Abstract
As a common additive in cigarette filters, nanosilica has been implemented to reduce the release of harmful substances in cigarette smoke. However, the potential risk of occupational exposure for cigarette factory workers is unknown. We collected physical examination data from 710 cigarette factory workers to evaluate the adverse effects of cigarette filter silica exposure. We also established mouse models induced by cigarette filter silica and crystalline silica separately to compare the lung inflammation, pulmonary function, apoptosis, and fibrosis of the two models. Workers in the rolling and packing workshop exposed to cigarette filter silica had a higher rate of abnormal lung function (17.75%) than those in the cutting workshop (0.87%). Animal experiments showed that compared with the same dose of crystalline silica, cigarette filter silica resulted in higher levels of inflammatory factors in the bronchoalveolar lavage fluid (BALF) of mice at day 7, and lower levels of total lung capacity (TLC), inspiratory capacity (IC), vital capacity (VC), and forced vital capacity (FVC) in mice at day 28. Additionally, both exposed groups of mice showed increased levels of caspase 3, collagen I (Col-Ⅰ), α-smooth muscle actin (α-SMA) and hydroxyproline (HYP) in the lungs, as well as collagen accumulation and fibrous nodules at day 28, with no significant difference between the two groups. The results suggested that cigarette filter silica caused more severe early lung inflammation and late ventilation impairment than the same dose of crystalline silica. In the future, we need to pay more attention to nanosilica protection in cigarette factories to prevent pulmonary dysfunction in workers.
Introduction
Cigarettes have become a daily necessity for many people. However, cigarettes contain a large number of harmful substances, such as nicotine and tar, making them a major health hazard. In order to reduce the harm caused by cigarettes, tobacco factories commonly use filter technology, with nanosilica being a frequently used material. Studies have shown that nanosilica added to cigarette filters could reduce the harmfulness of smoking by adsorbing or catalyzing some of the harmful components in cigarette smoke (Cao et al., 2006; Zeng et al., 2022). Notably, in the rolling and packing workshop where cigarette filters are manufactured, workers may be exposed to nanosilica, thus potentially leading to respiratory diseases. However, there are few studies on the respiratory effects of nanosilica in cigarette filters.
The potential risks of silica nanoparticles have received significant attention in recent years (Huang et al., 2022). Researchers have discovered that silica nanoparticles could enter the body through various routes, including respiratory, digestive, and dermal pathways, and subsequently circulate throughout the body, ultimately leading to harmful health effects (Oberdorster et al., 2005). These nanoparticles have been found to be toxic to multiple organ systems, including the respiratory, cardiovascular, digestive, nervous, reproductive, and immune systems (Huang et al., 2022). Particularly, in the respiratory system of mice, silica nanoparticles were found to cause lung inflammation, as well as apoptosis of lung epithelial cells, leading to structural destruction of alveoli and collagen deposition (Ko et al., 2020; Li et al., 2022). In vitro studies further demonstrated that silica nanoparticles induced apoptosis and cytokine secretion of alveolar macrophages, thus promoting the differentiation of lung fibroblasts (Li et al., 2021). Therefore, we inferred that exposure to cigarette filters silica might have an adverse effect on the respiratory system of cigarette factory workers.
The pulmonary toxicity of crystalline silica has been extensively studied, with findings suggesting that it caused the release of inflammatory factors from alveolar macrophages, leading to inflammation, apoptosis and fibrosis, impairing pulmonary ventilation (Song et al., 2022). However, the long-term effects of nanosilica in cigarette filters on lung health are still unknown. Consequently, it is necessary to investigate the pulmonary toxicity of silica nanoparticles in cigarette filters and to assess the occupational risks associated with exposure to these nanoparticles.
In this study, we collected physical examination data from 710 workers in cigarette factories to evaluate the adverse effects of nanosilica used in cigarette filters. Subsequently, we established two mouse models induced by cigarette filter silica and natural crystalline silica separately. By observing the differences in the inflammatory response, pulmonary function, apoptosis and fibrosis between the models, we aimed to explore the lung toxicity of cigarette filter silica. The findings of this study are significant in identifying the possible pulmonary toxicity of cigarette filter silica and revealing the occupational exposure hazards for workers in cigarette factories.
Materials and methods
Data collection
We collected the occupational health examination data from the Changsha Center for Disease Control and Prevention for 710 staff on duty in cigarette factories in 2021. This examination included 231 operators in the cutting workshop and 479 operators in the rolling and packing workshop.
Pulmonary function examination and criteria for determining abnormalities
Pulmonary function was examined using a spirometer (AS-407, Japan). The examination indexes include Forced Vital Capacity (FVC), Forced Expiratory Volume in the first second (FEV1), and the percentage of FEV1 in FVC (FEV1/FVC), with the first two items taking the percentage of the measured value in the predicted value as the numerical result.
The determination of abnormal pulmonary ventilation function: FVC%<80% or FEV1%<70% or FEV1/FVC%<70%. If any one of the three indexes is lower than the standard value, it is determined as abnormal pulmonary function (Ponce et al., 2022).
Reagents and antibodies
Cigarette filter silica particles (SPY ground silica; size distribution: 20-50 nm, 99.5%) were obtained from the Taihong Shengda New Material Co. Ltd (Caofeidian, Tangshan, China). Natural crystalline silica particles (Min-U-Sil five ground silica; size distribution: <5 μm, 97%) were obtained from the U.S. Silica Company (Frederick, MD, USA).
Cleaved caspase-3 antibody (AC033) was obtained from Beyotime Biotechnology Company (Shanghai, China); Antibodies of Collagen I (Col-Ⅰ) (AF7001) and GAPDH (AF7021) were obtained from Affinity Biosciences (Beijing, China); Antibodies of α-smooth muscle actin (α-SMA) (A17910) and HRP Goat Anti-Rabbit lgG (AS014) were obtained from ABclonal (Wuhan, China).
Animals and treatment
Male C57BL/6J mice, weighing 20-25 g and aged 6-8 weeks, were obtained from the Hunan SJA Animal Co. Ltd (Certificate No. 2019-0004). The animals were housed in rearing cages of dimensions 45 cm × 35 cm × 20 cm, with five mice per cage. The temperature and relative humidity were maintained at 21°C-25°C and 45%–65%, respectively, and a 12-h light/12-h dark cycle was provided. The animals were supplied with standard laboratory mouse maintenance diet (1010088, Jiangsu Xietong Pharmaceutical Bioengineering Co., Ltd) and sterile purified water ad libitum. The animals were acclimatized to the environment for 1 week before the experiment. The use of animals was approved by the ethics committees of biomedicine research, Hunan Normal University in Changsha, China (No. 2021-096).
A total of 30 mice were randomly divided into three groups with 10 mice in each group. The control group (Ctrl) was instilled intratracheally with 50 µL saline; The Silica group was instilled intratracheally with 50 µL saline containing 4 mg natural crystalline silica; The Cigarette filter silica group was instilled intratracheally with 50 µL saline containing 4 mg cigarette filter silica. The pretreatment of silica and the establishment of mice models was performed as previously described (Li et al., 2017). Briefly, silica particles were ground in saline for 3 h, boiled in 1 mol/L hydrochloric acid, washed, dried and suspended in sterile saline. The suspension was sonicated for 10 min before use. Mice were anesthetized by inhalation of isoflurane (1.5%) and placed on a platform. A 50 μL solution containing 4 mg silica particles was instilled by a single intratracheal instillation on the first day. The mice's conditions were observed daily and weights were monitored weekly. Five mice in each group were sacrificed at 7 or 28 days after instillation; both bronchoalveolar lavage fluid (BALF) and lung tissues were collected.
Enzyme linked immunosorbent (ELISA) assay
The levels of inflammatory factors tumor necrosis factor-α (TNF-α), interleukin (IL)-6, and IL-1β in BALF from mice were measured by ELISA (R&D Systems, Minneapolis, MN, USA) according to the manufacturer's instructions. Briefly, adding blanks, standards, and samples separately to a 96-well plate, with two replicates per sample. After gentle mixing, the plates were incubated for 30 min at 37°C, washed five times with PBS, and 50 µL of HRP-conjugated reagent was added to each well. After incubation for 30 min at 37°C and washing, chromogen solutions A (50 µL) and B (50 µL) were added and incubated for 10 min. Stop solution (50 µL) was then added to each well to end the reaction. Blank wells were set to zero, and the optical density (OD) of each well at 450 nm was measured within 15 min.
Pulmonary function test
Pulmonary function test was performed using the unrestrained pulmonary function plethysmograph (Buxco, FinePointeTM, USA) following the manufacturer’s protocol. Briefly, after tracheotomy, tracheas of all deeply anesthetized mice were connected with an auto-ventilator by an endotracheal tube. Then, the total lung capacity (TLC), inspiratory capacity (IC), vital capacity (VC), and forced vital capacity (FVC) were measured at day 28. Repeat the measurement 3 times for each mouse and take the average.
Quantitative PCR (qPCR)
Total RNA was extracted from lung tissues using TRIzol (R011-100, Genview, Beijing, China) according to the manufacturer's protocol. Reverse transcription was performed using the Prime Script RT kit (RK20429, ABclonal, Wuhan, China), followed by qPCR using the SYBR Green Master Mix Kit (RK21204, ABclonal, Wuhan, China). The data were normalized to GAPDH as the endogenous control. Relative expression was calculated using the 2−ΔΔCt method. The specific primer sequences were as follows: Caspase-3, F: GCTGACTTCCTGTATGCTTACTC, R: AATTCCGTTGCCACCTTCCT, Col-Ⅰ, F: CAGTGGCGGTTATGACTTCAG, R: GGCTGCGGATGTTCTCAATC, α-SMA, F: GAACACGGCATCATCACCAA, R: ATCTCCAGAGTCCAGCACAATA, GAPDH, F: AATGGTGAAGGTCGGTGTGA, R: CGCTCCTGGAAGATGGTGAT.
Western blot
The pulmonary tissues were lysed using RIPA Lysis Buffer (P0013 B, Beyotime, Shanghai, China) to extract the total protein measured by BCA Protein Assay Kit (P0010, Beyotime, Shanghai, China). Denatured proteins were separated by electrophoresis on 8%–15% sodium dodecyl sulfate-polyacrylamide gels (SDS-PAGE) and transferred onto polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with 5% non-fat milk and incubated with primary antibodies overnight at 4°C. Then, the blots were incubated with horseradish peroxidase-conjugated secondary antibodies for 1 h at room temperature. Immunoreactive bands were detected using an enhanced chemiluminescence (ECL) kit (BL520 A, Biosharp, Hefei, China) followed by quantitative estimation using Image J software.
Histology
The lungs of the mice were immersed using 4% neutral buffered formalin followed by paraffin embedding and cut into 5 μm sections. Then inflammation and fibrosis were assessed by hematoxylin and eosin (H&E) and Masson’s trichrome staining of paraffin lung sections, according to the manufacturer’s protocol. Fibrosis was scored using the Image J software. A mean value of the fibrosis scores was determined by evaluating three different fields of each section, with a total of three sections per animal (5 mice/group).
Measurement of HYP content
The levels of hydroxyproline (HYP) in lung tissue samples were quantified using the assay kit (A030-2-1, Jiancheng, Nanjing, China) according to the manufacturer’s instructions. The absorbance was measured at 550 nm, respectively. HYP content was calculated as μg/mg wet lung.
Statistical analysis
Measurement data were expressed as mean ± standard deviation (SD) and statistical significance was determined using one-way analysis of variance (ANOVA) and Student-Newman-Keuls test (for multiple groups). Count data were expressed as percentages and statistical significance was determined using the Chi-square test. All statistical analyses were performed using SPSS v.23.0 software. A value of p < 0.05 indicated statistical significance.
Results
Cigarette filter silica exposure decreased pulmonary function in cigarette factory workers
Comparison of detection rate of abnormal pulmonary function among workers in different workshops.
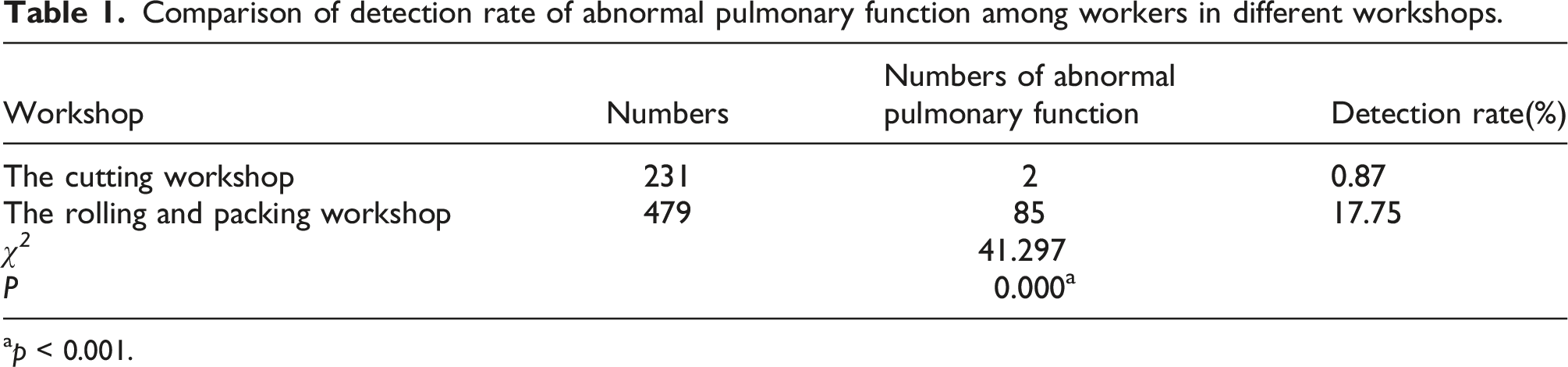
ap < 0.001.
Cigarette filter silica induced release of inflammatory factors in mice at the early stage
First, we measured the levels of inflammatory factors in BALF of each group by ELISA. The results in Figure 1 showed that the levels of TNF-α, IL-6, and IL-1β were significantly elevated of both exposed groups at day 7 and 28. Notably, the levels of inflammatory factors were higher in the cigarette filter silica group after 7 days of instillation, compared to the silica group (Figure 1(a)). However, there was no significant difference between the two groups at day 28 (Figure 1(b)). The data indicated that cigarette filter silica caused a more severe inflammatory response at the early stage. The expression levels of inflammatory factors TNF-α, IL-6, and IL-1β in BALF of mice. (a) The levels of inflammatory factors TNF-α, IL-6, and IL-1β in BALF of each group were detected by ELISA at day 7. (b) The levels of inflammatory factors TNF-α, IL-6, and IL-1β in BALF of each group were detected by ELISA at day 28. Significance was determined using one-way ANOVA. n = 5; **, p < 0.01.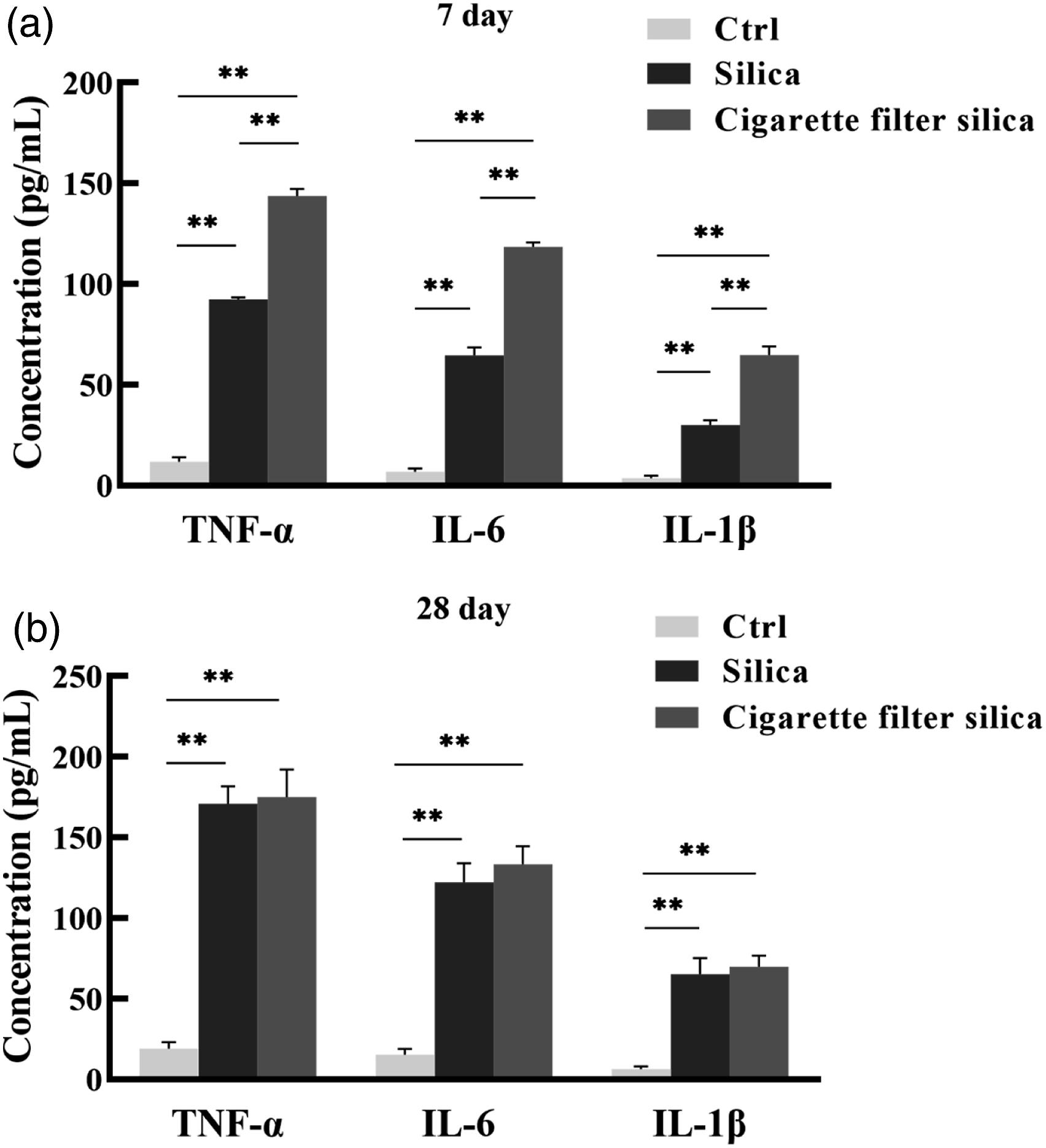
Cigarette filter silica decreased pulmonary function in mice at the late stage
Next, we evaluated the pulmonary function of mice through tests of TLC, IC, VC, and FVC at day 28. Our results in Figure 2 showed that both exposure groups had significantly lower TLC, IC, VC, and FVC. Moreover, these indexes decreased more in mice in the cigarette filter silica group compared to the natural crystalline silica group. These findings indicated that cigarette filter silica could cause more severe pulmonary dysfunction at the late stage. Cigarette filter silica reduced pulmonary function in mice. Anesthetized mice were endotracheally intubated with an airway catheter, and the parameters of pulmonary function were measured by a pulmonary function testing system. (a) TLC (total lung capacity), (b) IC (inspiratory capacity), (c) VC (vital capacity), (d) FVC (forced vital capacity). Significance was determined using one-way ANOVA. n = 5; **, p < 0.01.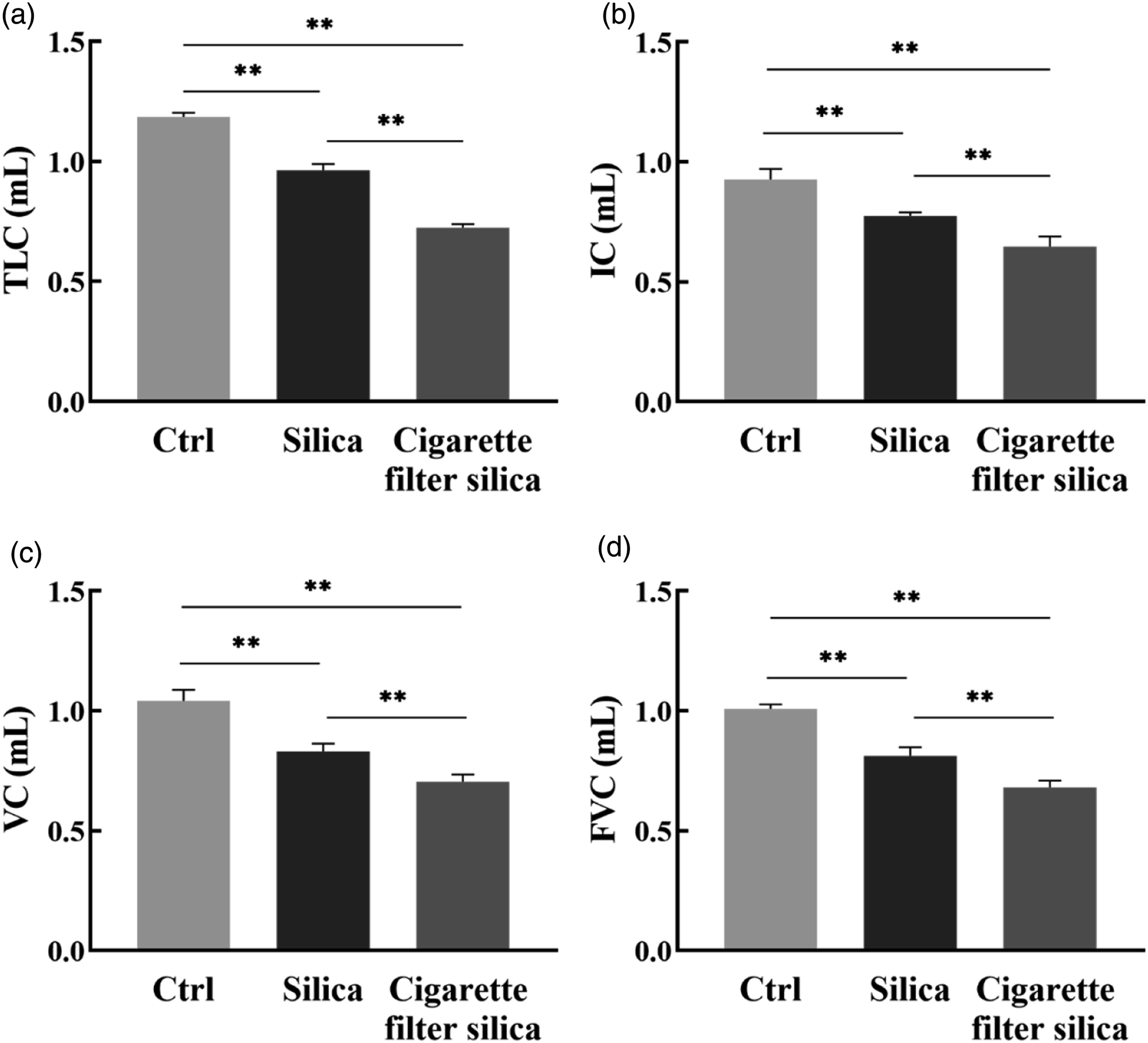
Cigarette filter silica induced apoptosis of pulmonary tissues in mice at the late stage
Caspase 3 regulates the terminal signaling pathway of apoptosis and is a key execution protein in apoptosis (Tan and Chen, 2021). Therefore, we analyzed the levels of caspase 3 in lung tissues at day 28 by Western Blot and qPCR. As shown in Figure 3, similar to the silica group, the levels of caspase 3 were significantly increased in the cigarette filter silica group. These results suggested that cigarette filter silica could induce apoptosis in mice lung tissues at the late stage. Cigarette filter silica induced apoptosis of pulmonary tissues in mice. (A-B) Western blot detection of cleaved caspase-3 expression levels in pulmonary tissues of each group at day 28, with GAPDH as the loading control. (C) qPCR detection of caspase-3 transcriptional levels in pulmonary tissues of each group at day 28, with GAPDH as the loading control. Significance was determined using one-way ANOVA. n = 5; **, p < 0.01.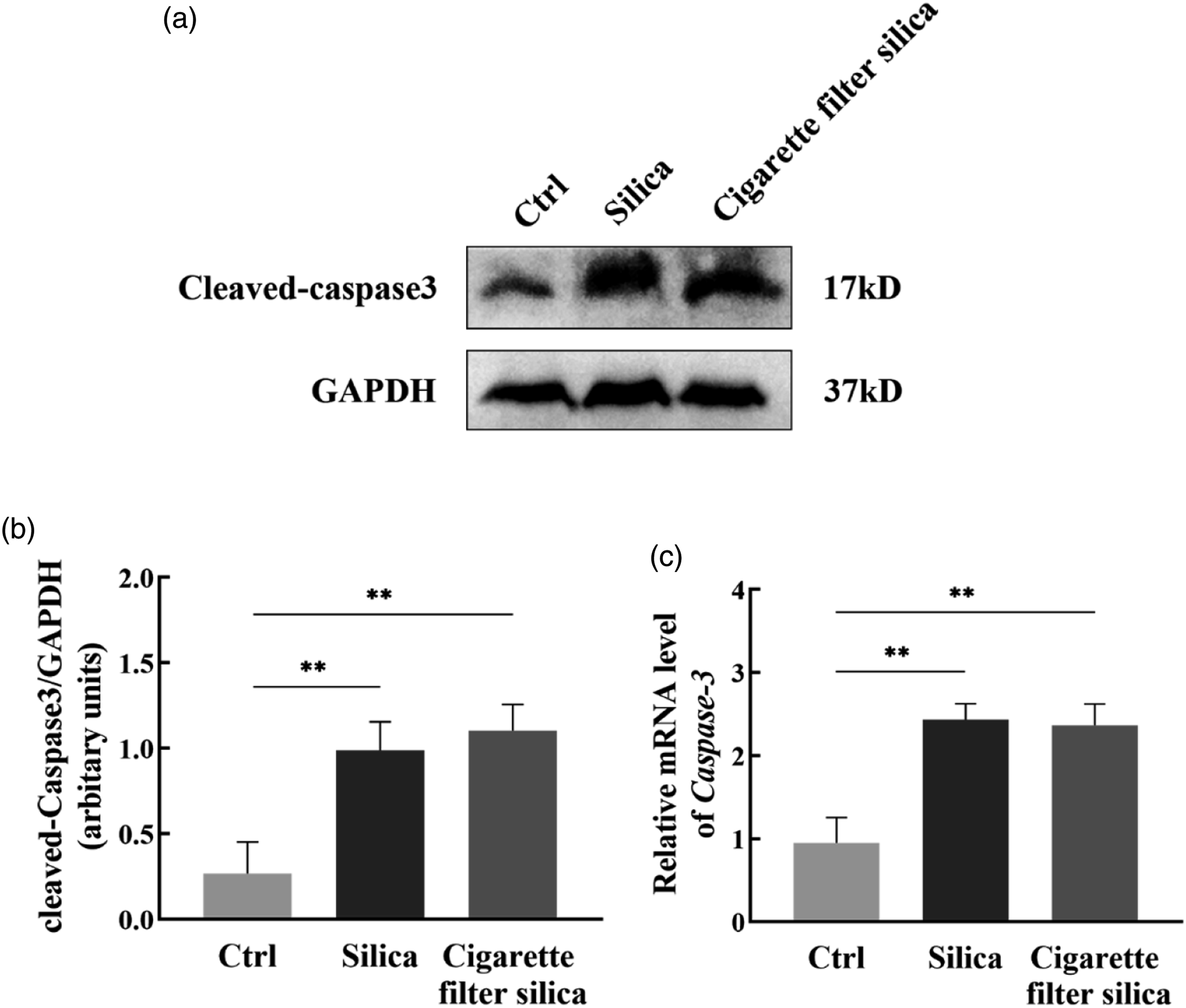
Cigarette filter silica induced collagen deposition and fibrosis of pulmonary tissues in mice at the late stage
To further assess the histopathological changes of mice, we stained lung tissues with H&E and Masson's trichrome after instillation 28 days. In Figure 4, H&E staining showed that the control group had no significant damage to the alveolar structure, while the exposed groups had significant damage to the alveolar walls, with inflammatory cells infiltrating and cellular nodules present (Figure 4(a)). In addition, Masson's staining revealed that both cigarette filters silica and silica exposure caused significant collagen deposition and enlarged fibrosis areas in the lungs (Figure 4(b)–(c)). Moreover, the levels of HYP, Col-Ⅰ and α-SMA were also significantly increased in the lung tissues of the exposed mice at day 28 and there was no significant difference between the two groups (Figure 4(d)–(f)). Similar results were obtained for the transcription levels of Col-Ⅰ and α-SMA by qPCR (Figure 4(g)–(h)). Our findings suggested that exposure to cigarette filter silica could lead to collagen deposition and fibrosis of lung tissues in mice at the late stage. Cigarette filter silica induced collagen deposition and fibrosis of pulmonary tissues in mice. (a) H&E staining of mice lung tissues in each group at day 28 (scale bar: upper×50 µm; lower×100 µm). (b)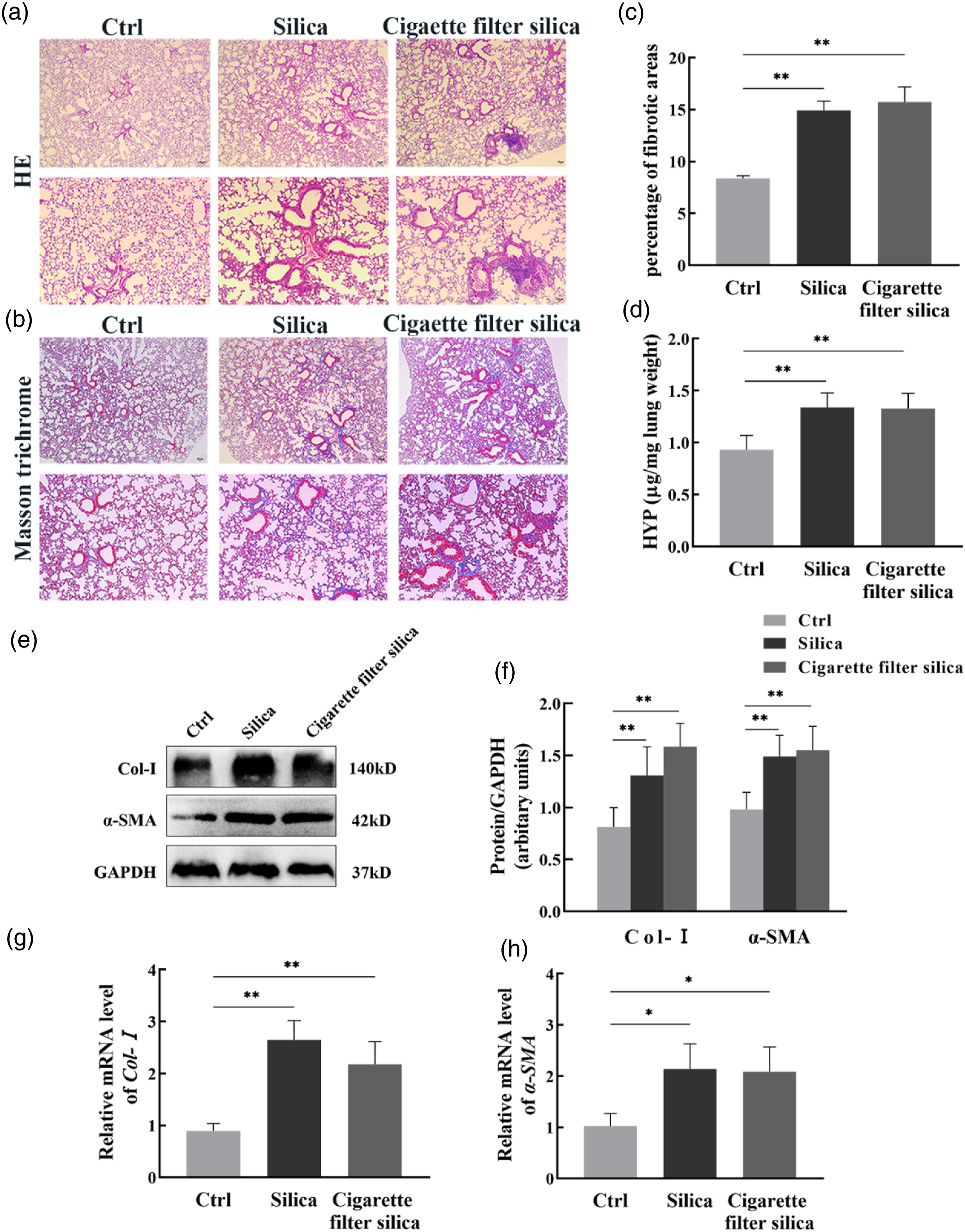
Discussion
Because of the wide range of industrial applications of silica nanoparticles, such as food additives, pharmaceutical carriers, and use in cigarette filters, there has been a dramatic increase in exposure to silica nanoparticles with growing concerns about their potential health effects (Huang et al., 2022). We collected medical data from cigarette factory workers and found pulmonary dysfunction in workers exposed to nanosilica in cigarette filters. Previous studies have revealed that crystalline silica induced inflammation, apoptosis of lung tissue, and particularly, occupational exposure to crystalline silica caused fibrosis of lung tissue (He et al., 2020; Tan et al., 2023). Therefore, we assessed the differences in pulmonary toxicity between cigarette filter silica and natural crystalline silica in this study.
Available studies have shown some differences in the pulmonary toxicity between nanosilica and micron silica. Several studies have reported that nanosilica induced more severe acute lung inflammation and oxidative damage in rats compared to micron silica (Jin et al., 2008); however, in terms of pro-fibrosis, nanosilica has not been found to induce more severe fibrotic damage compared to micron silica (Zhang et al., 2014; Yang et al., 2017, 2019). And it was found that prolonged exposure to nanosilica caused interstitial fibrosis in rat lungs, unlike the silicotic nodules induced by micron silica (Zhang et al., 2014). A recent study also showed granulomatous inflammation and large fibrotic nodules in the lung tissue of rats exposed to nanosilica (Guan et al., 2022). However, it is unclear whether there are such differences in the pulmonary toxicity between cigarette filter silica and natural crystalline silica. According to our previous study, the mouse model induced by silica showed characteristic inflammation and fibrosis at day 7 and day 28, respectively (Zhang et al., 2021). Thus, we compared the mouse models induced by cigarette filter silica and crystalline silica at both the early acute inflammatory stage (7 days post-instillation) and the late fibrosis stage (28 days post-instillation) to explore the potential pulmonary toxicity of cigarette filter silica.
Cytokines such as TNF-α, IL-6, and IL-1β played a crucial role in local lung injury and lung inflammatory response, which could trigger alveolar inflammation and pulmonary fibrosis through excessive repair of local damage (Hutyrova et al., 2002; Kusner et al., 2010; Tumelty et al., 2014). Our findings showed that cigarette filter silica caused more severe lung inflammation in the early stage, as evidenced by higher levels of inflammatory factors TNF-α, IL-6, and IL-1β in BALF. In addition, occupational exposure to silica leads to pulmonary fibrosis, which is usually accompanied by pulmonary ventilation dysfunction. We found that cigarette filter silica resulted in more severe pulmonary ventilation impairment of mice, as evidenced by lower levels of TLC, IC, VC, and FVC. Previous studies have shown that nanosilica triggered inflammation, with some degree of size-dependence (Kusaka et al., 2014; Tsugita et al., 2017; Nakayama, 2018). We inferred that the extremely small size and the increased surface energy of silica nanoparticles could alter the chemical activity of these particles, thus increasing their biological activity and leading to more serious damage. Additionally, research has demonstrated that, compared to micron particles at the same mass concentrations, nanoparticles had a much larger number of particles, which could reduce the ability of lungs to eliminate these particles and resulted in particle overload (Ferin et al., 1992). Then the acute overload of nanoparticles in the lungs might cause more severe damage in a short time. Accordingly, we could infer that cigarette filter silica is more likely to cause pulmonary ventilatory impairment of respiratory system.
Caspase 3 is a critical downstream regulator in both exogenous and endogenous apoptotic signaling pathways. When activated, caspase 3 cleaves and activates downstream effector caspases, which in turn subsequently cleave the cytoskeleton and nuclear proteins in a protein hydrolysis cascade, followed by apoptosis (Blocksom et al., 2004). HYP is a collagen-specific amino acid that reflects the extent of collagen proliferation (Gordon and Hahn, 2010). α-SMA and Col-Ⅰ are two important markers of fibrosis (Zhang et al., 2021). Our results revealed that cigarette filter silica induced apoptosis and fibrosis in mice lung tissues, as evidenced by significantly increased levels of caspase 3, HYP, Col-Ⅰ, and α-SMA. Histological analysis also showed inflammatory cell infiltration, alveolar wall damage, fibrous nodules, and collagen deposition. However, compared to the same dose of crystalline silica, cigarette filter silica did not show a more marked pro-apoptotic and pro-fibrotic effect. In terms of pro-apoptotic effects, most in vitro studies have reported that the pro-apoptotic toxicity of nanosilica exhibits size-dependence, which is inconsistent with our results (Lin et al., 2006; Wang et al., 2018). We believed that this contradiction might be attributed to the difference between in vivo and in vitro experiments and deserved further exploration. As for the pro-fibrotic effects, in vivo studies have shown that the pro-fibrotic potential of nanosilica was less pronounced than that of micron silica at the same concentration (Zhang et al., 2014; Yang et al., 2017, 2019). It has been observed that nanoparticles could transfer from the lungs to other organs via the circulatory system by using fluorescein or radiolabeling techniques (Nemmar et al., 2001; Furuyama et al., 2009). In addition, Qi et al. further suggested that silica nanoparticles might pass through the lungs into the circulatory system and be redistributed in the heart, causing myocardial damage and dysfunction (Du et al., 2019; Qi et al., 2022). Therefore, it was reasonable to assume that due to the smaller particle size and stronger transfer diffusion ability of cigarette filter silica, some of the particles could enter the circulatory system and then transit to other tissues, resulting in less exposure to the lungs, thus, avoiding more serious apoptosis and fibrosis of lung tissues.
The present study has certain limitations that need to be acknowledged. First, we did not measure the levels of cigarette filter silica in the cigarette factory workshops, so the actual exposure dose of cigarette filter silica from workers is unknown. Second, we have not yet explored the mechanisms underlying the pulmonary dysfunction caused by cigarette filter silica. Therefore, future research is necessary to shed more light on the mechanism of action of cigarette filter silica.
Overall, cigarette filter silica induced severe lung inflammatory injury in mice as it activated the release of pro-inflammatory cytokines. Also, cigarette filter silica induced apoptosis and fibrosis of lung tissues in mice, causing pulmonary dysfunction. Although the actual exposure dose of nanosilica in cigarette filters from cigarette factories may not be sufficient to cause pulmonary fibrosis in workers, our study also revealed that exposure to nanosilica in cigarette filters caused more severe lung inflammation and pulmonary ventilation impairment than the same doses of crystalline silica. In the future, we need to pay more attention to nanosilica protection in cigarette factories to prevent pulmonary dysfunction in workers.
Footnotes
Acknowledgements
We acknowledged all authors for their contributions to the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 82173493) and the Natural Science Foundation of Hunan Province (No. 2023JJ30423).
