Abstract
The present article describes an unusual proliferative islet finding observed incidentally in a young male Wistar rat in a 2-week toxicity study. Histologically, the islet lesion was characterized by diffuse enlargement of the islets, which consisted of peripheral proliferation of non-insulin-containing islet cells surrounding normal-appearing insulin-containing cells in the center. To the authors’ knowledge, this is the first report of spontaneous proliferative islet lesion composed of non-insulin-containing cells in young rats.
Keywords
Islet cell hyperplasias and adenomas of the pancreas are relatively common age-related spontaneous findings in the rat, while their malignant counterparts occur with low frequency (Germann et al. 1999). None of the spontaneous proliferative islet lesions commonly occur in rats younger than 8 months (Kroes et al. 1981; Son et al. 2010). The proliferative lesions, including hyperplasia, adenoma, and carcinoma, of the pancreatic islet in rats are predominantly composed of insulin-containing cells across strains (Germann et al. 1999; Stromberg, Wilson, and Capen 1983; Spencer, Andreu, and Greaves 1986) and are characterized by gradual transitions in cytology and tissue architecture. These lesions can be distinguished on the basis of growth size, growth pattern, and cellular pleomorphism (Riley, Boorman, and Hayashi 1990). Briefly, the hyperplastic islet can attain up to 500 µm in diameter and consist of a normal arrangement of normal to hypertrophic cells with peripheral distribution of alpha and delta cells. Islet cell adenomas are typically well circumscribed with a single expansile mass, which ranges from 1 to 10 mm in diameter and compresses adjacent acini. The adenomas may consist of solid clusters of cells and prominent cords or ribbons rather than the usual small nests, with occasional alpha and delta cells scattered throughout the mass. The neoplastic cells are usually uniform and normal appearing or enlarged. Islet cell carcinomas are generally distinguished from their benign counterparts by a prominent heterogenous growth pattern, marked cellular pleomorphism, local invasion, and/or metastases. We report here an unusual proliferative islet finding observed incidentally in a young male Wistar rat in a 2-week toxicity study.
The animal was a male Wistar Han IGS rat in a vehicle control group for a 14-day toxicity study. A group of Wistar Han IGS rats (5 males and 5 females), which was obtained from Charles River Laboratories when the rats were 10 to 12 weeks of age, was assigned to a vehicle control group for the 14-day toxicity study. These rats received 20.5% (w/v) hydroxypropyl β-cyclodextrin daily by subcutaneous administration for 14 days. The animal was observed daily for clinical signs, and body weight and food consumption were recorded weekly. Hematology and serum chemistry (alanine aminotransferase, albumin, albumin-globulin ratio, alkaline phosphatase, aspartate aminotransferase, blood urea nitrogen, calcium, chloride, insulin, cholesterol, creatinine, gamma-glutamyltransferase, globulin, glucose, lactate, phosphorus, potassium, sodium, total bilirubin, total protein, and triglycerides) measurements were conducted at the end of the study. A complete necropsy was performed, and a comprehensive set of tissues was collected in 10% neutral buffered formalin, 3% glutaraldehyde, or Modified Davidson’s solution. All procedures performed on these animals were in accordance with regulations and established guidelines and were reviewed and approved by an Institutional Animal Care and Use Committee or through an ethical review process.
Tissues were trimmed, embedded in paraffin, sectioned at approximately 5 µm, and stained with hematoxylin and eosin (H&E). In addition, sections of the pancreas were stained with Grimelius silver stain. Formalin-fixed, deparaffinized sections of the pancreas were also immunohistochemically stained with guinea pig polyclonal antibody specific for insulin (Dako, Carpinteria, CA), rabbit polyclonal antibody specific for glucagon (Abcam, Cambridge, MA), and rabbit antibody specific for somatostatin (Dako, Carpinteria, CA).
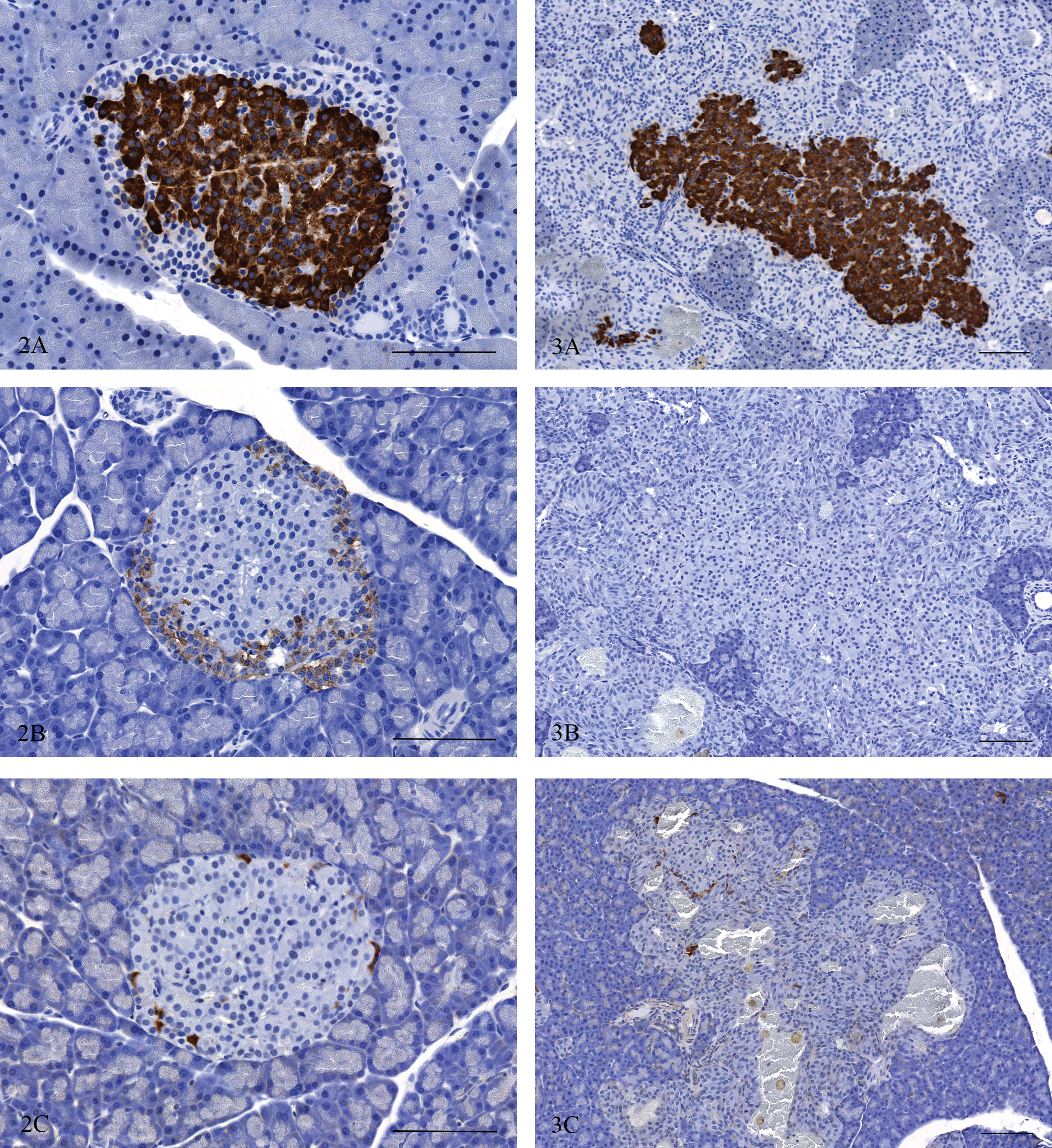
The animal had a lower body weight (231 g) at necropsy compared with those (262 g to 277 g) of the remaining four male rats in the same control group. No significant hematology or serum chemistry findings were observed. No gross pathology observations except for traumatic skin lesions at the injection sites were recorded. Microscopic examination revealed irregularly enlarged islets throughout the section of pancreas (Fig. 1A). The enlarged islets often contained dilated capillaries or blood-filled spaces. Almost all islets were variously enlarged up to 1 mm in the longest axis by peripheral proliferation of the fusiform to elongated cells, which had a moderate amount of slightly basophilic cytoplasm with a slightly enlarged oval nucleus (Fig. 1B). The cells were closely packed in cords or nests with a delicate fibrovascular stroma. Mitotic figures were occasionally noted. Compression of the adjacent pancreatic tissue was not evident. Groups of normal-appearing polyhedral cells were often embedded in the central zone of the enlarged islets, and these were exclusively positive for insulin. However, the proliferating fusiform cells that constitute the irregularly expanded peripheral zones of the islets were almost completely negative for both insulin and glucagon (Fig. 3A, B). A very few somatostatin-positive single cells were scattered in the peripheral zones of the islets (Fig. 3C). These immunohistochemical features were characteristic of the current case, given that islet cell neoplasms and hyperplasia in rats are predominantly composed of insulin-containing cells across strains (Germann et al. 1999; Stromberg, Wilson, and Capen 1983; Spencer, Andreu, and Greaves, 1986). The immunohistochemical findings of the islet from a normal rat pancreas islets (Fig. 2A–C) were consistent with those reported previously (Spencer, Andreu, and Greaves 1986). The proliferating cells in the peripheral zones contained argyrophil granules, which were stained with Grimelius silver procedure (Fig. 4). Argyrophil stain is a useful technique for visualizing cytoplasmic granules of neuroendocrine cells. It also stains the pancreatic polypeptide cells and A (glucagon) cells of the pancreatic islets (Wilander 1989). The silver-impregnated cells, however, were unlikely to be A cells because they were negative for glucagon immunohistochemically. A positive reaction of cytoplasmic granules against Grimelius silver stain could suggest that the proliferating cells possess some peptide hormone or hormone-like agent, but they were not plainly characterized in the current case. Although cellular atypia existed, the current case was diagnosed as hyperplasia based on a limited growth size (up to 1 mm) and a lack of compression of the surrounding tissues on H&E-stained sections. No remarkable changes were noted in other tissues examined, including thyroid, parathyroid, and pituitary gland. Multiple endocrine neoplasia type 1 (MEN1) is a genetically inherited cancer syndrome in humans, characterized by proliferative lesions involving the endocrine pancreas, parathyroid, and anterior pituitary gland. These tumors occur at a younger age. Humans with MEN1 almost always have loss-of-function mutations in the MEN1 gene, and inactivation of a single allele of the MEN1 gene in mice has resulted in a range of pancreatic islet lesions from hyperplasia to tumors as well as parathyroid adenomas by 9 months (Crabtree et al. 2000). The multiplicity of lesion formation across the endocrine organs was not evident in the current case. Although etiology was not determined, some specific genetic modification was suspected to underlie the proliferative islet lesion due to an early occurrence in the life span and a diffuse distribution pattern of the lesion development. To the authors’ knowledge, this is the first report of spontaneous proliferative islet lesion composed of non-insulin-containing cells in young rats.
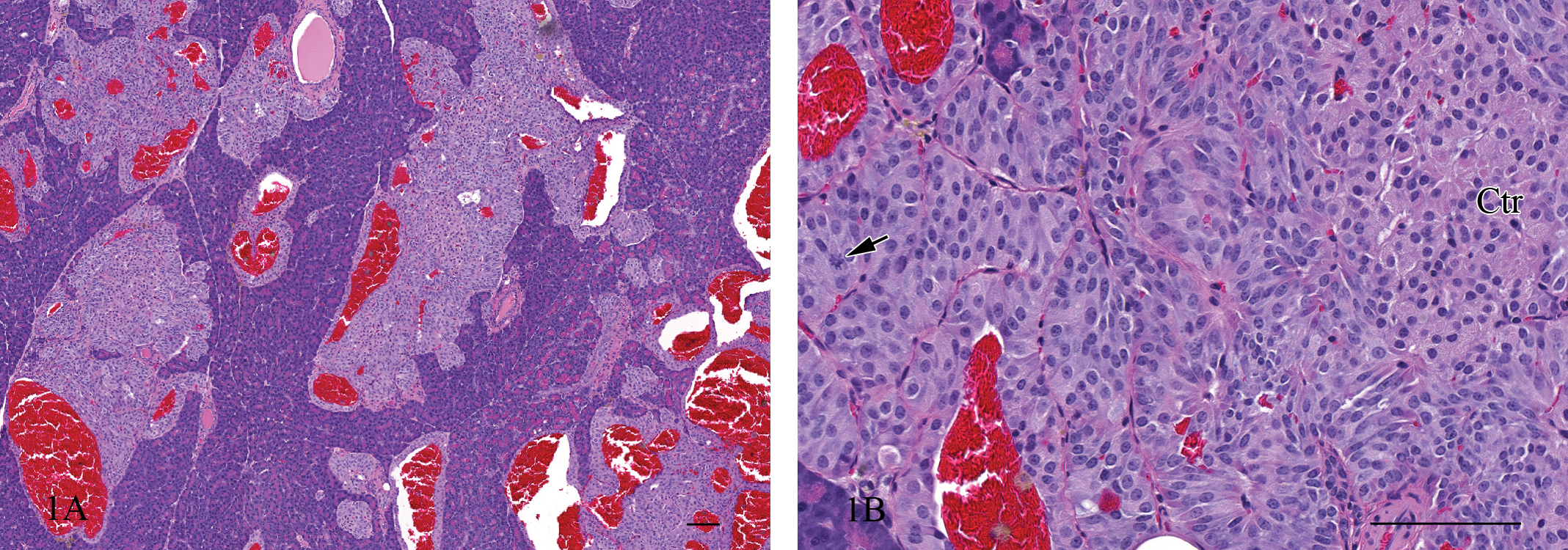
(A) Pancreas, affected rat. Irregularly enlarged islets with dilated capillaries or blood-filled spaces. Hematoxylin and eosin (H&E). Bar = 100 µm. (B) Pancreas, affected rat. Peripheral proliferation of the fusiform cells surrounding a group of normal-appearing polyhedral cells in the center (Ctr). A mitotic figure (arrow). H&E. Bar = 100 µm.
Figure 2. (A) Pancreas, normal rat. Insulin-containing cells and surrounding non-insulin-containing cells in the islet. Immunohistochemistry with hematoxylin counterstain. Bar = 100 µm. (B) Pancreas, normal rat. No glucagon-containing cells present. Immunohistochemistry with hematoxylin counterstain. Bar = 100 µm. (C) Pancreas, normal rat. A few somatostatin-containing cells in the islet. Immunohistochemistry with hematoxylin counterstain. Bar = 100 µm.
Figure 3. (A) Pancreas, affected rat. Insulin-containing cells in the islet. Immunohistochemistry with hematoxylin counterstain. Bar = 100 µm. (B) Pancreas, affected rat. Glucagon-containing cells in the islet. Immunohistochemistry with hematoxylin counterstain. Bar = 100 µm. (C) Pancreas, affected rat. Somatostatin-containing cells in the islet. Immunohistochemistry with hematoxylin counterstain. Bar = 100 µm.
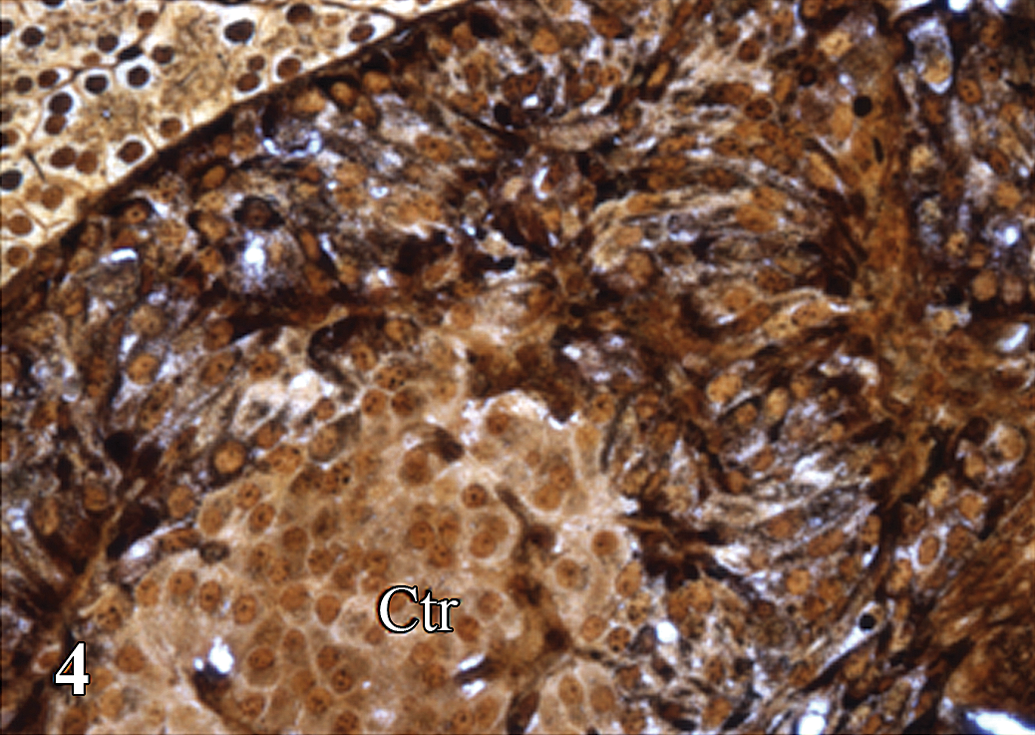
Pancreas, affected rat. The peripherally proliferating cells with cytoplasmic argyrophil granules surrounding a group of normal-appearing polyhedral cells in the center (Ctr). Grimelius silver stain.
Footnotes
Acknowledgment
The authors wish to thank the necropsy and histology laboratory staff at Pfizer (Groton, CT) for technical support.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
