Abstract
The immune system represents a key defense mechanism against potential pathogens and adverse non-self materials. During pregnancy, the placenta is the point of contact between the maternal organism and non-self proteins of the fetal allograft and hence undoubtedly fulfils immune functions. In the placenta bacteria, foreign (non-self) proteins and proteins that might be introduced in toxicological studies or by medication are barred from reaching the progeny, and the maternal immune system is primed for acceptance of non-maternal fetal protein. Both immunologic protection of the fetus and acceptance of the fetus by the mother require effective mechanisms to prevent an immunologic fetomaternal conflict and to keep both organisms in balance. This is why the placenta requires toxicological consideration in view of its immune organ function. The following articles deal with placenta immune-, control-, and tolerance mechanisms in view of both fetal and maternal aspects. Furthermore, models for experimental access to placental immune function are addressed and the pathological evaluation is elucidated. “The Placenta as an Immune Organ and Its Relevance in Toxicological Studies” was subject of a continuing education course at the 2012 Society of Toxicologic Pathology meeting held in Boston, MA.
Introduction
One of the major challenges of human gynecologic and toxicological research is to find an appropriate model for the human placenta. A constant, urgent need for such models arises from the most common placenta-based pregnancy-specific disorders like preeclampsia (Pennington et al. 2012; Redman et al. 2012) and intrauterine growth restriction. In addition, the placenta has to cope with toxicological challenges like medication in pregnancy or with other potentially harming substances like tobacco smoke which contains numerous compounds that can act as endocrine disruptors (Stasenko et al. 2010). In regard to morphological structure, the human placenta is hemochorial, discoidal, and contains fetal villi which are anchored into the basal plate of maternal endometrium and are surrounded by the maternal blood space (Leiser and Kaufmann 1994). The villous mesenchyme contains a complex, branching blood vessel system consisting of stem, intermediate, and terminal villous capillaries (Leiser et al. 1997). However, this specific placental type does not exist in most experimental animals. Therefore, a researcher has to carefully choose the experimental animal of choice depending on research aim of the planned study and on the aspect of placental function that should be most similar to the human placenta. The ideal animal model would have to be a small species, with a short reproduction cycle and most similar in outer placental form, fetomaternal interdigitation, and trophoblast invasion. The available animal model most similar to humans is the cynomolgus monkey (Macaca fascicularis). In recent years, this species, displaying hemochorial placenta, morphologically similar to the human placenta (De Rijk and van Esch 2008), represents the most frequently used nonhuman primate (NHP) model in reproductive toxicological studies. However, toxicologists have paid relatively little attention to the immunological role of the placenta and thus on its impact on medication. In this article, the placental morphology of relevant animal species, inclusive NHPs, is comparatively elucidated. Additionally, the expression of immunological tolerance molecules in the cynomolgus monkey placenta is presented, as these are vital for fetomaternal tolerance.
Material and Method
Tissue specimens included in the article are derived from different sources: Cynomolgus monkey (M. fascicularis) placentae were collected from cesarean sections from control and placebo cynomolgus macaques from toxicological preclinical safety studies carried out at Covance Laboratories GmbH in Münster, Germany. In detail material from gestational days (GD) 50, 80, 85, 100, and 150 (regular day of birth: GD 160–165) was collected. Bovine midgestational placenta (Haeger et al. 2011), rat placental tissue (GD 20), and human placental tissue at the end of pregnancy (39th week) were also included in the study and obtained at the Department of Anatomy, University of Veterinary Medicine Hannover, Hannover, Germany or at the Department of Obstetrics and Gynecology at the Justus Liebig University Giessen, Giessen, Germany. The collection of human tissue was approved by the local ethics committee and informed consent was given in all cases (Haeger et al. 2011). Porcine placental samples (GD 112) were a kind gift from Vibeke Dantzer (Department of Basic Animal and Veterinary Sciences, Copenhagen University, Copenhagen, Denmark). All procedures involving animals were approved by local ethics committees.
Histological evaluations were performed on formaldehyde- and paraformaldehyde-fixed tissues. Staining methods included were Delafield’s hematoxylin, periodic acid-Schiff, or immunohistochemical staining with antibodies. The antibodies used were obtained from the following sources: antihuman leukocyte antigen (HLA)-G and anti-annexin II antibodies from Abcam (Cambridge, UK), anti-CD95 from Medac (Wesel, Germany), and anti-indoleamine 2,3-dioxygenase (IDO) from Acris Antibodies (Herford, Germany). Anti-HLA-DR, anti-cytokeratin 17 and 18, antiprogesterone receptor, and anti-Ki-67 were all derived from Dako (Hamburg, Germany).
All antibodies were used according to the manufacturer’s recommendations. A staining control was routinely performed. Antibodies were tested before use in three different solutions. To obtain best results, immunostaining was performed with three different detection systems: (1) Envision/HRP-system (Dako, Hamburg, Germany), (2) Ultra Vision LP Detection System, large volume/HRP polymer, and (3) Ultra Vision LP Detection System, large volume/antipolyvalent/HRP. Unspecific staining was blocked with commercially available Hydrogen Peroxide Block (all Medac, Wedel, Germany) or with autologous serum collected from monkey specimens in our colony. As positive control tissue served specimens as suggested by the manufacturer, that is, HLA-G: placenta, HLA-DR: colon, Annexin: kidney, IDO, and CD95: tonsils. In view of antibody application on nonhuman target tissues, we furthermore have also used marmoset organs as additional controls. Specificity of further antibodies used in this article have been described (Buse, Zoeller, and van Esch 2008; Haeger et al. 2011; Martin and Buse 2008; van Esch et al. 2008).
Results
General Comparative Placental Morphology
Shape
The human placenta is discoid like that of the mouse and rat but in contrast to these small animals, it is composed of distinct, closely apposed cotyledons, which are located close to each other (Leiser and Kaufmann 1994). The other two placental types exhibiting a focal distribution of interdigitating structures are a zonary shape, found in carnivores, and a cotyledonary shape, found in ruminants (Bjorkman 1954, 1970). All these types contrast the species with diffuse placentae, like pigs and horses in which exchange units are distributed evenly all over the chorion (Amoroso 1952; Macdonald and Bosma 1985). However, the outer form of the placenta does not tell so much about its function, because placental efficiency largely depends on the fetomaternal interdigitating structures and the angioarchitecture found in those structures.
Fetomaternal Interdigitation
The human is sharing the feature of having fetal villous trees with many different species, especially large animals like ruminants (cow, sheep, and goat) and horses (Amoroso 1952; Steven 1975). This type invites studies where vascular architecture and blood flow are potentially affected, because the pattern of villous vascularization is comparable. A simpler type, found in pigs, dog, and cat, is equipped with folds or lamellae (Leiser and Kohler 1983; Macdonald and Bosma 1985). The most complex pattern of interdigitation is where fetal and maternal tissues are interwoven in a labyrinthine fashion. This type is found in the most favorite experimental animals: mouse, rat, and guinea pig (Kaufmann and Davidoff 1977; Mossmann 1987).
Degree of Trophoblast Invasion
The degree of trophoblast invasion is reflected by the number of tissue layers separating fetal and maternal blood stream. The human placenta has a highly invasive trophoblast, resulting in a degeneration of all maternal tissue layers including uterine epithelium, connective tissue, and maternal endothelial cells (Grosser 1927). Consequently, a hemochorial condition is established where fetal villi are immersed in maternal blood, allowing easy exchange of nutrients and metabolites, including maternal immunoglobulins. The hemochorial type is considered the most original/ancestorial type (Carter and Enders 2004). Unfortunately, most other species with hemochorial placentation, which are used as experimental animals, do not have fetal villi, but a labyrinthine interdigitation pattern. Additionally, this pattern can be subdivided into species with one, two, or three layers of trophoblast (Enders 1965). At term, the human shares a hemomonochorial placenta with caviomorphs, like the guinea pig and haplorhines as New World and Old World monkeys and apes (Carter and Pijnenborg 2011; Kaufmann and Davidoff 1977), while a hemodichorial condition is found in lagomorphs and the early human placenta. The most elaborate hemotrichorial form is observed in rodents, like mouse, rat, and hamster (Enders 1965).
In species with an intermediate degree of invasiveness, the uterine epithelium, and connective tissue degenerate, but the maternal endothelium remains, resulting in an endotheliochorial condition. This type is found in many carnivores (Bjorkman 1970). In the least invasive placental type, the maternal tissue layers remain intact, with microvilli of the intact uterine epithelium interdigitating with those of the trophoblast (Wooding 1992). This epitheliochorial condition is considered the latest evolutionary stage (Carter and Enders 2004) and has the advantage that parturition usually does not result in loss of blood. It is found in large animals, not typically used in experiments, like horses and pigs (Amoroso 1952; Macdonald and Bosma 1985). Yet it has to be emphasized that a subpopulation of equine trophoblast cells (chorionic girdle) destroys at very distinct sites the uterine epithelium in order to invade into the endometrial stroma, remaining there for more than half of equine gestation (Noronha and Antczak 2010). Ruminants also display a slightly more invasive type than the pig. In ruminants, mostly binucleated trophoblast giant cells (TGC) migrate toward the uterine epithelium and fuse with single uterine epithelial cells thus forming mostly trinucleated fetomaternal hybrid cells. This hybrid cell formation is reflected by the term synepitheliochorial (Wooding 1992). Since ruminant TGC never invade beyond the basement membrane, it has been referred to as restricted invasion (Pfarrer et al. 2003). Interestingly, certain prosimians such as lemurs also display an epitheliochorial placental type (Benirschke and Miller 1982).
Vascular Architecture
The blood flow interrelationship of fetal and maternal circulations has a major impact on theoretical exchange potential. While the least effective concurrent flow has not been reported for placentae, crosscurrent and countercurrent flow interrelationships and a combination of both have been described for several species. Early work involved analysis of vascular corrosion casts by scanning electron microscopy (Leiser and Kohler 1983). Together with histology, these casts could show that the vasculature responsible for exchange (capillaries) usually tends to lie close to the epithelial basal membranes. Therefore, the shape of the surface of a fetal cast mirrors the epithelial (trophoblast) surface and complements the one from the maternal side (Leiser and Pfarrer 2006). However, in hemochorial conditions, two ways exist to guide the maternal blood flow, which depend on the type of fetomaternal interdigitation. In the labyrinthine pattern found in rodents, the maternal blood flows in sinusoids, which are oriented in fetomaternal direction leading to a countercurrent flow relation (Dantzer et al. 1988; Kaufmann and Davidoff 1977). In contrast, in humans, the maternal blood circulates in blood channels leading to a so-called multivillous or pool flow system (Leiser et al. 1997). The development of the vascular system is regulated by many factors, among them oxygen. Hypoxia stimulates angiogenesis, while hyperoxia reduces growth of capillaries (Ferrara, Gerber, and LeCouter 2003). This has been shown in different model animals such as guinea pig and sheep. Guinea pigs had an increased placental capillarization when submitted to hypobaric chambers, and sheep showed similar results when kept at high altitude during pregnancy (Krebs, Longo, and Leiser 1997; Scheffen et al. 1990). Fetal adaptions such as increased placental capillarization of fetal villi have also been observed in human fetal placental villi of heavy smokers (Pfarrer et al. 1999). An opposite finding of reduced capillarization of fetal terminal villi is suggested to be a compensatory mechanism for a hyperoxic intervillous space due to insufficient extraction of oxygen (Krebs et al. 1996).
In Vitro Systems
In order to analyze human placental physiology or undertake toxicology testing in vitro, very different test systems are available ranging from more or less complex cell line–based tools to explants and placental perfusion models. In regard to cell-based assays, the use of 3-dimensional spheroids could improve reliability of toxicological studies and/or drug development (Friedrich, Ebner, and Kunz-Schughart 2007). Spheroids have been formed, for example, with trophoblast cells of human (Grümmer, Hohn, and Denker 1990), equine (de Mestre et al. 2008) and bovine (Haeger et al. 2011) origin. Recognizing the potential of 3-dimensional spheroids, new companies have been founded in the past years which offer spheroid-based assays for drug development and/or the development of 3-dimensional microtissues. In addition, more elaborated culture systems like Rotary Cell Culture Systems can be used in toxicological studies (Goodwin et al. 2000). Due to ethical barriers, certain experimental in vitro/in vivo setups (like ectopic injection of trophoblast spheroids) can hardly be carried out in humans. In such cases, researchers with a primary interest in human placentology and interactions of extravillous trophoblast (EVT) and immune cells could take a closer look at the horse placenta in which the HLA-class I positive chorionic girdle trophoblast encounters an accumulation of lymphocytes while invading the maternal endometrial stroma (Noronha and Antczak 2010).
Cynomolgus Monkey/NHP Placentae
Taking in account the aspects of placental similarity (shape, interdigitation, degree of trophoblast invasion, and vascular architecture) and the desire of using an animal model that comes closest to human placental structure, the use of NHP species appears as a good choice. Even though primates are our closest relatives, it has to be emphasized that NHP placentae vary considerably in placental type based on degree of trophoblast invasion. In fact, certain primates like lemurs possess an epitheliochorial type in contrast to the hemochorial type of New World and Old World Monkeys and the great apes (Benirschke and Miller 1982). New World Monkeys are also different from great apes and Old World Monkeys as they possess a “pseudolabyrinthine” placenta and seem to lack EVT. Due to ethical and judicial concerns, certain NHPs like great apes can virtually not be used as model species for the human placenta in reproductive toxicological studies even though they resemble its structure most closely as seen, for example, by interstitial invasion of the decidua (Carter and Pijnenborg 2011; Pijnenborg, Vercruysse, and Carter 2010). In Old World monkeys, interstitial EVT invasion of the decidua is very limited; yet, trophoblast invasion of the spiral arteries of the decidua basalis is similar (Blankenship and King 1996). Since Old World Monkeys like Macaques can be practically handled in industrial scale studies and additionally closely resemble human placental structure, they are commonly used for studies on reproductive toxicology. In particular, the cynomolgus monkey (M. fascicularis) is frequently used for such studies as an NHP animal model (Buse et al. 2003) since the placental structure is well known (De Rijk and van Esch 2008) and because extensive data are available on cynomolgus female reproductive biology and development of the fetal immune system (Buse 2005). For comparison, histological images of the placentae of pig (epitheliochorial), bovine (synepitheliochorial), rat (labyrinthine hemochorial), and human and cynomolgus monkey (villous hemochorial) are shown in Figure 1.
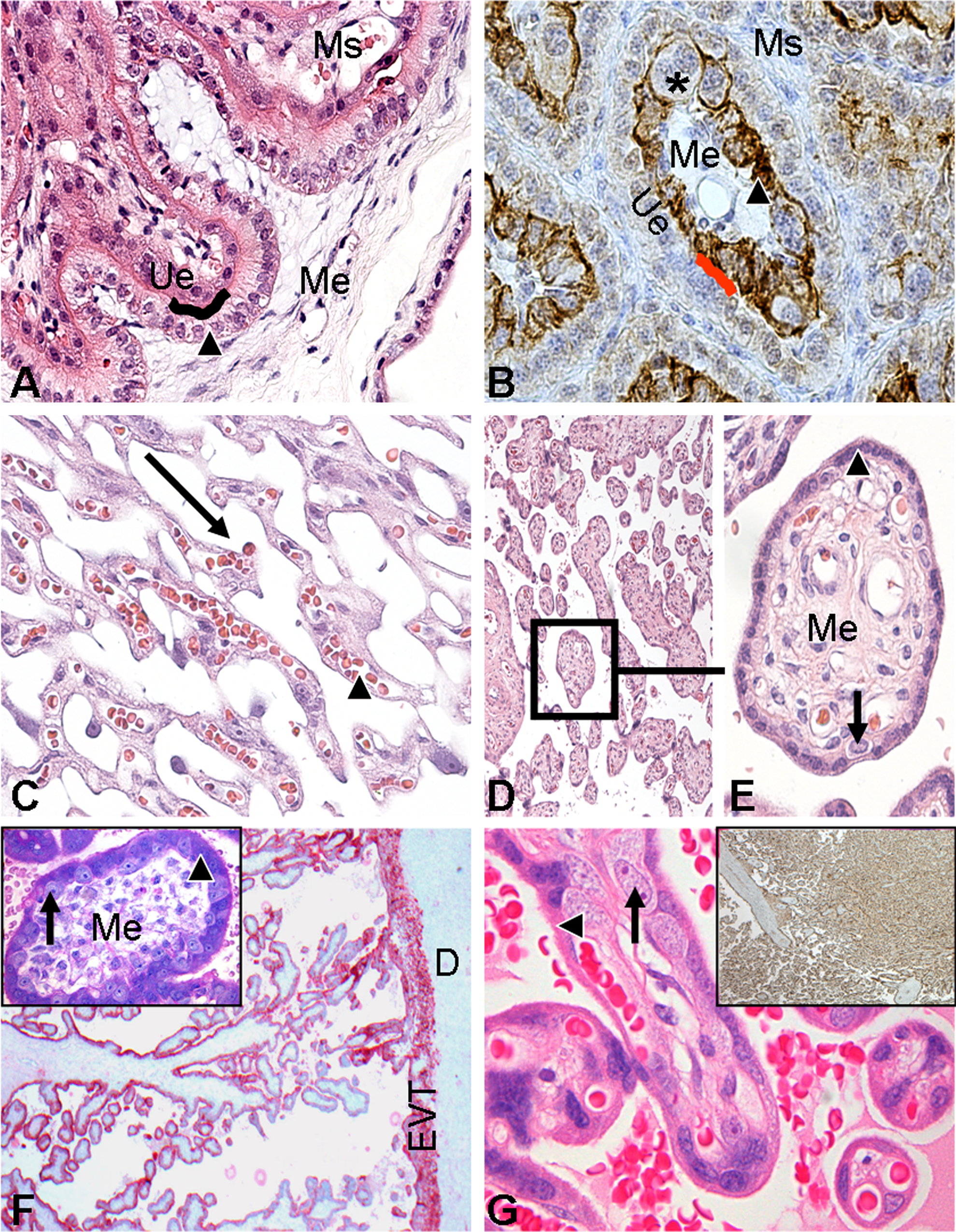
Placentae of pig (A), bovine (B), rat (C), human (D and E), and cynomolgus monkey (F and G). (A) H&E stain of porcine placenta on GD 112: Pale trophoblast (arrowhead) is lying apposed to the isoprismatic uterine epithelium (Ue), which persists throughout gestation (epitheliochorial type; 40×). Mesenchyme (Me) is located underneath the trophoblast, while endometrial maternal stroma (Ms) can be found underneath the Ue. The feto–maternal contact interface can be seen as a dark reddish line which is exemplarily indicated (black line). (B) Pan-cytokeratin staining of bovine midgestational placenta (synepitheliochorial, villous type): In contrast to the pig, ruminants have two trophoblast cell populations, namely 80% keratin-positive, uninucleate trophoblast (arrowheads) and invasive, keratin-negative trophoblast giant cells (*; TGC; 40×). (C) H&E stain of GD 20 rat placenta: in the labyrinthine exchange area, wide maternal blood channels (arrows) are neighbored by a very thin layer of syncytiotrophoblast (hemochorial). Fetal capillaries (arrowhead) are located in the mesenchyme. (D and E; 40×) H&E stain of human placental tissue (39th week; villous, hemochorial): Numerous fetal villi are present in the intervillous space which is in vivo filled with maternal blood (D): In single villi, the outermost trophoblast layer is the syncytiotrophoblast (arrowhead), which has apical microvilli (brush border) and is in contact with maternal blood. Below this layer at the end of gestation, only a few cytotrophoblast cells (arrow) can be found. Therefore, this type is referred to as hemomonochorial. (F) Cytokeratin 18 staining of cynomolgus monkey (villous, hemochorial) placental tissue at GD 50: All trophoblasts (villous and extravillous trophoblast (EVT)) are keratin positive. The EVT is distinctly separated from the decidua (D; 2×). Inset (periodic acid-Schiff staining): villus in early tertiary stage with a mesenchymal core covered with cytotrophoblast (arrow) and syncytiotrophoblast (arrowhead; 40×). (G) Cynomolgus monkey placenta on GD 150: in comparison to GD 50 the villous stroma now contains blood vessels. The outer syncytiotrophoblast (arrowhead) and singular cytotrophoblasts (arrow) can be seen (40×). Inset (CD95 staining): Overview of branching fetal villi in the same magnification as in (F). The maternal blood space is almost completely filled with highly branched tertiary villi (2×). GD = gestational days.
The Cynomolgus Monkey Placenta
Basic Morphology in Development
The cynomolgus monkey placenta is comparatively described at an early developmental stage on gestation day 50 (GD 50, e.g., the day of embryo–fetal transition) and a late developmental stage close to parturition, on GD 150 (average gestation length: 160–165 days).
On GD 50, the intervillous space appears as a relatively large, maternal blood-containing cavity between the chorionic plate (fetal oriented tissue; fetal origin) and the basal plate (maternal oriented tissue; maternal and fetal origin; Figure 1F). The fetal EVT cells and the maternal endometrium (decidua) are in close contact. Placenta villous trees extend from the chorion into the blood cavity and are anchored with the tips of trees into the basal plate. Whereas the EVT is organized as a solid and separate layer in the monkey placenta, it appears heavily intermingling with decidua cells in the human placenta (Kammerer, von Wolff, and Markert 2004; Poehlmann et al. 2006). Fetal tissues lining the blood cavity are covered with a syncytial epithelium, the so-called syncytiotrophoblast layer. Subjacent to the syncytiotrophoblast layer is a highly proliferative epithelium, the so-called cytotrophoblast layer. All trophoblasts (syncytio-, cyto-, extravillous-) can be identified histologically by intense cytokeratin 18 or cytokeratin 8 staining and thus distinguished from mesenchymal or decidual tissues (Daya and Sabet 1991; Uckan et al. 1997). The villi appear highly immature, characterized by poor arborization, loose mesenchyme, lack of vascularization, and an obvious lack of differentiated immune cells (Hofbauer cells, CD3, CD20, etc.). Structural proteins such as vimentin, actin, and S100 have been demonstrated in villi on GD 50 whereas desmin has not.
Placenta development advances significantly between days 80 and 100 as villous arborization and vascularization increase and the villous mesenchyme populates with a variety of cell types.
By GD 150 villi are highly arborized, displaying many distinct villi and narrow intervillous space in histological sections (Figure 1G). Detailed villous evaluation demonstrates a differentiated mesenchymal core occupied with a variety of immune cells (Hofbauer cells, macrophages, T-cells, and B-cells). Villous vascularization appears mature according to centrally located vessels branching into a dense network of capillaries that are separated from the intervillous maternal blood by a thin syncytiotrophoblast layer. At this stage of gestation, only singular cytotrophoblastic cells can be detected underneath the syncytiotrophoblast (Figure 1G).
The overall cynomolgus monkey placenta morphology is similar to human placenta but differs considerably from rodents (De Rijk and van Esch 2008; Kaufmann 1990; Lewis and Benirschke 1997).
Fetal Tolerance Mechanisms
On GD 50, the immune system of the fetal cynomolgus monkey is highly incomplete and unable to defend from external attacks and challenges (Buse 2005; Martin and Buse 2008). Therefore, at this age, an early fetal strategy with two key aspects is required: first, calming down the maternal immune cell attack potential and second evading resident maternal immune cells located in the placenta. The human fetus achieves this strategy with the help of certain fetal-borne molecules (Thellin et al. 2000). These molecules may have interspecies differences in specificity and source of synthesis and not all are necessarily described in the cynomolgus monkey. In this article, we describe the localization and discuss the potential roles and interactions of progesterone, annexin II, HLA, FAS–FAS-ligand (FAS-L), and IDO with respect to strategies related to calming or evading the maternal immune system with particular focus on the embryo–fetal transition stage on GD 50 and preparturition stage on GD 150.
Progesterone
Progesterone, annexin II, and the Ca+/− binding protein S100 are interactively connected in fetal immune suppressing functions. Progesterone is observed during the entire pregnancy. It is initially derived from the ovarian corpora lutea, which in cynomolgus monkeys are often active until birth of the fetus. Additionally, placenta-derived progesterone is synthesized post GD 50 (Figure 2A). This was concluded from pregnant female progesterone serum levels and from the villous expression of progesterone receptors on GD 100 but not on GD 50 (Figure 2B). On GD 150, the serum progesterone level has elevated significantly, similar to pregnant humans. Progesterone has many functions that include immunosuppression, interaction with the fetal adrenals, and relaxation of the myometrium. In the placenta, progesterone initiates linkage of the Ca++ donating protein S100 and annexin II thus activating immune relevant functions (Junqueira, Carneiro, and Kelley 2002; Lüllmann-Rauch 2003; Mjosberg et al. 2009).
Progesterone blood levels (A) and progesterone receptors (B) and annexin II (C) expression in cynomolgus monkey placenta. Blood progesterone in cycling and pregnant animals. Blue line: follicular and luteal phase (n = 200 females and females in early pregnancy): the peaking progesterone is considered of luteal origin. Red line, in advanced pregnancy, the progesterone declines until about pregnancy day 50 (arrow, GD 50) and then elevates continuously until birth (n = 77 females). Black line: progesterone level averaged from n = 4 pregnant females by weekly blood collection. The progesterone elevation in the 2nd and 3rd trimester is attributed to placental production more than luteal production. (B) GD 150: Progesterone receptors in nuclei of villous stroma cells (arrowheads; 40×). The progesterone receptor could be found in some syncytiotrophoblast nuclei only. No immunopositive nuclei have been observed in GD 50 placentae. (C) On GD 50, annexin II is strongly expressed on the brush border of the syncytiotrophoblast (arrow) and on the cell membranes of extravillous trophoblasts (20×). This pattern is also observed in later stages of gestation (not shown). Inset: In floating GD 50 villi, mesenchymal tissue is also strongly positive for annexin II (2×). (D) Annexin II in decidua and maternal endothelium of blood vessels (arrows; 40×). GD = gestational days.
Annexin II
The annexin II molecule is ubiquitous in the cynomolgus monkey placenta. It exists as a membrane-bound and soluble molecule and is involved in both immunosuppressive and allograft protecting functions. Bound to phospholipid Ca++ ions, annexin II is described as an effective inhibitor of lymphocyte proliferation and is moreover postulated to be a mediator of IgG/IgM synthesis in B-cells and of transmembrane ion channel transport (Kaczan-Bourgois et al. 1996; Aarli et al. 1997; Siever and Erickson 1997; Thellin et al. 2000; Zhang and Wu 2007). While the exact function of annexin II in early stage placenta development is unclear, its coexpression with S100 and presence in brush border membrane suggests a role in the cynomolgus monkey.
On GD 50, annexin II is significantly expressed in all cynomolgus monkey placenta tissues, similar to human and rodent placenta (Kim and Hajjar 2002; Siever and Erickson 1997). Cell membrane expression is particularly strong on syncytiotrophoblast, EVT, and decidual cells (Figure 2C). An observed coexpression of annexin II (membraneous expression) and S100 protein (cytoplasmic expression) strongly argues for early placenta immune-function involvement. In floating fetal villi, annexin II is also strongly expressed in the mesenchyme (Figure 2C).
On GD 150, annexin II expression is similar to annexin II expression on GD 50 as far as syncytiotrophoblast and EVT and decidual cells are concerned. In contrast to GD 50, annexin II is not found in the mesenchyme of floating villi (not shown). With the large number of functions attributed to annexin II and the significance of its expression, it is reasonable to assume similarity between cynomolgus monkeys and humans (Kim and Hajjar 2002; Siever and Erickson 1997).
HLA
The HLA (classes I and II) molecules are involved in the recognition of non-self protein and antigen presentation. Lack of class I molecules on the surface of the fetal syncytiotrophoblast or expression of non-self class I molecules inevitably activates the maternal placenta immune cells (i.e., class II carrying cells: natural killer (NK) cells [CD16], dendritic cells, macrophages [CD68], activated T-cells [CD3 and CD4], and B-cells [CD20]) for immediate elimination of the non-self cells/protein. HLA-DR is a class II molecule and a classical indicator of immune cell challenge (Challis et al. 2009; Fitzgerald et al. 2009).
In the cynomolgus monkey placenta of GD 50, there is generalized HLA-DR staining of placental villous tissue which is, however, absent on GD 100 (Figure 3A). On GD 100, maternal dendritic cells (positive for HLA-DR; negative for CD3, CD4, CD16, CD20, and CD68) are exclusively located adjacent to villous lesions as demarcated by fibroid structures. This argues strongly for both early integrity of the maternal HLA class II-system. Moreover, the nonself (i.e., at least paternal) HLA class I molecules on the syncytiotrophoblast are suspected to be fully downregulated effectively inhibiting maternal immune cell recognition.
Expression of HLA class I molecules in cynomolgus placenta. (A) HLA-DR staining on GD 100 (20×): trophoblast cells are negative for HLA-DR like most of the villous mesenchyme. HLA-DR positive cells cover a fibroid (asterisk). Inset (10×): on GD 50 HLA-DR immunostaining occurs almost ubiquitously in the fetal villous tissue. (B) Theoretical rationale for the placeholder role of placental HLA-G: foreign HLA class I molecules (upper example) or lack of HLA class I molecules (middle) lead to activation of maternal immune cells. The HLA-G molecule is accepted as a placeholder (lower bottom) and is considered an effective inducer of apoptosis (immune inhibitory activity). (C) HLA-G expression in GD 50 cynomolgus monkey placenta (10×): fetal anchoring (AV) and floating villi and extravillous trophoblasts are HLA-G negative, whereas a high number of maternal decidua cells are intensely stained. (D) HLA-G expression on GD 150: the syncytiotrophoblast displays an apical cytoplasmic HLA-G staining (arrowheads) whereas the villous stroma is devoid of HLA-G. Arrow: an immunopositive maternal white blood cell; asterisk: fibroid (40×). HLA = human leukocyte antigen. GD = gestational days.
On GD 150, all fetal tissues with the exception of a small villous stroma cell population is HLA immunonegative, and maternal immunoreactive immune cells are seen in the placental blood compartment in low numbers. Hence, the placenta obviously does not confront any immunologic challenge. In view of absent or downregulated classical HLA class I molecules on the syncytiotrophoblast–maternal blood border, a mandatory placeholder molecule is postulated.
HLA-G
HLA-G represents the placeholder molecule in the placenta. It is considered one of the major players in fetal immunologic evasion (Rouas-Freiss et al. 1997) by means of effectively increasing the activation threshold for maternal immune response. It exists in membrane bound and soluble isoforms and is described interacting with apoptosis inducing and inhibitory immune cell receptors (ILT2, ILT4, KIR4, and DL4; LeMaoult, Rouas-Freiss, and Carosella 2005a, 2005b; LeMaoult et al. 2005; Moreau et al. 1998). HLA-G thus represents the basis of resistance to NK-cell/activated immune cell induced cytotoxicity (Figure 3B).
On GD 50, the fetal cynomolgus monkey placenta tissue appears HLA-G immune-negative. Surprisingly, distinct HLA-G stain is expressed on the maternal tissue–derived decidual cell membranes (Figure 3C). This staining pattern changes on about GD 80 to GD 100 with cytoplasmic stain of the syncytiotrophoblast and the EVT. The stain shows a granular “rim phenomenon” that continuously intensifies until GD 150 (Figure 3D). The membranous decidual cell staining persists until birth.
HLA-G expression has frequently and controversially been described in humans and mostly emphasized on EVT cells, whereas immunoreactivity in syncytiotrophoblast and decidua are rarely mentioned (LeMaoult, Rouas-Freiss, and Carosella 2005a; McCormick et al. 2009). The differences between human and cynomolgus monkey HLA-G expression cannot be conclusively explained and may at least in part correlate with different trophoblast–decidua invasion patterns in both species (Figure 1E–F). Syncytiotrophoblast HLA-G immunoreactivity, with an obvious apical cytoplasmic stain was mentioned before in more immature (GD 36–42) NHP placentae (Bondarenko et al. 2009), but has in our study also been detected in more advanced stages of NHP gestation.
One of the described protective HLA-G mechanisms is the activation of the so-called programmed cell death receptor (FAS receptor), which is active in activated intervillous immune cells and considered to act via self-apoptosis induction.
FAS–FAS-L
The FAS receptor (synonymous: CD95) is a frequently expressed potent immune cell activator, with fatal action on non-self protein expressing cells. The FAS-L is synthesized in the placenta, binds to the FAS receptor, and is considered a key protective mechanism against activated lymphocytes. It inhibits reactive TH1 cytokine synthesis and induces self-apoptosis in activated immune cells (De Falco et al. 2005; Uckan et al. 1997; Figure 4A).
Role and expression of FAS-L (A, B) and IDO (C, D, E) in cynomolgus monkey placenta. (A) Theoretical background of FAS–FAS-L action: (1) FAS-L is synthesized in the syncytiotrophoblast cytoplasm, integrated into the syncytiotrophoblast membrane, and delivered into the maternal blood compartment. (2) A challenging molecule is recognized by an NK-cell. (3) The activated NK-cell synthesizes and expresses FAS-receptor prior to Th1 cytokine synthesis. (4) FAS-L binds to FAS-receptor and (5) inhibits the synthesis of Th1 cytokines and (6) activates an apoptosis cascade ending up with NK-cell death. (B) Intense syncytiotrophoblastic FAS-L (CD95) staining in the GD 50 placenta (2×). Inset: the FAS-L is not expressed in the decidua but in certain white blood cell (WBC) in the maternal blood (40×). (C–E) IDO expression in the cynomolgus monkey placenta on GD 50 (C), 85 (D), and 150 (E): on GD 50 (C) there is a general cytoplasmic stain in all fetal placenta cells, which is considered an undefined cross reactivity. IDO immunoreactive WBCs are frequent in the maternal blood (arrow). Inset: villus with Ki-67 positive cells predominantly underneath the syncytiotrophoblast (40×). (D) GD 85: the cytoplasmic staining of trophoblast has become considerably weaker, while fetal endothelial cells are immunopositive (40×). (E) GD 150: the endothelium of most fetal capillaries is strongly expressing IDO. Inset: representative villus displaying proliferation of few suspected cytotrophoblast cells (arrows; 40×). FAS-L = FAS-ligand; GD = gestational days; IDO = indoleamine 2,3-dioxygenase; NK = natural killer cells.
In the cynomolgus monkey placenta, the FAS-L is already in place by GD 50 (Figure 4B). It is strongly expressed in the cytoplasm of the syncytiotrophoblast and EVT as well as in certain (maternal) immune cells (Frangsmyr et al. 2005; Guleria and Sayegh 2007). The system is coexpressed with S100, whereas expression by no means is similar to HLA-G. In view of the topographic expression of FAS-L, there is sequential agreement with that described in late stage human placenta whereas there is apparently little information about younger placental tissue. FAS-L activity has been tested by the application of Tunel and caspase-3 staining in healthy and unchallenged placenta. Both 50-day-old and 150-day-old placentae show minimal apoptotic activity whereas apoptosis was significantly increased in post abortion placentae (unpublished data).
IDO
IDO is thought to be a very effective immune-modulating/inhibiting enzyme in the human and mouse placenta, which is released from the syncytiotrophoblast into the maternal blood compartment and from decidual macrophages (Baban et al. 2009; Larrea et al. 2007; Lei et al. 2007; Nagamatsu and Schust 2010; Sedlmayr et al. 2002). Upon catalizing tryptophan into N-formylkynurenine, it effectively creates a tryptophan-deficient microenvironment that stops both microorganism growth and cell proliferation by arrest in the G1 phase (Jasperson et al. 2008; Munn et al. 1998). Additionally, an inhibitory effect of IDO was reported on serotonin synthesis. Serotonin is in turn a well-known vasoconstrictive molecule.
In the GD 50 cynomolgus monkey placenta, there was microgranular immune staining in all cells, which is interpreted as nonspecific background staining (Figure 4C). The villous tissue is undergoing a high degree of proliferation as evidenced by increased Ki67 immune staining, which would be unexpected in IDO-derived tryptophan deficiency.
By GD 150 (beginning on about GD 85), nearly all capillary endothelia are IDO-positive, whereas the nonspecific background staining is entirely absent (Figure 4D). Endothelial IDO immunostaining is in good agreement with other studies that indicate immune negative endothelial cells in the first trimester and positive endothelia in the late third trimester in humans (Buttinger 2007; Lei et al. 2007). In view of fetal endothelia expression, IDO possibly plays a role as a targeted antiproliferative/anti-infective molecule, and in view of serotonin inhibition, IDO might play a role in continuous placental vasodilatation.
Discussion
Given the high prevalence of placenta-based gynecologic diseases (Pennington et al. 2012; Redman et al. 2012) and toxicologic challenges, the placenta has to deal with (Stasenko et al. 2010) a continuous need for reliable animal models for human placentation exists. Unfortunately an ideal, universal, model (small species, short reproduction cycle, and most similar in placental structure and physiology) does not exist. In fact, the labyrinthine, hemochorial mouse placenta, commonly used as a model, displays important differences to the villous human placenta (Leiser and Kaufmann 1994) as its interhemal barrier is comprised of 3 trophoblast layers (Enders 1965). Shallow trophoblast invasion and endocrine differences between mice and humans also have to be considered. Yet, the short reproduction cycle of mice renders them beneficial for gene targeting studies (Carter 2007). We suggest that a researcher, choosing his placental animal model based on his study aim, would also take into account animal species with very different, non-hemochorial placentation. If feto (villi)-maternal (crypts) interdigitation is the main criterion, epitheliochorial (Wooding 1992) ruminants could be used as their villi and its blood vessel branch similarly (Leiser et al. 1997) and show the same adaptations (e.g., capillarization) in response to lower oxygen levels (Krebs, Longo, and Leiser 1997) as in fetal villi of heavy smokers where vasoconstrictions were suspected as cause for hypoxia (Pfarrer et al. 1999). During human implantation and placentation, many possible interactions between immune cells and invading HLA class I positive EVTs are feasible (Huppertz et al. 2012). An intense lymphocytic response is also encountered by HLA class I positive equine chorionic girdle trophoblast in the endometrial stroma (Noronha and Antczak 2010). This response is also observed when chorionic girdle trophoblast is ectopically transplanted in nonpregnant mares (de Mestre et al. 2008). In addition, selective horse breeding for homozygosity at the HLA region could promote studies unraveling the role of paternal antigens in maternal immune response. For those reasons, the epitheliochorial horse placenta could also serve as a model for the human (Noronha and Antczak 2010). If many aspects of placental morphology (hemochorial and villous) and immunological background have to be most similar to humans, NHP are the experimental animals of choice. For various ethical reasons and for such based in placental structure (extent and route of interstitial trophoblast invasion, remodeling of uterine arteries; Benirschke and Miller 1982; Carter 2007; Carter and Pijnenborg 2011; Pijnenborg, Vercruysse, and Carter 2010), Old World monkeys like the cynomolgus monkey are commonly used as animal models in toxicological research. Morphological analysis of the cynomolgus placenta demonstrates relatively few, but nonetheless probably meaningful differences, to the human placenta like a less prominent decidual response and an altered pattern of trophoblast–decidual interdigitation (De Rijk and van Esch 2008; Kaufmann 1990). Undisputed structural differences are also observed in comparison to rodents and even with other NHP species, for example, the mamorset (Mellor and Munn 1999).
The mature cynomolgus placenta (GD 150) represents a late stage of gestation and is by no means representative for the whole gestation period as far as the development of villous blood vessels is concerned. Undifferentiated GD 50 villi show a lack of capillarization while differentiated GD 150 villi possess numerous blood vessels. It is clear that putative toxic agents within the intervillous space could exhibit very different transfer from the fetal mesenchyme into other parts of the fetus. As this decision of applying substances on GD 50 or GD 150 can be of effect, toxicologist should be knowledgeable that the mature cynomolgus placenta (GD 150) represents a late stage of gestation and is by no means representative for the whole gestation period.
In our study, most tolerance molecules (HLA-G, IDO, and FAS-L) are detectable in their final position in mid-(GD 100) and late gestation (GD 150) but not at early stages of placental development (GD 50). This also argues for a consideration of the profound structural and immunological placental differences between the phases of placental development used (GD 50–150). In regard to expression of tolerance molecules, for example, IDO, we were able to detect IDO exclusively in the endothelium of fetal villous capillaries and in an increasing pattern to the end of gestation. This corresponds to human studies that have spotted IDO exclusively in the endothelium of blood vessels in fetal villi (terminal- and stem-villi and chorionic plate) but not in trophoblast (Blaschitz et al. 2011). Yet, it has to be mentioned that other studies of human placental tissue have detected IDO in trophoblast populations (Honig et al. 2004).
From the findings of our study, we have prepared a summarizing scheme of the fetomaternal tolerance molecules at the contact of fetal villi and maternal blood in the cynomolgus monkey (Figure 5A and B).
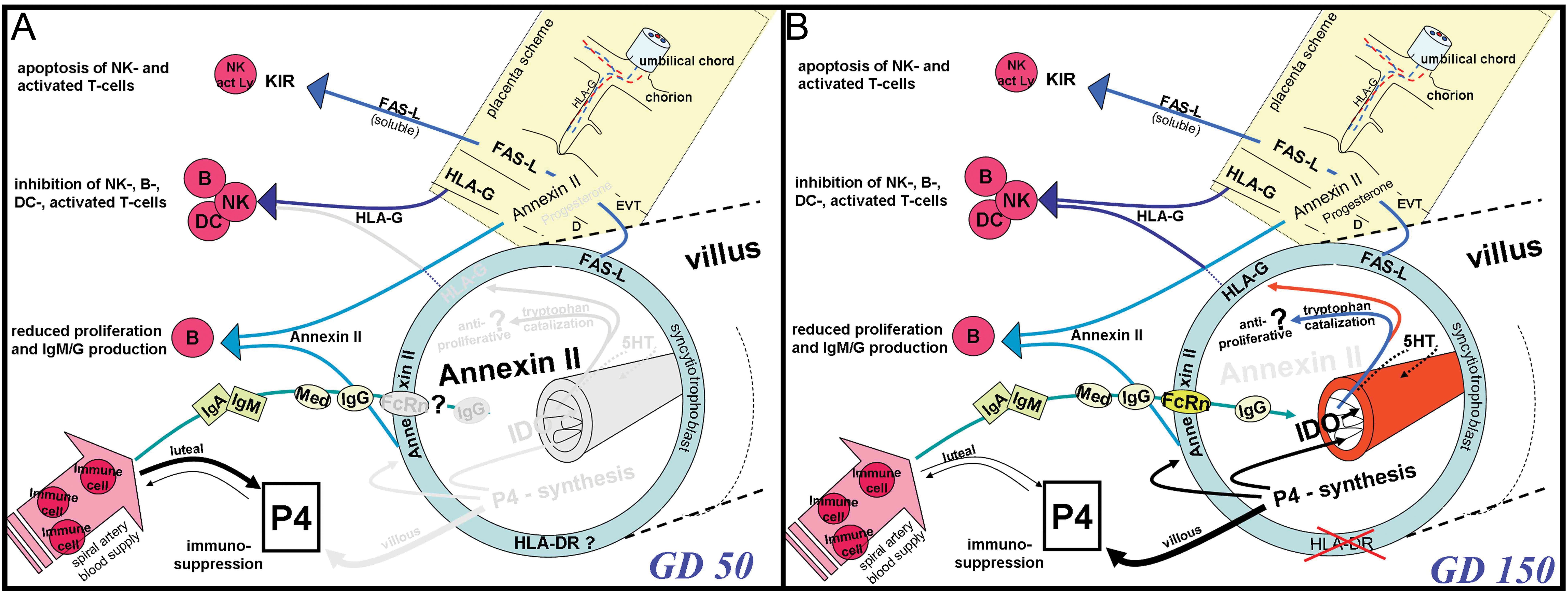
Summarizing scheme of the expression of immune tolerance mediating molecules in the cynomolgus monkey placenta on GD 50 (A) and GD 150 (B). The legend points out the differences between scheme (A) and (B). Exclusive morphologic detail shown is the lack of vascularization on GD 50, whereas lack of immune cells on GD 50 contrasting villous stroma population on GD 150 is not included. The GD 50 progesterone (P4) is of luteal but not of villous origin, whereas a vast surplus derives from villous mesenchyme, extravillous trophoblast (EVT), and decidua (D) on GD 150. The arrows indicate the different progesterone actions targeting to fetal adrenals, annexin II, and mymometrium. Annexin II is expressed on GD 50 in decidua, EVT, and the syncytiotrophoblast with high prevalence on the brush border membrane. Additionally, and different from GD 150, there is strong and intense expression in the villous mesenchyme. HLA-G is expressed on decidua cells on GD 50 but not in any other trophoblast tissue. On GD 150, it is additionally expressed in the syncytiotrophoblast. HLA-DR displays an unclear stain on GD 50 (see ?), which is most prominent in the syncytiotrophoblast. In advanced development (GD 150), the HLA-DR is downregulated. The FAS-L displays similar trophoblast expression on GD 50 and GD 150. From GD 85 to GD 150, the blood vessels are well developed (B) and IDO is expressed in vascular endothelial cells. Stimulating activity is postulated on syncitial HLA-G (red arrow), whereas further activities are hypothetical (blue arrow for tryptophan deficient fluid; black arrow—fetus directed action; dotted arrow: 5HT—mediating action. HLA = human leukocyte antigen. FAS-L = FAS-ligand; GD = gestational days; IDO = indoleamine 2,3-dioxygenase.
Finally, the immune cell population in the villi (not yet published) roughly coincides with the immune system development in the fetal cynomolgus monkey in which macrophages have been described from post embryonic day 40 to 50 and peripheral T-lymphocytes and B-cells from GD 70 to GD 80 on (Buse 2005). In view of controversial interspecies descriptions concerning cellular expression of tolerance molecules, a conclusive interpretation of the mechanisms of immunological tolerance in cynomolgus monkeys needs more detailed analysis and thus must challenge toxicologists for more, rather than less, attention in the near future.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
Acknowledgments
The authors gratefully acknowledge the skilled assistance of the technical staff at Covance Laboratories GmbH in Münster, Germany, and at the Department of Anatomy, University of Veterinary Medicine Hannover, Hannover, Germany.
