Abstract
As a cause of proteinuria in diabetic nephropathy, a decrease in anionic charge on the glomerular basement membrane (GBM) is considered to be related to protein leakage. However, the constancy of the anionic charge has been reported in several types of nephropathy. To elucidate the relation between glomerular protein leakage and anionic charge, we examined the distribution of anionic sites on the GBM and podocytes in diabetic rats induced by a single intravenous injection of 60 mg/kg of streptozotocin (STZ). Five months after the treatment with STZ, urinalysis for glucose and protein levels was conducted, and the kidneys were examined using electron microscopic cytochemistry for the assessment of anionic charge with two cationic probes. The distributions of anionic sites on the GBM demonstrated by two kinds of cationic markers in the diabetic rats were similar in density to those seen in the control animals. The distributions of anionic sites on the foot processes and cell membrane of podocytes were regular and also similar in density to that of the control group. From these results, we consider that the charge barrier of the GBM and podocytes is irrelevant to the protein leakage in diabetic rats.
As a site attributable to protein leakage through the glomerular wall, the glomerular basement membrane (GBM), which possesses an anionic charge consisting mainly of heparan sulfate proteoglycans, has been suggested to play an important role on the permeability of plasma proteins as a charge barrier in the glomerular capillary wall (Kanwar 1984; Kanwar et al. 1991; Raats, Van Den Born, and Berden 2000). The anionic charge of the GBM has been shown to decrease during proteinuria in various kinds of nephropathy (Arai et al. 1992; Barnes et al. 1984; Schneeberger, Stavrakis, and McCarthy 1983; Suzuki et al. 1983; Weening and Rennke 1983). In diabetic nephropathy, many investigators have suggested that the loss of anionic sites on the GBM is closely related to protein leakage in the glomerular wall (Goode et al. 1995; Ina et al. 1991; Isogai et al. 1999; Moriya et al. 1993; Ward-Cook et al. 1999). However, inconsistent results have been reported showing that the anionic charge in the GBM was irrelevant to protein leakage during proteinuria (Kanwar and Jakubowski 1984; Rada and Carlson 1991; Shimoi, Harada, and Watanabe 2000). This discrepancy has been suggested to be due to the differences in the methods applied for the detection of anionic sites (Quatacker 1986). No conclusions have been reached yet on the relation between the alteration of anionic sites in the GBM and protein leakage.
In addition to the GBM, glomerular epithelial cells (podocytes), another fundamental component of the glomerular permselectivity, have also been demonstrated to possess anionic charge, consisting mainly of sialic acids, which also act as a charge barrier (Kerjaschki 1994; Pavenstadt 1998, 2000; Seiler et al. 1977; Weening and Rennke 1983). Several investigators suggested that the decrease in anionic sites of podocytes not only precedes but also plays a greater role in causing proteinuria than that of the GBM (Kerjaschki, Vernillo, and Farquhar 1985; Quatacker 1986; Shimoi, Harada, and Watanabe 2000). Thus, the importance of podocytes in glomerular filtration has become emphasized by the many studies that had been performed on the GBM. However, it is not fully understood whether the alteration of podocyte anionic sites is related to protein leakage in diabetic nephropathy.
To detect anionic sites on the glomerular wall, various kinds of cationic molecules such as polyethyleneimine (PEI), cationized ferritin, and poly-L-lysine-gold (PLG) have been used as markers. The anionic sites demonstrated by PEI distribute only on the GBM. By PLG method, anionic sites in the glomerular wall can be detected both on the cell membrane of podocytes and on the GBM (Russo, Gingras, and Bendayan 1993). Many studies using a cationic marker have reported on anionic charge density on the GBM; however, the comparison of the different methods for the detection of anionic sites is not fully studied.
In this study, we reevaluated the anionic charge density on the GBM by using two kinds of markers, PLG and PEI, in rats with streptozotocin-induced diabetic nephropathy. In addition, we also examined the distribution density of anionic sites on the podocytes with respect to the relation between podocytes and proteinuria in diabetic nephropathy.
Materials and Methods
Animals
Five-week-old female Wistar rats, weighing 125 to 140 g, were purchased from Charles River Laboratories Japan, Inc. (Atsugi, Kanagawa, Japan). The animals were maintained with a 12-hr light-dark cycle at 23°C, 55% humidity. The rats had free access to a standard laboratory diet and tap water. The animal care and experimental procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals (1996) by the Institute for Laboratory Animal Research.
Induction of Diabetes in Rats
After the 7-day quarantine period, four rats were treated with a single intravenous administration of 60 mg/kg of streptozotocin (STZ; Sigma, St. Louis, MO) after an overnight fasting. STZ was dissolved in 10 mM citrate buffer solution (pH 4.5) and immediately injected into the tail vein. Control rats were injected with citrate buffer alone.
Urinalysis
Urine samples were collected for 24 hr from animals in individual metabolic cages, and the total urine volume was measured. Glucose levels were determined with Ames reagent strips (Multistix, Bayer Medica Ltd., Tokyo, Japan). Protein levels were measured by the pyrogallol red method.
Tissue Preparation for Immunocytochemistry
Five months after the STZ injection, rats were anesthetized by intraperitoneal administration of sodium pentobarbital (50 mg/kg). After opening the abdomen, the left renal artery and vein were clamped. A small amount (0.15–0.2 mL) of fixative consisting of 0.1% glutaraldehyde and 2% paraformaldehyde in 0.15 M cacodylate buffer solution (pH 7.4) was injected directly into the cortex of the left kidney with a thin needle (26G). After leaving it for about 30 s, the kidney was removed, and a discolored portion by the fixation was excised out with a razor blade. Small blocks from the cortical tissue were cut and immersed in the fixative for 1 hr. Then the blocks were washed with the cacodylate buffer and immersed in 50 mM NH4Cl in the cacodylate buffer for 1 hr to remove the free aldehyde group. After washing with the buffer, the blocks were dehydrated through graded ethanol at –20°C and embedded in LR-White (London Resin Co., UK) at the same temperature. The resin was polymerized by UV light radiation for the first 24 hr at –20°C and then for another 24 hr at room temperature in a box filled with nitrogen gas.
Tissue Preparation for Conventional Electron Microscopy
For conventional electron microscopy, several pieces of the right kidney were fixed with 2% glutaraldehyde and 1% osmium tetroxide solutions. The blocks were dehydrated through ethanol and embedded in Epon. The sections were stained with uranyl acetate and lead citrate.
Tissue Preparation for PEI Staining
PEI (MW 45,000; Sigma) was used as a cationic probe for staining anionic sites of the GBM. A dose of 0.08 mL/100 g body weight of 0.5% PEI in saline adjusted to pH 7.4 with 1 N HCl and to 400 mOsm with sucrose was injected into the jugular vein under anesthesia with sodium pentobarbital. After 10 min, the animals were euthanized by exsanguination, and the right kidney was removed. Small blocks from cortical tissue were immersed in a fixative containing 2% phosphotungstic acid and 0.1% glutarldehyde in 0.1 M cacodylate buffer (pH 7.4) for 15 min. Then, the blocks were fixed in a fixative of 2% phosphotungstic acid, 1% glutarldehyde, and 0.1 M cacodylate buffer for 3 hr. After washing overnight in 0.1 M cacodylate buffer containing 7% sucrose, the blocks were postfixed with 1% osmium tetraoxide solutions, dehydrated through ethanol, and embedded in Epon.
Labeling with PLG
Cationic poly-L-lysine conjugated with colloidal gold (15 nm, PLG) was purchased from British BioCell (UK). The reaction method with PLG was described previously (Shimoi, Harada, and Watanabe 2000). Briefly, thin sections mounted on nickel grids were immersed in 1% bovine serum albumin (BSA) in 0.01 M phosphate buffered saline (PBS) for blocking nonspecific staining. The sections were then incubated overnight with PLG complex diluted 1/50 in 0.01 M phosphate-HCl-glycine buffer (pH 3.3) containing 0.2% BSA. After washing with PBS and then with water, the sections were counterstained with uranyl acetate and lead citrate.
Digestion with Neuraminidase and Heparitinase
Before the incubation with PLG complex, thin sections were treated with neuraminidase or heparitinase for 1 hr at 37°C. Neuraminidase (Sigma, Germany) and heparitinase (Kanto Chemical Co., Inc., Tokyo, Japan) were adjusted to be 5 mU/µL and 2 mU/µL in 0.1 M phosphate-K buffer of pH 6.5 and 7.0, respectively.
Immunostaining for Proteins
Thin sections mounted on nickel grids were incubated overnight at 4°C with the first antibodies, rabbit anti-rat albumin (diluted 1:500; Biogenesis, UK), and then with colloidal gold (15 nm)–conjugated goat anti-rabbit IgG (EY Lab, San Mateo, CA) diluted 1:20 in 0.05 M Tris-HCl buffer containing 0.1% Tween 20 for 60 min at room temperature. After washing with PBS and then with water, the sections were counterstained with uranyl acetate and lead citrate.
Morphometry
At least two photographs from each animal were taken at 10,000×. The labeled anionic sites located within the lamina rare externa were counted over 10,000-nm lengths of the GBM on photographs. The number of anionic sites on the GBM was represented per 1,000-nm lengths. Oblique sections through the GBM were excluded from counting, and straight lengths of the GBM were examined. Since the anionic sites on the lamina densa and lamina rara interna distributed irregularly, counting was not conducted in these areas.
In the PLG method, gold particles on the GBM were counted on photographs taken from the sections after the digestion with neuraminidase to exclude the labeling on the podocyte cell membrane. The photographs were enlarged at a final magnification of 30,000×. In the PEI method, anionic sites were counted on photographs enlarged at a final magnification of 40,000×.
Statistical Analysis
Data were expressed as mean ± SD. Differences were statistically analyzed by Student’s t-test. Statistical evaluation was conducted at a level of p < 0.05.
Results
Urinalysis
In the diabetic rats, the urinary volume and glucose level were significantly elevated compared with the control group. The urinary total protein level in the diabetic rats was about four times higher than that in the control animals (Table 1).
Urinalysis in rats 5 months after induction of diabetes.
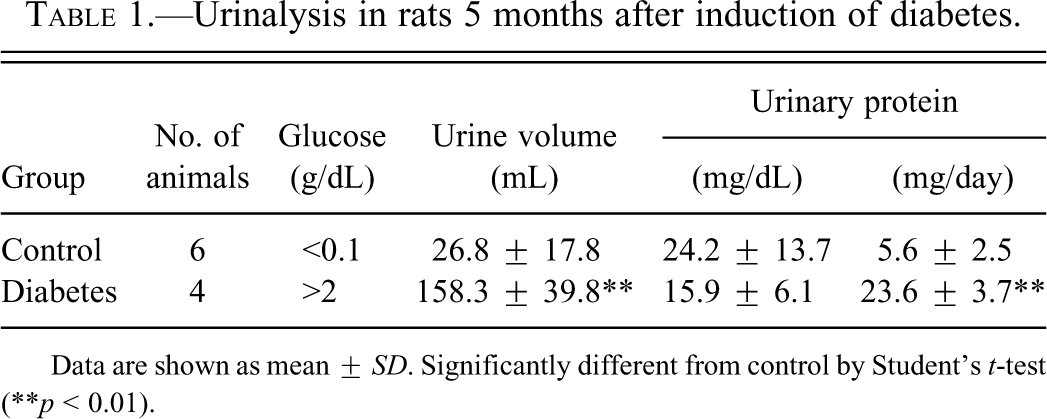
Data are shown as mean ± SD. Significantly different from control by Student’s t-test (**p < 0.01).
Conventional Electron Microscopy
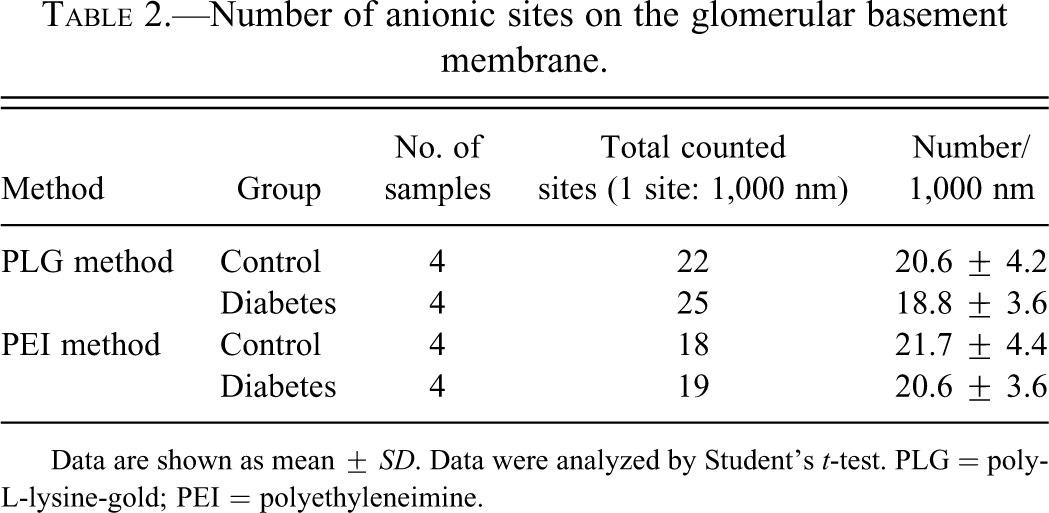
In the diabetic rats, the glomeruli showed apparent flattening of foot processes of podocytes (Fig. 1b).
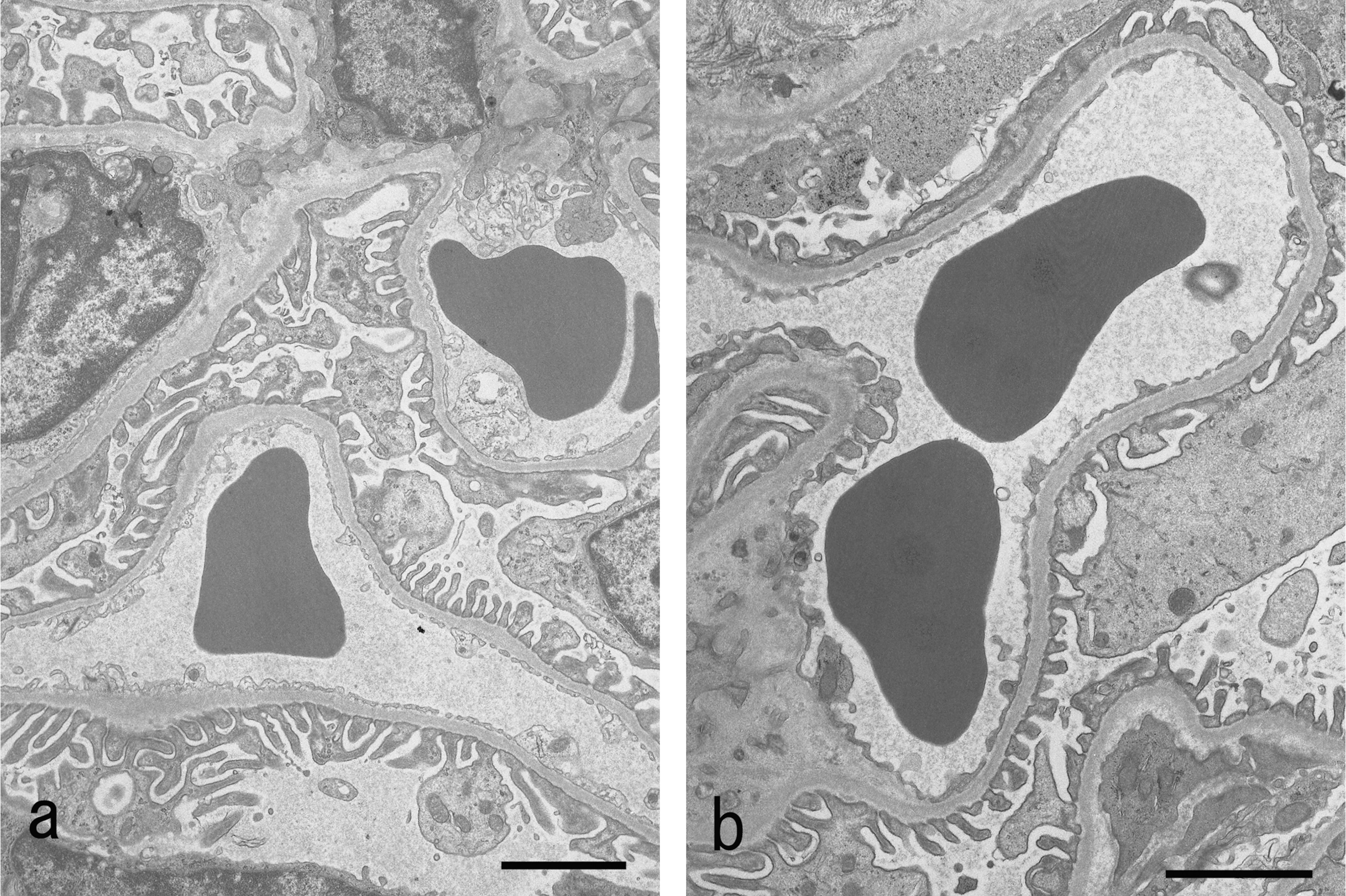
Transmission electron micrograph of the glomeruli from a control rat (a) and from a diabetic rat induced by streptozotocin (b). Flattening of foot processes is seen in the podocytes. Bar = 2 µm.
Anionic Sites Distribution by PLG
In the control rats, the distribution of gold particles detected by the PLG reaction method at pH 3.3 in the glomerular wall showed highly dense distribution both in the podocytes and the GBM (Fig. 2a). In the podocytes, the gold particles were distributed on the surface of the cell membrane of the cell body and foot processes. The gold particles in the podocytes were not seen when the sections were treated with neuraminidase, which digests sialic acid (Fig. 3a), but they remained after the treatment with heparitinase, which digests heparan sulfate. In the GBM, the gold particles were distributed diffusely with a higher density in the lamina rara externa. This distribution was lost by the treatment with heparitinase but remained by the treatment with neuraminidase (photo not shown).
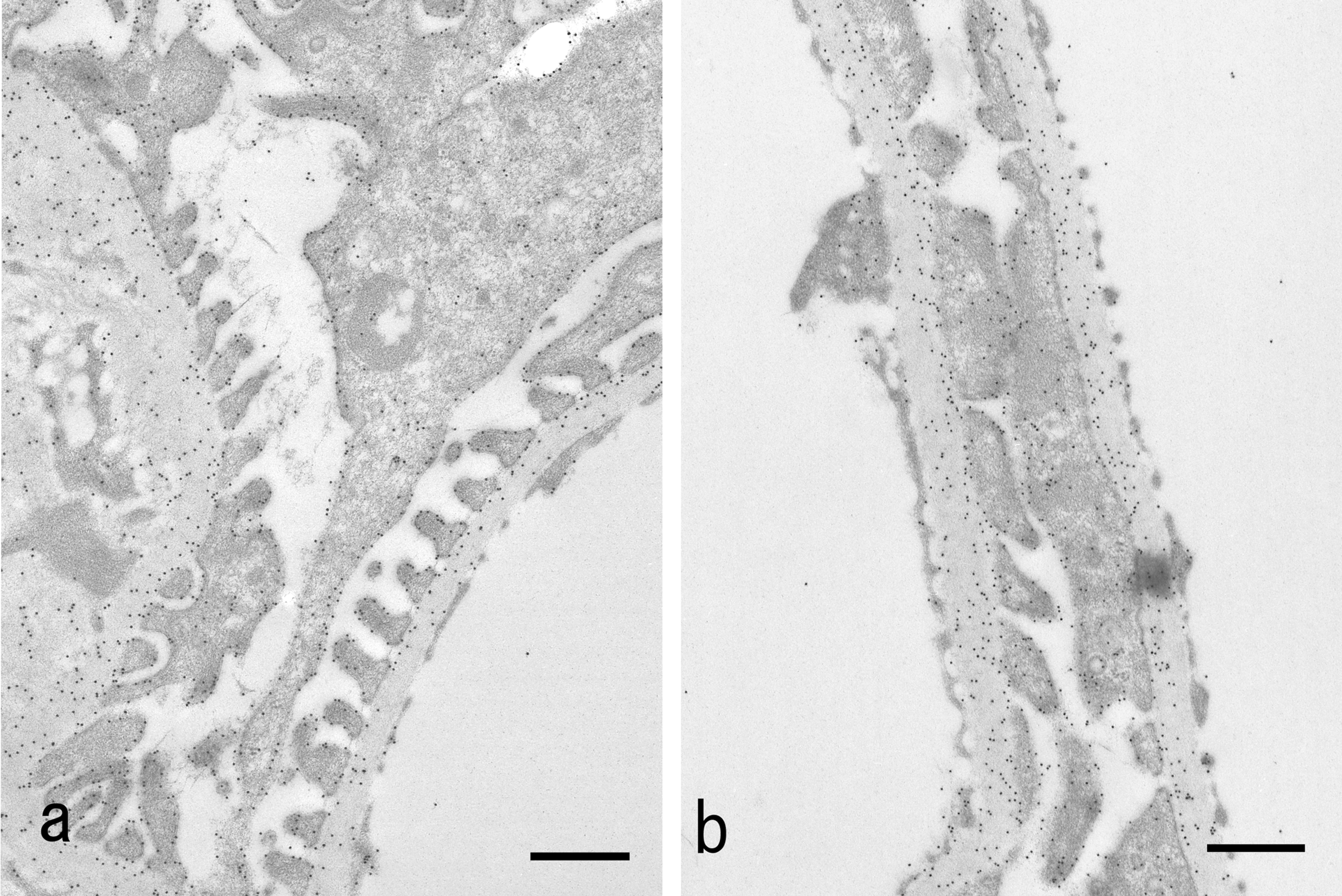
Transmission electron micrographs of the glomerulus treated with poly-L-lysine cationic colloidal gold probe for anionic sites. Incubated with poly-L-lysine-gold (PLG) at pH 3.3 for 16 hr at 4°C. (a) Control rat. Gold particles (15 nm) are present on the cell membrane of podocytes and on the basement membrane. (b) Diabetic rat. Gold particles are distributed in a similar pattern to the control rat. Bar = 0.5 µm.
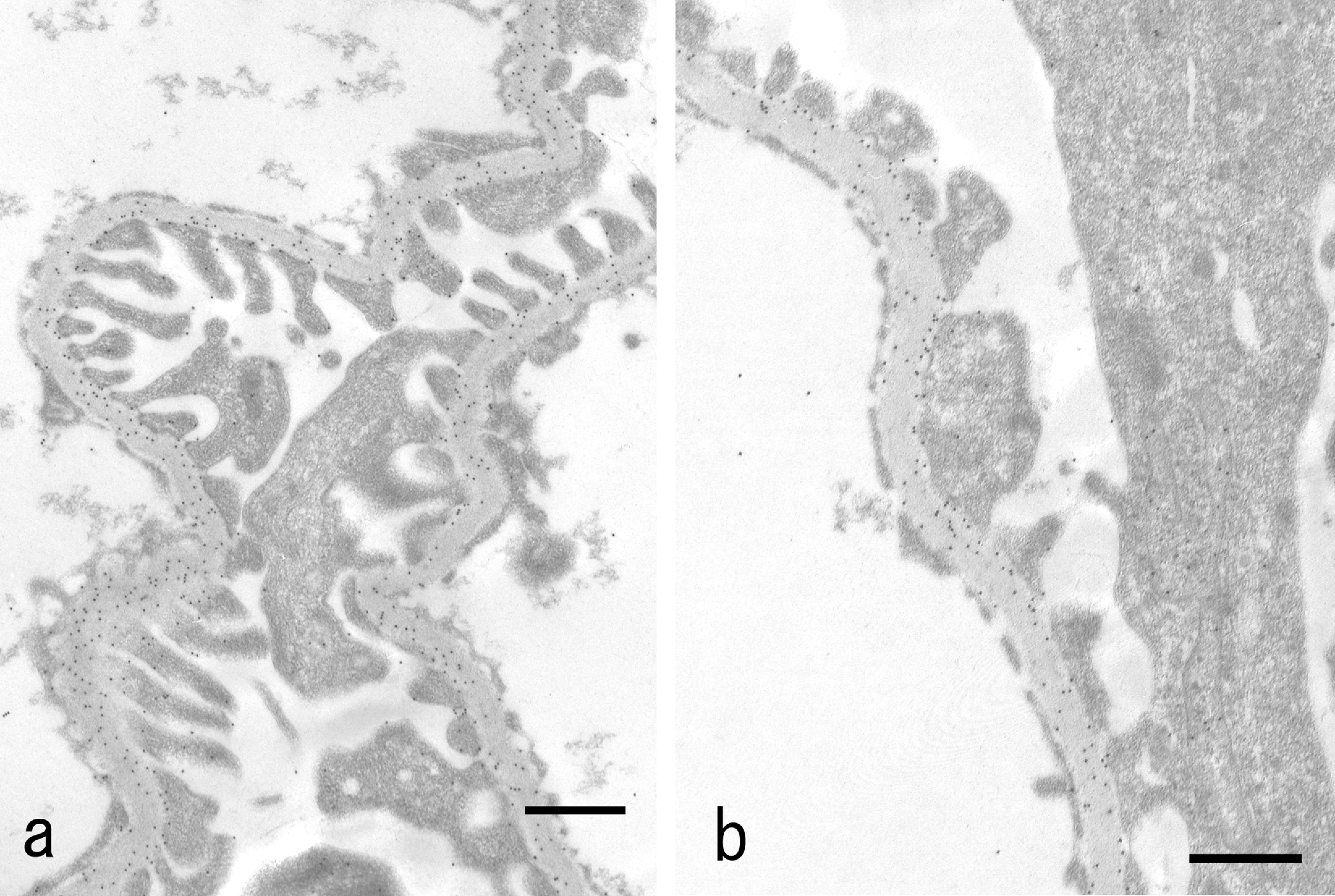
Transmission electron micrographs of glomerulus treated with poly-L-lysine cationic colloidal gold probe for anionic sites after digestion by neuraminidase. (a) Control rat. Gold particles (15 nm) are present on the basement membrane. (b) Diabetic rat. Gold particles are distributed in a similar pattern to that of the control rat. Bar = 0.5 µm.
In the diabetic rats, the distributions of PLG in the foot processes and cell bodies of podocytes, including the portion of flattening of foot processes, were regular and similar in density to those of the control animals (Fig. 2b). In the GBM, the distribution of PLG particles after the digestion by neuraminidase was similar in density to that seen in the control group (Fig. 3b). There were no statistically significant differences in the number of PLG particles per 1,000 nm in the GBM between the diabetic and control animals (Table 2).
Number of anionic sites on the glomerular basement membrane.
Data are shown as mean ± SD. Data were analyzed by Student’s t-test. PLG = poly-L-lysine-gold; PEI = polyethyleneimine.
Anionic Site Distribution by the PEI Method in the GBM
In the control animals, anionic sites were distributed in a regular linear pattern in lamina rara externa of the GBM (Fig. 4a). In the diabetic rats, the distribution density of anionic sites was similar to that of control animals (Fig. 4b). The number of anionic sites per 1,000 nm in the GBM was not significantly different from that in the control group (Table 2).
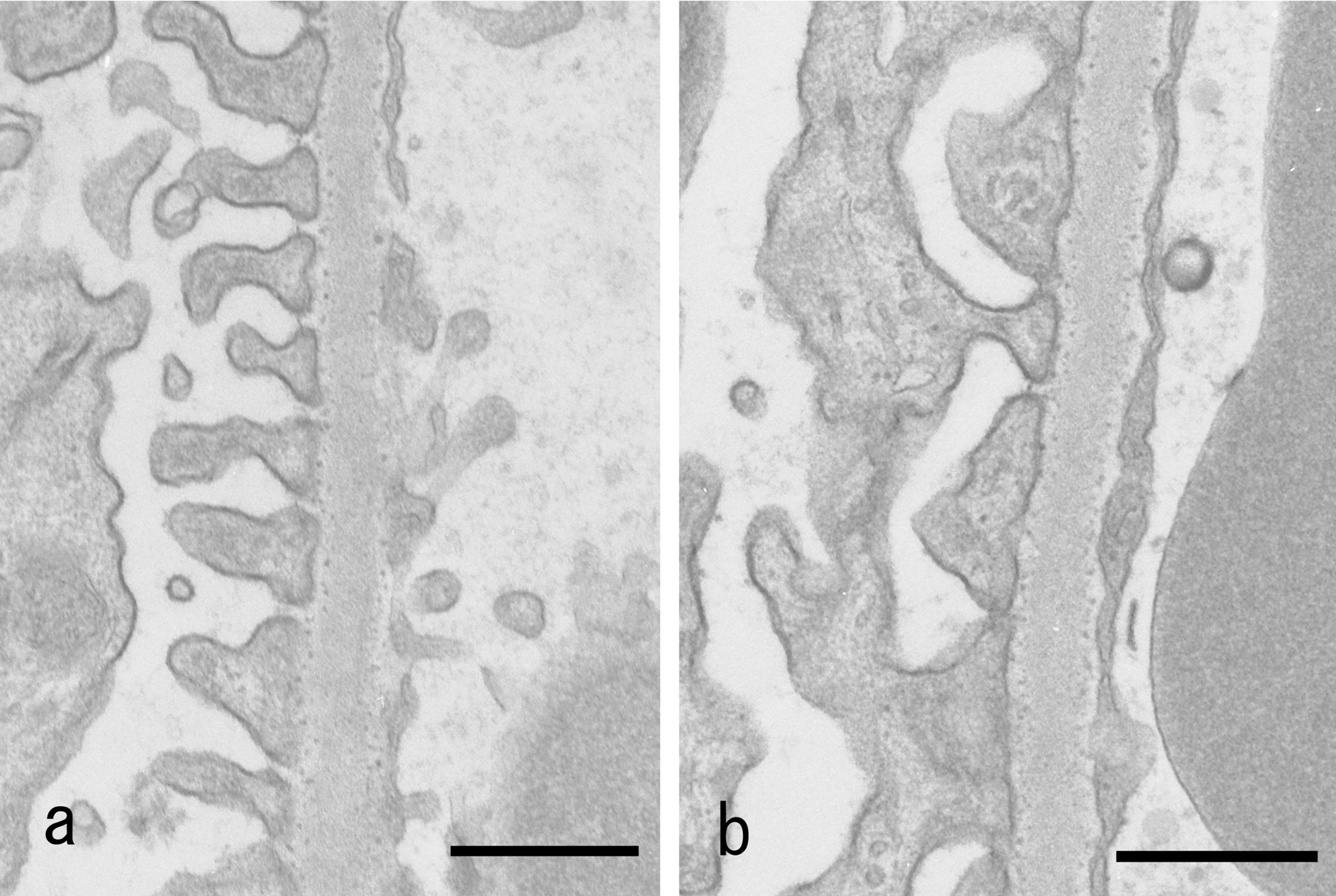
Transmission electron micrographs of glomerulus treated with polyethyleneimine for anionic sites. In the control animal, anionic sites are distributed in a regular linear pattern in lamina rara externa of the GBM. (a) In the diabetic rats, the distribution density of anionic sites was similar to that of the control animals. Bar = 0.5 µm.
Immunocytochemistry of Plasma Proteins
In the control animals, the gold particles demonstrating albumin were distributed densely on the homogenous substance in the capillary luminae and on the GBM. Not a few gold particles were seen in the cytoplasm of major foot processes as well, and only a few particles were distributed on the intact foot processes (Fig. 5a). In the diabetic rats, the distribution of gold particles on the GBM and podocytes is similar to that of the control animals (Fig. 5b).
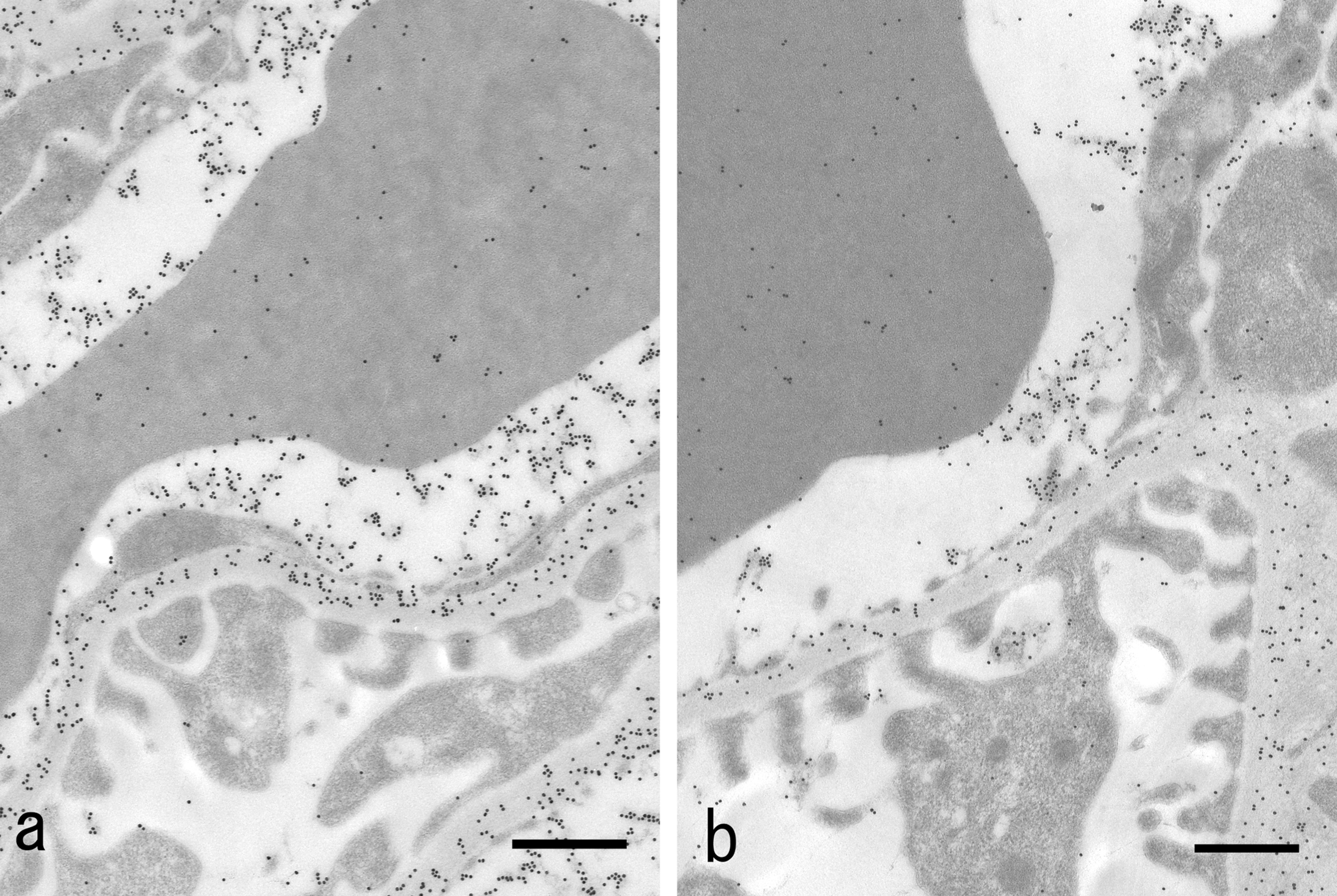
Electron microscopic immunocytochemistry with anti-rat IgG. In the control animals, the gold particles are distributed densely on the homogenous substance in the capillary luminae and on the glomerular basement membrane (GBM). Not a few gold particles were seen in the cytoplasm of major foot processes as well, and only a few particles were distributed on the intact foot processes. (a) In the diabetic rats, the distribution of gold particles on the GBM and podocytes is similar to that of the control animals. Bar = 0.5 µm.
Discussion
Not a few reports have indicated that protein leakage through the glomerular wall is related to the decrease in anionic charge density of the GBM in several kinds of nephropathy, including diabetic nephropathy (Goode et al. 1995; Ina et al. 1991; Moriya et al. 1993; Van Den Born et al. 1995). However, inconsistent results were obtained from other investigators (Kanwar and Jakubowski 1984; Rada and Carlson 1991). We examined the distribution of the anionic charge density on the GBM and podocytes in relation to protein leakage in rats with diabetic nephropathy induced by STZ administration using two kinds of markers (PLG and PEI). The anionic sites on the GBM and podocytes were distributed regularly in the diabetic rats, and no differences were observed from the control animals. From these findings, the anionic charge on the GBM is considered to be irrelevant to the protein leakage in diabetic rats. This consideration is supported by our previous experiment demonstrating that no changes in the anionic charge density on the GBM were observed in the puromycin-induced nephritic rats, which showed more severe proteinuria than the animals in this study (Shimoi, Harada, and Watanabe 2000).
The flattening of foot processes of podocytes has been discussed in relation to protein leakage in several types of nephropathy (Pavenstadt 1998, 2000; Ryan and Karnovsky 1975). In this study, the flattening of foot processes was also observed in the diabetic rats, which may suggest close relation to proteinuria. This finding is consistent with the reports that demonstrated the decrease in the flattening of foot processes and the amelioration of proteinuria in diabetic rats treated with antidiabetic drugs (Mifsud et al. 2001). However, it is hard to conceive how this morphological change plays a role in protein leakage. Many ultrastructural studies have suggested that the detachment of foot processes from the GBM is the cause of protein leakage in proteinuria (Ryan and Karnovsky 1975; Whiteside et al. 1989). However, no detachment of foot processes was observed in this study. Therefore, we consider that the detachment of foot processes is not the trigger to induce protein leakage in diabetic nephropathy. Protein leakage without detachment of foot process has been demonstrated in mice treated with Vibrio cholerae sialidase (Gelberg et al. 1996).
In summary, we consider protein leakage on the glomerular wall to be not caused by the decrease in anionic charge density on the GBM in diabetic rats induced by STZ. In the examination of albumin distribution by immunological technique, no differences in the albumin quantity in the glomerular wall were observed between the diabetic rats and the control animals. This result may indicate that the amounts of albumin that existed in the glomerular wall, the GBM, and podocytes were normally constant even in the diabetic animals, but the passage through the glomerular wall was increased. As the sites of passage, the pore in the GBM and slit diaphragm between foot processes are considered. The change in pore size of the GBM in relation to the protein leakage has been demonstrated in diabetic nephropathy (Friedman et al. 1983; Isogai et al. 1999; Ota, Shikata, and Ota 1994). The slit diaphragm also has been indicated to be responsible for the protein leakage in various glomerular lesions. Further studies are necessary to elucidate the exact site(s) of protein leakage through the glomerulus.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
