Abstract
Subacute inhalation study (1 week or 2 weeks) is an important process for screening out inhaled compounds causing lung irritation. To investigate whether the lung weight can be used as an indicator for acute lung injury, we have analyzed retrospectively the lung weight data from 30 studies in rats exposed to dry powder inhalation. The lung weight change was correlated with lung histopathology in the majority of studies (25 of 30), showing as either both changed or both unchanged. The sensitivity and specificity of using the weight change in lungs as biomarker for predicting lung histopathology in these studies were over 80%. The pattern of lung weight change was often similar in the 1- to 2- week studies and the 4-week studies. Our analysis of covariance model showed that a study with 40 rats (5 males + 5 females/group and 4 groups) could detect lung weight change greater than 10% to 20% of control value. These results suggested that lung weight is a useful indicator for identifying acute lung toxic effect by inhaled compounds in these subacute inhalation studies.
Introduction
For inhaled drugs to reach clinical trials and market, it is mandatory to test the drug candidates in toxicological studies using the intended clinical administrative route. Such toxicological tests are usually done through inhalation studies using the rodent species rats and the nonrodent species dog. Within AstraZeneca (AZ), single dose of dry powder inhalation or intratracheal administration is often applied as a first step in testing a broader selection of chemistry leads. The next inhalation study is normally a subacute (1 week or 2 weeks) rat inhalation study. It is carried out to determine the suitable dose ranges and to further screen out compounds with potent irritant properties, which could cause local adverse effects in the respiratory system at relevant clinical doses. In these studies, histopathology is usually the most vital measure of adverse effects, although the lung weight data are also collected.
Organ weight in general has been accepted as a valuable readout in toxicological studies (Bailey, Zidell, and Perry 2004; Michael et al. 2007). According to the recommendation by the Society of Toxicologic Pathology (STP), many organs, including the liver, heart, kidneys, brain, testes, and adrenal glands, should be weighed in all species used in good laboratory practice (GLP) toxicology studies; while certain organs, such as the lung, should be weighed on a case-by-case basis rather than routinely assessed in every inhalation study (Sellers et al. 2007). Despite this recommendation, lung weight data are often collected in the inhalation studies, but a comprehensive review of lung weight as one assessment of lung toxicity in inhalation studies is still lacking.
In our daily practice, we have noticed that lung weight changes were often but not always associated with lung histopathology, as observed by others (Michael et al. 2007). The variations in lung weights, together with relatively small group sizes, made it difficult to interpret the lung weight changes occurring in inhalation studies, particularly in the 1- to 2-week studies in rat. Further literature searching did not help us find sufficient information discussing the application of lung weight as a biomarker of lung toxicity and its correlation with lung histopathology. We therefore collected lung weight data from 30 inhalation studies with AZ compounds performed in recent years and analyzed the data using different approaches to evaluate lung weight change as an independent end point, using their relation to lung histopathology as a comparator.
We analyzed the relationships of lung to body weight, and lung to brain weight, to determine an appropriate indicator for the lung assessment of adverse changes. We calculated the minimal group size for such studies for detecting 5%, 10%, and 20% difference in lung weight change. We further compared the lung weight change between 1- to 2-week and 4-week studies with the same test compound to explore the predictivity of lung weight change.
Materials and Methods
Thirty rat inhalation studies with test compounds from different pharmacological and chemical classes administered in dry powder formulation were included in the analyses. The majority of the studies were the early non-GLP studies with a 1-week or 2-week exposure time; the minority were GLP studies with a 4-week exposure time using the same test compounds as in the previous group. All studies included were performed at AZ Safety Assessment Södertälje during the years 2005 to 2009.
Animals and Dose Groups
Within the laboratory, the animals were housed in transparent plastic cages. The bedding material consisted of aspen wood chips. Plastic tunnels and aspen chew blocks were provided. The animals had free access to pelleted RM1 (E) SQC diet purchased from Special Diets Services, Ltd., England, and water from the site drinking water supply. The animal rooms were illuminated by artificial light from fluorescent tubes on a 12-hr light/dark cycle. The temperature and relative humidity were about 17°C to 23°C and 30% to 80%, respectively. All experiments were approved by the regional ethical committee and conducted in compliance with national legislation and guidelines for the care and use of laboratory animals.
Typically the studies had 4 groups, that is, control/air, low-dose, intermediate-dose, and high-dose groups, with equal animal numbers in both genders. There were 40 rats (5 males and 5 females/group) for the 1- to 2-week studies, and 80 (10 males and 10 females/group) for the 4-week studies. The animals were acclimated to the inhalation exposure conditions by placing them in the exposure chamber for 3 days before dosing. They were dosed by dry powder inhalation once daily for 1 or 2 weeks or for 4 weeks and were terminated for scheduled necropsy 1 day after the final dose.
Dry Powder Formulation
For all inhalation studies, the test compound was formulated as a micronized dry powder containing particles. The test compound was compressed to a powder cake and a dry particle aerosol was produced using a modified Wright Dust Feeder mechanism (L. Adams Ltd., London, UK). The aerosol generation and the animal exposure were operated via a computerized inhalation system (FIBA, AstraZeneca R&D, Södertälje, Sweden) together with nose-only “flow-past” exposure chambers (Münster AG, Switzerland).
The particle size distribution of the aerosols was determined using a 7-stage Mercer cascade impactor (model 02 110, In-tox Products, USA) and determined by high-performance liquid chromatograpgy (HPLC). The mass median aerodynamic diameter (MMAD) and geometric standard deviation of the aerosol were derived for each occasion of measurement. The results from the particle size samplings indicated essentially log-normal distributions on the majority of samplings occasions. The MMAD values for these studies were within the range of 1.2 µm to 3.3 µm (except for one high dose group which was 0.8 µm, one vehicle group which was 3.9 µm), indicating that the aerosols were highly respirable to the rat. The sizes are in ideal range and can reach the alveoli of the lungs (Anjilvel and Asgharian 1995; Darquenne 2012) as many other inhaled drugs (Sutherland and Martin 2005).
Necropsy and Tissue Processing
The necropsy and tissue processing were performed according to the AstraZeneca’s standard operating procedures (SOPs) in a standardized fashion. In brief, the rats were terminated by exsanguinations from common carotid arteries under isoflurane and N2O anesthesia. The organs were examined macroscopically and collected according to the study plans. For the 1- to 2-week studies the organ list usually included the core organs (heart, liver, spleen and kidneys), respiratory organs (nose, larynx, trachea, carina, and lung), and other possible target organs depending on previous experience with the test substances. Many of the organs were weighed as well. For the 4-week studies, all organs (about 40 organs in each animal) were collected and weighed (many of them). All collected organs were sliced and fixed in buffered formalin, and further processed to paraffin-embedded hematoxylin & eosin (H&E) sections for microscopic examination.
After opening of thoracic cavity and removing the heart, the thymus, and surrounding fat and vessel tissues, the trachea was separated from the esophagus and then was cut at 1.5 cm above the bronchial branch. The lower trachea was gently lifted together with connected lung lobes and removed out of the thoracic cavity. After being weighed with an adjusted weighing scale, the lung was inflated manually with 10% buffered formalin using a 5-ml syringe inserted in the trachea. After 48-hr fixation, the lung was trimmed and 5 slices (1 from each lobe) were processed further and embedded into one paraffin block together. The sections (3µm) were stained with H&E for microscopic examination.
Lung Histopathology Evaluation and Terminology
The H&E slides from each study were examined microscopically by a study pathologist. Morphologic alterations were diagnosed and recorded, using a four-grade severity scores (
As the studies were examined by different pathologists, several diagnostic terms might have been applied for similar histopathologic alterations. The terms for common histopathologic alterations in these studies were standardized in accordance with the standardized nomenclature of the respiratory tract lesions (Renne et al. 2009). The term perivascular inflammation covered all similar morphologic changes as perivascular inflammation, perivascular infiltration, and perivascular neutrophils; and the term macrophages covered increase in alveolar macrophages, histiocytosis, alveolar histiocytosis, macrophage cluster, and alveolar foamy cells. The term inflammation covered all pulmonary inflammatory lesions, including perivascular inflammation, macrophages, alveolitis, pneumonitis, and pneumonia.
Analysis of Lung Weight in Relationship to Body Weight and Brain Weight in Control Rats from All Studies
To evaluate the linear relationship between lung weight and body/brain weight, lung weights of all animals from control groups in the studies were analyzed using the simple regression model as
Comparison of Mean Lung Weights with Analysis of Covariance
For each study, lung weights were log transformed and fitted to an analysis of covariance model (ANCOVA) with log transformed body weights as covariate and treatment group as a fixed factor. Least squares means (LSmeans) of lung weights from each treated group were compared against the corresponding control group using Dunnett’s
If the interaction term, that is Treatment × Body Weight, was not found to be significant, it suggested the treatment did not affect the relationship between body and lung weights. Therefore, the lung weights adjusted of body weights were calculated as the residual after subtraction of the ANCOVA regression line from the original lung weight values and were used for correlation analysis against histopathologic findings with Spearman’s rank correlation coefficient and for plotting purposes.
For those studies which showed that treatment significantly affected the relationship between body and lung weights (where the interaction was significant), an ANCOVA model with brain weight as the covariate was fitted for the males and female separately.
Power Analysis for Detection of Mean Lung Weight Changes in Treated Groups
To determine the minimal group size for detecting a real difference in lung weight change, the following approaches were applied (Gad and Rousseaux 2002; Shirley 1977). (1) ANCOVA, using treatment as the fixed factor and body weight as the covariate, analyzed on both genders simultaneously; (2) ANCOVA, using treatment as the fixed factor and either body or brain weight as the covariate, analyzed on male and female rats separately; (3) ANOVA, using lung weight and body or brain weight ratios with treatment as a fixed factor, analyzed for male and female rats separately.
Power was calculated using the Glmpower procedure in SAS 9.1 (SAS Institute, Cary, NC) to compare the control group with the three treated groups for a deviation from the mean of 5% (1.05-fold), 10% (1.1-fold), and 20% (1.2-fold). Sample sizes were assumed to be equal for all treatment groups. Standard deviations were calculated from the root mean square error of an ANOVA model of the lung weights from control animals with the individual studies as the fixed factor. The correlation between the lung weight and the covariate (body or brain weights) was estimated with Pearson’s correlation coefficient (Quinn and Keough 2002). Total numbers of animals was calculated for a power of 0.8 using significance level α = .05. The results were reported with a Bonferroni correction (αsingle test = .0167).
Correlation of Lung Weight Changes and Histopathologic Alterations in the Lung
To investigate whether the lung weight changes can be an indicator for lung histopathology, the sensitivity and specificity of the adjusted lung weight change for histopathologic alterations were calculated (Altman and Bland 1994). Sensitivity is the proportion of true positives that were correctly identified by the test, and specificity is the proportion of true negatives that were correctly identified by the test. With the definition of “true” being a lung weight change correlating with lung histopathology or an absence of lung weight change correlating with nonappearance of lung histopathology.
Results
Correlation of Lung Weight Change with Lung Histopathology
All studies were grouped into four categories according to their histopathology and weight responses to the treatment (Table 1). The first group included 12 of 30 studies, which had both treatment-related lung weight change and lung histopathology. The second group (2 of 30 studies) had no lung weight change but lung histopathology. The third group (13 of 30 studies) had neither lung weight change nor lung histopathology. The last group (3 of 30 studies) had lung weight change but no lung histopathology. These results indicated that the lung weight change and lung histopathology were concurrent in the majority of studies (25 of 30), showing as either both changed or both unchanged. The lung weight change and lung histopathology were not agreed in the minority of studies (5 of 30), showing as one of them changed while the other was unchanged.
Correlation of lung weight change with lung histopathology.
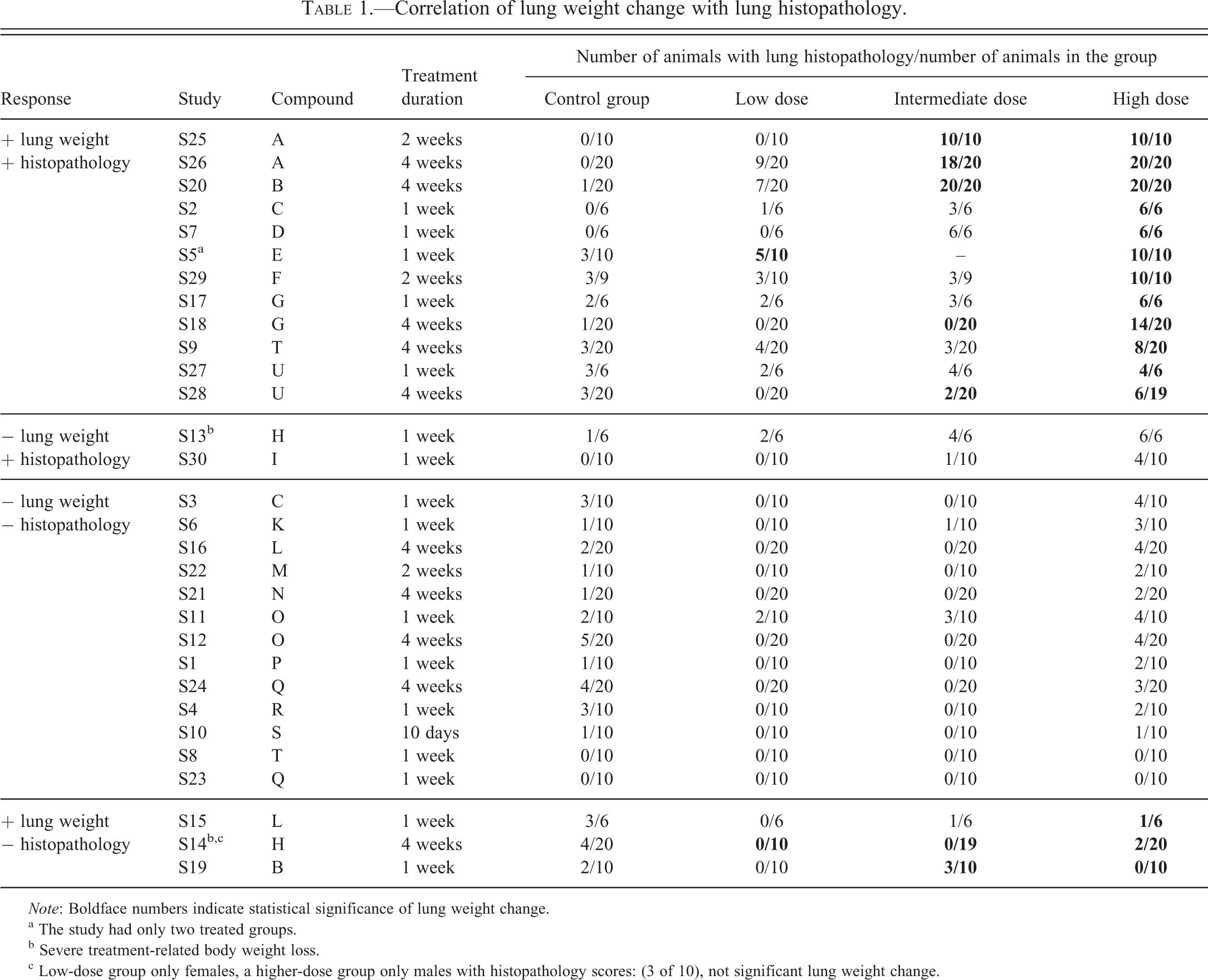
a The study had only two treated groups.
b Severe treatment-related body weight loss.
c Low-dose group only females, a higher-dose group only males with histopathology scores: (3 of 10), not significant lung weight change.
The relationship of lung weight change to lung histopathology was further analyzed in individual animals as shown in Figure 1. The box plots displayed all individual lung weights (Y-axis) in relation to different lung histopathologic alterations (X-axis). The central mark of each box represented the median, the edges of the box shows the 25th and 75th percentile. The central line extended to the extreme values, and individual circles were outliers that extended more than 1.5 times of the interquartile range (the size of the box) from either edge of the box. The amount of outliers was proportional to the group size.
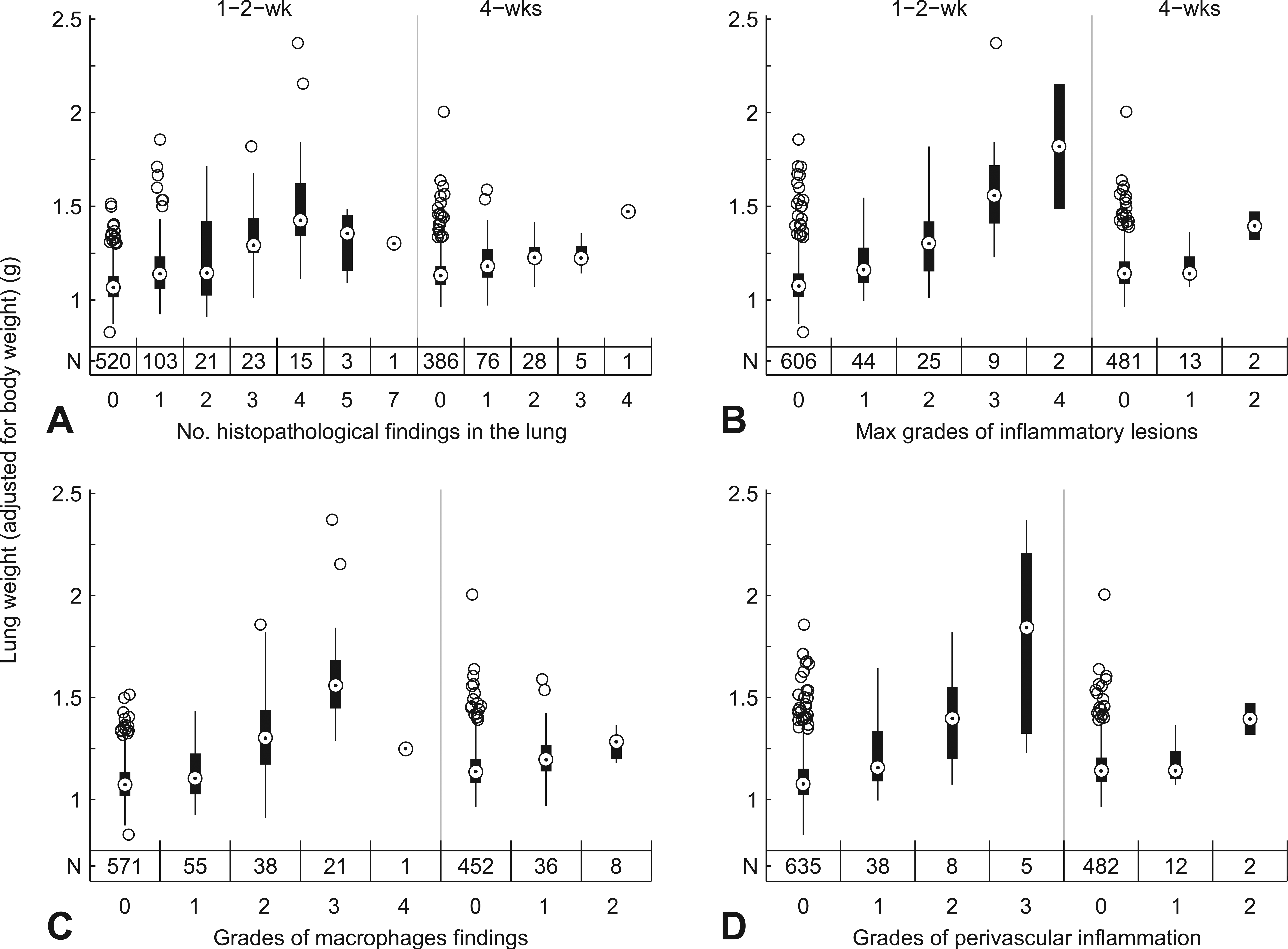
Box plots show the adjusted lung weights (Y-axis) and different types of morphological lesions (X-axis) from all of individual animals in the studies. The number of animals included in the analyses is presented at the bottom of each plot (N). These results indicated that the lung weight increase was associated with the total number of lung histopathologic observations (A), severity of any inflammatory lesions (B), grade of macrophage changes (C), and grade of perivascular inflammation (D) in both 1- to 2-week studies and 4-week studies.
Figure 1A demonstrates the relationship of the lung weight to the amount of histopathologic findings, confirming that the more events (different histopathologic alterations) present in the lung, the greater is the increase in lung weight, when there were sufficient individuals in the group. Figure 1B demonstrates the lung weight changes related to the maximal grades (severity) of any inflammatory lesions. The two most common lung alterations in early inhalation studies, macrophages (Figure 1C), and perivascular inflammation (Figure 1D) were correlated with lung weight change in both the 1- to 2-week and the 4-week inhalation studies. Spearman’s correlation coefficients were calculated between the adjusted lung weights and the respective grading and found significant (
The calculated sensitivity and specificity of using lung weight change as a marker for histopathologic change in the lung are shown in Figure 2. The sensitivity of lung weight change as a marker for histopathologic changes was 86% in this investigation, indicating that in the majority of cases the lung weight predicted the presence of histopathology. There were 3 of 30 studies that had identified lung weight change but showed no appreciable lung histopathology on the examined sections. The specificity of lung weight as a marker of lung histopathology was 81%. This relative high presentation suggested that if there was no relative lung weight change, the likelihood of finding histopathologic changes in the lung should be low.
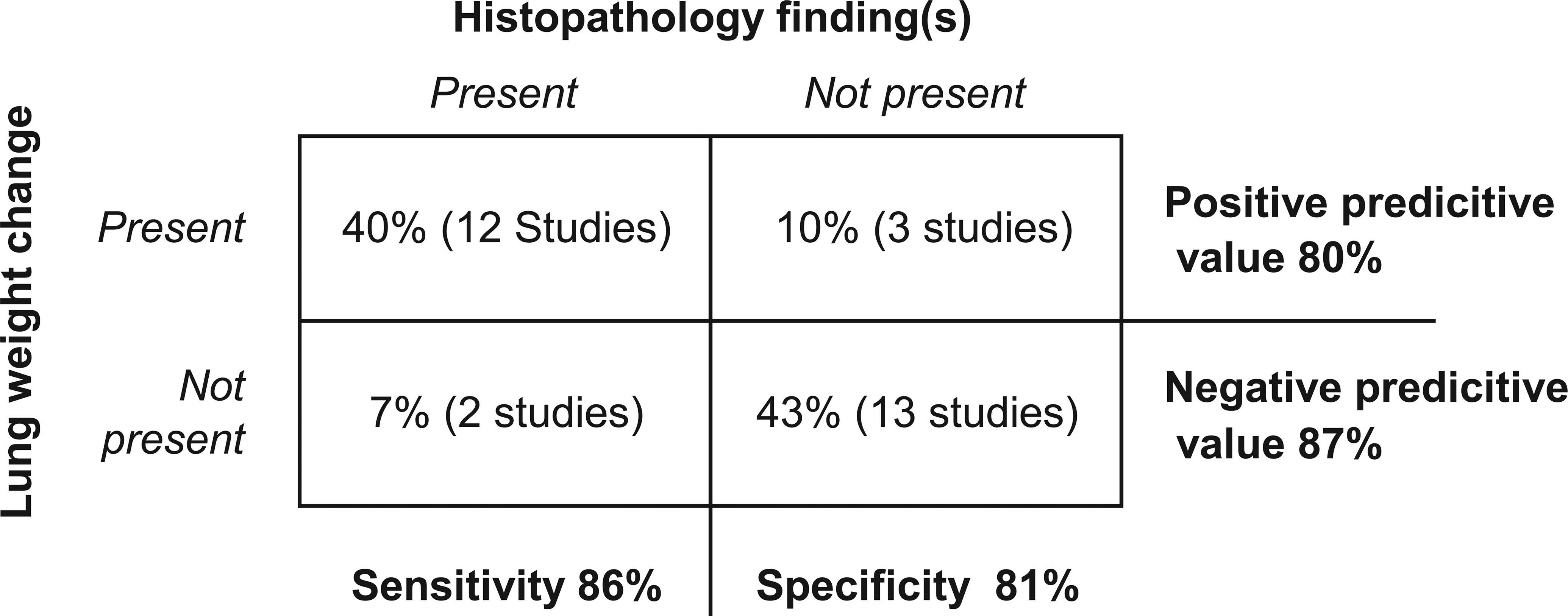
Sensitivity and specificity of using lung weight change as biomarker for predicting lung histopathology in the dry powder inhalation studies were calculated. The sensitivity was 86% indicating that the lung weight change predicted lung histopathology in most cases. The specificity of lung weight as a biomarker for lung histopathology was 81%, suggesting that if there was no lung weight change the likelihood of having lung histopathology is lower.
Relationship of Lung versus Body Weight and Lung versus Brain Weight
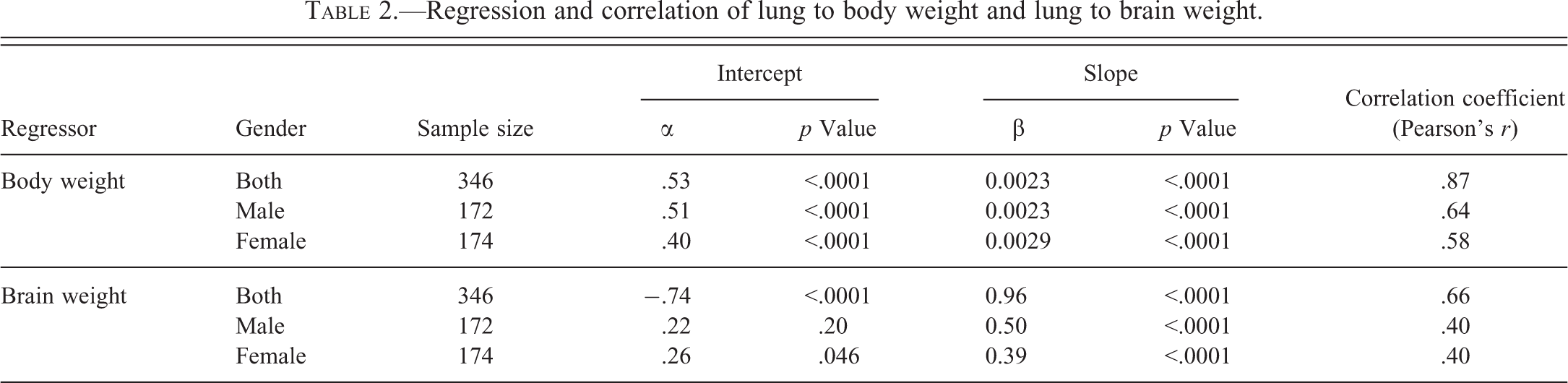
The linear relationships of lung weight to body weight, and lung weight to brain weight, were analyzed using regression and correlation analysis (Table 2). All regressions showed a linear relationship by statistically significant slopes β, but the linear relationship were nonproportional as shown by statistically significant intercepts α, except male group for lung to brain regression, rendering the lung and brain weight relationship somewhat ambiguous. Higher correlation coefficients,
Regression and correlation of lung to body weight and lung to brain weight.
The relationships of log transformed organ weights were further analyzed using scatter plots and regression analysis (Figure 3). Scatter plots with linear regression lines of lung weight versus body weight are shown in the Figure 3A and lung weight versus brain weight are shown in Figure 3B. The lines represent separate regression models for both genders (solid lines), for male subjects only (dashed lines), and for female subjects only (dotted lines). The relationship of lung to body weight was similar for both males and females, while the relationship of lung to brain weight was clearly different between the two genders.
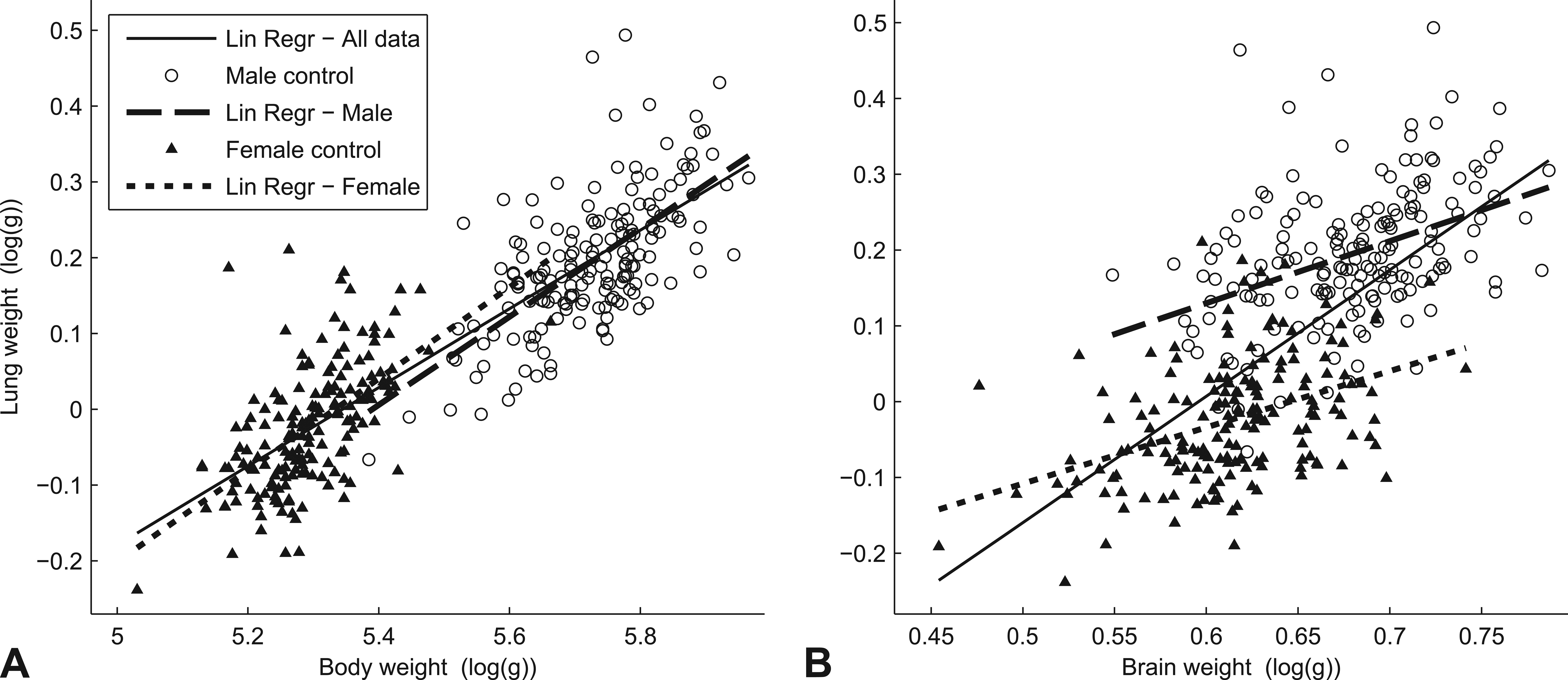
Scatter plots with linear regression lines of log transformed organ weights. Lung weight versus body weight (A) and lung weight versus brain weight (B). The solid line represents the regression for mixed subjects, the dashed line for male subjects, and the dotted line for female subjects. The relationship of lung to body weight was similar between males and females (A), while the relationship of lung to brain weight was more divergent between genders (B).
Comparison of Lung Weight Changes in the 1- to 2-week and the 4-week Studies
Among the 30 studies, there were 10 test compounds including data from both the 1- to 2-week study and the 4-week study. The results in Table 3 represented the mean fold change (MFC) of the adjusted lung weight in the inhalation studies (S) with different test compounds (A to U). Several patterns of lung weight change were found when the data were compared from the 1- to 2-week and the 4-week studies. There were significant lung weight change both in the 1- to 2-week and 4-week studies (compounds A, B, G, U; 4/10), no lung weight change in both durations (compounds N, O, Q; 3/10), change only in the 1- to 2-week study (compound L; 1/12), and change only in the 4-week study (compound T; 1/12). Of the 10 compounds, 7 showed similar results in lung weight changes from the 1- to 2-week and the 4-week studies. Results for compound H are difficult to interpret since the lung weight results of study S14 were hard to verify due to severe systemic body weight loss.
Treatment durations and lung weight changes.
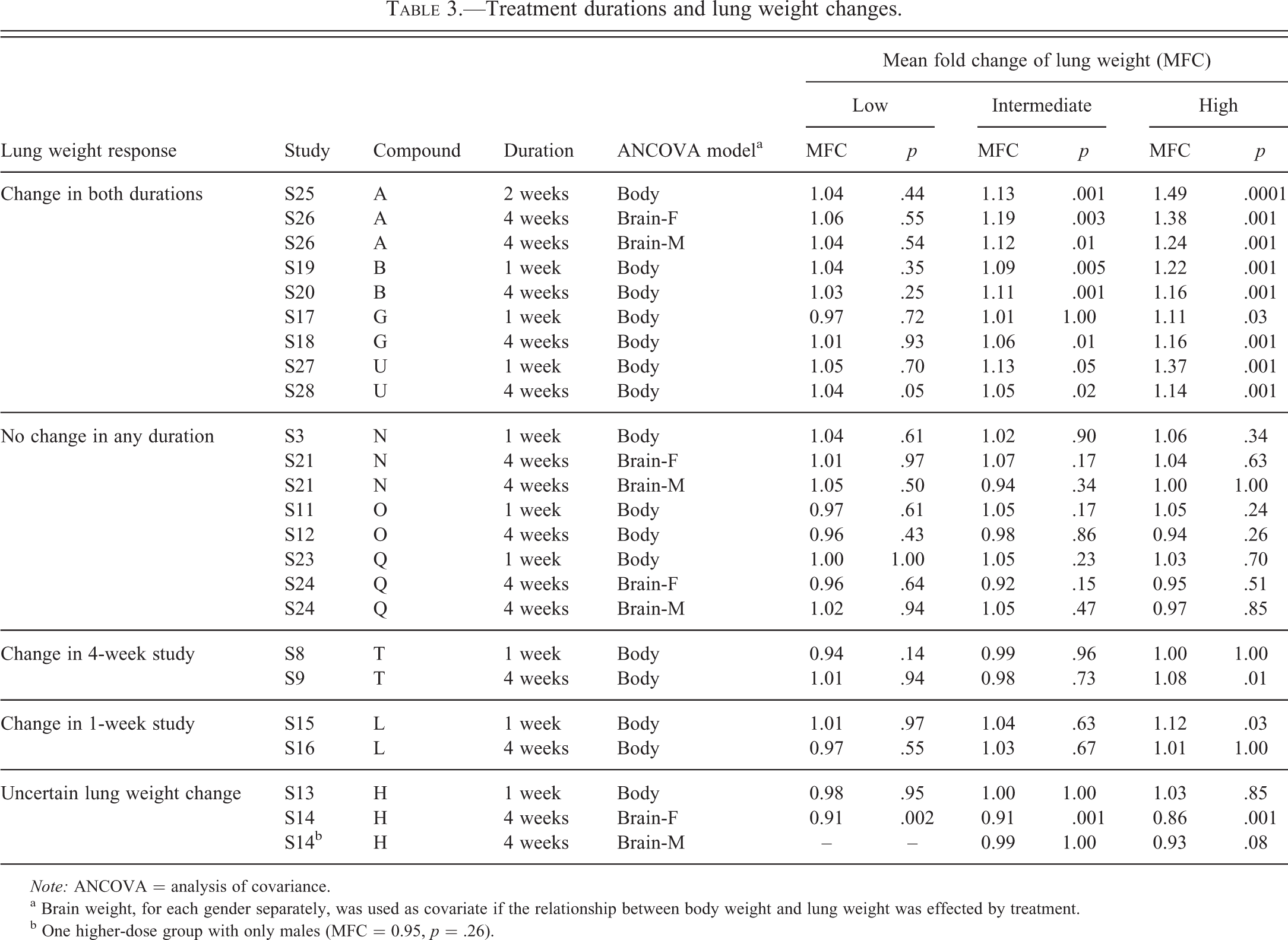
a Brain weight, for each gender separately, was used as covariate if the relationship between body weight and lung weight was effected by treatment.
b One higher-dose group with only males (MFC = 0.95,
Lung Weight Increase versus Lung Histopathology in 1- to 2-week and 4-week Studies
Lung weight and lung histopathology were compared between the 1- to 2-week studies and the 4-week studies as shown in Figure 4.
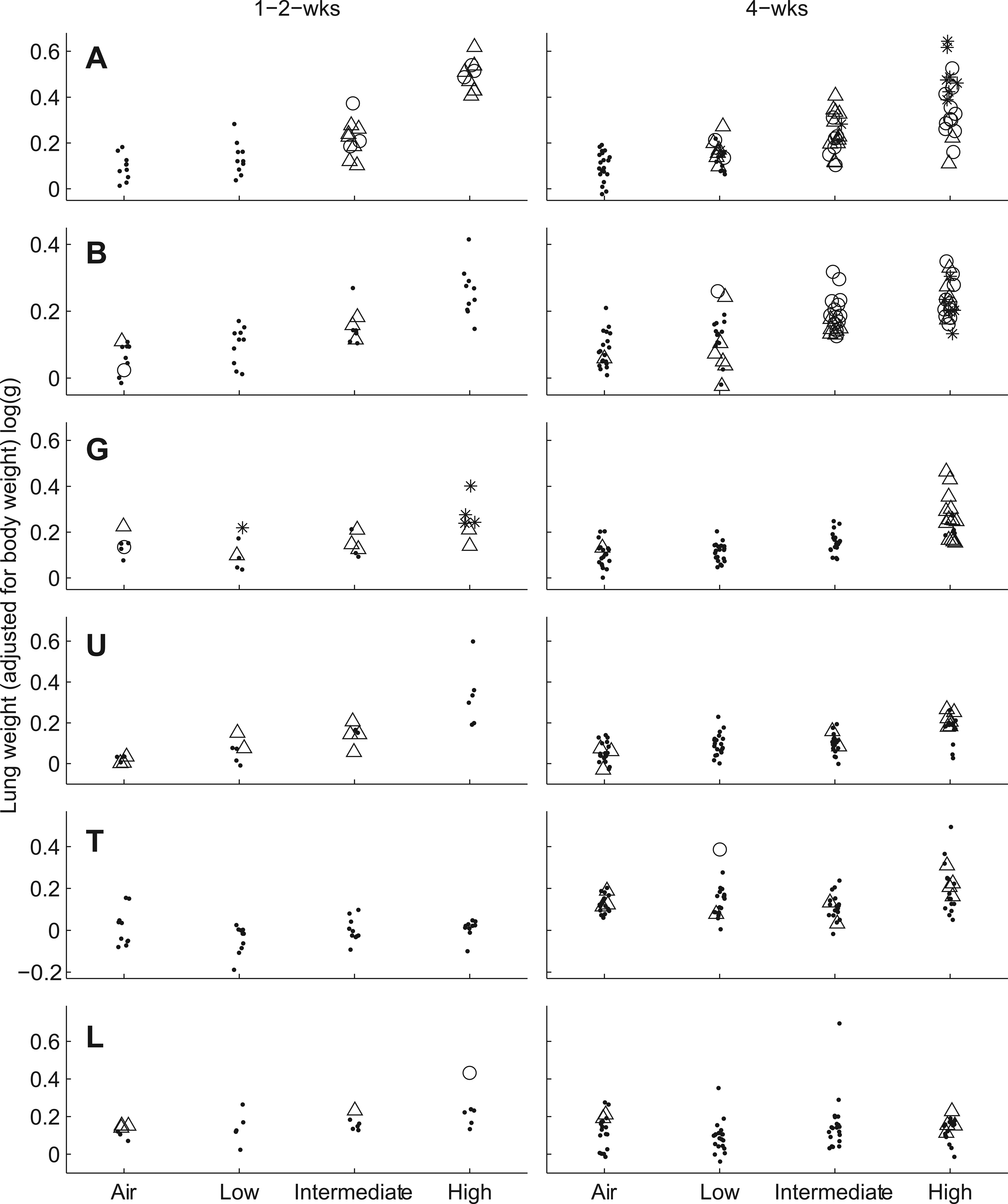
Body weight adjusted lung weights from the 1- to 2-week and the 4-week studies with compounds A, B, G, U, T, and L. Doses are indicated on the X-axis and the log-transformed lung weights (adjusted for body weights) on the Y-axis. Each marker represents one animal, shape of the markers indicates different number of histopathologic findings in the lung for each animal, (•) – no effects, (▵) – one effect, (^) – two effects, and (⋆) – three or more effects.
Compound A (S25 and S26 in Table 3) had dose-related lung weight increases in both the 1- to 2-week and in the 4-week studies, for example the MFC of lung weight was 1.15 (1- to 2-week) and 1.13 (4-week) in the intermediate-dose group and 1.49 (1- to 2-week) and 1.31 (4-week) in the high-dose group. Similar degrees of lung weight change were seen in studies where compound B was administered (S19 and S20), 1.09 (1- to 2-week) and 1.11 (4-week) in the intermediate-dose group and 1.22 (1- to 2-week) and 1.16 (4-week) in the high-dose group. Interestingly, the similar pattern of lung weight change was not as concordant with degree of lung histopathology observed in the two studies. The incidence of lung histopathologic alterations appeared higher in the 4-week studies than that in the 1- to 2-week studies.
Compound G (S17 and S18) and compound U (S27 and S28) showed similar pattern of lung weight change in both the 1- to 2-week and the 4-week studies. Again, lung histopathology was missing in the 1- to 2-week study as evidently showing the high-dose group of compound U study.
For compound L (S15 and S16) and compound T (S8 and S9) showed similar negative changes in lung weight and lung histopathology in both the 1-week and the 4-week studies.
Minimal Group Size for Detecting 5%, 10%, and 20% of Lung Weight Change
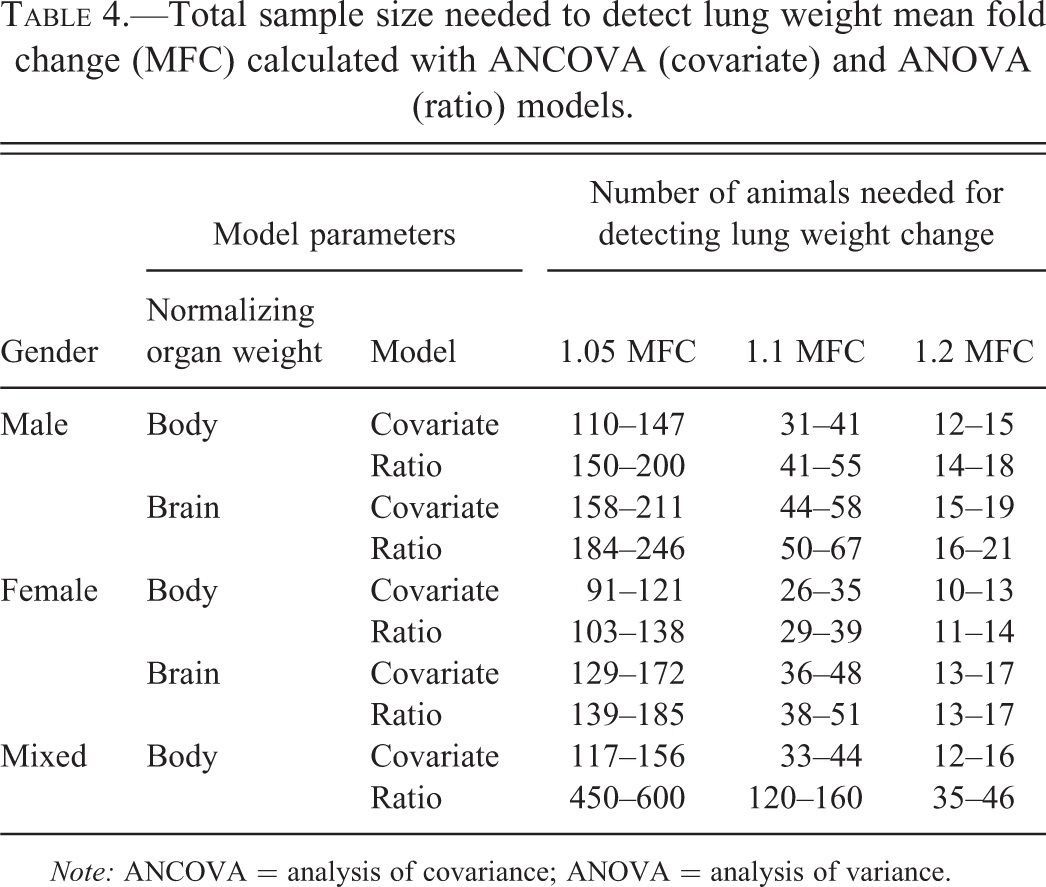
For detecting three effect levels of lung weight change, that is 5% (1.05 MFC), 10% (1.1 MFC), and 20% (1.2 MFC), the minimal total animal number in a study was calculated with a power of 0.8. That is, for the specific fold changes, the estimated standard deviations, and the selected significance level, the probability of detecting a change in lung weight should be at least 0.8 (Quinn and Keough 2002). Power calculations were conducted for a significance level of .05 and .0167, which corresponds to a Bonferroni correction for multiples comparisons.
As shown in Table 4, when body weight was used as the covariate, the total number of animals (all groups together) was 33 to 44 animals for detecting a 1.1 fold (10%) lung weight change in one study. If the genders were separated, 31 to 41 male or 26 to 35 female animals were needed for detecting a similar degree (10%) of lung weight change. When brain weight was used as the covariate, the power calculations had to be conducted with genders separated, as males and females had different linear relationship between lung weight and brain weight (Figure 3). Using brain weight as covariate a total number of 44 to 58 for male animals or 36 to 48 for female animals were needed to detect a 1.1 fold (10%) change when lung to brain weight was used. Power calculations for ANOVA on ratios are included in Table 4 for comparison.
Total sample size needed to detect lung weight mean fold change (MFC) calculated with ANCOVA (covariate) and ANOVA (ratio) models.
Discussion
An international survey by STP of current practices in organ weight evaluation in toxicity studies showed that many organizations including pharmaceutical, agrochemical, veterinary, chemical, food/nutritional, and consumer product industries believed that lung weight was a useful indicator particularly in inhalation studies. Some of them pointed out that lung weights were often correlated with histopathologic changes related to toxicity or enzyme induction (Michael et al. 2007). However, 20% of pharmaceutical industry respondents considered lung weight less useful due to various reasons, for example, a lower frequency of finding weight changes that correlated with toxicity and less sensitivity to predict toxicity compared to histopathology.
In this study, we demonstrated a good coincidence of lung weight change and lung histopathology in the majority of dry powder inhalation studies in rat. The lung weight change, mostly increasing, was proportional with the incidents of histopathologic findings (events) and with the maximal grades of all inflammatory lesions, the grades of alveolar macrophages, or the grades of perivascualar inflammation (severity). The sensitivity of using lung weight change as biomarker for predicting lung histopathology in these studies was 86%, indicating that the lung weight is a sensitive marker for lung histopathology.
However, we also noticed a few studies that had a miscorrelation of lung weight change and lung histopathology, showing as either only lung histopathology (2 of 30 studies) or only lung weight changes (3 of 30 studies). The discrepancy between lung weight change and lung histopathology could be related to the limitations of these two methods.
Lung weight change is an objective observation, in contrast to a histopathology assessment, and is not affected by the experience level of the pathologist. But lung weight change is relatively less sensitive to detect mild or focal lesions. The individual and group variations of lung weight may cover up mild lung weight change, especially when number of animals in each group is small. As the calculation showed that, the minimal detection level for a lung weight change must be greater than 10 to 20% of the control value in the 1- to 2-week inhalation studies. In addition, if the test compound had strong effect on body weight and/or multiple organ weights, the lung weight loss became part of systemic rather than lung specific effect. In such case, as study S14, the lung weight data should be interpreted with caution.
Pulmonary responses in the inhalation studies could be related to chemical features, pharmacological effects, toxicological effects, or the particle accumulation in the lung (David et al. 2006; Jones and Neef 2012). Histopathologic evaluation in such studies is based upon microscopic examination on H&E slides of five sections in each animal. Very focal and mild lesions might be not present in the examined sections and or not be easily detected by pathologists. Moreover, some less characteristic lesions as edematous changes are not easily detected on a routine H&E section, especially when it is of low grade and diffuse. Such changes can be seen in the early phase of lung toxicity (Dungworth, Hahn, and Nikula 1995) and may cause abnormalities after resolution of the edematous process (Witschi et al. 2008). For that reason, lung wet weight and dry weight have been introduced in many investigative studies for detecting acute pulmonary toxicities caused by phosgene (Currie, Hatch, and Frosolono 1987; Pauluhn 2006), bleomycin (Yamauchi et al. 2011), and ozone (Prows et al. 1999). Taken together, both lung histopathology and lung weight changes have pros and cons, they should be applied as complementary methods for assessing lung toxicity in the inhalation studies.
Individual variation in organ weights is typically normalized by body weight, expressing as ratio, organ weight as a fraction percentage of body weight (Gad and Rousseaux 2002). In many cases, the body weights are affected by treatment of the test compound and therefore organ-to-brain weight ratio is frequently applied in toxicological studies as well (Michael et al. 2007; Sellers et al. 2007). We analyzed the relationship between lung weight to body weight and lung to brain weight, in order to choose an appropriate adjusted lung weight for studying lung weight changes. To use a ratio as a proper measurement, the relationship between weights should be proportional, meaning that the regression line should pass through zero, so an increase in one of the weights should be followed by increase in the other weight proportionally (Bailey, Zidell, and Perry 2004). We conducted regression analysis on control rats from all 30 studies and found that the lung-to-body-weight relationship was not proportional, evident by significantly nonzero slope and intercept parameters for both genders (Table 2). This indicates that ANCOVA is the better alternative (Bailey, Zidell, and Perry 2004; Jarvis et al. 2011). The lung-to-brain-weight relationship appeared differently in the male and female, that is slope and intercept were significant in females but only the slope parameter was significant in males. One could argue that lung-to-brain weight ratio might be an acceptable end point for males only. Due to the ambiguity of the lung-to-brain-weight relationship, and the fact that lung weight correlates substantially stronger with body weight than with brain weight, ANCOVA analysis model with body weight as covariate was considered more proper for analysis of lung weight changes.
The ANCOVA method was early suggested for measuring organ weights (Angervall and Carlström 1963) and was shown to be superior to the analysis of variance on ratios (Shirley 1977; Shirley and Newnham 1984). In a recent assessment of statistical methods, ANCOVA was recommended to use for measuring organ weights (Jarvis et al. 2011). However, care must be taken to avoid violation of the underlying assumptions of ANCOVA (Gad and Rousseaux 2002; Shirley and Newnham 1984). In this study, an ANCOVA model was built using treatment as a fixed factor and body weight as the covariate. The interaction between the covariate (body weight) and the fixed factor (treatment: administration of test compound in each individual study) was analyzed for each individual study data set, to evaluate the dependence of the covariate (body weight) with the treatment factor. When the interaction was found to be significant, or close to significant, such as in studies S14, S21, S24, and S26, the assumption of homogenous regression slopes was considered to be violated and therefore the brain weight, instead of the body weight, was used as covariate and males and females were analyzed separately.
Using smaller number of animals is a laudable objective vigorously pursued today in toxicology (Abbott 2010; Stokes 2011; Wells 2011). Obtaining meaningful and conclusive data is a challenge for these early toxicological studies where group sizes are usually small. One of the objectives of the current study was to establish, in a scientific rigorous way, the minimum group size for the 1- to 2-week inhalation studies. Our 1- to 2-week inhalation studies often had 40 animals together and we routinely measured the relative ratio of lung to brain weight (ANOVA models). This study design allowed us to detect the lung weight changes greater than 10 to 20% of control values. If an ANCOVA analysis model is applied, the animal number in one study could be reduced considerably, for example from 50 to 67 male animals to 44 to 58 male animals, for detecting similar level of lung weight change (Table 4).
Another purpose of this review was to investigate whether one could predict lung toxicity from the early 1- to 2-week toxicological studies. Among 10 test compounds, 7 of them showed the same pattern of lung weight changes between the 1- to 2-week and the 4-week studies, suggesting that the lung weight change observed in the 1- to 2-week studies were helpful in predicting the outcome of the 4-week study, irrespective of the presence of lung histopathology.
In conclusion, the lung weight change and lung histopathology in the inhalation studies reviewed showed concordance in the majority of studies. The lung weight change in the 1- to 2-week studies was often predictable to the lung weight change in the 4-week studies with the same test compound. Under current design with 5 males and 5 females per group, the lung weight change greater than 10 to 20% of control values can be detected. The lung weight should be applied as a complementary end point to lung histopathology for assessing lung toxic effect by inhaled compounds in acute/subacute inhalation studies in rat.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
We thank John Foster for critical reading and valuable comments on the manuscript, Per Åberg for continuous support and critical comments, Magnus Söderberg, and Kristian Königsson for valuable comments, Niklas Sjögren for advice on statistical issues, and Gunnar Nordahl for comments on the manuscript.
