Abstract
Gingival lesions of squamous hyperplasia, cystic keratinizing hyperplasia (CKH), and squamous cell carcinoma (SCC) can be induced in rats treated by chronic gavage with 10–100 mg/kg 3,3′,4,4′-tetrachloroazobenzene. We evaluated gingival squamous hyperplasia (GSH), CKH, and SCC for the immunohistochemical pattern of expression of carcinogenesis-associated markers. The 3 types of lesions and controls were stained with proliferation markers (proliferating cell nuclear antigen [PCNA] and cyclin-D1), tumor-suppressor markers (β-catenin and mammary serine protease inhibitor [maspin]) and stroma-related markers (α-smooth muscle actin [SMA] and osteonectin/SPARC). The lesions had common immunohistochemical characteristics that differed in their expression patterns among the various diagnoses. PCNA and cyclin-D1 expression was higher in GSH, CKH, and SCC than in controls. The normal membranous expression of β-catenin was lower in GSH, and almost absent in CKH and SCC. Maspin expression was similar in GSH and controls, whereas both CKH and SCC showed decreased expression. SMA and/or osteonectin/SPARC were seen in stromal cells in CKH and SCC. Collectively, there appears to be a progression from hyperplastic and cystic lesions toward malignancy based on the morphological changes, supported by the expression of carcinogenesis-associated proteins. The exact sequence of events leading to SCC remains to be defined in a time-dependent manner.
Introduction
The U.S. National Toxicology Program (NTP) has recently performed a series of two-year bioassays in Harlan Sprague-Dawley rats in order to evaluate the pathology and carcinogenicity induced by chronic gavage exposure to dioxin and dioxin-like compounds (DLCs). These assays revealed an array of neoplastic and non-neoplastic effects, including, for example, cholangiocarcinoma and/or hepatocellular adenoma of the liver and cystic keratinizing epithelioma of the lungs (Brix et al. 2004; Nyska et al. 2004; Tani et al. 2004; Walker et al. 2006; Yoshizawa et al. 2005). In addition, oral lesions, including gingival squamous hyperplasia (GSH) and/or squamous cell carcinoma (SCC) were increased in treated rats in all studies except polychlorinated biphenyl (PCB) 153 (Yoshizawa et al. 2005). Oral tumors in rats have also been reported with other chemicals examined by the NTP, thus highlighting the oral mucosa as a prominent target organ in toxicity studies (e.g., benzene, chloroprene, and others; Yoshizawa et al. 2005).
The NTP also conducted a two-year chronic gavage study in rats using 3,3′,4,4′-tetrachloroazobenzene (TCAB), a DLC formed as a byproduct during the manufacture of 3,4-dichloroaniline or its herbicidal derivatives, and following 3,4-dichloroaniline photolysis and biolysis (Hill et al. 1981; Poland et al. 1976; Ramot et al. 2009; Singh et al. 2010). Humans can be exposed to TCAB either as occupational exposure during the manufacture of 3,4-dichloroaniline or application of herbicides containing TCAB, or as non-occupational exposure during consumption of food contaminated with TCAB (Ramot et al. 2009; Singh et al. 2010).
The molecular factors involved in oral carcinogenesis are extensively evaluated and reviewed by Choi and Myers (2008). In the present study, we intended to employ a panel of markers known to operate within the epithelial and subjacent stromal cells. The former included proliferating cell nuclear antigen (PCNA), which is important for both DNA synthesis and DNA repair (Kato et al. 2011; Maga and Hubscher 2003); cyclin D1, which controls the cellular mitotic activity (Diehl 2002; Hall and Peters 1996); β-catenin, which in normal conditions is located at the cell membrane and acts as a key factor in the intercellular adherence mechanism, whereas in pathological circumstances it is found in a nuclear location and acts as a transcription factor for a number of pro-tumorogenic genes (Daa et al. 2004; Oyama et al. 1994; Pollack et al. 1997; Shiozaki et al. 1996; Zhurinsky et al. 2000); and mammary serine inhibitory protein (maspin), which is considered as a tumor suppressor factor (Bello et al. 2011). The stroma-related markers included α-smooth muscle actin (α-SMA), which is expressed in cells with a myofibroblastic phenotype (cancer-associated fibroblasts) that are involved in tumor proliferation and invasion (Vered et al. 2007; Vered et al. 2010); and osteonectin/cystine-rich acidic secreted protein (SPARC), which is beneficial to tumor cells in terms of angiogenesis, proliferation, and migration (Chlenski and Cohn 2010; Podhajcer et al. 2008).
Therefore, we aimed to use this TCAB model to immunohistochemically characterize, for the first time, the different types of lesions diagnosed in the gingiva of the experimental rats.
Materials and Methods
Chemical
3,3′,4,4′-Tetrachloroazobenzene (CAS No. 14047-09-7) was obtained from AccuStandard, Inc. (New Haven, CT) in one lot. 3,3′,4,4′-Tetrachloroazobenzene was identified by infrared and proton nuclear magnetic resonance spectroscopy, by gas chromatography (GC) coupled with mass spectrometry, and by melting point analysis. The purity of the TCAB was determined by Karl Fischer titration to determine moisture content; elemental analysis for carbon, hydrogen, nitrogen, and chlorine; and GC with flame ionization detection, which indicated purity of 99.8% or greater. Purity of TCAB was analyzed several times during the study; no change was observed over the duration of the study. Dose formulations were prepared for administration by gavage by mixing TCAB in a corn oil vehicle containing 1% USP-grade acetone; they were prepared fresh approximately once a month and were shown to be stable during that period of time. Pre- and post-administration analyses were performed using a GC assay with electron capture detection. All analyzed dose formulations were within 10% of the target concentrations.
Animals and Housing
The studies were conducted in the AAALAC-accredited facility of Battelle Columbus Laboratories (Columbus, OH). Animal use was in accordance with the United States Public Health Service policy on humane care and use of laboratory animals and the Guide for the Care and Use of Laboratory Animals (National Research Council 1996). In addition, these studies were conducted in compliance with the United States Food and Drug Administration (FDA) Good Laboratory Practice Regulations (FDA 1987). Animals were male and female Harlan Sprague-Dawley rats (Harlan Sprague-Dawley, Inc., Indianapolis, IN); they underwent health screening during a quarantine period of thirteen (males) or fourteen (females) days and were released for study when about five weeks old. They were randomly assigned to their respective experimental groups and permanently identified by tail tattoo. Male rats were housed three per cage, and female rats were housed five per cage; all were kept in solid-bottom polycarbonate cages (Lab Products, Inc., Seaford, DE) suspended on stainless steel racks. Filtered room air was supplied at the rate of at least ten room-changes per hour. The rats were maintained at 72°F ± 3°F, with a relative humidity of 50% ± 15% and a light/dark cycle of twelve hours each. Tap water and feed (irradiated NTP-2000 pelleted diet, Zeigler Bros., Inc., Gardeners, PA) were available ad libitum.
Experimental Design
Groups of fifty male and fifty female rats were administered 10, 30, or 100 mg TCAB/kg by gavage in corn oil and acetone (99:1), five days per week for two years. The total dosing volume was 2.5 mL/kg body weight. The dose levels were selected based on the results from NTP three-month toxicity studies (NTP 2009; van Birgelen et al. 1999). All rats were observed twice daily for morbidity and once a month for clinical findings. Moribund animals were sacrificed and necropsied. Health monitoring via sentinel animals showed no evidence of any significant rodent pathogens.
Pathology
Animals were euthanized by carbon dioxide asphyxiation. Complete necropsies were performed on all animals, whether they died early or were sacrificed at termination
Immunohistochemical Analysis
As the incidence of the treatment-related gingival lesions was relatively comparable between the sexes, we used samples from five control male rats and from twelve high-dose male rats: five different rats that showed GSH, five different rats that showed CKH, and five rats that showed SCC (two different rats, and three that had also either GSH or CKH). All sections were stained for previously characterized immunohistochemical markers for squamous cell proliferation and carcinogenesis (Table 1, http://www.niehs.nih.gov/research/atniehs/labs/lep/path-support/immuno/protocols.cfm). These particular markers were the following: SPARC (Santa Cruz Biotechnology, Santa Cruz, CA), maspin (Santa Cruz Biotechnology), α-SMA (Abcam, Inc., Cambridge, MA), β-catenin (Santa Cruz Biotechnology), cyclin D1 (Cell Marque, Rocklin, CA), and PCNA (Santa Cruz Biotechnology). All the markers were stained manually using the the avidin-biotin complex (ABC) technique except β-catenin, which was performed on the Discovery XT Automated System (Ventana Medical Systems, Tucson, AZ) using the OmniMap anti-Rabbit Polymer Detection Kit. The system’s CC1 solution was used for antigen retrieval, and the primary antibody was incubated at 1:500 for sixty minutes without heat. For the manual stains, formalin-fixed, paraffin-embedded tissue sections were deparaffinized in xylene and rehydrated through graded ethanol. Endogenous peroxidase blocking was then performed by immersing the sections in 3% H2O2 for fifteen minutes.
Staining Protocols for the Immunohistochemical Markers Used in the Current Study
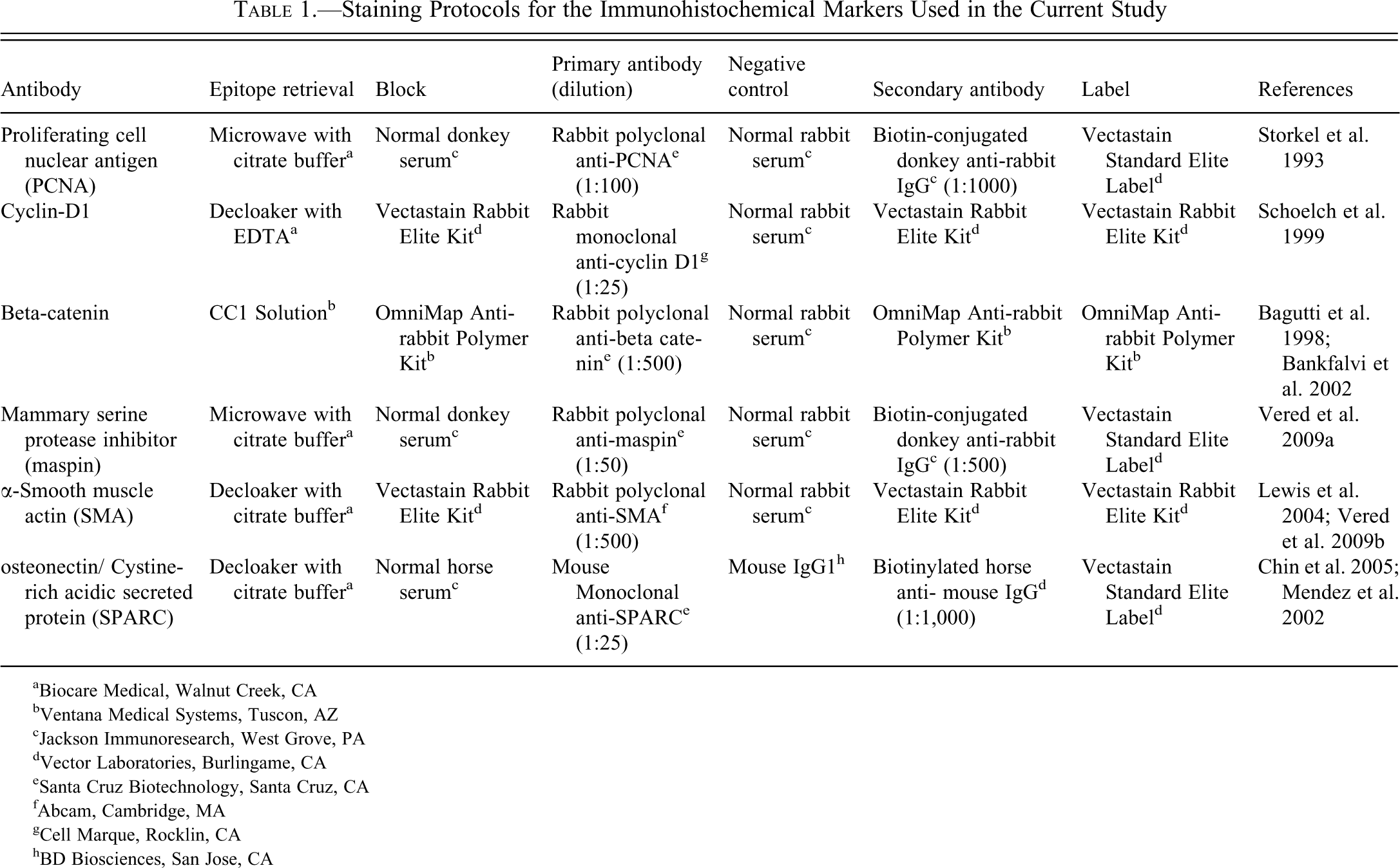
aBiocare Medical, Walnut Creek, CA
bVentana Medical Systems, Tuscon, AZ
cJackson Immunoresearch, West Grove, PA
dVector Laboratories, Burlingame, CA
eSanta Cruz Biotechnology, Santa Cruz, CA
fAbcam, Cambridge, MA
gCell Marque, Rocklin, CA
hBD Biosciences, San Jose, CA
For the SPARC and SMA antibodies, antigen retrieval was carried out using a decloaking chamber and citrate buffer pH 6.0 solution. Cyclin D1 staining also required the decloaker, but EDTA pH 8.5 was used as the retrieval solution. Retrieval for both the maspin and PCNA markers was performed using a microwave with citrate buffer solution. After antigen retrieval, nonspecific sites were blocked using normal sera matching the host species of the secondary antibody. The tissues were then incubated with the various primary antibodies at these specified dilutions: SPARC, 1:25; maspin, 1:50; SMA, 1:500; cyclin D1, 1:25; and PCNA, 1:100. All antibodies were incubated at room temperature for sixty minutes except SMA, which had a thirty-minute incubation. Biotin-conjugated donkey anti-rabbit secondary was applied to the tissues for the PCNA and maspin stains at 1:1,000 and 1:500, respectively, for thirty minutes. A biotinylated horse anti-mouse secondary was also used for the SPARC staining at a 1:500 dilution for thirty minutes. After these secondary incubations, the slides were treated with the Vectastain Elite ABC Standard Kit for thirty minutes. For SMA and cyclin D1, sections were incubated with Vectastain Elite ABC Rabbit IgG Kit (Vector Laboratories, Burlingame, CA). The antigen of interest was visualized using 3-diaminobenzidine (DAB) chromagen. After counterstaining with hematoxylin, all sections were dehydrated through graded ethanol, cleared in xylene, and coverslipped. For negative controls, the antibody was replaced with a nonimmune serum diluted to match the protein concentration of the respective antibody.
Assessment of the Immunostains
Assessment of the expression of the epithelial-related markers within the oral lining epithelium (GSH and control) was performed separately for the basal, suprabasal, and upper layers. Similarly, in the CKH lesions, the cystic epithelium was divided into the basal, suprabasal, and upper layer, where the latter referred to the luminal layers. In regard to SCC, assessment was done in the tumor at the front of invasion versus its more superficial areas.
Staining, assessed according to the extent of the positively stained cells, was classified as (–), no staining; (+), 1%–10% cells positively stained; (++), 11%–50%; and (+++), > 50% positive cells. Proliferating cell nuclear antigen and cyclin D1 were assessed as nuclear stainings. In regard to β-catenin, only membranous staining was considered positive. Maspin was assessed in both the cytoplasmic and nuclear compartments. Expression of α-SMA and SPARC was assessed as cytoplasmic staining in cells adjacent to the oral lining or cystic epithelia or close to the tumor.
Statistical Analysis
The incidences of microscopic lesions were evaluated statistically by the poly-3 test (Bailer and Portier 1988; Portier and Bailer 1989), which makes adjustments for survival differences among groups. Severity scores were compared among dose groups using the Kruskal-Wallis analysis of variance for an overall comparison, and using Mann-Whitney tests to compare each dose group to the control group (Hollander and Wolfe 1973).
Results
Microscopic Evaluation of the Gingivae
The frequencies of the different types of gingival lesions for each dose group of animals are summarized in Table 2. All lesions were confined to nasal section level III.
Incidence and Mean Severity (in parentheses) of Neoplasms and Non-neoplastic Lesions of the Oral Cavity in Rats in the Two-Year Gavage Study of TCAB
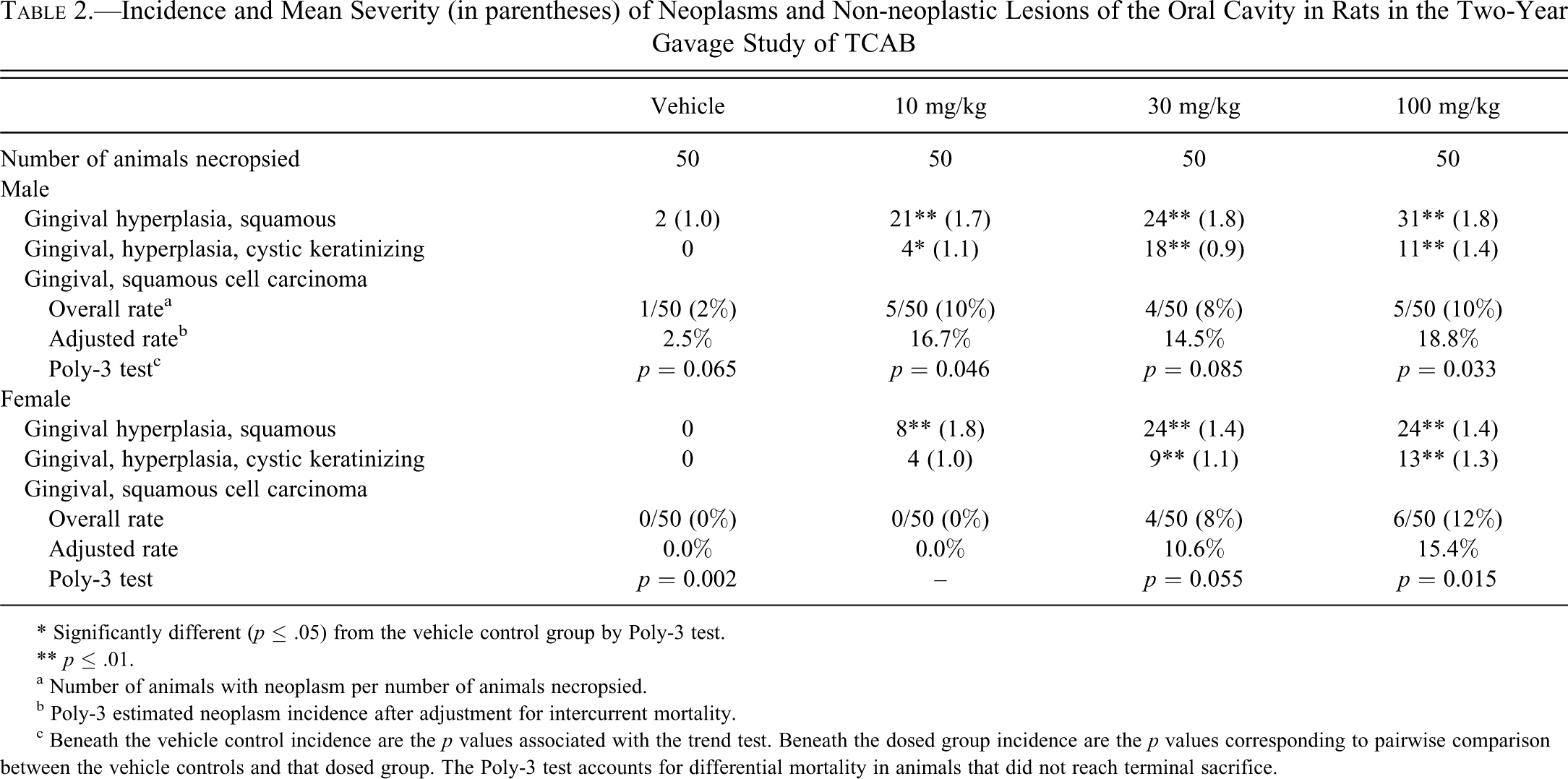
* Significantly different (p ≤ .05) from the vehicle control group by Poly-3 test.
** p ≤ .01.
a Number of animals with neoplasm per number of animals necropsied.
b Poly-3 estimated neoplasm incidence after adjustment for intercurrent mortality.
c Beneath the vehicle control incidence are the p values associated with the trend test. Beneath the dosed group incidence are the p values corresponding to pairwise comparison between the vehicle controls and that dosed group. The Poly-3 test accounts for differential mortality in animals that did not reach terminal sacrifice.
Increased incidence of gingival SCC, single or multiple, was noted in both sexes treated with 30 and 100 mg/kg, and in the males treated with the 10 mg/kg compared to control. The highest incidence was seen in the females treated with the 100 mg/kg compared to control. Generally, a dose-related increased incidence of gingival squamous cell hyperplasia and gingival CKH was noted in all treated groups of both sexes, except for the incidence of CKH in the high-dose males.
The following histopathological descriptions summarize findings from all study groups. Squamous hyperplasia occurred in the stratified squamous epithelium of the gingival oral mucosa adjacent to the molar teeth in nasal section III, and it consisted of varying degrees of thickening of the epithelium, generally with the formation of epithelial rete ridges that extended into the underlying connective tissue. Minimal lesions, located directly adjacent to the molar teeth, consisted of slight thickening of the epithelium. As severity increased, the epithelium became thicker, usually demonstrating formation of multiple papillary projections, accompanied by abundant keratin accumulation and prominent rete ridges. In some of the more severe cases of hyperplasia, the hyperplastic epithelium formed prominent invaginations into the underlying palate, displacing normal tissue, thus somewhat resembling invasive lesions (Figure 1). It was noted in some sections containing the root of a molar tooth (i.e., the periodontal ligament) that the hyperplastic epithelium tended to grow down along the root.
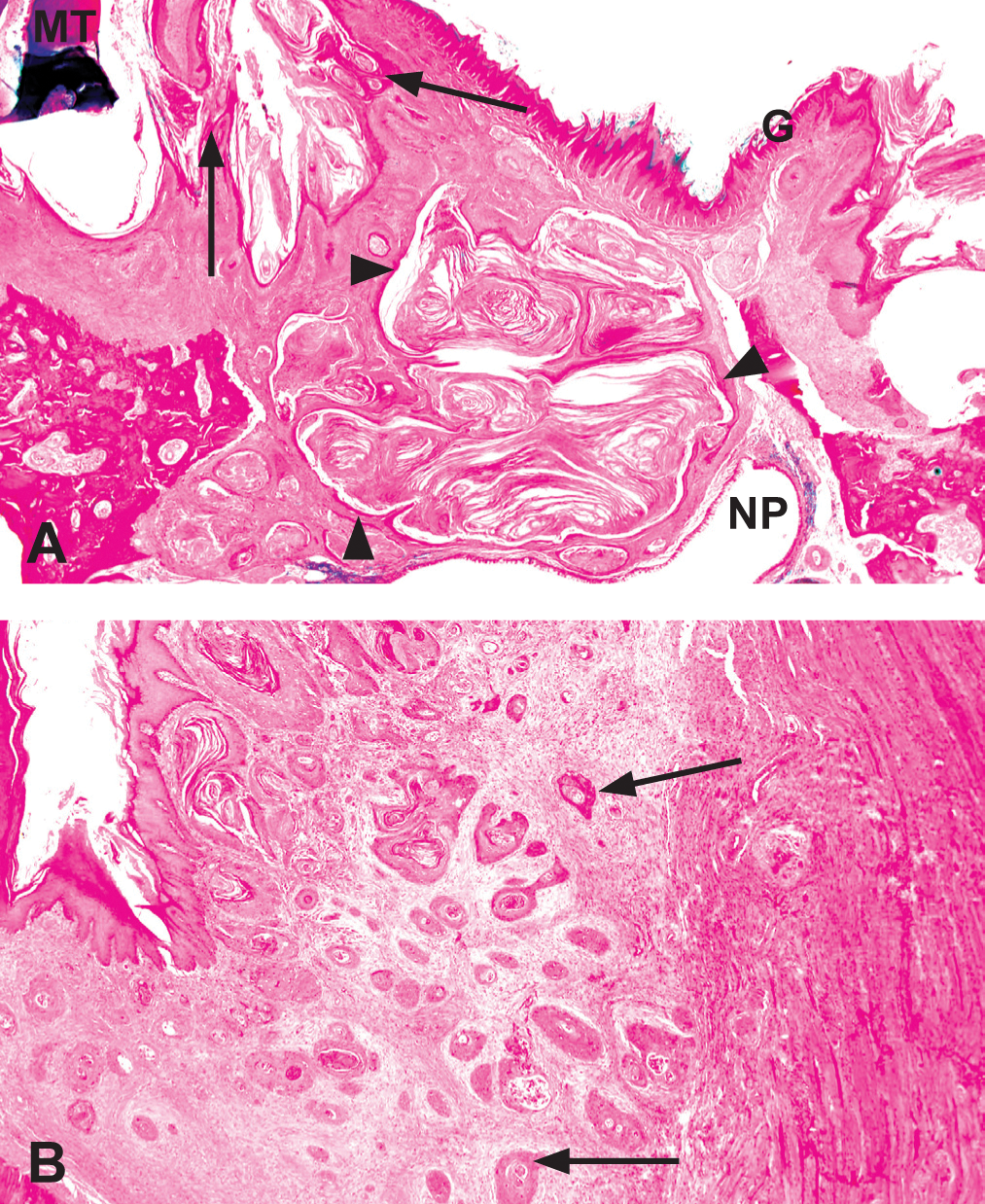
Gingival lesions of 3,3′,4,4′-tetrachloroazobenzene (TCAB)–treated rats. (A) Oral cavity mucosa at nasal section III (at the level of the molars) in a male Harlan Sprague-Dawley rat administered 100 mk/kg TCAB by gavage for two years. Note the presence of gingival squamous hyperplasia (arrows) and gingival cystic keratinizing hyperplasia (arrowheads). Hematoxylin and eosin. G, gingiva; MT, molar tooth; NP, nasopharynx. (B) Squamous cell carcinoma in the oral cavity mucosa at nasal section III (at the level of the molars) in a female Harlan Sprague-Dawley rat administered 100 mk/kg TCAB by gavage for two years. Note the irregular cords and clusters of stratified squamous epithelial cells that invade deeply into the underlying connective tissue (arrows). Hematoxylin and eosin.
Cystic keratinizing hyperplasia also occurred adjacent to the molar teeth in section III. It was characterized by variably sized cavities lined by stratified squamous epithelium and filled with keratin located deep to the oral surface. Minimal lesions consisted of one to a few small, ovoid cavities, lined by a thin layer of epithelium and containing small amounts of keratin, lying within the connective tissue of the palate a short distance below the oral lining epithelium. Some of these lesions occurred in isolation, whereas others occurred in association with diffuse hyperplastic lesions more typical of squamous hyperplasia and appeared to be arising from the more diffuse hyperplasia. As the severity of the cystic lesions increased, the size of the lumina and the depth of penetration into underlying tissues increased. In moderate to severe cases, the cystic structures penetrated deep into surrounding tissues, including into the nasal cavity, and into the soft tissues attached to the maxilla (Figure 1A). The lining of more severe lesions often tended to have areas of irregular thickening and formed multiple, adjacent cystic structures, giving the lesion a multilocular appearance. Generally, a connection could be seen between the cystic structure and the overlying gingival oral mucosa, suggesting that the CKH may have arisen from squamous hyperplasia. In some cases of severe CKH with penetration into the nasal cavity, variably sized islands and keratin pearls of stratified squamous epithelium, embedded within dense fibrous tissue, were seen lying deep to the cystic structure and were considered indicative of invasion. These lesions were diagnosed as SCC.
Squamous cell carcinoma occurred within the oral mucosa adjacent to the molar tooth in nasal section III. It was characterized by irregular cords and clusters of stratified squamous epithelial cells that invaded deep into the underlying connective tissue, usually accompanied by abundant fibrous tissue proliferation (Figure 1B).
Immunohistochemical Stains
A summary of immunohistochemical stains is presented in Table 3. All negative controls showed absence of staining (results not shown).
Immunohistochemical Expression of the Examined Markers According to the Various Diagnoses a

a (−) no staining; (+) 1% - 10% stained cells; (++) 11% - 50% stained cells; (+++) >50% stained cells]
b Blood vessel walls served as control.
c Cells within tumor microenvironment.
Proliferating cell nuclear antigen
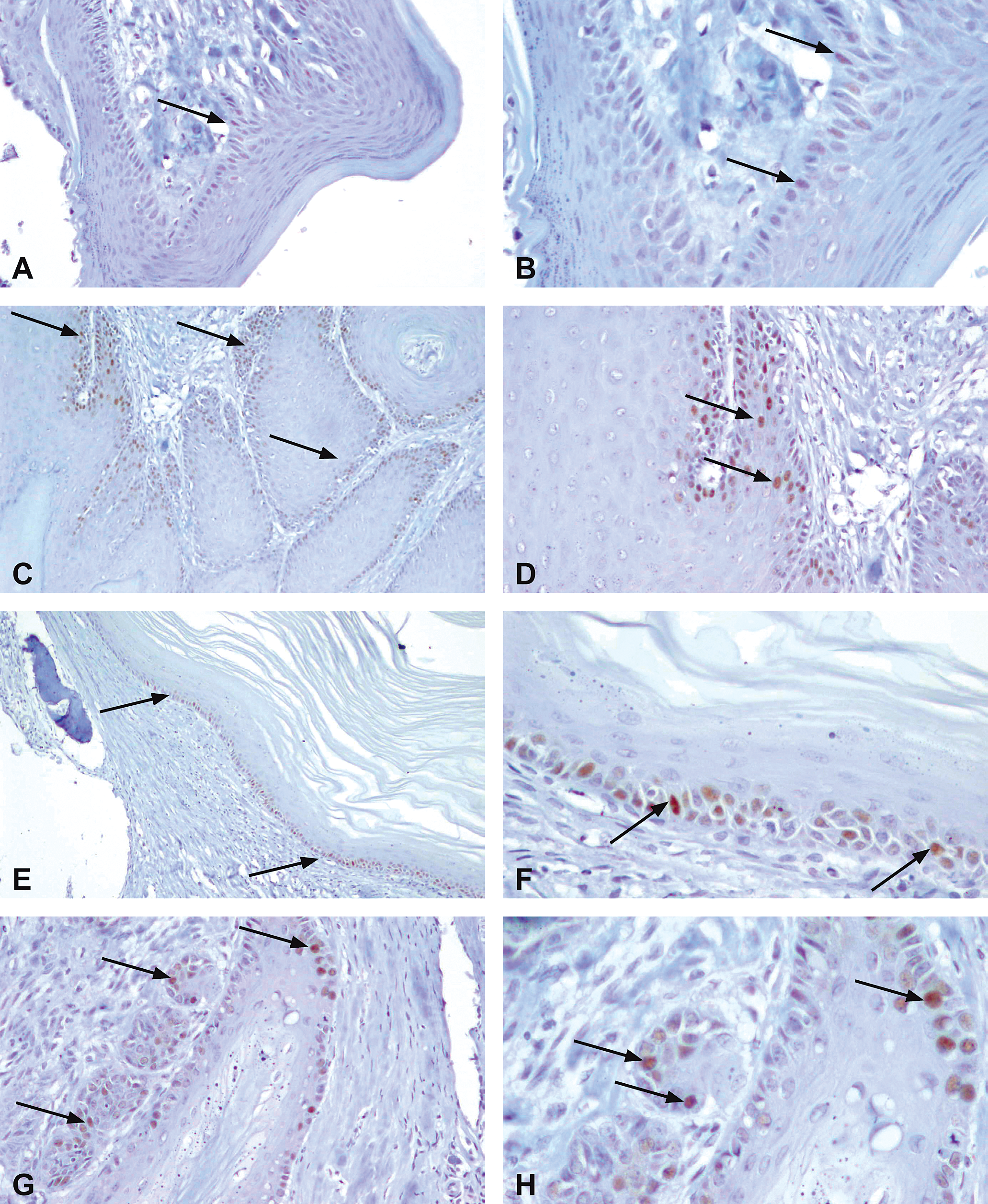
In normal epithelium, expression was seen in scattered basal cells of the lining epithelium of the gingival papillae (Figure 2A and 2B). In GSH, expression of PCNA was frequent and involved both basal and suprabasal layers (Figure 2C and 2D). In CKH, expression was mainly in the basal layer (Figure 2E and 2F). In SCC, expression was most evident at the front of invasion (Figure 2G and 2H).
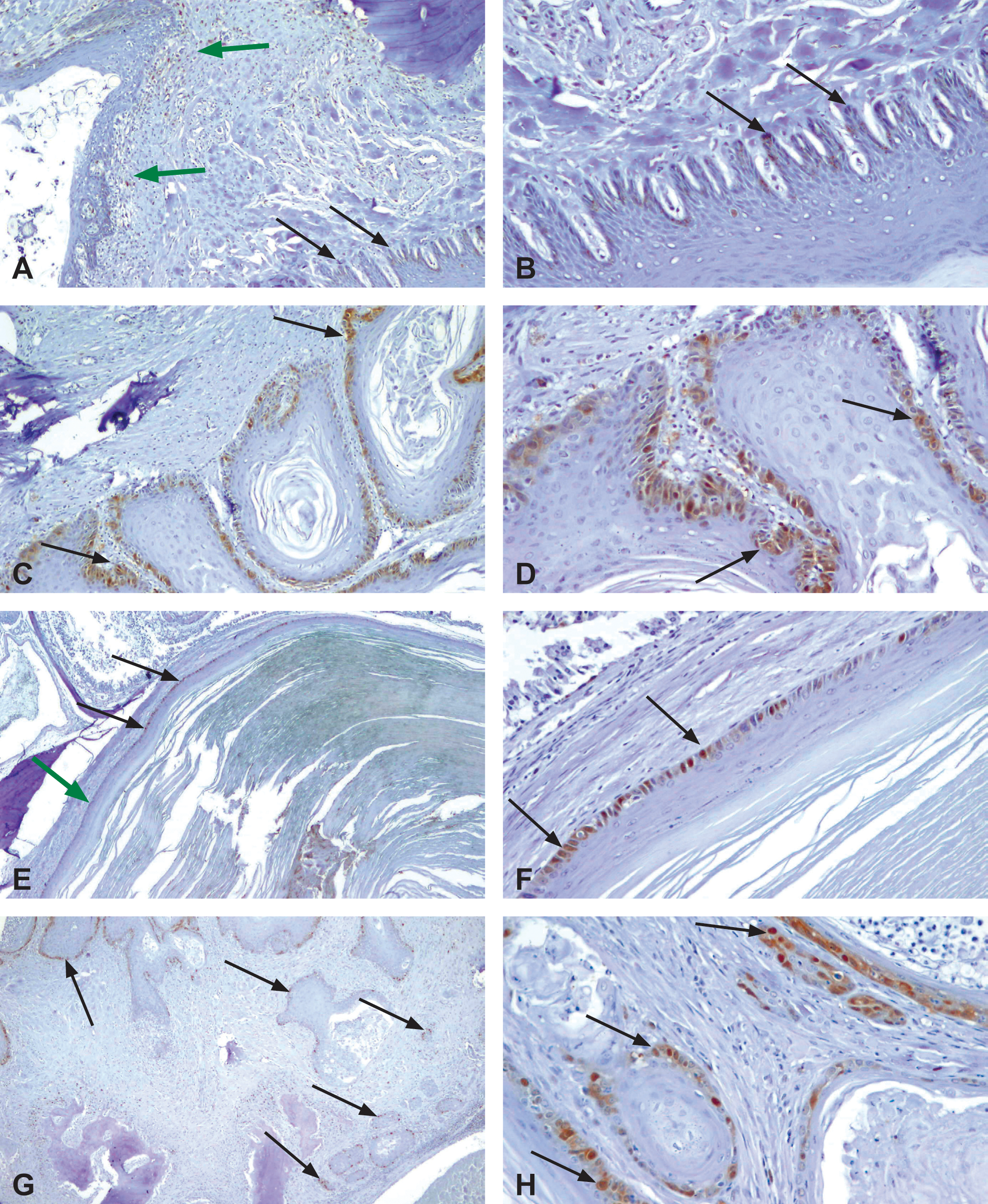
Proliferating cell nuclear antigen (PCNA) immunohistochemical profile of 3,3′,4,4′-tetrachloroazobenzene (TCAB)–induced gingival lesions. (A). Epithelium lining the gingival papilla from a control male rat. Note the PCNA positive staining of the basal cells (black arrows). There is mild gingivitis in the mucosa close to the molar tooth, indicated by PCNA-positive inflammatory cells (green arrows); ×100. (B) Epithelium lining the gingival papilla from a control male rat. Magnification of a region from (A), further showing the PCNA-positively stained basal cells (arrows); ×200. (C) Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. Note the strong, continuous staining within cells located at the basal and suprabasal layers (arrows); ×100. (D) Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. A magnified area of (C), showing the strong positively stained nuclei within cells located at the basal and suprabasal layers (arrows); ×200. (E) Cystic keratinizing gingival hyperplasia from a high-dose TCAB–treated male rat. Note a segment of the cystic epithelium demonstrating strong positively stained nuclei within basally located cells (arrows). The adjacent segment of cystic epithelium is negative to PCNA (green arrow); × 40. (F) Cystic keratinizing gingival hyperplasia from a high-dose TCAB–treated male rat. Note a segment of the cystic epithelium demonstrating strong positively stained nuclei within cells located at the basal layer (black arrows). Note the higher intensity of staining comparing to the control (A and B); × 200. (G) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. Note strong positively stained nuclei at the periphery of the tumor islands, which are most evident in the invading islands (arrows); ×40. (H) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. Higher magnification from (G), lower right corner, demonstrating strongly positive nuclei, which are most evident at the invasion front (arrows); ×200.
Cyclin D1
In normal gingival epithelium, only a minority of basal cells showed positively stained nuclei (Figure 3A and 3B). In GSH, expression of cyclin D1 was frequent and involved both the basal and suprabasal layers (Figure 3C and 3D). CKH demonstrated a similar pattern of expression as GSH (Figure 3E and 3F). Squamous cell carcinoma showed high expression, especially in the invading and less differentiated areas (Figure 3G and 3H).
Cyclin D1 immunohistochemical profile of TCAB-induced gingival lesions. (A) Epithelium lining the gingival papilla from a control male rat. There are only sparse faintly cyclin D1–stained nuclei within the basal layer (arrow); ×200. (B) Epithelium lining the gingival papilla from a control male rat. Magnification of the bottom region of (A), showing a few faintly stained nuclei within the basal layer (arrows); ×400. (C) Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. Note strong positively stained nuclei within cells located at the basal and suprabasal layers (arrows); ×100. (D) Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. Magnification of the upper left region from (C) showing the strong positively stained nuclei within cells located at the basal and suprabasal layers (arrows); ×200. (E) Cystic keratinizing gingival hyperplasia from a high-dose TCAB–treated male rat. Note continuous and strong nuclear staining within cells located at the basal and suprabasal layers (arrows); ×100. (F) Cystic keratininzing gingival hyperplasia from a high-dose TCAB–treated male rat. Magnification of an area (bottom right) from (E) further showing the strong positively stained nuclei within cells located at the basal and suprabasal layers (arrows); ×400. (G) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. Note strongly stained nuclei, which are most evident in the invading islands (arrows); ×200. (H) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. Magnification of the top region from (G), which emphasizes the strong and frequent nuclear staining of the invading islands (arrows); ×400.
β-Catenin
In the normal gingiva, membranous expression was mainly in the suprabasal and basal side of the upper layer (Figure 4A). Gingival squamous hyperplasia had a similar pattern of expression as the normal gingiva (Figure 4B and 4C). In superficially located CKH, membranous expression was evident throughout the epithelium (Figure 4D), whereas in deeper located CKH, there was overall loss of membranous β-catenin (Figure 4E). In SCC, membranous expression was gradually lost as the tumor invaded, and at the invasion front, no membranous staining was present (Figure 4F and 4G).
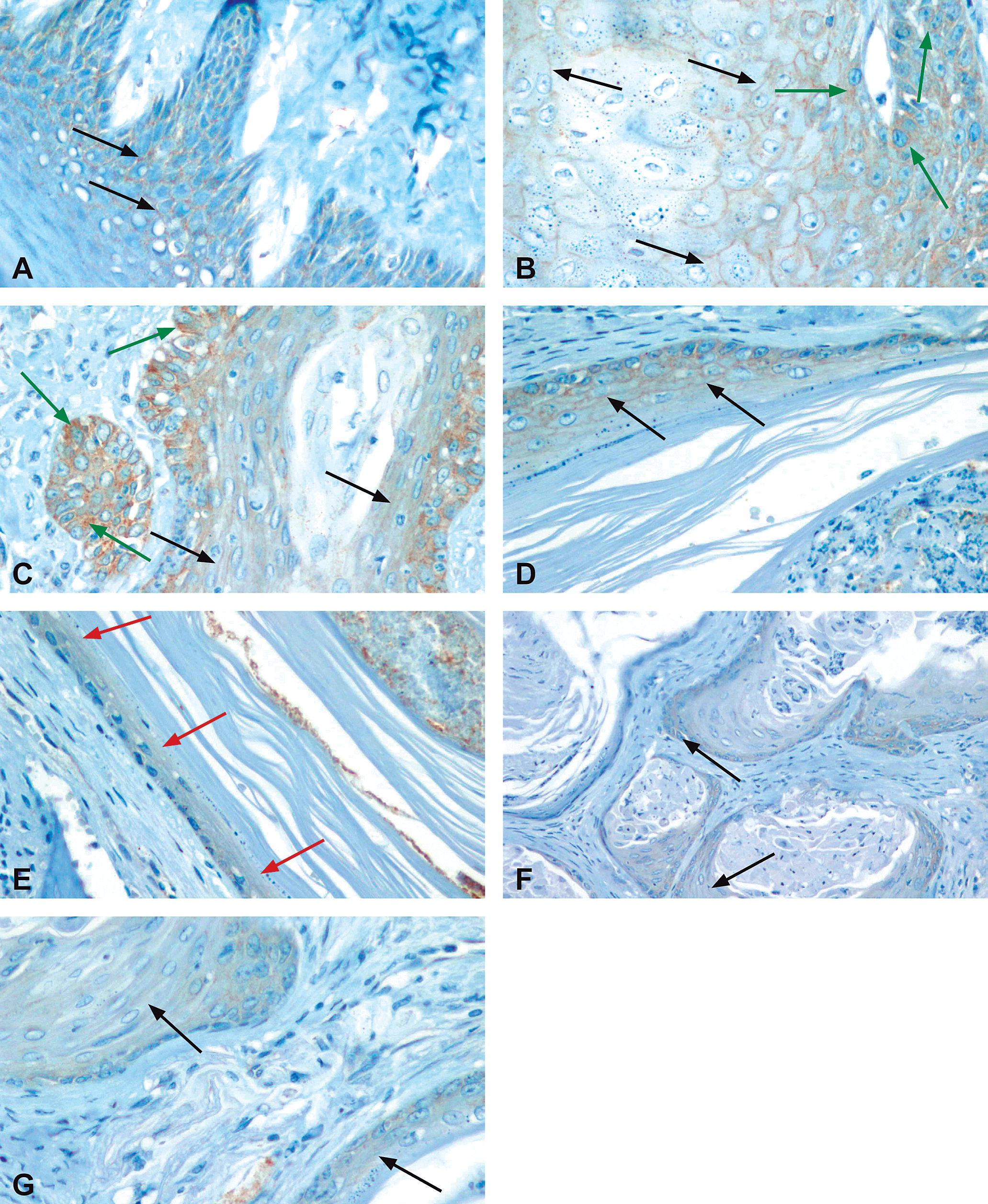
β-catenin immunohistochemical profile of 3,3′,4,4′-tetrachloroazobenzene (TCAB)–induced gingival lesions. (A) Lining epithelium of the gingival papilla from a control male rat showing extensive normal membranous β-catenin staining in the spinous layer (arrows); ×400. (B) Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. Note that there is membranous staining in the entire width of the epithelium (black arrows). However, some cytoplasmatic staining is also observed, especially within cells from the more basal layers (green arrows); ×400. C. Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. In this example, cytoplasmatic staining is predominant (green arrows) over the membranous staining, which is mainly constrained to cells from the upper epithelium (black arrows); ×400. (D) Cystic keratinizing gingival hyperplasia from a high-dose TCAB–treated male rat. There is membranous staining throughout the width of the cystic epithelium (arrows); ×400. (E) Cystic keratinizing gingival hyperplasia from a high-dose TCAB–treated male rat. Another area from the cystic lesion presented in (D) showing lack of membranous staining (arrows); ×400. (F) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. Overall, there is loss of membranous staining in the deep infiltrating invasive islands with only some residual, weak cytoplasmic staining (arrows); ×200. (G) Higher magnification of a region in (F) further demonstrating loss of membranous staining (arrows); ×400.
Maspin
In the normal gingiva, maspin was expressed in all layers of the epithelium as a homogenous membranous - cytoplasmatic staining (Figure 5A). In GSH, expression was observed also at a nuclear level (Figure 5B and 5C). In contrast, in CKH, staining was gradually lost (Figure 5D and 5E), and in SCC, expression of maspin decreased in parallel to the depth of invasion (Figure 5F and 5G).
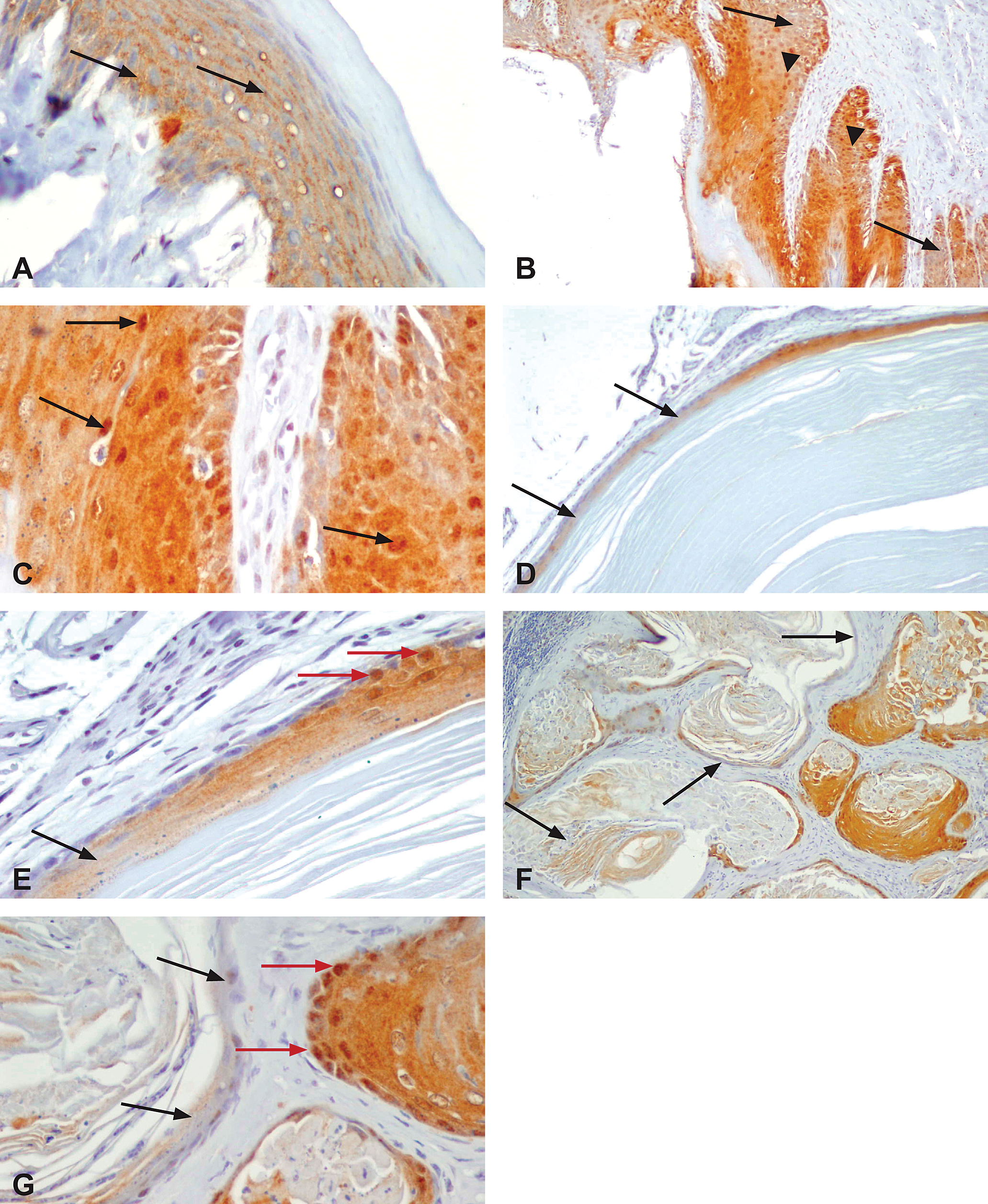
Maspin immunohistochemical profile of 3,3′,4,4′-tetrachloroazobenzene (TCAB)–induced gingival lesions. (A) Epithelium lining the gingival papilla from a control male rat. The staining is homogenous in all layers, and mostly membranous and cytoplasmatic (arrows); ×400. (B) Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. The membranous/cytoplasmatic staining is usually preserved, however, there are areas almost lacking this pattern of staining (arrows). Concurrently, several cells with nuclear staining can be observed (arrowheads); ×100. (C) Gingival hyperplasia (moderate) from a high-dose TCAB–treated male rat. Note the cytoplasmic staining, which is usually preserved. In addition, many of the nuclei in this area are also positively stained (arrows); ×100. (D) Cystic keratinizing gingival hyperplasia from a high-dose TCAB–treated male rat. Note the partial loss of staining (arrows), which is more pronounced in deeper and invasive regions; ×100. (E) Cystic keratinizing gingival hyperplasia from a high-dose TCAB–treated male rat. A magnification of a region from (D), showing the transition between strong membranous and nuclear staining (red arrows) to an area almost devoid of any staining (black arrow); ×400. (F) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. Note the partial loss of staining in the deepest invading neoplastic islands (arrows). (G) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. A magnified area from Figure F, showing partial loss of staining (black arrows, left side) versus strong cytoplamic and nuclear staining (red arrows, right side); ×100.
α-Smooth muscle actin
In normal and GSH lesions, expression was confined to the smooth muscle cells located within the blood vessel walls (Figure 6A). In CKH (Figure 6B and 6C) and SCC (Figure 6D), sporadic, spindle-shaped, positively stained stromal cells were observed running in parallel to the cystic epithelium and tumor islands, respectively.
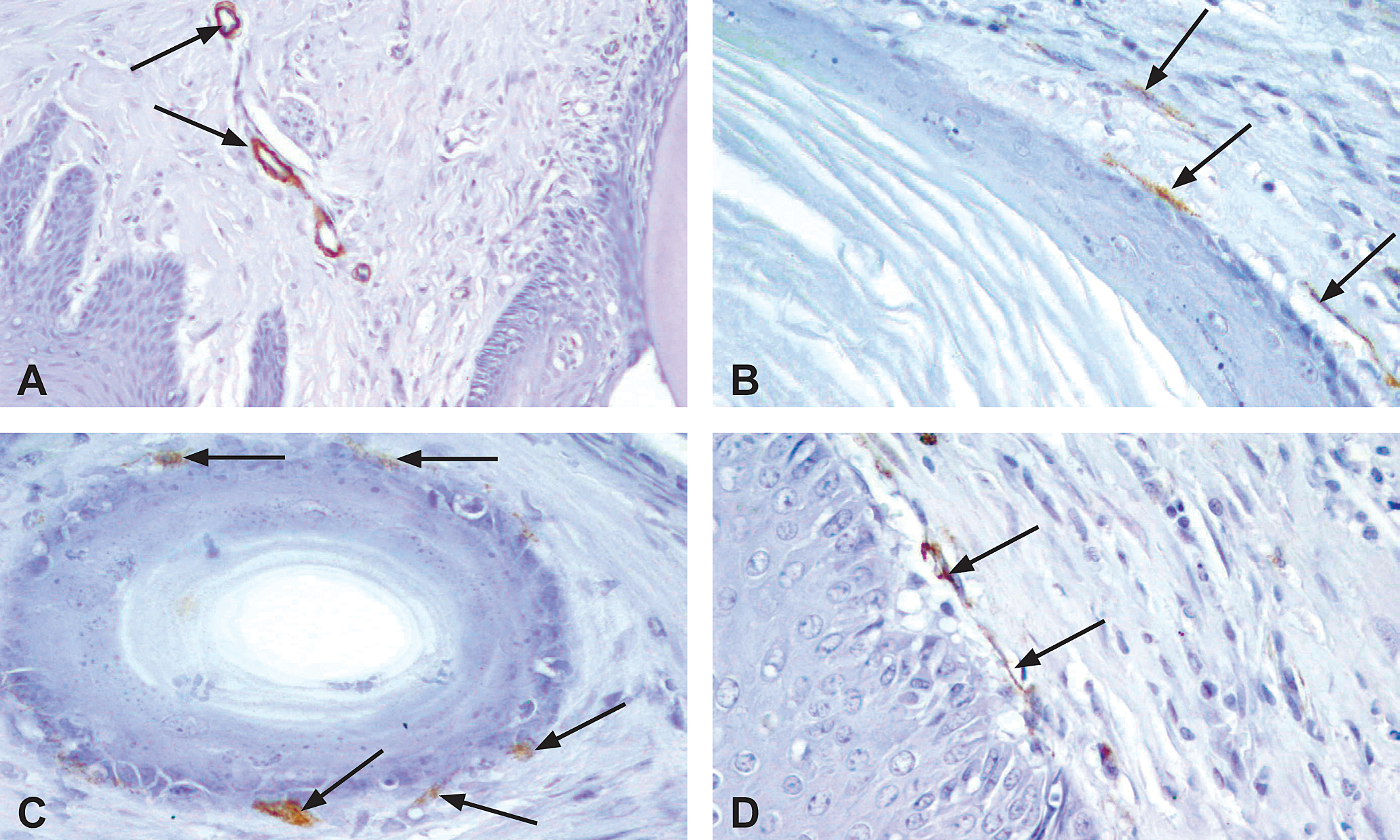
α-Smooth muscle actin immunohistochemical profile of 3,3′,4,4′-tetrachloroazobenzene (TCAB)–induced gingival lesions. (A) Gingival papilla from a control male rat. The smooth muscle cells located within the blood vessel walls (arrows) are αSMA positive. There are no positive stromal cells close to the lining gingival epithelium; ×200. (B,C) Cystic keratinizing gingival hyperplasia from a high-dose TCAB -treated male rat. Note sporadic, spindle-shaped, a SMA-positive stromal cells, usually located adjacent to the cystic epithelium (arrows); ×400. (D) Gingival squamous cell carcinoma from a high-dose TCAB–treated male rat. Note sporadic, spindle-shaped, αSMA-positive stromal cells usually located adjacent to the tumor (arrows); ×400.
Cystine-rich acidic secreted protein /osteonectin
No expression was detected in normal gingiva, except for sporadic stromal cells in areas of minimal gingivitis. In CKH (Figure 7A) and GSH (Figure 7B), sporadic positive stromal cells located close to the hyperplastic epithelium were seen.
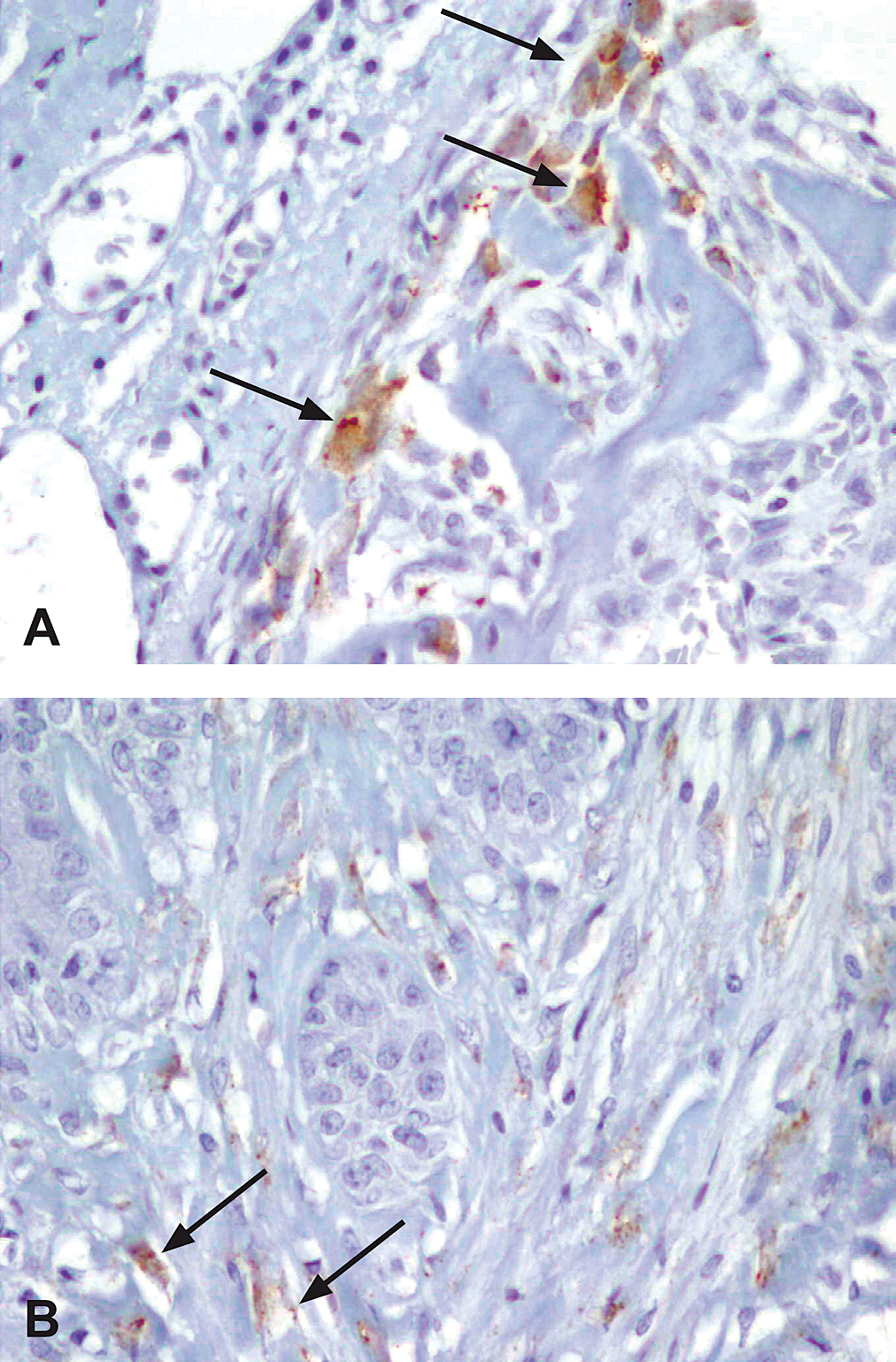
Osteonectin/cystine-rich acidic secreted protein (SPARC) immunohistochemical profile of 3,3′,4,4′-tetrachloroazobenzene (TCAB)–induced gingival lesions. (A) Stromal SPARC-positive cells (arrows) located adjacent to reactive bone, in close proximity to cystic keratinizing hyperplasia from a high-dose TCAB–treated male rat. (B) Gingival hyperplasia from a high-dose TCAB–treated male rat. Note the SPARC-positive stromal cells adjacent to the epithelial islands (arrows); ×400.
Discussion
Recently, the effects of chronic exposure of rats to dioxin and dioxin-like compounds (DLCs) have been reported in detail following the two-year chronic gavage studies performed by the National Toxicology Program with 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD); 3,3′,4,4′,5-pentachlorobiphenyl (PCB126); 2,3,4,7,8-pentachloro-dibenzofuran (PeCDF); 2,2′,4,4′,5,5′-hexachlorobiphenyl (PCB153); the Toxic Equivalency Factor (TEF) tertiary mixture of TCDD, PCB126, and PeCDF; and the binary mixtures of PCB126 and 153 and PCB126 and 2,3′,4,4′,5-pentachlorobiphenyl (PCB118; Yoshizawa et al. 2005). In these experiments, the incidence of gingival squamous hyperplasia was significantly increased following exposure to the tested compounds, and exposure to TCDD, PCB126, and the mixture of PCB126 and 153 led to a significant increase in gingival SCC incidence (Yoshizawa et al. 2005). The effect of DLCs on the oral cavity has been demonstrated before in a number of reports in humans and animals (Yoshizawa et al. 2005). For example, in humans, elongation of the depth of periodontal pockets and gingival pigmentation was observed following exposure to PCBs and PeCDFs (Hashiguchi et al. 1995). In monkeys, an array of oral changes was observed, including GSH, hyperkeratosis, dyskeratosis, keratocystic formation, squamous metaplasia of the ameloblast surrounding unerupted teeth, and/or hyperpigmentation (Hashiguchi et al. 1983; Hashiguchi et al. 1987; McNulty 1985; Tryphonas et al. 1986; Yoshihara et al. 1979). However, the oral carcinogenicity properties of DLCs were not demonstrated before the current long-term studies conducted by the NTP (Yoshizawa et al. 2005).
Here we report that TCAB, an additional DLC, may also predispose to gingival SCC. In addition, we report for the first time the development of a new type of DLC-associated gingival lesion, cystic keratinizing hyperplasia (CKH), which is characterized by variably sized cavities lined by stratified squamous epithelium and filled with keratin located deep to the overlying gingival epithelium. It is not clear why TCAB was the only DLC to cause CKH in the gingivae. However, our studies with different DLCs suggest that the range of neoplastic and non-neoplastic lesions seen in the same organ may vary among the DLC compounds. For example, in the reproductive system, uterine SCC was caused only by TCDD and the binary mixture of PCB126 and 153, whereas no change was noted with all other DLCs (Yoshizawa et al. 2009a). This finding suggests that more than one mechanism is involved in DLC-induced carcinogenesis in rats, including in the gingivae. Additionally, some of the carcinogenic effects of TCAB might be induced by one or more of its dichloroaniline metabolites (Pillai et al. 1996), and not directly by TCAB.
3,3′,4,4′-Tetrachloroazobenzene, like other DLCs, binds to its cytosolic aryl hydrocarbon receptor (AhR; NTP 2009). The latter dissociates from the chaperones, dimerizes with AhR nuclear translocator, and ultimately translocates into the nucleus, where it induces changes in gene transcription (Martinez et al. 2002). Indeed, the AhR has previously been shown to be expressed in oral tissues, including the molar teeth buds and palatal epithelial cells, and its activation has been shown to change epithelial cell differentiation and proliferation (Yoshizawa et al. 2005). It should be noted, however, that there are many different factors that can influence the effects of various DLCs by altering the responsiveness of downstream pathways (Harper et al. 2006). Therefore, although presumably all DLCs act by binding to the AhR, it is hard to predict the end effect of each DLC as a result of this complex set of mediators. This is especially the case when considering the recently gained knowledge on the AhR-mediated activities that are not mediated by the canonical dioxin response element–dependent gene expression (Murray et al. 2010). 3,3′,4,4′-Tetrachloroazobenzene, delivered over a long period of time, appears to exert a hyperplastic and neoplastic effect on the lining epithelium of the gingivae, in particular in the region of the maxillary molars. There are several possible explanations for the increased sensitivity of this region to TCAB administered by gavage (Yoshizawa et al. 2005). The gingival epithelium, especially in the dento-gingival junction of molars, possesses a high mitotic activity (Loe et al. 1972; Shimono et al. 2003; Watanabe et al. 2004). Together with its high metabolic activity (Vondracek et al. 2001) and high rate of absorption (Shojaei 1998), the molar gingival epithelium becomes a location that is very sensitive to xenobiotic effects. Another explanation can be based on the anatomy of this region. It can be assumed that a certain amount of the gavage containing the chemical agent repeatedly and persistently flows toward the posterior maxillary sulcus (vestibule), where it is kept in tight contact with the gingival epithelium, which in time reacts to the chronic irritation with a hyperplastic response. Furthermore, some of the chemical probably penetrates into the gingival sulcus, where it delivers a chronic stimulus to the junctional epithelium, which in time is translated into hyperplastic lesions that were observed to run along the tooth root. However, it is not likely that the lesions occur solely as a result of gavaging, because these types of tumors are rarely seen in vehicle control animals in other gavage studies. In male F344 rats, in which background tumor incidence is well established, the mean incidence of SCC seen in vehicle controls from corn oil gavage studies is 0.33% (range 0%–2%) which is similar to the mean background incidence for SCC for all routes of administration (http://ntp.niehs.nih.gov/ntpweb/index.cfm?objectid=92E705C7-F1F6-975E-72D23026B1645EB9
The TCAB model is partly similar to other animal models (rats and mice) in which the carcinogenic potential of different chemicals was tested, in particular 4-nitroquinoline 1-oxide (4NQO; Dayan et al. 1997; Kanojia and Vaidya 2006). This is a water-soluble carcinogen that, when administered in drinking water in very small concentrations for long periods of time, simulates the action of carcinogenic agents within the oral cavity of humans (e.g., tobacco smoke). In a time-dependent manner, under the action of 4NQO, a range of lesions develop on the animals’ tongues that are both morphologically and cytologically analogous to the precursor lesions seen in human patients, namely, hyperplasia and escalating dysplasia, which finally culminate in invading SCC (Dayan et al. 1997; Vered et al. 2005). This also may be the case with the DLCs / TCAB, where GSH is the earliest morphological type of lesion to be observed, CKH seems to be a more severe lesion in terms of penetration into the depth of the surrounding tissues than GSH, and SCC is the most invasive
By employing a panel of epithelial markers related to the cell cycle and cellular differentiation status, we showed that changes in their expression in GSH was different from the control and that in CKH it was very similar to SCC. In general, we found that the carcinogenesis-associated immunohistochemical profile followed the morphological changes of increased extent of GSH and CKH and their depth of invasion, suggesting that they can be considered as lesions with potential for malignant transformation.
The pattern of expression of PCNA was more extensive in the GSH lesions compared to control and most remarkable in the SCCs, and in general, it followed that of cyclin-D1. Upon DNA damage signal and activation of p53, one of the G1-S controllers induced by p53 is GADD45 (Hartl and Jones 2009). The latter forms a complex with PCNA that reduces the processivity of DNA polymerase with an outcome of slowing down DNA synthesis, allowing for DNA repair. 3,3′,4,4′-Tetrachloroazobenzene, through its receptor, AhR, has an inhibitory effect on the transcription activity of p53 (Tijet et al. 2006), which can result in loss of control on PCNA, and as a result, its expression increased in the TCAB-induced hyperplastic lesions and culminated in SCC. It should be noted, however, that TCAB exposure may lead to increased expression of PCNA by other mechanisms than direct effect on the AhR, for example, by disrupting retinoid homeostasis and causing vitamin A deficiency, which can in turn lead to altered growth and differentiation of the oral gingival epithelium (Yoshizawa et al. 2005). It has been demonstrated that higher expression of PCNA is a marker for the malignant potential of the oral mucosa and serves as an indicator for poor prognosis (Tsuji et al. 1995; Vered et al. 2007).
In the present study, cyclin-D1 showed a similar trend of expression compared to PCNA. This finding is somewhat analogous to the 4NQO model, in which its expression was found to increase with the severity of the dysplastic lesions (Kanojia and Vaidya 2006). It has been shown that AhR can activate a series of genes whose protein products are critical in the G1-S transition of the cell cycle, namely, a series of cyclin-dependent kinases (CDK), among them CDK-6 (Martinez et al. 2002). Cyclin-D1 works together with CDK6 and CDK4 and jointly increases cell proliferation (Diamandis 2002). Furthermore, the gene encoding for cyclin-D1 has a binding region for the β-catenin after it leaves the membranous location, and in a complex with Tcf-4, it transports to the nucleus to become a transcription factor (Diamandis 2002). In the present study, we found loss of membranous expression of β-catenin; however, we could not detect any nuclear expression. Presumably, activation of the cyclin-D1 is not performed by β-catenin in the TCAB model, but perhaps rather by the AhR route. In carcinomas of the head and neck, oral mucosa, larynx, and nasopharynx, higher expression of cyclin-D1 has been reported (Das et al. 2011; de Rosa et al. 1999; Dong et al. 2001; Gimenez-Conti et al. 1996; Lamb and Ewen 2003; Lin et al. 2006; Staibano et al. 2001; Xu et al. 1998). In addition, expression of membranous β-catenin was previously found to be reduced in oral SCC cases (Bagutti et al. 1998; El-Rouby 2011; Lo Muzio et al. 1999; Williams et al. 1998).
We have found that the expression of maspin was the highest in control animals, almost similarly high in GSH, less in CKH, and the lowest in SCC. Maspin is known to have a critical role in maintaining cellular homeostasis, in particular to guard cells from oxidative stress and keep them from going into a metabolism of glycolysis (Cai et al. 2009; Yin et al. 2005). Its expression in counteracting increased oxidative stress is demanded when cells are exposed to TCAB, since AhR is one of the main reasons for the activation of the gene encoding for the cytochrome P450 (CYP450) family that has a strong effect of increasing intracellular oxidative stress (Martinez et al. 2002).
As the lesions of GSH, CKH, and SCC in the present study showed decreased expression of maspin, it can be assumed that the guardian effect of maspin is overruled, especially in CKH and mostly in SCC. From a study on one DLC on normal lung and lung cancer cell lines, it has been shown that AhR has the ability to suppress the transcriptional activity of the cell cycle protein p53 (Martinez et al. 2002), which is known to be one of the major activators of the maspin gene (Hendrix 2002). This can be an explanation of why the more extensive and penetrating lesions in the present study demonstrated less expression of maspin. In general, loss of maspin was correlated with poor prognosis in colon, uterus, breast, prostate, skin, thyroid, and oral SCC (Amir et al. 2005; Bettstetter et al. 2005; Biliran and Sheng 2001; Denk et al. 2007; Shams et al. 2006; Xu et al. 2005; Yin et al. 2006; Yoshizawa et al. 2009b; Xia et al. 2000; Yasumatsu et al. 2001).
α-Smooth muscle actin and SPARC were not a prominent feature of the stroma adjacent to TCAB-induced SCC. In contrast, α-SMA–positive stromal cells (myofibroblasts) are a remarkable finding of the stroma surrounding 4NQO-induced tongue SCC (Fracalossi et al. 2011; Vered et al. 2007) as well as in human patients, where their presence is associated with tumor stage, metastasis, and poor prognosis (Kawashiri et al. 2009; Vered et al. 2010), which further enhances the molecular differences between these animal models.
In summary, this is the first set of studies to report that rat gingivae exposed for a long time to doses of TCAB, a nongenotoxic compound, respond by developing epithelial hyperplastic lesions, which include benign GSH, benign but locally aggressive CKH, and malignant lesions in the form of low-grade, well-differentiated SCC. There appears to be a progression from the hyperplastic and cystic lesions toward malignancy based on the morphological changes that is supported by the relative expression of proteins associated with carcinogenesis. In order to prove that SCC develops from progression of CKH, or that CKH is a more advanced stage of GSH, studies showing the relationship among these lesions in a time-dependent manner have to follow.
Footnotes
Acknowledgments
We gratefully acknowledge the expert review of Dr. Mamta Behl and Dr. Janardhan Kyathanahalli from NIEHS, RTP, NC. Dr. Mamta Behl from NIEHS and Dr. Kyathanahalli Janardhan from ILS, RTP, NC.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the intramural program of the National Institute of Environmental Health Sciences (NIEHS), Research Triangle Park, NC. However, the statements, opinions, or conclusions contained herein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the United States government.
