digestive tracts of the experimental frogs were fixed in Bouin’s fixative for 24 hr, dehydrated in ethanol, cleared in xylol, and embedded in paraffin. Thus, fixed tissues were then serially sectioned at 5 μm and stained with Mayer’s Hematoxylin and Eosin. Sections of all digestive tract parts were entirely examined by light microscopy and then photographed with Olympus CX31 (Tokyo/Japan).
Results
The current study was designed to determine histopathologic changes in the digestive tract of P. bedriagae, which was exposed to different carbaryl concentrations. After exposure to carbaryl, slowing down of the movements especially in high-dose animal group was observed. But death did not occur during the experiment.
Light microscopic examinations demonstrated that epithelial layer of the esophagus was formed of ciliated columnar cells and goblet cells in the control group (Figure 1A). In addition, esophageal glands were observed (Figure 1B). In the low-dose group, the appearance of the histologic view of the esophagus was similar to that of the control group. In the medium-dose group, vacuolization and separation of epithelial cells were observed (Figure 2A and B). In the high-dose group, esophageal cells exhibited cellular swelling and necrosis characterized by nuclear pyknosis and karyolysis (Figure 3A and B), and vacuolization was also observed in these regions of the esophagus (Figure 3C). In some esophageal glands, cellular swelling, nuclear pyknosis, karyolysis, and necrosis were detected (Figure 4A). Esophageal glandular atrophy was observed, but it was only lined by flattened epithelial cells (Figure 4B). In addition, hemorrhage and infiltration of inflammatory cells around esophageal glands were determined (Figure 4C).
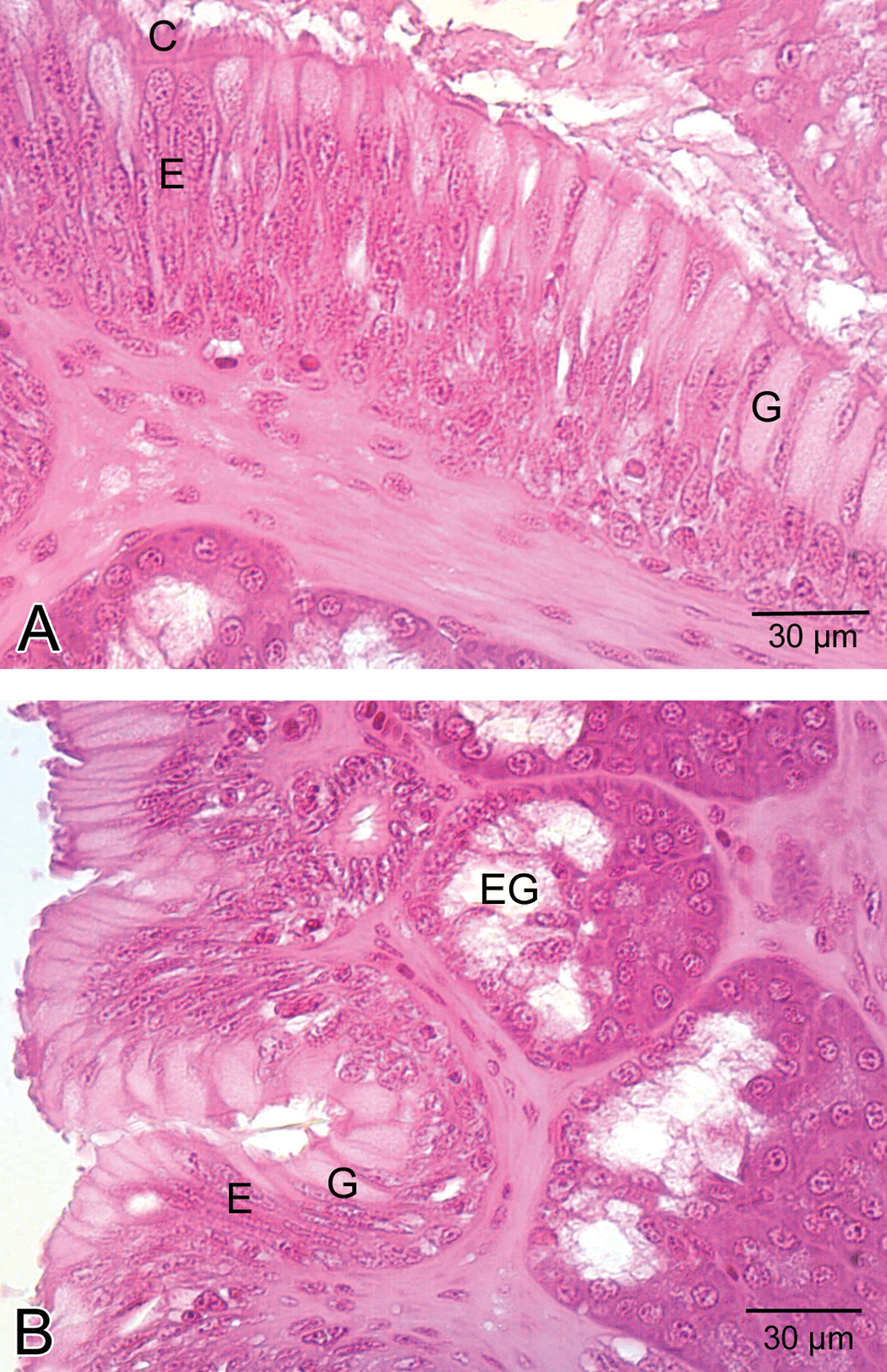
General view of esophagus in the control group. A. Note that normal appearance of columnar epithelial cells (E), cilia (C), goblet cells (G). B. esophageal glands (EG).
Histological view of the esophagus in the medium-dose group A. General view of esophagus. B. Detailed view of epithelial layer. Note the separation (S) of epithelial layer and the vacuolization (asterisk) in the epithelial layer.
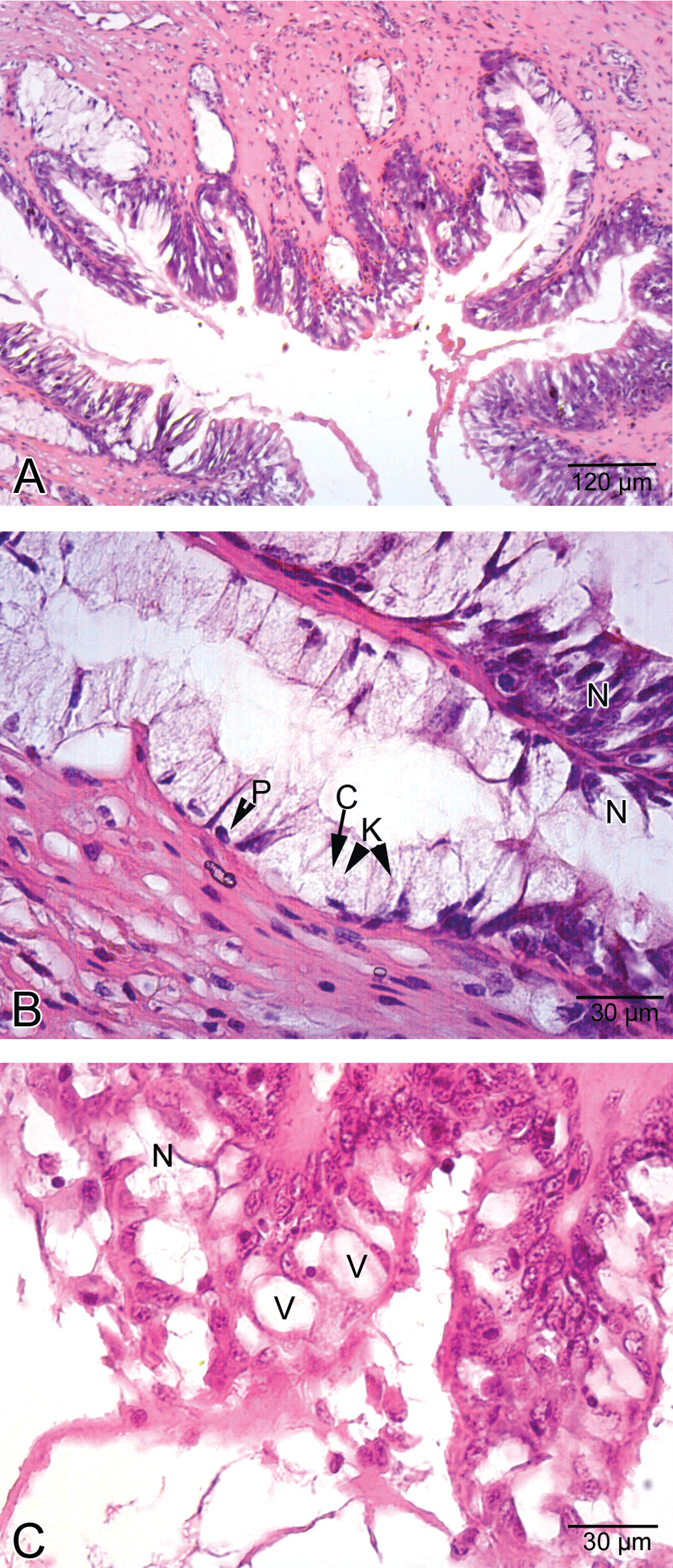
Histological view of esophagus in high-dose group. A. General view of esophagus. B. Esophageal cells showed cellular swelling (C) and necrosis (N) characterized by nuclear pyknosis (P) and karyolysis (K), C. Necrosis (N) and vacuolization (V) in epithelial cells.
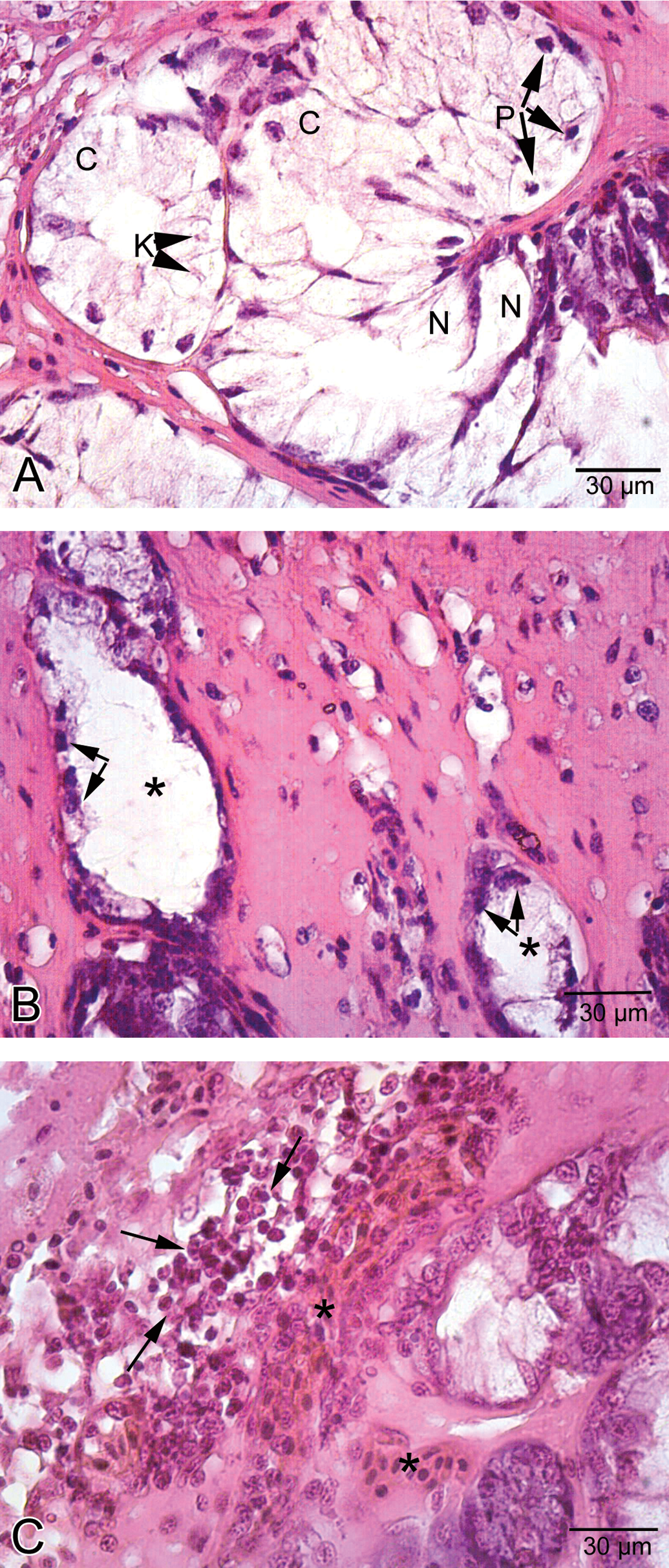
Histological view of esophagus in high-dose group. A. Cellular swelling (C), nuclear pyknosis, karyolysis (K), and necrosis (N) in esophageal glands. B. Atrophy (asterisk) in esophageal glands. Note the flattened cells (arrow) lining esophageal gland. C. Hemorrhage (asterisk) and infiltration of inflammatory cells (arrow).
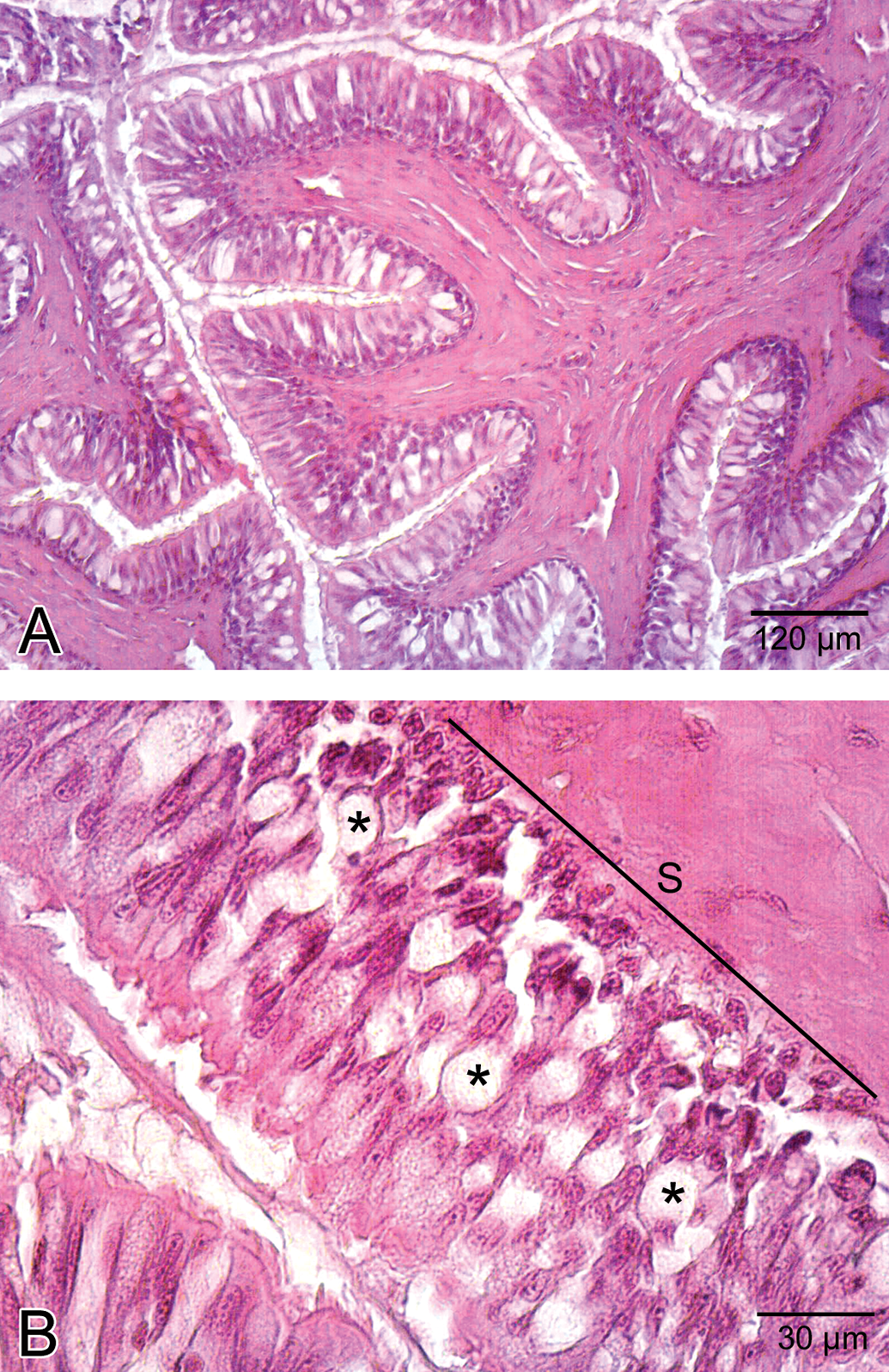
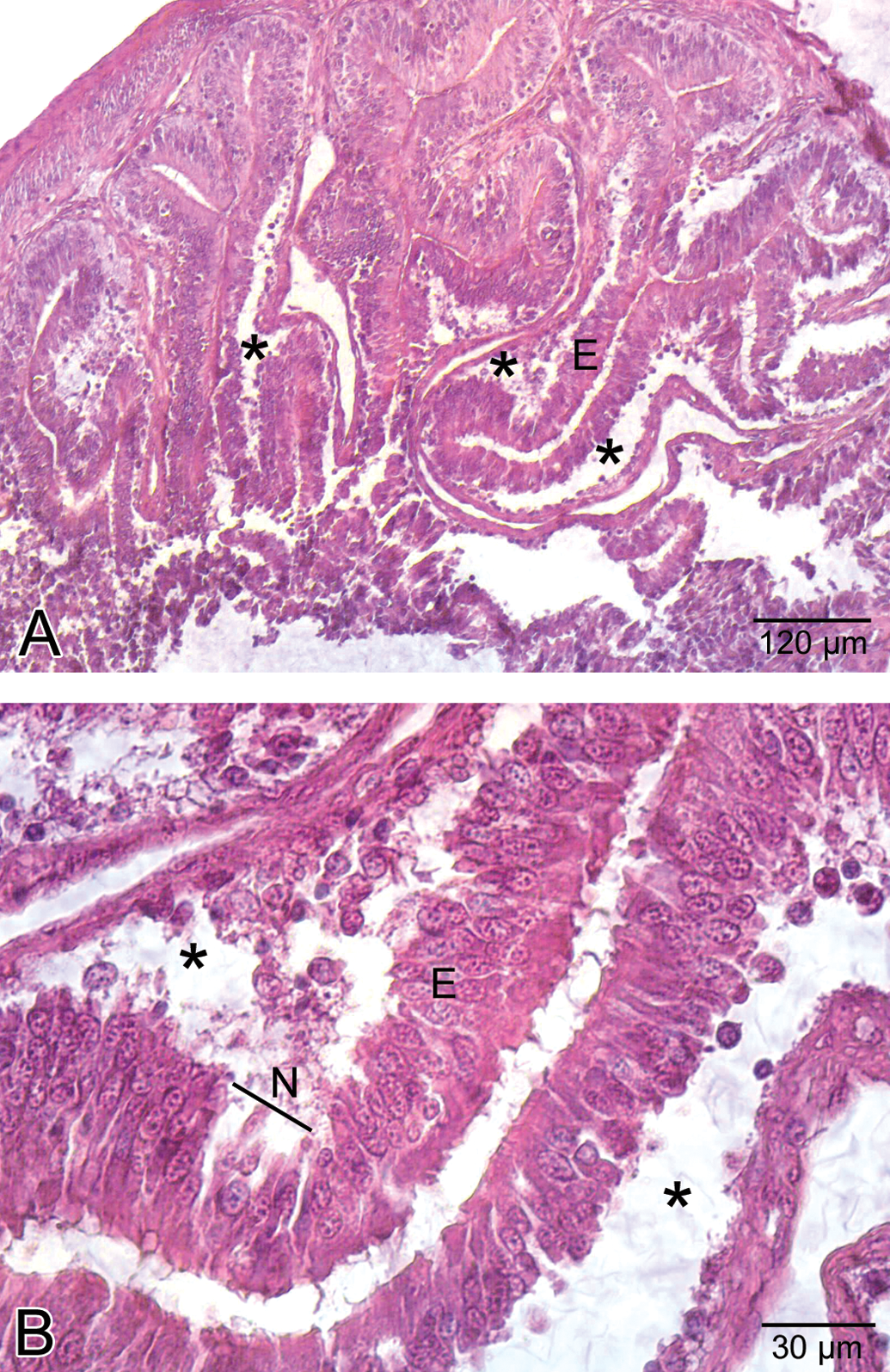
The stomach was lined with columnar epithelium (Figure 5A), which displayed numerous invaginations and gastric pits that gave rise to gastric glands (Figure 5B). In low-dose group, congested vessels were clearly seen within mucosa (Figure 6). In medium-dose group, a congested vessel (Figure 7A) and venous congestion (Figure 7B) were detected in submucosa layer. In addition, vacuolization and necrosis in gastric glandular epithelium and necrotic cells within the interstitial spaces were determined (Figure 7C). In high-dose group, separation of epithelial cell layer and edema among gastric glands were observed (Figure 8A). Cellular swelling in gastric glandular epithelium (Figure 8B) and prominence of hemorrhage in submucosa layer (Figure 8C) were also determined. Necrosis in glandular epithelium and epithelial cells lining stomach were prominent histopathologic changes; in addition, eroded epithelial layer was clearly seen (Figure 8D). Due to the edema, separations in tunica muscularis were obvious (Figure 8E).
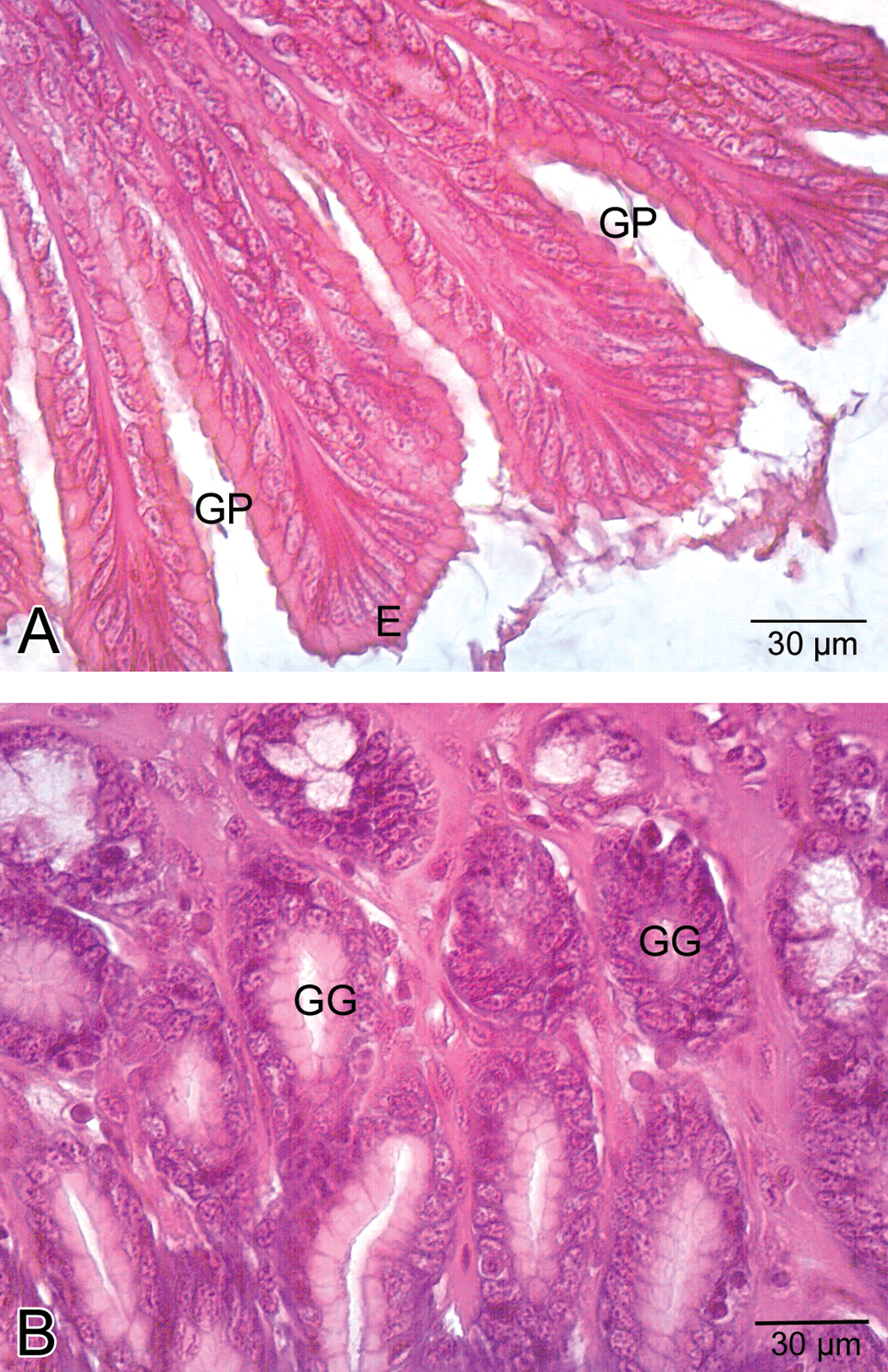
General view of stomach in control group. A. Epithelial cells (E). Note the gastric pits (GP). B. Gastric glands (GG) of stomach.
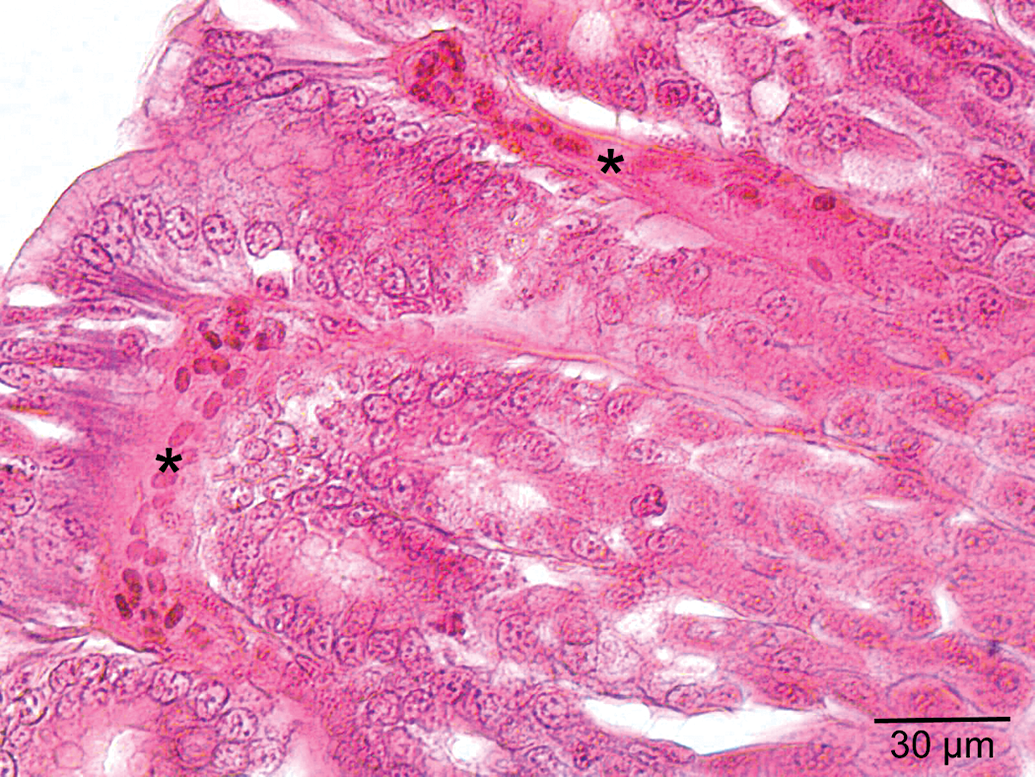
Congested vessels (asterisk) within mucosa in the low-dose group.
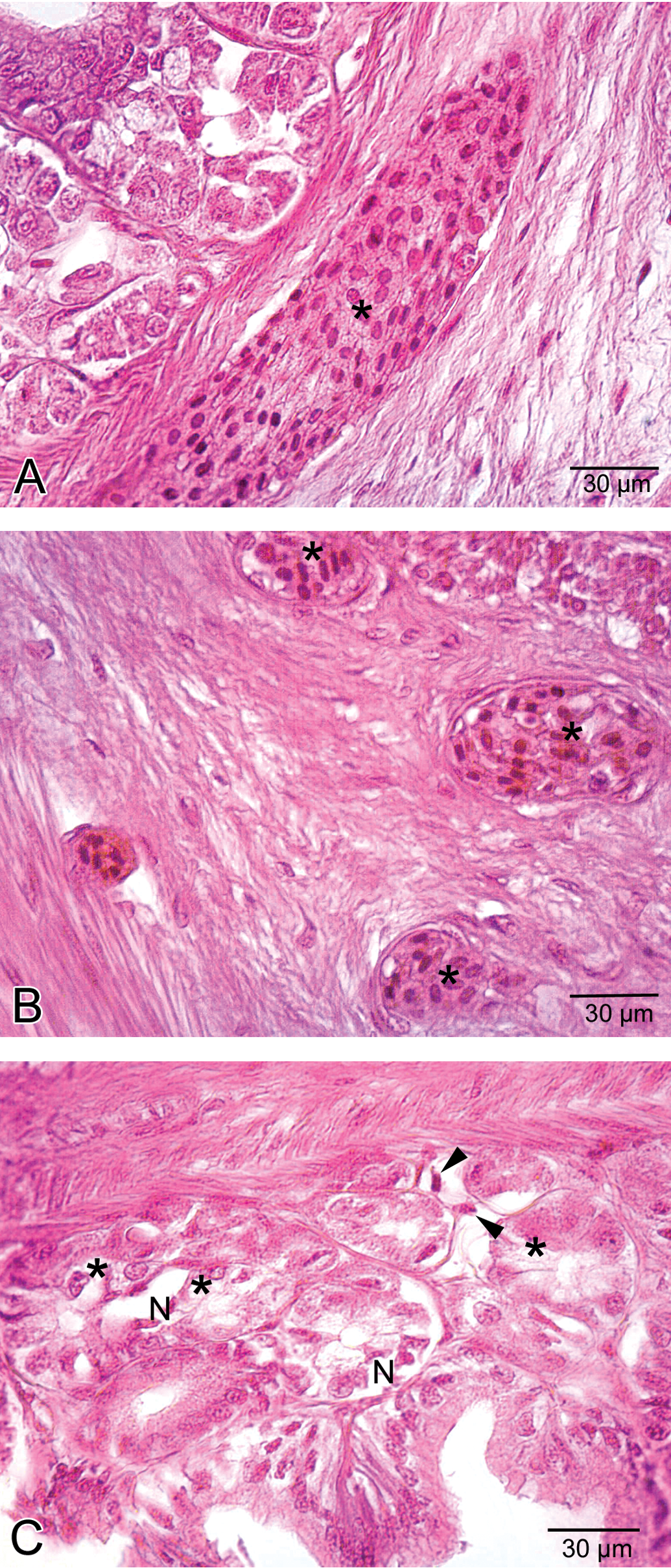
Histological view of the stomach in the medium-dose group. A. A congested vessel (asterisk) in submucosa layer. B. Venous congestion (asterisk) in submucosa layer. C. Vacuolization (asterisk) in gastric glands and necrosis (N) and necrotic cells (arrowhead) within the interstitial spaces.
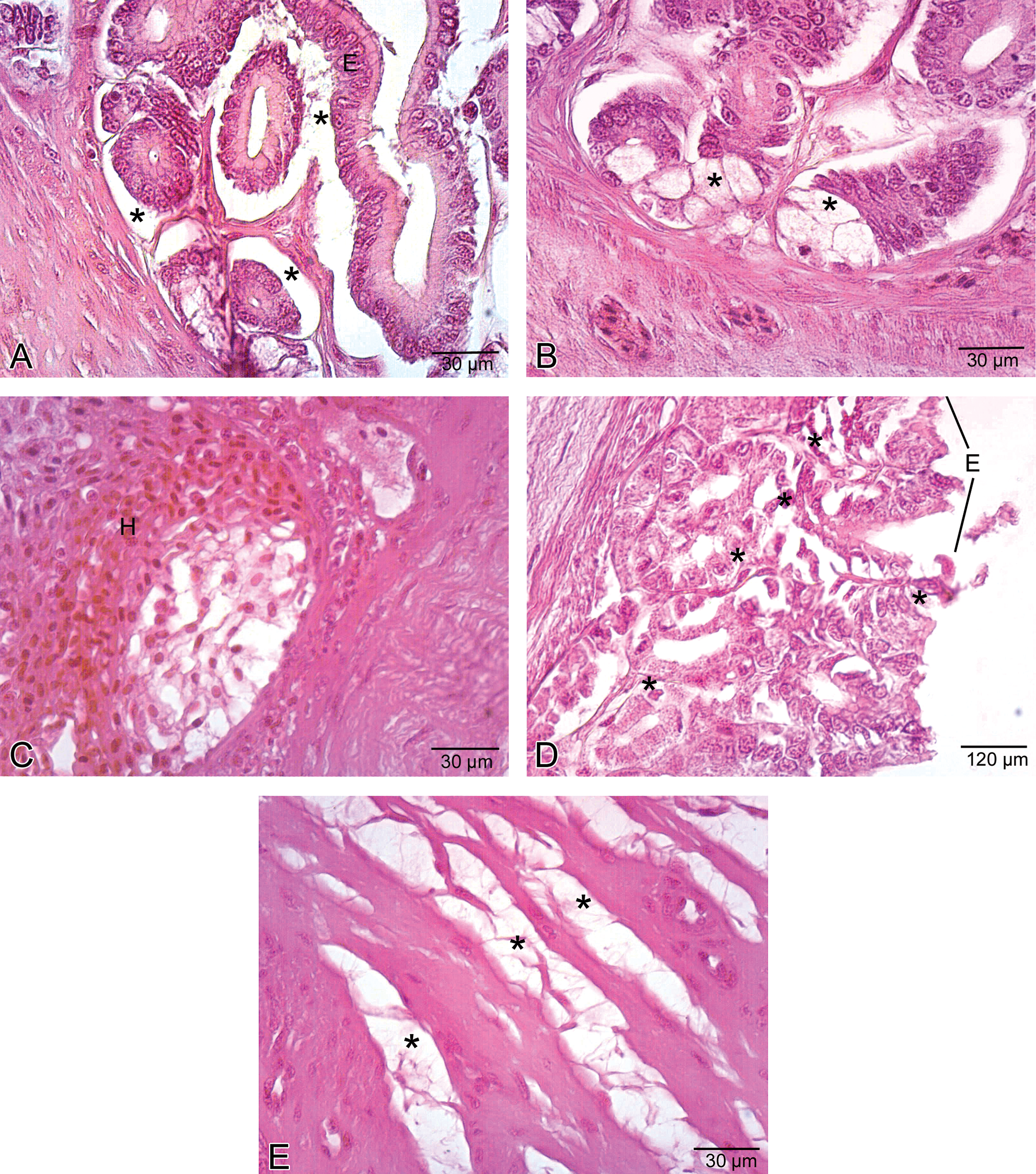
Histological view of the stomach in the high-dose group. A. Separation of epithelial layer (E) and due to the edema, wide spaces (asterisk) among gastric glands. B. Cellular swelling (asterisk) in gastric glandular epithelium. C. Hemorrhage (H) in submucosa layer. D. Necrosis (asterisk) in glandular epithelium and epithelial cells lining the stomach and eroded epithelial layer (E). E. Edema (asterisk) in tunica muscularis.
Epithelial layer of intestine contains two types of cells: absorptive epithelial cells and goblet cells (Figure 9). In low-dose group, there were no important defects compared to control. In medium-dose group, some degenerative changes in villi were seen (Figure 10A). Inflammation and necrotic cells were detected in villus structure (Figure 10B). In high-dose group, wide spaces as a result of the detachment of epithelial layer were observed. In addition, epithelial cell disorganization and necrosis were detected (Figure 11A and B).
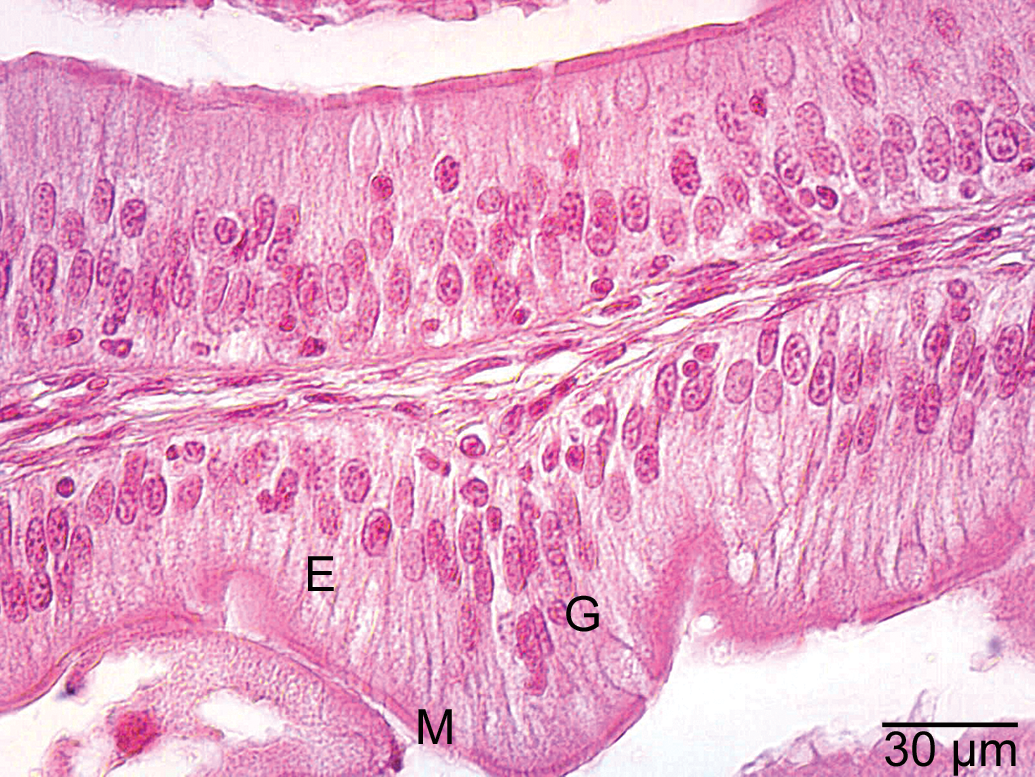
Epithelial cells (E), microvilli (M), and goblet cells (G) in the intestine of the control group.
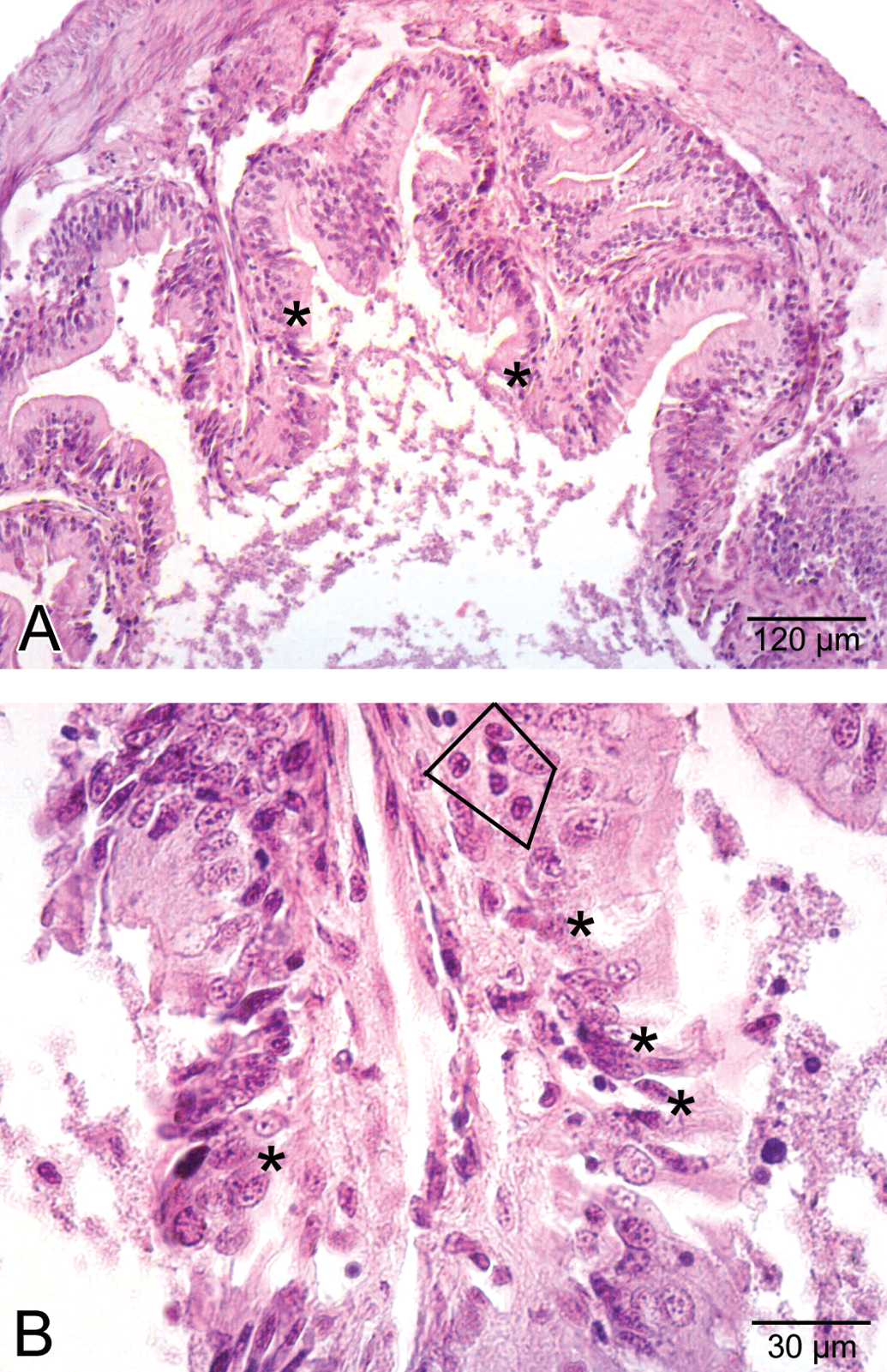
Histological view of the intestine in the medium-dose group. A. Some degenerative changes (asterisk) in villi. B. Inflammation (rectangulated) and necrotic cells (asterisk) in villi.
Histological view of the intestine in the high-dose group. A. Wide spaces (asterisk) due to the detachment of epithelial layer (E). B. Detailed view of wide spaces (asterisk), epithelial cell (E) disorganization, and necrosis (N).
Briefly, histopathologic changes observed in this study were also demonstrated in Table 1.
Histopathological findings of P. bedriagae after exposing to 0.05-, 0.1-, or 0.2-mg/g carbaryl for 96 hr.

Note: Lesions were scored based on their severity (0: none; 1: mild; 2: moderate; and 3: severe).
Discussion
The present study was conducted to examine histopathologic changes in the digestive tract of P. bedriagae, which was exposed to different carbaryl concentrations. According to the results of this study, histopathological changes were more prominent in medium- (0.1 mg/g) and high-dose (0.2 mg/g) groups than in the low-dose (0.05 mg/g) group.
Because there are a few study on the histopathologic effects of pesticides on amphibians, comparison to the current study is limited. Cakici and Akat (2012) studied the effects of carbaryl on the digestive tract of snake-eyed lizard, Ophisops elegans. The closest comparison can be made with this particular study. Authors stated that in the low-dose group, while a weak hemorrhage was observed in the connective tissue of esophagus, prominence of hemorrhages was found in the medium-dose group. Also, hemorrhage was noted in high-dose group in P. bedriagae. When compared to both species, epithelial layer was normal in appearance in low-dose groups. However, while the separation of muscle layer was determined in high-dose group of O. elegans, there were some histopathologic changes in epithelial cells such as vacuolization, cellular swelling, nuclear pyknosis, karyolysis, and necrosis in P. bedriagae at high dose. As different from than O. elegans, there are esophageal glands in P. bedriagae. In addition to glandular atrophy, cellular swelling, nuclear pyknosis, karyolysis, and necrosis were observed in esophageal glands in the high-dose group. Histopathologic examination of stomach showed several histopathologic changes in both O. elegans and P. bedriagae. In the low-dose group, while gastric gland cells had notable vacuolization in O. elegans, hemorrhage was detected in P. bedriagae. In the medium-dose groups, gastric gland atrophy was determined in O. elegans. However, vacuolization and epithelial cell necrosis were observed in gastric glands of P. bedriagae. When compared to P.bedriagae, O. elegans showed more severe lesions in the high-dose groups. Progressive gastric glandular atrophy was seen and finally these glands completely disappeared. In addition, epithelial cell necrosis was prominent. In P. bedriagae, there were necrosis in glandular epithelium and epithelial layer cells. Separations within tunica muscularis were also determined. Intestine of P. bedriagae showed hemorrhage in the low-dose group, whereas no histopathologic change was observed in O. elegans. In both medium and high doses, both species showed necrosis in epithelial cells of villi.
There are some studies related to different pesticide effects on the digestive tract of various animals. Sastry and Malik (1979) examined the effects of a sublethal concentration (0.32 mg/l) of dimecron for 20 days on the digestive system of a fresh water teleost fish, Channa punctatus. Authors found the degenerative changes in mucosa and gastric glands of the stomach in the medium and high doses. Banerjee and Bhattacharya (1995) examined the histopathologic changes in the intestine of C. punctatus induced by chronic nonlethal levels of Elsan (211 ppb), mercury (16.7 ppb), and ammonia (15.64 ppm) at 7-day intervals for 90 days. According to this study, destruction of the structure of villi was prominent in C. punctatus. Velmurugan et al. (2007) stated that infiltration of eosinophils into the lamina propria and atrophy of epithelial cells were observed in the intestine of Cirrhinus mrigala, which was exposed to sublethal concentrations (0.3 and 0.6 ppb) of lambda-cyhalothrin for 10 days. Özelmas and Akay (1995) examined the effects of malathion on the dwarf lizard, Lacerta parva. Authors stated villi degeneration, atrophy, and ruptured villi in the small intestine of the medium- and high-dose groups. Similar observations were observed in P. bedriagae. Also, there are some studies related to the effects of polluted water on the different organisms. Kumari and Kumar (1997) reported that intestinal tissue of Channa striatus and Heteropneustes fossilis inhabiting the polluted water showed degenerative alterations such as focal necrosis, proliferation, and desquamation of the superficial parts of villi. Önen, Gündüz, and Üçüncü (2011) studied the effects of water-soluble fractions of crude oil on the intestine of Pelvicachromis pulcher. Authors stated that the intestine of P. pulcher exhibited several histopathologic changes such as prominent ulceration, desquamation, lymphocyte infiltration, and necrosis after exposure to water-soluble fractions of crude oil.
As mentioned previously, pesticides have adverse effects on the digestive tract of many organisms. This is in accordance with the results of the study that revealed important histopathologic changes in digestive tract of Levantine frog, P. bedriagae after exposure to carbaryl. Due to the fact that there is no sufficient histopathologic study related to amphibians, it should be noted that the findings of this study could be of importance in terms of providing direction for other amphibian toxicological studies.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
