Abstract
Senna is a pod or leaf of
Introduction
Senna is a pod or leaf of
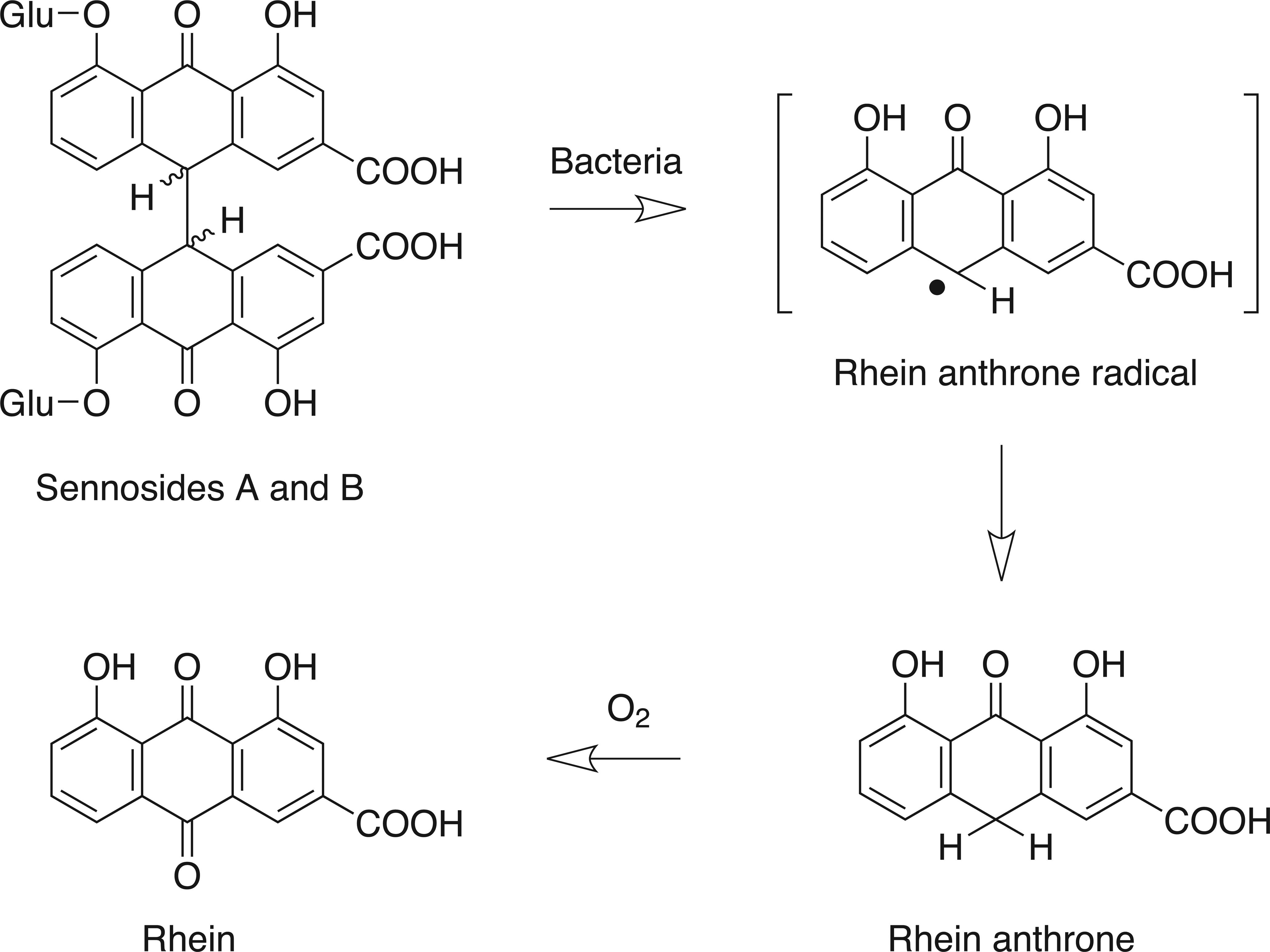
Reduction of sennosides A and B to pharmacologically active rhein anthrone by gut microflora and oxidation to rhein. (Adapted from Lemli and Lemmens 1980 and Dreessen, Eyssen, and Lemli 1981.) Glu = glucose.
Most of the published genotoxicity studies with senna reported negative results, but some positive results for senna extracts were reported in bacterial gene mutation test systems as well as in mammalian cells
Toxicity of senna has been reported in literature. Subacute exposure to senna (10% senna in diet) resulted in signs of liver and kidney toxicity in rats (Al-Yahya, Al-Farhan, and Adam 2002). In a subchronic study, reversible hyperplastic changes in the forestomach and the large intestine have been observed in rats (Mengs et al. 2004). In human, case studies of hepatitis and allergic reactions associated with excessive use of senna have been reported (Beuers, Spengler, and Pape 1991; Marks, Salome, and Woolcock 1991; Helin and Makinen-Kiljunen 1996; Spiller et al. 2003; Seybold et al. 2004; Vanderperren et al. 2005). Sennosides increased colonic epithelial cell proliferation in humans (Kleibeuker et al. 1995; van Gorkom et al. 2000).
Studies reported in the literature suggest that there may be an association between laxative use and colon cancer in humans (Siegers et al. 1993; Satia et al. 2009). For example, findings from an epidemiology study revealed that more patients with gastrointestinal cancer were senna users than patients without cancer and patients without gastrointestinal disease (Boyd and Doll 1954). However, the relationship between senna use and colon cancer has not been clearly demonstrated. Experimental animal models have not been able to resolve this association between senna use and cancer. Several studies showed senna or sennosides did not affect incidences of neoplastic lesions (Lyden-Sokolowski, Nilsson, and Sjoberg 1993; Siegers, Siemers, and Baretton 1993; Borrelli et al. 2005; Mitchell et al. 2006). However, in the literature senna extract or sennosides have been shown to increase tumor formation (Mereto, Ghia, and Brambilla 1996; Mascolo et al. 1999).
The
Laxative use is widespread in the United States and there is concern about potential toxicity from long-term use, including commonly used products containing senna (U.S. Food and Drug Administration 1999). Additional characterization of the toxic potential of senna is needed for risk assessment in humans. Therefore, we conducted toxicological and carcinogenicity studies of senna in the C3B6.129F1-
Materials and Methods
Chemicals
Senna was obtained from Madaus AG (Köln, Germany) in one lot (1999000). Senna was identified by chromatographic comparisons to purchased reference standards using two high-performance liquid chromatography with ultraviolet detection (HPLC/UV) analytical systems. Weight percentages of active components were estimated: 0.7% sennoside A, 1.3% sennoside B, 0.06% sennidin A, and 0.03% sennidin B. The senna was sterilized by exposure to cobalt-60 ionizing radiation. Comparison of the irradiated material with the nonirradiated material indicated that all 10 major peaks present in the nonirradiated material were present in the irradiated material at a relative concentration of ≥90.7%. Examination of the chromatograms before and after irradiation indicated no differences in significant peaks.
Animals
Male and female C57BL/6NTac mice as well as male and female C3B6.129F1-
An outcross between C3H/HeNTac (C3, Taconic Model P53N12-M) female mice homozygous for the wild-type
Five-Week Study
Groups of 5 male and 5 female C57BL/6NTac mice at 6 to 7 weeks of age were used. NTP-2000 diets containing 0, 625, 1,250, 2,500, 5,000, or 10,000 ppm senna were provided
Forty-Week Study
Groups of 25 male and 25 female p53+/− mice at 6 to 8 weeks of age were used. NTP-2000 diets containing 0, 100, 300, 1,000, 3,000 or 10,000 ppm senna were provided
Pathology
Hyperplasia of the large intestinal mucosa was graded similarly in the 5-week and 40-week studies. In the colon, normal crypt depth in controls was up to 175 µm. Colonic hyperplasia was graded based upon crypt depth, as follows: in minimal (grade 1) hyperplasia, crypt depth was approximately 200 µm (from basement membrane to surface of crypt epithelium, measured where an entire crypt was in longitudinal section) but only focally or multifocally; in mild (grade 2) hyperplasia, crypt depth was ≥200 µm over much of the circumference and was focally approximately 300 to 350 µm; in moderate (grade 3) hyperplasia almost the entire circumference of the section examined was involved with crypts 300 to 350 µm deep. The same criteria were used for the cecum but due to the naturally thicker mucosa found in the rectum severity grading for the rectum was done more subjectively.
Statistical Analysis
The Fisher exact test was used to determine statistical differences in incidences of lesions between treatment groups.
Results
Five-Week Study in C57BL/6NTac Mice
A 5-week study was designed to investigate toxicological effects of senna in C57BL/6NTac mice. All mice survived to the end of the study. Final mean body weights of all exposed groups were within 5% of the controls. Feed consumption by exposed groups was similar to that by the controls. Concentrations of 625, 1,250, 2,500, 5,000, and 10,000 ppm senna in feed resulted in average daily doses of 115, 245, 490, 975, and 2,075 mg senna/kg body weight to males and 160, 310, 625, 1,190, and 2,570 mg/kg to females. There were no apparent treatment-related clinical findings. Significant decreases in absolute heart weights occurred in all exposed groups of males. Relative heart weights were significantly decreased in 625, 5,000, and 10,000 ppm males.
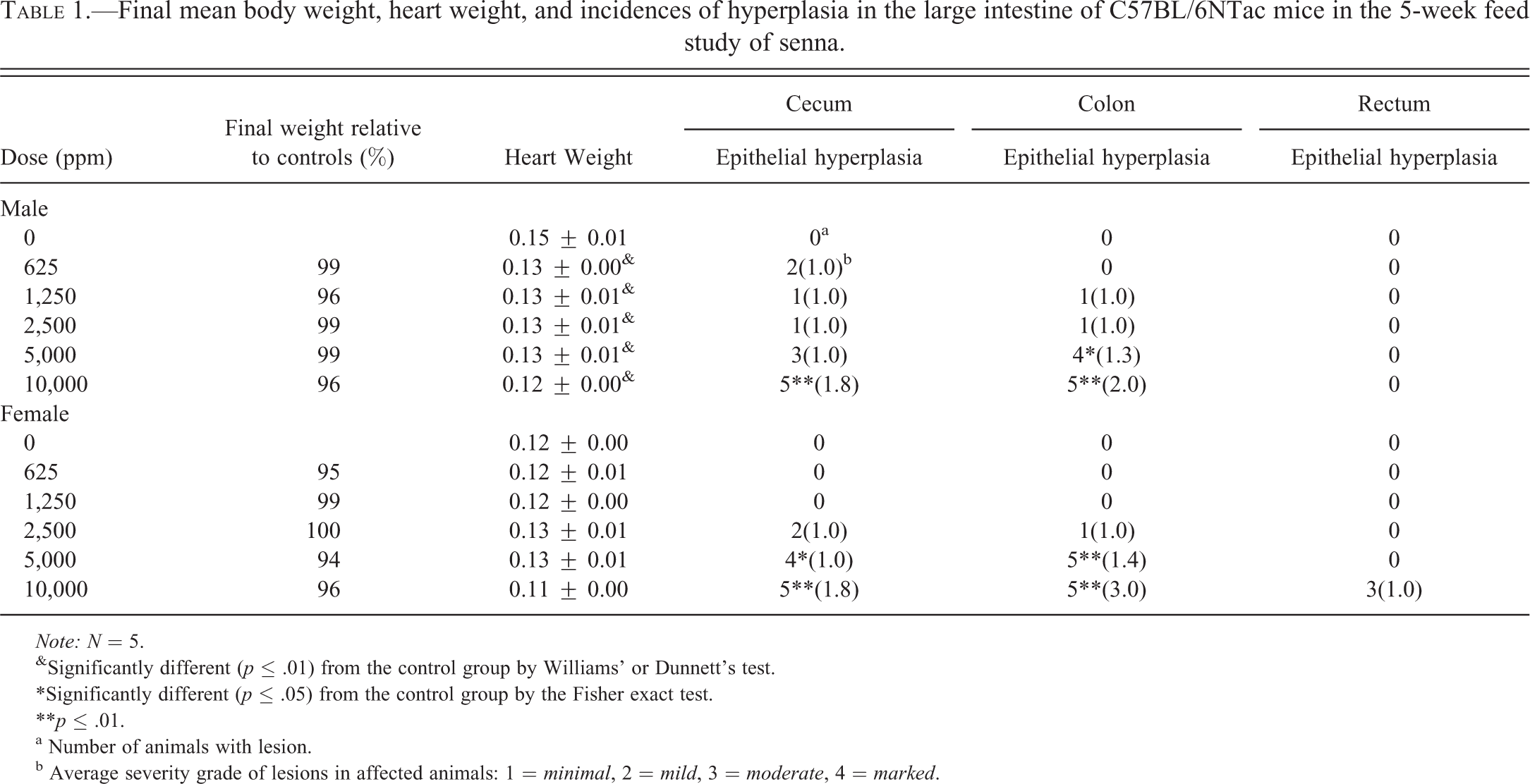
Exposure to senna resulted in epithelial hyperplasia of the cecum and colon in males and of the cecum, colon, and rectum in females (Table 1 and Figure 2). A no-observed-effect level (NOEL) could not be determined for the male mice because the 625 ppm group had two mice with minimal hyperplasia of the cecum, and one animal in the 1,250 ppm group had hyperplasia of the cecum and colon. The NOEL for female mice was 1,250 ppm based upon the lack of lesions in that group and the presence of two animals in the 2,500 ppm group with cecal hyperplasia and one with hyperplasia of the colon. The hyperplasia was characterized by a thickening of the mucosa by increased depth of the crypts (Figure 2). This was due to increased numbers of epithelial cells, which were small, with basophilic cytoplasm and round to oval nuclei, which became more vesicular toward the luminal surface of the glands. Goblet cells were interspersed within these cells but did not constitute an increased proportion of the epithelial cells in the colon or cecum. Mitotic figures were numerous.
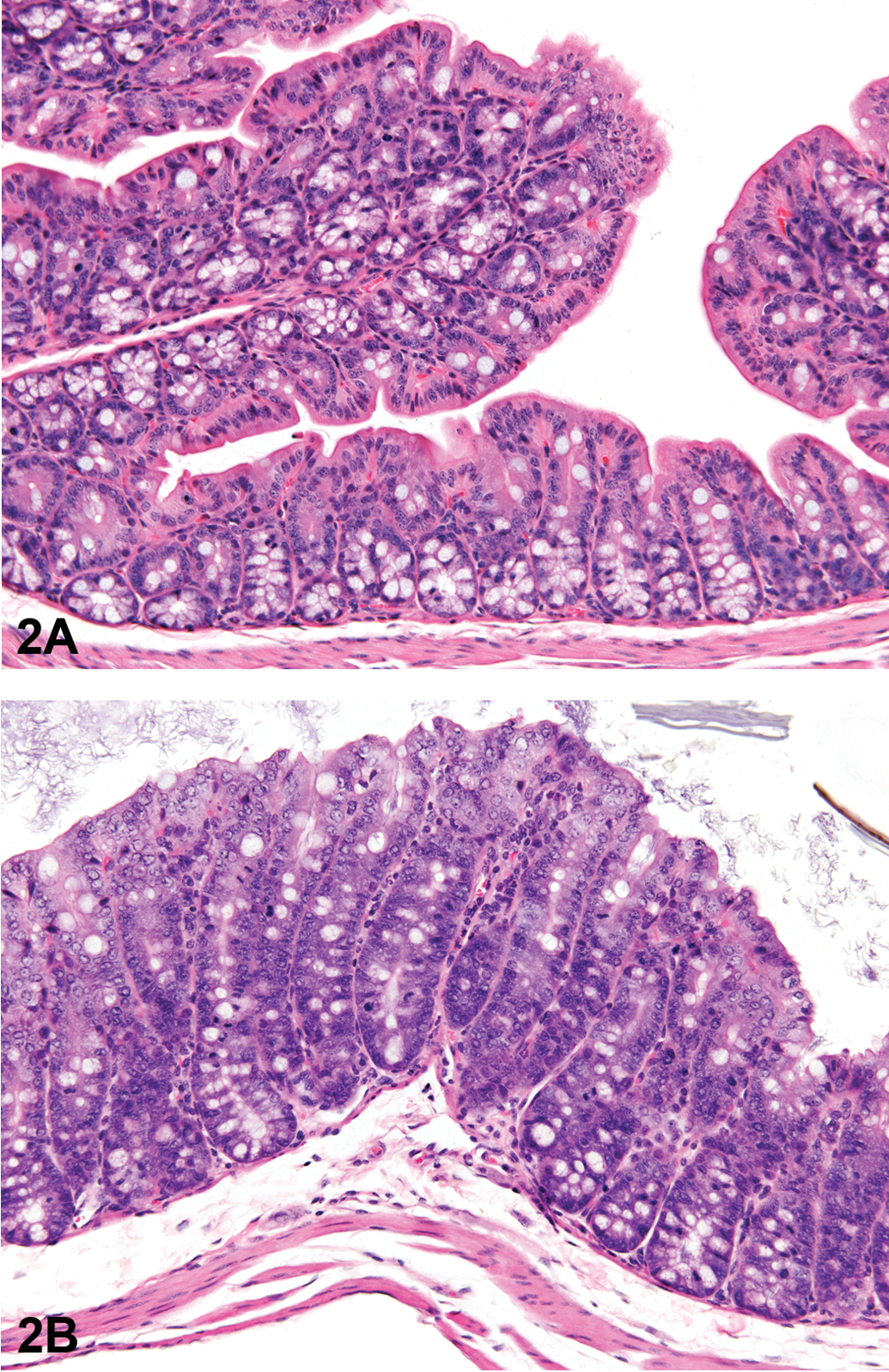
Colon from the 5-week study of senna. (A), A section of colon from a control female mouse; 25×, hemotoxylin and eosin (H&E). (B), A section of colon with moderate hyperplasia from a female mouse exposed to 10,000 ppm senna. The depths of the crypts and the number of epithelial cells are increased, as evidenced by crowding, increased basophilia, and increased mitotic figures; 25×, H&E.
Final mean body weight, heart weight, and incidences of hyperplasia in the large intestine of C57BL/6NTac mice in the 5-week feed study of senna.
&Significantly different (
*Significantly different (
**
a Number of animals with lesion.
b Average severity grade of lesions in affected animals: 1 =
40-Week Study in p53+/− Mice
A 40-week study was conducted to investigate the toxic and/or carcinogenic potential of senna in p53+/− mice. In the 5-week study in C57BL/6NTac mice, no effect on survival or body weights was observed. In addition, intestinal lesions were not considered to be of a sufficient severity to cause mortality in a 40-week study in p53+/− mice. Therefore, the high dose of 10,000 ppm was selected. Due to increased sensitivity in p53+/− mice and increased duration of exposure, a broader range of exposure (0, 100, 300, 1,000, 3,000, and 10,000 ppm) was selected for the 40-week study.
Survival of all exposed groups was similar to that of the control groups (Table 2). Mean body weights of exposed male and female mice were within 10% of those of the controls throughout the study. Feed consumption by exposed mice was generally similar to that by the controls throughout the study. Concentrations of 100, 300, 1,000, 3,000, and 10,000 ppm in feed resulted in average daily doses of 12, 36, 120, 365, and 1,260 mg/kg to males and 14, 42, 140, 435, and 1,520 mg/kg to females. There were no apparent exposure-related clinical findings. The absolute and relative liver weights of 10,000 ppm males were significantly less than those of the controls.
Significant increases in the incidences of epithelial hyperplasia of the colon and cecum occurred in 10,000 ppm males and females, and the incidence of hyperplasia of the colon was significantly increased in 3,000 ppm females (Table 2, Figures 3 and 4). Three 3,000 ppm males had epithelial hyperplasia of the colon and single occurrences of epithelial hyperplasia of the rectum were recorded in the 10,000 ppm males and females; although lacking statistical significance, these lesions are considered biologically noteworthy. The changes were characterized by a thickening of the mucosa by a lengthening of the crypts (Figures 3 and 4). This was due to increased numbers of epithelial cells that appeared crowded together. These cells were small and had basophilic cytoplasm and round to oval nuclei, which became more vesicular toward the luminal surface of the glands. Goblet cells were interspersed with these cells but did not constitute an increased proportion of the epithelial cells in the colon or cecum. Mitotic figures were numerous. The cecum was usually less severely affected than the colon.
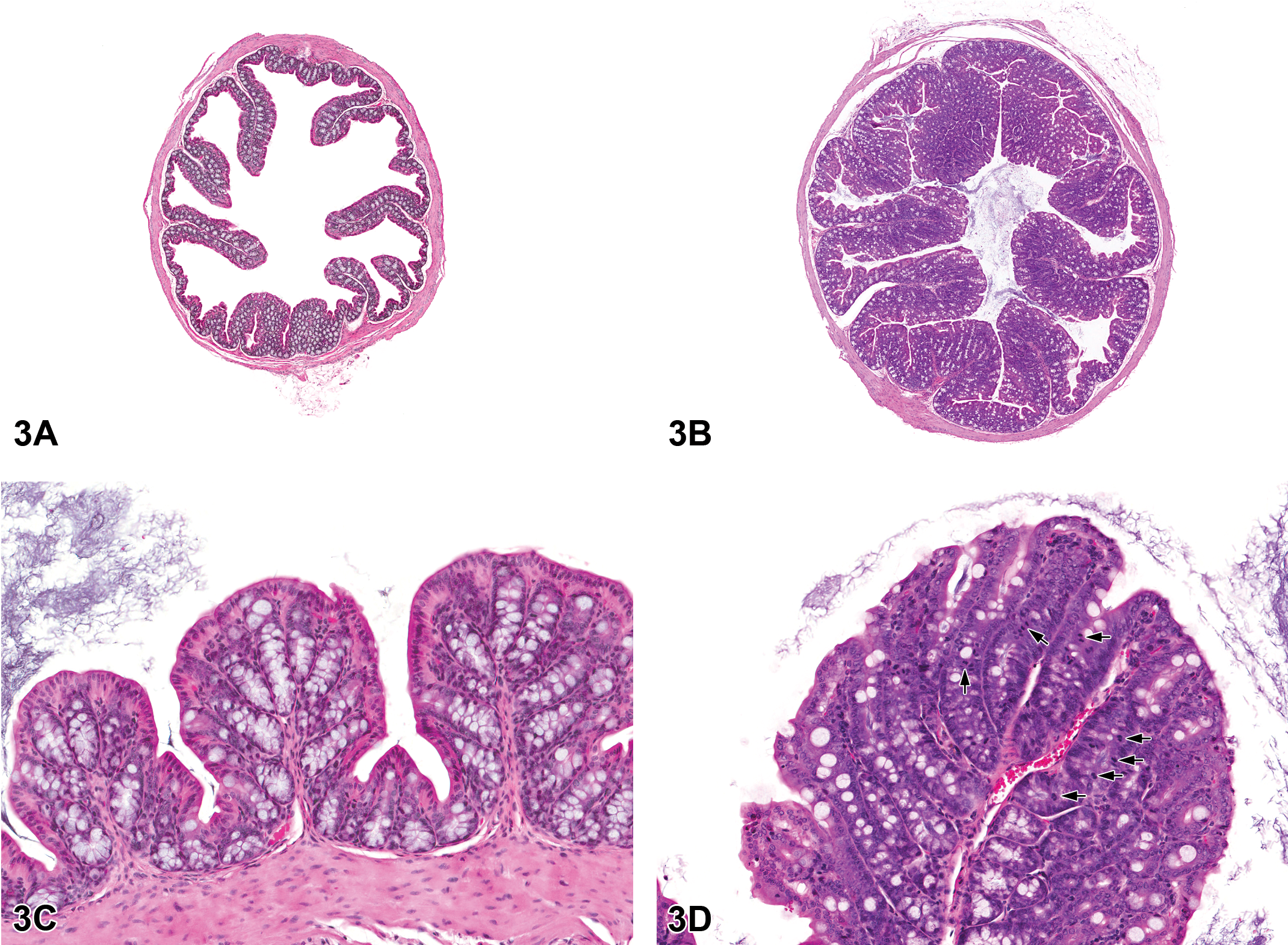
Colon from the 40-week study of senna. (A), A cross-section of the colon from a control male mouse; 3×, hemotoxylin and eosin (H&E). (B), A cross-section of the colon from a male p53+/− mouse exposed to 10,000 ppm. There is moderate epithelial hyperplasia, with diffuse thickening of the epithelium. 3×, H&E. (C), Higher magnification of colonic epithelium from a control male mouse. 20×, H&E. (D), a section of colon from a male mouse exposed to 10,000 ppm senna. The epithelium is thickened as evidenced by increased distance from the luminal surface to the base of the crypts. Epithelial cells are increased in number and are basophilic and crowded, the cells lack the orderly arrangement seen in the control mouse, and there are numerous mitotic figures (arrows); 20×, H&E.
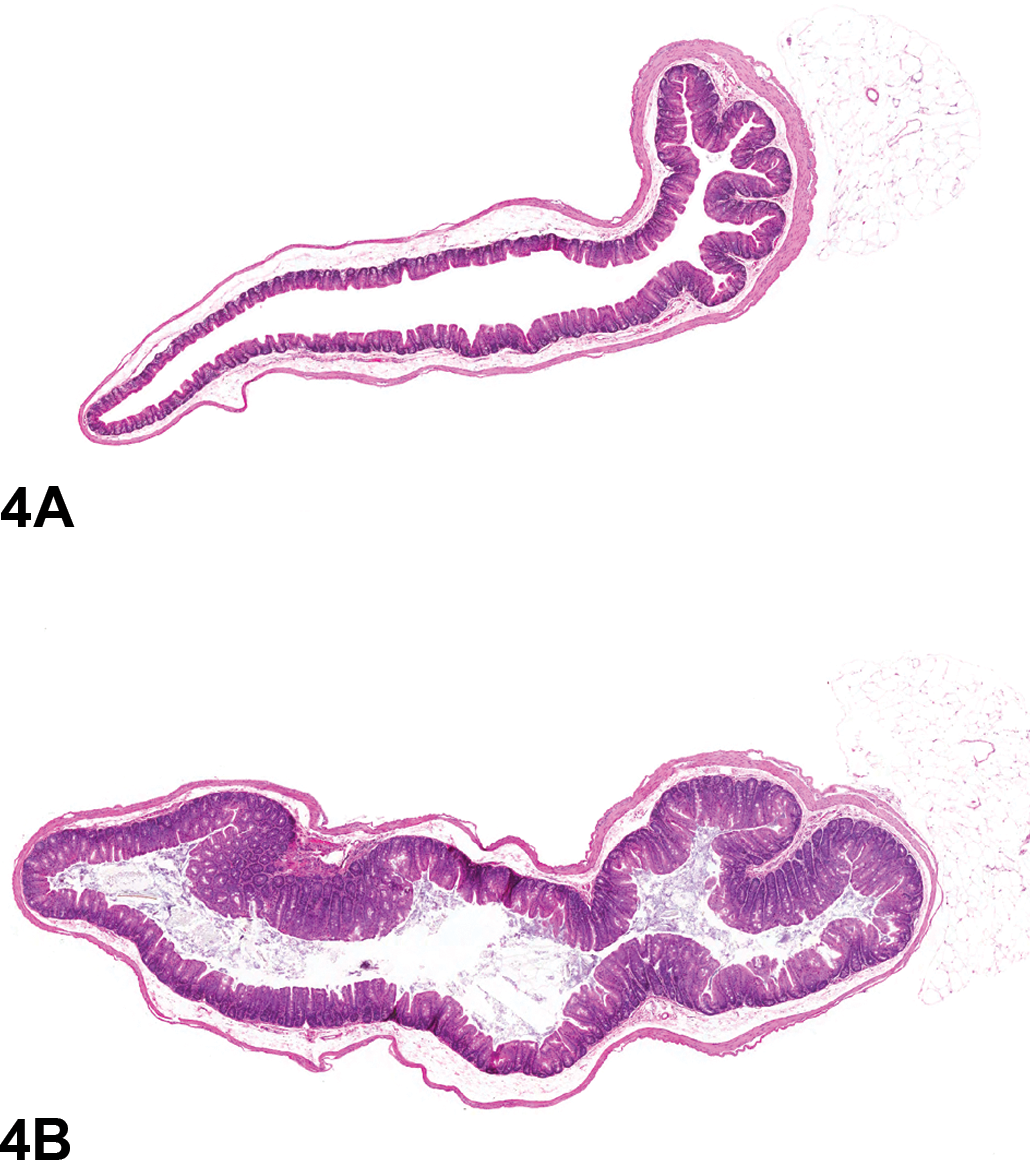
Cecum from the 40-week study of senna. (A), Low magnification of the cecum from a control male; 2.3×, hemotoxylin and eosin (H&E). (B), Cecum from a male mouse exposed to 10,000 ppm senna. There is moderate hyperplasia; the epithelium of the cecum is diffusely thickened in the treated animal when compared to the control animal; 2.3×, H&E.
Survial, final mean body weights and incidences of hyperplasia in the large intestine of p53+/− mice in the 40-week feed study of senna.
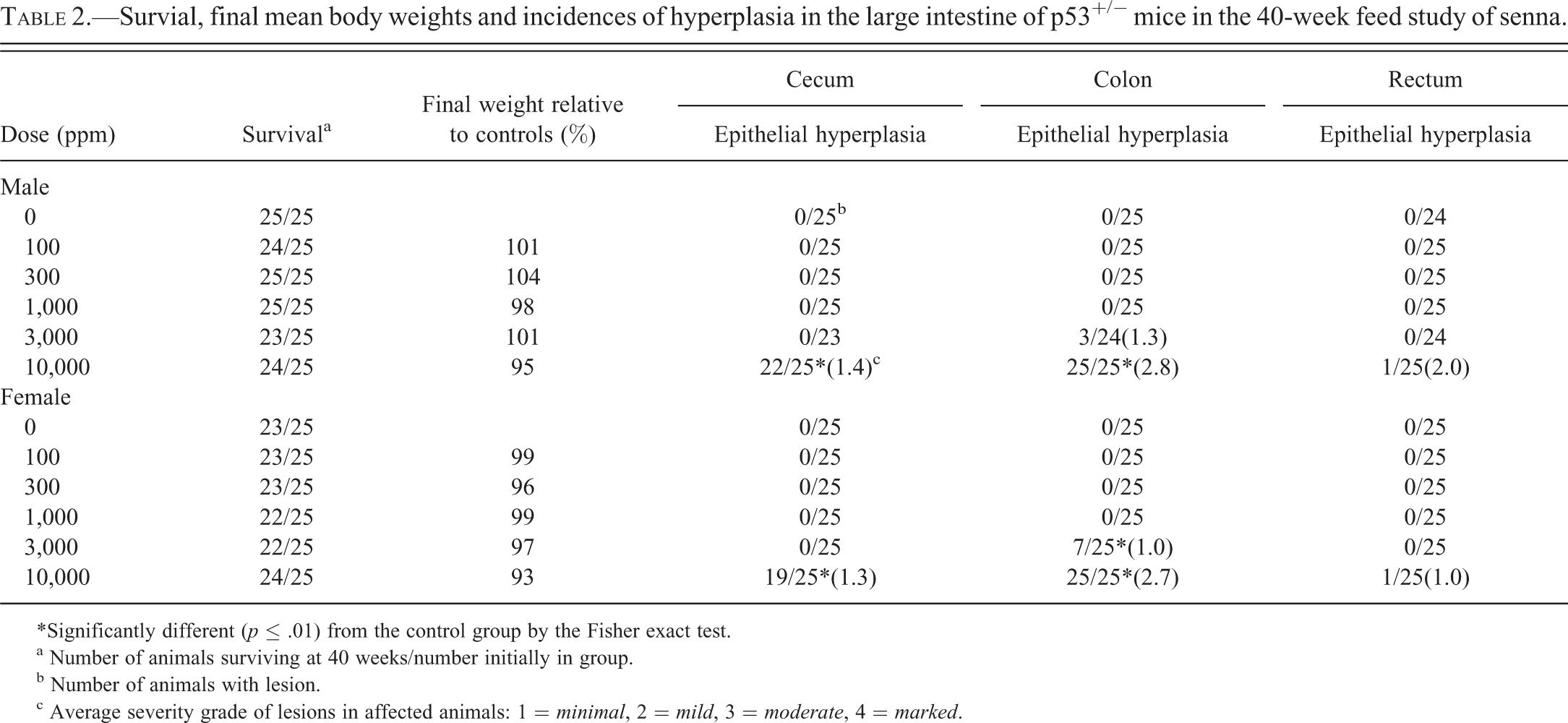
*Significantly different (
a Number of animals surviving at 40 weeks/number initially in group.
b Number of animals with lesion.
c Average severity grade of lesions in affected animals: 1 =
Osteosarcomas occurred in 1,000 and 3,000 ppm males but not in the control group; the incidences in these groups were within the historical control range (Table 3). In 1,000 ppm females, the incidence of osteosarcoma exceeded the historical control range by one tumor but was not significantly greater than the incidence in the concurrent control group. Osteosarcomas in the mice were found in various sites, including femur (one male and one female), maxilla (four males and two females), rib (one female), tibia (one female), vertebra (one male and four females), and unspecified (one female). The incidence of hepatocellular adenoma in 300 ppm males was increased, which exceeded the historical control range but not significantly (Table 3). The biological relevance of these findings is unknown.
Incidences of osteosarcoma and hepatocellular adenoma in p53+/− mice in the 40-week feed study of senna.
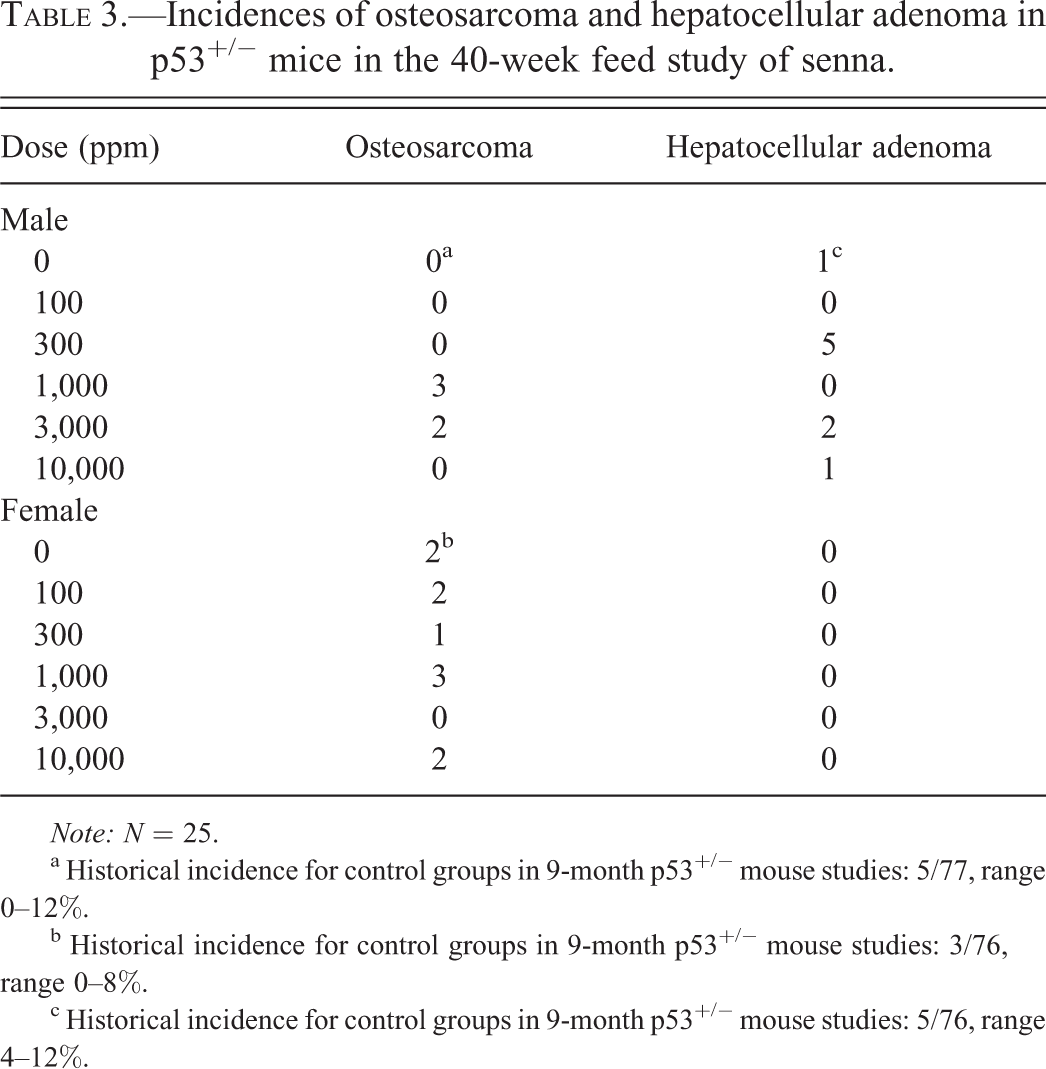
a Historical incidence for control groups in 9-month p53+/− mouse studies: 5/77, range 0–12%.
b Historical incidence for control groups in 9-month p53+/− mouse studies: 3/76, range 0–8%.
c Historical incidence for control groups in 9-month p53+/− mouse studies: 5/76, range 4–12%.
Discussion
The daily senna exposure concentrations in the current studies overlapped with daily exposures in humans taking senna in laxative preparations. In the 40-week study, based on the percentage total weight values for sennosides A and B (approximately 2% of senna by weight), the sennosides A and B consumption was approximately 0.24, 0.72, 2.4, 7.3, or 25.4 mg/kg or 0.72, 2.2, 7.2, 21.9, or 76.1 mg/m3 (on a body surface area basis) per day in males (Freireich et al. 1966). Senna consumption by females was similar to that by males. For a typical over-the-counter senna laxative preparation taken twice daily, the amount of sennosides consumed is approximately 1.4 mg/kg per day or 51.8 mg/m3 (body surface area basis) per day (Novartis 2010). In some cases, laxatives may be taken in greater than recommended amounts (Cance, Ashley, and Penne 2005; Bryant-Waugh et al. 2006; Mond et al. 2006; Tozzi et al. 2006).
Senna had a laxative effect in rats receiving daily doses of 300 mg/kg or greater by oral gavage for 13 consecutive weeks (Mengs et al. 2004). Also, senna extract or sennosides had a laxative effect in rats (Lyden-Sokolowski, Nilsson, and Sjoberg 1993; Toyoda et al. 1994; Mascolo et al. 1999). However, in the current studies no laxative effect was observed at comparable or higher doses in mice, possibly indicating species-dependent differences in sensitivity or kinetics in the formation of rhein anthrone, primarily responsible for the purgative activity of senna (Leng-Peschlow 1992; Yamauchi, Yagi, and Kuwano 1993). Additionally, a vehicle effect cannot be ruled out due to possible interaction of sennoside-derived species with the feed particles. Rhein anthrone is formed by intestinal bacteria at the site of action and is purportedly metabolized through a free radical (Lemli and Lemmens 1980; Dreessen, Eyssen, and Lemli 1981; Lemli 1988), a possible mechanism in the formation of the intestinal hyperplastic response observed at the high dose in the current studies.
Significant decrease in heart weights was observed from the 5-week study. However, no gross lesions were found in heart tissues. In the 40-week study, there was no change in heart weights or no treatment-related lesions in the heart. Therefore, decrease in heart weight was not considered treatment related. In the 40-week study, there were changes recorded in the liver weights (reduction in the mean absolute and relative liver weight in the 10,000 ppm males and an increase in the relative liver weight in the 10,000 ppm females), but the incidences of chronic active inflammation, or any other lesion in the liver, could not explain the liver weight changes.
There were increases in the incidences of osteosarcoma in 1,000 and 3,000 males. However, the incidences were within historical control range. Increased incidence of osteosarcoma in 1,000 females was not significantly increased but greater than the historical control range. Increased incidence of hepatocellular adenoma in 300 ppm males exceeded the historical control range, but not significantly. Therefore, increased incidences of osteosarcoma and hepatocellular adenoma were not considered as treatment-related finding.
Treatment-related epithelial hyperplasia in the large intestine occurred in both the 5-week senna study in C57BL/6NTac mice and in the 40-week senna study in p53+/− mice. These results are consistent with other rodent studies. In rats receiving up to 0.05% sennoside A in feed for 7 days, a dose-dependent increase in proliferation index in the large intestine (cecum and colorectum) was observed (Toyoda et al. 1994). However, in contrast to our results, inflammatory changes (neutrophilic leukocytes infiltration and crypt abscess formation) were observed in that study. In rats receiving up to 1,500 mg/kg senna for 13 weeks, minimal to slight hyperplastic changes in the mucosa of the large intestine in rats receiving more than 100 mg/kg of senna were observed. These hyperplastic lesions were not observed after 8 weeks of recovery period (Mengs et al. 2004). Also, in rats receiving 25, 100, or 300 mg/kg senna for 2 years, dose-dependent, minimal to slight mucosal epithelial hyperplasia of the large intestine (colon and cecum) was observed (Mitchell et al. 2006). In addition, in human, 2 mg/kg sennoside A and B administration increased the proliferative index in the large intestine (cecum, colon, and rectum; van Gorkom et al. 2000).
In agreement with our results, exposure to senna, senna extract, or sennosides to rodents in previous studies was not associated with tumor formation. Mitchell et al. (2006) conducted a 2-year oral gavage study of senna in Sprague-Dawley rats (0, 25, 100, or 300 mg/kg/day). There were no treatment-related carcinogenic effects in any target organ and there was only a slight intestinal hyperplasia in the treated groups in that study. In another 2-year study in Sprague-Dawley rats administered a senna extract (reported to contain up to 35% to 42% sennosides) in drinking water (to deliver 0, 5, 15, or 25 mg/kg/day of the test article), no intestinal lesions were observed in treated or control groups (Lyden-Sokolowski, Nilsson, and Sjoberg 1993). Also, administration of 30 or 60 mg/kg senna extract by oral gavage (six times per week) for 110 weeks in male Wistar rats did not induce aberrant crypt foci or tumors (Borrelli et al. 2005). Mascolo et al. (1999) showed that a laxative-producing dose of senna pod extract (10 mg/kg) administered to male Wistar rats for 13 to 28 weeks did not promote the formation of tumors induced by azoxymethane. In addition, a diet containing 0.03% sennoside fed to male NMRI mice for 20 weeks with concurrent administration of dimethylhydrazine (20 mg/kg, subcutaneous) for 10 weeks did not promote the formation of colorectal tumors (Siegers, Siemers, and Baretton 1993). The numbers and multiplicities of aberrant crypt foci were not affected in dimethylhydrazine-treated male Sprague-Dawley rats fed a 0.1% sennoside-containing diet (Mereto, Ghia, and Brambilla 1996).
Results of NTP bacterial mutagenicity tests with senna and some of its constituents (sennosides, rhein, and chrysophanic acid) were mixed, with some samples giving negative results and others, including rhein and the senna sample used in the 40-week mouse study, giving positive results (NTP 2011). Despite the observed activity with some of these compounds
In contrast to our result, a diarrhea-producing dose (100 mg/kg, by gavage) of senna pod extract administered for 13 weeks to rats increased the incidence and multiplicity of tumors induced by azoxymethane (Mascolo et al. 1999). In addition, Sprague-Dawley rats fed a diarrhea-inducing level of sennoside in the diet (0.2%) exhibited a significantly increased number of crypts/foci compared to controls (Mereto, Ghia, and Brambilla 1996). However, oral administration of 30 or 60 mg/kg senna pod extract for 2 years to male Wistar rats treated with the initiating agent azoxymethane (7.5 mg/kg, intraperitoneal) decreased the formation of aberrant crypt foci and colon tumors compared to animals that received azoxymethane alone (Borrelli et al. 2005).
In conclusion, the major effect of senna administered in feed to mice was a dose-related increase in the incidence of intestinal epithelial hyperplasia. There was no neoplastic change in this experimental situation, when senna was administered to p53+/− mouse; however, it remains to be determined whether the observance of senna-induced intestinal hyperplasia in rodents indicates potential risk to senna-exposed humans.
Footnotes
Acknowledgements
This article may be the work product of an employee or group of employees of the National Institute of Environmental Health Sciences (NIEHS), National Institutes of Health (NIH). However, the statements, opinions, or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the U.S. government. We greatly acknowledge the expert review by Ms. Kristine Witt and Dr. Arun Pandiri. The in life phase of the study was conducted under NIEHS contracts N01-ES-65406 and N01-ES-55536. The authors would like to thank Drs. Milton Hejtmancik, Diane Gerken, Daphne Vasconcelos, and other staff at Battelle Memorial Institute for helping to conduct these studies.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
