Abstract
We evaluated immunohistochemistry (von Willebrand Factor [vWF] or fibrinogen) and systemic and coronary arterial physiological parameters in beagle dogs to investigate early arterial lesions induced by the potassium channel opener, ZD6169, or the endothelin receptor antagonist, ZD1611. Dogs given an oral dose of ZD6169 (experiment 1) were terminated 1 day later and showed arterial and myocardial lesions. Minimal arterial lesions exhibited few condensed medial smooth muscle cells only, with others showing segmental medial necrosis occasionally with medial/adventitial acute inflammation. Intercellular immunostaining was seen in ostensibly normal tissue, where no pathology was present in conventionally stained sections. vWF and fibrinogen are valuable tools for detecting disruption of arterial integrity. In experiment 2, 2 dogs were given a single high dose of ZD6169 or ZD1611 and BP/HR monitored by conventional measures or telemetry. Substantially reduced systolic/diastolic BP and increased HR occurred within 10 min of ZD6169 infusion: ZD1611 caused minor BP decrease and HR increase. In experiment 3, both drugs given to anaesthetized dogs induced markedly exaggerated systolic phasic forward and reverse flow in left descending and right coronary arteries. Diastolic coronary artery flows were unaffected with ZD1611 and increased slightly with ZD6169. In both coronary arteries, the ZD1611-induced increase in flows paralleled decreased resistance.
Keywords
Introduction
Administration of numerous vasoactive substances to beagle dogs has resulted in a relatively small number of cardiac and vascular pathological changes that often show a predictable pattern related to drug class (Albassam et al. 2001; Greaves 1998; Herman et al. 1989; Isaacs, Joseph, and Betton 1989; Jones et al. 2003) and include cardioactive drugs such as endothelin receptor antagonists (ETRAs) or potassium channel openers (KCOs: Albassam et al. 2001; Dogterom, Zbinden, and Reznik 1992; Isaacs, Joseph, and Betton 1989; Jones et al. 2003; Louden & Morgan, 2001; Louden et al. 1998; Metz et al. 1991). Cardiac pathology consequent to KCO administration is diverse and often substantial with left ventricular and papillary muscle myocardial necrosis, left ventricular subendocardial hemorrhage, right atrial epicardial hemorrhage, and bilateral, intra- and extramural coronary arterial damage being frequently seen. KCOs also induce such pathological changes in miniature pig and monkeys (Belin et al. 1996; Herman et al. 1989), although the greater doses required to induce lesions in these latter two species have led to the dog being considered as especially sensitive to these molecules (Food and Drug Administration [FDA] 2002). By contrast, ETRAs induce coronary (and some systemic) arterial damage alone. The histopathological appearances of coronary arterial lesions induced by different substances, for example inotropic vasodilators (e.g., ETRAs, KCOs, or phosphodiesterase inhibitors), are often strikingly similar and are indicative of physical disruption of vessels. Coronary arterial damage induced by these drugs is characterized in early lesions by medial smooth muscle necrosis often in a segmental or diffuse circumferential pattern, hemorrhage, and inflammatory cell infiltration, and later, by intimal proliferation, chronic inflammatory cell infiltrates, adventitial fibrosis, and neovascularization and is present at multiple sites and often at high incidence in dosed animals (Isaacs, Joseph, and Betton 1989; Jones et al. 2003; Joseph, Jones, and Kerns 1996).
While the cardiovascular pathology that occurs as a consequence of acute and chronic drug administration is well documented, its pathogenesis is not understood but widely regarded to be due to significant perturbations in cardiovascular physiology. It has been suggested that in clinical studies, conventional, noninvasive peripheral blood pressure and pulse measures are adequate for monitoring drug-induced effects. With regard to KCOs, this may be so but we know that some vasoactive drugs, (e.g., ETRAs) cause coronary arterial damage with only minor alterations in these monitorable peripheral cardiovascular parameters. When given to dogs, single or multiple doses of KCOs produce a marked, dose-dependent systemic hypotension accompanied by reflex tachycardia often resulting in peripheral blood pressure of 50 to 60% predose values and heart rates of 180 to 200 bpm (Carlson and Feenstra 1977; Mesfin et al. 1995). Our choice of ZD6169 ((
In the search for biological indicators of vascular injury induced by vasoactive pharmaceuticals in humans, correlation between arterial pathology obtained from studies in dogs and detection of lesion-associated molecules in plasma is a required first step. While some advance has been made in identifying and investigating potential biological indicators of drug-induced vascular injury (DIVI; Kerns et al. 2005), investigations directed at clarifying the fundamental cardiovascular physiological events that form the basis for subsequent disruption of arterial structure have not been undertaken. It is clear that without an understanding of the pathogenesis of DIVI, no accurate correlation can be constructed of the temporal relationship between the administration of a cardioactive drug and the appearance of the first changes in plasma concentration of such markers. Examination of the morphological hallmarks of DIVI would suggest that an early event is likely to be disruption of the structural integrity of the blood vessel, with associated leakage of plasma from the vascular lumen into the vessel wall. Conventional tissue processing does not allow for the sensitive detection of plasma ingress into the vessel wall and more sensitive markers than H&E-stained sections are needed. In attempting an understanding of the nature of the earliest lesions associated with intrusion of plasma into the artery we employed fibrinogen and von Willebrand Factor (vWF) as indicators of the tissue distribution of plasma.
In addition to the cardiovascular physiological changes that occur acutely following dosing of either ZD6169 or ZD1611, this article focuses on description of the early structural changes in canine coronary arteries that occur following a single cardiotoxic dose of these drugs as assessed by conventional histopathological examination and describes the changes in tissue distribution of fibrinogen and vWF using immunohistochemistry: changes due to ZD6169 are reported primarily to avoid reiteration of the same vascular changes due to ZD1611. We chose to investigate such a model rather than disease models, for example, of atherosclerosis or spontaneous canine vascular disease (Beagle Pain Syndrome; Bishop 1989; Hayes et al. 1989), which, because of its acute nature, offered insights into the early pathogenesis of induced vascular damage in a simple experimental scenario that was free of the adaptive and reparative modulation of vascular structure manifest in chronic vascular disease.
Materials and Methods
Early Markers of Vascular Injury
Animals and Study Design
Beagle dogs were obtained from the breeding unit (AstraZeneca Pharmaceuticals, Alderley Park, Macclesfield, UK) aged between 51 and 65 weeks and were housed in standard conditions except during telemetry recordings. Water from the site drinking water supply was freely available and a set quantity of diet (Special Diets Services Ltd, UK) was provided 1 hr pre-dose and removed 2 h later.
Animals (all females) obtained from two experiments were employed. In the first experiment, the doses administered to 2 animals per dose were 15, 60, or 240 mg/kg ZD6169 (AstraZeneca, Macclesfield, UK) while in the second, either 240 mg/kg ZD6169 or 200 mg/kg ZD1611 (AstraZeneca, Macclesfield, UK) was administered by gavage to 2 animals per drug. All animals were given a single oral dose of ZD6169 by gavage formulated as a suspension in 0.5% w/v hydroxypropyl-methylcellulose in 0.1% w/v polysorbate. ZD1611 was formulated as a 50% mixture with lactose and administered by gavage.
No additional control (vehicle-dosed) animals were used in this study. However, in order to determine the distribution of fibrinogen and vWF in untreated dog coronary arteries, sections of heart from 12 control dogs from a separate study were immunohistochemically stained for fibrinogen and vWF. The animals in this latter study were housed, maintained, and examined in the same way as the ZD6169-treated animals but were dosed with the same vehicle only.
Physiological Measurements
Experiment 1
All animals were assessed as follows. Three separate electrocardiograms (ECG) and direct arterial blood pressure (BP) measurements were made on all dogs: once during the pre-test period and 1 or 4 hr post dose for ZD6169 and ZD1611, respectively (at estimated Cmax obtained from previous studies). The 2 dogs per group dosed with 15, 60, or 240 mg/kg ZD6169 were also monitored for the same parameters once pre-dose. Multiple concentrations of ZD1611 were not evaluated. Arterial blood pressure was measured directly from the cannulated central ear artery (intermediate auricular) of the conscious dog with a full bridge transducer (Statham P23XL manometer transducer, Spectramed, CA, USA) under local anesthesia with 2% lidocaine hydrochloride (which has no or negligible associated systemic cardiovascular effects at the dose and administrative route used). Systolic and diastolic blood pressures were obtained from the blood pressure recordings. All animals were terminated at 24 hr post dose.
In experiment 2, all animals were continuously monitored telemetrically for 24 hr via a noninvasive, ECG system (EMKA Technologies, Paris, France) and terminated at the end of the evaluation period. Lead II ECG only was recorded continuously from jacketed dogs, singly housed in a double-pen, fitted with appropriate signal receivers (antennae). Recordings were made from both animals on day 1 for approximately 1 hr, and then for approximately 10 min pre-dose and 24 hr post dose on the day of dosing. Dogs were removed from the pens for dosing and at prescribed time points for conventional recordings (as above).
On day 1, the data were sampled every 10 min during the pre-test recording, for approximately 1 hr, then averaged and reported (Schofield et al. 2006). For the pre-dose data on day 1, a single sample was recorded for 1 min approximately 10 min prior to dosing. For the post-dose values, data were sampled for 1 min at 10-min intervals for the first 2 hr post dose, and thereafter at intervals of 20 min for the remaining 24 hr post dose. The data from these samples (up to 100 beats/sample) were averaged and reported in Table 1. The telemetry data were compared to conventional data (obtained as for experiment 1) using averaged data at comparable times prior to removal of the animal from its pen for conventional recordings (Davis 1999; Fredericia 1920; Prior et al. 2009).
Cardiovascular physiological measurements following ZD6169 or ZD1611 administration

Note: Cardiovascular physiological measurements (Heart rate [beats/min]) and blood pressure (systolic/diastolic, mm Hg) from animals given a single oral dose of 15, 60, or 240 mg/kg ZD6169 and measured pre-test and 1 hr post dose. In a separate experiment, 2 animals given a single oral dose of 240 mg/kg ZD6169 were telemetrically monitored for 24 hr. In these dogs (*), data were recorded pre-test and 1 hr pre- and post-dose. Note the elevated heart rates and reduced blood pressures in all animals 1 or 4 hr post dose compared with pre-test or pre-dose values. The physiological changes are most apparent in animals given 60 mg/kg ZD6169 and above.
*Experiment 2.
Necropsy and Histopathology
All animals were terminated 24 hr post dose, by intravenous injection of an overdose of a solution of 200 mg/ml pentobarbital sodium into the left jugular vein. At necropsy, the heart was examined and tissues sampled as described below.
All studies were compliant with UK Home Office requirements.
Cardiovascular Physiological Studies (Experiment 3)
Animals
All experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the U.S. National Institutes of Health (NIH Publication No. 85-23, revised 1996), and approved by the ethical committee for animal research at the University of Göteborg, Sweden. Eighteen beagle dogs with a weight range between 14.0 and 19.6 kg were used in this study. The dogs were all healthy and purchased from the same breeder (AstraZeneca Rååhöjden, Örkelljunga, Sweden). They were stalled in house for at least 1 week before entering the experimental protocol and overnight fasted before surgery.
Anaesthesia and Ventilation
The dogs were anesthetized with 6 to 8 mg/kg Propofol and when necessary titrated with 2 to 3 mg (Diprivan®, AstraZeneca Ltd., Macclesfield, Cheshire, UK). The anaesthesia was maintained with 100 mg/kg a-chloralose as an intravenous bolus injection and then 25 to 50 mg/kg/hr as a continuous infusion. After induction of anesthesia, the dogs were intubated with a cuffed tracheal tube and ventilated with room air supplemented with 10% oxygen by means of a positive-pressure respirator (15 cycles/min). During the preparation period, the anesthesia was supplemented with 1.8% isoflurane (Florene®, Abbott Scandinavia AB, Solna, Sweden) added to respiratory air. Arterial blood gases and pH were checked every 30 min and adjusted to physiological levels (average values were: pH 7.39 ± 0.003; pCO2 5.3 ± 0.04 kPa; pO2 12.0 ± 0.2 kPa). Deviations were corrected by regulating the tidal volume and by continuous infusion of a solution of sodium bicarbonate and Ringer solution (10 ml/kg × hr) to replace fluid losses.
Animal Preparation
Catheters were inserted into the saphenous and brachial veins for infusion of drugs, anesthetics, sodium bicarbonate, and Ringer solution. Aortic blood pressure was measured through a catheter inserted into the saphenous artery and connected to a pressure transducer. Central venous pressure and body temperature were measured through a catheter and a thermistor in the right external jugular vein. A dual sensor pressure transducer was inserted via the right carotid artery, with the proximal transducer placed in the ascending aorta (mean arterial pressure [MAP]) and the distal transducer in the left ventricle (left ventricular pressure, LVP and max dP/dt). The left femoral artery was exposed and femoral artery blood flow (FAF) measured using an ultrasonic transit-time volume flow sensor (T206 Transonic System Inc., Ithaca, NY, USA). The heart was exposed through a mid-sternal split and suspended in a pericardial cradle. One segment, between the first and second diagonal branches of the left anterior descending coronary artery (LAD), and one segment of right coronary artery (RCA), 10 to 20 mm from its origin, were carefully freed from the surrounding tissue to allow placements of closely fitting flow probes. LAD and RCA flows, as well as the flow in the ascending aorta were continuously recorded by transit-time volume flow sensors (T206 Transonic System Inc, Ithaca, NY, USA). Body temperature was monitored and held between 37°C and 38°C using a thermal table and a heating lamp. The urinary bladder was catheterized and urine drained by gravity. A custom-designed computer program was used for data sampling and for analysis of ECG and hemodynamic signals (PharmLab V5b, AstraZeneca R&D, Mölndal, Sweden).
Experimental Protocol
During stable hemodynamic conditions, about 1 hr after finishing the surgical procedure and the use of isoflurane anesthesia, a baseline period of 90 min was started with automatic recordings of hemodynamic conditions throughout the entire experimental period. After completion of the baseline period, drug infusions began. The vehicle solution and 5 mg/kg ZD6169 were administered for 30 min whereas the 10 mg/kg ZD1611 was given for 10 min. The vehicle solution contained either 1 ml/kg mannitol, 5% (v/v) (3 animals) or 30% (w/w) cyclodextrin at 1 ml/kg (3 animals). Three hours after initiating drug infusions the experiment was terminated, and the dogs received an overdose of pentobarbital sodium. The heart was examined for macroscopic lesions (hemorrhage) and sampled as detailed below.
Data Presentation and Statistical Analysis
Data are mean value ± SEM. The Student paired or unpaired t test was used to assess differences between pre- and post-dose values. A probability value of p < .05 was considered statistically significant. Data in the two vehicle groups are pooled because no differences between them were apparent.
Histopathological and Immunohistochemical Assessments
After euthanasia, the heart was excised and external surfaces carefully examined for the appearances of vascular lesions and when discovered, their position noted and tissue samples taken. The heart was then opened revealing all chambers and further examined closely. Tissue sampling was comprehensive, with any lesions and standard samples of right and left atria, ventricles, and papillary muscles taken and immersed in 10% neutral buffered formaldehyde and fixed for 24 to 48 hr. Following conventional tissue processing into paraffin wax, 4 µm tissue sections were cut, stained with H&E, examined by light microscopy, and evidence of vascular and myocardial lesions recorded. Whenever possible, serial sections (3 to 5 µm thick) were obtained in order to examine the same arterial lesion by H&E and immunohistochemical staining.
Where tissue damage was clearly associated with surgical procedures, all lesions were disregarded. However, lesions that were located deeper in the myocardium, for example, papillary muscle and/or myocardial necrosis or arterial medial damage, were regarded as being drug-induced injuries.
In order to demonstrate arterial lesions, serial sections were immunohistochemically stained to demonstrate either vWF or fibrinogen. The methods used are as follows.
Sections were stained using appropriate primary antibody followed by the DAKO EnVision + polymer System (Ely, Cambridge, UK), which uses 3,3′-diaminobenzidine (DAB) as chromogen. Briefly, 4-µm thick paraffin-embedded tissue sections were mounted on slides, deparaffinized, and rehydrated by sequential immersion in aqueous solutions containing decreasing amounts of ethanol (100 to 70%) and finally into running tap water. Antigen retrieval was achieved by proteolytic digestion using DAKO Pronase solution (DAKO, Glostrup, Denmark) for 15 min prior to the vWF procedure only. Fibrinogen required no pretreatment. Endogenous peroxidase activity was quenched with an aqueous solution of 3% (v/v) hydrogen peroxide and nonspecific antibody binding was blocked by incubating sections with DAKO Protein Block Serum-Free solution or by incubation in 100 µl of a 5% (w/v) BSA solution in 20 mM Tris-buffered saline, pH 7.4, containing 0.2% (v/v) Tween (TBST) for 20 min at 20°C. Sections were incubated in appropriate dilutions of primary antibody for 1 hr at 20°C. Dilutions and wash steps were carried out using TBST. Incubation in pre-immune serum or incubation in the absence of primary antibody served as negative controls. Tissue incubation in primary antibodies used rabbit anti-human fibrinogen or rabbit anti-human vWF (DAKO, Glostrup, Denmark) at 1:800 for 1 hr at 20°C in a Labvision automated immunostainer. After primary antibody incubation, sections were washed in TBST for 5 min at 20°C and incubated with DAKO EnVision + system-labeled polymer (peroxidase-labeled polymer conjugated to goat anti-rabbit or goat anti-mouse immunoglobulins) for 30 min at 20°C. Sections were then washed in TBST for 5 min at 20°C. Peroxidase activity was developed with a buffered DAB chromogen solution (DAB kit, DAKO, Glostrup, Denmark) in Tris-buffer at room temperature for 10 min. Tissues were counterstained in Carazzi's hematoxylin for 1 min before being dehydrated, cleared, and mounted.
Results
Early Markers of Vascular Injury
Physiological Measurements (Table 1)
ZD6169
In experiment 1, conventional measures showed a large, rapid increase in heart rate compared with pre-study values in animals dosed with 60 or 240 mg/kg of ZD6169 of 70% or 69%, respectively, 1 hr after dosing, with lesser increase seen following 15 mg/kg ZD6169. Doses of either 60 or 240 mg/kg ZD6169 generated substantially reduced BP of a similar degree (approximately 50% 1 hr post dose), but those following dosing with 15 mg/kg ZD6169 were changed to a much lesser degree (BP 22%; HR 22%).
In experiment 2, telemetry data showed clear shortening of PR and QT interval (data not shown) during the period of increased HR, which was particularly noticeable between approximately 30 and 90 min post dose and associated with the degree of increased HR. HR remained elevated for approximately 14 hr post dose. Mean systolic and diastolic blood pressures for these two animals were reduced by 38% and 49%, respectively, 1 hr post dose by comparison with 1 hr pre-dose values.
ZD1611
No consistent changes were observed in any physiological parameters monitored in animals dosed with 200 mg/kg ZD1611 in either the conventional or the telemetry data. Conventionally recorded pre-test HRs were higher than those observed in the telemetry data, which is considered to be normal for this invasive technique.
Histopathology
Histopathological features of the dog heart following dosing with ZD6169 had two aspects. First, cardiac myocardial necrosis, notably of the left ventricle and of the left ventricular papillary muscle in animals given either 60 or 240 mg/ kg ZD6169, and second, coronary arterial injury that was present in both extra- and intramural arteries, in small-to-large vessels in atria and ventricles on both sides of the heart. In association with the arterial and some arteriolar damage, right atrial hemorrhage was observed and also, less commonly, some subendocardial hemorrhage. It is important to note that the arterial lesions seen in this study were variable in type and extent at the only sampling time employed of 24 hr post dose: several lesions were seen in the heart of any one animal and varied from those showing smooth muscle necrosis only, to those that were more advanced inflammatory lesions. In contrast to the dose-dependency of the incidence of myocardial necrosis, the arterial lesions were more variable in appearance, independent of dose.
The pathological changes in H&E-stained sections of coronary arteries 24 hr post dosing with ZD6169 were numerous and illustrate the range of changes that take place within the vessel prior to inflammatory cell ingress that clearly occur within a few hours of dose administration. Some arteries showed only a few condensed medial smooth muscle cells that appeared to be separated from their neighbors by an expanded intercellular space. Most often, several such changes were present in coronary arteries at 24 hr post dose (Figures 1b–d, 4a, and 5a). These minor lesions contrasted with those which exhibited substantial medial smooth muscle cell condensation (seen as focal areas of hyperchromasia) that were often present in a single segment of the media (Figures 1b and 5a ), while others showed additional features of intramedial erythrocytes (Figures 1c, 1d, and 5a) and rarely, a neutrophilic infiltration (Figure 1d). Indeed, the multiple, intramedial aggregates of erythrocytes present in numerous locations within a single arterial section emphasizes the severity and extent of the structural changes that take place within 24 hr permitting blood penetration deep into the vessel wall. At this time post dose, it was relatively uncommon to observe inflammatory cell infiltrates in the arterial media associated with these lesions. More frequently, a few neutrophils or, extravascular hemorrhage were seen in extra-adventitial connective tissue. Smooth muscle cell necrosis was most often seen in the outermost medial layers and less frequently in subintimal layers, although occasionally, transmedial lesions were observed that extended from intimal to adventitial locations. In a few instances, vacuolated smooth muscle cells occurred immediately adjacent to areas of medial necrosis (Figure 1c). No overt degeneration of intimal endothelial cells was seen, although the cells were often rounded and projected into the luminal space in contrast to their normally flattened appearance. Apoptotic cell death was not a feature of the coronary arterial pathology observed in these experiments.
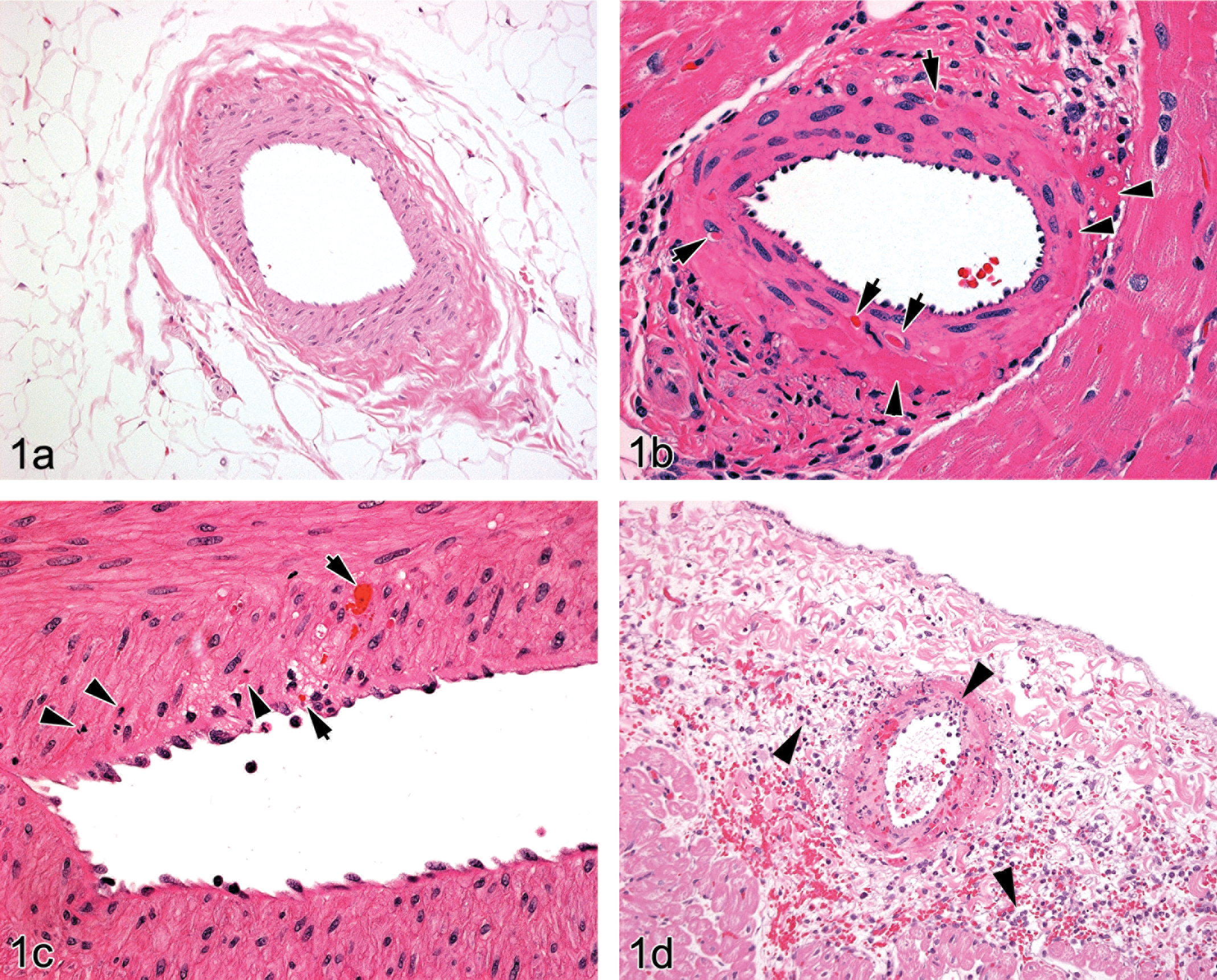
(a). Canine coronary artery—control (untreated) showing the features of an undamaged vessel (H&E stain, original magnification ×100) and compared with drug-induced arterial injury at 24 hr post dose. (b). 24 hr post ZD6169 dose: left atrial, medium-sized intramural artery—multifocal, segmental medial and adventitial hyperchromasia (arrowheads) with single, condensed smooth muscle cells (arrows). Note the different appearances of luminal erythrocytes and condensed medial smooth muscle cells. (H&E stain, original magnification ×400). (c). 24 hr post ZD6169 dose: right atrioventricular groove, medium-sized artery—focal medial vacuolation with inflammatory cell infiltration adjacent to disrupted internal elastic lamina with some local endothelial cell reaction and deeper, focal medial hemorrhage (arrows): medial smooth muscle cells in this area show nuclear pyknosis/cytoplasmic hyperchromasia (arrowheads) by comparison with others which appear normal (H&E stain, original magnification ×400). (d). 24 hr post ZD6169 dose: right atrial, medium-sized, extramural artery—note the medial and adventitial hyperchromasia indicative of cellular degeneration/necrosis with medial and adjacent tissue hemorrhage and acute inflammatory cell infiltration (arrowheads). The media of this vessel is almost entirely necrotic. Endothelial cell reaction apparent as raised and rounded cell profiles is also present (H&E stain, original magnification ×200).
Immunohistochemistry
Background (artifactual) staining with either of the immunostaining methods was absent or minimal. Positive staining was always distinct from this and showed as a moderate to intense brown coloration.
vWF
In vessels from untreated dogs, following anti-vWF immunostaining, only endothelial cells stained uniformly in arteries, veins, and capillaries throughout the heart and acted as an internal positive control (Figure 2a). The media and adventitia were devoid of vWF positivity. In many vessels, plasma was also positively stained when retained in lumina following tissue processing. Of the 12 heart samples examined from control dogs, vWF staining in addition to that normally present in the endothelial cells was seen only in those vessels showing spontaneous arteritis (one dog), in which case it was present predominantly in the media and associated with necrosis and inflammatory cell infiltrates. In all other vessels in this animal and in all others in this group, vWF staining was restricted to the vascular endothelium.
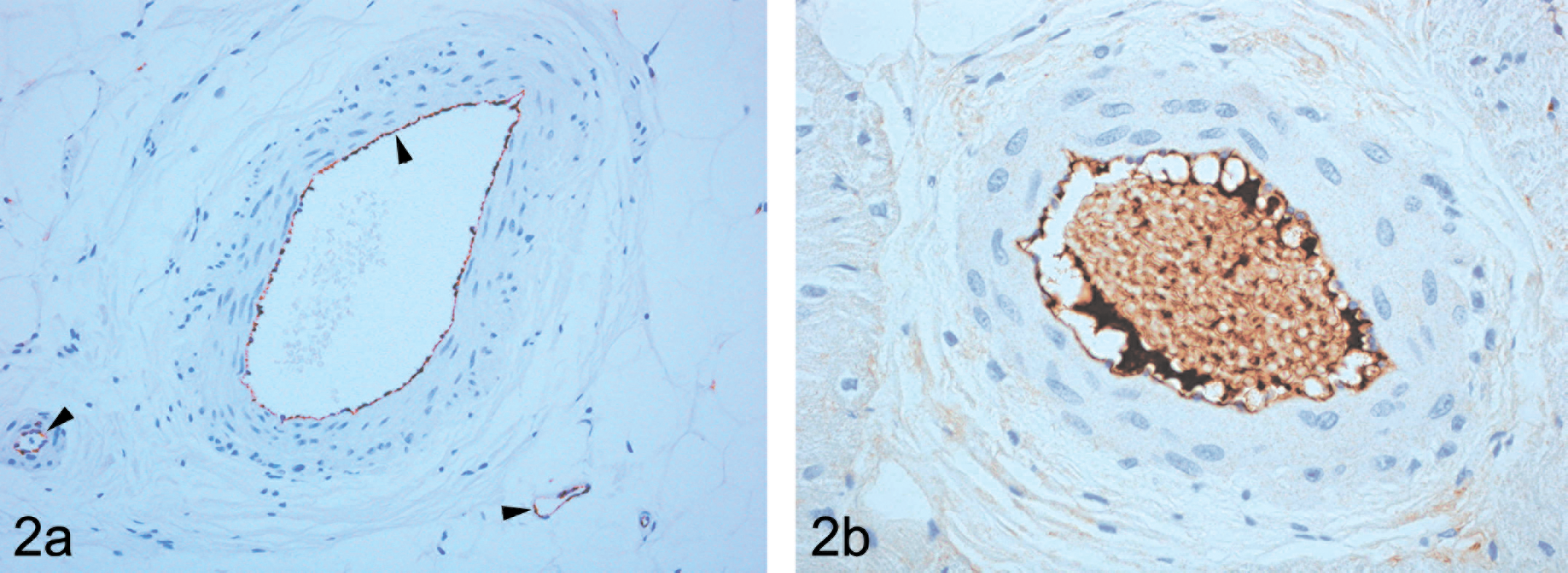
Control animal—(a). extramural large and small arteries and vein, immunohistochemically stained with von Willebrand Factor (vWF), showing endothelial cell staining only (indicated by arrowheads) and illustrating the uniform tissue distribution of vWF in the vasculature (original magnification ×200); (b). fibrinogen present only as immunopositive material in an adherent layer adjacent to the endothelial cell luminal surface and in plasma between luminal blood cells. Note, that no medial or adventitial staining is present in either image, which is a characteristic feature of structurally intact vessels (original magnification ×400).
In arteries from dogs given ZD6169 showing small medial lesions, vWF staining of the arterial media consisted of two types, namely, a generally homogeneous type with staining of mild intensity and another of a distinctly heterogeneous nature with moderate/marked staining intensity (Figures 3, 4b, and 5b). Both types were usually present together, normally orientated in the plane of the medial smooth muscle cells, and it was apparent from examination of the adjacent H&E-stained sections that hyperchromatic condensed smooth muscle cells as well as fragmented and condensed elements of necrotic smooth muscle cells were both stained, a useful artifact of immunostaining (Figures 4b and 5b). The homogeneous vWF staining appearance was present in smooth muscle cells showing early necrotic changes (mild hyperchromasia on H&E staining) without fragmentation. Adjacent, intact smooth muscle cells were frequently separated by narrow ribbons of immunopositive material (Figures 4b and 5b). In some instances, these appeared to track from sites immediately subjacent to the intimal layer and could be traced deep into the medial layer (Figures 3, 4b, and 5b). Any attempt at distinction between artifactual background staining of necrotic debris and staining of intramedial plasma in combination with this debris was difficult, the probable reality being a combination of the two in most circumstances. In numerous instances, adventitial and extra-adventitial (perivascular) immunostaining was also present, usually, but not exclusively adjacent to medial lesions. Substantial adventitial immunopositivity was frequently observed around arteries indicative of plasma (vWF) diffusion into this layer from adjacent minor segmental medial lesions or possibly, other adjacent lesions situated out of the plane of section.
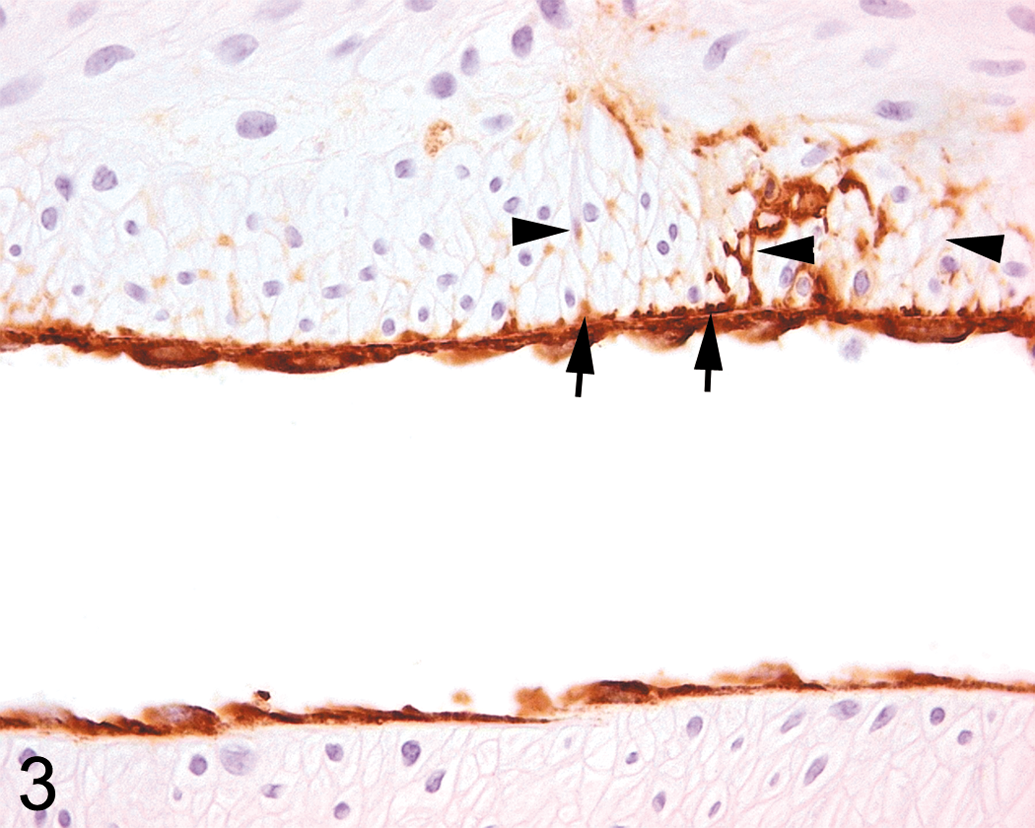
Right atrial extramural artery (LS) from dog 24 hr post ZD6169 dose—vWF immunohistochemical staining shows the characteristic uniform staining pattern of the endothelial cell layer only, with no medial staining on one side of the artery which is typical of the staining pattern in uninjured vessels. By contrast, on the contralateral side, minimal, focal medial staining between smooth muscle cells extends deep into the media (arrowheads). An area of locally intense staining penetrating between smooth muscle cells connects a subintimal pool of positively stained material (arrows, likely plasma, possibly with some necrotic cellular debris) to deeper medial locations (original magnification ×400).
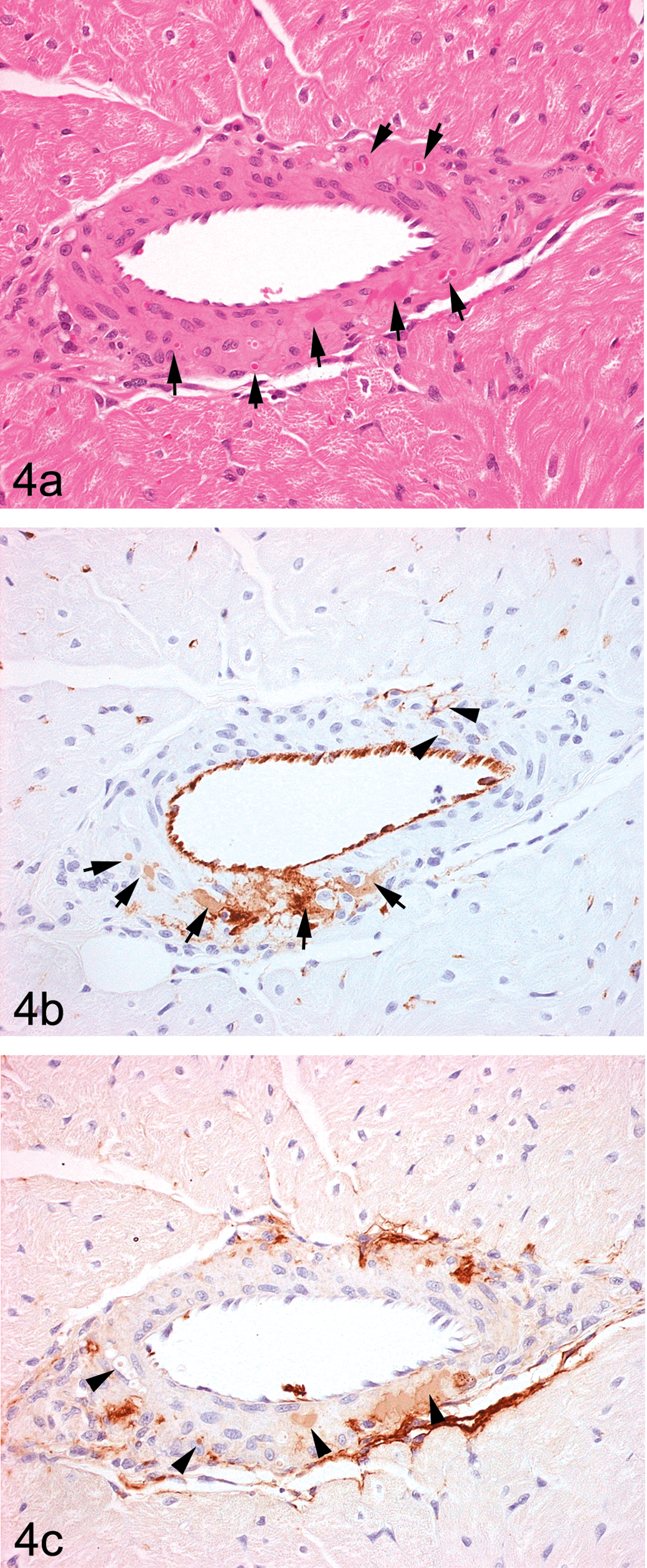
Left atrial intramural, medium-sized artery from dog 24 hr post ZD6169 dose: (a). minimal, multifocal medial hyperchromatic, condensed smooth muscle cells (arrows) illustrative of an early lesion (H&E stain); (b). semiserial section of the same vessel showing intensely staining heterogeneous and paler homogeneous medial staining
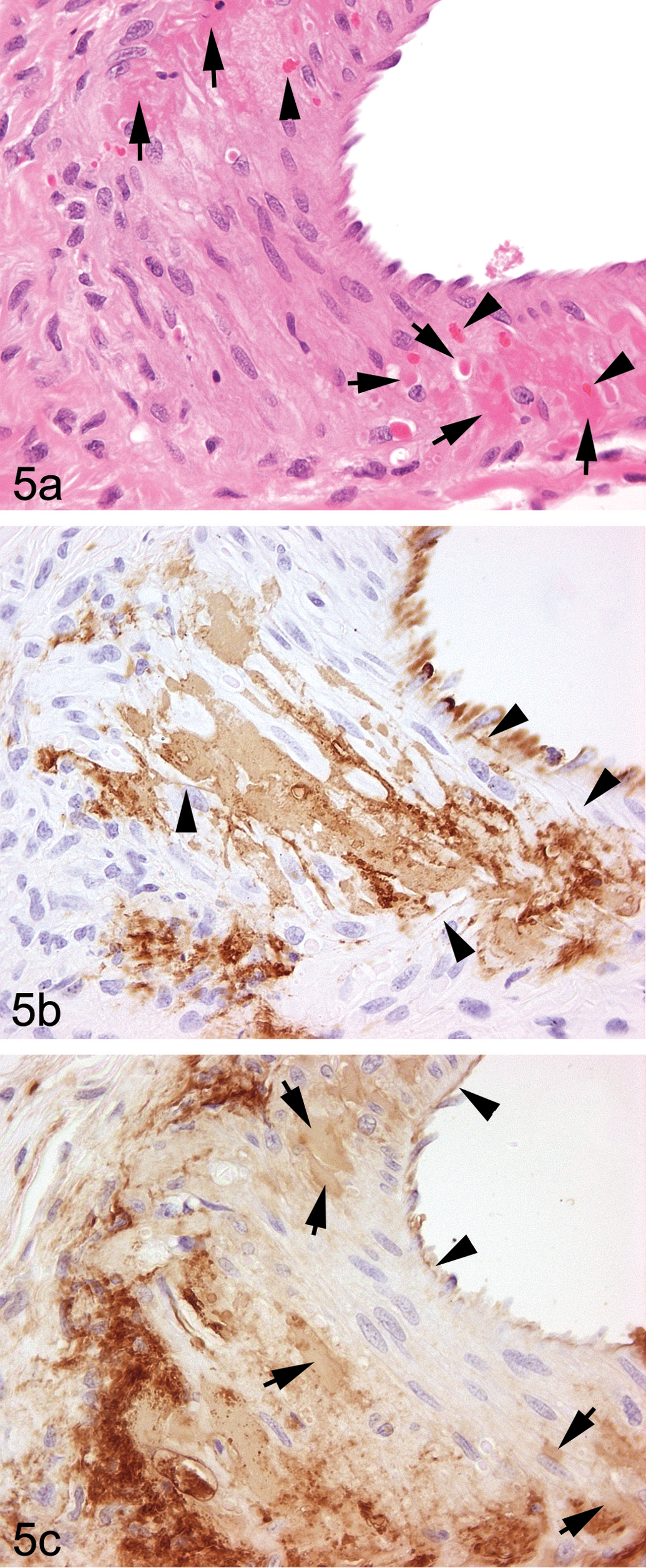
Right atrioventricular groove, extramural medium-sized artery from dog 24 hr post ZD6169 dose: Oblique, semiserial sections. (a). An area of arterial injury showing smooth muscle cells of normal histological appearance lying immediately subjacent to the intima and surrounded by areas of tissue disruption characterized
Fibrinogen
Fibrinogen is not a constitutive component of arterial compartments and within the vasculature is present only in plasma (Haidaris et al. 1989). In immunostained sections from untreated dogs (Figure 2b), fibrinogen was evident either as an adherent layer on the luminal aspect of endothelial cells or in association with erythrocytes when present. In these dogs, as with vWF staining, fibrinogen immunopositivity was present neither in the media nor in the adventitia of undamaged arteries. It was seen only in those vessels in one dog showing spontaneous arteritis where it was present predominantly in only those parts of media associated with necrosis and inflammatory cell infiltrates.
In ZD6169-induced arterial lesions, the distribution of fibrinogen immunopositivity was similar to that of vWF with some important exceptions. A significant benefit of the use of fibrinogen immunostaining was that plasma ingress immediately subjacent to the endothelial cell layer was readily observable because of the absence of the confounding influence of the constitutive vWF staining in this layer. While fibrinogen staining paralleled closely the vWF staining in damaged arterial media, the staining of the adventitia and extra-adventitial tissues was often substantial, being considerably greater than that shown by vWF (Figures 4c and 5c).
No detailed description of the appearances of coronary arteries 24 hr post dose with ZD1611 is given in order to avoid repetition, due to their characteristic features being identical to those lesions in ZD6169-dosed animals. For detailed description of lesions induced by ETRAs, please see Jones et al. (2003).
Cardiovascular Physiological Studies (Experiment 3)
General Haemodynamics (Figure 6a–c)
ZD6169 (Potassium Channel Opener)
Immediately after beginning 30 min intravenous administration of 5 mg ZD6169, MAP significantly decreased by 40% with a contemporaneous 30% decrease in total peripheral resistance (TPR) and cardiac inotropy measured as max dP/dt. Mean aortic pressure, TPR, and cardiac inotropy recovered to within 10% of baseline levels within 60 min after initiation of ZD6169 infusion with a simultaneous, approximately 20% increase in heart rate.
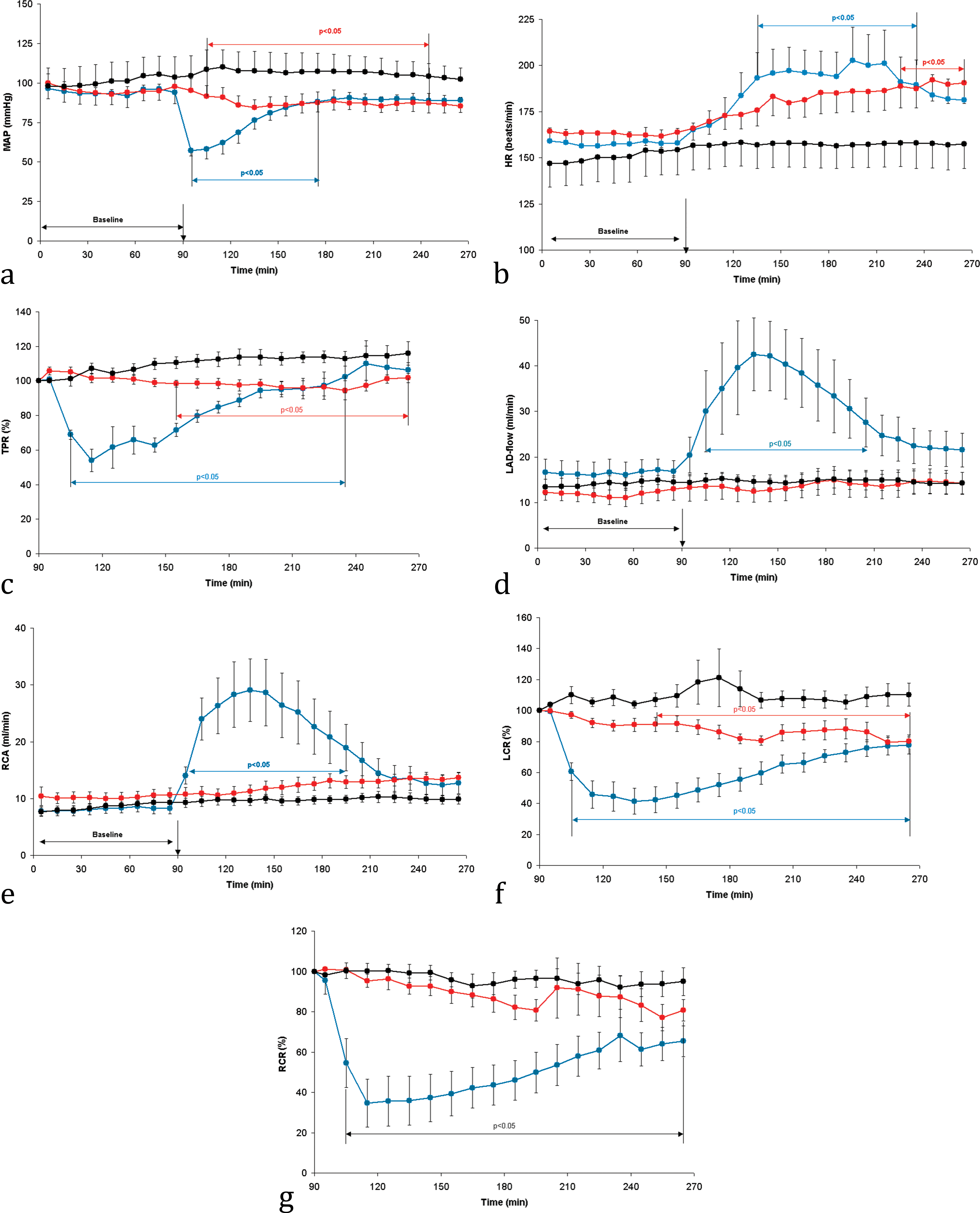
Cardiovascular physiological parameters of anesthetized dogs infused with vehicle (black, 1 ml/kg for 10 min), ZD1611 (red, 10 mg/kg for 10 min), or ZD6169 (blue, 5 mg/kg for 30 min) monitored for 3 hr following initiation of infusion at end of baseline period of 90 min. Data are mean values ± 1 SEM (n = 6 per group) and Student’s paired or unpaired t-test used to assess differences between pre- and post-dose values. For the two vehicle groups, (n = 6 (2 × 3)), data were pooled as no differences between them were evident. (a). mean arterial blood pressure (MAP, mm Hg), (b). heart rate (HR, beats/min), (c). total peripheral resistance (TPR, % baseline), (d). left anterior descending coronary artery blood flow (LAD flow, ml/min), (e). right coronary artery blood flow (RCA-flow, ml/min), (f). left anterior descending coronary artery resistance (LCR, % baseline), (g). right coronary artery resistance (RCR, % baseline).
ZD1611 (Endothelin Antagonist)
Administration of 10 mg/kg ZD1611 caused a minor post-infusion decrease in MAP (10 to 20%) lasting for more than 2 hr and, following a gradual HR rise beginning at initiation of infusion, a small statistically significant increase in HR (8%) compared with predose values occurred at about 3.5 hr.
Coronary Flow Hemodynamics (Figures 6D–G and 7)
ZD6169
A marked rise in both LAD and RCA flows appeared almost immediately following initiation of ZD6169 administration and lasted for about 1.5 hr. The maximal increase in LAD and RCA flows amounted to about 150% and 250% of pre-dose infusion values, respectively. A minor decrease in TPR of <10% of baseline occurred from approximately 1 hr post-initiation of infusion to the end of the experiment. Both LAD and RCA resistance were statistically significantly reduced from about 10 min post-initiation of infusion and maintained for the remainder of the experiment.
Substantial changes in phasic coronary arterial flow appeared both in the right and left coronary arteries (Figure 7a), such that 30 min following initiation of ZD6169 infusion, early systolic flow in the LAD increased 7-fold and the characteristically small, negative systolic flows observed under control conditions, became more pronounced (30- to 50-fold increase) in mid- to late systole. The systolic flow pattern changes in both coronary arteries had reduced substantially from these large increases at 2 hr and at 3 hr post-infusion initiation were at normal levels. Diastolic flows in both coronary arteries were increased slightly at 30 min and 2 hr post-initiation of infusion but at neither time were the increases comparable to those seen at systole.
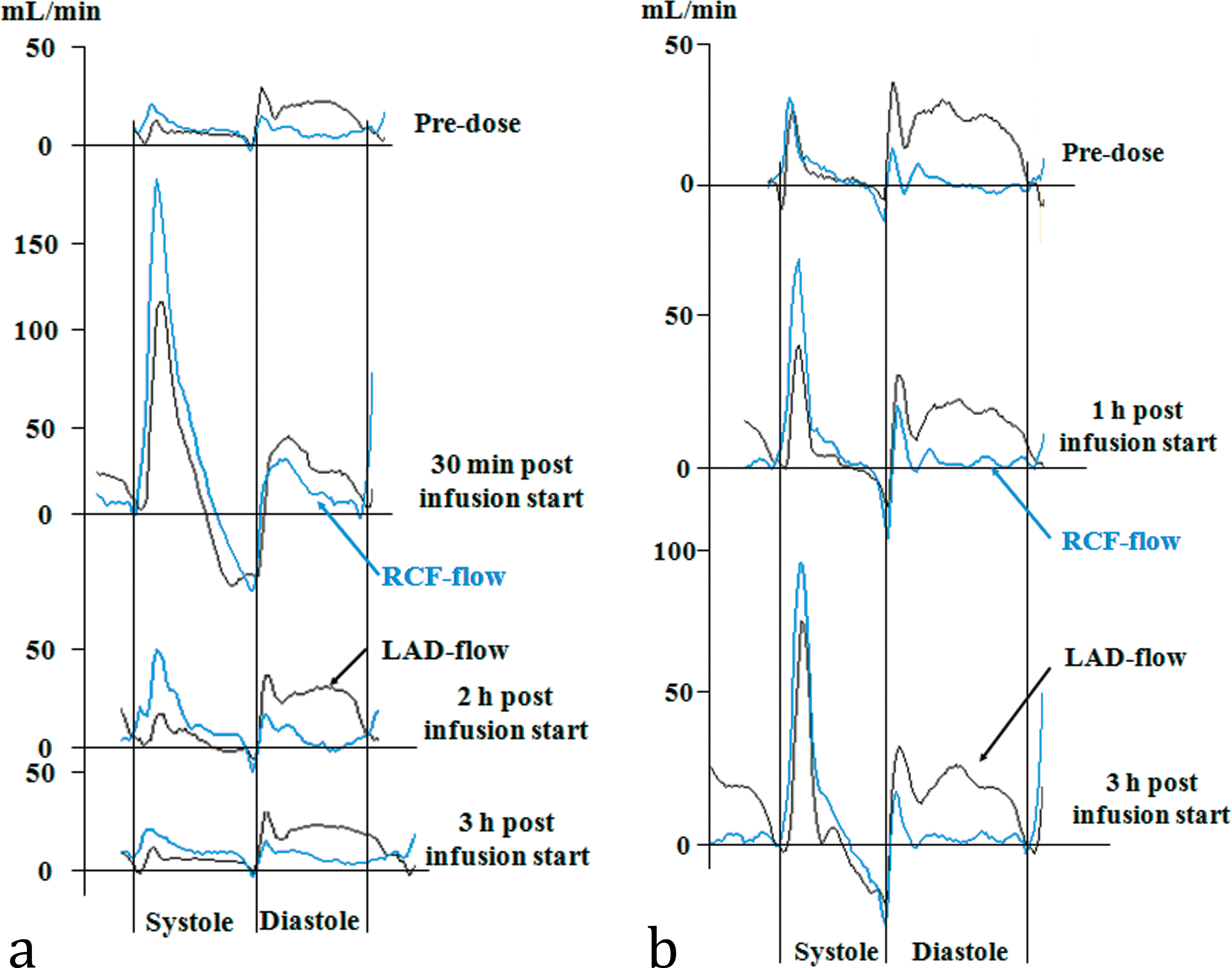
Single cardiac pulse cycle showing flows (ml/min) in right coronary artery (RCA flow, blue) and left anterior descending artery (LAD flow, black) pre- and post-dose of A. ZD6169 and B. ZD1611. In (a). systolic and diastolic flows are markedly and mildly elevated, respectively, in both right and left coronary arteries at 30 min following initiating drug infusion. At 2 hr, systolic and diastolic flows in the left anterior descending artery only are slightly elevated but much reduced by comparison with earlier maximum flows. It is noteworthy that right coronary arterial flow has returned to pre-dose values at this time. Both arterial blood flows have returned to pre-dose magnitude and profiles at 3 hr. In (b). both coronary arterial flows were elevated during systole 1 hr post-initiation of infusion with greater increases apparent 2 hr later. No changes in coronary arterial blood flows during diastole occurred at any time following initiation of ZD1611 infusion.
ZD1611
ZD1611 infusion did not induce increased LAD-flow. However, RCA flow increased more in animals treated with ZD1611 than animals in the vehicle control or pre-infusion groups, with maximum increase present at the end of the experiment, i.e., 3 hr after initiation of drug administration.
Left and right coronary arterial resistance declined gradually from initiation of ZD1611 infusion to the end of the experiment and was only statistically significant in the former from about 1 hr post-initiation of infusion to the end of the experiment. Significant changes in phasic coronary arterial flows occurred following infusion of ZD1611, which by comparison with those due to ZD6169 occurred gradually and were maximal at the end of the experiment 3 hr later. Early, increased positive and late reverse systolic flows in both arteries appeared first at 1 hr post-initiation of infusion and were greater at 3 hr with diastolic flows being unaffected at any time. Maximal but not statistically significant increases in LAD and RCA blood flows of about 17% and 31%, respectively, occurred. After treatment with ZD1611, the coronary artery flow pattern slowly changed and was more pronounced at 3 hr than at 1 hr, such that early systolic flow increased 29% and 45% for LAD and RCA, respectively, with a 9- to 12-fold increase in late systolic back flow (Figure 7b).
Histopathology
Several animals showed unequivocal evidence of cardiac or coronary arterial damage at 3 hr following the initiation of drug infusion.
ZD6169
Left ventricular myocardial necrosis in the ventricular wall, papillary muscle, and subendocardial locations were observed after infusion of this agent only. Vascular injury was noted in left ventricular, intra- and extramural coronary arteries of medium-sized or large dimensions. Characteristically, these lesions presented as small, focal areas of medial smooth muscle cell hyperchromasia (hypereosinophilia) and represented localized cytoplasmic condensation (degeneration/necrosis) as illustrated in Figures 1b and 4a. In some cases, minimal acute inflammatory cell infiltration and medial hemorrhage were also present. Immunohistochemical staining for vWF or fibrinogen effectively demonstrated the penetration of plasma into the media of coronary arteries similar to that shown in Figure 4b and c.
ZD1611
The only pathological changes observed were vascular lesions as described above for ZD6169 that were located in extramural vessels in the right atrioventricular groove and left atrium and ventricle (Figures 1b and 4a).
No myocardial necrosis was observed following infusion of ZD1611.
Discussion
During normal function, myocardial contraction impedes vascular perfusion of the heart. A gradient of tissue pressure between epicardial and endocardial regions occurs that reduces blood flow and tissue oxygenation, most significantly in subendocardial areas. Most coronary vascular flow occurs during systole but the inner (subendocardial) tissues are perfused during a small fraction of the cardiac cycle, namely, during diastole when coronary arterial pressure exceeds local myocardial pressure (Greaves 1998). The coronary circulation is an autoregulated vascular bed directly influenced by mechanical, metabolic, anatomic, and myogenic factors and myocardial perfusion is balanced sensitively and controlled by vasoconstriction and vasodilatation of microvascular elements (Feigl 1987). Greaves (1998) indicates numerous factors that predispose to myocardial ischemia. Failure of autoregulation of medial tonal decreases during ZD6169 exposure likely leads to poor perfusion of susceptible vascular beds during diastole such as in subendocardial and papillary muscles due to coronary steal, leading to ischemic myocardial necrosis. The lesser degree of arterial dilatation and gradual onset of minor coronary vascular flow and resistance changes with potential accommodation of flow due to other mechanisms (Wilcken 1983) may account for the absence of ischemic myocardial necrosis following ZD1611 infusion, while being of sufficient magnitude to induce multiple coronary arterial lesions. ZD6169 administration resulted in decreased peripheral cardiovascular resistance associated with reduced BP, tachycardia, increased cardiac output, and coronary arterial dilatation, which, taken together, indicate that coronary arterial flow is likely to be significantly perturbed, with vessels dilating during diastole, retaining an abnormal blood volume such that the systolic reflection wave results in an abnormal reversed flow of blood. Indeed, such conditions are conducive to the development of a “water (or blood) hammer effect,” i.e., the rapid generation and propagation of a pressure pulse wave due to sudden stoppage of flow. It is probable that depending on their magnitude, such pressure waves will rapidly contribute to and promote coronary arterial wall damage.
While the influences of pro-inflammatory mechanisms induced by shear stress in atherosclerosis have been recognized (Malek et al. 1999), the effects of drug-induced coronary vascular flow changes on endothelial cell function are less well known. In our study, both drugs induced very substantial changes in bidirectional oscillatory flow patterns with increased velocity during the cardiac cycle, particularly during systole, that potentially generated pro-inflammatory processes (Sucosky et al. 2009). However, the rapid induction of coronary arterial damage occurring within 3 hr of initiation of infusion but showing no or little inflammatory cell infiltration argues against such a primary route to vascular damage bolstering a view of mechanical disruption due to medial dilatation as the most plausible cause.
An objective of our studies of this model was to determine whether the use of two plasma-borne proteins, namely vWF and fibrinogen, shows potential as sensitive morphological markers of disruption of the wall of the coronary artery by indicating the presence of non-luminal plasma, changes that may prove helpful in ascertaining timing of lesion generation related to potential plasma biomarkers of DIVI. While fibrinogen is produced predominantly in the liver and not constitutively expressed in arteries (Haidaris et al. 1989), vWF is located in Weibel-Palade bodies found in the cytoplasm of endothelial cells and released into plasma (Mannucci 1998). Our rationale for the use of immunohistochemical staining for these two molecules was that they are integral constituents of plasma and are therefore appropriate indicators of plasma penetration into vascular and extravascular tissues at sites of arterial injury. Immunohistochemical staining demonstrated that even in minimal arterial lesions, and those that were generated within 3 hr of drug exposure, substantial penetration of immunopositive material into the media was present at sites in H&E-stained sections where no damage was apparent. The presence of fibrinogen immunopositivity can only be due to the leakage of plasma into the vessel wall since this protein is only expressed in plasma. By contrast, the presence of vWF within coronary arterial tissue may be due to a combination of plasma ingress and/or leakage from damaged endothelial cells. The immunostaining between medial smooth muscle cells that was visible as thin, brown lines represents plasma ingress into the arterial wall. The appearance and pattern of distribution of these immunopositive tracks indicated that they represent plasma ingress (and subsequently, probably blood cells also) into the expanded interstitial space between smooth muscle cells due to disrupted structural and functional integrity of the endothelial cell layer. The pulsatile movement of the artery in the living animal with frequent expansion and recoil is likely to serve as a means of pumping plasma into the media via this damaged endothelial cell layer and, perhaps, lead to expansion of the initial lesion by further, hydrostatically induced separation of medial smooth muscle cells. As the pharmacological effect of a KCO such as ZD6169 results in medial smooth muscle relaxation, it is likely that damage may progress more rapidly in arteries that display flaccid media. Frequently, lesions were encountered that showed remarkable, substantially diffuse, intercellular medial immunostaining, supportive of the notion of hydrostatically induced expansion of initially, rather minor lesions. In several instances, in both fibrinogen and vWF-immunostained arteries, dense positively immunostained homogeneous material lay immediately subjacent to the endothelial cell layer and may be indicative of a local “pool” of plasma immediately adjacent and able to enter the media as a consequence of endothelial cell compromise (Figure 3). In these cases, the adjacent undamaged tissue and that on the contralateral side of the artery showed no such change.
The disruption of the normal, structurally contiguous, endothelial cell layer to yield a porous, functionally compromised barrier is not unique to our findings in this study of a KCO. Indeed, ultrastructural investigations by Joseph, Jones, and Kerns (1996) and Jones et al. (2003) have demonstrated similar morphological compromise following administration of single doses of either SKF95654 (phosphodiesterase III inhibitor) or ZD1611 (ETRA) to dogs. Following a single, 2 hr infusion of SKF 95654, Joseph, Jones, and Kerns (1996) reported similar histological lesions to those described in our study, 24 hr later in perfusion-fixed hearts. In their study, in control animals, coronary arteries showed a flattened, intact endothelial cell layer with neither visible gaps between the tightly apposed cells nor adherent inflammatory cells by scanning and transmission electron microscopy. At lesion sites, the endothelial cell layer was markedly different from this appearance and showed raised and rounded endothelial cells with numerous intercellular gaps and adherent inflammatory cells, some of which were present within these gaps and indicative of post-adhesion migration. Some of these endothelial cells were necrotic. Not unexpectedly, plasma was not visible in these perfusion-fixed tissues as a potential indicator of an unusual distribution at lesion sites. Jones et al. (2003) demonstrated similar changes 24 hr following a single oral dose of ZD1611 and also noted breaks in the internal elastic lamina at lesion sites.
In attempting to identify biological indicators of vascular damage that will be relevant in ensuring the safety of human volunteers and patients in clinical assessment, the gold standard biomarker is one that can be detected prior to induction of physical injury. In the absence of such an ideal biomarker, the appearance in plasma of tell-tale molecules at the time of early tissue damage may, on close monitoring, provide an opportunity to cease drug administration and permit tissue repair of minimal damage. A strategy based upon biomarker monitoring in circumstances of rigorously monitored human clinical drug assessment requires that alterations in plasma concentration of a target molecule truly reflect a change in tissue integrity (Brott, Jones et al. 2005). A biomonitoring process that has no basis in thorough validation is, at best, of no value and, at worst, potentially hazardous to humans. In this circumstance, and in the absence of human coronary artery biopsy samples obtained from drug evaluation studies in the clinic, it is vital that a selected biomarker has been validated and is known to represent arterial damage in vivo, albeit in animals. Fundamentally, it is an issue of transference of understanding of biological mechanisms between species obtained from data derived through animal studies. For biomarkers of acute DIVI, an expectation might be that cellular constituents that are associated with the major cell types that make up vascular structure, namely, endothelial and smooth muscle cells should be present in plasma. Newsholme et al. (2000) showed that fenoldopam-induced increases of plasma vWF in rats at 2 and 6 hr post dose returned to control (saline infusion) values at 24 hr. These plasma biochemical changes did not correlate with the appearance of morphological evidence of arterial damage in mesenteric arteries at these early times but did so at 24 hr: no immunohistochemical assessments were employed in their study. They also reported elevated plasma vWF values in saline-treated rats after the minimal disturbance of repeated venipuncture and others have demonstrated plasma vWF increases following noninjurious stimuli namely, vascular catheterization (Abu-Zidan et al. 1999; Wagner 1990), exercise, or adrenalin administration in humans (Prentise, Forbes, and Smith 1972). Brott, Gould, et al. (2005) demonstrated that animals given either 60 or 240 mg/kg ZD6169 showed increases in plasma vWF of more than 60% of prestudy values, as early as 1.5 hr post-dosing that remained at 6 hr and gradually declined below pre-study levels thereafter to 24 hr post dose. Brott, Gould, et al. (2005) and Newsholme et al. (2000) have suggested that vWF plasma levels fall quickly post-dosing due to several possible causes, and the conclusions of these latter publications and those of Brott, Gould, et al. (2005) and Louden et al. (2006) were that vWF plasma levels alter due to numerous stimuli, are rapidly transient, and thus render this molecule inappropriate for use as a specific biomarker of DIVI. However, we have demonstrated in our studies that its presence within plasma makes it a useful diagnostic indicator of plasma penetration into damaged arteries caused by ZD6169 and ZD1611.
Footnotes
Acknowledgments
The contributions made by staff of the Histology, Analytical Morphology and Safety Pharmacology Groups, AstraZeneca Pharmaceuticals, Alderley Park, Macclesfield, UK, are much appreciated.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
