Abstract
A 12-week-old Swiss Albino mouse was presented with unilateral (left) testicular enlargement of approximately 1.5 cm in diameter and the right testicle mildly reduced in size and weight. Histopathology evaluation revealed three distinct neoplasms in the left testicle: choriocarcinoma, yolk sac carcinoma, and embryonal carcinoma. Teratoma was diagnosed in the right testicle. The histomorphological and immunohistochemical characteristics of the tumor are presented here. To the best of the authors’ knowledge, this is the first report of spontaneous nonmetastasizing choriocarcinoma, yolk sac carcinoma, embryonal carcinoma, and teratoma in testes of a Swiss albino mouse.
Introduction
The nonseminomatous germ cell tumors of the testis arise from totipotent cell population derived from primitive embryonal cells. Such totipotent cells may give rise to largely undifferentiated form of tumor called embryonal carcinomas, may differentiate to form yolk sac tumors and choriocarcinomas, or may differentiate along somatic cell lines to produce teratomas (Burns 2001).
Choriocarcinoma is a rare tumor in laboratory animals and has been reported in the deutsche Maus at Denken (DD) mouse (Madrame, Sakurai, and Konno 1989), B6C3F1 mouse (Alison, Lewis, and Montgomery 1987), Sprague-Dawley (SD) rat (Pirak et al. 1991), Donryu rat, (Yoshida et al. 1997) rabbit (Kaufmann-Bart and Fisher 2008), and also in rhesus monkey (Farman et al. 2005).
Yolk sac tumors are the most common primary testicular neoplasm in children younger than 3 years (Burns 2001). Yolk sac carcinoma can be produced experimentally in the abdomen of female Wistar King Aptekman (WKA) rats (Sakashita et al. 1977); however, reports of its spontaneous occurrence in the testis are rare in rodents and other domestic animals. Testicular yolk sac carcinoma has been reported in the SD rat (Nakazawa et al. 1998) and in a Japanese black calf (Kagawa et al. 1998). Recently, a case of highly metastatic ovarian yolk sac carcinoma in a Crl:CD (SD) rat has been reported (Sakamoto et al. 2011). Ovarian choriocarcinoma occurring with yolk sac carcinoma and teratoma has been reported in B6C3F1 mice (Frith and Evans 1993).
Among laboratory rodents, teratocarcinoma (teratoma and embryonal carcinoma together) of the testis is commonly seen only in mouse strain 129 (Stevens and Little 1954). The authors have recently reported a spontaneous case of teratocarcinoma in a Swiss albino mouse (Jamadagni et al. 2011). The few other reports of teratocarcinoma or teratoma in the testis of rats and mice include teratocarcinomas in two SD IGS rats (Sawaki et al. 2000), a teratoma in an ICR strain mouse (Tani et al. 1997), and teratocarcinomas in DBA/2J and A/HeJ mice (Meier et al. 1970). A case of spontaneous ovarian teratoma has been reported in a Swiss albino mouse (Fawcett 1950).
To our knowledge, spontaneous concurrent testicular choriocarcinoma, yolk sac carcinoma, and embryonal carcinoma along with teratoma in contralateral testis have not been reported in mice. Herein, we describe the histomorphology and immunohistochemical features of a spontaneous choriocarcinoma, yolk sac carcinoma, embryonal carcinoma, and teratoma in the testes of a Swiss albino mouse.
Materials and Method
A Swiss albino mouse (National Institute of Nutrition, Hyderabad, India) aged 12 weeks was found to have a unilateral left testicular enlargement. The mouse was from a colony maintained at the animal housing facility of the National Research Institute of Ayurvedic Drug Development, Kolkata, India. All mice in the colony were housed according to gender in groups of five, in polycarbonate cages (27 × 19 × 14 cm3) with rice husk bedding and a stainless steel grill lid. The cage environment was maintained at a temperature range of 20–26°C, relative humidity of 40–70%, and a 12-hr light–dark cycle. The animals were fed standard laboratory mice feed (National Institute of Nutrition) and provided ad libitum drinking water purified by reverse osmosis. All animals in the colony were maintained and cared for as per the guidelines of the Committee for the Purpose of Control and Supervision on Experiments in Animals, Government of India. The animal showing testicular enlargement was euthanized by CO2 asphyxiation followed by exsanguination and a detailed necropsy was performed. Both the testes were preserved in Modified Davidson’s Fixative for the first 24 hr and later transferred to 10% neutral buffered formalin along with all other organs and tissues collected (i.e., heart, lungs, liver, spleen, kidneys, brain, prostate, epididymides, seminal vesicles and coagulation glands, skin, and gastrointestinal tract). Fixed and processed organs were embedded in paraffin, sectioned at 4–5 µm, and stained with H&E for histopathological evaluation. After evaluating the H&E-stained slides, immunohistochemical staining was performed on charged sections of the testicular mass. Alpha-fetoprotein (AFP) immunostaining was performed using polyclonal rabbit antihuman α-1-fetoprotin antibodies (1:240, A0008, Dako, CA). Detection was performed using VIP peroxidase (SK-4600, Vector Laboratories, Burlingame, CA) as chromogen, and Mayer’s hematoxylin as counterstain. Human yolk sac carcinoma served as external positive control. For cytokeratin staining, monoclonal mouse antihuman cytokeratin, clone AE1/AE3, ready-to-use antibody (IS053, Dako, Denmark) was used, and detection was performed by one-step polymer detection system (K80023, Envision Flex Mini Kit, Dako, Denmark) and Mayer’s hematoxylin as counterstain. Human yolk sac tumor tissue was used as positive control. β-Human chorionic gonadotropin (β-hCG) immunostaining was performed by polyclonal rabbit anti-hCG ready-to-use antibodies (IR508, Dako, Denmark) and detection was performed by one-step polymer detection system (K80023, Envision Flex Mini Kit) and Mayer’s hematoxylin as counterstain. Human choriocarcinoma tissue was used as positive control. The markers for immunostaining were chosen as per the prevalent literature in the human pathology (Burns 2001; Ulbright 2005).
Results and Discussion
The mouse did not show any abnormal clinical signs indicating disease except left-sided scrotal enlargement. The duration and rate of tumor growth are unknown, because mice in this facility do not undergo daily physical examination. At necropsy, the left testis was enlarged and measured approximately 15 × 15 × 10 mm3, with normal shape and smooth surface having intact tunica albuginea. The cut surface of the testis was blackish red. The left epididymis was increased in length. The right testis was mildly reduced in size, and the gross appearance of the right epididymis was normal. No other lesion was found in any other organ or tissue.
Histologically, nearly 90% of the section from the enlarged left testis showed complete replacement of testicular parenchyma by large confluent areas of hemorrhage and necrosis with vague retention of stromal elements. Multiple neoplastic foci consisted of mainly three types of cells: large pleomorphic polyhedral cells with hyperchromatic nuclei, polygonal cells with round-to-ovoid nuclei and eosinophilic cytoplasm, and cuboidal cells arranged around collagenous and hyaline stroma (Figure 1).
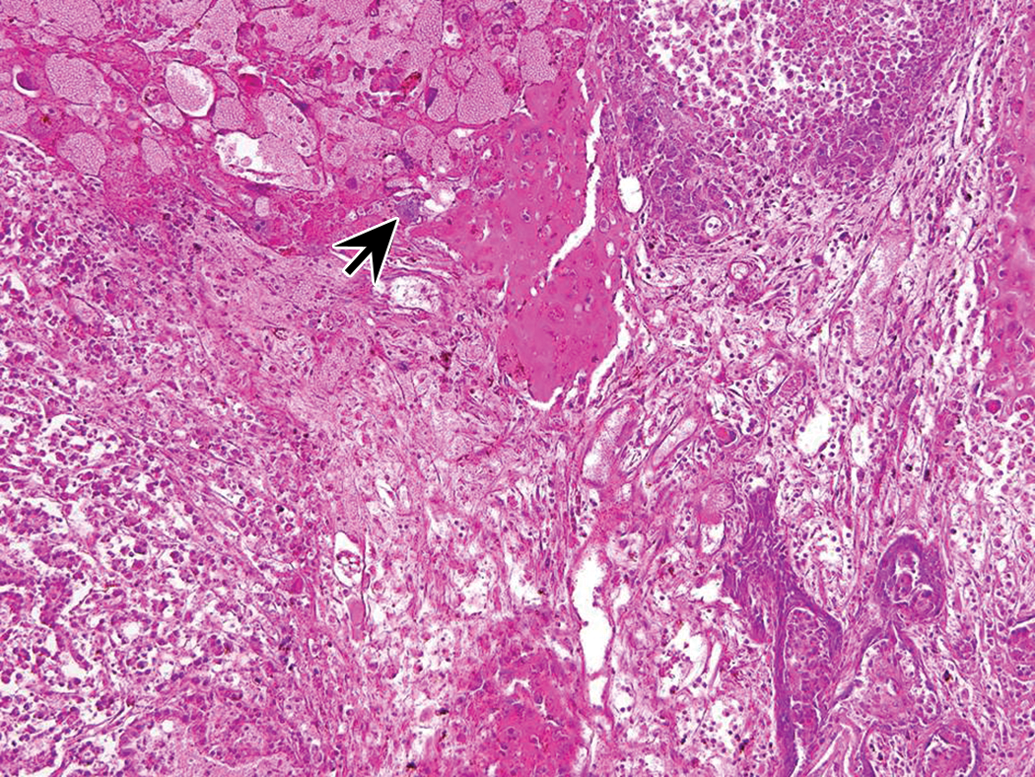
Low magnification image showing relationship between confluent areas of hemorrhages and choriocarcinoma (upper left) with syncytiotrophoblast (arrow), yolk sac tumor (upper right and lower left), and stratified squamous epithelium component (lower right). H&E, 10× objective magnification.
The choriocarcinoma was composed of the first two types of the cells arranged haphazardly in solid cellular areas. Large, multinucleated, pleomorphic, polyhedral cells with abundant eosinophilic cytoplasm were syncytiotrophoblasts and smaller polygonal cells with round-to-ovoid nuclei and pink cytoplasm were cytotrophoblasts (Figures 2 and 3). The syncytiotrophoblasts showed marked anisokaryosis, karyomegaly, and anisocytosis with occasional mitoses. Numerous trophoblasts were also observed, which were large sized, mononucleated polyhedral cells with abundant cytoplasm present. However, immunostaining of the syncytiotrophoblasts, trophoblasts, and cytotrophoblasts for β-hCG that is produced by placental trophoblasts was found to be negative. No metastasis of neoplastic cells or invasion into surrounding tissues was observed. In humans, choriocarcinoma is generally a highly metastatic tumor and always shows positive immunostaining for β-hCG (Burns 2001). In case of animals, choriocarcinomas in rhesus monkey, SD rat, Donryu Rat, and rabbit were positive for β-hCG immunostaining and also metastasized (Farman et al. 2005; Pirak et al. 1991; Yoshida et al. 1997; Kaufmann-Bart and Fischer 2008). The choriocarcinoma in this case was similar to previous reports, except for remarkable absence of detectable metastatic disease and negative immunostaining for β-hCG.
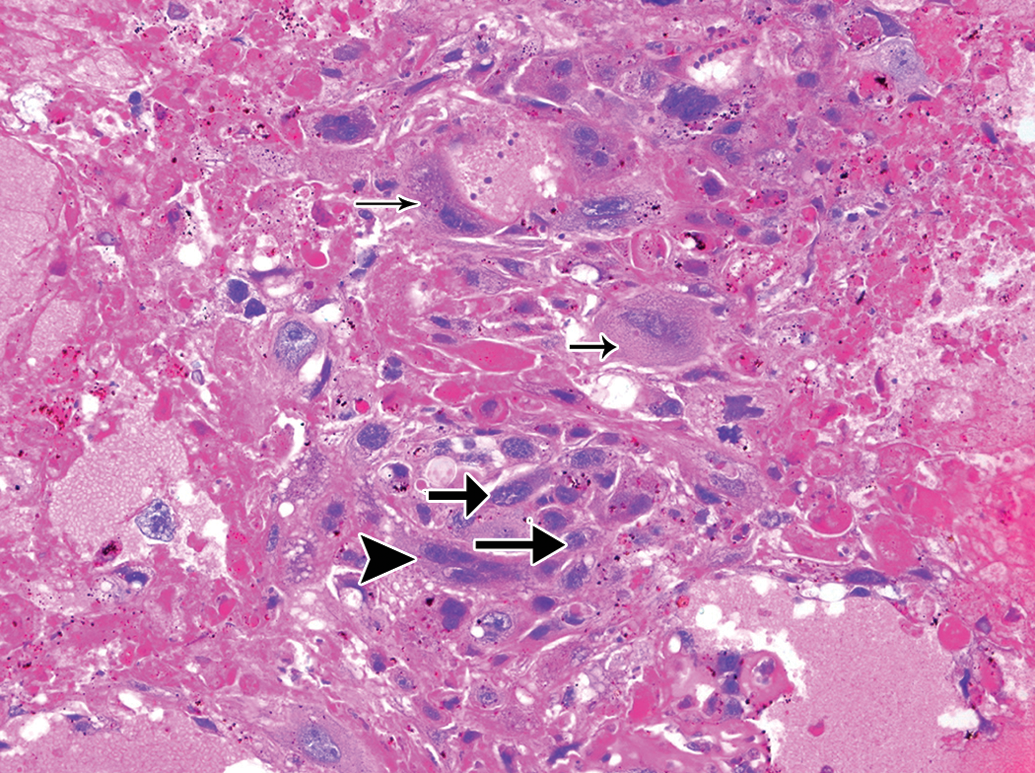
Choriocarcinoma showing syncytiotrophoblast (arrowhead), cytotrophoblasts (arrow), and trophoblasts (smaller arrows in upper and upper-mid portion). H&E, 20× objective magnification.
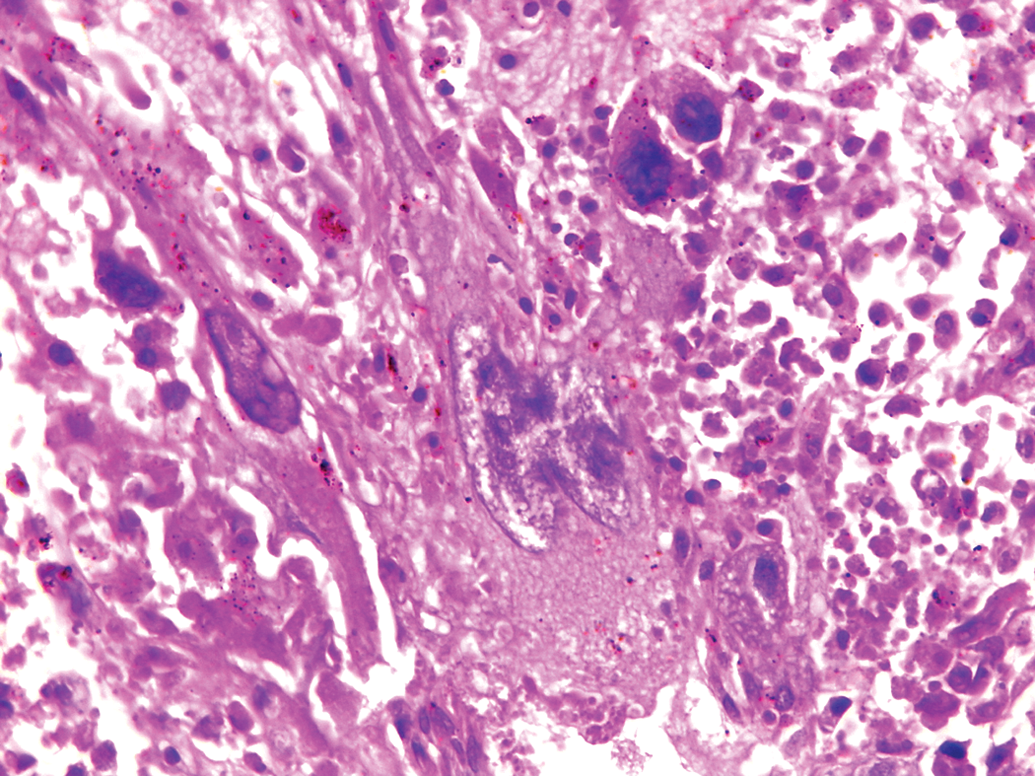
A Syncytiotrophoblast with double nuclei. H&E, 40× objective magnification.
The second neoplasm was yolk sac carcinoma, composed mainly of cuboidal cells arranged in ribbons and cords supported on fine collagenous stroma and occasionally surrounded by eosinophilic, Periodic Acid Schiff (PAS)-positive hyaline ground substance (Figures 4 and 5). The hyaline ground substance was reminiscent of Reichert’s membrane, the basement membrane separating the visceral and parietal layers of the rodent yolk sac (Majeed et al. 1986).These neoplastic cells featured abundant, eosinophilic cytoplasm containing droplets of hyaline proteinaceous material. No mitoses were apparent in this population. Positive immunostaining for cytokeratin and AFP further supported the diagnosis of yolk sac carcinoma (Figure 6A and B). Ovarian yolk sac carcinoma in B6C3F1 mouse (Frith and Evans 1993) and Crl:CD(SD) rat (Sakamoto et al. 2011) showed metastasis; however, in the present case, no metastasis of yolk sac carcinoma was detected. Seminiferous tubules with one to two layers of germinal cells were observed at one end of the section.
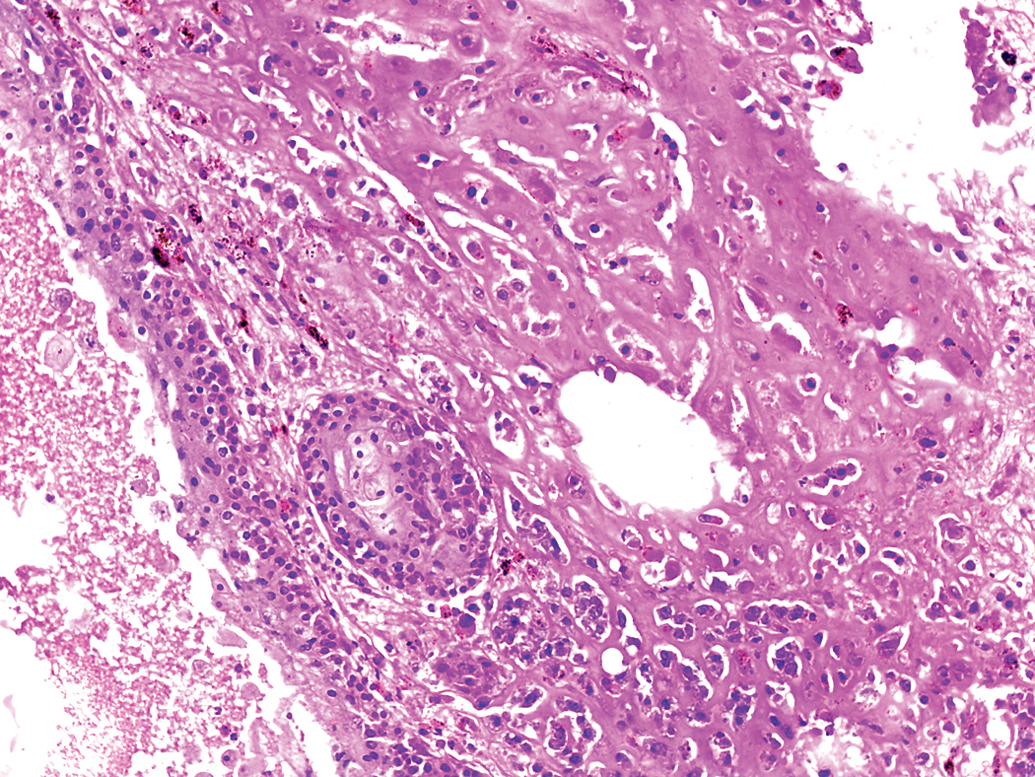
Cuboidal cells of yolk sac tumor supported on fine collagenous stroma and eosinophilic matrix. H&E, 10× objective magnification.
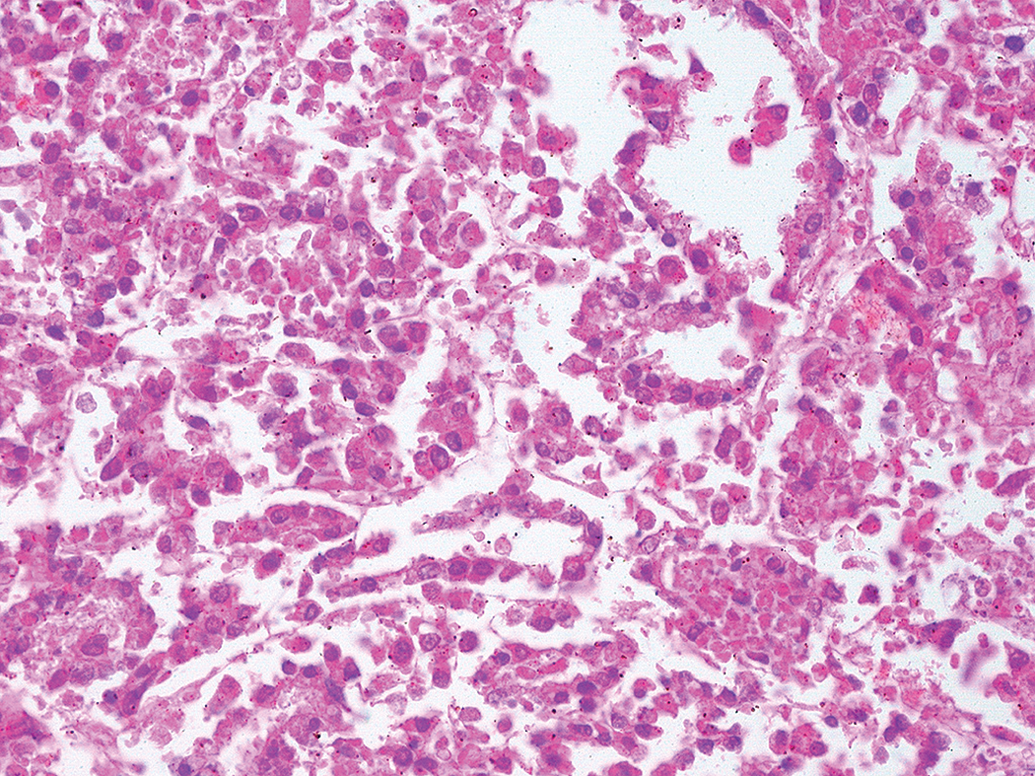
Cuboidal cells of yolk sac tumor forming ribbons and supported on fine collagenous stroma. H&E, 20× objective magnification.
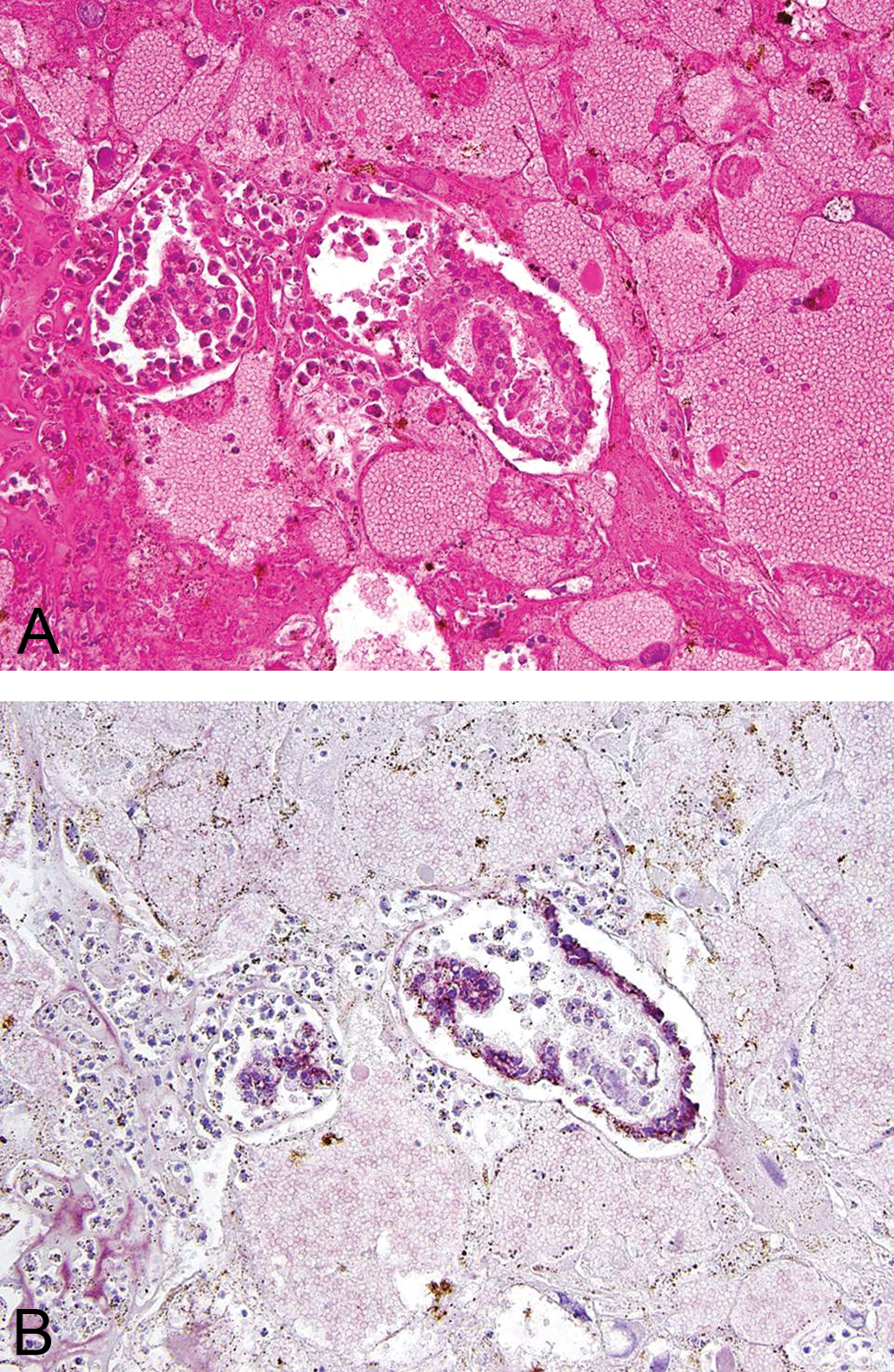
A: Yolk sack carcinoma. H&E, 20× objective magnification. B: Same region in (A) with positive cytoplasmic immunoreactivity for alpha-fetoprotein (AFP). AFP immunohistochemistry, counterstain Mayer’s Hematoxylene 20× objective magnification.
Serial sectioning of the tumor tissue revealed few foci of a third neoplasm—embryonal carcinoma—in the left testis. The embryonal carcinoma was composed of solid sheets of primitive embryonic cells with crowded, large, oval, basophilic nuclei along with prominent nucleoli, scant cytoplasm, and indistinct cytoplasmic borders (Figure 7). These cells were occasionally arranged in vague rosette-like structures. In the left testis, there were few foci of polygonal cells forming islands undergoing central squamous differentiation with keratinization resembling with hair follicles. Rare mitoses were observed in this population. The cellular morphology of the embryonal carcinoma was similar to that of a previously published report of embryonal carcinoma in the testis of a Swiss albino mouse (Jamadagni et al. 2011). However, in the present case, only one type (i.e. ectodermal) of well-differentiated somatic cell line, in the form of keratinized stratified squamous epithelium was found.
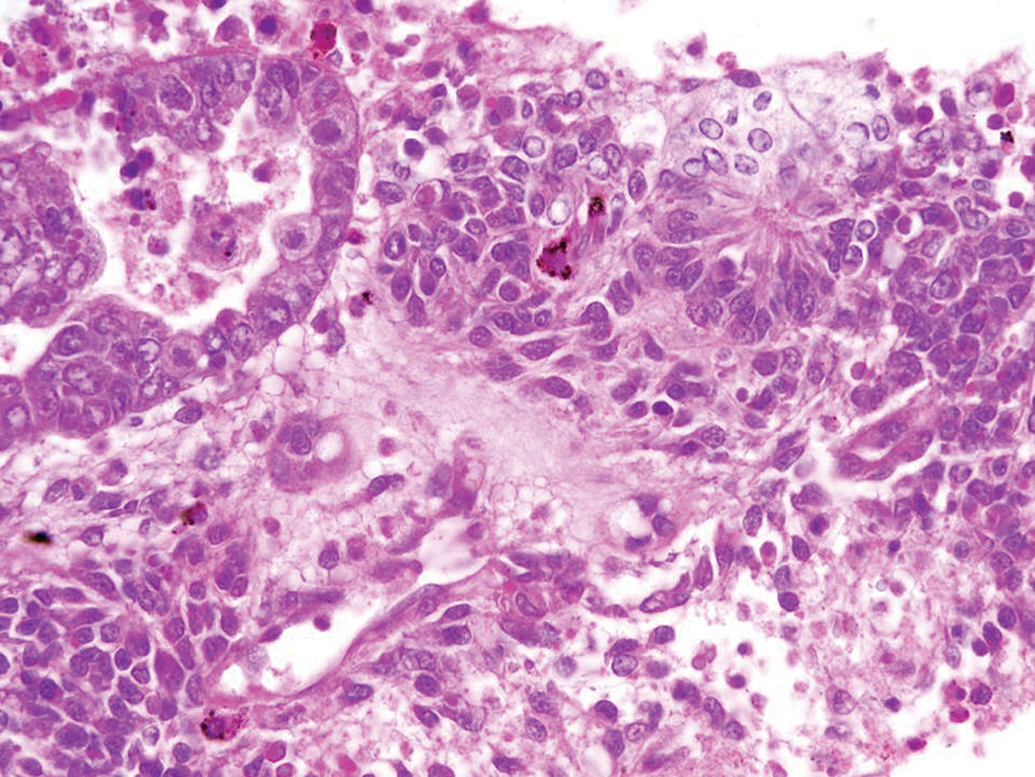
Embryonal carcinoma cells (upper left) trying to form rosette. H&E, 20× objective magnification.
The right testis, although grossly reduced in size, revealed typical teratoma features, including foci of well-formed adipose tissue, cavities lined by ciliated columnar epithelium similar to respiratory epithelium, mesenchymal tissue resembling smooth muscle, cartilage, and bone (Figure 8).
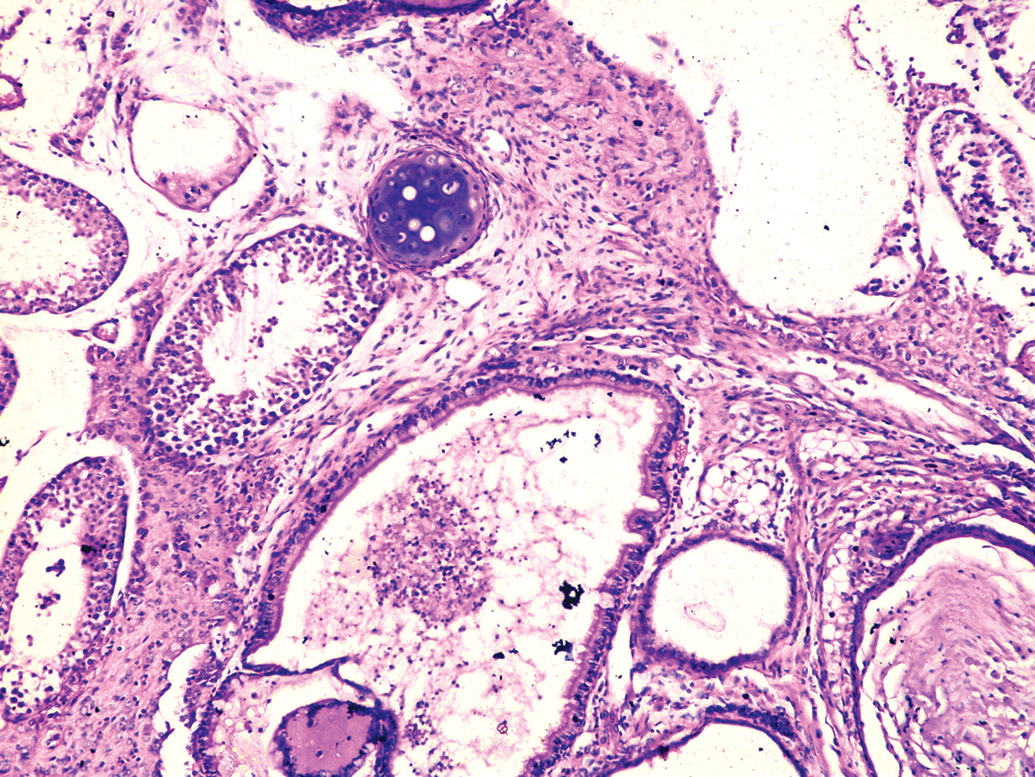
Teratoma in right testis showing area of cartilage tissue and cavity lined by ciliated columnar epithelium. H&E, 10× objective magnification.
Microscopic evaluation of the left epididymis revealed complete absence of spermatozoa in the tubules, which was attributed to the replacement of spermatogenic seminiferous tubules by the tumor mass in the left testis. Other collected organs including heart, lung, kidneys, brain, liver, spleen, and other visceral organs did not show any lesion on microscopic evaluation.
In the left testis, majority area of the sections was occupied by tissues of choriocarcinoma and yolk sac carcinoma. Small foci of embryonal carcinoma were observed in the last few sections of the testis. Hence, choriocarcinoma, yolk sac carcinoma, and embryonal carcinoma were identified as three distinct tumors. Taken together, the diagnoses were primary spontaneous non-metastatic choriocarcinoma, yolk sac carcinoma, and embryonal carcinoma in the left testis and teratoma in the right testis of a Swiss albino mouse. The presence of mixed histologic pattern of nonseminomatous germ cell tumor in left testis supports the hypothesis of histogenesis of the various neoplastic cells from the primitive totipotent cells. In human pathology literature, which is based on World Health Organization classification, such a tumor may be termed as “non-seminomatous mixed type germ cell tumor with differentiation to choriocarcinoma, yolksac carcinoma and embryonal carcinoma” of testis (Burns 2001). However, we found no such type of nomenclature in veterinary pathology literature. To the best of the authors’ knowledge, the current report is the first description of four concurrent nonmetastatic, nonseminomatous germ cell tumors in the testes of a Swiss albino mouse, suggesting that its occurrence is extremely rare in laboratory mice.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
The authors gratefully acknowledge Dr. Isabel Sesterhenn from the Joint Pathology Centre for her advice and assistance with confirmation of the diagnoses, immunostaining, and photography. We thank the Director General and Deputy Director (Technical) of the Central Council for Research in Ayurvedic Sciences, New Delhi for providing necessary facilities to conduct the research.
Authors' Note
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, Army, or Air Force, the Department of Defense, nor the U.S. Government.
