Abstract
The efferent ducts represent an important site of toxicity in the male reproductive tract but are not routinely examined in toxicity studies. This article describes a primary efferent duct toxicity that resulted in secondary testicular changes in rats. Male rats were administered LTI-1, a leukotriene A4 hydrolase inhibitor, at doses up to 250 mg/kg/d for 3 month or 150 mg/kg/d for 6 month. At the highest dose levels, testicular changes were predominantly unilateral and characterized by diffuse dilation or atrophy of the seminiferous tubules. These testicular changes correlated with granulomatous inflammation in the corresponding efferent ducts, suggesting that the mechanism for the testicular changes involves obstruction and impaired fluid reabsorption in the efferent ducts. Subsequent buildup in fluid volume and back-pressure upstream of the blockage cause dilation of the seminiferous tubules, which, in its late stages, progress to tubular atrophy. There are important differences in efferent duct anatomy between rats and larger mammals, including humans, such that the latter are less susceptible to testicular injury by this mechanism. Because of the limited relevance of this rat-specific finding to humans, it is important to distinguish testicular changes secondary to efferent duct toxicity from primary drug-induced testicular toxicity.
Keywords
Introduction
Testicular atrophy is an end-stage lesion that can occur by many different mechanisms, including effects on Sertoli cells, Leydig cells, germ cells, disturbance of blood flow, and fluid balance disturbance (Creasy 2001). Regardless of the primary site of injury or mechanism of toxicity, most testicular toxicants cause degeneration and depletion of germ cells from the seminiferous tubules, culminating in tubules lined only with Sertoli cells (tubular atrophy). Identifying the primary target and mechanism of toxicity requires a detailed examination of testes, efferent ducts, and epididymides at an early stage of lesion development. This article describes a primary efferent duct toxicity that resulted in testicular injury, secondary to buildup of fluid (pressure atrophy) in the seminiferous tubules. An important characteristic of the testicular lesion was its predominantly unilateral occurrence, which is unusual for most xenobiotic-induced lesions.
LTI-1, a proprietary compound with an arbitrary identifier, is a leukotriene A4 hydrolase inhibitor that is being investigated as an anti-inflammatory therapeutic agent. Repeat-dose toxicity studies were conducted in rats and dogs as part of drug development. In a 3-month study in rats, unilateral seminiferous tubular dilation, degeneration, and atrophy were observed at relatively high incidences in the high-dose (250 mg/kg/d) group. Test article–related unilateral testicular changes are unusual, but this pattern of changes has been described in the testis as a secondary effect of obstruction and impaired fluid reabsorption in the efferent ducts (Hess 1998; Gotoh et al. 1999; Creasy 2001). The presence of granulomatous inflammation in the efferent ducts can cause blockage of fluid outflow and subsequent dilation of the upstream seminiferous tubules and end-stage progression to tubular atrophy from increased fluid volume and backpressure. In this study, rats were administered LTI-1 for up to 6 months to test the hypothesis that the testicular changes observed in the previous 3-month study were secondary to primary effects in efferent ducts. The characteristic morphological profile and the implications of this mechanism of testicular toxicity for human risk assessment are discussed.
Materials and Methods
This study was conducted at Huntingdon Life Sciences (East Millstone, NJ), which is fully accredited with the Association for Assessment and Accreditation of Laboratory Animal Care International. The study was conducted under U.S. Food and Drug Administration Good Laboratory Practice standards (21 CFR 58) for preclinical laboratory studies. All procedures were reviewed by the Institutional Animal Care and Use Committee. The study assessed toxicity in male and female rats following LTI-1 administration for up to 6 months, but only data related to the male reproductive tract are provided and discussed.
Animals and Compound Administration
Male Sprague-Dawley (Crl: CD[SD]IGS BR) rats were purchased from Charles River Laboratories (Kingston, NY), were acclimated for 2 weeks, and were approximately 7.5 weeks old at initiation of dosing. Rats were individually housed in elevated stainless-steel cages. The study room was maintained on a 12-hour light-dark cycle and within temperature and humidity ranges of 18°C to 26°C and 30% to 70%, respectively. Rats were fed with Certified Global 16% Protein Rodent Diet No. 2016C (Harlan Teklad, Madison, WI), and water was provided ad libitum by an automatic watering system.
Rats (n = 15 per group) were randomized to receive vehicle (distilled deionized water) or 10, 50, or 250 mg/kg/d of test article for 3 months for the interim phase analysis (Table 1). For the terminal phase analysis, rats (n = 20/group) were randomized to receive vehicle or 10, 50, or 150 mg/kg/d of test article for 6 months. An additional five animals in the vehicle and 150 mg/kg/d groups dosed for 6 months were assessed for reversal of effects after completion of an additional 3-month test article–free recovery period.
Study design and group sizes
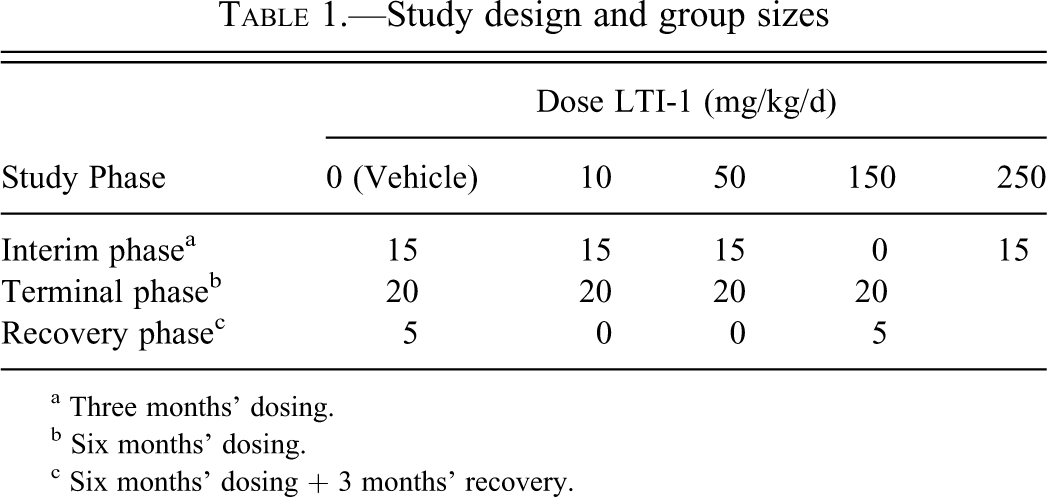
a Three months’ dosing.
b Six months’ dosing.
c Six months’ dosing + 3 months’ recovery.
The 250 mg/kg/d dose group was included in the 3-month interim phase of the study to confirm an apparent treatment relationship of tubular atrophy and tubular dilation that had occurred in an earlier 3-month study. The dose levels included in the 6-month phase of the study were chosen to establish a no-observed adverse effect level (NOAEL) for the testicular changes following 6 months of dosing.
LTI-1 was formulated in vehicle (water) and administered to rats once daily by oral gavage. Animals in the vehicle groups were administered water by gavage. Various in-life, clinical pathology, and toxicokinetic parameters were assessed but not included in this article.
Endocrine Assessment
Sera for endocrinology were collected from all animals at necropsy after 3 and 6 months of dosing and at the end of the recovery period. Follicle-stimulating hormone (FSH) was determined by ELISA (rat FSH ELISA, IDS/Biocode-Hycel, Liege, Belgium). Testosterone and luteinizing hormone (LH) were determined by radioimmunoassay (Coat-a-Count Testosterone, Siemens Medical Solutions Diagnostics, Los Angeles, CA; and Rat LH [125I] RIA kit, IZOTOP, Budapest, Hungary, respectively).
Necropsy
Necropsy was performed on 15 animals per group after 3 months of treatment (interim phase necropsy), up to 20 animals per group after 6 months of treatment (terminal phase necropsy), and 5 animals per group after 6 months of treatment followed by a 3-month recovery period (recovery phase necropsy). Animals were fasted overnight and euthanized by isoflurane anesthesia followed by exsanguination. Various tissues and organs were collected for macroscopic and microscopic examination, but only data for testes, epididymides, and efferent ducts are reported here. Testes were preserved using Modified Davidson’s solution for at least 48 h followed by storage in 10% neutral buffered formalin. Efferent ducts and epididymides were preserved using 10% neutral buffered formalin.
Collection of Efferent Ducts
The right and the left epididymal fat pads were collected at necropsy with their corresponding testis and epididymis still attached. Dissection of the efferent ducts began with careful cutting of the ligament that attaches the epididymal head to the testis (Fig. 1A). Once the epididymis was free of the testis, it was possible to view the efferent ductules and blood vasculature entering the testicular hilus (Fig. 1B). Using a dissecting microscope with transillumination, the efferent ducts were identified within the right and left epididymal fat pads, and then most of the fat around the length of the efferent ducts was removed using small scissors, being careful not to cut or squeeze the efferent ductules. The anterior end of the efferent ducts was then carefully sectioned from the testis and the posterior end removed from the epididymis. Tissue ink was used to identify the orientation of distal and proximal ends. After allowing the ink to fully dry, the efferent ducts were placed between tissue sponges in small-tissue cassettes to keep them flat during fixation and processing.
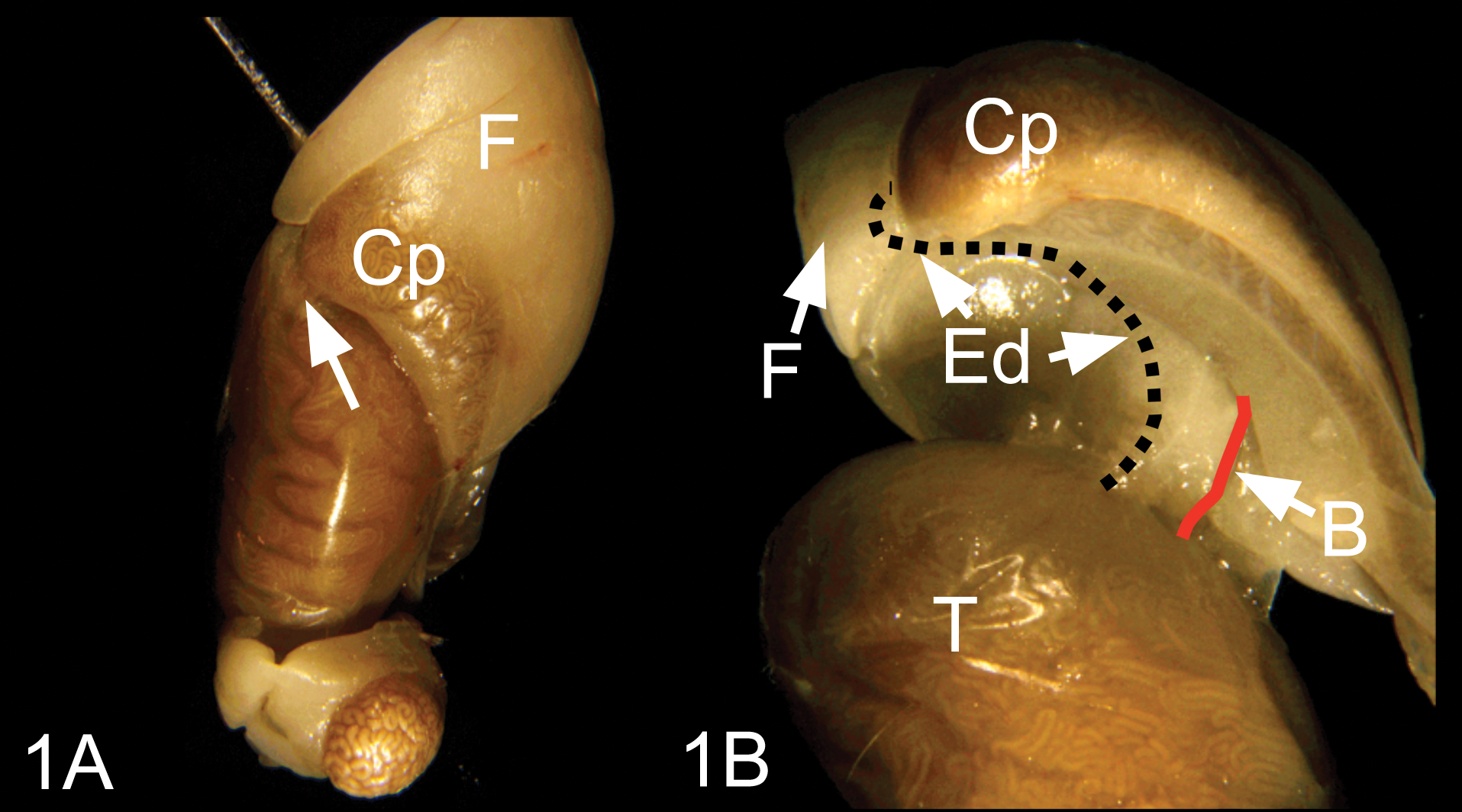
(A) Gross dissection of the testis and attached epididymis, with the epididymal fat pad (F). To dissect the efferent ductules, first cut the fine ligament (arrow) that attaches the caput epididymis (Cp) to the tunica albuginea of the testis. (B) After cutting the thin ligament, lay open the caput epididymis (Cp) with attached fat pad (F) to reveal the efferent ductules (Ed) and blood vasculature (B) areas, which are best observed with transilluminated light. T, testis.
The above methodology was used successfully to sample the efferent ducts in this current study. Alternative modifications of the dissection technique may also be tried. These include (1) leave the efferent ductules within the epididymal fat pad and attached to the epididymis while cutting them away from the testis at the tunica albuginea and (2) leave the efferent ductules attached to the testis and to the head of the epididymis by cutting through the caput epididymis. This will give only a very slight increase in testis weight but gives an excellent view of rete testis and proximal efferent ductules. When the efferent ductules are left attached to the head of the epididymis, it is difficult to slice the tissues in a way to align the efferent ductules with the head of the epididymis. The ductules are tortuous and enter the epididymal capsule from an angle, rather than a straight line from the rete testis. Therefore, it is best to cut the epididymis first by transverse plane at the mid caput region and then turn the caput epididymis, with attached ductules, so that a sagittal section can be made, leaving two half-sections for embedding.
Histopathology
Tissues were routinely processed, embedded in paraffin, sectioned at approximately 5 µm, mounted on glass slides, and stained with hematoxylin and eosin. Step serial sections were taken through the efferent duct samples to ensure that proximal and distal regions of the ducts were available for examination (Fig. 2). Efferent duct sections also were stained using periodic acid–Schiff (PAS). Light microscopy examination was performed by a pathologist experienced in evaluating the rodent male reproductive tract.
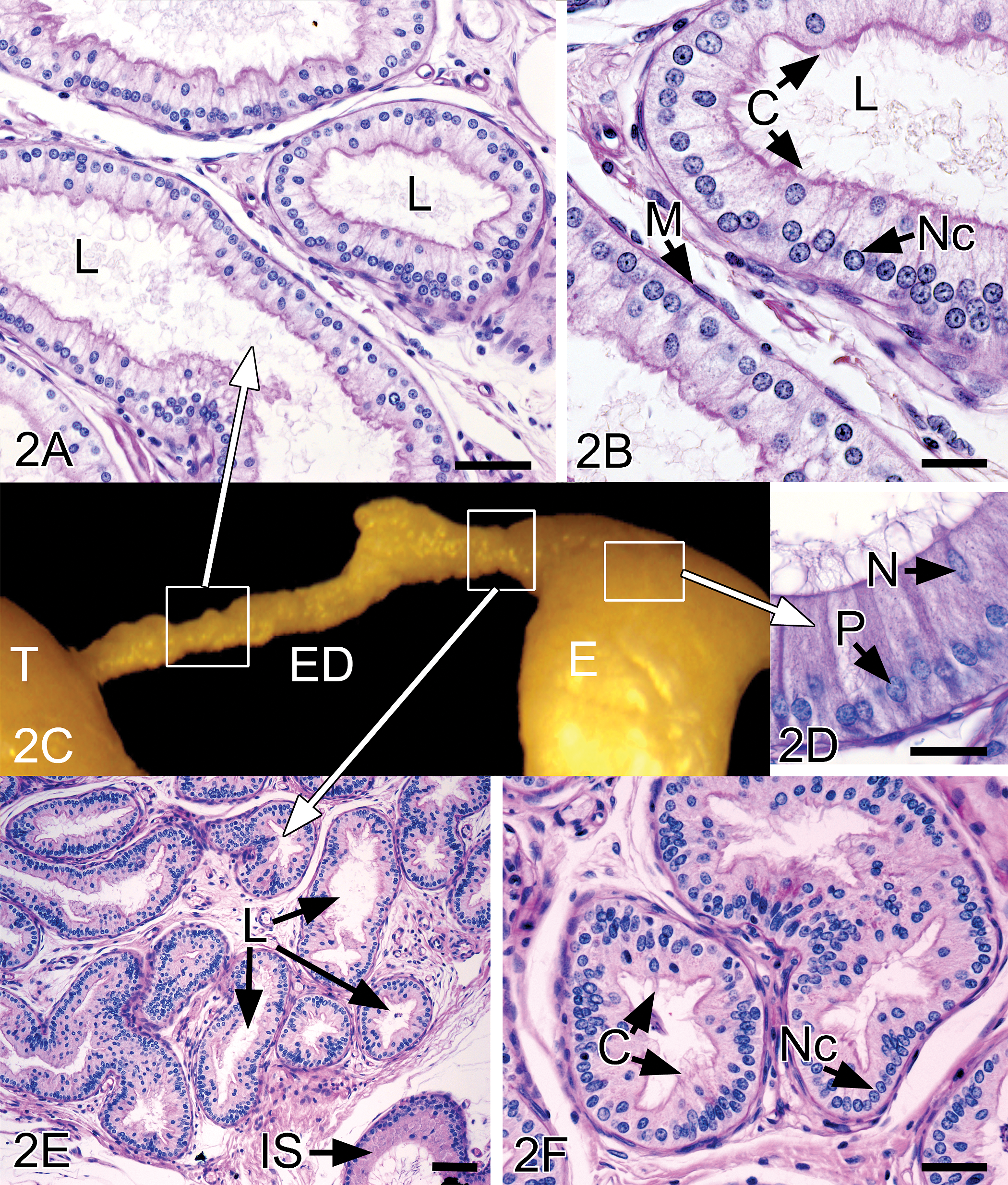
(A) Proximal efferent ductules from vehicle group rat showing normal histomorphology with open, wider lumens (L) than distal regions Bar = 25 µm. (B) Intermediate magnification shows a lumen (L) lined with epithelium consisting of nonciliated (Nc) and ciliated (C) cells. Periodic acid/Schiff’s staining is observed along the microvilli lining the lumen. The ductules are lined by a single layer of flattened smooth muscle cells (M). The epithelium consists of nonciliated (Nc) and ciliated (C) cells resting on a single layer of flattened smooth muscle cells (M). Periodic acid/Schiff's staining is observed along the microvilli lining the lumen. Bar = 10 µm. (C) Gross dissection of the testis (T), efferent ductules (ED), and head of the epididymis (E), after removal of the epididymal fat pad. Boxes and arrows indicate the regions from
Results
Hormone Analysis
Detectable levels of FSH and testosterone were present in sera from vehicle and LTI-1 groups. Levels of LH were below the lower limit of quantification of 0.78 ng/mL in all groups at the interim analysis but detectable at the terminal analysis. FSH, LH, and testosterone levels in male rats administered LTI-1 were generally similar to those of the vehicle-treated controls and showed no dose-related changes (data not presented).
Histomorphologic Evaluation: 3-Month Interim Phase
Macroscopic and microscopic findings were present in the testes, efferent ducts, and/or epididymides in the 250 mg/kg/d group following 3 months of dosing. Macroscopic findings included unilateral small and/or soft testis, unilateral enlarged testis, abscess in efferent duct, and small epididymis. On microscopic examination, severe granulomatous inflammation in the efferent duct correlated with the unilateral abscess, severe unilateral tubular atrophy correlated with unilateral small and/or soft testis, and tubular dilation correlated with unilateral enlargement of the testis.
Efferent ducts
Microscopic examination revealed unilateral or bilateral granulomatous inflammation in the proximal region of the efferent ducts in 9 of 15 males in the 250 mg/kg/d group (Table 2). The granulomatous inflammation was characterized by infiltration of the efferent ductal epithelium, lumen, and interstitial tissue by macrophages and multinucleated giant cells mixed with plasma cells, lymphocytes, and eosinophils; the severity of the lesions ranged from slight to severe (Fig. 3). Other efferent duct changes included minimal to slight dilation of the duct lumens and/or vacuolar degeneration of the epithelium lining. The granulomatous inflammation often resulted in occlusion of ducts, but this was frequently observed in only one ductule, with other ductules appearing to have patent or open lumens.
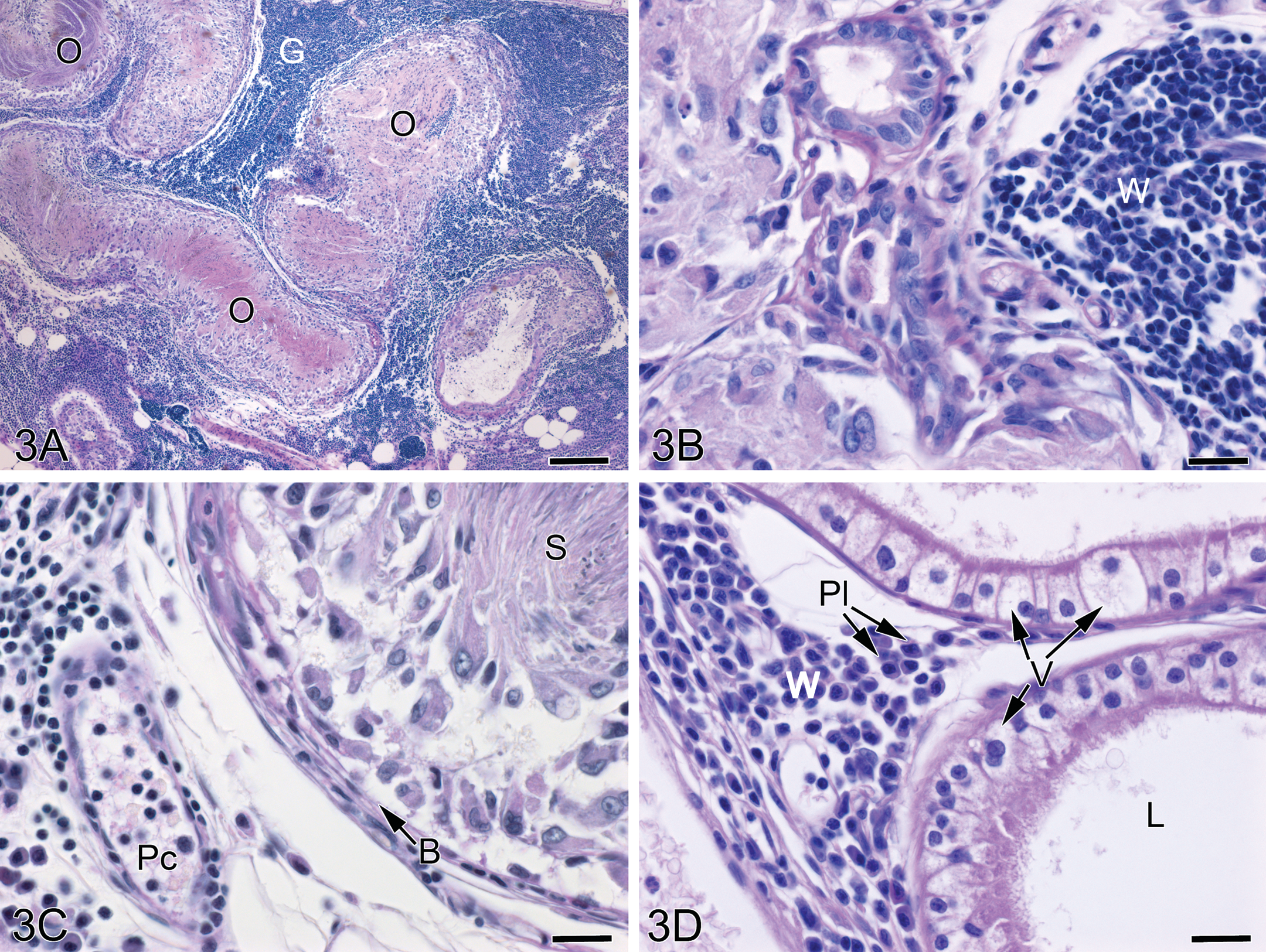
Efferent ductules from the 250 mg/kg/d group rats after 3 months of dosing showing marked granulomatous inflammation. (A) Occluded efferent ductule lumens (O) are seen with compacted sperm, and large interstitial spaces between the ductules are filled with inflammatory cells of the granuloma (G). Bar = 200 µm. (B) White blood cells (W) of the granuloma are found adjacent to the ductule epithelium. Bar = 20 µm. (C) An occluded ductule is noted by the basement membrane (B) with an aggregate of sperm (S) in the lumen surrounded by macrophages and epithelial cells that have moved away from the basement membrane. Numerous white blood cells are found in the interstitium and within the postcapillary venule (Pc). Bar = 20 µm. (D) The lumen (L) of efferent ductules devoid of sperm is apparently downstream of occlusions. The epithelial cells appear to be highly vesiculated (V). White blood cells, including plasma cells (Pl), are seen in a small aggregate. Bar = 20 µm.
Incidence of histomorphologic changes in the testes and efferent ducts at the Interim (3-Month) necropsy.
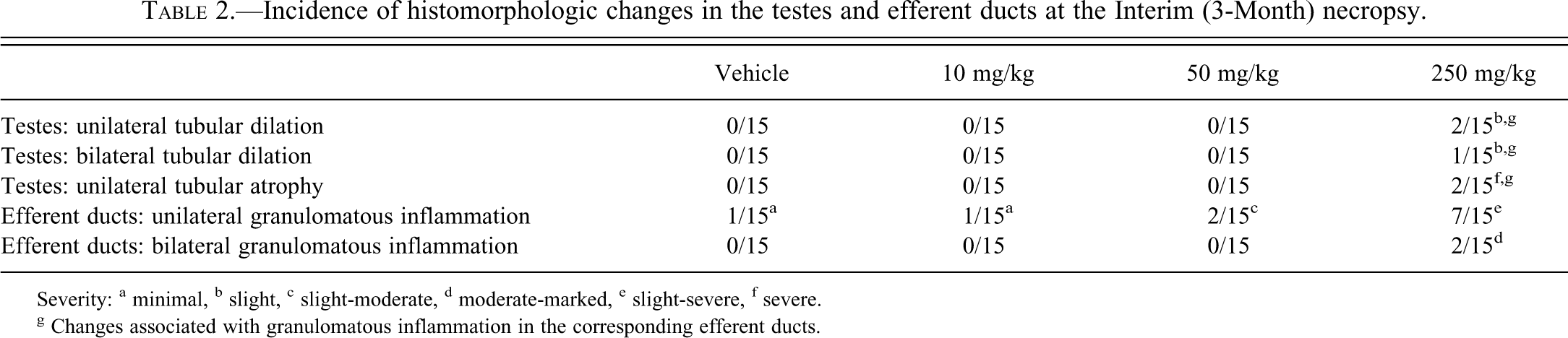
Severity: a minimal, b slight, c slight-moderate, d moderate-marked, e slight-severe, f severe.
g Changes associated with granulomatous inflammation in the corresponding efferent ducts.
Testes
In 5 of the 9 affected males in the 250 mg/kg/d group, the presence of granulomatous inflammation in the efferent ducts was associated with secondary changes in the corresponding testis (mostly unilateral), which included severe seminiferous tubular atrophy or slight diffuse tubular dilation (Table 2). Tubular dilation was characterized by tubules with dilated lumens surrounded by a seminiferous epithelium that was thinned but that contained a normal complement of germ cell layers with normal cell associations (Fig. 4). In testes with severe tubular atrophy, there was loss of all germ cells from the seminiferous epithelium, leaving tubules lined only by Sertoli cells. An unusual characteristic of the atrophic tubules was that the lumens were dilated, whereas atrophic seminiferous tubules in most other situations are shrunken and have closed lumens. All animals with testicular atrophy or tubular dilation had granulomatous inflammation in the corresponding efferent ducts, but not all animals that had granulomatous inflammation of the efferent ducts had testicular changes. This suggests that the efferent duct inflammation is the primary toxicological lesion that leads to secondary testicular changes in only a proportion of cases.
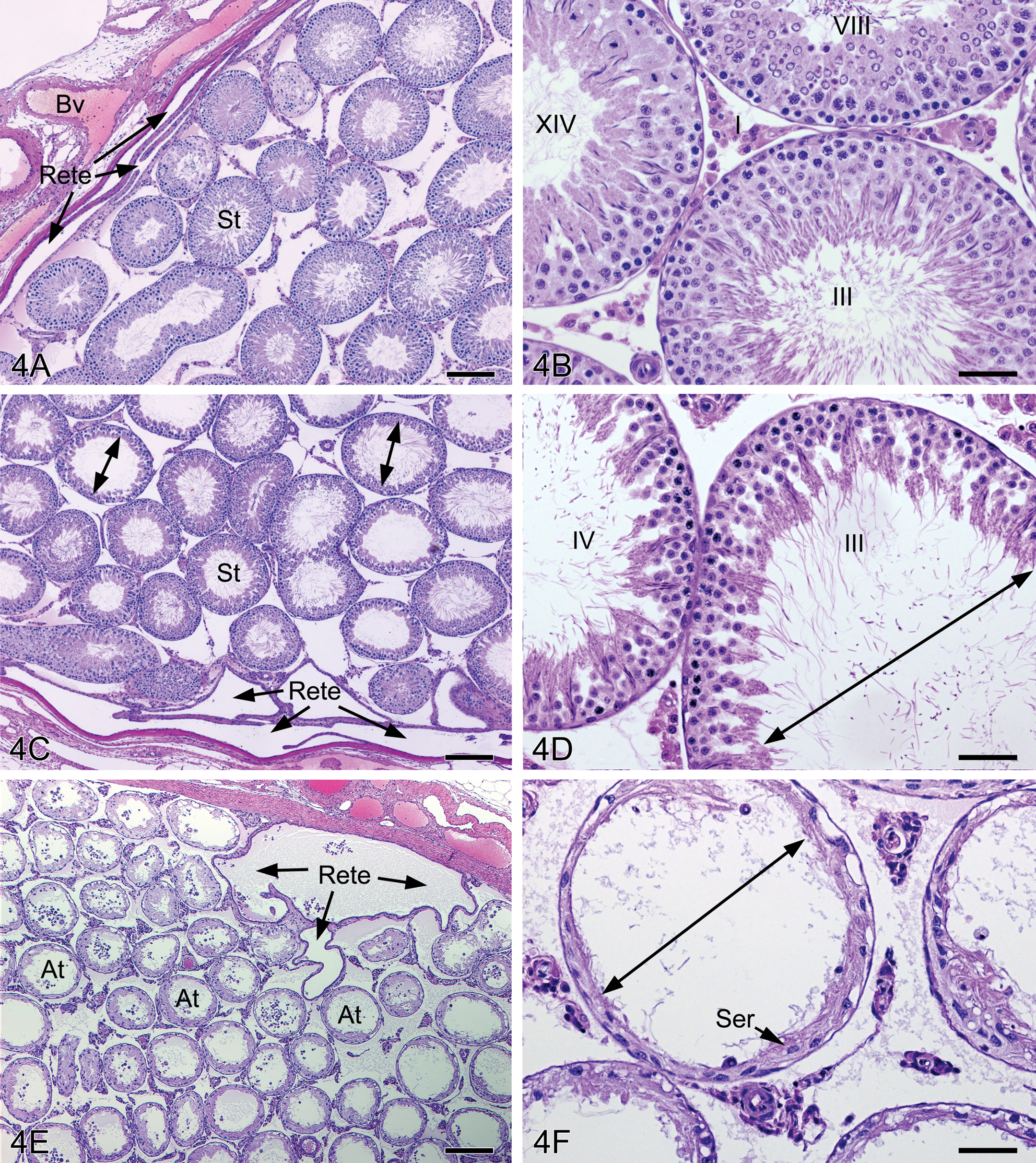
(A) Control testis at the rete testis junction. The rete is typically flattened cavernous spaces adjacent to thickened connective tissue and branches of the blood vasculature (Bv); seminiferous tubule (St). Bar = 200 µm. (B) Normal spermatogenesis in the control testis (stages III, VIII, XIV) shows a thick seminiferous epithelium. I = interstitium. Bar = 50 µm. (C) High-dose group testis at the rete testis junction showing dilated rete and diffusely dilated seminiferous tubules. Bar = 200 µm. (D) Seminiferous tubular dilation in which the seminiferous epithelium is thinned but contains normal spermatogenesis surrounding tubular lumens that are diffusely dilated. Bar = 50 µm. (E) Rete testis region from high-dose group animal showing dilated rete and numerous cross sections of atrophic seminiferous tubules (At). Bar = 200 µm. (F) Atrophic seminiferous tubules from high-dose group animal showing dilated lumens (double arrow) and a seminiferous epithelium lined by Sertoli cells (Ser) and rare spermatogonia. Bar = 50 µm.
Epididymides
Slight to marked ductal atrophy, moderate to marked reduction of sperm, and slight luminal cellular debris were present in the epididymides attached to those testes showing severe tubular atrophy, but sperm content was normal in the epididymides attached to testes with tubular dilation. This suggests that although granulomatous inflammation was present in the efferent ducts, they were still sufficiently patent to be able to transport sperm into the epididymis.
Incidental findings
A low incidence and low severity of granulomatous inflammation also was observed in efferent ducts of the vehicle and 10 and 50 mg/kg/d groups; these changes were not associated with any testicular effects and were considered incidental. Bilateral, moderate degeneration of the seminiferous tubules was observed for one male in the 250 mg/kg/d group, which was characterized by segmental degeneration of the germ cells accompanied by spermatid retention. Since a similar lesion was not observed for any other animals of this group, the lesion was considered an incidental finding unrelated to the test article.
Histomorphologic Changes: 6-Month Terminal Phase
The terminal phase analysis included macroscopic and microscopic findings from three unscheduled decedents in the high-dose group (found dead on days 128, 159, and 168) in addition to those of scheduled sacrifice animals following 6 months of dosing. Test article–related macroscopic changes included one high-dose animal with a unilateral small soft testis and a small corresponding epididymis and another with bilateral soft testes. The small testes correlated microscopically with tubular atrophy/degeneration and the small epididymis with reduced sperm content.
Efferent ducts
Unilateral or bilateral granulomatous inflammation of the efferent ducts was present in 10 of 20 males in the 150 mg/kg/d group (Table 3). The severity of the granulomatous inflammation ranged from minimal to marked. Other findings in this group included minimal to slight dilation of the ductular lumen and/or minimal vacuolar degeneration of the epithelium lining the proximal efferent ducts.
Incidence of histomorphologic changes in the testes and efferent ducts at the terminal (6-Month) necropsy.
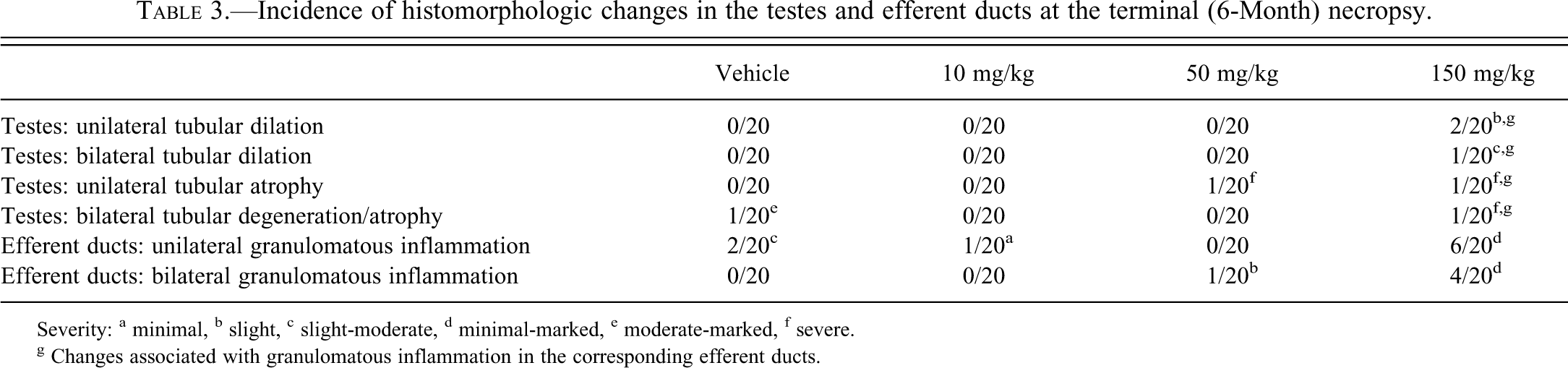
Severity: a minimal, b slight, c slight-moderate, d minimal-marked, e moderate-marked, f severe.
g Changes associated with granulomatous inflammation in the corresponding efferent ducts.
Testes
LTI-1–related changes in the testes from the high-dose group included one rat with severe tubular atrophy in one testis and severe degeneration in the other testis. Another rat had severe atrophy in one testis and slight tubular dilation in the other testis. Two additional rats had tubular dilation: one was unilateral and the other bilateral. The characteristics of the tubular atrophy and tubular dilation were similar to those seen at 250 mg/kg/d after 3 months. In the testis with severe tubular degeneration, most of the germ cells formed multinucleate syncytial cells, which were being sloughed into the lumen. All rats with testicular lesions in this dose group had granulomatous inflammation of the corresponding efferent ducts, but as with the interim time point, not all rats with efferent duct lesions had testicular changes.
Epididymides
Sloughed germ cells and cellular debris as well as reduced sperm content were present in the epididymides associated with degenerate or atrophic testes. There was no cell debris or reduced sperm in epididymides associated with testes that had tubular dilation.
Incidental findings
Granulomatous inflammation also was observed occasionally as an incidental lesion in the efferent ducts of vehicle and 10 and 50 mg/kg/d group males. One rat from the vehicle group had bilateral moderate to severe segmental tubular degeneration in the testis, and one rat from the 50 mg/kg/d group had unilateral severe tubular atrophy; neither was associated with any granulomatous change in the corresponding efferent ducts, and both were considered incidental findings.
Recovery Phase Analysis
After a 3-month recovery period, changes were still present in the testes, efferent ducts, and epididymides of rats administered 150 mg/kg/d. Testicular lesions were present in two of five males. Tubular dilation observed in one rat was associated with granulomatous inflammation in the corresponding efferent duct. Unilateral severe tubular atrophy was observed in one rat. Insufficient tissue was available to evaluate the corresponding efferent ducts for this animal, but the corresponding epididymis had reduced sperm in the caput.
Discussion
In this study, dosing with LTI-1 at 150 mg/kg/d for 6 months or 250 mg/kg/d for 3 months to rats was shown to cause testicular changes (tubular dilation and tubular atrophy) that were predominantly unilateral in distribution. With the exception of one incidental testicular finding in the 250 mg/kg/d group, all other testicular lesions in these two dose groups, whether unilateral or bilateral, were associated with granulomatous inflammation in the corresponding efferent ducts. The high incidence of efferent duct inflammation and the strong correlation between the occurrence of efferent duct inflammation with all of the testicular lesions in these groups suggest that the efferent ducts are the earliest and primary site of toxicity and the testicular lesions are secondary.
Efferent ducts in rats comprise a series of fine ductules connecting the testis to the epididymis. Approximately 90% of seminiferous tubular fluid secreted in the testis is reabsorbed in the efferent ducts during the transport of sperm to the epididymis (Clulow et al. 1998). Disruption of this resorptive function has been shown to result in seminiferous tubular dilation and severe tubular atrophy (Hess 1998; Creasy 2001; Piner et al. 2002). Dilation and swelling of the testis appear to be early responses of the seminiferous tubules to increased fluid volume, which progress to tubular degeneration and atrophy following prolonged backpressure. This progression of testicular changes has been demonstrated experimentally by obstruction of efferent ductules (Harrison 1953; Smith 1962; Cooper and Jackson 1972; Kuwahara 1976; Hess et al. 1991; Nakai et al. 1992). Inhibition of fluid reabsorption in efferent ducts of estrogen receptor-α knockout mice also leads to dilation of seminiferous tubules and subsequent tubular atrophy (Hess et al. 1997; Hess 2003). There are few examples of chemically induced efferent duct toxicity. Benomyl and its metabolite carbendazim have been the most characterized of efferent duct toxicants and were shown to induce testicular changes consistent with impaired fluid reabsorption (Hess et al. 1991; Nakai et al. 1992; Gotoh et al. 1999; Hess and Nakai 2000). Tubular dilation in response to disturbances in fluid reabsorption by the rete and efferent ducts also has been described following administration of a single dose of a 5HT1 agonist (Piner et al. 2002). In this case, the tubular dilation seen at 12 to 24 hours after dosing was preceded by dilation of the efferent ducts and rete, with the earliest changes (at 4 hours postdosing) being vasoconstriction of the veins in the mediastinal plexus overlying the proximal efferent ducts. The change in blood flow was considered the main cause of the inhibited fluid reabsorption in this example.
Although the cause of the fluid disturbances with LTI-1 is unknown, the nature of the changes in the efferent ducts and testes are consistent with a primary effect on the efferent ducts and secondary changes in the testes. These changes do not appear to be related to the pharmacological target, as they were not observed with other leukotriene A4 hydrolase inhibitors in similar toxicity studies (data not shown). There were no changes in serum FSH, LH, or testosterone concentrations, indicating that the efferent duct and testicular changes were not mediated through or associated with disturbances in the hypothalamic-pituitary-testicular axis.
Granulomatous inflammation in efferent ducts generally occurs due to breakdown of the tight junctions (blood-epithelial barrier) that separate the antigenically foreign sperm from the immunocompetent cells of the host (Creasy 2002). The observed changes in the efferent ducts (epithelial vacuolization, ductal dilation, and granulomatous inflammation) suggest disturbed fluid reabsorption followed by breakdown of the blood-epithelial barrier, resulting in sperm stasis, granulomatous inflammation, and obstruction of the efferent duct lumen. Once sufficient proximal ductules become blocked or the single distal duct becomes blocked, the growing pressure of the fluid and sperm causes tubular dilation, which rapidly progresses to atrophy. Whether or not the testes show an effect, and the severity of that effect, depends on the number of efferent ducts obstructed (Gotoh et al. 1999). Because of the technical difficulties of obtaining a complete section through the entire length of the efferent ducts and the lack of clear morphological distinction among the proximal, conus, and distal regions, it was not always possible to accurately locate where the inflammatory lesions were located. As a general rule, lesions were in the proximal region of the ducts but closer to the conus than the rete testis. The unilaterality of the testicular lesions can be explained when the granulomatous obstruction occurs on only one side of the efferent ducts, but there was no evidence to indicate that the left or right side was more predisposed to these changes. In those testes with tubular atrophy, the corresponding epididymides also showed ductal atrophy and reduced sperm content, which is an expected effect of reduced or absent spermatogenesis. In those testes with tubular dilation, the epididymides contained normal amounts of sperm, suggesting occlusion of only a limited number of efferent ducts. For animals with unilateral lesions, the contralateral tissues were normal.
The NOAEL in this study was considered to be 50 mg/kg/d. Although one male in the 50 mg/kg/d group had severe unilateral tubular atrophy, it was not accompanied by efferent duct inflammation that could be identified and was considered an incidental finding, similar to the marked/moderate tubular degeneration seen in the testes of one rat from the vehicle group. Unilateral and bilateral severe tubular degeneration and atrophy as well as tubular dilation are occasionally seen as background lesions in control Sprague-Dawley rats (Huntingdon Life Sciences historical control data; Lee et al. 1993, Creasy 2012). Since the efferent ducts are rarely taken for examination in these situations, it is unknown whether these background lesions might also involve efferent duct obstruction. The presence of efferent duct granulomatous inflammation in several vehicle group rats confirms that such changes occur in control rats and also suggests that the low incidence of efferent duct changes seen in the 10 and 50 mg/kg/d group rats are probably within the normal background range for this finding. However, historical data for efferent duct lesions are not available because efferent ducts are not routinely collected and examined in toxicity studies. Incidental granulomatous inflammation may be due to sperm accumulation at blind-ending ductules (Foley, Bassily, and Hess 1995; Hess 1998; Hess and Nakai 2000).
There are important species differences in efferent duct anatomy such that rats are more likely to develop testicular effects following efferent duct obstruction than are larger mammals, including humans (Fig. 5). Rats have fewer ductules compared with humans and most other large mammals. Rat efferent ductules take a long and tortuous route through the epididymal fat pad before merging into a single duct that enters the initial segment of the epididymis (Saitoh and Hatakeyama 1990; Yeung et al. 1991; Ilio and Hess 1994). By contrast, efferent ductules in large mammals, including humans, are much shorter and do not merge into a single duct but rather enter the epididymis at several sites. This latter anatomical feature enables the outflow of fluid even if several efferent ductules become occluded or fluid reabsorption is inhibited. Because the rat has fewer and longer efferent ductules that all converge into a single duct, there is much greater likelihood for blockage, backpressure, and buildup of fluid. There also are important species differences in rete testis anatomy, with peripheral organization in rats but mediastinal location in dogs and humans (Dym 1976). This difference affects fluid reabsorption by the rete testis epithelium and sensitivity to fluid backpressure. In dogs and humans, it is difficult to induce total atrophy of the testis even with ligation of the efferent ducts (Kothari et al. 1979), while blockage of the efferent ductules in rats leads to total atrophy (Van Wagenen 1924; Hess et al 1991). Consistent with these differences in efferent duct and rete testis anatomy, there were no testicular changes observed in dog repeat-dose toxicity studies with LTI-1 (data not shown). Humans have a redundant outflow system comparable to dogs and would likely not be at risk for the testicular toxicity described here in rats.
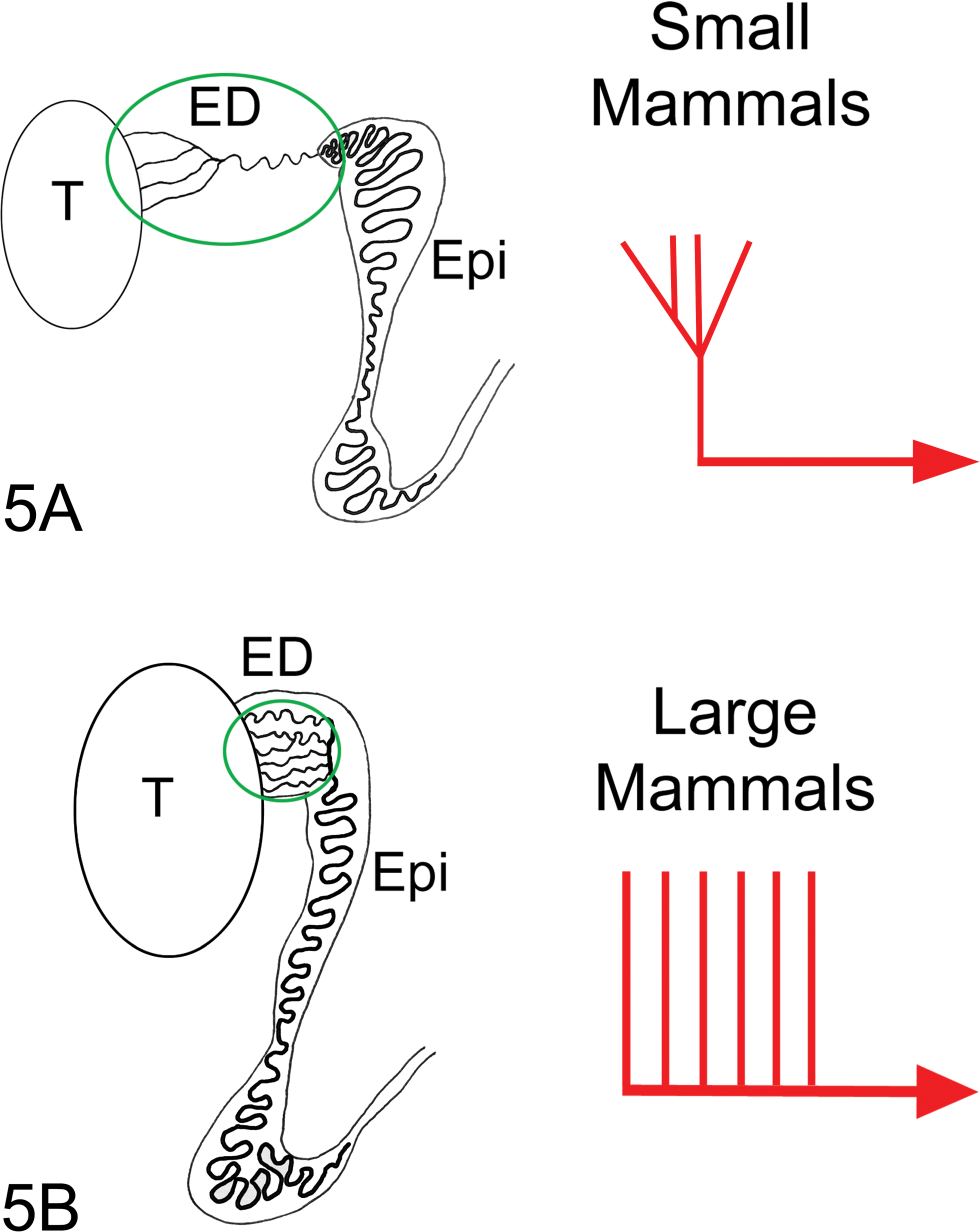
In mammals, there are two basic designs associated with the terminal region of efferent ductules (ED) where they enter the epididymis (Epi). (A) The first design is associated with smaller mammals, including rat, mouse, guinea pig, hamster, and cat, and effectively functions as a funnel in which the proximal efferent ductules merge to form a single, common duct that enters the head of the epididymis (Reid and Cleland 1957; Cooper and Jackson 1972; Guttroff, Cooke, and Hess 1992). (B) The second type of organization involves parallel coils of efferent ductules that form multiple entries into the head of the epididymis, as seen in larger mammals, including man, monkey, dog, goat, pig, sheep, bull, horse, and rooster (MacMillan 1953; Hemeida, Sack, and McEntee 1978; Hoffer and Greenberg 1978; Hess 2002). In larger mammals, some proximal efferent ductules may merge, but there remain numerous independent entrances into the epididymis.
Data from this study support the hypothesis that testicular changes observed in rats administered large doses of LTI-1 occur secondary to obstruction of efferent ducts. It is important to recognize this mechanism of rat testicular injury in toxicity studies since it has limited relevance for human risk assessment because of the differences in efferent duct anatomy that make obstruction of the efferent ducts less likely to occur and the testis less likely to experience pressure changes due to fluid buildup. It is therefore important to distinguish it from a primary drug-induced testicular toxicity. The characteristics that can be used to recognize this secondary mechanism of toxicity are (1) high incidence or dose-related incidence of testes with tubular dilation and testes with severe tubular atrophy, (2) both lesions frequently exhibiting a unilateral distribution, and (3) the severely atrophic tubules often contain a dilated lumen. Efferent duct toxicity is likely underreported because efferent ducts are not routinely collected or examined in toxicity studies. Unfortunately, there is no simple method for the collection and sectioning of efferent ducts from rodents. Because of the thinness and tortuous pathway of the ducts within the epididymal fat pad, dissection is time-consuming and requires practice, and sectioning requires step serial sections to ensure adequate representation of the proximal and distal regions of the ducts. Because it is important to collect and examine the efferent ducts in situations in which testicular injury is suspected to relate to disruption of fluid dynamics, a number of different dissection techniques have been provided in the Methods section of this article to aid the investigator.
Footnotes
Acknowledgments
The authors would like to acknowledge the expert technical assistance of the necropsy and histology staff of Huntingdon Life Sciences in the collection and sectioning of the efferent ducts. Special thanks go to Aleksandra Gondek, Joel Mwangi, Amanda Carolan, Cyndi Pillsbury, Karen Osgood, Diane Vanterpool, and Halina Gondek.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding for this work was provided by Janssen Research & Development.
