Abstract
Testicular tubular dilatation and degeneration and epididymal sperm granulomas were frequently seen in 4-week toxicity studies using different phosphodiesterase-4 (PDE4) inhibitors in Wistar rats, including the prototypic PDE4 inhibitor BYK169171. To investigate the pathogenesis of testicular and epididymal lesions, a time course study with BYK169171 was conducted with sequential necropsies after 7, 14, 21, and 28 days of treatment. After 7 days, a dilatation of efferent ducts and of the initial segment of the epididymis and a subacute interstitial inflammation were seen followed by a diffuse dilatation of seminiferous tubules in the testis. Dilatation and inflammation were most pronounced after 14 days. Single animals also exhibited vascular necrosis in the inflamed interstitium. Although dilatation decreased later in the study, the incidence and severity of tubular degeneration increased from 14 days onward. Sperm granulomas developed in efferent ducts and in the caput and cauda of the epididymis after 14 days. Our results demonstrate a clear time course of PDE4 inhibition-induced lesions, with dilatation preceding sperm granuloma formation. We conclude that the most likely mechanism of toxicity is a disturbance of fluid homeostasis in efferent and epididymal ducts resulting in abnormal luminal fluid and sperm contents, epithelial damage at specific sites of the excurrent duct system, sperm leakage, and granuloma formation.
Keywords
Introduction
Phosphodiesterases (PDEs) constitute a family of cyclic nucleotide–degrading enzymes. Currently, 11 members of this family have been identified (PDE1–11) among which phosphodiesterase-4 (PDE4), is the major cyclic adenosine monophosphate (cAMP)-hydrolyzing enzyme in mammalian cells. The PDE4 gene encodes four different isoenzymes: A, B, C, and D with multiple splice variants (Richter, Jin, and Conti 2005). Inhibition of PDE4 augments the cellular level of cAMP, thus enhancing biological effects as a consequence of cAMP downstream signaling. In immune cells, inhibition of cAMP degradation has been found to suppress certain inflammatory processes, and selective inhibitors of the PDE4 are currently in clinical development for treatment of inflammatory diseases such as chronic obstructive pulmonary disease (COPD), asthma, rheumatoid arthritis, and psoriasis (Pagès, Gavaldà, and Lehner 2009). Besides its antiinflammatory properties in targeting immune cells, PDE4 inhibitors have recently demonstrated efficacy in the treatment of type 2 diabetes (Rabe 2011).
Published reports on preclinical toxicity studies with PDE4 inhibitors indicate that inflammation and vascular lesions are major toxicities in animals (Hanton et al. 2008; Larson et al. 1996; Losco et al. 2004; Peter et al. 2011). In rats, the most frequently reported target site is the mesentery. Further target sites in rats are the heart, gastrointestinal tract, pancreas, liver, kidney, and the soft tissue of hind limbs (Dietsch et al. 2006; Larson et al. 1996; Mecklenburg et al. 2006; Peter et al. 2011; Sheth et al. 2011; Slim et al. 2003; Zhang et al. 2002, 2008). Although differences exist in localization and degree of vascular injury, which might be related to different doses, potencies, or binding affinities (Wang et al. 2007), lesions seem in general to be related to the pharmacodynamic action of PDE4 inibition in rats. In addition to inflammatory lesions, cortical hypertrophy of adrenal glands (Giembycz 2006) and lymphoid atrophy of the thymus and spleen (Peter et al. 2011) have been described. To our knowledge, no reports have been published on the effects on male gonads after administration of PDE4 inhibitors to rats. There is only indirect evidence that the rodent testes might be a target for PDE4 inhibitors. Theophylline, an unselective inhibitor of PDEs, has induced sperm stasis in dilated tubules of the rete testis in rats (Foley 2001).
Testicular and epididymal toxicity have repeatedly been observed with PDE4 inhibitors of different chemical classes in Wistar rats at our facility. The aim of this article is to report on a 4-week repeat-dose toxicity study with the selective PDE4 inhibitor BYK169171. Toxicity was detected by organ weight measurement and histopathology and was further characterized in a time course study. Efferent ducts that connect the rete testis and the epididymis were identified as an additional target structure of male gonad toxicity. We demonstrate a sequence of events characterized by an initial dilatation of efferent ducts and caput epididymis followed by dilatation of seminiferous tubules in the testis. Sperm granulomas developed subsequently in proximal parts of efferent ducts and in the cauda and caput epididymis.
Materials and Methods
Animals
Male Wistar rats (Crl/WU) were purchased from Charles River Laboratories (Sulzfeld, Germany) and acclimatized for 2 weeks before study initiation. Rats were maintained in polycarbonate cages, 2 rats in each cage, and held under conventional housing conditions. Provimi Kliba Nr. 343 GLP-diet and tap water were provided ad libitum throughout the study. All experiments were approved by local authorities and were performed to International Standards in Laboratory Animal Science.
Test Article and Administration
BYK169171 is a selective PDE4 inhibitor with an IC50 of 0.9 nM for in vitro activity in the human recombinant PDE4B2 enzyme. No active metabolite is formed in rats. The test compound was prepared as a suspension in 4% methocel (w/w) and the control animals received an aqueous 4% methocel suspension. All rats were dosed at a volume of 10 mL/kg orally by gavage.
Four-week Toxicity Study
Animals were 6 weeks old at the start of dosing. Ten animals/sex/group were dosed once daily by gavage with 0, 1, 2, or 5 mg/kg of BY169171. An additional 8 animals/sex/group were treated with either placebo or 5 mg/kg for 28 days followed by a 28-day recovery period. All rats were sacrificed by diethyl ether inhalation and underwent a complete necropsy. Organ weights were obtained from fixed tissues for a standard panel of organs including seminal vesicles, prostate gland, adrenal glands, and testes. Except for testis and epididymis that were fixed in a modified Davidson’s solution, all organs were preserved in 8% neutral-buffered formalin. For histological evaluation of testis and epididymis, a transverse section through the testis containing the area of the rete testis and a longitudinal section through the whole epididymis were made. All tissues were embedded in paraffin, sectioned at 3 µm, and stained with H&E.
Time Course Study
The follow-up time course study was conducted with one dose level only. Based on the findings from the 4-week toxicity study, where 5 of the 10 animals had shown testicular toxicity at the highest dose of 5 mg/kg, the dose for the follow-up study was increased to 10 mg/kg to demonstrate toxicity in a higher number of animals. A total of 45 rats aged 6 weeks at study initiation were treated with 10 mg/kg/day, and groups of animals were sacrificed on study day 7 (n = 10), 14 (n = 10), 21 (n =10), or 28 (n = 15). Age-matched control animals (n = 6) were included for each time point. For evaluation of intratesticular hormone levels, a separate set of 5 animals received either placebo or 10 mg/kg for 28 days.
Hormone analysis
On the day of necropsy, blood was taken from the retro-orbital venous plexus. Blood samples were always taken at the same time of day, in order to limit the influence of circadian rhythms. Testosterone was measured by a solid-phase radioimmunoassay (Diagnostic Products Corporation, Los Angeles, CA), based on an immobilized antibody that specifically reacts with testosterone with little cross-reactivity against dihydrotestosterone (<3.3%), androstenediol (0.4%), androstenedione (0.5%), and other steroids. Inhibin B was determined by an enzymatically amplified 2-site, 2-step sandwich-type immunoassay (Diagnostic Systems Laboratories, Webster, TX) that had been previously validated for measurement of inhibin B in the rat (Woodruff et al. 1996) and whose precision, accuracy, and linearity were verified internally.
In addition, on study day 28, pieces of rat testes were analyzed for intratesticular concentrations of androgens (testosterone and dihydrotestosterone) by radioimmunoassay (RIA) in a subset of animals. Animals were euthanized by diethyl ether inhalation, the inguinal sac was opened, the testis removed, and immediately deep frozen at −80°C. Frozen tissue samples were homogenized and the homogenates were extracted with ethyl ether. The recovery was monitored by the addition of [1β, 2β, 6β, 7β-3H]testosterone. After ether evaporation, extracts were reconstituted in assay buffer and testosterone was measured by a double antibody RIA technique using commercial iodinated tracer and a rabbit antiserum against testosterone-3(carboxymethyloxime)-BSA. This antiserum reacts with rat DHT (100%) and shows little cross-reactivity against androstenediol (1.6%), androstenedione (0.3%), and other steroids (<0.1%; Nieschlag and Lorieaux 1972).
Histology
At necropsy, the abdominal cavity and inguinal sac were opened, and right and left testes and epididymes removed. Organ weights were obtained for seminal vesicles, prostate gland, and adrenal glands. Testis and epididymis were left together for further preparation, and therefore testicular weights were not taken. Efferent ducts were dissected using a light box with a magnifying glass. The epididymal tail was separated from the testis, cut through with a razor blade at the corpus, and corpus and cauda were put into fixative. The fat pad around the epididymal head was carefully removed and the efferent ducts were dissected free of fat while left connected to the rete testis and epididymal head (Figure 1A). Epididymal head, efferent ducts, and testis were fixed together on cardboard to avoid rotation. After fixation in a modified Davidson’s solution for 2 days, a cross section through the middle of the testis was made for evaluation of seminiferous tubules. For evaluation of the excurrent duct system, a longitudinal section was made through the remainder of the testis including rete testis, efferent ducts, and epididymal head (Figure 1B). Finally, a longitudinal section was made through the epididymal corpus and cauda. All sections were embedded in paraffin, sectioned at 3 µm, and stained with H&E.
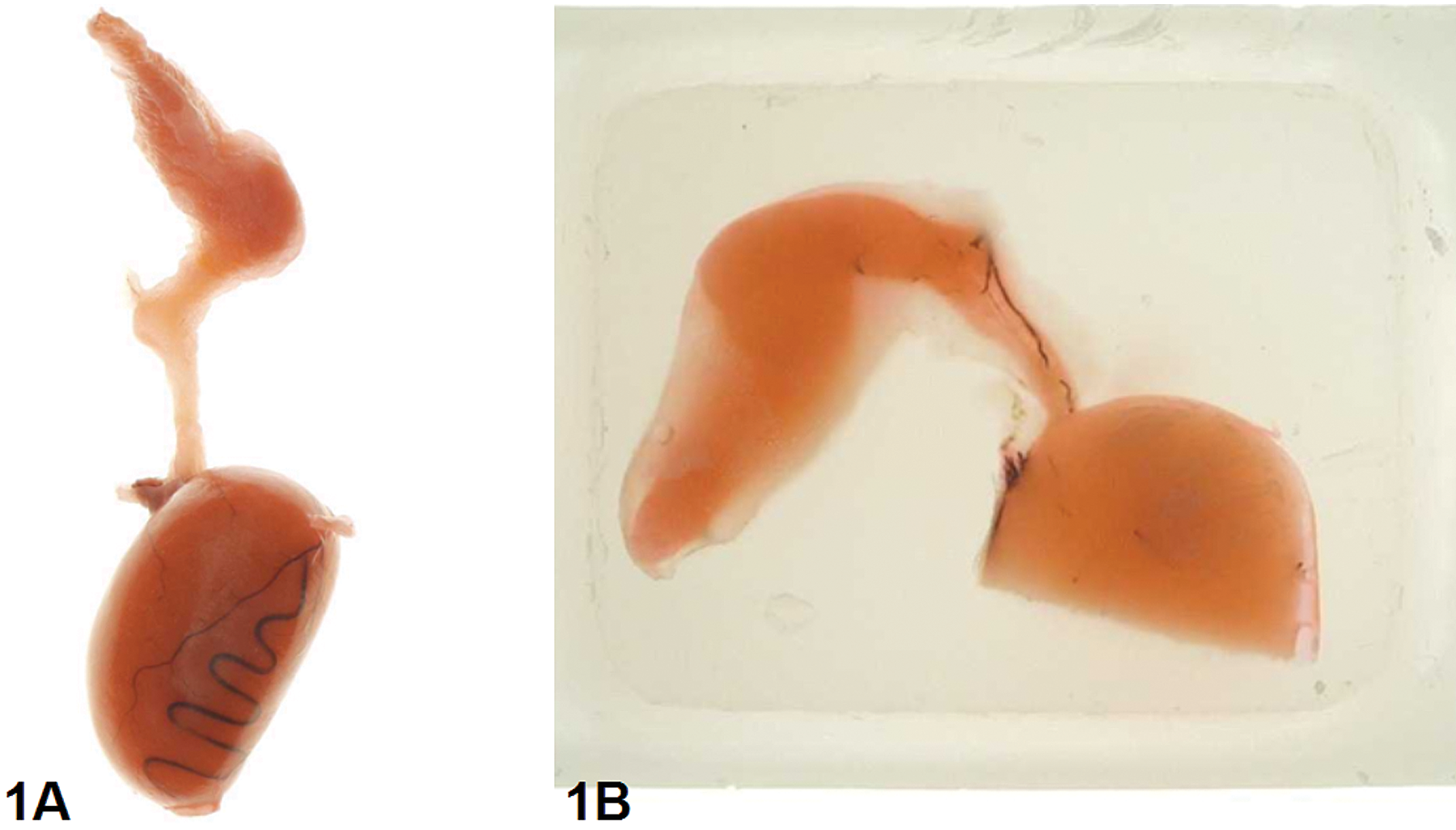
Preparation of efferent ducts. (A) Fixed tissue: The epididymis has been cut through with a razor blade at the corpus. Efferent ducts were dissected free of fat and left connected to the rete testis and epidiymidal head. (B) Paraffin block: A longitudinal section was made through the upper part of the testis including rete testis, efferent ducts, and epididymidal head. Tissue stained with 2% eosin for better differentiation of efferent ducts from surrounding fat tissue.
Nomenclature of testicular lesions
In addition to “germ cell apoptosis,” and “tubular atrophy” (the latter describing tubules with only Sertoli cells left), changes in the germinal epithelium comprised exfoliation of germ cells, germ cell depletion, and spermatid retention. Since no stage or cell specificity of these lesions was seen, the terms exfoliation of germ cells, germ cell depletion, and spermatid retention were summarized under the generalized term tubular degeneration.
Severity grading
The generalized terms tubular degeneration and tubular atrophy were graded according to the proportion of tubules affected into 1 (minimal, <10%), 2 (mild, 10–25%), 3 (moderate, 25–50%), or 4 (marked, >50%). Dilatation of seminiferous tubules and efferent ducts was graded according to the extent of dilatation from minimal to moderate (grade 1–3). For focal or multifocal changes, 0.5 or 0.75 scores were subtracted from the respective grading. Left and right sides were evaluated separately. Group mean values were calculated for individual findings by dividing the sum of all gradings including left and right side by the number of tissues evaluated. Incidences of findings were presented per animal.
Results
Four-week Toxicity Study
Body and organ weights
Animals receiving the high dose of 5 mg/kg/day gained weight more slowly than control animals, resulting in a total body weight gain of 57% after 28 days of treatment versus 80% body weight gain in age-matched controls. Deviations in mean relative organ weight changes for seminal vesicles, prostate, and adrenal glands were within a 10% range. No deviations in mean absolute testicular weights were noted when comparing treated animals with controls. However, on an individual animal basis 3 animals displayed absolute testicular weights that were increased by 30% to 40%, compared to the mean value of concurrent controls.
Histopathology
Increases in testicular weights correlated with tubular dilatation. In addition to dilatation, minimal to moderate degeneration of the seminiferous tubule epithelium was associated with mild oligospermia in the epididymis. Four animals treated with 2 or 5 mg/kg/day developed sperm granulomas in the cauda or distal caput of the epididymis. However, no obstruction of semen outflow was evident. Pertinent BYK169171-related histopathological findings in other organs comprised minimal atrophy of seminal vesicles and prostate glands.
Time Course Study
Mortalities
Compound-related mortalities occurred in single animals on treatment days 15, 16, 21, and 26. At necropsy, these animals had enlarged fluid-filled small intestines and thickened mesenteries correlating histologically with a subacute diffuse mesenteritis as described in a previous article (Mecklenburg et al. 2006). Autolysis of testicular tissue did not allow a detailed histopathological evaluation and animals were excluded from investigations.
Body and organ weights
A dose of 10 mg/kg/day induced high systemic toxicity as indicated by body weight and organ weight changes. Body weight gain reached a plateau after 3 treatment days, and body weight remained constant until the end of treatment week 3. A slight increase in body weight gain was observed during the last treatment week resulting in a total body weight gain of 18% after 28 days of treatment versus 59% body weight gain in age-matched controls. Mean relative weights were significantly reduced for seminal vesicles (−41% vs. control mean) and prostate gland (−18% vs. control mean) and significantly increased for adrenals (+100% vs. control mean).
Histopathology
In 7- and 8-week-old control animals that served as the age-matched control group for the 7- and 14-day treatment groups, seminiferous tubules in the testis were fully populated by germ cells, no exfoliation of germ cells was observed and the epididymes were entirely filled with sperm. However, compared with animals aged 9 weeks and older, sperm content and luminal diameter of the caudal epididymal duct were slightly reduced, indicating that spermiogenesis was quantitatively not fully developed in 7- and 8-week-old animals.
Dilatation of Efferent Ducts and Testicular Tubules
The main finding after 7 days of treatment was dilatation of efferent ducts, the epididymal head, and seminiferous and rete tubules of the testis. In 9 of the 10 animals, dilatation involved the initial segment of the epididymal head and the distal parts of the efferent ducts (Table 1). Although spermatozoa are only rarely detectable in the proximal efferent ducts where they are still extensively diluted within testicular fluids, they become visible in the lumen of the single common distal duct as highly concentrated entangled eosinophilic filaments after more than 90% of testicular fluids have been removed. However, in animals with efferent duct dilatation, spermatozoa remained diluted. Consequently, the lumen of the common distal duct appeared empty and the sperm density of the initial segment of the epididymis was decreased (Figure 2D and F). Testicular tubular dilatation was noted in 6 of the 10 animals as a diffuse change affecting tubules in all stages of the spermatogenesis cycle (Figure 3C). A dilatation of rete tubules was obvious in 3 animals. After 14 days, incidence and severity of dilatation reached its maximum before it gradually declined after 21 and 28 days of treatment (Figure 4). In rats treated for 14 days, dilatation involved proximal and distal parts of efferent ducts as well as seminiferous tubules in 8 of the 10 animals. In rats treated for 21 and 28 days, fewer animals displayed dilatation in the efferent ducts and epididymal head, but the number of animals displaying testicular dilatation was comparable to that seen in rats of the 14-day group. However, the severity of testicular dilatation was much lower after 28 days of treatment, with most animals showing only minimal to mild changes. In addition, the distribution pattern of testicular tubular dilatation was changed from diffuse to focal or multifocal. Concomitantly, dilatation of efferent ducts and epididymal head was only minimal to mild in animals of the 28-day treatment group, and in some animals concentrated spermatozoa were again visible in the lumen of the common distal duct (Figure 2H).
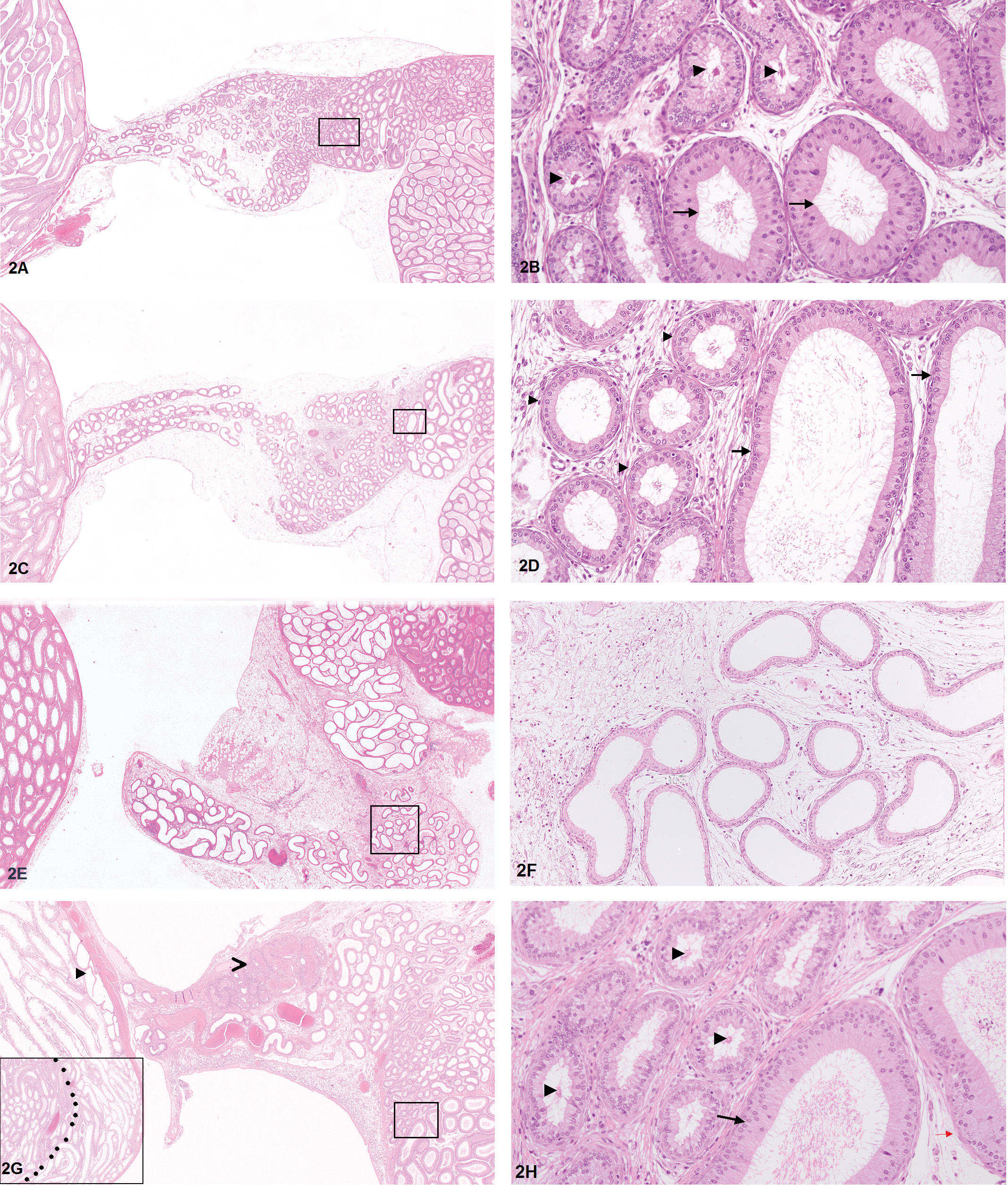
Histopathological findings in efferent ducts, rete testis, and initial segment epididymis of rats dosed with 10 mg/kg/day BYK169171 for 7, 14, and 28 days. (A) Excurrent duct system of 7-week-old control animal (H&E, 1.25×). (B) Higher magnification of boxed area in A: concentrated spermatozoa are visible in the lumen of the distal end of efferent ducts (arrowheads) before transition into the initial segment of epididymidal head (arrows; H&E, 20×). (C) Animal treated for 7 days. Mild dilatation of distal end of efferent ducts and initial segment of epidiymidal head (H&E, 1.25×). (D) Higher magnification of boxed area in C: dilatation of distal segment of efferent ducts (arrowheads) and initial segment of epididymidal head (arrows). Note decreased density of spermatozoa in lumen of efferent ducts (H&E, 20×). (E) Animal treated for 14
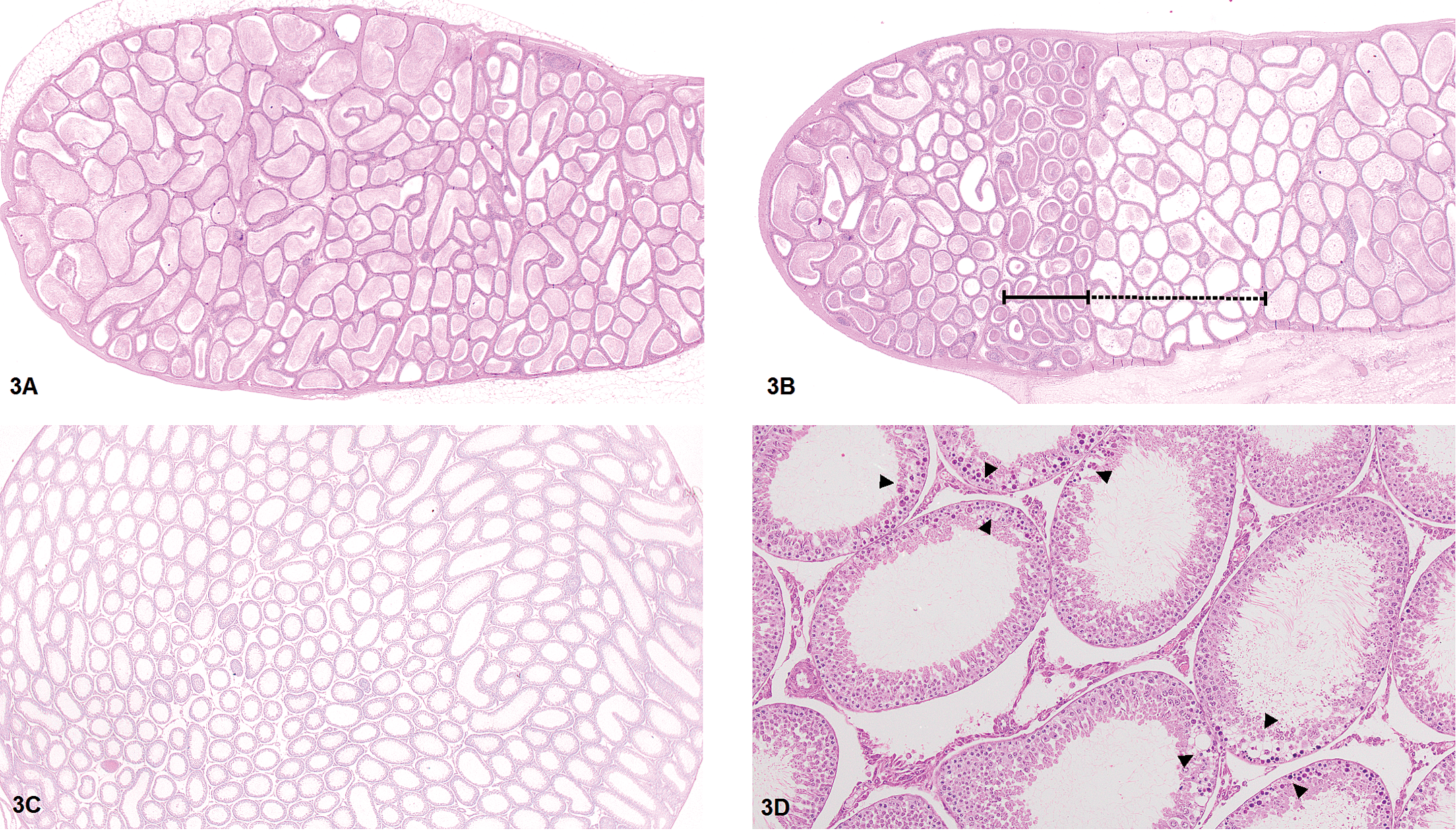
Histopathological findings in testis and epididymis in animals treated with 10 mg/kg/day BYK169171. (A + B) Cauda epididymis of an animal treated for 21 days compared with an age-matched control animal. (A) Normal sperm density in the control animal (H&E, 2×). (B) In the treated animal, segments with increased sperm density (continuous line) and decreased sperm density (dashed line) were noted (H&E, 2×). (C) Diffuse seminiferous tubular dilatation of the testis of an animal treated for 7 days (H&E, 1.25×). (D) Focal apoptosis of germ cells of seminiferous tubules (arrowheads). Only those parts of the germinal epithelium of neighboring tubules are affected that face a common interstitial space (H&E, 1.25×).
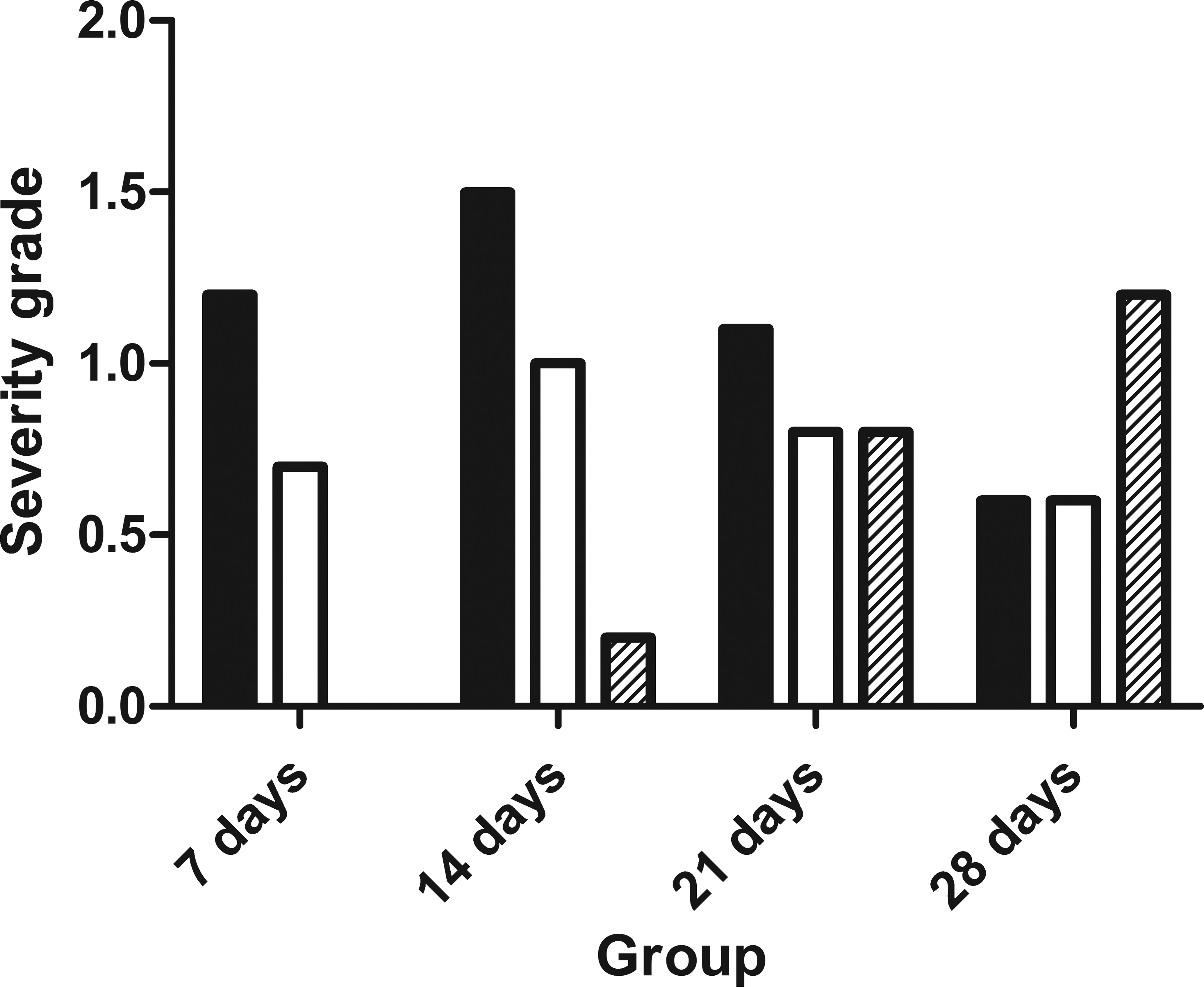
Mean severity grade of dilatation of efferent ducts (black bars), of dilatation of seminiferous tubules (white bars), and of degeneration of seminiferous tubules (striped bars) after 7, 14, 21, and 28 days of treatment with 10 mg/kg/day BYK169171. N = 10 (7 and 14 days), n = 9 (21 days), n = 12 (28 days). Dilatation peaked after 14 days of treatment and declined thereafter, whereas tubular degeneration increased toward the end of treatment. For calculation of severity grades see Materials and Methods.
BYK169171-related histopathological findings of male gonads after 7, 14, 21, and 28 days.
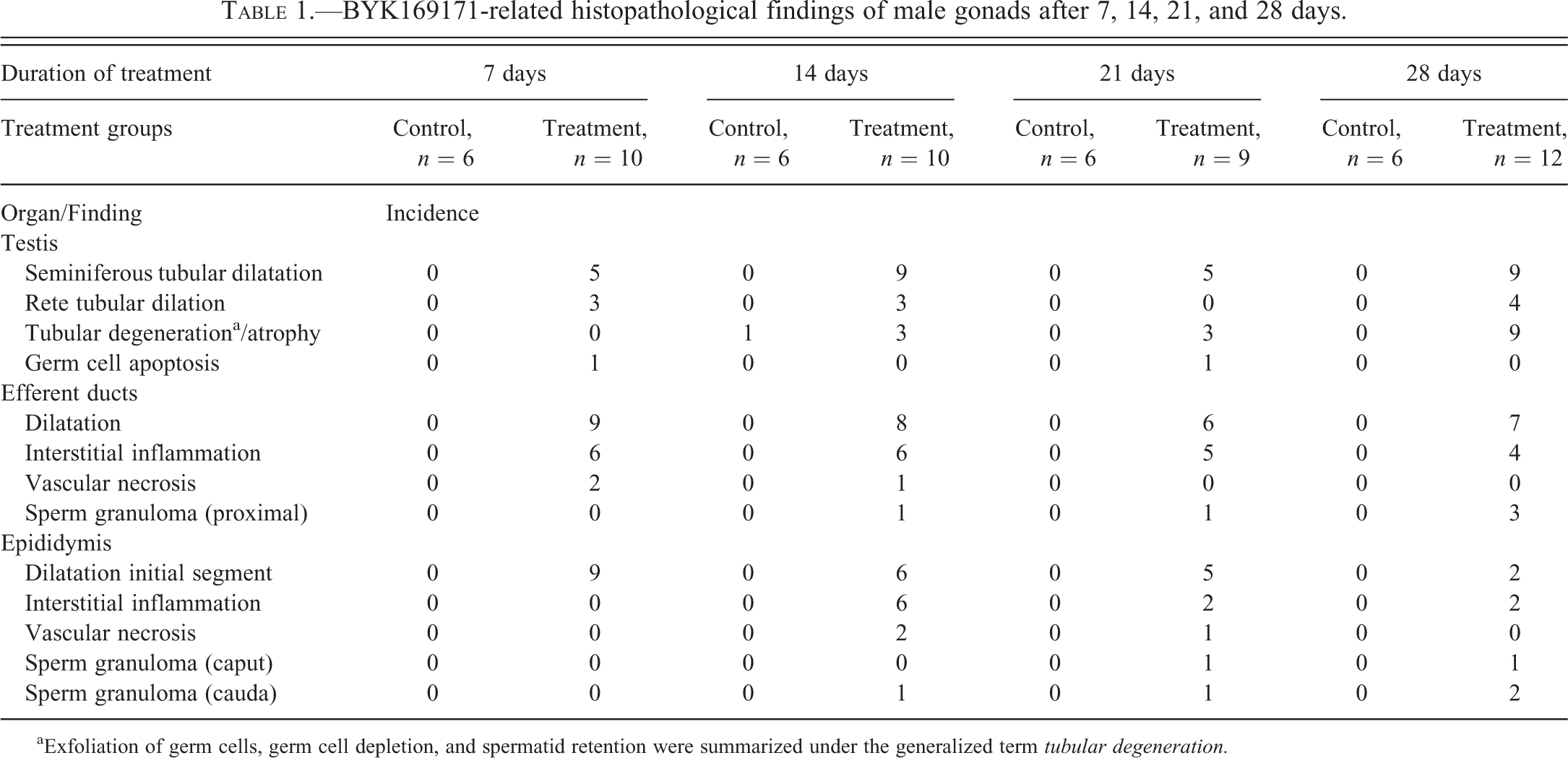
aExfoliation of germ cells, germ cell depletion, and spermatid retention were summarized under the generalized term tubular degeneration.
At all time points, the majority of animals developed dilatation of efferent ducts and epididymidal heads bilaterally with only 1 to 3 animals per group showing dilatation only unilaterally. However, dilatation of efferent ducts did not extend to involve seminiferous tubules of the testis on each side to the same extent, and the majority of animals with bilateral efferent duct dilatation exhibited seminiferous tubular dilatation only unilaterally (Figure 5).
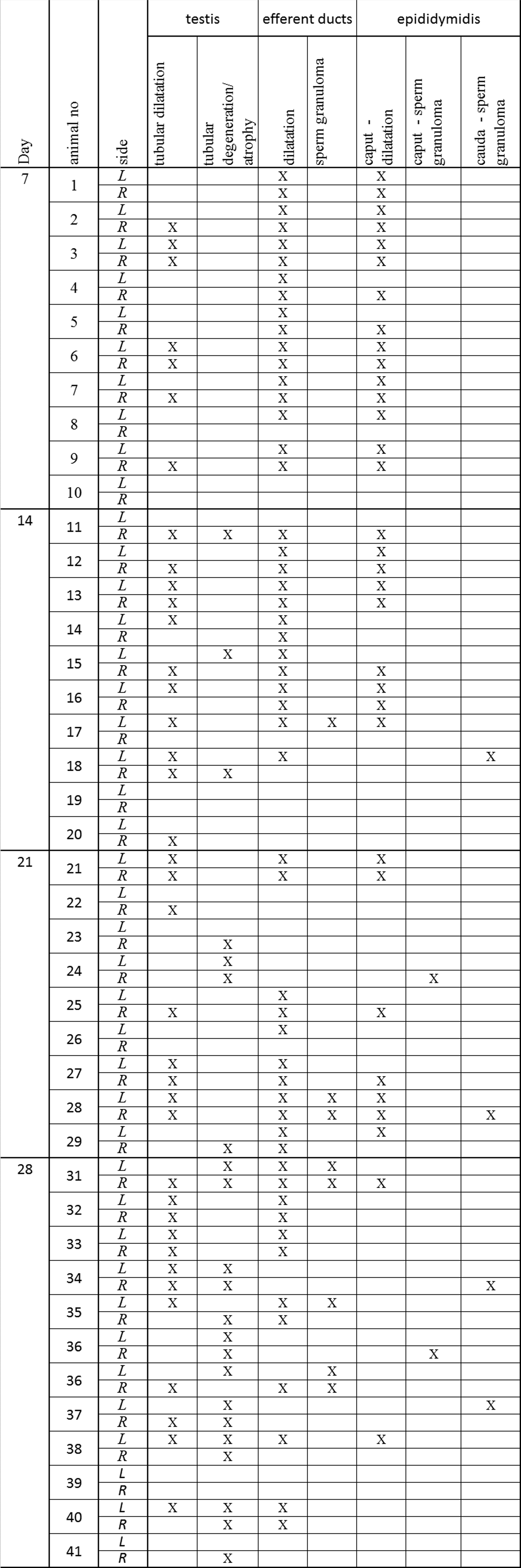
Incidence of findings in testis, efferent ducts, and epididymis in individual animals treated with 10 mg/kg/day BYK169171 for 7, 14, 21, and 28 days. Incidences are listed for right and left side separately. L = left side, R = right side, x = finding present.
Seminiferous Tubule Degeneration
Dilatation of seminiferous tubules caused a mild exfoliation of germ cells. Germ cell apoptosis was apparent in each of the animals after 7 days and 21 days of treatment. Germ cell apoptosis followed a specific focal pattern, namely only those parts of the germinal epithelium were affected that faced the triangular interstitial space between neighboring tubular cross sections (Figure 3D). Minimal to marked seminiferous tubule degeneration and atrophy were evident in 3 rats after 21 days of treatment, and it increased in incidence and severity after 28 days. Although degeneration in animals treated for 21 days did not display any stage or cell specificity, degeneration in rats treated for 28 days affected mainly elongating spermatids throughout all stages. Spermatid retention was evident in 2 of the 15 rats. In addition, in 4 of the 12 animals, the germ cell depletion displayed the same focal pattern as seen with germ cell apoptosis. In all animals, the most severe changes were seen in seminiferous tubules close to the rete testis (Figure 2G, inlet).
Interstitial Subacute Inflammation/Vascular Changes
In addition to dilatation, diffuse interstitial subacute inflammation was another early finding seen already after 7 days of treatment. Inflammation was characterized by mild to moderate edema and inflammatory cell infiltrates that were mainly composed of histiocytes, with fewer lymphocytes, and rare neutrophils. The time response of subacute interstitial inflammation was similar to that of dilatation, reaching a maximum after 14 days of treatment and decreasing gradually thereafter. In rats treated for 7 days, subacute inflammation was evident in the connective tissue encasing the dilated efferent ducts only. In rats of the 14-day group, it was additionally evident in the epididymal cauda (in 3 of the 10 animals), caput (in 2 of the 10 animals), and corpus (in 2 of the 10 animals).
Single animals treated for up to 21 days exhibited lesions of small interstitial arteries characterized by endothelial activation, eosinophilic necrosis of the media, and perivascular fibroblast proliferation. These vascular changes occurred either in the inflamed interstitium of the efferent ducts or in the interstitium of the caput or cauda epididymis. Two animals exhibited vascular changes in the serosal tissue flanking the epididymis at the level of transition from the distal caput to the corpus of the epididymis. No vascular changes were noted in the interstitium of the testis.
Changes in Epididymal Sperm Content
Changes in sperm content were apparent in the epididymal duct of animals treated for 14 and 21 days with segments showing increases or decreases in sperm density (Figure 3B) adjacent to segments with aspermia and reduced ductual diameter.
Sperm Granulomas
Because sperms are immunologically privileged, meaning that they elicit an inflammatory response if present outside of their physiological habitat, sperm granulomas develop as a local immune reaction to sperm that escape the excurrent duct system. The formation of sperm granuloma was first evident after 14 days of treatment when dilatation and interstitial inflammation were most pronounced. Sperm granulomas increased over time from 21 to 28 days of treatment, with the highest incidence found in efferent ducts followed by the epididymis. No sperm granulomas were found in seminiferous tubules of the testes including the rete tubules. Preferred sites of granuloma formation were the proximal parts of efferent ducts. In the epididymis, sperm granulomas were either located in the cauda or in the distal segment of the epididymal head. In efferent ducts, sperm granulomas developed bilaterally in 3 and unilaterally in 2 animals, but all sperm granulomas of the epididymidis were unilateral. Acute changes preceding granuloma formation in efferent ducts were evident in rats treated for 14 and 21 days and comprised a marked focal accumulation of spermatozoa within dilated portions of the proximal efferent ducts. Neutrophils were found to infiltrate the periductular interstitium and the dilated tubules, thereby causing epithelial necrosis and sperm leakage. Acute changes preceding granuloma formation in the epididymis were restricted to 1 animal of the 21-day treatment group. This animal displayed segmental epithelial necrosis of the epididymal duct in the distal caput region. Epithelial necrosis was associated with interstitial neutrophilic inflammation and vascular necrosis. The same animal exhibited sperm leakage admixed with neutrophils in the cauda epididymis of the contralateral site. Acute changes were no longer present in rats of the 28-day treatment group. All sperm granulomas that were evident after 28 days of treatment were organized and displayed the typical composition with a dense central core of entangled spermatozoa encapsulated by layers of radially arranged epithelioid macrophages. In some of the rats, a recanalization of occluded efferent ducts was evident. On an individual animal basis, there was no correlation between sperm granuloma formation in efferent ducts and incidence of seminiferous tubule degeneration (Figure 5).
Hormone Levels
Serum levels of testosterone were slightly decreased on day 21 and day 28 of the study but without reaching statistical significance. The decrease in systemic testosterone levels was accompanied by a marked decrease in androgen concentrations of both testicles. Inhibin B serum levels declined over time, both in control animals and in BYK169171-treated rats. When animals with seminiferous tubular atrophy were compared to those without atrophy, animals with atrophy had lower serum levels of Inhibin B (data not shown).
Discussion
Several target organs after administration of PDE4 inhibitors to rats have been described (Dietsch et al. 2006; Larson et al. 2006; Mecklenburg et al. 2006; Peter et al. 2011; Sheth et al. 2011; Slim et al. 2003; Zhang et al. 2002, 2008), but to our knowledge no detailed reports about changes in male reproductive organs are available so far. Testicular dilatation and epididymal sperm granulomas were repeatedly seen in the 4-week toxicity studies with PDE4 inhibitors of different chemical classes at our facility. To better understand the pathogenesis and sequence of lesions, a mechanistic study over 4 weeks with sequential necropsies in weekly intervals was performed with BYK169171, a highly selective PDE4 inhibitor. A high dose of 10 mg/kg/day was selected based on results of a routine 4-week toxicity study with BYK169171 where 5 mg/kg/day had induced lesions in only 5 of the 10 animals. A special focus was on the investigation of efferent ducts, which had not been part of the investigations in the routine toxicity study. In addition, serum levels of testosterone and inhibin B as well as intratesticular androgen concentrations were determined.
The Earliest Detectable Lesion Is a Dilatation of Efferent Ducts and Initial Segment Epididymis That Extend Rapidly to Involve Seminiferous Tubules of the Testis
The earliest lesion after 7 days of treatment was a dilatation of efferent ducts and the initial segment of the epididymis, followed by a diffuse dilatation of seminiferous tubules in the testis. Although measuring testicular weight was impossible in this study, it is assumed that the dilatation is associated with an increase in testicular weight, as was observed in the previously performed 4-week study with BYK169171. Although, we cannot entirely exclude that an increase in interstitial fluid content may have added to the increase in testicular weights, the severity of tubular dilatation let us conclude that intratubular fluid accumulation accounted for most of the weight increase.
Increases in intratubular fluid content can derive from an increased rate of fluid production by Sertoli cells, a decreased fluid reabsorption in efferent ducts, a decreased rate of fluid transport (e.g., by reduced contractions of peritubular myofibers), or an outflow obstruction along the rete testis, efferent ducts, or epididymal duct. From the results of our study, an obstruction of outflow by sperm granulomas could clearly be excluded since sperm granulomas only developed after dilatation. A decreased rate of fluid transport caused by relaxation of myoid cells is possible, if a higher sensitivity to relaxation is assumed for myoid cells around the efferent ducts compared to myoid cells surrounding testicular tubules. Recently, the expression of the PDE5 enzyme in myoid cells around the epididymal duct has been investigated by Mietens and coworkers (2012). The authors could localize the PDE5 enzyme to the smooth muscle layer of the epididymal duct in caput, corpus, and cauda and to vascular smooth muscle cells. Similar investigations for the PDE4 enzyme would be helpful to investigate the potential impact on regional smooth muscle relaxation on the dilatation observed under treatment with BYK169171. Discrimination between increased fluid production by Sertoli cells and decreased fluid reabsorption by efferent ducts is difficult based on histology alone, because both mechanisms may result in early dilatation of efferent ducts. An increase in fluid production with unchanged outward transport may overload the normal capacity of fluid reabsorption of efferent ducts causing dilatation primarily at the site of overload, whereas a decrease in fluid reabsorption by normal fluid production and outward transport would have the same effect. Although experimental evidence is lacking, we consider it more likely that a disturbed reabsorption in efferent ducts constitutes the primary effect, because on an individual animal basis the incidence of dilatation of efferent ducts was higher than the dilatation of testicular tubules. Moreover, in the first 2 weeks testicular dilatation was always associated with efferent duct dilatation and did not occur as an isolated finding. A potential explanation for the efferent duct dilatation based on changes in fluid homeostasis may be derived from the work of Man, Clulow, and Jones (2000) who demonstrated a role for cAMP in the regulation of fluid reabsorption in an in vivo microperfusion study in isolated efferent ducts from rats. Increasing levels of cAMP first reduced net fluid reabsorption and at higher concentrations increased net secretion, an effect that they attributed to two ion pumps, the sodium hydrogen exchanger 3 (NH3) and the cystic fibrosis transmembrane regulator (CFTR). The NH3 is an electroneutral Na/H antiport system that is located in the apical membrane of efferent ducts and is the main driver for sodium-coupled water reabsorption (Hansen, Clulow, and Jones 1999; Leung, Tse, et al. 2001). The CFTR is a cAMP-regulated chloride channel that upon activation drives an inward flux of chloride and water. It is also located at the apical membrane of efferent ducts (Leung, Gong, et al. 2001) and counterbalances fluid reabsorption under physiological conditions to avoid dehydration and excessive sperm concentration. cAMP was found to exert its effect on net fluid increase by both inhibition of the NH3 and stimulation of the CFTR. Similar results were obtained when using the nonspecific PDE inhibitor pentoxifylline. Although to our knowledge the expression of the PDE4 enzyme in efferent ducts has not been investigated yet, the results demonstrate a possible interaction of BYK169171 with ion transport mechanisms based on its ability to increase intracellular levels of cAMP. Disturbance of fluid homeostasis may thereby not only account for changes seen in efferent ducts but also account for changes seen in the epididymis. Although differences existed between sodium reabsorption in efferent ducts and epididymis, the CFTR is involved in the regulation of water secretion in both organs (Cheung et al. 2003; Leung, Gong, et al. 2001) suggesting a potential for interaction with BYK169171. Fluid reabsorption continues during epididymal passage, especially in the anterior segment. In the mid-caput region, sperm concentration is 20-fold higher than in the rete testis (Turner 1984). The dilatation of the initial segment of the epididymal head together with a decreased sperm density suggests that the disturbance of fluid reabsorption extends to the epididymal head. However, changes in sperm density were not restricted to the caput but also occurred in the corpus and cauda epididymis where segments with increased sperm density alternated with segments of decreased sperm density or even with segments of aspermia and reduced ductual lumen. Based on the work of Wong (1986) who showed that fluidity of the microenvironment influenced not only sperm maturation but also sperm transport, the changes in sperm contents in the corpus and cauda were interpreted as a disturbance in normal movement of sperm through the epididymis as a result of impaired fluid reabsorption.
In addition to dilatation, inflammation of the connective tissue of efferent ducts and of the epididymis was another early finding. However, a direct relationship between dilatation and inflammation could not be established. It can be speculated that extensive stretching of efferent duct epithelium and widening of epithelial tight junctions exposed the immunogenic spermatozoa to the interstitium, thereby breaking the immune privilege. However, since inflammation in the epididymis was not associated with dilatation and since single animals showed vascular lesions in the inflamed areas, it appears more likely that inflammation had a vascular origin. Investigations of Korkmaz et al. (2009) on regional blood flow in different tissues after administration of the PDE4 inhibitor CI-1044 might support this hypothesis. The authors could demonstrate an increase in the mesenteric blood flow (up to 190% vs. control), which was associated with perivascular and interstitial inflammation. Slight and transient increases in blood flow were also noted in the epididymis (at 4 hr postdose up to 38% vs. control). However, no inflammation was noted upon histological investigation. Nevertheless, the results indicate that changes in vascular tone induced by PDE4 inhibitors may result in inflammation in sensitive vascular beds. The observed increases in blood flow in the epididymal tissue in the experiment of Korkmaz and coworkers might have been too low to result in inflammation. Alternatively, efferent duct interstitium that was not part of the investigation might be more sensitive for developing inflammation than epididymal tissue.
Another experiment conducted by Piner and coauthors (2002) points to a possible involvement of vascular changes in the pathogenesis of testicular and efferent duct dilatation. In their experiment, local vascular constriction of veins of the mediastinal plexus or of arteriovenous anastomoses in the spermatic cord were found to proceed or accompany dilatation of efferent ducts, rete testis, and seminiferous tubules after single oral administration of a high dose of a 5-hydroxytryptamine agonist to male rats. The effect could be antagonized by minoxidil, a potent vasodilator. However, minoxidil alone had no significant effect on arteriovenous anastomoses in the spermatic cord vessels demonstrating that not vasodilation per se but local changes in especially sensitive vascular beds are required to initiate toxicity. However, although there are many indications that vascular changes are involved in the histological lesions observed in our experiment, a direct pathogenetic link needs to be established. Further investigations on effects of BYK169171 on regional blood flow in vessels supplying efferent ducts and epididymidis might be helpful to explain a potential vascular involvement in interstitial inflammation or dilatation or both.
Sperm Granulomas Develop Secondarily in the Proximal Parts of Efferent Ducts and the Distal Caput or Cauda Epididymis
When dilatation of efferent ducts reached a maximum score, sperm granulomas appeared in some but not in all animals. Overall, the number of animals with dilatation of efferent ducts was much higher than the number of animals with sperm granulomas. Only in single animals did an accumulation of highly concentrated spermatozoa within proximal parts of dilated efferent ducts appear, first visible after 14 days of treatment. Obviously, an occlusion of the ductal lumen by inspissated sperm and not an overstreching of the epithelium by fluid accumulation initiated granuloma formation. Periductual infiltration of neutrophils, transepithelial neutrophil migration, and focal rupture of the epithelium were seen at the site of occlusion suggesting that neutrophil attraction and activation were the driving forces for granuloma formation. A similar response of efferent duct epithelium to occlusion has been described for benzimidazol carbamates (Hess and Nakai 2000). All sperm granulomas of efferent ducts in our experiment were located in the proximal parts. However, due to the small number of affected animals it remains questionable whether or not the proximal parts are an especially vulnerable site to BYK169171-induced toxicity. In addition, since no serial sections of the efferent ducts were performed, the total incidence of sperm granulomas in the proximal parts might actually be higher. However, in none of the animals with sperm granulomas in efferent ducts was a complete outflow obstruction observed. Unaltered ducts allowing a passage of spermatozoa were always present. Interestingly, the reabsorption capacity of efferent ducts and epididymal head seems to recover in animals treated for 28 days as indicated by a decrease in the severity of dilatation. In addition, concentrated spermatozoa were again visible in the lumen of the common distal duct of some of the animals (Figure 2H).
In addition to sperm granulomas in efferent ducts, a total of 6 sperm granulomas developed in the epididymis under treatment with BYK169171. A variety of compounds are known to induce sperm granulomas in the epididymis. A direct toxic effect on the epithelium was thought to be the mechanism of granuloma formation in the rat epididymal cauda after inhalation of methyl chloride (Chapin et al. 1984). Long-lasting blockade of adrenergic innervation induced epithelial rupture with subsequent granuloma formation at the epididymal–vas deferens junction after administration of guanethidine (Bhathal et al. 1974). Defective development of the epididymal duct resulting in obstruction of sperm outflow were suggested as underlying causes for
Testicular Damage Results Mainly from Pressure Atrophy and Hormonal Changes Are Secondary to Testicular Toxicity and Stress
In the testis, moderate tubular dilatation was associated with exfoliation of germ cells. After 28 days of treatment, tubular atrophy was obvious in seminiferous tubules close to the rete testis. The tubular atrophy as well as the degeneration of elongating spermatids was interpreted to result from back pressure atrophy. In some animals, a specific pattern of germ cell apoptosis or germ cell depletion was observed. Germ cell depletion or apoptosis was only evident in those parts of the germinal epithelium of neighboring tubular cross sections that faced the common triangular interstitial space. We think that this distribution pattern might be related to changes in local blood supply resulting in germ cell apoptosis in adjacent tubules and when apoptotic cells have been removed in germ cell depletion.
Reductions in serum testosterone levels and intratesticular androgen concentrations were regarded as nonspecific and attributed to high systemic toxicity and stress as reflected by threefold lower body weight gain and twofold higher adrenal weights in treated animals compared with controls (Hu et al. 2008; Rehm et al. 2008; Rivier and Rivest 1991). Inhibin B is secreted from Sertoli cells and inhibits follicle-stimulating hormone (FSH) production. It may be used as a biomarker of male fertility. Indeed, reduced inhibin B concentrations were found in the serum of those animals that exhibited testicular atrophy. However, it is assumed that the decrease in inhibin B is a consequence rather than a cause of testicular degeneration.
In summary, the changes observed in male gonads appear to have multifactorial causes including effects on vascular supply and fluid transport mechanisms. Interestingly, perfusion-mediated changes as well as changes in fluid reabsorption were only recognized in animals treated for up to 21 days but were no longer evident in animals treated for 28 days. This observation let us speculate that BYK169171 induced a biphasic response in efferent ducts and epididymis: after an acute phase with accumulation of fluid within the system some kind of adaption allowed a recovery of fluid reabsorption and secretion, although treatment with BYK169171 was continued. However, the basis for this adaptive response remains unclear. Pharmacokinetic data did not yield an explanation since steady state of the compound was achieved rapidly, exposure was stable over time, and no accumulation in testis or epididymis was seen (data not shown). All findings observed after 28 days were regarded as secondary changes including nonreversible findings like sperm granulomas or findings requiring probably longer recovery periods like seminiferous tubular degeneration. However, repeat-dose toxicity studies with treatment periods longer than 4 weeks would be helpful to assure that acute lesions do not reappear under chronic treatment with PDE4 inhibitors.
Another important aspect for the interpretation of routine 4-week repeat-dose toxicity studies with PDE4 inhibitors involves the distribution pattern of lesions. Although the dilatation of efferent ducts and epididymal head developed as a bilateral change, subsequent changes like testicular tubular dilatation and sperm granulomas were often evident only unilaterally. Similar results were reported by Längle et al. (1994) who treated rats with a dual selective PDE3/4 inhibitor. The compound induced unilateral inflammation and new bone formation in the hind limbs of treated animals. Unilateral findings in studies with PDE4 inhibitors should therefore be considered as toxicological significant. Why different sensitivities to developing lesions existed between right and left side remains to be investigated.
In conclusion, based on the timing of the various changes seen, we propose the following sequence of events: (1) transient dilatation of efferent ducts, initial segment epididymis, and seminiferous tubules of the testis, (2) sperm granulomas develop secondarily in the proximal parts of efferent ducts and the distal caput or cauda epididymis, and (3) testicular damage results mainly from pressure atrophy and is not associated with outflow obstruction by granuloma formation.
We propose that the mode of BYK169171-induced toxicity is multifactorial. It involves a disturbance in fluid homeostasis in efferent and epididymal ducts, resulting in abnormal luminal fluid and sperm contents, epithelial damage, and sperm leakage. In addition, it involves vascular changes resulting in interstitial inflammation and local ischemia. Although our study was not designed to detect the underlying mechanism of BYK169171-induced toxicity, we speculate that interaction with ion transporters or local changes in blood flow might have initiated the observed histopathological changes. As no reports on gonadal toxicity after administration of PDE4 inhibitors to rats are available so far, and since we have always used Wistar rats from the same supplier for our toxicity studies with PDE4 inhibitors, it would also be interesting to investigate whether similar morphological changes occur in other strains of rats.
Footnotes
Acknowledgments
The assistance of Prof. Nieschlag and Prof. Simoni in determination of testicular hormone concentrations is greatly acknowledged.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
