Abstract
Studies assessing the effects of partial-hepatic ischemia/reperfusion (I/R) injury focused on the damage to the ischemic-lobe, whereas few data are available on non-ischemic lobe. This study investigated whether acute liver I/R does affect non-ischemic lobe function via modulation of extracellular matrix remodeling. Male Sprague-Dawley rats underwent left lateral- and median-lobe ischemia for 30 min and reperfusion for 60 min or sham operation. After reperfusion, blood samples and hepatic biopsies from both the ischemic (left-lobe, LL) and the non-ischemic lobe (right-lobe, RL) were collected. Serum hepatic enzymes and TNF-alpha, tissue matrix metalloproteinases (MMP-2, MMP-9), liver morphology, malondialdehyde (MDA), and myeloperoxidase (MPO) were evaluated. Liver I/R injury was confirmed by altered increased hepatic enzymes and TNF-alpha. I/R induced an altered morphology and an increase in MMP-2 and MMP-9 activity not only in left-ischemic lobe (LL) but also in the right-non-ischemic (RL) lobe. A lobar difference was detected for MDA formation and MPO activity in both sham and I/R submitted rats, with higher levels in the left lobe for both groups. This study indicates that an increase in MMPs, which may be TNF-alpha-mediated, occurs in both the ischemic- and the non-ischemic lobes; the heterogeneous lobe concentrations of MDA and MPO suggest that the random sampling of liver tissue should be avoided.
Introduction
Prolonged oxygen deprivation corresponding to the ischemic period is observed during liver resection, transplantation, and trauma; the subsequent oxygen restoration always leads to reperfusion injury. In particular, warm ischemia occurs in hypovolemic shock or inflow occlusion during liver surgery. Experimental and clinical studies have been mainly focused on the cellular alterations taking place in the ischemic lobes, whereas little is known on the changes affecting the non-ischemic lobes. Indeed, the non-ischemic lobe has been almost invariably considered as the “control” lobe in experimental studies involving rat (Hsu et al. 2002) or pig experimental models (Helling et al. 1994). However, there is increasing evidence of a functional heterogeneity between the individual liver lobes that revealed a striking and yet unexplained inter- and intra-lobular variability of acute hepatic necrosis as showed by heterogeneous damage distribution within a single lobe or among different lobes (Malarkey et al. 2005). Several studies reported marked lobe variation in the extent and distribution of tissue injury during chemical carcinogenesis (Richardson et al. 1986), acetaminophen hepatoxicity (Irwin et al. 2005), copper distribution (Faa et al. 1995), and cirrhosis (Regev et al. 2002). One possible explanation may be associated to the distribution of the liver vascular system: there is an incomplete mixing of blood coming from the gastrointestinal tract and spleen, leading to variation in the delivery of nutrients and toxins to the liver lobes. For example, venous portal blood draining the stomach and the spleen tend to be directed to the left side of the liver. Another possible mechanism that may play a central role in such hepatic variability is a different lobar gradient of gene expression profiles (Malarkey et al. 2005) as documented for acetaminophen hepatoxicity, that is, a situation in which the variability of lobar damage has been correlated to different regional gene expression (Ruepp et al. 2002). Some reports have also described inter-lobar differences in the levels of malondialdeyde (MDA), and along this line, Singh et al. (2001) reported a lobe-specific increase in MDA-DNA adduct formation in the mouse liver following
The matrix metalloproteinases (MMPs) are a family of at least 25 well-characterized proteases that use zinc-dependent catalysis to break down several components of the extracellular matrix (ECM), thereby allowing cell movement and tissue reorganization. Most MMPs are secreted from cells as pro-enzymes (pro-MMPs) that are activated extracellularly (Visse and Nagase 2003). Following liver I/R injury, MMPs have profound effects on tissue integrity. A key role seems to be played by MMP-2 (gelatinase A; EC 3.4.24.24) and MMP-9 (gelatinase B; EC 3.4.24.35), since they are critically involved in the degradation of collagen IV and fibronectin (Ram et al. 2006), two main components of the space of Disse (Khandoga et al. 2006). Recently, increased liver MMP-9 expression has been described following I/R injury (Moore et al. 2007), and a correlation between serum MMP-9 and the severity/progression of liver damage has been found in the setting of I/R injury (Kuyvenhoven et al. 2003), acute allograft rejection (Kuyvenhoven et al. 2004), and chronic viral hepatitis (Leroy et al. 2004). Inflammatory cytokines such as TNF-alpha participate to ECM degradation following liver injury and hepatic TNF-alpha expression has been demonstrated to parallel MMPs induction (Khandoga et al. 2006). Some experimental evidence has suggested MMP inhibition as a promising strategy in the setting of the pharmacological interventions aimed at limiting the post-ischemic hepatic damage not only in whole liver transplantation but also in acute small-for-size liver graft injury (Ma et al. 2010).
Moreover, although previous studies reported an involvement of myeloperoxidase activity in the I/R injury (Jaeschke et al. 1990), no data are available on the inter-lobe distribution of this enzyme.
The aim of the present study was therefore to investigate whether acute liver ischemia-reperfusion injury does affect the function and the structure of the non-ischemic lobe via modulation of ECM remodeling, MDA formation, and MPO activation. These aspects have been evaluated in both the ischemic and the non-ischemic lobe.
Material and Methods
Animals
The use of animals in this experimental study was approved by the National Institute for Research, and the animals were cared for according to its guidelines. Fourteen male Sprague-Dawley rats (200–250 g; Charles River, Calco-Lecco, Italy) with free access to water and food were used.
Materials
All reagents were of the highest grade of purity available and were obtained from local suppliers.
Ischemia-Reperfusion (I/R) Procedure
The effects of I/R were studied in vivo in a partial normothermic hepatic I/R model. Briefly, the abdomen was opened by a median incision in pentobarbital anesthetized rats (50 mg/kg). Ischemia to the left and median lobe (LL and ML) was induced by clamping for 30 min with microvascular clips the branch of portal vein and the branch of the proper hepatic artery (Figures 1 and 2) after the bifurcation to the right lobe, with the abdomen temporary closed with a suture. After 30 min of ischemia, the abdomen was reopened, the clips were removed, the abdomen was closed again, and the liver was allowed to reperfuse for 60 min. By using partial, rather than total hepatic ischemia, portal vein congestion and subsequent bacterial translocation into the portal venous blood was avoided. To prevent postsurgical dehydration and hypotension, 1 ml of saline was injected in the inferior vena cava. During I/R the animals (
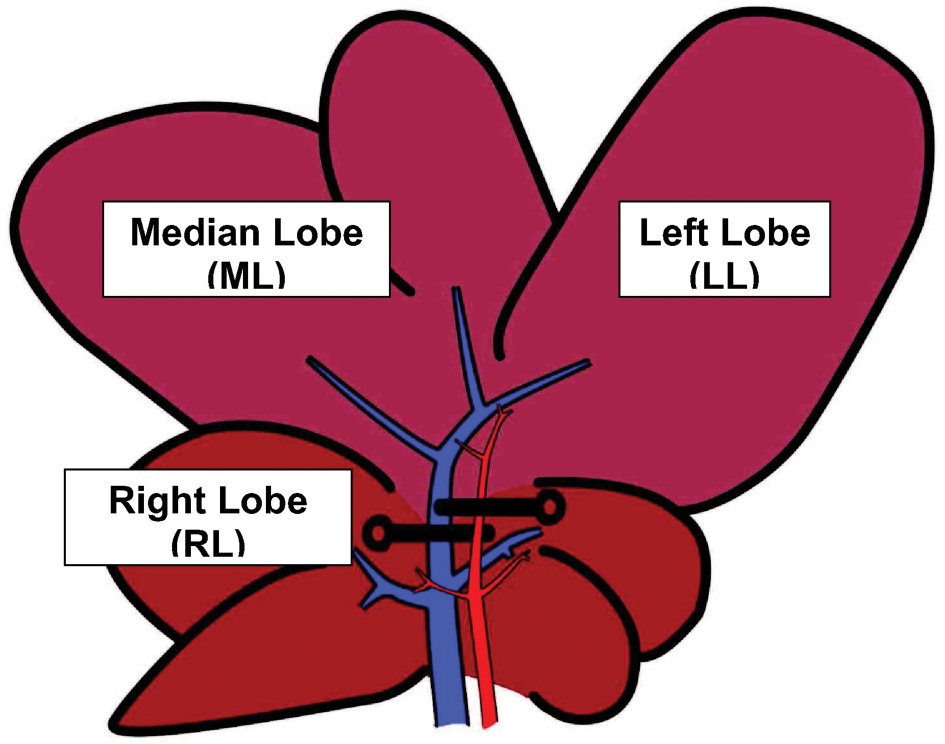
Graphic (schematic) representation of hepatic lobes in Sprague-Dawley rats. Ischemia was induced to the median lobe (ML) and left lobe (LL) but not to the right lobe (RL).
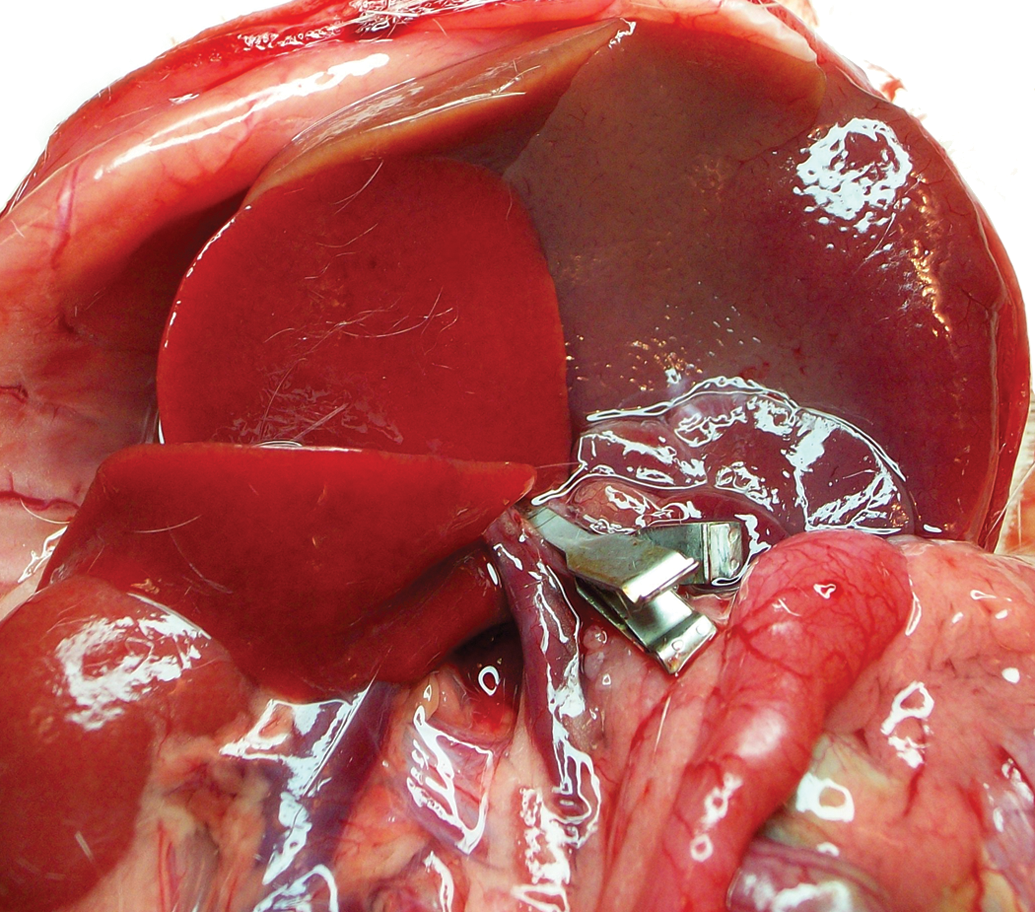
Surgical image of hepatic lobes in Sprague-Dawley rats. Ischemia was induced to the median lobe (ML) and left lobe (LL) but not to the right lobe (RL).
Biochemical Assays
Liver injury was assessed by serum release of alanine transaminase (ALT), aspartate transaminase (AST), and lactate dehydrogenase (LDH) by an automated Hitachi 747 analyzer (Roche/Hitachi, Indianapolis, IN, USA).
The amounts of malondialdehyde (MDA) formation were quantified by HPLC method using the Chromosystems assay kit (Chromsystems GmbH, München, Germany). The assay was performed according to manufacturer instructions with some modifications: briefly, the derivatized samples were incubated for 60 min at 95°C and finally used after centrifugation.
Polymorphonuclear leukocyte (PMN) accumulation was assessed via tissue myeloperoxidase (MPO) activity: after tissue extraction with hexadecyltrimethylammoniumbromide (HTAB) (Kuebler et al. 1996), the MPO activity was evaluated using a Cayman Kit according the manufacturing procedures.
Serum levels of TNF-alpha have been evaluated by ELISA kit according to the manufacturing procedures (R&D Systems, Minneapolis, MN, USA).
Tissue Sources for MMPs Analysis
After sacrifice, hepatic lobes were quickly excised and placed in cold (4°C) buffer (30 mM Histidine, 250 mM sucrose, 2 mM EDTA, pH 7.2) to remove blood. Liver was weighed and subsequently cut, frozen in liquid nitrogen, and stored at –80°C until use.
Hepatic Protein Isolation
Hepatic protein were extracted by homogenization (IKA-Ultraturrax T10) of frozen liver tissue, in an ice-cold extraction buffer (1:10 wt/vol) containing 1% Triton X-100, 500 mmol/L Tris-HCl, 200 mmol/L NaCl, and 10 mmol/L CaCl2, pH 7.6 (Kossakowska et al. 1998). The homogenate was then centrifuged (30 min. at 12,000 rpm at 4°C), and the protein concentration of the supernatant was measured with the colorimetric Lowry method (Lowry et al. 1951). Samples were stored at –20°C before use.
MMPs Zymography
To detect MMPs activity present in the samples, the homogenate protein content was normalized by a final concentration of 400 µg/ml in sample loading buffer (0.25 M Tris-HCl, 4% sucrose w/v, 10% SDS w/v and 0.1% bromphenol blue w/v, pH 6.8). After dilution, the samples were loaded onto electrophoretic gels (SDS-PAGE) containing 1 mg/ml of gelatin under nonreducing conditions (Kleiner and Stetler-Stevenson 1994) followed by zymography as described previously (Tozzi et al. 2007).
The zymograms were analyzed by densitometer (GS 710 Densitometer BIORAD, Hercules, CA, USA), and data were expressed as optical density (OD), reported to 1 mg/ml protein content.
Tissue Morphology
Hepatic lobes were fixed in 2% p-paraformaldehyde in 0.1 M phosphate buffer at pH 7.4 for 24 hours, processed routinely, and embedded in Paraplast wax. Sections (8-µm thick) were stained with hematoxylin-eosin (H&E). To appraise the severity of hepatic injury, H&E-stained sections were evaluated as follows: Grade 0, minimal or no evidence of injury; Grade 1, mild injury consisting of cytoplasmic vacuolation and focal nuclear pyknosis; Grade 2, moderate-to-severe injury with extensive nuclear pyknosis, cytoplasmic hypereosinophilia, and loss of intercellular borders; and Grade 3, severe necrosis with disintegration of hepatic cords, hemorrhage, and occasional granulomas (Serafín et al. 2004).
Statistical Analysis
Results are expressed as mean ± standard error. Comparisons between groups were performed by unpaired
Results
Liver I/R Injury
As expected, serum AST, ALT, and LDH increased in animals submitted to ischemia (30 minutes) and reperfusion (60 minutes) (
Serum enzymes and TNF-alpha levels after 30 min ischemia and 60 min reperfusion.
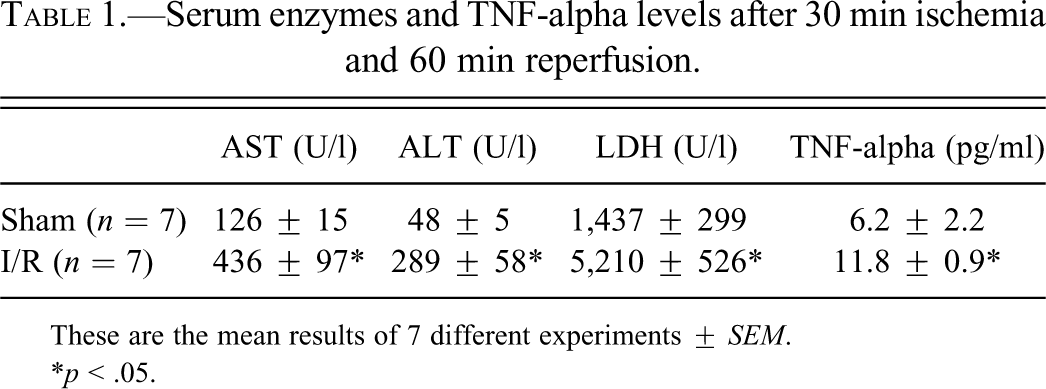
These are the mean results of 7 different experiments ±
*
MDA Formation
In the ischemic left lobe (LL) of the I/R rats, MDA formation was comparable with sham-operated rats (nmol/mg prot: 0.29 ± 0.04 versus 0.29 ± 0.06, respectively,
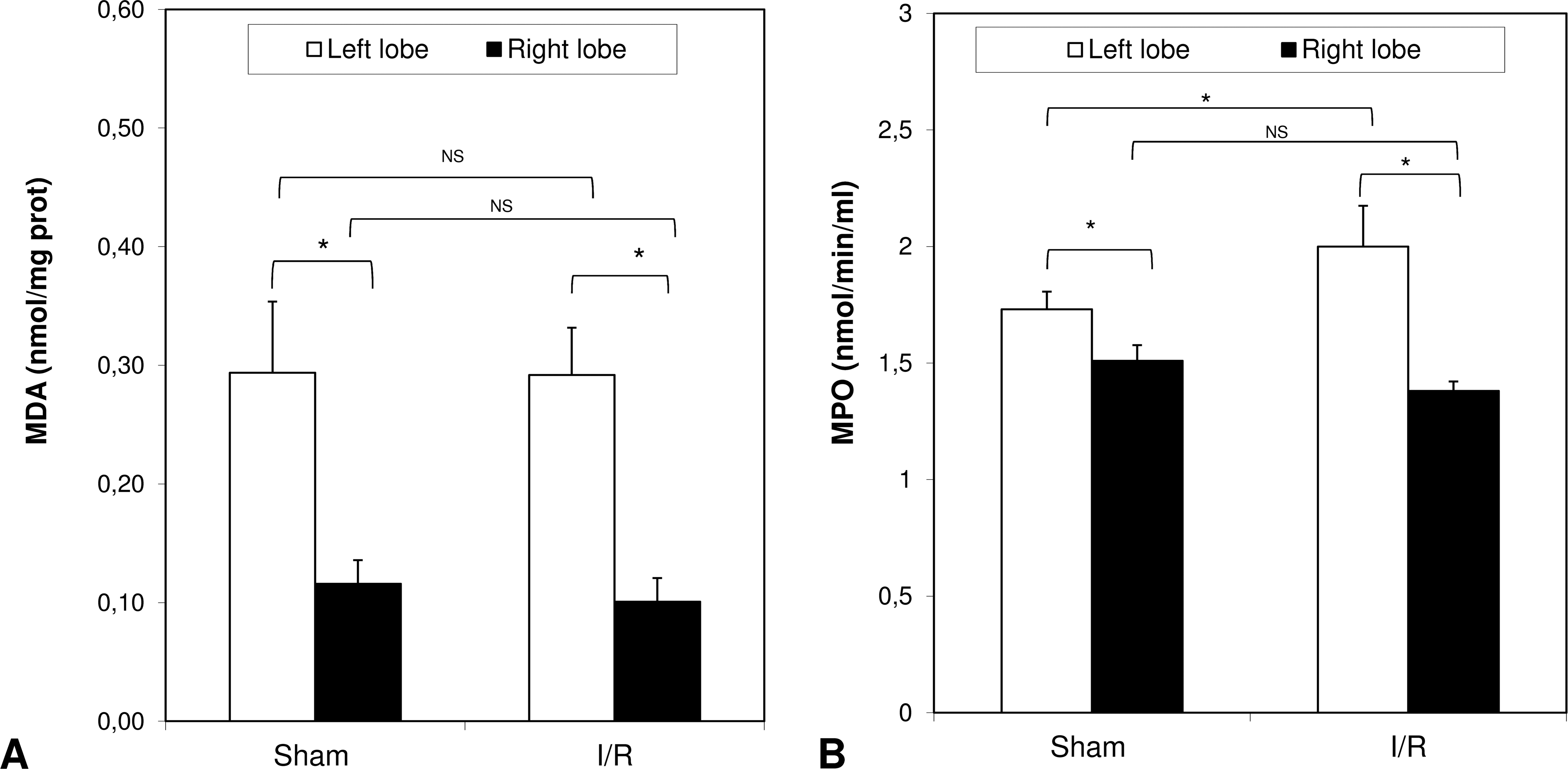
Hepatic levels of MDA and MPO at the end of reperfusion. Livers were submitted to 30 min ischemia to the left and median lobe (LL and ML) but not to the right lobe (RL). Sham-operated control animals had similar manipulation without vascular occlusion. After 60 min, reperfusion samples from LL and RL were collected from both groups. The results are reported as the mean ±
MPO Levels
Liver MPO activity, an index of neutrophil infiltration after I/R injury was 1.73 ± 0.076 nmol/min/ml protein in the LL of the control group (Figure 3B). After 30 min ischemia and 60 min reperfusion, these values increased to 2.0 ± 0.175 nmol/min/ml (*
Liver Histology
A semiquantitative evaluation of liver lesions is shown in Figure 4. A statistically significant difference of liver damage was observed between right non-ischemic and left ischemic lobe in animals subjected to I/R. Although there was a reduction of injury in right non-ischemic lobe, the damage was significantly higher respect to sham-operated animals.
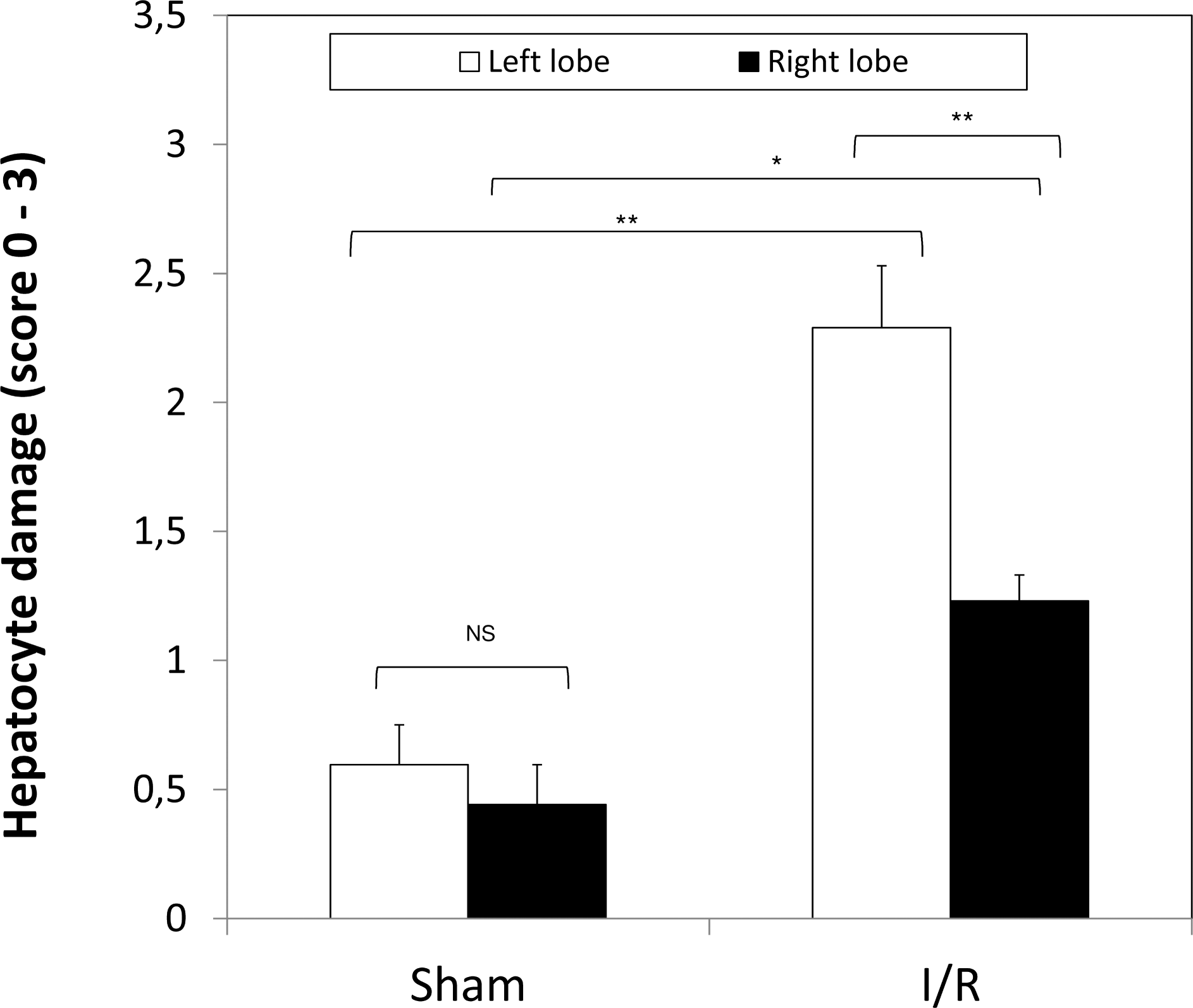
Hepatocellular damage measured by a semiquantitative score from 0 to 3 in the left and in the right hepatic lobes obtained from sham animals and animals submitted to I/R. Values are reported as mean ±
In the sham-operated animals, no significant morphological difference was observed between the right and left lobes (Figure 4 and Figure 5a–b). In animals submitted to I/R, the left ischemic lobe showed cytoplasm vacuolation of hepatocytes scattered through the entire parenchyma, intra- and extra-cellular edema, and sinusoid blood congestion mainly in peri-portal and midzone area (Figure 5d, f). Although much less severe than in the ischemic lobe, several alterations were also observed in the right non-ischemic lobe when compared with sham-operated animals. In particular, the sinusoids appeared dilated (Figure 5c) or even highly dilated in a few centrolobular areas (Figure 5e), and intracellular vacuolation was seen in few scattered hepatocytes (Figure 5e).
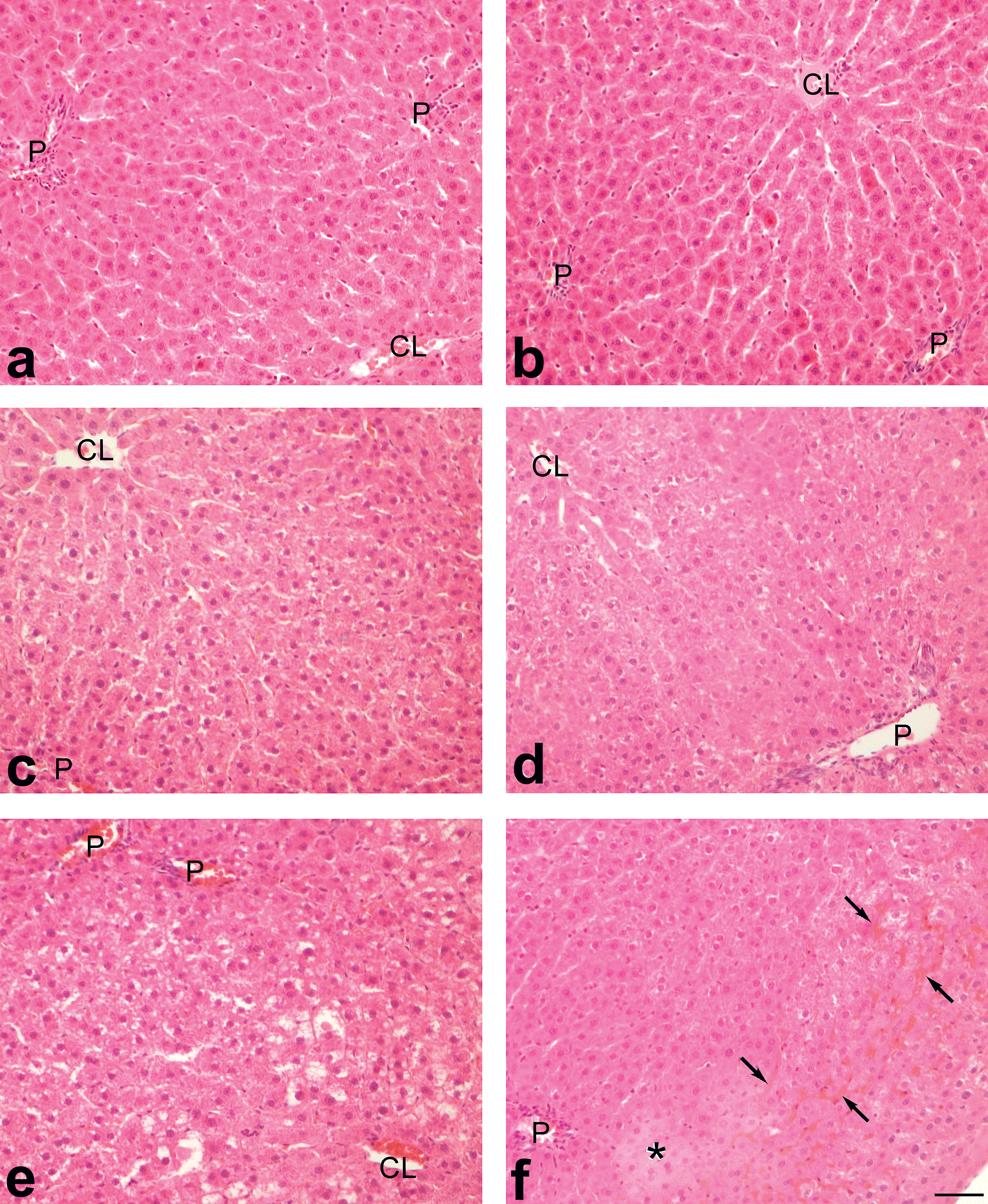
Representative light micrographs of liver samples in sham-operated rats with no I/R (Livers a and b) and in rats whose left lobes were first submitted to 30 min of ischemia and hence reperfused for 60 min (Livers c–f). Livers a, c, e: right lobe. Livers b, d, f: left lobe. P: portal vein; CL: centrolobular vein; asterisk: edematous area; black arrows: congested sinusoids. Hematoxylin & eosin. Scale bar: 50 µm.
Gelatinolytic Ativity
MMP-2 (Gelatinase-A) and MMP-9 (gelatinase-B) activities were evaluated to investigate the degree of MMP-induced hepatic extracellular matrix degradation after I/R injury and its variability between the different lobes. No difference between left and right lobes was seen for MMP-2 and MMP-9 activity for sham or for I/R submitted rats. However, the activity was altered differently in the left and right lobes when animals submitted to I/R were compared to sham animals. In particular, I/R induced a significant increase in MMP-2 and MMP-9 activity in respect to control animals not only in ischemic lobe but also in the right non-ischemic lobe (Figure 6).
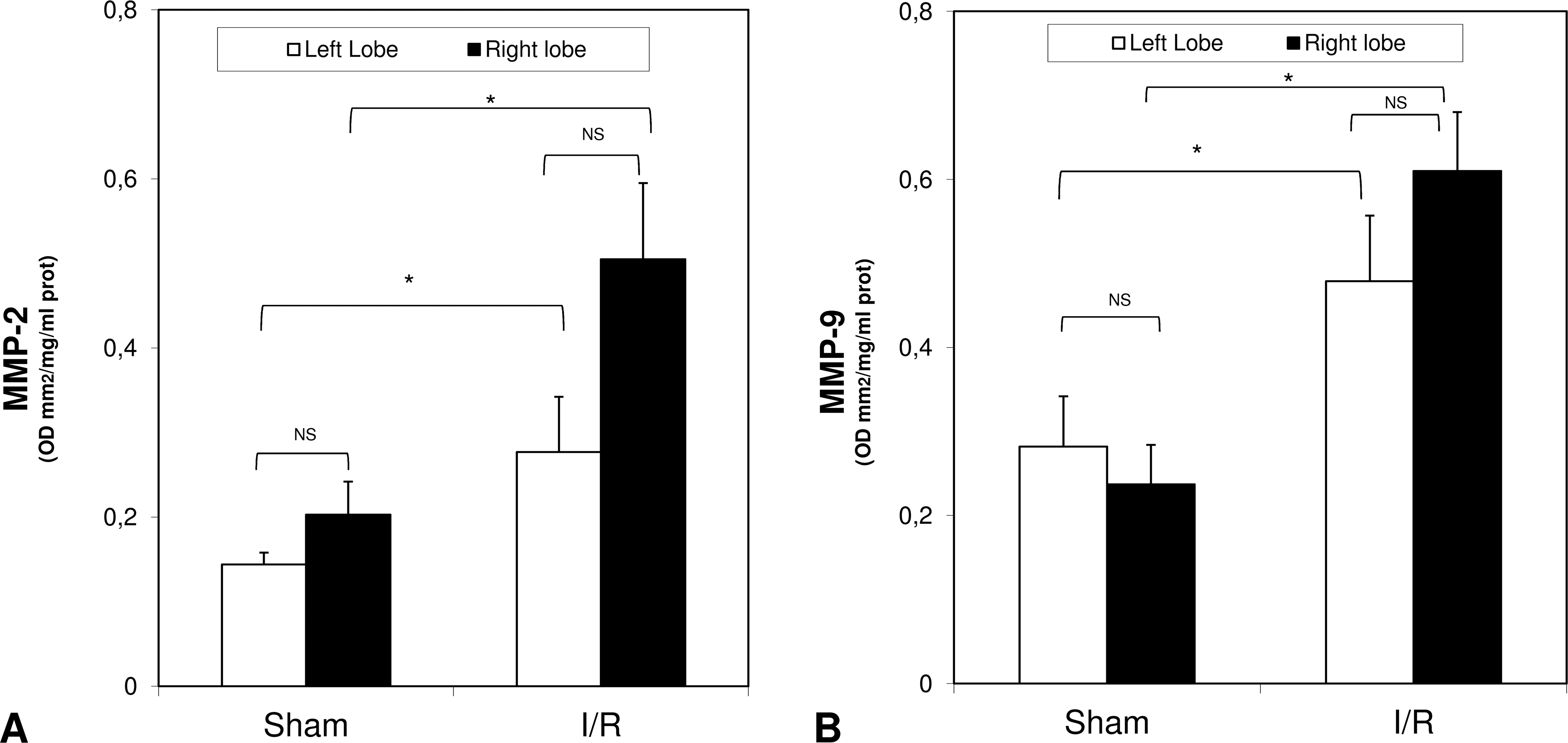
Bar graphs of MMP-2 (panel A) and MMP-9 (panel B) activity and gelatin zymography (panel C) at the end of reperfusion. Livers were submitted to 30 min ischemia to the left and median lobe (LL and ML) but not to the right lobe (RL). Sham-operated control animals had similar manipulation without vascular occlusion. After 60 min, reperfusion samples from LL and RL were collected from both groups. MMP-2 and MMP-9 gelatinase activity is expressed as optical density (OD) for mm2, reported to 1 mg/ml protein content. Data are shown as mean values ±
Discussion
These results support previous data on the lobar functional heterogeneity of the liver. In addition, we found that, under the reported I/R conditions, the right non-ischemic lobe does not exhibit oxidative stress-related damage, as evaluated by lipid peroxidation (MDA) and enzyme myeloperoxidase (MPO) secretion by neutrophils that were superimposable in sham and I/R rats. Therefore, our data suggest that I/R injury is not associated with oxidative damage in the right non-ischemic lobe, despite early signs of tissue damage. The latter is shown by stromal morphological alterations and disarranged sinusoids, possibly related to the concomitant increase in MMPs associated with I/R in the remote ischemic lobe. TNF-alpha from the I/R lobe may have circulated systemically and, reaching the non-ischemic right lobe, upregulated the MMPs during the reperfusion period.
The differential blood supply could be related to higher MPO and MDA in the left lobe as compared to the right lobe. The portal blood drains from mesenteric, gastric, splenic, and pancreatic veins and travels to the liver, where it branches into the right and left sides of the liver. It might be that an incomplete mixing of blood coming from the gastrointestinal tract and spleen occurs leading to variation in delivery of different nutrients or toxins to the liver lobes; this event is also called Portal Streamlining (Malarkey et al. 2005).
To the best of our knowledge, this is the first evidence showing that MPO content is significantly lower in the right compared with the left lobe. The MPO levels obtained are connected with the experimental model used: in the present work, we observed a limited increase in MPO because we induced a short ischemia (30 min) and evaluated the reperfusion injury after 60 min. A few neutrophils were occasionally seen in edematous portal spaces, and/or forming small granulomas around necrotic cells, especially in ischemic lobes. Miranda et al. (2010) found higher levels of MPO after 60 min ischemia and 2 or 6 hours reperfusion. Interestingly, the present work underlined that already after a short ischemia/reperfusion period, a change in MPO content occurs. The hepatic expression of MPO was a matter of debate, but a recent work of Amanzada et al. (2011), using both normal and inflamed liver, showed that MPO-expression is identified only in granulocytes and not in the parenchymal and non-parenchymal cells.
In Wistar rats, the right and the caudate lobes represent a functional unit that differs from the middle and the left lobes in terms of protein synthesis (Garcia-Moreno et al. 1994). Each lobe is organized into three-dimensional vascular units (Teutsch et al. 1999), although the left lobe has only one primary portal branch at variance with the median lobe that appears to have two portal branches (Duchen 1961). In this study we used a model of partial ischemia comparing the ischemic lobe with non-ischemic one. We did not make the right lobe ischemic because the total hepatic ischemia induced intestinal congestion (Coelho et al. 2011).
Previous reports indicated a yet unexplained interlobular variability in acute hepatic necrosis indicating the liver as an heterogeneously complex organ (Malarkey et al. 2005). Diethylnitrosamine-treated rats show a higher incidence of carcinoma in the left and median lobe as compared to the right one (Richardson et al. 1986). Also DNA damage is greater in the left lobe (Richardson et al. 1986), and the present study supports the hypothesis of a higher hepatocyte activity in this lobe when compared with the right one. Moreover, a different expression of positive and negative regulators of hepatocyte growth occurs in the different hepatic lobes after portal vein branch ligation in rats (Uemura et al. 2000).
The involvement of all liver lobes after partial regional I/R injury has been documented by other authors. For example, Lentsch et al. (1998) reported that after 90 min of hepatic ischemia in mice, edema was maximal after 3 hours of reperfusion in both the ischemic and non-ischemic lobes. The mechanism involved in this inter-lobe variation is largely unknown, although factors such as portal streamlining of blood to the liver (Duchen 1961) and differences in the metabolic capacity of each lobe have been proposed for explaining the heterogeneous liver lobe response (Lawson et al. 1974). Our study further supports the metabolic heterogeneity among the hepatic lobes.
Degradation of Interstitial Matrix Could Be Attributed to the MMPs Activity
The study of MMPs expression and activity following liver I/R injury is a developing field. Increased MMP activity is associated with hepatocellular damage as indicated by increased ALT levels and by hepatic microcirculatory disturbances such as reduced sinusoidal perfusion (sinusoid blood congestion). It is known that Kupffer cells and hepatic stellate cells (HSCs) can secrete MMPs (Knittel et al. 1999); sinusoidal endothelial cells (SEC) are another source of MMPs in the rat liver (Deleve et al. 2003). In particular, the ability of the HSCs to express significant amount of all matrix degradating enzyme and their inhibitors has been clearly demonstrated in Knittel et al. (1999). We support the possibility that the HSCs could play a role in the TNF-alpha and MMP induction (Knittel et al. 1999). We reported an increase in TNF-alpha levels, and it is possible that in the present study the MMP expression by HSCs was regulated in a TNF-alpha specific pattern. TNF-alpha and HSCs might be involved in the initial matrix breakdown essential for the early repair reactions that occur after the liver injury.
The ischemic/reperfusion injury is associated with an early MMPs secretion (Cursio et al. 2002); MMP-2 and MMP-9 seem to be particularly relevant in the liver, since they are critically involved in the degradation of key components of the basement membrane such as collagen IV and fibronectin, which are largely represented in the space of Disse (Khandoga et al. 2006). Therefore, increased activity of these gelatinases may cause hepatocellular damage, with alterations of the sinusoidal lining and remodeling of the stromal structure. The present study demonstrates that a moderate acute hepatic I/R injury is able to increase MMPs activity not only in the ischemic region, as previously reported (Hamada et al. 2008), but also in the non-ischemic lobe. Moreover, this event is associated with several histological signs of interstitial and cellular damage. During the standard experimental model of normothermic hepatic I/R, only the left and the median lobe are occluded by vascular clamp, whereas the remaining lobes are usually considered the control lobes (Matsui et al. 2005) because they retain an intact portal and artery blood supply, at a time when the venous outflow prevents intestinal venous hypertension. Our results demonstrate that a significant modification of MMPs activity also occurs in the non-ischemic lobe, possibly due to the release of TNF-alpha that takes place during the early phase of reperfusion, as recently reported (Duenschede et al. 2007).
Interestingly, previous studies reported that changes in endothelin receptors (ETs) identically occur in both the ischemic and non-ischemic lobes: ET(A) receptors were significantly decreased whereas ET(B) receptors were increased (Yokoyama et al. 2000). To support the results of this study, it has been recently reported that ET-1 is likely to promote an unfavorable matrix turnover through the stimulation of collagen 1, TGFβ-1, and MMP-2 in hepatic stellate cells (Koda et al. 2006). Furthermore, molecular evidence suggested that ET-1 and MMP-1 actively participate to the same molecular pathway in the pathogenesis of inflammation-associated preterm birth (Wang et al. 2010).
Our data indicate the I/R injury also affects the non-ischemic lobe, as clearly shown by the analysis of MMP levels. Although the involved mechanism is still unclear, the upregulation of MMP-9 is directly connected with the I/R damage (Ma et al. 2010) and is involved in the modulation of cell activation through altering cytokine levels such as TNF-alpha (Atkinson and Senior 2003). These considerations should be taken into account when comparing the ischemic and the non-ischemic lobe. It is important to note that the present results demonstrate that biochemical and histological changes do occur also in the non-ischemic lobe and that they are traceable very early during reperfusion after a brief ischemia.
The goal of the present work was the comparison of ischemic lobe with the non-ischemic one in a model of partial ischemia; among the ischemic lobes we decided to use the left lobe because it has only one primary portal branch, while the median lobe appears to have two portal branches (Malarkey et al. 2005). The left lobe appears a more defined unit, as does the right. Comparing the results using both sham and I/R rat, we found new information regarding the hepatic lobe heterogeneity. Unlike in the reports of Uehara et al. (2004), the functional lobe heterogeneity has been evaluated in the CCl4 toxicity induced to the whole liver. The functional differences between the right and median lobes have not been evaluate in case of I/R injury but will be considered in the future experiments.
In conclusion, our data reported, for the first time, that MMPs activation in the ischemic lobe is associated with a parallel MMP upregulation in the non-ischemic lobe; this event is probably TNF-alpha-mediated. These results support the hypothesis that a direct connection exists between the events that happen in the damaged lobe and in the non-ischemic liver. In addition, the previously described lobe-specific heterogeneity has been confirmed and supported by the present study. While sampling the liver, the hepatic lobar functional heterogeneity should be considered, and the comparison of the heterogeneous hepatic lobes should be avoided.
Footnotes
Acknowledgments
We thank Mr. Gaetano Viani for skillful technical assistance, Dr. Enrico Scoglio for technical support during the HPLC assay, and Mrs. Nicoletta Breda for editing assistance.
Giuseppina Palladini and Andrea Ferrigno contributed equally to this article.
This work was funded by F.A.R. 2009–2010, University of Pavia. E.B. was partially funded by the Superpig Program, Fund for Promoting Institutional Agreements, Regione Lombardia, Italy.
