Abstract
Connexin 32 (Cx32) is a major gap junction protein in the liver. The authors previously demonstrated that transgenic rats carrying a dominant negative mutant of Cx32 (Cx32ΔTg) have much decreased capacity for gap junctional intercellular communication (GJIC) and increased susceptibility to diethylnitrosamine (DEN)–induced hepatocarcinogenesis as compared to littermate wild-type (wt) rats. To evaluate the age-dependent susceptibility to DEN-induced hepatocarcinogenesis and alteration of GJIC function, male Cx32ΔTg and wt rats at 10, 30, or 85 weeks old were given a single intraperitoneal administration of DEN (40 mg/rat) and sacrificed 12 weeks later. The number and area of glutathione S-transferase placental form (GST-P)–positive preneoplastic foci were significantly increased in the liver of 10- and 30-wk-old Cx32ΔTg rats compared with age-matched wt. However, in the 85-wk-old rats, both Cx32ΔTg and wt rats had similarly large number and area of GST-P-positive foci, and the difference was not significant. Interestingly, function of hepatic GJIC was reduced and protein and mRNA expression of Cx32 were decreased with aging in wt rats. These results suggest that a decline of hepatic intercellular communication through gap junction results in increased susceptibility to DEN-induced hepatocarcinogenesis in aged rats.
Introduction
Cancer incidence rises with age in humans and mice. From this phenomenon, it seems advancing age is one of the most important risk factors for malignancy (Peto et al. 1975; Gloeckler Ries et al. 2003; DePinho 2000). Several factors have been implicated. For example, molecular, cellular, and physiological changes with age may lead to decline in organismal fitness, increase of susceptibility to carcinogen, and accumulation of critical DNA alterations for carcinogenesis (Balducci and Ershler 2005). However, there is little experimental evidence to establish the link between aging and carcinogenesis.
Gap junctions are a particular type of cell junction that specializes in the direct exchange of small molecules, such as ions, second messengers, and cellular metabolites between contacting cells (Bennett et al. 1991; Gilula et al. 1972; Loewenstein 1981). They are composed of 2 connexons in adjacent cells, which in turn are formed by 6 connexin proteins (Evans and Martin 2002; Vinken et al. 2008). Intercellular communication through gap junction is essential for maintenance of tissue environment, the control of cell growth, and differentiation (Loewenstein 1981). In the liver, connexin 32 (Cx32) is a major gap junctional protein (Paul 1986). With regard to hepatocarcinogenesis, Cx32 protein levels decrease during chemical hepatocarcinogenesis in the rat liver, and hexachlorobenzen-induced liver tumor in female rats is associated with decreased gap junctional intercellular communication (GJIC) caused by dysregulation of Cx32 transcription (Krutovskikh et al. 1995, 1991; Plante et al. 2002, 2007). The down-regulation of Cx32 is also observed during progression of human chronic liver disease, including cirrhosis and hepatocellular carcinomas (Nakashima et al. 2004). In other organs, altered localization of Cx32 from plasma membrane to cytoplasm plays a role in formation of gastric adenomas and adenocarcinomas in human and mice (Jee et al. 2011), and Cx32 regulates cell proliferation, invasion, and metastasis of renal cell carcinoma via inhibition of hypoxic adaptation (Sato et al. 2007, 2008). We had previously established transgenic rats carrying a dominant negative mutant of Cx32 under the control of the albumin promoter (Cx32ΔTg), resulting in decreased capacity for and increased susceptibility to diethylnitrosamine (DEN)–induced hepatocarcinogenesis as compared to littermate wild-type (wt) Sprague-Dawley (SD) rats (Asamoto et al. 2004; Hokaiwado et al. 2005). These findings indicate that GJIC composed of Cx32 have essential functions in protection against hepatocarcinogenesis. A few papers reported that the function of gap junctions was altered with age in the nervous system (Cotrina et al. 2001; Huang et al. 2006). However, the function of GJIC in the aged organ, particularly liver, has not yet been reported. Moreover, the relationship between age-promoted cancer development and GJIC is unclear.
In this study, we examined how aging affects susceptibility to DEN-induced hepatocarcinogenesis by using the Cx32ΔTg rats and their littermate wt rats. Furthermore, alteration of GJIC capacity and Cx32 expression with aging in wt rats were confirmed by in vivo dye loading assay and immunohistochemistry.
Materials and Methods
Production and Screening of the Transgenic Rats
The establishment, production, and screening of Cx32ΔTg rats carrying mutated Cx32 gene were as previously described in detail (Asamoto et al. 2004). Animals having high copy numbers (~50 copies) of the transgene were used in this study. Cx32ΔTg male rats were produced by mating heterozygous Cx32ΔTg males with wt SD females (Japan SLC, Shizuoka, Japan). Rats were maintained in plastic cages on hardwood chips, in an air-conditioned, specific pathogen-free (SPF) animal room at 22 ± 2°C and 50% humidity with 12 h/12 h light-dark cycle. All animal experiments were performed under protocols approved by the Institutional Animal Care and Use Committee of Nagoya City University School of Medical Sciences.
Animal Treatments
Cx32ΔTg rats and littermate wt rats at 10, 30, and 85 weeks of age were given a single i.p. injection of 40 mg DEN (Tokyo kasei Kogyo Co. Ltd, Tokyo, Japan), dissolved in saline (Wako, Osaka, Japan). All rats were sacrificed at the 12th week following injection. The livers were immediately excised, weighed, and cut into slices of 2–3 mm thickness, then fixed in 10% buffered formalin, paraffin-embedded, and routinely processed for histological evaluation (2–3 µm thickness). Sections were routinely stained with hematoxylin and eosin.
Identification of Preneoplastic Foci
Deparaffinized slide sections of liver tissues that were fixed with 10% formalin were incubated with 1:4,000 diluted anti-glutathione S-transferase placental form (GST-P) polyclonal antibody (MBL, Nagoya, Japan). Antibody binding was visualized by a conventional immunostaining method using an auto-immunostaining apparatus (VENTANA HX SYSTEM, VENTANA Japan, Tokyo, Japan). The average number and area of GST-P-positive foci > 0.2 mm in diameter, and total area of the liver section were measured with an image analyzer (IPAP; Sumica Technos Co., Osaka, Japan).
Measurement of GJIC Capacity in Liver In Vivo
This procedure was performed according to the method of Sai et al. (2000). Briefly, liver samples were obtained from 10-, 40-, 70-, 100-wk-old wt and 100-wk-old Cx32ΔTg rats (4 rats per group). Lucifer Yellow (LY), a stain that can pass through the gap junction channel, and rhodamine-dextran (RhD), which does not transfer through the channel, were obtained from SIGMA-Aldrich Corp (Dt. Louis, MO) and used for measurement of GJIC capability. Five-mm-thick liver slices were cut, and 3 to 4 incisions (1 mm depth) were made with a blade. A mixture of fluorescent dyes containing 0.05% LY and 0.05% RhD in PBS were added drop-wise onto the liver slices. After 3 minutes, the slices were washed 3 times with PBS, then embedded and frozen in the Tissue-Tek Optimal cutting temperature (OCT) compound 4583 (Sakura Finetechnical Co. Ltd., Tokyo, Japan). Frozen sections 6 µm thick were made and photographed by fluorescence microscope. Spread of the dye was measured using an image analyzer (IPAP-WIN, Sumika Technos Co.). The adjacent liver slices were used for immunohistochemical staining for Cx32, Western blotting, and quantitative RT-PCR.
Immunohistochemcal Staining for Cx32
Detailed methods for fluorescence immunohistochemistry are as described previously (Asamoto et al. 2004). Frozen sections were cut to 5 µm thickness and fixed in cold acetone and 10% buffered formalin. A polyclonal rabbit antibody against Cx32 (INVITROGEN, Carlsbad, CA) was used with biotin-conjugated anti-rabbit IgG and TRITC-labeled streptavidin (INVITROGEN) to visualize the endogenous proteins under fluorescence microscopy (Olympus AX-70, Tokyo, Japan).
Western Blotting
Liver tissues were prepared as described previously (Asamoto et al. 2004). Briefly, 10 µg samples were loaded par lane, separated in 12% acrylamide gels, and electroblotted onto nitrocellulose membranes (Hybond-ECL, GE Healthcare UK Ltd., Buckinghamshire, UK). Cx32 expression levels were assessed using the same antibodies as applied for immunohistochemical staining. β-actin expression was evaluated with a monoclonal anti-β-actin, AC-74 (SIGMA-Aldrich), to confirm equal protein loading.
Quantitative RT-PCR Analysis of Cx32
One microgram of RNA was converted to cDNA with avian myoblastosis virus (AVM) reverse transcriptase (Takara, Otsu, Japan) in a 20 µl reaction mixture. Aliquots of 2 µl of cDNA samples were subjected to quantitative PCR in 20 µl using SYBR Premix ExTaq (Takara) in a light Cycler apparatus (Roche Diagnostics, Mannheim, Germany). The primers used were 5′-AACGTGGCGGAGGTGGTGTA-3′ and 5′-GGGAAGGTTTGATGGAGTAAT-3′ for Cx32 and 5′-GCATCCTGCACCACCAACTG-3′ and 5′-GCCTGCTTCACCACCTTCTT-3′ for GAPDH. Initial denaturation was at 95°C for 60 sec, followed by 40 cycles with denaturation at 95°C for 5 sec, annealing at 55°C for 15 sec, and elongation at 72°C for 30 sec. GAPDH mRNA levels were used as internal controls, and the values for Cx32 were normalized against GAPDH and compared.
Results
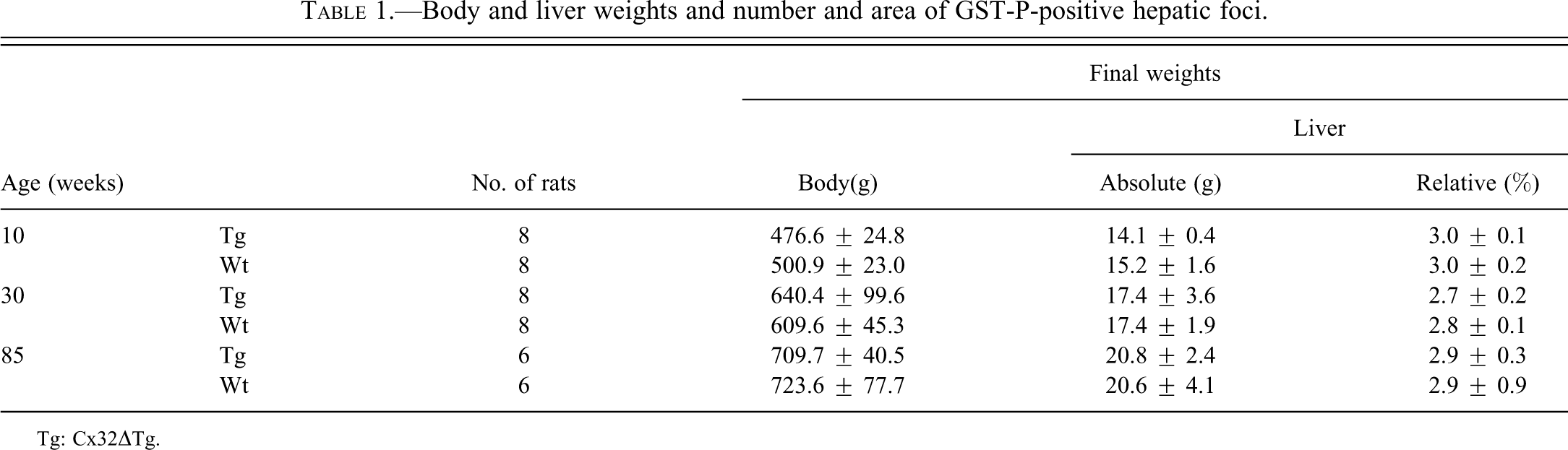
At the start of the carcinogenic test, body weights of 10-wk-old Cx32ΔTg and wt rats were 360.6 ± 18.7 g and 391.9 ± 14.8 g, 30-wk-old were 597.9 ± 79.2 g and 580.4 ± 37.9 g, and 85-wk-old were 722.0 ± 43.5 g and 736.0 ± 50.0 g, respectively. There was no significant difference in final body and liver weights between each group of age-matched Cx32ΔTg and wt rats (Table 1). DEN exposure induced more numbers and larger preneoplastic GST-P-positive foci in the livers of the 10- and 30-wk-old Cx32ΔTg rats as compared to wt (Figure 1 and Figure 2). These results suggest that knockout of Cx32 increased the susceptibility to DEN-induced hepatocarcinogenesis. On the other hand, both 85-wk-old Cx32ΔTg and wt rats showed a high susceptibility to DEN, and there was no significant difference in the number and area of GST-P-positive foci between Cx32ΔTg and wt rats (number: p = .1, area: p = .7) (Figure 2). By age comparison of wt rats, both number and area of GST-P-positive foci were significantly decreased in 30-wk-old rats (number: p = .003, area: p = .007), and increased in 85-wk-old animals (number: p = .0004, area: p = .01) as compared to those in the 10-wk-olds. In Cx32ΔTg rats, the area of GST-P-positive foci tended to increase with age, but there were no significant differences (10- vs. 30-wk-old; p = .5, 10- vs. 85-wk-old; p = .3). The numbers of foci decreased with age, and this tendency was significant between 10- and 30-wk-old Cx32ΔTg rats (10- vs. 30-wk-old; p = .02, 10- vs. 85-wk-old; p = .08). This decreasing trend in number of GST-P-positive foci in 30-wk-old rats of both genotypes may be related to the lower dose of DEN (on a mg/kg basis) as compared to 10-wk-old animals. In 85-wk-old rats, significant increases in number and area of GST-P-positive foci were shown in wt; however, this trend was not observed in the Cx32ΔTg. These results suggest that older wt rats are more sensitive to DEN-induced hepatocarcinogenesis, and depression of Cx32-associating GJIC function is assumed to contribute to this phenomenon. Therefore, we examined alteration of GJIC function with age in the livers of wt rats.
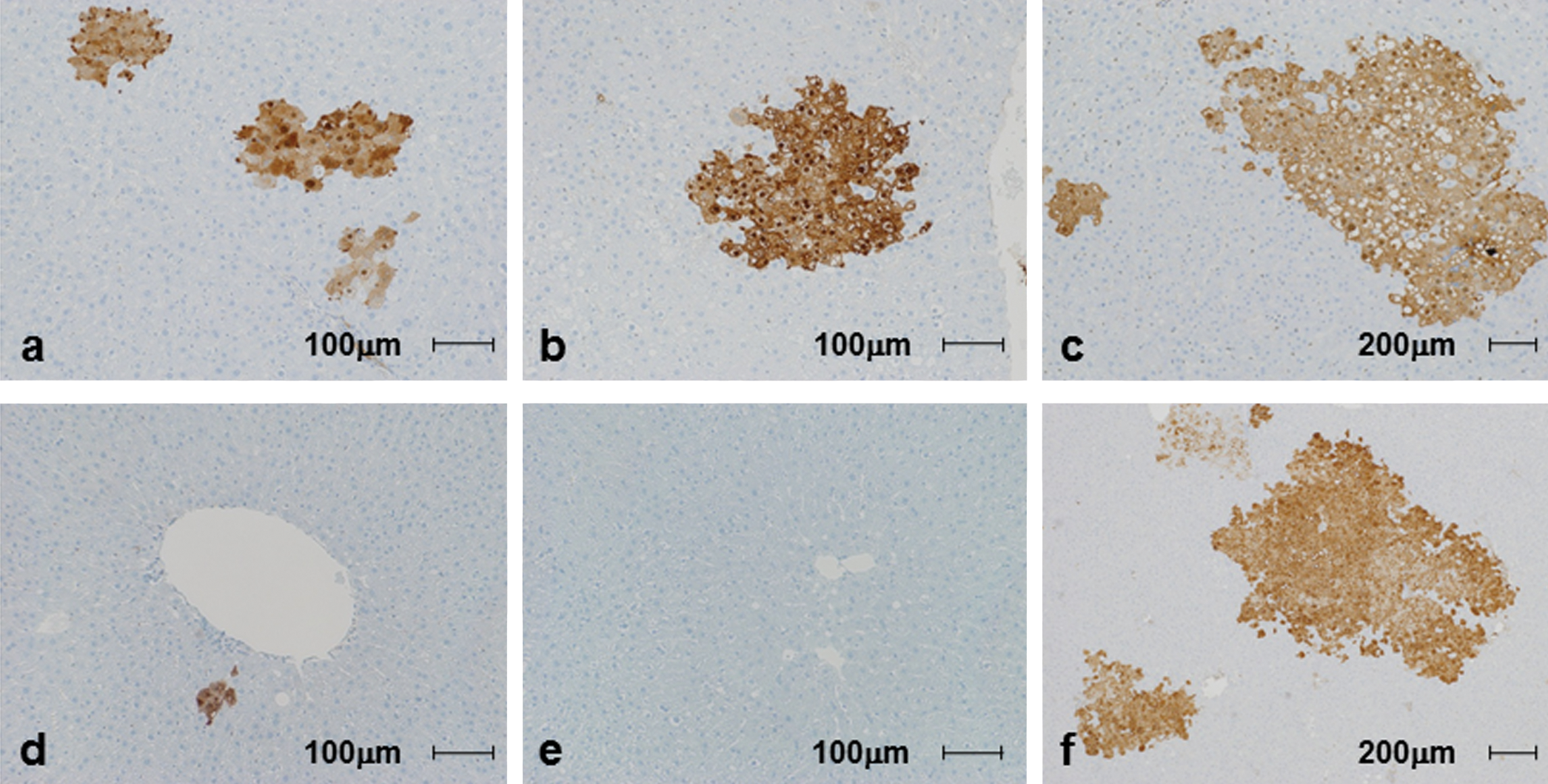
GST-P-positive preneolpastic foci induced by diethylnitrosamine in Cx32ΔTg and wt rats at various ages. a, d: 10-wk-old Cx32ΔTg (a) and littermate wt rats (d). b, e: 30-wk-old Cx32ΔTg (b) and littermate wt rats (e). c, f: 85-wk-old Cx32ΔTg (c) and littermate wt rats (f).
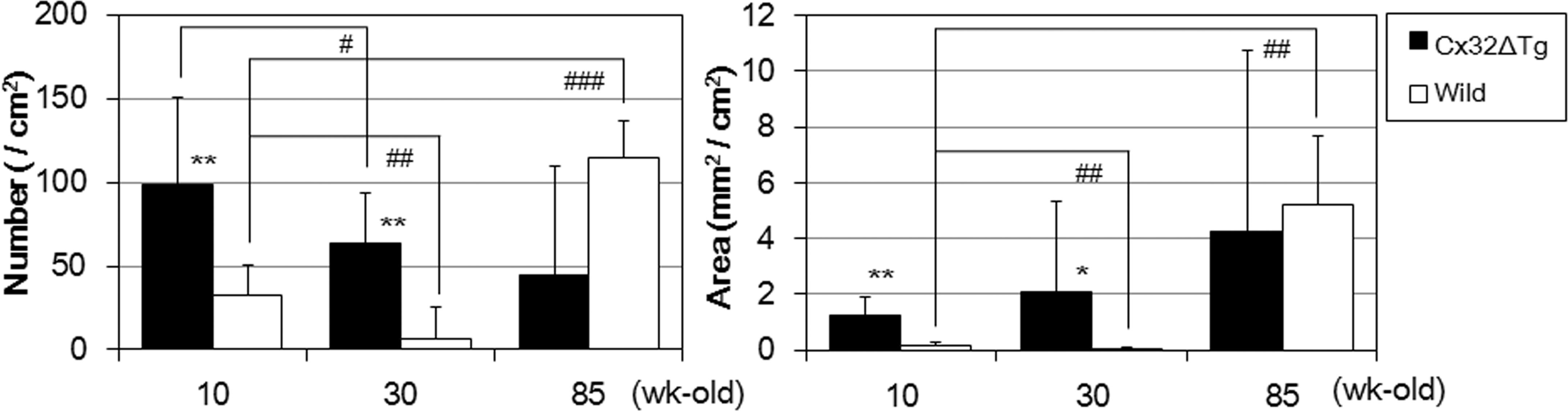
Number and area of GST-P positive hepatic foci induced by DEN. Number is represented in the left graph and area in the right. Statistical significance as compared to age-matched wt rats: *p < .05, **p < .001. Statistical significance as compared to 10-wk-old, genetically-matched rats: # p < .05, ## p < .01, ### p < .0005.
Body and liver weights and number and area of GST-P-positive hepatic foci.
Tg: Cx32ΔTg.
The scrape dye-loading method in vivo revealed that transfer of LY was significantly reduced in the liver of aged wt rats (40-, 70-, and 100-wk-old) as compared to the 10-wk-old rats (Figure 3A and 3B). Especially in 100-wk-old wt rats, LY spread was reduced to about 60% of the 10-wk-olds, this was not significantly different from that in age-matched Cx32ΔTg rats, p = .4 (Figure 3B).
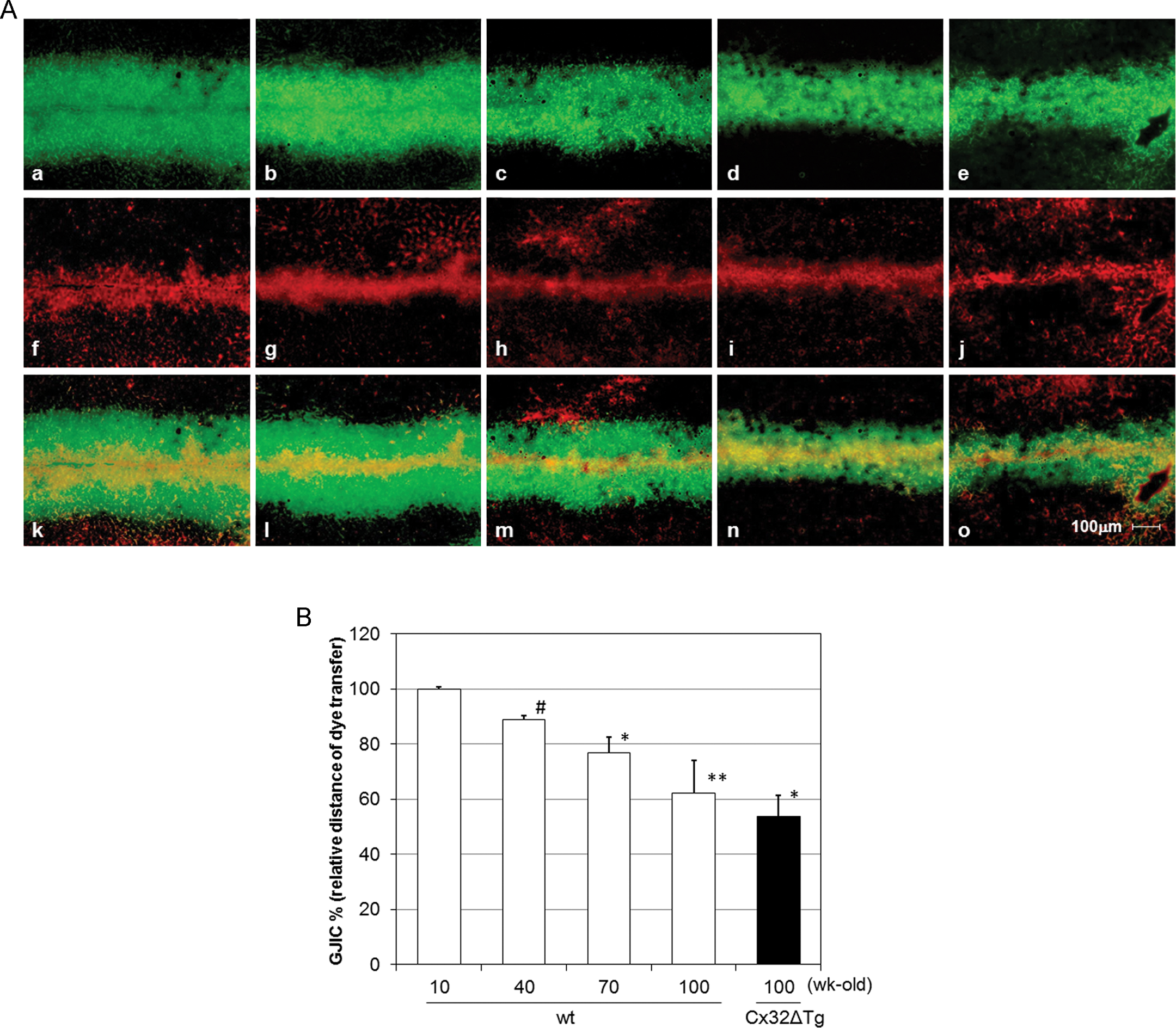
Gap junctional intercellular communication in the rat livers of various ages revealed by in vivo dye loading assay. (A) a–e: Spread of LY through gap junction; f–j: RhD used for non-gap junctional signaling; k–o: merged pictures of LY and RhD; f and k: 10-wk-old wt; b, g and l: 40-wk-old wt; c, h and m: 70-wk-old wt; d, i and n: 100-wk-old wt; e, j and o: 100-wk-old Cx32ΔTg rats. (B) Relative distance of LY spread (%). The values represent the means ± SD of data from four rats. *p < .05, **p < .005 and # p < .001 versus 10-wk-old wt group (Student’s t-test).
To explore the manner of reduction of GJIC by aging in wt rats, we examined the expression level and localization of Cx32 protein in aged livers. Immunohistochemical staining revealed that expression of Cx32 protein was located similarly in the plasma membrane of hepatocytes at every age of wt rats. However, reduction of Cx32 plaques and of its density with aging in wt rats is evident as seen in Cx32ΔTg rats (Figure 4). Western blotting analysis indicated that expression of Cx32 protein was decreased in the liver of 100-wk-old wt rats as compared to those of the 10-wk-olds (Figure 5A). Therefore, we confirmed the expression level of Cx32 mRNA by quantitative RT-PCR, and we showed that Cx32 transcript level was lower in 100-wk-old wt rats than in 10-wk-olds (Figure 4B; Figure 5B).
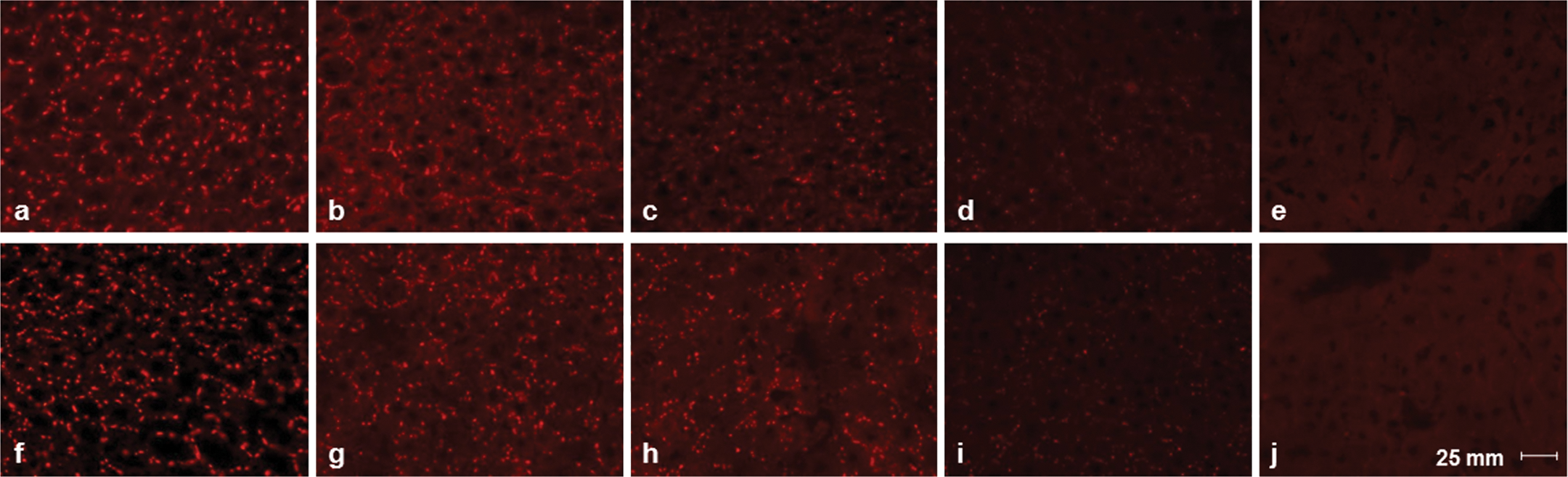
Immunofluorescence staining using Cx32 antibody. a, f: 10-wk-old wt; b, g: 40-wk-old wt; c, h: 70-wk-old wt; d, i: 100-wk-old wt; e, j: 100-wk-old Cx32ΔTg rats. Images of each age group were obtained from separate rats.
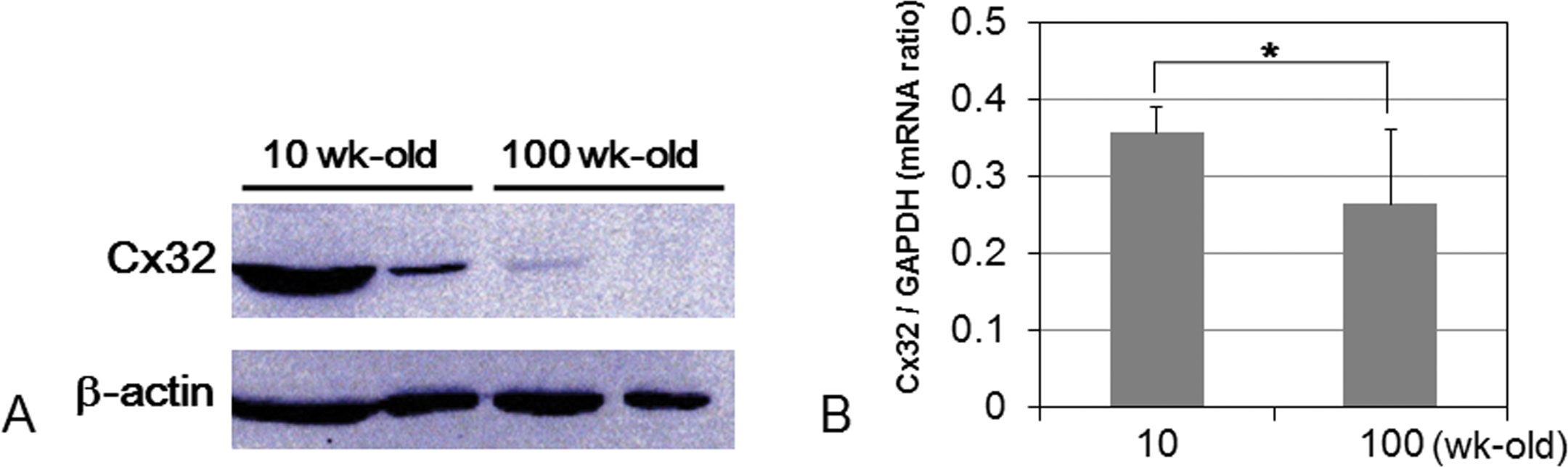
Protein and mRNA expression level of Cx32. (A) Western blotting for Cx32 protein, and (B) quantitative RT-PCR analysis of Cx32. *p < .05 compared with 10-wk-old group (Student’s t-test).
Discussion
In the present study, we examined alteration of susceptibility to DEN-induced hepatocarcinogenesis by aging in rats. Aging is a critical factor for carcinogenesis. Previous reports suggested that susceptibility to carcinogens in old animals is elevated as compared to those in young ones (Fukushima et al. 1987; Pasciu et al. 2006). In contrast, the reverse has also been reported (Reuber 1976; Hasegawa et al. 1991). In our carcinogenic test, we applied the same dosage (40 mg; this dose corresponds to approximately 100 mg/kg for 10-wk-old rats) to 10-, 30-, and 85-wk-old rats. In the 10- and 30-wk-old rats, the number and area of GST-P-positive foci in the liver were significantly increased in the Cx32ΔTg rats as compared to wt (Figure 1 and Figure 2). Similar findings were observed in our previous study in which Cx32ΔTg rats demonstrated a high susceptibility to DEN, phenobarbital, and N-nitrosomorpholine (Hokaiwado et al. 2007, 2005). Because liver weights increase proportionally to body weights with age, older animals received a lower dose of DEN in general (Table 1). This confounding factor may be the reason why the number of GST-P-positive foci was significantly fewer in 30-wk-old than 10-wk-old Cx32ΔTg and wt rats. On the other hand, despite a lower dose of DEN, induction of preneoplastic foci increased in 85-wk-old wt rats (Figure 1 and Figure 2). These results suggest that aging promotes hepatocarcinogenesis in wt rats.
In humans, most hepatocellular carcinomas arise from chronic liver disease, for example, chronic hepatitis and cirrhosis. In addition, expression of Cx32 is gradually decreased during the progression of hepatocarcinogenesis not only in rodents but also in human (Krutovskikh et al. 1995, 1991; Nakashima et al. 2004). These reports suggest that GJIC capacity is crucial for regulation of liver cancer initiation and progression. In the present study, elevation of carcinogenic susceptibility to DEN with age in wt rats was not shown in Cx32ΔTg (Figure 2). Therefore, we assumed that a high susceptibility to chemical hepatocarcinogenesis in old rats is caused by reduction of GJIC.
As expected, we showed here that GJIC capacity gradually reduced with age in wt rats (Figure 3A and 3B). Furthermore, the levels of GJIC in 100-wk-old wt rats were almost the same as those in age-matched Cx32ΔTg rats. With regard to the high sensitivity to DEN in both Cx32ΔTg and wt rats at an older age such as 85-wk-old, it is reasonable to think that GJIC capacity and development of hepatocarcinogenesis are inversely associated. Cx32, which is a major protein that forms gap junctions in the liver, was immunohistochemically localized in the plasma membrane of hepatocytes, and the amount of plaques was decreased by age in wt rats (Figure 4). Moreover Western blotting and quantitative RT-PCR revealed that both protein and mRNA expression of Cx32 were reduced in old wt rats as compared to younger ones (Figure 5A and 5B). These results suggest that decreased levels of Cx32 and GJIC in older wt rats may be similar to the effective levels of Cx32 in Cx32ΔTg rats, resulting in the lack of a difference between the 85-wk-old Cx32ΔTg and wt rats. Moreover, low effective levels of Cx32 lead to an absence of significant increase in GST-P-positive foci with age in Cx32ΔTg rats.
Another gap junctional protein in the liver, Cx26, was also localized in the membrane at the periportal zone of the lobules, and we found that there was no significant difference in expression with age (data not shown). Cx26 is preferentially expressed in the periportal acinar area, and the relative ratio of Cx26 to Cx32 protein in rat liver is 1:10 (Nicholson et al. 1987; Sakamoto et al. 1992; Vinken et al. 2008). In addition, downregulation of Cx32 expression occurs earlier than Cx26 during rat hepatocarcinogenesis (Sakamoto et al. 1992). These findings suggest that decrease of Cx32 expression is essential for GJIC and early hepatocarcinogenesis, rather than Cx26. These results also indicate that reduction of Cx32 expression contributes to deterioration in GJIC function and cancer development in aged liver. To our knowledge, this is the first report to confirm GJIC capability and its effect to carcinogenesis in aged liver. However, further studies are necessary to understand how GJIC signaling acts as a tumor suppressor and to apply this knowledge for prevention of hepatocarcinogenesis.
In summary, the present study clearly shows that GJIC function is decreased with aging in rats, and it may cause promotion of hepatocarcinogenesis in older rats. In addition, Cx32ΔTg may be a good model to study roles of gap junctions in aged liver.
Footnotes
This work was supported in part by a Grant-Aid from the Ministry of Education, Culture, Sports Science and Technology of Japan (Grant Number 23790449) and by a Grant-in-Aid for Research at Nagoya City University.
