Abstract
The advancement of technology and the growth of international commerce underscore the need for global harmonization of regulatory safety requirements and their assessment pertaining to consumer products such as drugs, medical devices, and food. This need is particularly relevant when safety requirements involve time-intensive and costly animal safety studies. Here we present the current regulatory requirements in Europe, the United States, and Japan for flavoring substances (FSs) used in foods and point out significant differences relevant to the international standardization for safety assessments that in our opinion need to be addressed and overcome. The safety assessments that are carried out for FSs in various countries are influenced by divergent definitions of FS, by the information required and available for regulatory submission, and by different regulatory procedures, including the use of decision tree approaches. The European Food Safety Authority (EFSA), the Expert Panel of the U.S. Flavor and Extract Manufacturers Association (FEMA), and the Joint Food and Agriculture Organization (FAO)/World Health Organization (WHO) Expert Committee on Food Additives (JECFA) are making efforts to improve and harmonize the safety assessment of FSs. The application of in silico methods such as quantitative structure–activity relationships and read-across strategies relying on expert input are useful as a first-step screening of the assessment. Application of the Threshold of Toxicological Concern (TTC) approach permits conclusions that are compatible with the risk assessment approaches currently used by international advisory committees.
The Japanese Regulatory Authority, on the other hand, does not yet consider in silico methods but still requires in vivo and in vitro genotoxicity test data as well as repeat-dose 90-day toxicity data in at least one species, to be submitted as the first step in the safety assessment of FSs. With this article, we echo requests that have been made for xenobiotics by the pharmaceutical industry worldwide, extending them to food-related products, especially FSs. We encourage regulatory agencies to adopt globally harmonized safety assessment procedures, regulatory guidelines, and review practices for FSs to foster global trade and to reduce costs and laboratory animal use.
Keywords
Introduction
Currently, consumers are increasingly interested in ethnic food products, and this has resulted in greater international exchange of ethnic and country-specific foods and beverages that were formerly limited in distribution. Each of these foods contain various ingredients, including flavoring substances (FSs), which are essential for foods and beverages since they impart, enhance, and/or modify flavor and/or aroma. As with all added food ingredients, FSs require a safety assessment by regulatory agencies. At present, about 3,250 FSs are in use in Japan; 2,500 in Europe; and 2,300 in the United States. About 1,800 FSs are commonly used in all three of these geographic regions.
To bring the need for international harmonization of the safety assessment for FSs specifically pertaining to the need of conducting repeat-dose animal toxicity studies to the attention of a broader audience, we compared the current regulatory situation for FS in Europe, the United States, and Japan. The safety assessment of FSs in the European Union (EU) is conducted by the EFSA and in the United States by the Expert Panel of the FEMA. In Japan, FSs come under the purview of the Japanese Food Safety Commission (JFSC) as part of the Cabinet Office of the Japanese Government since 2003.
Consequently, the regulatory definition of FSs differs among countries/regions. In Japan, FSs are considered food additives that are added during the manufacturing process to provide or enhance flavor (Japanese Consumer Affairs Agency 2010). In the Codex Alimentarius and in Europe, FSs are distinct from food additives and food enzymes (Joint FAO/WHO Food Standards Program 2008; Regulation [EC] 2008). The independent Expert Panel supported by FEMA has been performing the safety assessment of FSs by utilizing the U.S.-specific Generally Recognized as Safe (GRAS) process for over 50 years. In the United States, FEMA member companies initiate the review of flavor ingredients, through assessment by the FEMA Expert Panel, resulting in a GRAS status determination for the use of the substance as a flavor ingredient. The outcomes of GRAS assessments are then published in the public domain to make the information available to the U.S. Food and Drug Administration (FDA), the food and flavor industries, and the consumer. Through these GRAS evaluations, more than 2,300 FSs have been determined “safe” under conditions of use as an FS (Taylor and Adams 2012). Assessments of FSs by the FEMA Expert Panel as substances that are GRAS under conditions of intended use are generally accepted in more than 70 countries outside the United States, either explicitly within a regulation or by other official or informal notification.
In this article, we describe similarities and differences con-cerning regulatory requirements for the safety assessment of FSs in different countries and propose a direction for international harmonization to foster global trade and reduce unnecessary additional testing.
Categorical Difference of FSs
Common characteristics of FSs are as follows. Most are naturally occurring, many are potent and self-limiting; thus, they are used in food and beverages at very low levels (parts per million to parts per trillion). Moreover, most FSs have relatively simple chemical structures made up of carbon, hydrogen, oxygen, nitrogen, and sulfur.
Within JECFA, the United States (GRAS) and the EU, the approach to safety assessment of FSs is in many ways different from the one for food additive safety evaluations, while in Japan the safety assessment of FSs is handled in the same manner as for other food additives (JFSC 2010). We believe there is an urgent need for the international harmonization of approaches toward the safety assessment of FSs. A critical first step in this direction is the standardization of the documentation and data required for regulatory safety assessments. However, at present, there are no globally accepted documentation standards.
In the United States, Europe, and particularly in considerations by JECFA, in silico and expert approaches that utilize structure–activity relationships, such as quantitative structure–activity relationship (QSAR), read-across procedures, and the Threshold of Toxicological Concern (TTC), are used in the regulatory evaluation of FSs. The TTC approach establishes a human exposure threshold value for any chemical based on its anticipated toxic potential. If the intake of that substance is below the TTC, it is considered to present no appreciable risk to human health. Fundamentally, this approach proposes that a safe intake level can be identified for many chemicals, including those with unknown toxicity, based upon their chemical structures. Recently, the TTC approach was evaluated by international advisory committees, and it was concluded that this method is consistent with and valuable for conventional risk assessment approaches (Dewhurst and Renwick 2013). QSAR identifies quantitative or qualitative relationships between biological activity (i.e., toxicity), which may be categorical or quantitative, and one or more molecular that are used to predict that activity. The qualitative read-across approach relies on the expert judgment that substances that are chemically similar (i.e., common structure) to other chemicals with known metabolism and target-organ toxicity will likely behave similarly also, when in the body. A quantitative read-across approach utilizes a mathematical analysis of specific values from more than 2 substances with similar chemical structures, to categorize the safety of a chemically related FS (Cronin et al. 2003; Bassan et al. 2011).
Although the Japanese regulatory agency has recently shown interest in QSAR and reported the QSAR combination approach for mutagenicity prediction of FSs (Ono et al. 2012), it currently still requires in vivo and in vitro genotoxicity data as well as repeat-dose 90-day toxicity studies in at least one species as the first step in the safety assessment of FSs. Notably, in Japan, there is no regulatory provision for the use of in silico data as a first prioritization step and, as a result, 2 stages of genotoxicity testing are automatically required. In the first stage, in vitro tests using bacterial cells and mammalian cells are conducted. If the results are ambiguous or clearly positive, a follow-up in vivo micronucleus test is carried out in the second stage to confirm or refute that the in vitro results are relevant in vivo.
Since 2003, the Japanese Ministry of Health, Labour, and Welfare (JMHLW) has requested the JFSC to conduct a safety assessment on 54 FSs. For 45 of these 54 FSs, published genotoxicity data were not available; therefore, JMHLW performed these genotoxicity tests to provide the respective data to JFSC for the requested safety assessment. Similarly, JMHLW conducted 90-day rodent studies on 35 of the 54 FSs that did not have published data from repeat-dose toxicity studies, using doses based on recently available assumed daily intake in humans and preliminary information on no-observed adverse effect levels (NOAELs; Someya 2012). In the absence of displayed genotoxic potential, and since FSs in food and beverages are present at extremely low levels, the safety of FSs was approved when the Margin of Safety based on the NOAEL value from the 90-day study and the estimated daily intake was more than 1,000-fold (Someya 2012).
Difference in Decision Tree/Procedures
A variety of decision tree schemes are utilized by risk assessors for safety assessments of FSs. The stepwise procedure employed by JECFA is shown in Figure 1. In the first step, chemical structures of FSs are classified using the Cramer/Ford Hall Decision Tree into Structural Class I, II, or III (Cramer, Ford, and Hall 1978). Class I is an FS with a simple chemical structure that is efficiently metabolized to innocuous products with anticipated low oral toxicity. Class II contains FSs that are less innocuous than Class I, but without structural features indicative of toxic potential. A Class III substance is one with no presumption of safety and may have reactive chemical structures suggestive of toxicity. The corresponding TTC levels are 1,800, 540, and 90 μg per day for Classes I, II, and III, respectively. The JECFA assessment proceeds from the initial categorization to steps A4, B4, A5, and B5, and ultimately reaches a conclusion on the safety of the FS. In addition to consideration of TTC levels, depending upon how some substances move through the assessment procedure, there is a consideration of whether the intake of the substance would be anticipated to exceed 1.5 μg/day (shown in step B5), which is a recognized threshold of regulation that is utilized by the FDA (1995) for food contact materials.
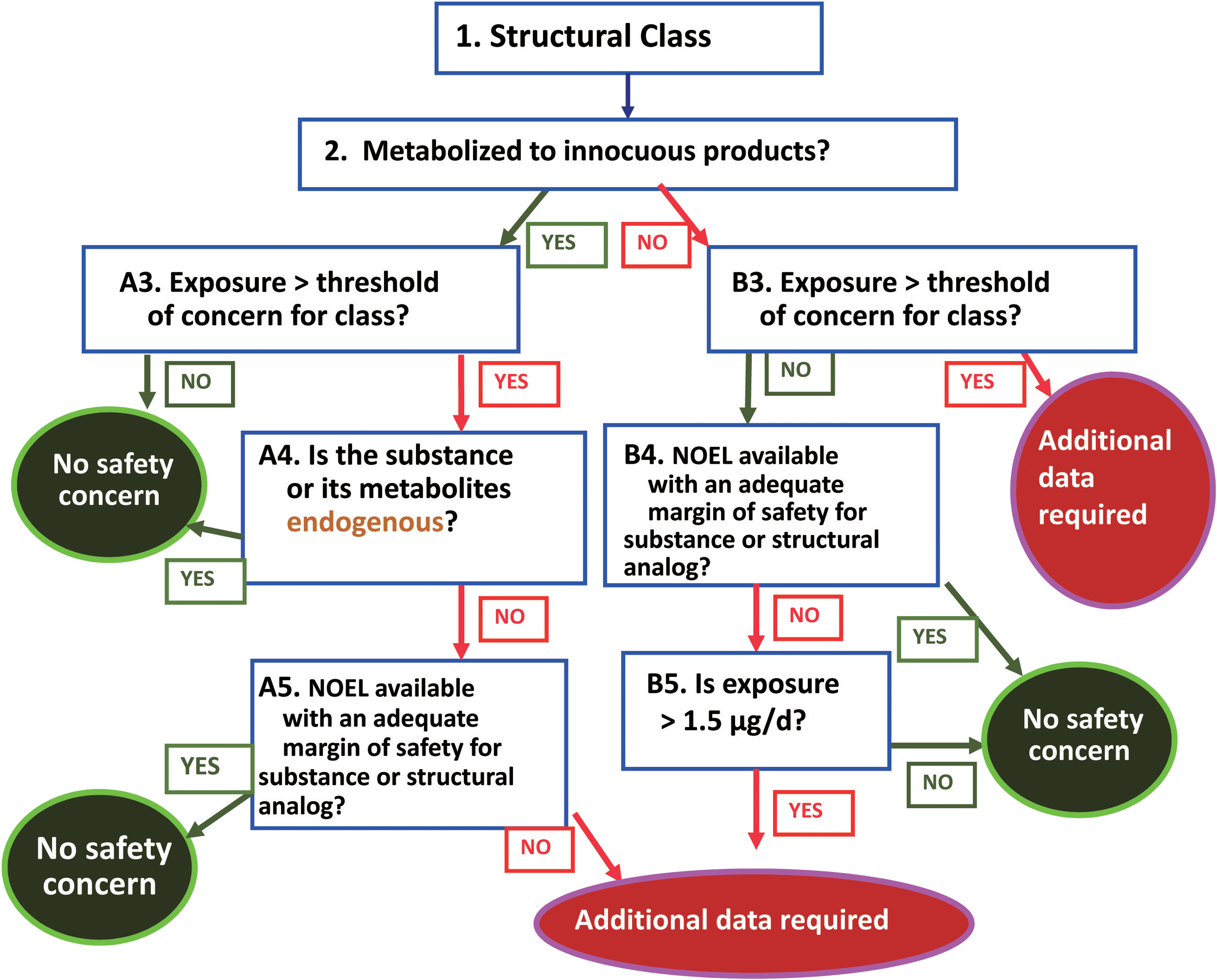
JECFA decision tree for flavoring agents. JECFA = Joint Food and Agriculture Organization (FAO)/World Health Organization (WHO) Expert Committee on Food Additives.
The assessment procedure used by the FEMA Expert Panel in the United States and EFSA in the EU follows approaches similar to those described for JECFA above. FSs with uses determined to be GRAS by the FEMA Expert Panel may be distributed in the United States under the conditions of the intended use. Purity, physicochemical characteristics, and production methods are also considered during the assessment procedure. In contrast, the assessment of FSs in Japan (Figure 2) is dependent upon the positive or negative results in a preliminary genotoxicity hazard assessment step, and upon the NOAEL from a 90-day repeated-dose animal toxicity study. A threshold of regulation for intake greater than 1.5 μg/day is not adopted for substances of unknown toxicity in Japan.
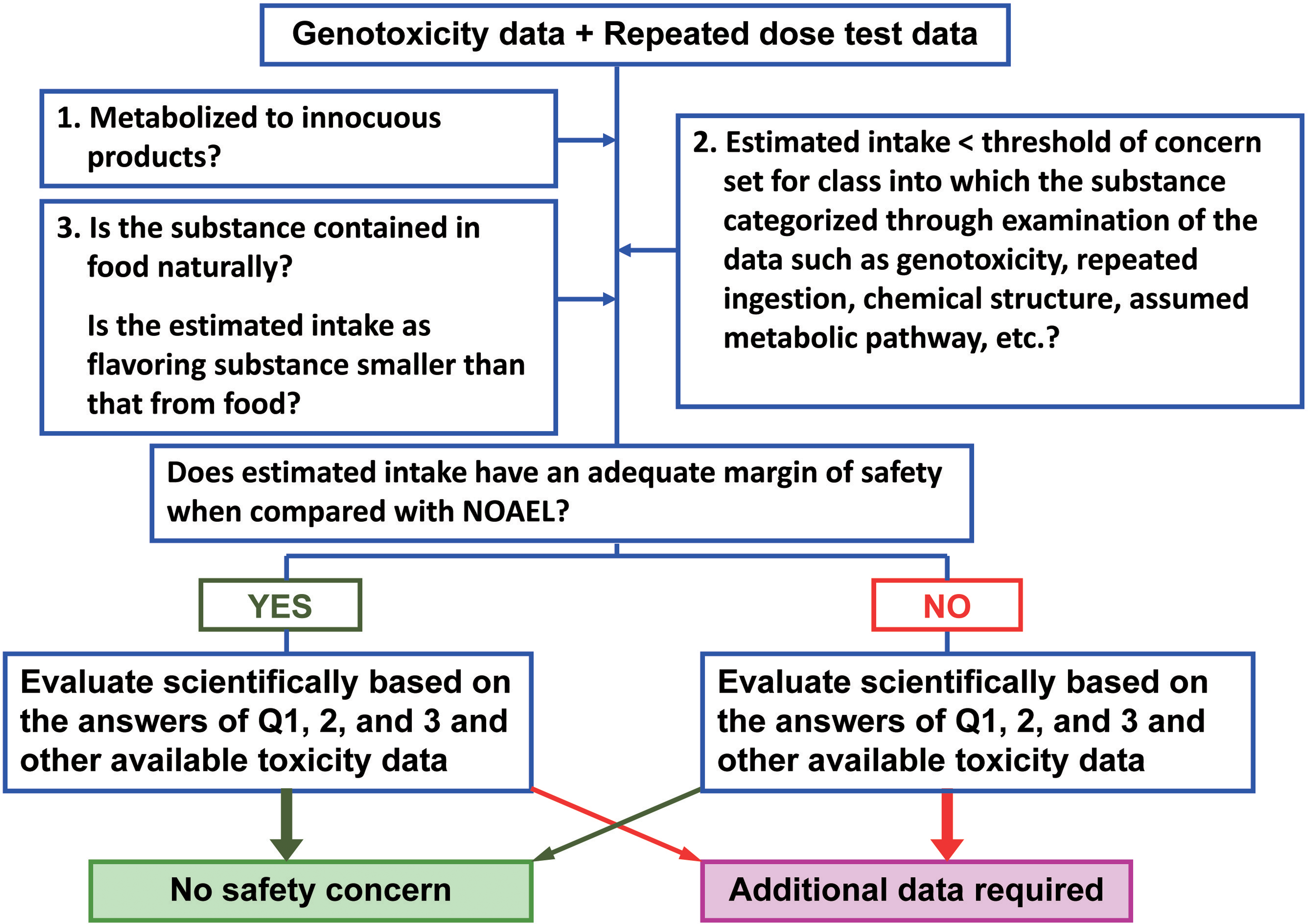
Flow chart of safety evaluation method applied in Japan for globally used flavoring substances.
Searching for Direction toward International Harmonization
In our opinion, there are several factors worth considering in charting a direction for international harmonization of regulatory requirements for the assessment of FSs. Harmonization is expected to provide a global list of safe FSs that will lead to a greater number of globally consistent approvals with reduction of cost, review times, and a need for animal testing, as well as increased consumer confidence in the global food supply.
The assessment approach used by JECFA is widely accepted and similar approaches are already in use in the United States and EU.
The FEMA Expert Panel and EFSA are constantly moving forward to further evolve the present procedures for the safety assessment of FSs.
The JFSC completed safety evaluation for 50 of the 54 FSs, and their results are consistent with those done by JECFA, the FEMA Expert Panel, and EFSA (Someya 2012).
Therefore, considering these international circumstances, it is our opinion that attempts should be made by the Japanese Regulatory Authorities aiming to solve the disparity by harmonizing global approval requirements through adopting international, scientifically rigorous and time-tested procedures.
Footnotes
*This is an opinion article submitted to the Society of Toxicologic Pathology (STP) Regulatory Forum and does not constitute an official position of the STP or Toxicologic Pathology. The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of respective agencies and organizations. The Regulatory Forum is designed to stimulate broad discussion of topics relevant to regulatory issues in toxicologic pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to
Acknowledgments
The authors greatly appreciate Dr. Robert R. Maronpot, Dr. Sean V. Taylor, and Mr. Toshinao Baba for their assistance in the preparation of this article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported in part by a grant from the Japan Food Chemical Research Foundation.
