Abstract
The authors evaluated dermal phototoxicity using the world smallest minipig (MMPig: Microminipig). MMPigs were administered 100 mg/kg ciprofloxacin hydrochloride with an infusion pump. The dorsal area of each animal was irradiated with ultraviolet-A irradiation. The left dorsal skin was irradiated at intensities of 5, 10, 15, and 20 J/cm2, and the right dorsal back skin was set as a nonirradiated site. Gross and histopathological examinations were conducted before irradiation and from 1 to 72 hr after irradiation. Initial changes in the skin were necrosis of the basal and/or prickle cell layer and cellular infiltration from 24 hr after irradiation. Vesicle formation observed from 48 hr after irradiation was considered similar to bullous eruptions, a known side effect of fluoroquinolones in humans. Therefore, the authors suggest that the MMPig may be a useful experimental animal model for dermal phototoxicity studies.
Dermatitis has been found to occur in both laboratory animals and humans exposed to ultraviolet radiation following the administration of photoreactive chemicals and drugs, and antibiotics, psoralens, nonsteroidal anti-inflammatory drugs, and tranquilizers are reported to have induced this undesirable adverse effect (Marrot et al. 2003). Among these, fluoroquinolone antibiotics are recognized as a group associated with drug-induced phototoxicity from interaction with ultraviolet light. In vivo models are preferred for estimating fluoroquinolone phototoxicity to humans because they incorporate photoreactivity and toxicity to the skin (Mayne et al. 1997; Owen 1999; Yabe et al. 2005). Therefore, in vivo models have been developed in a variety of animals, such as mice, rabbits, guinea pigs, and swine. Among laboratory animals, the pig has relatively hairless skin that enables the clinical evaluation of surface alterations. The skin of minipigs is also considered a good model for human skin because it is morphologically, physiologically, and pharmacologically similar (Yabuki et al. 2007). Recently, the world's smallest minipig (MMPig: Microminipig; registered as a novel variety of swine with the Japanese Ministry of Agriculture, Forestry, and Fisheries) has emerged as a possible experimental animal model for nonclinical pharmacological/toxicological use (Miyoshi et al. 2010; Murayama et al. 2009; Sugiyama et al. 2011). The MMPig is docile with body weight (BW) at young mature of less than 7 kg, a good manageable size for an experimental animal (Kaneko et al. 2011; Kawaguchi et al. 2011; Kawaguchi et al. 2012; Takeishi et al. 2012). However, dermatological investigations have not been conducted in the MMPig and no basic dermal research data are available. In this study, we evaluated the MMPig following intravenous administration of a second-generation fluoroquinolone (CPFX: ciprofloxacin hydrochloride) and showed the possibility of its use as an animal model for assessing phototoxicity.
Five female MMPig (aged 5–7 months, BW approximately 8–15 kg: Fuji Micra Inc., Shizuoka, Japan) were used. Each animal was provided with mashed diet (Kodakara 73; Marubeni Nisshin Feed Inc., Tokyo, Japan) equivalent to approximately 2% of BW once daily. Tap water was available ad libitum. The animals were singly housed in stainless steel cages [90 (D) × 90 (W) × 80 (H) cm] for beagle dogs in an air-conditioned room (temperature, 22–28°C; humidity, 40–80%; 12-hr light/dark cycle; ventilation 15 times per hour). This study was approved by the Institutional Animal Care and Use Committee of Shin Nippon Biomedical Laboratories, Ltd., Drug Safety Research Laboratories (Kagoshima, Japan) and was conducted in accordance with the ethics criteria contained in the bylaws of the committee.
The cervical region of the skin was incised and one side of the carotid vein exposed under anesthesia. A polyurethane tube was inserted and located in the sinus venarum cavarum after confirmation of regurgitation of blood. A plug for administration set at the end of the tube was filled with physiological saline with heparin (50–100 U/mL). All pigs were fasted overnight before the procedure. On the day on which the catheter was set in place and on the following 3 days, a postoperative analgesic and an antimicrobial prophylactic were administered for pain relief and infection prevention, respectively.
CPFX (Wako Pure Chemical Industries, Ltd., Osaka, Japan) was diluted to 20 mg/mL in physiological saline. CPFX 100 mg/kg was set as the dose level expected to induce phototoxicity, as previously reported in mice (Yabe et al. 2005).
In a pilot experiment, a female MMPig was dosed at a rate of 5 ml/kg/hr for 60 min to determine the time for CPFX concentration to reach the steady-state level. The plasma concentration level increased in a linear manner immediately after the end of the administration. In simulations at 2.5 ml/kg/hr, half the pilot dosing rate, at least 2 hr were required for the concentration to reach the steady-state level, and it was considered that prolonged continuous dosing would overload the MMPig. Therefore, to shorten the time for the concentration to reach the steady-state level, the initial dosing rate was set at 10 ml/kg/hr for 10 min followed by 2.5 ml/kg/hr for 80 min. To confirm this dosing rate and time, three MMPigs were used to calculate the plasma concentrations of ciprofloxacin. Blood was drawn from the sinus venarum cavarum with a syringe containing heparin sodium. Blood was drawn before dosing, and at 10, 15, 20, 30, 40, 55, 90, 105, and 150 min after dosing. As shown in Figure 1, maximum plasma drug concentration was reached by 10 min after the start of administration in three animals and the mean concentration was 48.8 ± 6.1 µg/mL. Although the plasma concentration level decreased thereafter, it was confirmed that the steady-state level was maintained under continuous dosing. From these results, the initial dosing rate was set at 10 ml/kg/hr for 10 min followed by 2.5 ml/kg/hr for 80 min and the irradiation time of ultraviolet-A (UVA) was set from 15 to 75 min after the start of dosing.
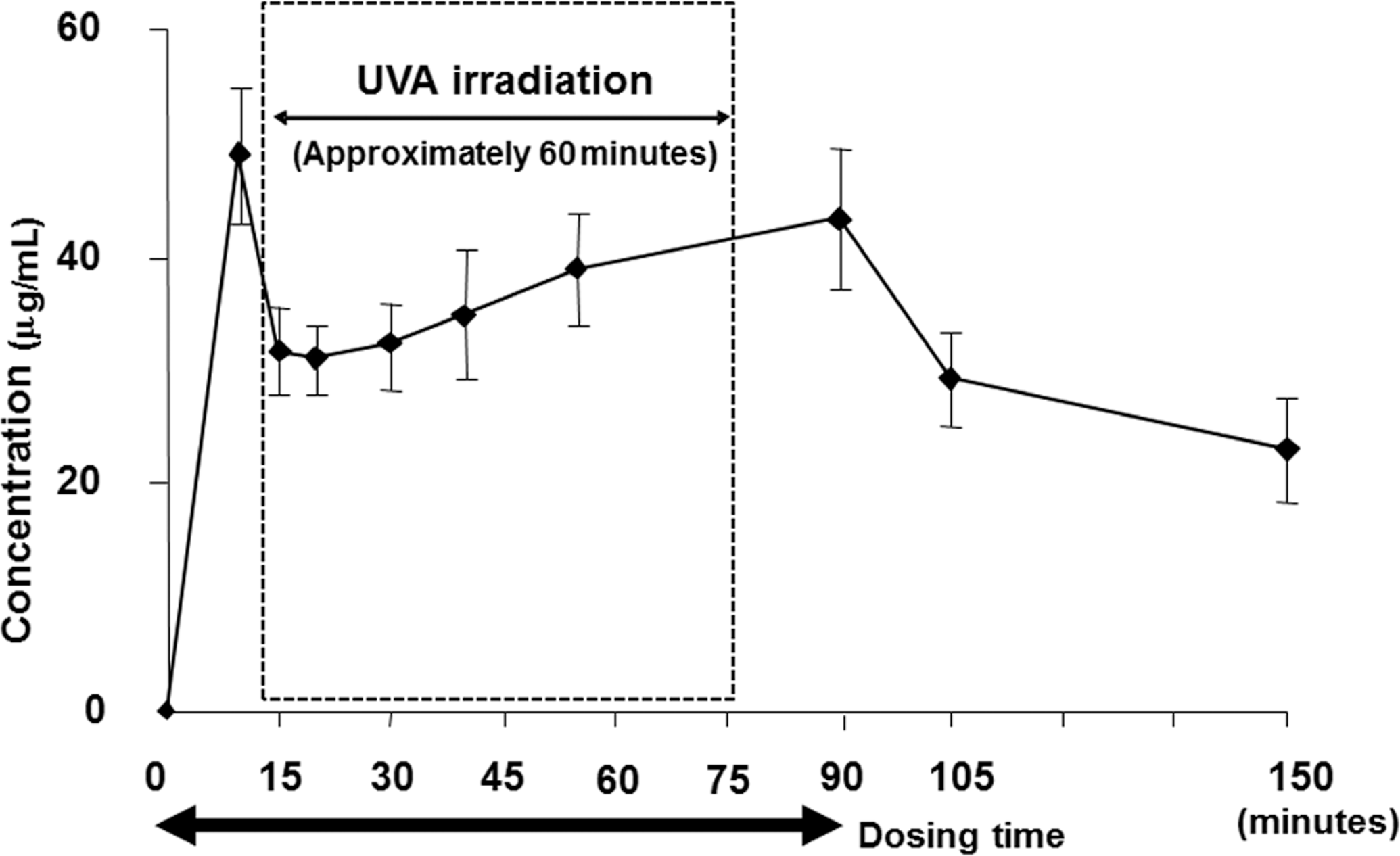
Mean plasma concentrations of CPFX after single intravenous infusion at a dose of 100 mg/kg to three MMPigs in a pilot experiment.
In this study, fur on the back of each animal was shaved off with an electric clipper on the day before administration. The dorsal area of each animal was divided into eight parts (A–H) [1 part; 2.5 cm × 2.5 cm] (Figure 2a). The left back skin (A–D) was irradiated with UVA. The right back skin (E–H), as the control site, was not irradiated. The right back skin (E–H) was covered with a sheet of aluminum foil affixed with adhesive tape. Each animal was held in a restraint without sedation or anesthesia, and the dosing formulation was administered via the tube, using an infusion pump (Braintree BSP-99 M, Braintree Scientific Inc., MA). Approximately 15 min after the start of administration, when the concentration had reached the steady-state level, irradiation of the animals with UVA at approximately 5, 10, 15, and 20 J/cm2 (5.6 W/cm2, approximately 15, 30, 45, and 60 min) was initiated, using an ultraviolet irradiation apparatus (Dermaray, M-DMR- 50, Eisai Co., Ltd., Tokyo, Japan). The foil was removed sequentially after 0, 15, 30, and 45 min from the 20, 15, 10, and 5 J/ cm2 irradiation sites, respectively (Figure 2b).
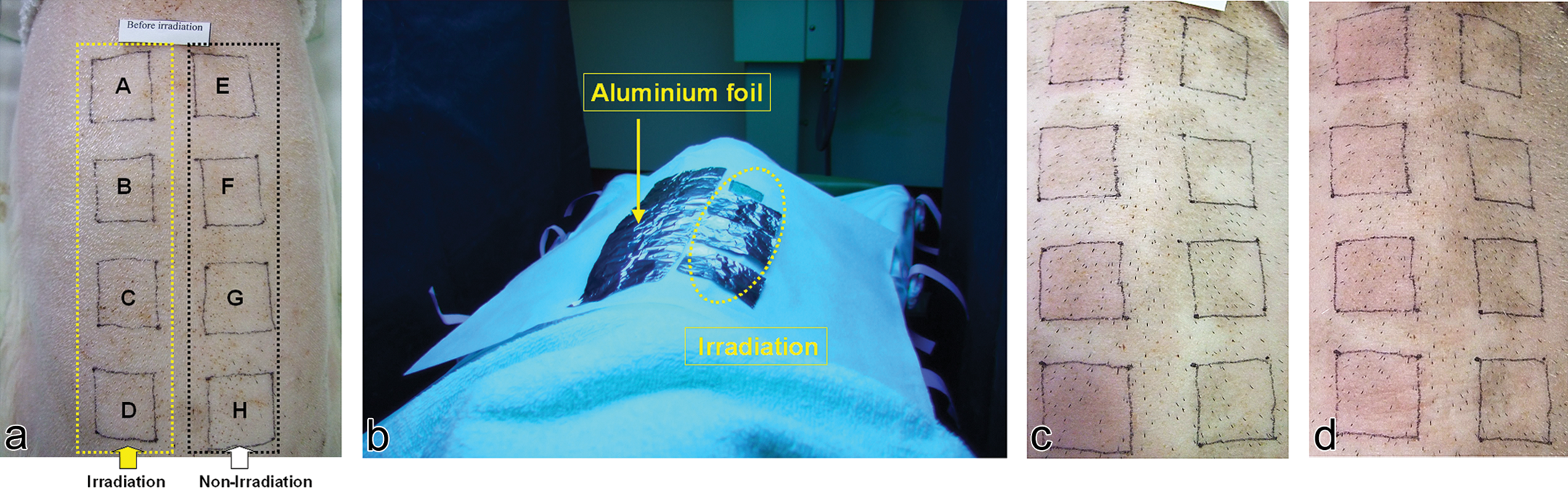
(a) Back skin of the microminipig in the Animal No. 5. Irradiation site is A–D and nonirradiation site is E–H. (b) Irradiation was initiated with all sites other than the 20 J/ cm2 ultraviolet-A (UVA) site covered with aluminum foil. Macroscopic examination. (c) At 24 hr after irradiation, well-defined erythema (score 2) was observed at the 10(C), 15(D), and 20(A) J/cm2 irradiation sites in the Animal No. 5. (d) At 72 hr after irradiation, well-defined erythema (score 2) was observed at the 20(A) J/cm2 irradiation sites in the Animal No. 5.
The back skin was observed before UVA irradiation and at 1, 24, 48, and 72 hr after UVA irradiation (Animal Nos. 2–5). One animal was observed at 1, 4, 8, 12, and 24 hr (Animal No. 1). Skin reactions at the irradiation sites were evaluated in accordance with Draize’s criteria (Draize, Woodard, and Calvery 1944) for erythema and edema.
Histopathological examinations of the back skin were conducted after euthanasia and necropsy. Animal Nos. 2 through 5 were necropsied 1, 24, 48, and 72 hr, respectively, after exposure to ultraviolet radiation. Animal No.1 was not necropsied. The animals were anesthetized and euthanized by exsanguination. The back skin was removed and fixed in 10% neutral buffered formalin for histopathological examination. The specimens were embedded in paraffin, sectioned, stained routinely with Hematoxylin–Eosin (HE) stain, and examined histopathologically.
For immunohistochemical examination, the sections were deparaffinized in xylene and rehydrated through graded alcohol. The primary antibodies and concentrations were as follows: Iba1 (polyclonal, 1:500 dilution; Wako Pure Chemical Industries, Ltd., Osaka, Japan) for macrophage marker, CD3 (F7.2.38; mouse monoclonal, 1:400 dilution; Dako Cytomation Co., Ltd., Kyoto, Japan), and CD79α (HM47/A9; mouse monoclonal, 1:200 dilution; Abcam plc., Cambridge, UK). The sections treated with primary antibody were incubated with the appropriate biotinylated secondary antibody with EnVision (Dako Cytomation Co., Ltd.). Immunoreactivity was visualized with 0.075% 3,3′-diaminobenzidine tetrachloride. The sections were then washed, counterstained, dehydrated, cleared in xylene, and mounted. The liver of MMPig was set as a positive control specimen to Iba1 and the spleen of MMPig as a positive control specimen to CD3 and CD79α.
At 1, 4, and 8 hr after irradiation, no skin reaction was grossly observed at the 5–20 J/cm2 site. At 12 hr after irradiation, very slight erythema was observed at the 5–20 J/cm2 site, and no edema was observed at the 5–15 J/cm2 site. At 24 hr after irradiation, very slight erythema was observed at the 5 J/cm2 site, well-defined erythema was observed at the 10–20 J/cm2 site and no edema was observed at the 5–20 J/cm2 site. At 48 hr after irradiation, very slight erythema was observed at the 5–15 J/cm2 site, well-defined erythema was observed at the 15–20 J/cm2 site, no edema was observed at the 5–15 J/cm2 site, and very slight edema was observed at the 15–20 J/cm2 site. At 72 hr after irradiation, very slight erythema was observed at the 5–15 J/cm2 site, well-defined erythema was observed at the 20 J/cm2 site, no edema was observed at the 5–10 J/cm2 site, and very slight edema was observed at the 15–20 J/cm2 site (Tables 1, Figure 2c and d). Clinical manifestations of erythema and edema in this study were consistent with those reported previously in humans (Lipsky and Baker 1999). Macroscopic examination showed that erythema first appeared from 12 hr after irradiation. While erythema at the low-intensity irradiation sites decreased with time, edema at the high-intensity sites was observed at 48 and 72 hr after irradiation.
Macroscopic findings from 1 to 72 hr.
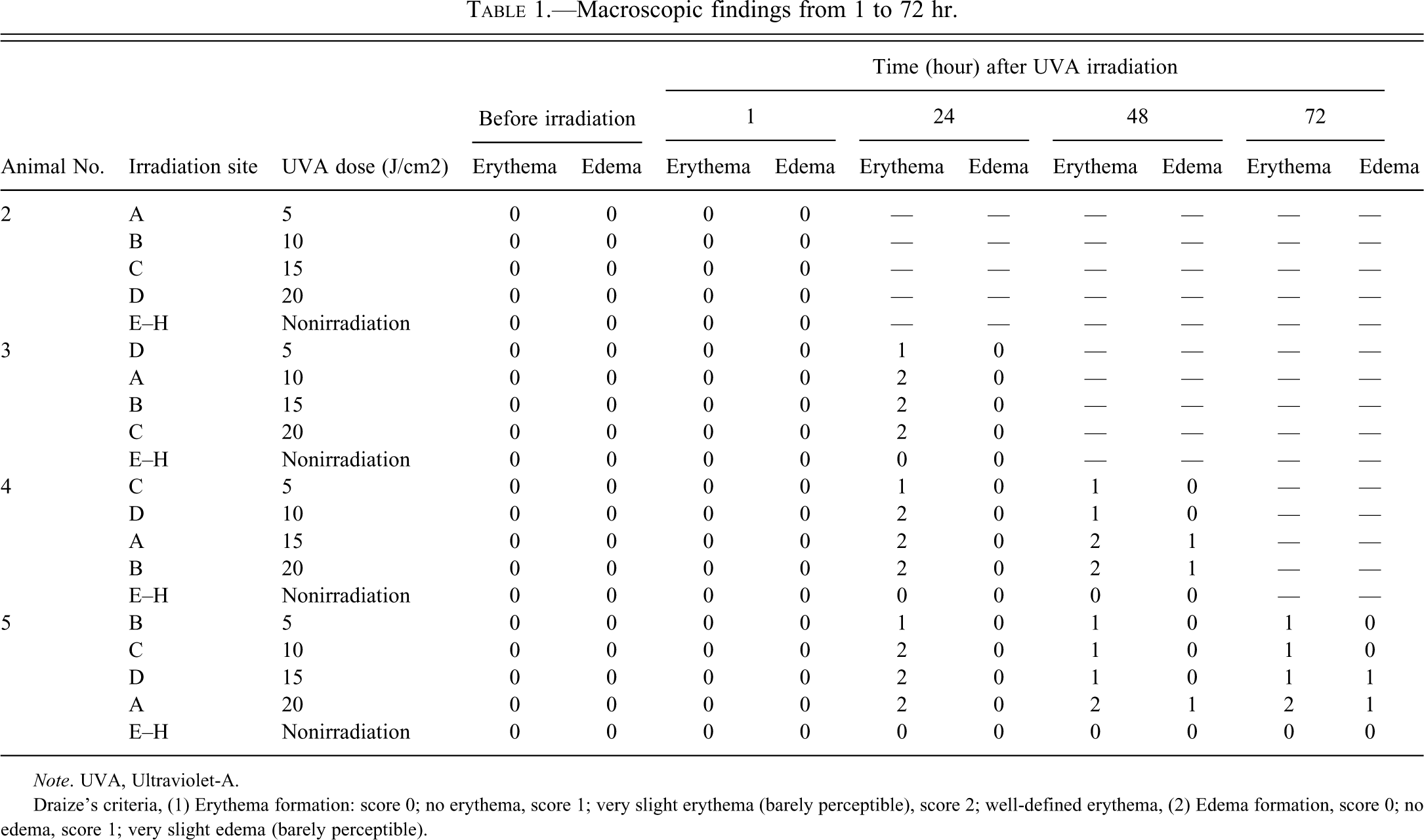
Note. UVA, Ultraviolet-A.
Draize’s criteria, (1) Erythema formation: score 0; no erythema, score 1; very slight erythema (barely perceptible), score 2; well-defined erythema, (2) Edema formation, score 0; no edema, score 1; very slight edema (barely perceptible).
Histopathological findings from 1 to 72 hr after irradiation are shown in Table 2. At 1 hr after irradiation, no histopathological changes were observed in any animal (Figure 3a). Necrosis of the basal and/or prickle cell layer of the epidermis was observed from 24 hr after irradiation (Figure 3b). This lesion was considered an initial change from dermal phototoxicity. Vesicle formation in the dermal–epidermal junction and cellular infiltration in the vesicle was observed from 48 hr after irradiation (Figure 3c). These changes tended to be more severe at high-intensity sites than at low-intensity sites. Regeneration of the epidermis observed from 48 hr after irradiation was considered a reactive change against necrosis of the basal and/or prickle cell layer of the epidermis.
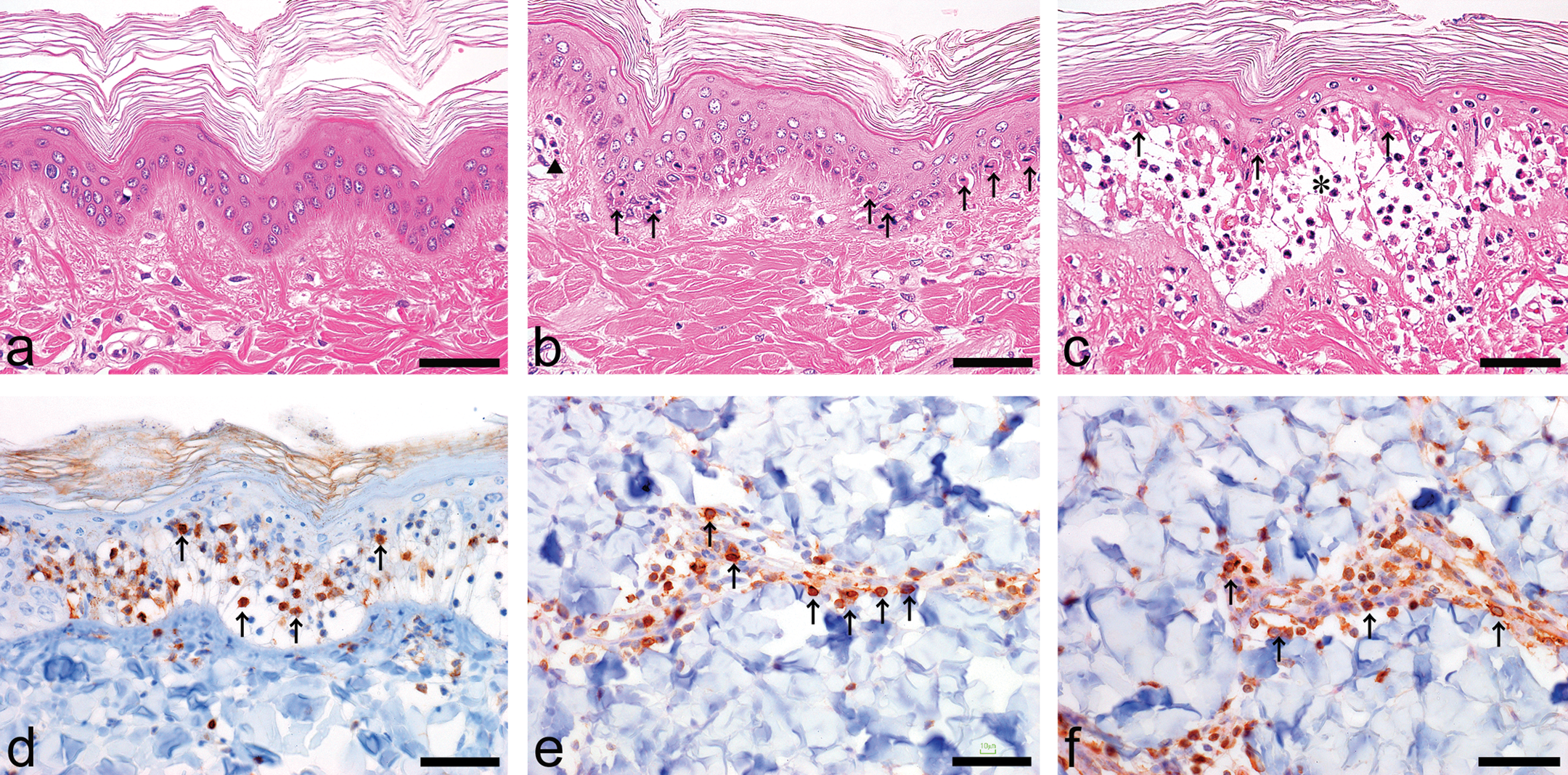
Histopathological examination (a–c: Hematoxylin–Eosin [HE] stain). (a) At 1 hr after irradiation, no abnormal changes were observed at the 20 J/cm2 irradiation sites. (b) At 24 hr after irradiation, necrosis of the basal and/or prickle epidermal cell layer (arrows), and cellular infiltration in the dermis (triangle) were observed at the 20 J/cm2 irradiation sites. (c) At 72 hr after irradiation, necrosis of basal and/or prickle epidermal cell layer (arrows), vesicle formation in the subepidermis (*), and cellular infiltration in the epidermis/dermis were observed at the 20 J/cm2 irradiation sites. Immunohistochemical examination (d–f). At 72 hr after irradiation, (d) Iba-1, (e) CD3, and (f) CD79α (each arrows) were detected, suggesting macrophages, T-cells, and B-cells, respectively. Bars: 50 µm.
Histopathological findings from 1 to 72 hr.
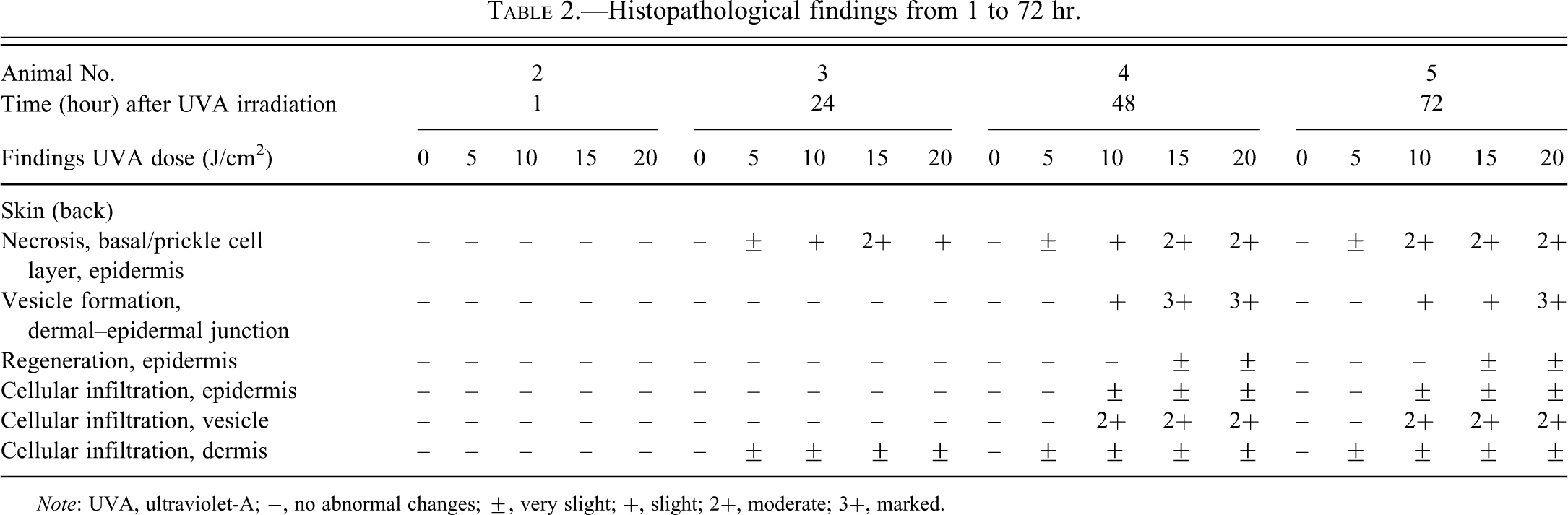
Note: UVA, ultraviolet-A; −, no abnormal changes; ±, very slight; +, slight; 2+, moderate; 3+, marked.
Similar histopathological lesions, such as cellular infiltration in the auricle, had been observed in mice that received a single intravenous administration of 100 mg/kg ciprofloxacin at 96 hr after irradiation (Yabe et al. 2005). To our knowledge, time-dependent histopathological examinations of other experimental animals have not been conducted and vesicle formation in the subepidermis has not been reported. It is reported that photosensitivity reactions may generally appear within a few days and these vesicular lesions are similar to bullous eruption, a known dermatological side effect associated with fluoroquinolone in humans (Lipsky and Baker 1999).
Cellular infiltration observed in the epidermis, vesicles, and dermis at the 20 J/cm2 UVA irradiation sites at 72 hr was examined immunohistochemically. Iba-1 positive cell infiltration was observed mainly in the epidermis and vesicles, suggesting mainly macrophages (Figure 3d) because positive staining was detected in macrophages such as Ito cells in the liver. CD3 positive cell infiltration was observed mainly in the dermis, suggesting T-cell (Figure 3e) because positive staining was detected in lymphocytes in the region around splenic lymph nodule and/or white pulp arteries such as sheathed artery. CD79α positive cell infiltration was also observed mainly in the dermis, suggesting B-cells (Figure 3f) because positive staining was detected in lymphocytes in the splenic lymph nodule regions. To our knowledge, immunohistochemical staining techniques for macrophages, T-cells, and B-cells using antibodies against Iba-1, CD3, and CD79α have not been previously reported for the MMPig. Therefore, this immunohistochemical information is considered helpful for further experiments with the MMPig.
In conclusion, this first report of a dermal phototoxicity study in the MMPig demonstrates its potential suitability as a new experimental animal model.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by Health Labour Sciences Research Grant (No. 33361105) from the Ministry of Health, Labour and Welfare of Japan (to AT and HK), Adaptable and Seamless Technology transfer Program (A-Step No. AS2316907E) from the Ministry of Education Culture, Sports, Science and Technology of Japan (to AT and HK), Suzuken Memorial Foundation (to NM and AT) and SENSHIN Medical Research Foundation (to HK, AT, NM).
Abbreviations
Acknowledgments
The authors are grateful to Mr. T. Motokado, Mr. T. Okazaki, Dr. Y. Ooshima, and Dr. T. Sukamoto (Shin Nippon Biomedical Laboratories, Ltd.) for their valuable technical assistance and advice.
