Abstract
Inorganic arsenic (arsenite and arsenate) at high exposures is a known human carcinogen, inducing tumors of the urinary bladder, skin, and lungs. In two experiments, we examined the urothelial proliferative effects of treatment with 173 ppm sodium arsenite (100 ppm arsenic) in the drinking water for 6 and 24 hr, and 3, 7, and 14 days in female F344 rats and 43.3 ppm sodium arsenite (25 ppm arsenic) in female C57BL/6 wild-type and arsenic (+3 oxidation state) methyltransferase knockout (As3mt KO) mice that are unable to methylate arsenicals. In the rat and both mouse genotypes, scanning electron microscopy showed cytotoxic urothelial changes as early as 6 hr after the start of arsenic exposure. The severity of AsIII-induced cytotoxic urothelial changes increased over time in the rat and in the As3mt KO mouse. Light microscopy showed an increase in urothelial hyperplasia in the rat. No significant increases in bromodeoxyuridine-labeling index were observed. The data support the hypothesis that the sequence of events in the mode of action for urothelial effects of orally administered inorganic arsenic in the rat and mouse involves superficial cytotoxicity with consequent regenerative increased cell proliferation similar to the findings associated with the administration of dimethylarsinic acid (DMAV) in rats.
Introduction
Inorganic arsenic (arsenite and arsenate) is a known human carcinogen at high exposures, associated with the induction of cancer of the urinary bladder, skin, and lungs, and possibly other organs (National Research Council [NRC] 1999, 2001). Exposure to inorganic arsenic usually occurs through the drinking water and food, but exposure can also occur via various pharmaceutical products, and inhalation exposure can occur in some occupational settings, such as mining. Of the tumors that are related to arsenic exposure, the urinary bladder has received the most attention, primarily because of the availability of animal models and extensive epidemiologic investigations. Hyperplasia of the urothelium, a preneoplastic change, was initially observed in mice administered sodium arsenite by Simeonova et al. (2000). Subsequently, hyperplasia of the urothelium in rats and in mice administered either arsenite or arsenate in the drinking water or in the diet has been observed (Suzuki, Arnold, Ohnishi, et al. 2008). Considerable evidence has accumulated during the past decade strongly supporting the hypothesis that the trivalent forms of arsenic are responsible for its toxic and carcinogenic effects. We have hypothesized that the key events in the urothelium involve the induction of cytotoxicity with consequent cell death and regenerative cell proliferation resulting in hyperplasia. The goal of the study reported in this article was to investigate the specific sequence of events in the urothelium to confirm this hypothesis.
Dimethylarsinic acid (DMAV) induced urinary bladder tumors in rats but did not induce an increased incidence of any tumors in mice (Cohen et al. 2006). Extensive research demonstrated that the sequence of events leading to these tumors involved reduction of DMAV to dimethylarsinous acid (DMAIII) leading to urothelial cytotoxicity and to cell death with consequent regenerative hyperplasia and the ultimate formation of tumors. The specific effect of DMAIII on the urothelium likely involves the interaction of the trivalent arsenic with sulfhydryl groups of critical proteins, similar to other effects of arsenicals.
For inorganic arsenic, it has been proposed that the trivalent methylated forms of arsenic are primarily responsible for the toxic and carcinogenic effects of the administered inorganic arsenic (NRC 1999, 2001). Inorganic arsenic undergoes a sequence of reductions and methylations all catalyzed by the enzyme arsenic (+3 oxidation state) methyltransferase (As3mt), proceeding through the addition of methyl groups to the arsenic yielding monomethyl, dimethyl, and trimethyl forms of arsenic. The first two methylations occur readily in rats and mice as well as in humans, but the third methyl group is rarely found in humans, primarily because DMA appears to be a very poor substrate for As3mt. Although in vitro, the trivalent methylated forms of arsenic are more cytotoxic than inorganic arsenic, the difference is only 2- to 5-fold. Inorganic arsenic, particularly arsenite (AsIII), is also highly cytotoxic (Suzuki, Arnold, Ohnishi, et al. 2008). In arsenic (+3 oxidation state) methyltransferase knockout (As3mt KO) mice administered arsenate or arsenite, urothelial changes are also produced, although the methylated forms of arsenic are present at negligible levels (Yokohira et al. 2010, 2011). In these mice, the urine contains very high concentrations of arsenite, well above concentrations that are expected to produce cytotoxicity.
In the study described in this article, the sequence of events leading to hyperplasia of the urothelium was investigated in rats and in mice, as well as in As3mt KO mice to evaluate whether the cytotoxicity precedes urothelial cell proliferation or if there is a mitogenic response with consequent cytotoxicity.
Methods and Materials
Chemicals
Sodium arsenite (NaAsIII; AsNaO2, anhydrous), 94% pure, was purchased from Sigma (St. Louis, MO) and was stored desiccated at room temperature. It was used as received without additional analysis.
Animals
Female rats and mice were used since they have the same or greater urothelial response to arsenicals compared to males of both species (Suzuki, Arnold, Ohnishi, et al. 2008). Female F344 rats and female C57BL/6 wild-type (WT) mice were purchased from Charles River Breeding Laboratories (Kingston, NY). Female As3mt KO mice were obtained from the F4 generation of a breeding colony at the University of Nebraska Medical Center (Omaha, NE). The colony originated from 4 female and 2 male mice homozygous for the disrupted As3mt gene (Drobna et al. 2009) obtained from Dr. David Thomas (U.S. Environmental Protection Agency, Research Triangle Park, NC). To generate the As3mt KO homozygous mice, exons 3 through 5 were deleted by homologous recombination. The altered gene was introduced and maintained in strain 129S6 mice before being introduced into strain C57BL/6 mice by marker-assisted accelerated backcrossing performed by Charles River Laboratories (Wilmington, MA). The mice were fertile, so brother/sister matings were used to maintain the homozygous As3mt KO genotype.
Study Design
All animals were housed in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The study protocol was approved by the University of Nebraska Medical Center Institutional Animal Care and Use Committee, and the level of care provided to the animals met or exceeded the basic requirements outlined in the Guide for the Care and Use of Laboratory Animals (NIH publication #86–23, revised 2011). The animals were group housed (4–5/cage) in polycarbonate cages with dry corncob bedding. The cages housing the mice were equipped with microisolator tops. Nestlets™ (Ancare, Bellmore, NY) were added to the mouse cages, and Nylabones® (Nylabone Products, Neptune, NJ) were added to the rat cages for environmental enrichment. The animal rooms were on a 12-hr light/dark cycle at a targeted temperature of 22 ± 2°C and humidity of 50 ± 5%. All animals were fed pelleted Certified Purina 5002 diet (Dyets Inc., Bethlehem, PA) with arsenic < 0.2 ppm. Food and drinking water were available ad libitum at all times during the study. Rats were approximately 8 weeks old and mice were approximately 9 weeks old at the start of treatment. Following quarantine for at least 7 days, all animals were randomized into treatment groups by species, strain, and weight (Martin et al. 1984) and treated for 6 or 24 hr, or 3, 7, or 14 days. Via drinking water, rats were treated with 173 ppm NaAsIII (100 ppm AsIII) and mice were treated with 43.3 ppm NaAsIII (25 ppm AsIII). NaAsIII was mixed into the drinking water using the tap water in the animal rooms (arsenic levels < 5 μg/L). Based on stability studies done for Dr. Dori Germolec’s laboratory at National Institute of Environmental Health Sciences (Reed, Fuciarelli, and Graves 2001), the NaAsIII-containing water was prepared fresh twice per week. The same schedule was used to change the drinking water on the control cages. Water and food consumptions were measured over a 7-day period for animals treated for 7 or 14 days. Water and food consumptions were measured over the entire treatment periods for animals treated for less than 7 days. Body weights (BW) of all animals were measured on study day 0, on the last day of each 7-day consumption period, and on the day of sacrifice. Detailed clinical observations of each animal were conducted on day 0 and just prior to sacrifice.
One hour prior to sacrifice, all animals were injected with bromodeoxyuridine (BrdU; 100 mg/kg; Sigma, St. Louis, MO). At necropsy, the urinary bladder was inflated in situ with Bouin’s fixative, and after removal, the bladders were placed in Bouin’s fixative (Cohen et al. 2007). A small section of intestine was removed and placed in Bouin’s fixative with the bladder. Kidneys were removed from all mice and fixed in formalin. Following fixation, the Bouin’s-fixed bladders and intestinal tissue were rinsed in 70% ethanol, the bladders were weighed, and all tissues were observed macroscopically. The entire surface of one-half of the bladder was examined by scanning electron microscopy (SEM) and classified into one of the five categories as previously described (Cohen et al. 1990, 2007). Briefly, class 1 bladders have polygonal superficial urothelial cells; class 2 bladders have occasional small foci of superficial urothelial necrosis; class 3 bladders have numerous small foci of superficial urothelial necrosis; class 4 bladders have extensive superficial urothelial necrosis, especially in the dome of the bladder; and class 5 bladders have necrosis and piling up (hyperplasia) of rounded urothelial cells. Normal rodent urinary bladders are usually class 1 or 2, or occasionally class 3. The other half of the bladder was cut longitudinally into strips and with a slice of intestinal tissue was embedded in paraffin, stained with hematoxylin and eosin (H&E), and examined histopathologically (Cohen 1983; Cohen et al. 1990, 2007). A diagnosis of mild simple hyperplasia was made when there were 4 to 5 cell layers in the bladder epithelium and moderate hyperplasia when 6 to 8 layers were present. Kidneys were also embedded in paraffin and H&E-stained sections were examined histopathologically.
Unstained bladder and intestinal tissue slides were processed for immunohistochemical detection of BrdU (Cohen et al. 2007). Intestinal tissue served as a positive staining control and to verify the validity of the injection. Anti-BrdU (Millipore Corporation, Temecula, CA) was used at a dilution of 1:200. The BrdU-labeled cells per bladder in at least 3,000 urothelial cells were counted to determine a labeling index.
Statistics
The group means for BWs, food and water consumptions, labeling index, and tissue weights were evaluated using analysis of variance followed by the Duncan’s multiple range test for groupwise comparisons. Histopathology was compared using the 2-tailed, Fisher’s exact test. SEM data were analyzed using 1-way nonparametric procedures followed by a chi-square test. P values less than 0.05 were considered significant. These statistical analyses were performed using SAS for Windows (Version 9.1).
Results
BWs, Food and Water Consumptions, and Tissue Weights
Although the ages of the C57BL/6 WT mice and the As3mt KO mice were similar, BWs of the As3mt KO mice on study day 0 (17.8 ± 0.1 g) were significantly lower than the BWs in the WT mice (22.2 ± 2.3). Therefore, data for BWs, food and water consumptions, and tissue weights were not compared statistically between the two genotypes.
The mean BW of the AsIII-treated rats on study day 14 (Supplemental Table S1) and of the As3mt KO mice on study days 7 and 14 (Supplemental Table S2) were significantly less than the BWs of the respective control groups. The BWs of the AsIII-treated WT mice were similar to those of the respective controls throughout the study (Supplemental Table S2). The food consumption of the AsII-treated rats measured at 24 hr as g/rat/day was significantly increased compared to that of the rat control group, but it was similar to that of the control group at 3 and 7 days (Supplemental Table S1). The food consumption of the As3mt KO mice measured as g/rat/day at 6 hr and 3 days was significantly decreased compared to the respective controls (Supplemental Table S2). The food consumption of the WT mice was similar to that of the respective control group. The water consumption of the AsIII-treated rats measured as g/rat/day or g/kg BW/day was similar to that of the control group at 24 hr and 3 days, but slightly lower at 7 days. The decrease was not statistically significant (Supplemental Table S1). Water consumption was not statistically analyzed in the mice due to numerous water bottle leaks. Absolute and relative weights of bladder tissue from AsIII-treated rats (Supplemental Table S1) and those of As3mt KO and WT mice (Supplemental Table S2) were similar to the respective control groups at all time points. The relative kidney weights of the AsIII-treated As3mt KO and WT mice were significantly increased compared to the respective control groups from day 3 and continuing through days 7 and 14 (Supplemental Table S2).
Histopathological Examination of Rat and Mouse Urinary Bladder and Mouse Kidneys
Simple hyperplasia of the urinary bladder epithelium was detected in the AsIII-treated rats in one bladder at 24 hr and at 3 days, in two bladders at 7 days, and in all bladders at 14 days (Table 1 and Figure 1). In contrast, in the AsIII-treated As3mt KO mice, there was 1 bladder with simple hyperplasia on day 3, and 2 on day 7, but no hyperplasia of the urinary bladder epithelium was detected at 6 or 24 hr or on day 14 (Table 2 and Figure 2). In the WT mice, one mouse with urothelial hyperplasia was detected on days 7 and 14 (Table 2 and Figure 2). There were no treatment-related changes observed in the kidneys of any As3mt KO or WT mice (data not shown).
Effects of treatment with 100 ppm AsIII in the drinking water on the bladder epithelium of female rats.
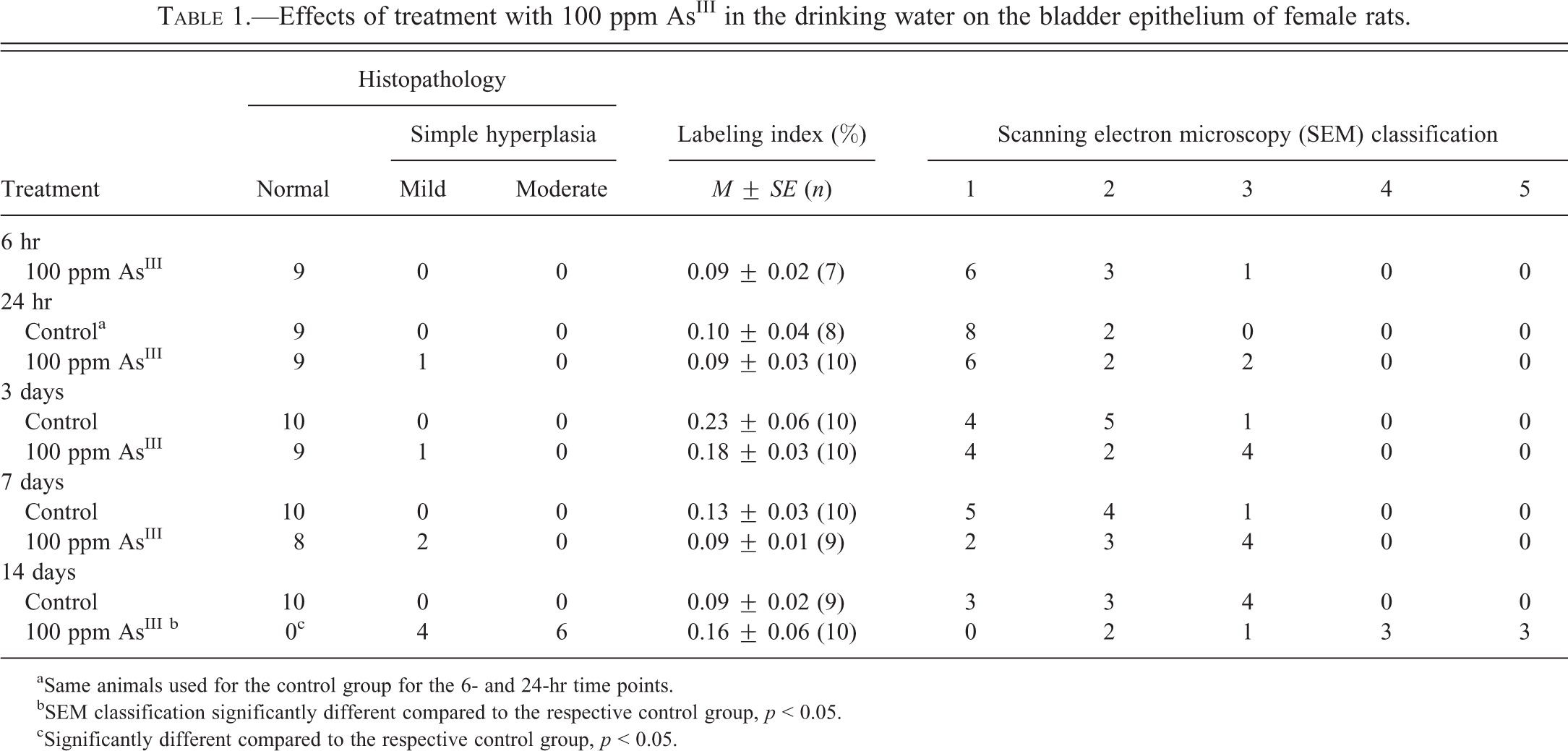
aSame animals used for the control group for the 6- and 24-hr time points.
bSEM classification significantly different compared to the respective control group, p < 0.05.
cSignificantly different compared to the respective control group, p < 0.05.
Effects of treatment with 25 ppm AsIII in the drinking water on the bladder epithelium in mice.
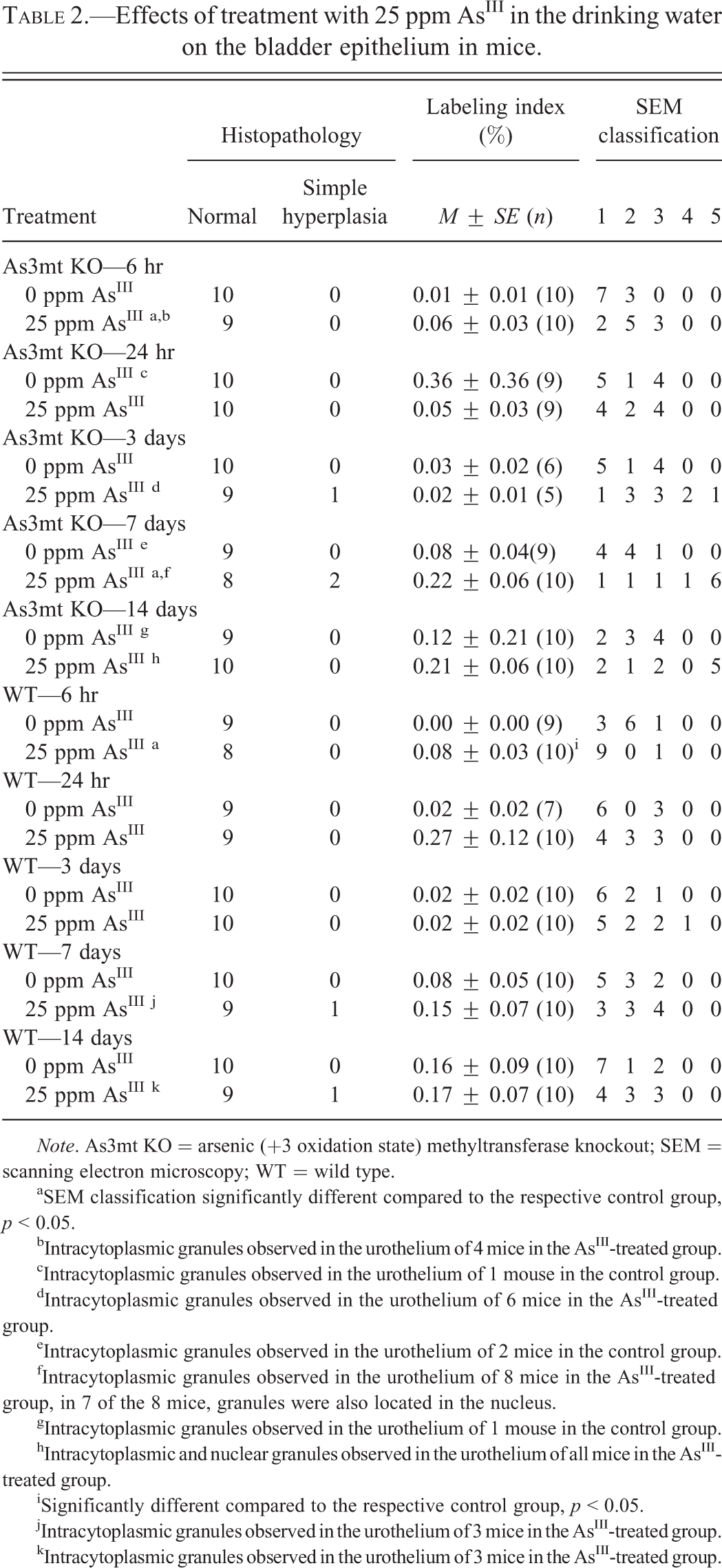
Note. As3mt KO = arsenic (+3 oxidation state) methyltransferase knockout; SEM = scanning electron microscopy; WT = wild type.
aSEM classification significantly different compared to the respective control group, p < 0.05.
bIntracytoplasmic granules observed in the urothelium of 4 mice in the AsIII-treated group.
cIntracytoplasmic granules observed in the urothelium of 1 mouse in the control group.
dIntracytoplasmic granules observed in the urothelium of 6 mice in the AsIII-treated group.
eIntracytoplasmic granules observed in the urothelium of 2 mice in the control group.
fIntracytoplasmic granules observed in the urothelium of 8 mice in the AsIII-treated group, in 7 of the 8 mice, granules were also located in the nucleus.
gIntracytoplasmic granules observed in the urothelium of 1 mouse in the control group.
hIntracytoplasmic and nuclear granules observed in the urothelium of all mice in the AsIII-treated group.
iSignificantly different compared to the respective control group, p < 0.05.
jIntracytoplasmic granules observed in the urothelium of 3 mice in the AsIII-treated group.
kIntracytoplasmic granules observed in the urothelium of 3 mice in the AsIII-treated group.
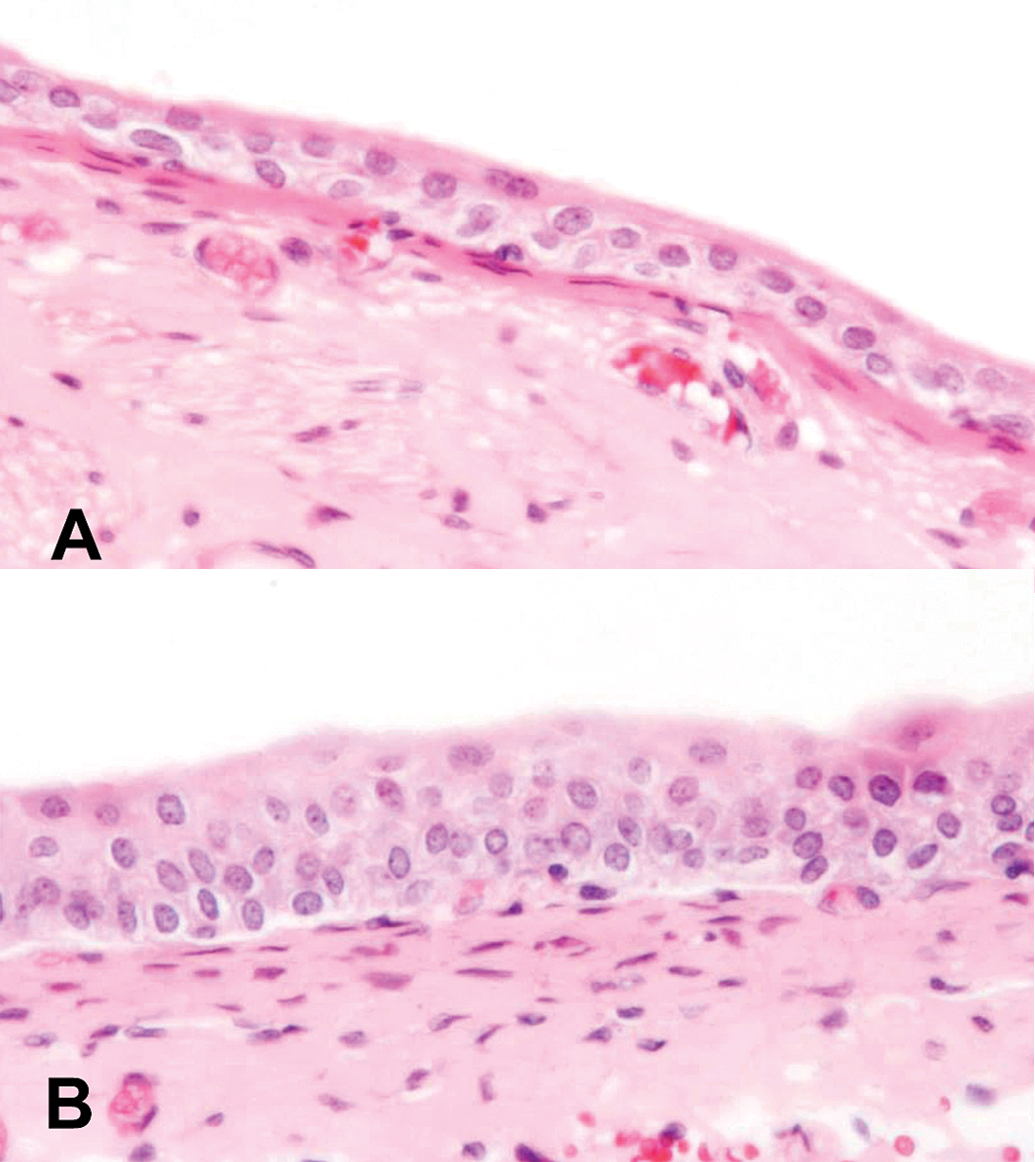
Light microscopic images of (A) normal epithelium of a urinary bladder from a control rat and (B) epithelium of a urinary bladder from a rat treated with 100 ppm AsIII for 7 days showing mild simple hyperplasia. Hematoxylin and eosin (H&E) stain, 400×.
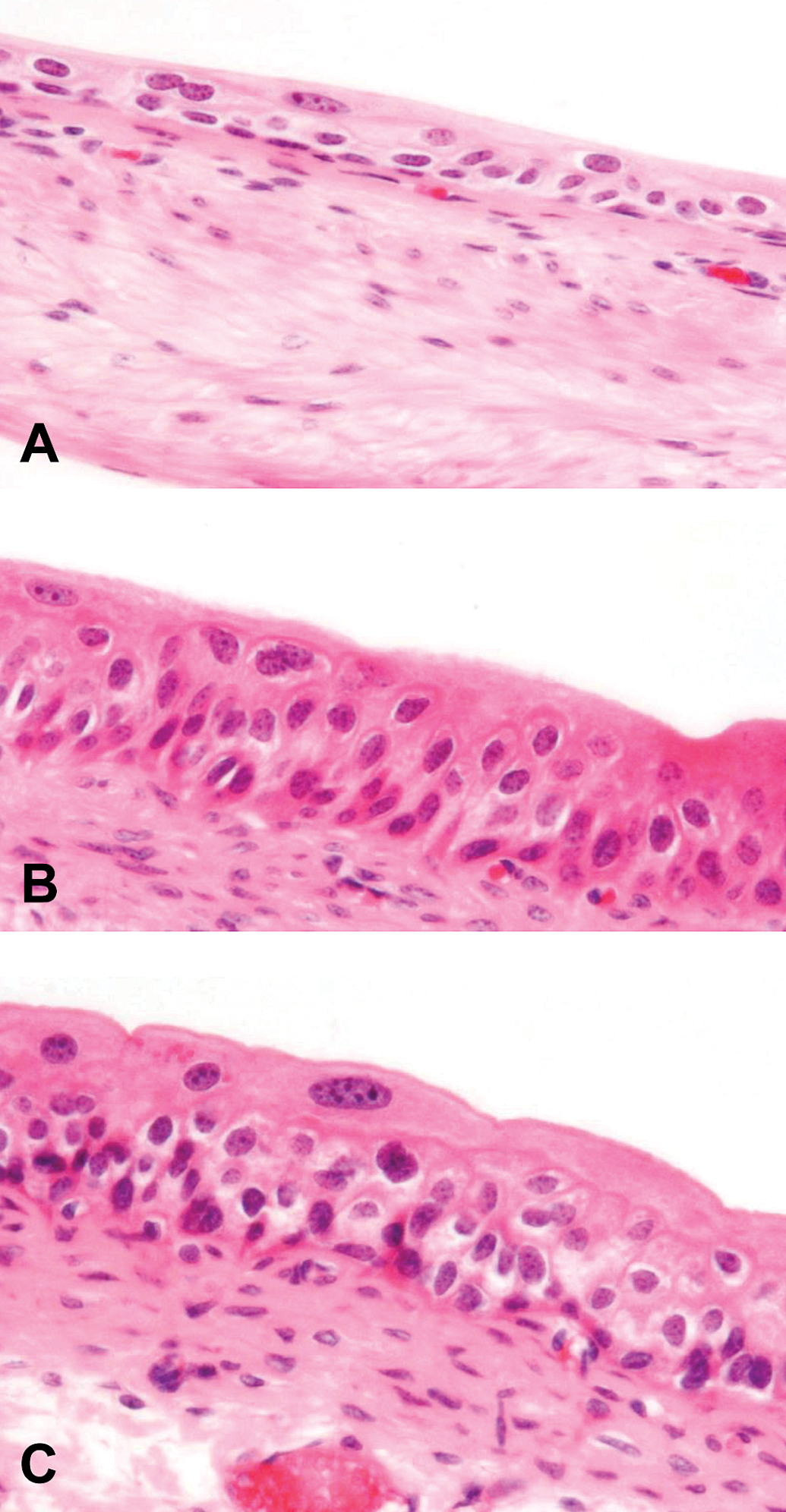
Light microscopic images of (A) normal epithelium of a urinary bladder from a wild-type control mouse, (B) epithelium of a urinary bladder from an As3mt KO mouse treated with 25 ppm AsIII for 7 days showing mild simple hyperplasia, and (C) epithelium of a urinary bladder from a wild-type mouse treated with 25 ppm AsIII for 7 days showing mild simple hyperplasia. Hematoxylin and eosin (H&E) stain, 400×. As3mt KO = arsenic (+3 oxidation state) methyltransferase knockout.
We have previously reported the presence of intracytoplasmic granules in the urothelial cells of WT and As3mt KO mice treated with arsenic (Suzuki, Arnold, Muirhead, et al. 2008; Yokohira et al. 2010, 2011), which contain arsenic predominantly as AsIII (Suzuki, Arnold, Muirhead, et al. 2008). In the present study, intracytoplasmic granules were observed in the urothelial superficial cells of As3mt KO mice as early as 6 hr after the start of treatment and 7 days after the start of treatment in the WT mice (Table 2). Seven days after the start of the treatment, intracytoplasmic granules were observed in all layers of the urothelium of the As3mt KO mice, and granules were also present in the nucleus of the urothelial cells.
SEM Examination of the Bladder Epithelium
Examination by SEM showed foci of cytotoxicity and necrosis in 1 bladder in the AsIII-treated rat group at 6 hr after starting treatment (Table 1 and Figure 3). Over time, there was an increase in the number of rats with AsIII-induced cytotoxic urothelial changes as well as in the severity of the changes (Table 1 and Figure 3). At day 14, the classification of bladder cytotoxic changes in the treated rats was significantly different from that of bladders in the rat control group (Table 1). In the As3mt KO and WT mice, AsIII-induced cytotoxic changes were present in the urothelium as early as 6 hr after the start of treatment (Table 2 and Figure 4). In the As3mt KO mice as in the rat, the number of mice with cytotoxic changes in the bladder and the severity of the changes increased as exposure time increased (Table 2 and Figure 4). In the WT mice, the number of animals with AsIII-induced changes increased after treatment for 24 hr (Table 2), but no change was observed at later time points in the number of animals with cytotoxic changes, or in the severity of lesions.
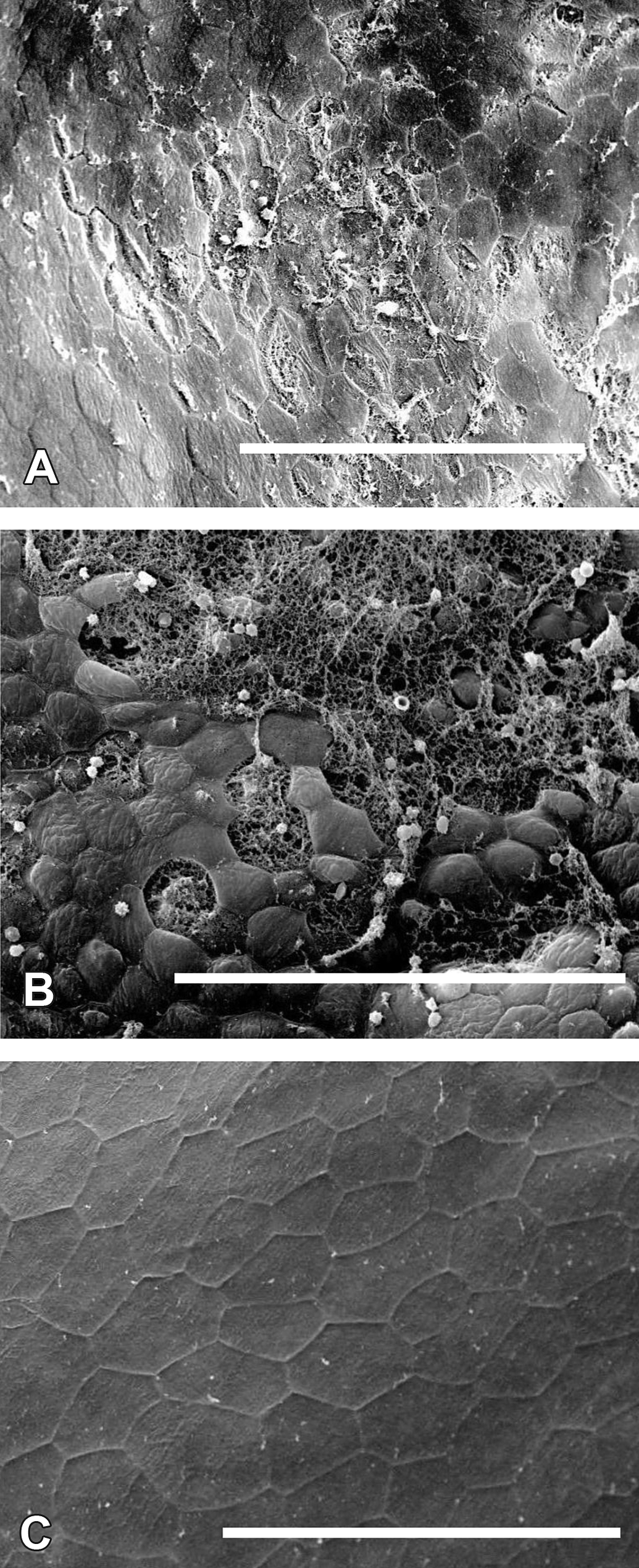
SEM photographs showing (A) a bladder from a rat treated with 100 ppm AsIII in the drinking water for 6 hr with an area of necrosis and exfoliation of some of the superficial cells, bar = 100 µm; (B) a bladder from a rat treated with 100 ppm AsIII in the drinking water for 14 days with a large area of extensive necrosis and exfoliation of the superficial cells, bar = 100 µm; and (C) a bladder from a control rat showing flat, polygonal superficial cells, bar = 200 µm. SEM = scanning electron microscopy.

SEM photographs showing (A) a bladder from an As3mt KO mouse treated with 25 ppm AsIII in the drinking water for 6 hr showing necrosis and exfoliation of the superficial cells, bar = 400 µm; (B) a bladder from a WT mouse treated with 25 ppm AsIII in the drinking water for 6 hr with an area of necrosis and exfoliation of the superficial cells, bar = 300 µm; (C) a bladder from an As3mt KO mouse treated with 25 ppm AsIII in the drinking water for 7 days showing piling up of round cells indicative of hyperplasia, bar = 100 µm; and (D) a bladder from a control WT mouse showing large flat, polygonal superficial cells, bar = 100 µm. As3mt KO = arsenic (+3 oxidation state) methyltransferase knockout; WT = wild type; SEM = scanning electron microscopy.
Immunohistochemical Determination of BrdU
There was no increase in the BrdU-labeling index in treated rats at any time point (Table 1). There was an increase in the labeling index in the AsIII-treated As3mt KO mice on day 7, and in the AsIII-treated WT mice on days 1 and 7; however, none of these increases were statistically significant (Table 2).
Discussion
The results of the present studies provide support for the hypothesis that the sequence of events leading to urothelial hyperplasia in rats and mice treated with AsIII is initiated by cytotoxicity with consequent cell proliferation. The findings were similar in rats and mice. The extent of cytotoxicity in knockout mice was slightly more pronounced as detected by SEM than the effect in the WT mice, in agreement with the results of previous studies comparing these two strains (Yokohira et al. 2010, 2011), but the changes overall by labeling index and light microscopy were similar. The slightly greater response in the knockout mice is likely because these mice are less able to eliminate the inorganic arsenic due to their inability to methylate the arsenic (Drobna et al. 2009). Evidence of cytotoxicity was observed by SEM as early as 6 hr in the rat and in the WT mice. By 24 hr, there was significant evidence of cytotoxicity in rats and both strains of mice. In the rats, there was no increase in the labeling index during the first 14 days of this study, but there was evidence of hyperplasia as detected by light microscopy as early as 24 hr after administration, although it was significant only by 14 days.
Hyperplasia, by definition, is an accumulation and increase in the number of cells. This occurs due to a combination of increased production of cells as well as their prolonged survival. Hyperplasia can be detected by light microscopy as an increase in the number of cell layers. For mild simple hyperplasia, this is an increase from the usual 3 cell layers to 4 or 5, and moderate hyperplasia involves 6 to 9 cell layers. The labeling index, in contrast, only measures the proliferation, and only at a single point in time. To fully assess the changes that are occurring in the urothelium, it is important to evaluate the findings by SEM, light microscopy, and labeling index to put in perspective what biologically is occurring in the urothelium (Cohen et al. 1990, 2007).
SEM is the most sensitive technique for detecting changes in the urothelium for a number of reasons. The changes can be observed over the entire bladder lumen rather than in thin slices seen by light microscopy or immunohistochemistry for labeling index. This is important since the initial mild changes that occur in response to arsenic administration are very focal and could easily be missed by light microscopy. Furthermore, the cytotoxicity caused by arsenic is primarily superficial, involving the luminal superficial cell layer of the urothelium and not the underlying layers. This superficial cell layer is very thin and is not always easily observed by light microscopy. Furthermore, there are artificial tears that occur during processing of tissue for light microscopy, making distinction between true cell death and artifact difficult (Cohen et al. 2007). However, these are readily distinguished by SEM.
As has been reported in mice (Suzuki, Arnold, Muirhead, et al. 2008), intracytoplasmic eosinophilic granules were detected in the present study following the administration of AsIII in As3mt KO mice as early as 6 hr after the start of treatment. We have demonstrated by electron microscopy that these granules are intramitochondrial. In the knockout mice, more of these granules accumulate, and they are present not only in the superficial cells but also in the intermediate and basal cell layers. In the knockout mice, in addition to the intracytoplasmic granules, intranuclear granules were observed. At this point, we have demonstrated that these granules contain arsenic, mainly as arsenite (Suzuki, Arnold, Muirhead, et al. 2008), and it is bound by protein. Interestingly, similar granules have been observed in mice administered DMAV, but without any evidence of cytotoxicity (Arnold et al. 2006). It would appear that these granules function as a storage form of arsenic and are not of any adverse toxicologic significance. It is interesting to note that similar granules were observed in exfoliated urothelial cells of acute promyelocytic leukemia patients treated with high levels of arsenic trioxide (Wedel et al., in press).
In summary, the data presented here in combination with data reported previously (Suzuki, Arnold, Ohnishi, et al. 2008; Yokohira et al. 2010, 2011) strongly support the hypothesis that the sequence of events in the mode of action for the urothelial effects of orally administered inorganic arsenic involves superficial cytotoxicity with consequent regenerative increased cell proliferation. This occurs in the rat and in the mouse, and is similar to the findings associated with the administration of DMAV in rats (Cohen et al. 2006).
Supplemental Material
Supplemental Table S1. Effects of treatment with 100 ppm AsIII on terminal body and bladder weights and food and water consumptions in rats.
Supplemental Table S2. Effects of treatment with 25 ppm AsIII on terminal body and bladder weights and food and water consumptions in mice.
Statement of Interests
Supported in part by grants from the Organic Arsenic Products Task Force and the Arsenic Science Task Force.
Footnotes
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported in part by grants from the Organic Arsenic Products Task Force and the Arsenic Science Task Force.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
